Phytochemicals and Enzyme Inhibitory Capacities of the Methanolic Extracts from the Italian Apple Cultivar Mela Rosa dei Monti Sibillini
Abstract
1. Introduction
2. Results
2.1. Polyphenols and Triterpenes Composition
2.2. Enzymatic Inhibitory Effects
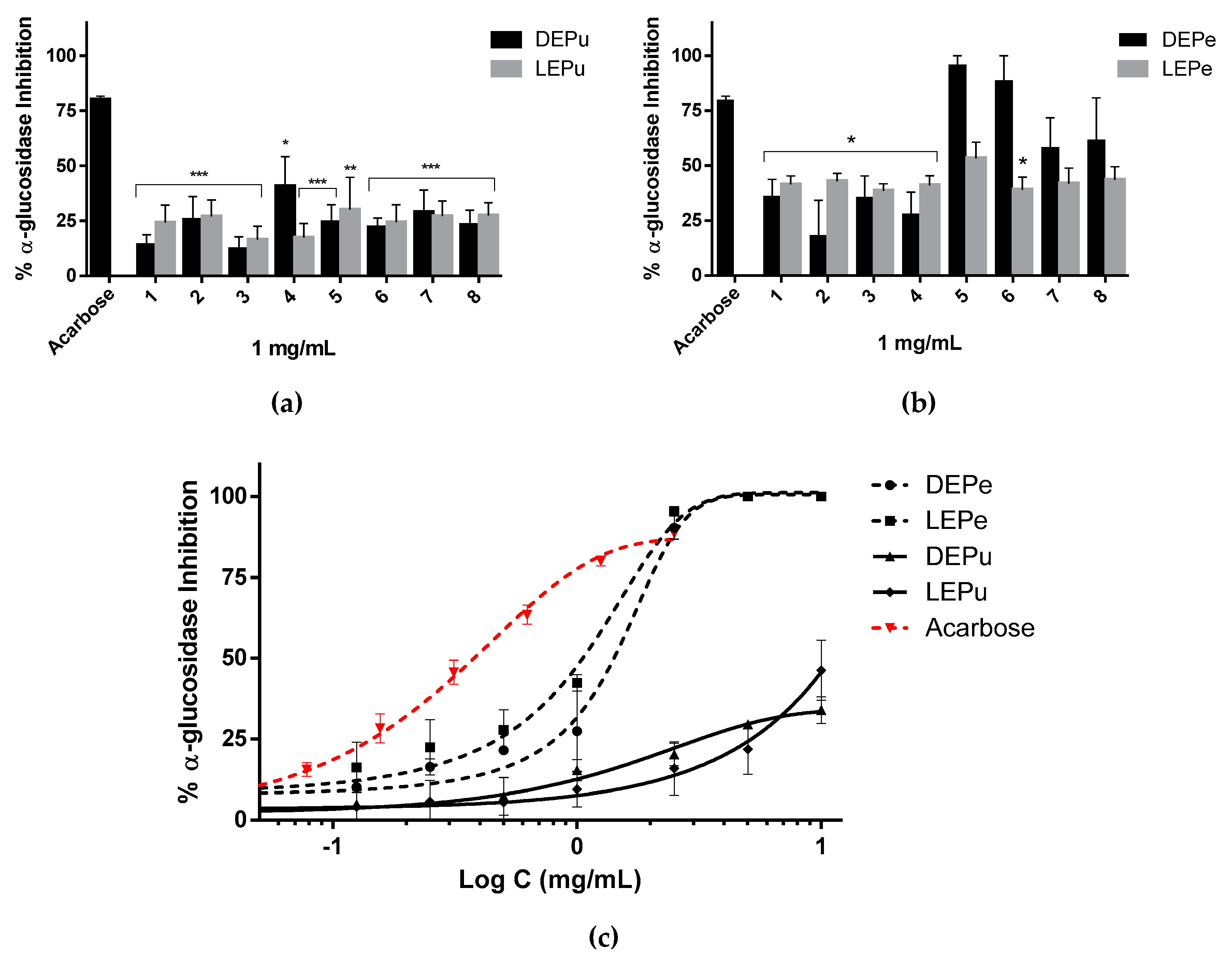
2.2.1. α-Glucosidase Inhibition
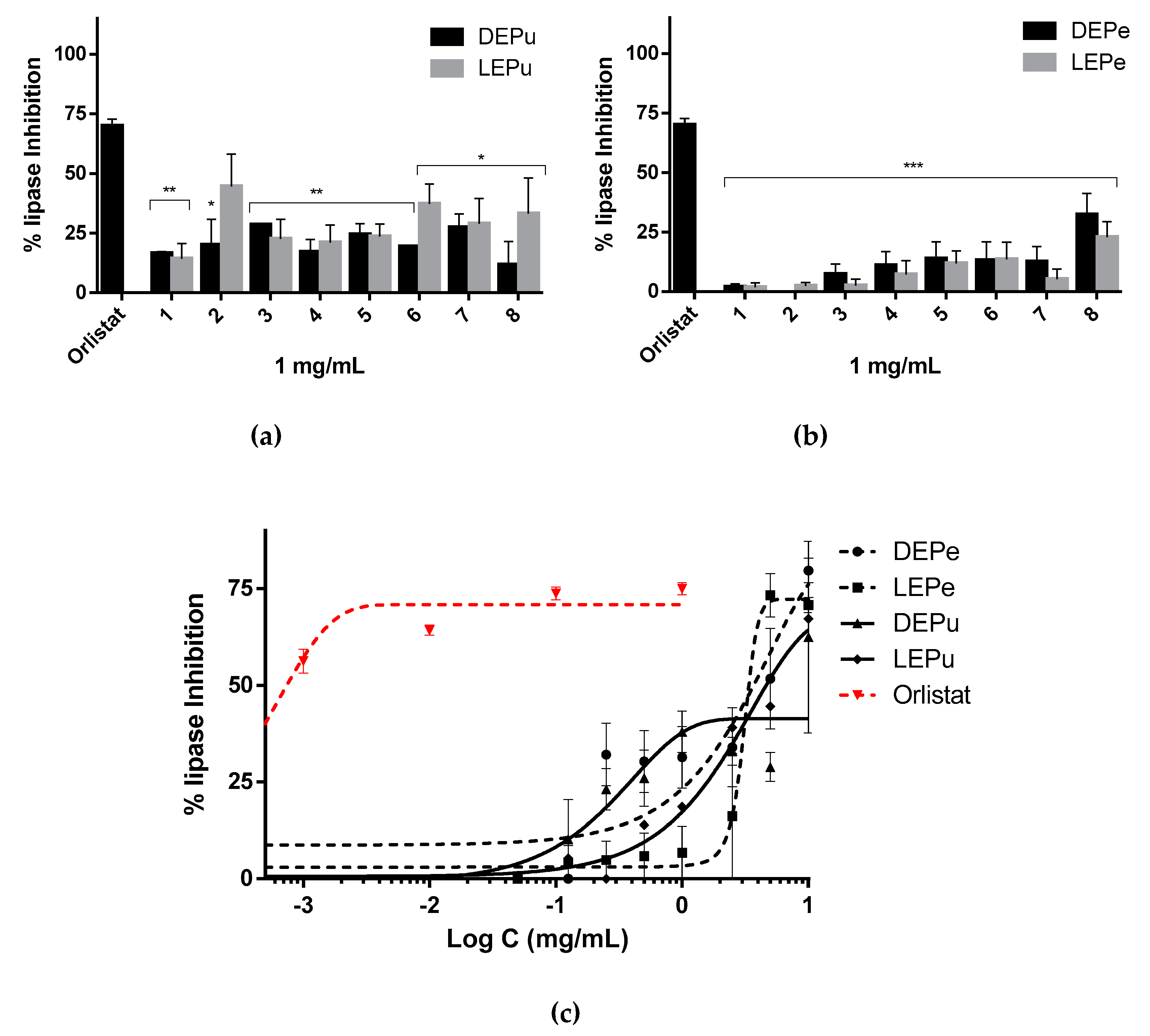
2.2.2. Lipase Inhibition
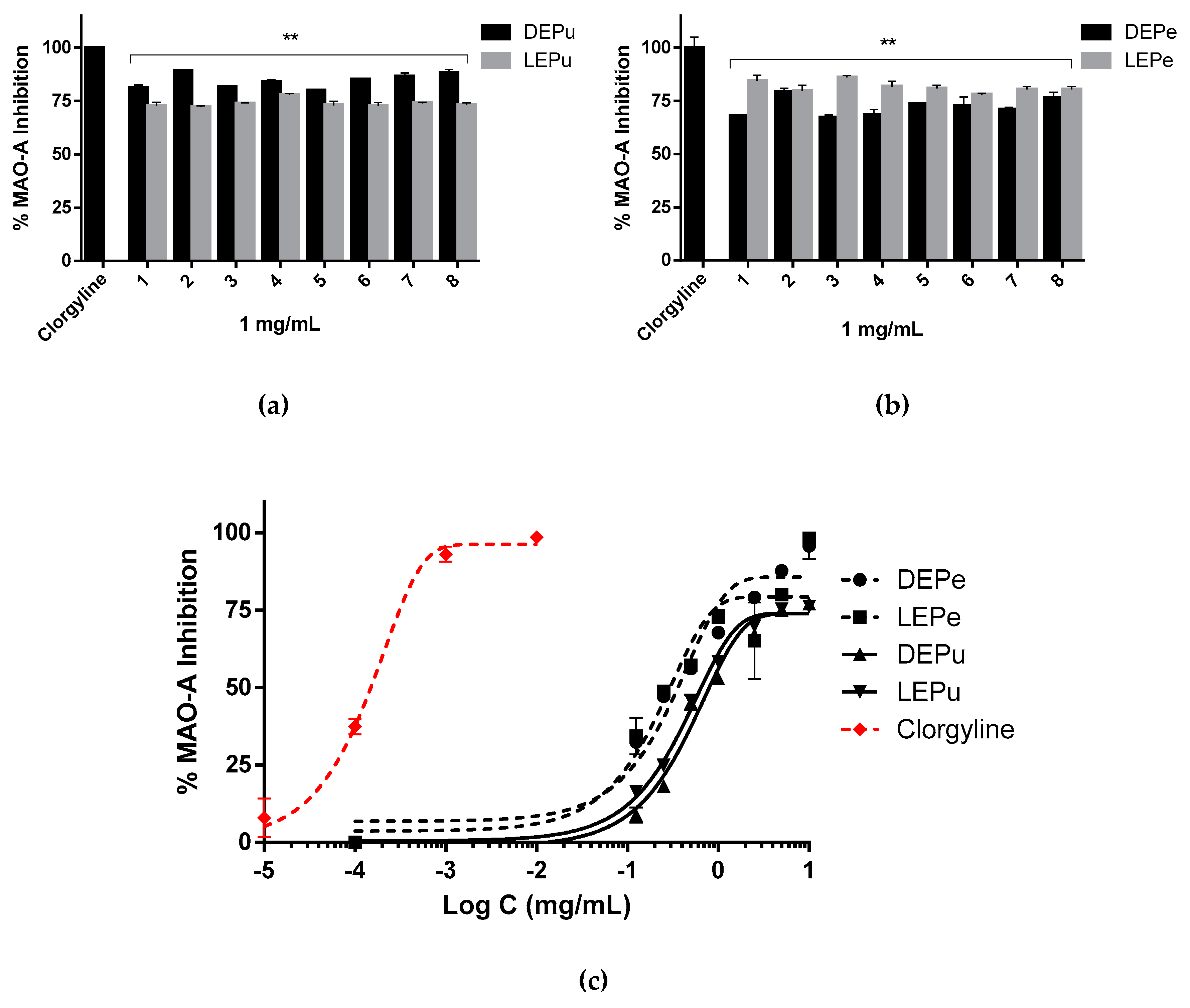
2.2.3. MAO-A Inhibition
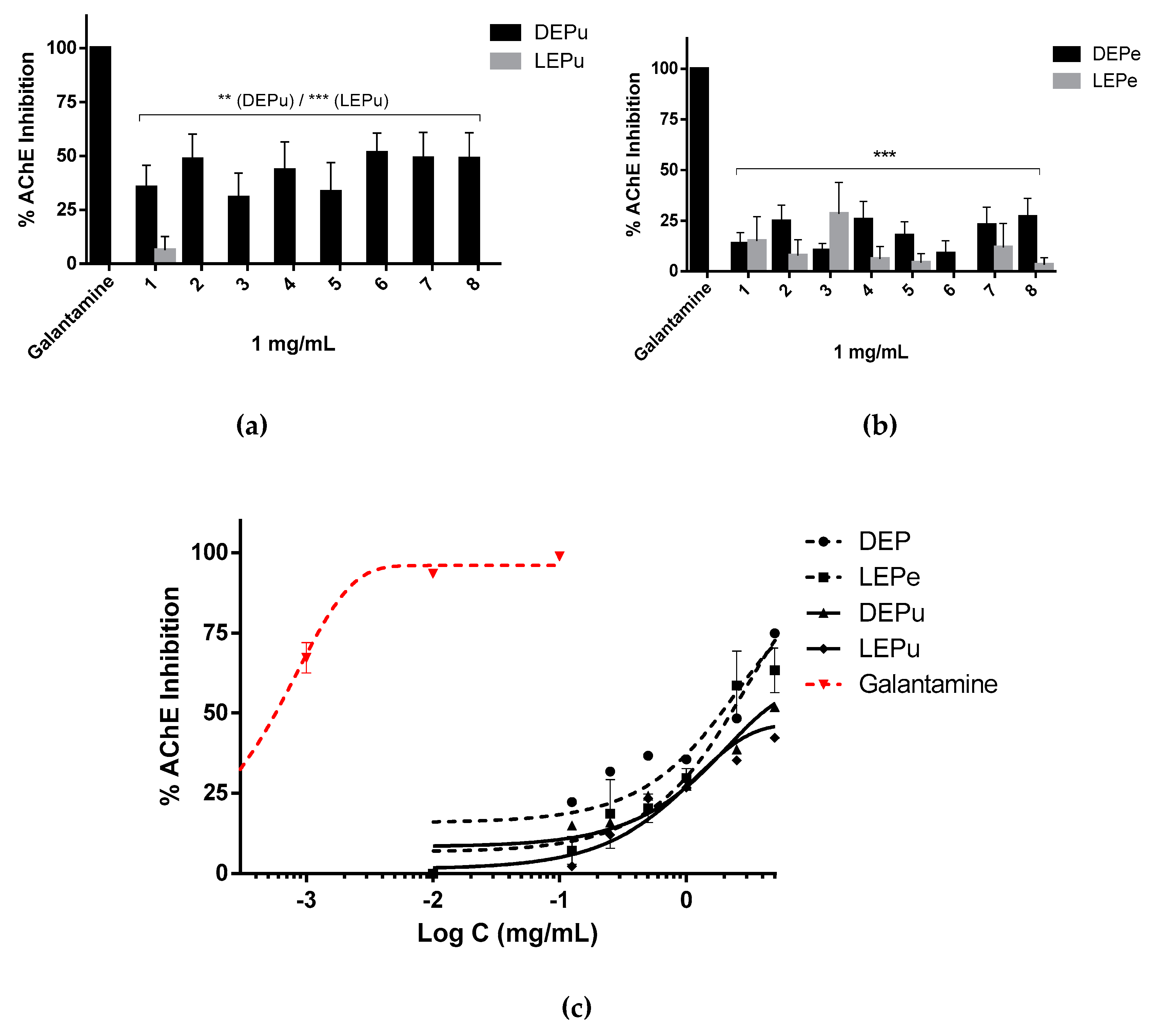
2.2.4. AChE Inhibition
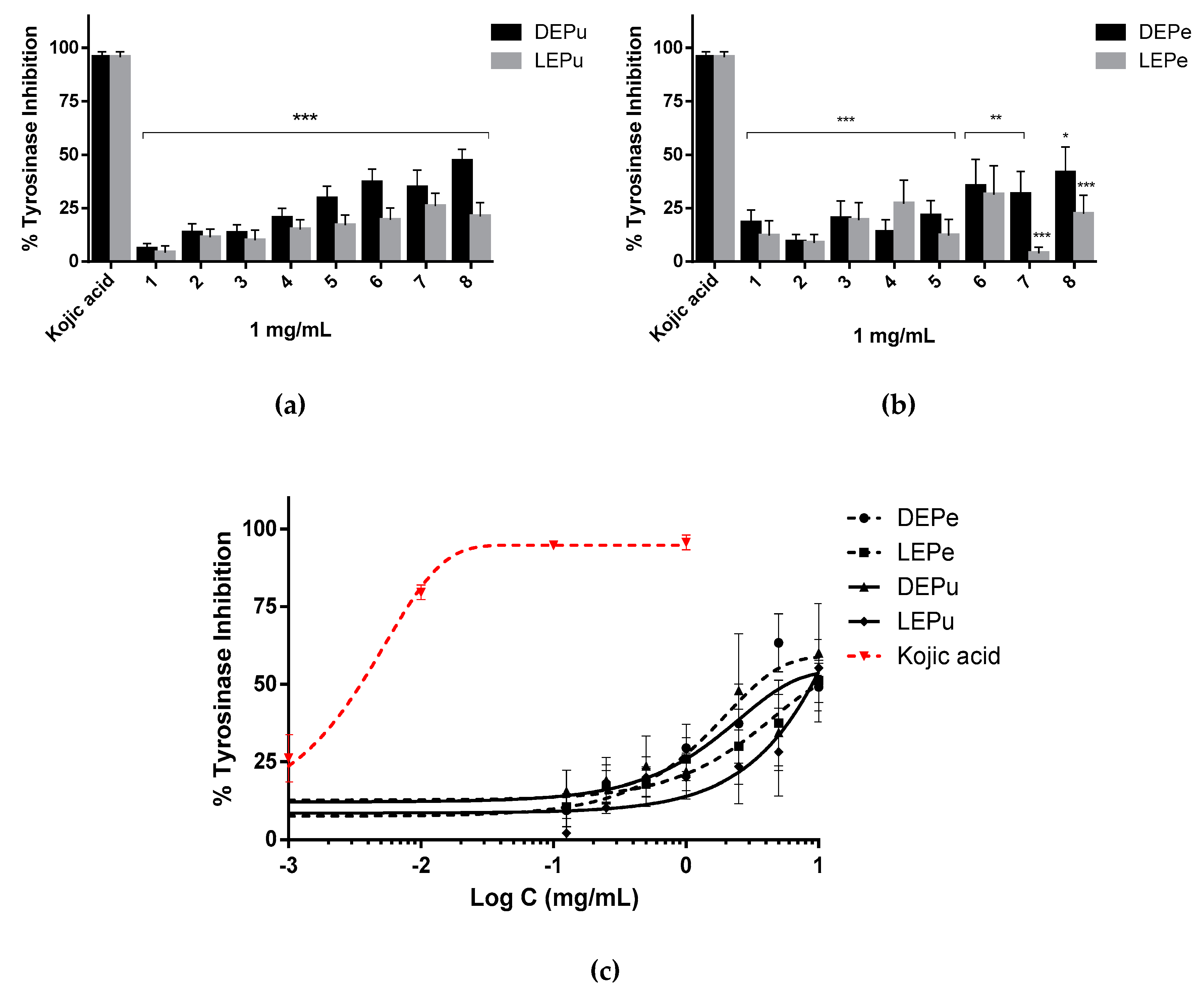
2.2.5. TYR Inhibition
2.3. Correlation Analysis between Phytochemical Composition and Bioactivity
3. Discussion
4. Materials and Methods
4.1. Sampling and Preparation of Apple Extracts
4.2. HPLC-DAD Analysis
4.3. Enzyme Inhibitory Activities
4.3.1. Reagents and Chemicals
4.3.2. α-Glucosidase Inhibition Assay
4.3.3. Lipase Inhibition Assay
4.3.4. Monoamine oxidase (MAO-A) Inhibition Assay
4.3.5. Acetylcholinesterase (AChE) Inhibition Assay
4.3.6. Tyrosinase Inhibition Assay
4.3.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–516. [Google Scholar] [CrossRef]
- Boyer, J.; Liu, R.H. Apple phytochemicals and their health benefits. Nutr. J. 2004, 3, 5. [Google Scholar] [CrossRef]
- Feliciano, R.P.; Antunes, C.; Ramos, A.; Serra, A.T.; Figueira, M.E.; Duarte, C.M.; de Carvalho, A.; Bronze, M.R. Characterization of traditional and exotic apple varieties from Portugal. Part 1–Nutritional, phytochemical and sensory evaluation. J. Funct. Foods 2010, 2, 35–45. [Google Scholar] [CrossRef]
- Nkuimi Wandjou, J.G.; Sut, S.; Giuliani, C.; Fico, G.; Papa, F.; Ferraro, S.; Caprioli, G.; Maggi, F.; Dall’Acqua, S. Characterization of nutrients, polyphenols and volatile components of the ancient apple cultivar ‘Mela Rosa Dei Monti Sibillini’from Marche region, central Italy. Int. J. Food Sci. Nutr. 2019, 70, 796–812. [Google Scholar] [CrossRef] [PubMed]
- Nkuimi Wandjou, J.G.; Mevi, S.; Sagratini, G.; Vittori, S.; Dall’Acqua, S.; Caprioli, G.; Lupidi, G.; Mombelli, G.; Arpini, S.; Allegrini, P.; et al. Antioxidant and Enzyme Inhibitory Properties of the Polyphenolic-Rich Extract from an Ancient Apple Variety of Central Italy (Mela Rosa dei Monti Sibillini). Plants 2020, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of polyphenols in different apple varieties. J. Agric. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef]
- D’Abrosca, B.; Fiorentino, A.; Monaco, P.; Oriano, P.; Pacifico, S. Annurcoic acid: A new antioxidant ursane triterpene from fruits of cv. Annurca apple. Food Chem. 2006, 98, 285–290. [Google Scholar] [CrossRef]
- Szakiel, A.; Pączkowski, C.; Pensec, F.; Bertsch, C. Fruit cuticular waxes as a source of biologically active triterpenoids. Phytochem. Rev. 2012, 11, 263–284. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Rupasinghe, H.P. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxidative Med. Cell. Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Azizi, F. Dietary polyphenols as potential nutraceuticals in management of diabetes: A review. J. Diabetes Metab. Disord. 2013, 12, 43. [Google Scholar] [CrossRef]
- de la Garza, A.L.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Olsen, H.T.; Stafford, G.I.; Van Staden, J.; Christensen, S.B.; Jäger, A.K. Isolation of the MAO-inhibitor naringenin from Mentha aquatica L. J. Ethnopharmacol. 2008, 117, 500–502. [Google Scholar] [CrossRef] [PubMed]
- Saaby, L.; Rasmussen, H.B.; Jäger, A.K. MAO-A inhibitory activity of quercetin from Calluna vulgaris (L.) Hull. J. Ethnopharmacol. 2009, 121, 178–181. [Google Scholar] [CrossRef]
- Hanamura, T.; Uchida, E.; Aoki, H. Skin-lightening effect of a polyphenol extract from Acerola (Malpighia emarginata DC.) fruit on UV-induced pigmentation. Biosci. Biotechnol. Biochem. 2008, 72, 3211–3218. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.W.; Chiang, H.M.; Lin, Y.C.; Wen, K.C. Natural products with skin-whitening effects. J. Food Drug Anal. 2008, 16, 2. [Google Scholar]
- Hiipakka, R.A.; Zhang, H.Z.; Dai, W.; Dai, Q.; Liao, S. Structure–activity relationships for inhibition of human 5α-reductases by polyphenols. Biochem. Pharmacol. 2002, 63, 1165–1176. [Google Scholar] [CrossRef]
- Innocenti, A.; Gülçin, I.; Scozzafava, A.; Supuran, C.T. Carbonic anhydrase inhibitors. Antioxidant polyphenols effectively inhibit mammalian isoforms I–XV. Bioorganic Med. Chem. Lett. 2010, 20, 5050–5053. [Google Scholar] [CrossRef]
- Yousefi-Manesh, H.; Hemmati, S.; Shirooie, S.; Nabavi, S.M.; Bonakdar, A.T.; Fayaznia, R.; Asgardoon, S.H.; Dehnavi, A.Z.; Ghafouri, M.; Nkuimi Wandjou, J.G.; et al. Protective effects of hydroalcoholic extracts from an ancient apple variety ‘Mela Rosa dei Monti Sibillini’against renal ischemia/reperfusion injury in rats. Food Funct. 2019, 10, 7544–7552. [Google Scholar] [CrossRef]
- Yousefi-Manesh, H.; Dehpour, A.R.; Ansari-Nasab, S.; Hemmati, S.; Sadeghi, M.A.; Shahraki, R.H.; Shirooie, S.; Nabavi, S.M.; Nkuimi Wandjou, J.G.; Sut, S.; et al. Hepatoprotective Effects of Standardized Extracts from an Ancient Italian Apple Variety (Mela Rosa dei Monti Sibillini) against Carbon Tetrachloride (CCl4)-Induced Hepatotoxicity in Rats. Molecules 2020, 25, 1816. [Google Scholar] [CrossRef]
- D’Abrosca, B.; Pacifico, S.; Cefarelli, G.; Mastellone, C.; Fiorentino, A. ‘Limoncella’apple, an Italian apple cultivar: Phenolic and flavonoid contents and antioxidant activity. Food Chem. 2007, 104, 1333–1337. [Google Scholar] [CrossRef]
- He, X.; Liu, R.H. Triterpenoids isolated from apple peels have potent antiproliferative activity and may be partially responsible for apple’s anticancer activity. J. Agric. Food Chem. 2007, 55, 4366–4370. [Google Scholar] [CrossRef] [PubMed]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclic triterpene distribution in various plants–rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [PubMed]
- Veeresham, C. Natural products derived from plants as a source of drugs. J. Adv. Pharm. Technol. Res. 2012, 3, 200–201. [Google Scholar] [CrossRef]
- Goncalves, S.; Romano, A. Inhibitory Properties of Phenolic Compounds Against Enzymes Linked with Human Diseases; IntechOpen: London, UK, 2017; p. 100. [Google Scholar]
- Birtic, S.; Régis, S.; Le Bourvellec, C.; Renard, C.M. Impact of air-drying on polyphenol extractability from apple pomace. Food Chem. 2019, 296, 142–149. [Google Scholar] [CrossRef] [PubMed]
- DeGeeter, M.; Williamson, B. Alternative agents in type 1 diabetes in addition to insulin therapy: Metformin, alpha-glucosidase inhibitors, pioglitazone, GLP-1 agonists, DPP-IV inhibitors, and SGLT-2 inhibitors. J. Pharm. Pract. 2016, 29, 144–159. [Google Scholar] [CrossRef] [PubMed]
- Ikarashi, N.; Takeda, R.; Ito, K.; Ochiai, W.; Sugiyama, K. The inhibition of lipase and glucosidase activities by acacia polyphenol. Evid. Based Complement Altern. Med. 2011, 2011, 272075. [Google Scholar] [CrossRef]
- McDougall, G.J.; Stewart, D. The inhibitory effects of berry polyphenols on digestive enzymes. Biofactors 2005, 23, 189–195. [Google Scholar] [CrossRef]
- Sugiyama, H.; Akazome, Y.; Shoji, T.; Yamaguchi, A.; Yasue, M.; Kanda, T.; Ohtake, Y. Oligomeric procyanidins in apple polyphenol are main active components for inhibition of pancreatic lipase and triglyceride absorption. J. Agric. Food Chem. 2007, 55, 4604–4609. [Google Scholar] [CrossRef]
- Herraiz Tomico, T.; Guillén Fuerte, H. Monoamine oxidase—A inhibition and associated antioxidant activity in plant extracts with potential antidepressant actions. BioMed Res. Int. 2018, 2018, 4810394. [Google Scholar] [CrossRef]
- Bouayed, J. Polyphenols: A potential new strategy for the prevention and treatment of anxiety and depression. Curr. Nutr. Food Sci. 2010, 6, 13–18. [Google Scholar] [CrossRef]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517S–520S. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.Z.; Hwang, I.W.; Chung, S.K. Enhancing polyphenol extraction from unripe apples by carbohydrate-hydrolyzing enzymes. J. Zhejiang Univ. Sci. B 2009, 10, 912. [Google Scholar] [CrossRef] [PubMed]
- Karaman, Ş.; Tütem, E.; Başkan, K.S.; Apak, R. Comparison of antioxidant capacity and phenolic composition of peel and flesh of some apple varieties. J. Sci. Food Agric. 2013, 93, 867–875. [Google Scholar] [CrossRef]
- Carpéné, C.; Les, F.; Cásedas, G.; Peiro, C.; Fontaine, J.; Chaplin, A.; Mercader, J.; López, V. Resveratrol anti-obesity effects: Rapid inhibition of adipocyte glucose utilization. Antioxidants 2019, 8, 74. [Google Scholar] [CrossRef]
- Rhee, I.K.; van de Meent, M.; Ingkaninan, K.; Verpoorte, R. Screening for acetylcholinesterase inhibitors from Amaryllidaceae using silica gel thin-layer chromatography in combination with bioactivity staining. J. Chromatogr. A 2001, 915, 217–223. [Google Scholar] [CrossRef]
- Senol, F.S.; Orhan, I.E.; Ozgen, U.; Renda, G.; Bulut, G.; Guven, L.; Karaoglan, S.E.; Sevindik, H.G.; Skalicka-Wozniak, K.; Caliskan, U.K.; et al. Memory-vitalizing effect of twenty-five medicinal and edible plants and their isolated compounds. S. Afr. J. Bot. 2016, 102, 102–109. [Google Scholar] [CrossRef]
| Dried Samples | Lyophilised Samples | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Hydroxybenzoic Acids | ||||||||||||||||
| Gallic Acid | 8.8 | 6.8 | 25.5 | 7.9 | 6.7 | 8.4 | 3.9 | 10.2 | 6.5 | 3.9 | 4.1 | 4.1 | 8.2 | 0.0 | 0.0 | 0.0 |
| Flavan-3-ols | ||||||||||||||||
| Catechin | 333.5 | 136.1 | 190.4 | 223.1 | 309.7 | 301.2 | 221.2 | 364.2 | 470.0 | 235.9 | 260.8 | 260.4 | 552.0 | 426.2 | 320.4 | 403.0 |
| Epicatechin | 2042.9 | 938.1 | 1280.1 | 1257.0 | 1772.0 | 2083.5 | 1630.1 | 2049.9 | 2735.5 | 1740.2 | 3059.2 | 3053.9 | 3999.0 | 3976.4 | 3007.5 | 2869.5 |
| Procyanidin B2 | 2041.4 | 855.0 | 858.5 | 874.0 | 1324.2 | 1177.9 | 1204.6 | 1201.6 | 2089.8 | 1655.9 | 2266.4 | 2262.5 | 2732.7 | 2421.4 | 2386.6 | 1995.1 |
| Procyanidin A2 | 252.4 | 230.1 | 209.0 | 280.9 | 224.6 | 174.0 | 174.9 | 222.1 | 301.5 | 272.7 | 715.7 | 714.4 | 565.0 | 503.8 | 562.8 | 267.5 |
| Anthocyanins | ||||||||||||||||
| Cyanidin 3-glucoside | 27.7 | 7.1 | 16.9 | 19.7 | 19.3 | 9.8 | 7.7 | 9.8 | 50.8 | 51.9 | 103.5 | 103.3 | 79.4 | 123.7 | 43.9 | 39.2 |
| Flavonols | ||||||||||||||||
| Rutin | 306.2 | 572.2 | 509.9 | 656.8 | 342.0 | 687.2 | 553.2 | 545.1 | 0.0 | 882.0 | 1034.5 | 1032.7 | 972.9 | 1384.9 | 1422.4 | 917.6 |
| Quercetin-3-O-galactoside | 101.1 | 235.6 | 359.3 | 496.8 | 360.4 | 444.0 | 220.2 | 363.3 | 11.5 | 429.8 | 751.3 | 749.9 | 1313.8 | 1158.8 | 922.3 | 650.6 |
| Kaempferol-3-glucoside | 258.2 | 293.0 | 408.9 | 253.6 | 340.8 | 197.2 | 126.3 | 272.1 | 358.6 | 486.9 | 811.8 | 810.3 | 1015.7 | 472.4 | 336.6 | 296.7 |
| Quercetin | 6.2 | 5.2 | 16.6 | 6.5 | 6.7 | 3.8 | 8.0 | 14.6 | 21.3 | 9.1 | 14.4 | 14.3 | 8.5 | 4.3 | 14.4 | 20.9 |
| Kaempferol | 2.8 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 2.6 | 6.9 | 0.0 | 0.0 | 0.0 | 0.0 | 7.9 | 0.0 | 6.0 |
| Hydroxycinnamic Acids | ||||||||||||||||
| Neochlorogenic Acid | 6.0 | 0.0 | 0.0 | 7.2 | 4.4 | 4.5 | 4.2 | 4.3 | 12.0 | 0.0 | 0.0 | 0.0 | 9.8 | 0.0 | 4.2 | 5.8 |
| Chlorogenic Acid | 120.9 | 1551.7 | 84.0 | 90.2 | 143.6 | 163.7 | 137.1 | 196.0 | 240.9 | 1726.3 | 97.8 | 97.7 | 154.9 | 167.2 | 109.8 | 156.6 |
| Caffeic Acid | 21.0 | 0.0 | 13.6 | 18.7 | 11.7 | 13.0 | 18.8 | 18.6 | 26.2 | 0.0 | 0.0 | 0.0 | 14.9 | 5.5 | 17.4 | 19.9 |
| p-Coumaric Acid | 4.8 | 7.6 | 0.0 | 4.7 | 3.4 | 2.2 | 0.0 | 0.0 | 2.3 | 0.0 | 0.0 | 0.0 | 6.2 | 3.7 | 2.1 | 0.0 |
| trans-Ferulic Acid | 3.7 | 2.2 | 9.6 | 6.0 | 7.3 | 2.9 | 6.1 | 8.0 | 13.8 | 5.1 | 39.7 | 39.7 | 15.3 | 13.0 | 10.7 | 5.9 |
| Dihydrochalcones | ||||||||||||||||
| Phloridzin | 1315.0 | 507.3 | 527.1 | 387.7 | 600.5 | 872.8 | 398.5 | 562.9 | 1809.5 | 892.0 | 1628.3 | 1625.5 | 1228.1 | 2012.1 | 730.2 | 773.8 |
| Phloretin | 16.1 | 0.0 | 10.9 | 3.2 | 3.2 | 10.4 | 11.7 | 0.0 | 3.7 | 0.0 | 27.4 | 27.4 | 3.3 | 6.1 | 3.1 | 0.0 |
| Total Polyphenols | 6868.8 | 5347.9 | 4520.3 | 4593.9 | 5480.3 | 6156.5 | 4726.6 | 5845.2 | 8160.7 | 8391.5 | 10,814.9 | 10,796.1 | 12,679.8 | 12,687.5 | 9894.5 | 8427.8 |
| Triterpenes | ||||||||||||||||
| Oleanolic Acid | 6301.5 | 3996.2 | 598.5 | 3361.9 | 2340.1 | 3132.9 | 6527.3 | 3543.2 | 811.6 | 10,835.5 | 321.1 | 320.5 | 1398.0 | 6194.2 | 4658.1 | 4278.2 |
| Ursolic Acid | 15,088.8 | 10,719.5 | 1361.7 | 5383.3 | 2121.7 | 7188.4 | 9006.2 | 7307.5 | 1911.8 | 7667.8 | 584.4 | 583.4 | 3366.3 | 12,541.8 | 8722.0 | 9069.4 |
| Total Triterpenes | 21,390.4 | 14,715.8 | 1960.2 | 8745.2 | 4461.8 | 10,321.4 | 15,533.5 | 10,850.6 | 2723.5 | 18503.3 | 905.5 | 903.9 | 4764.3 | 18,736.0 | 13,380.1 | 13,347.6 |
| Dried Samples | Lyophilised Samples | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Hydroxybenzoic Acids | ||||||||||||||||
| Gallic Acid | 4.8 | 5.7 | 3.9 | 3.8 | 5.6 | 6.4 | 4.1 | 11.8 | 2.4 | 2.2 | 0.0 | 2.7 | 2.0 | 2.0 | 5.7 | 4.9 |
| Flavan-3-ols | ||||||||||||||||
| Catechin | 183.5 | 131.7 | 261.8 | 144.9 | 285.3 | 336.6 | 271.3 | 366.1 | 214.5 | 164.8 | 176.2 | 184.9 | 340.1 | 236.7 | 195.1 | 416.9 |
| Epicatechin | 586.9 | 616.5 | 791.7 | 676.1 | 982.9 | 1208.7 | 820.5 | 1410.9 | 833.7 | 494.5 | 881.1 | 814.3 | 1292.2 | 738.7 | 769.3 | 1725.2 |
| Procyanidin B2 | 338.9 | 325.6 | 455.4 | 429.2 | 827.5 | 714.0 | 472.0 | 1015.6 | 431.0 | 387.0 | 538.6 | 471.5 | 784.3 | 503.7 | 535.1 | 1262.2 |
| Procyanidin A2 | 30.0 | 27.2 | 38.0 | 41.0 | 68.9 | 65.2 | 39.3 | 88.0 | 66.5 | 32.6 | 49.3 | 53.0 | 97.6 | 33.8 | 55.9 | 106.7 |
| Anthocyanins | ||||||||||||||||
| Cyanidin 3-glucoside | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Flavonols | ||||||||||||||||
| Rutin | 88.9 | 63.5 | 82.9 | 65.3 | 99.5 | 125.8 | 86.0 | 123.9 | 108.7 | 84.5 | 109.7 | 71.4 | 117.4 | 82.7 | 61.7 | 124.7 |
| Quercetin-3-O-galactoside | 7.1 | 3.8 | 8.7 | 8.1 | 14.8 | 8.0 | 9.0 | 13.1 | 9.9 | 4.1 | 8.8 | 9.1 | 14.8 | 6.8 | 10.7 | 20.4 |
| Kaempferol-3-glucoside | 24.6 | 17.5 | 36.9 | 24.7 | 48.5 | 30.3 | 38.3 | 36.4 | 37.0 | 19.3 | 49.2 | 27.4 | 73.1 | 32.2 | 22.7 | 55.4 |
| Quercetin | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Kaempferol | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Hydroxycinnamic Acids | ||||||||||||||||
| Neochlorogenic Acid | 2.7 | 2.4 | 3.0 | 1.9 | 4.3 | 3.3 | 3.1 | 5.8 | 3.1 | 3.5 | 2.3 | 4.0 | 4.3 | 4.3 | 4.7 | 7.1 |
| Chlorogenic Acid | 311.9 | 1438.7 | 306.5 | 136.0 | 260.8 | 492.0 | 317.7 | 529.6 | 384.2 | 1946.8 | 244.6 | 228.2 | 273.6 | 387.5 | 167.9 | 338.8 |
| Caffeic Acid | 14.0 | 0.0 | 9.8 | 16.1 | 15.2 | 21.5 | 10.1 | 0.0 | 14.6 | 0.0 | 14.6 | 14.3 | 26.5 | 15.9 | 21.0 | 16.5 |
| p-Coumaric Acid | 1.0 | 59 | 0.0 | 1.0 | 1.1 | 1.1 | 0.0 | 1.0 | 0.0 | 6.1 | 0.0 | 1.1 | 1.1 | 1.0 | 1.0 | 0.8 |
| trans-Ferulic Acid | 1.1 | 1.2 | 1.1 | 1.5 | 1.1 | 1.6 | 1.1 | 1.8 | 2.0 | 2.5 | 4.5 | 1.6 | 1.8 | 1.6 | 1.2 | 1.8 |
| Dihydrochalcones | ||||||||||||||||
| Phloridzin | 72.1 | 73.1 | 61.3 | 49.6 | 64.5 | 101.0 | 63.5 | 64.5 | 136.9 | 135.5 | 98.5 | 68.8 | 122.4 | 97.3 | 58.7 | 103.7 |
| Phloretin | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| TOTAL Polyphenols | 1667.6 | 2712.9 | 2060.9 | 1599.0 | 2679.7 | 3115.4 | 2135.9 | 3668.5 | 2244.3 | 3283.4 | 2177.4 | 1952.3 | 3151.2 | 2144.0 | 1910.7 | 4185.0 |
| Triterpenes | ||||||||||||||||
| Oleanolic Acid | 47.7 | 123.6 | 144.6 | 71.9 | 85.3 | 95.0 | 149.9 | 32.7 | 39.7 | 107.7 | 107.2 | 142.5 | 75.8 | 56.6 | 178.9 | 19.7 |
| Ursolic Acid | 40.6 | 29.8 | 77.4 | 29.4 | 137.7 | 36.2 | 80.2 | 48.3 | 35.5 | 17.3 | 47.2 | 82.9 | 90.7 | 58.0 | 23.3 | 37.5 |
| Total Triterpenes | 88.3 | 153.4 | 222.0 | 101.2 | 223.0 | 131.1 | 230.0 | 81.0 | 75.2 | 125.0 | 154.4 | 225.3 | 166.5 | 114.6 | 202.2 | 57.2 |
| IC50 Value in µg/mL (Mean ± SEM) | |||||
|---|---|---|---|---|---|
| Bioassay | DEPe | LEPe | DEPu | LEPu | Reference Inhibitor |
| α-glucosidase | 1417 ± 195 | 1056 ± 168 | - | - | 379 ± 74 (Acarbose) |
| Ns | - | **/*/-/- | |||
| Lipase | 3690 ± 2503 | 3375 ± 3690 | - | 4783 ± 237 | 0.90 ± 0.01 (Orlistat) |
| Ns | - | ns/ns/-/ns | |||
| MAO-A | 361 ± 11 | 291 ± 26 | 797 ± 4 | 650 ± 14 | 0.15 ± 0.02 (Clorgyline) |
| # | &&& | ****/****/****/**** | |||
| AChE | 1887 ± 211 | 2261 ± 574 | 3963 ± 431 | - | 0.56 ± 0.01 (Galantamine) |
| Ns | - | */*/***/- | |||
| TYR | - | 3419 ± 145 | 8953 ± 124 | 3690 ± 1154 | 3.81 ± 0.22 (Kojic acid) |
| - | &&& | -/*/****/** | |||
| Bioassay | DEPe | LEPe | DEPu | LEPu | ||||
|---|---|---|---|---|---|---|---|---|
| Phenol. | Triterp. | Phenol. | Triterp. | Phenol. | Triterp. | Phenol. | Triterp. | |
| α-GLU | 0.1142 | −0.4741 | 0.2428 | −0.09464 | −0.2745 | −0.0469 | 0.3865 | −0.2265 |
| ns | ns | ns | ns | ns | ns | ns | ns | |
| Lipase | −0.07635 | −0.3406 | 0.1867 | 0.2813 | −0.3416 | 0.9568 | 0.4261 | −0.1890 |
| ns | ns | ns | ns | ns | *** | ns | ns | |
| MAO-A | 0.1567 | 0.1037 | −0.2331 | −0.8241 | 0.5092 | −0.2526 | −0.4146 | 0.6902 |
| ns | ns | ns | * | ns | ns | ns | ns | |
| AChE | −0.2922 | 0.2110 | −0.1816 | −0.5390 | 0.4546 | −0.1949 | −0.1910 | −0.4477 |
| ns | ns | ns | ns | ns | ns | ns | ns | |
| TYR | 0.1816 | −0.1827 | 0.3528 | −0.3741 | 0.7924 | −0.1270 | 0.1498 | 0.02118 |
| ns | ns | ns | ns | * | ns | ns | ns | |
| Sample N. | Farmers | Variety | Altitude (m) | Graft | Municipality |
|---|---|---|---|---|---|
| 1 | Traini | classica | 500 | M111 | Montedinove |
| 2 | Botticelli | classica | 390 | M111 | Montottone |
| 3 | Orsolini | classica | 480 | M111 | Montedinove |
| 4 | Galli Stefano | classica | 250 | M9 M26 | Montedinove |
| 5 | Geminiani I | classica | 450 | M111 | Montedinove |
| 6 | Geminiani II | classica | 480 | M111 | Montedinove |
| 7 | Orsolini | pianella abruzzese | 480 | M111 | Montottone |
| 8 | Acciarri | classica | 350 | M 26 | Monterinaldo |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López, V.; Les, F.; Mevi, S.; Nkuimi Wandjou, J.G.; Cásedas, G.; Caprioli, G.; Maggi, F. Phytochemicals and Enzyme Inhibitory Capacities of the Methanolic Extracts from the Italian Apple Cultivar Mela Rosa dei Monti Sibillini. Pharmaceuticals 2020, 13, 127. https://doi.org/10.3390/ph13060127
López V, Les F, Mevi S, Nkuimi Wandjou JG, Cásedas G, Caprioli G, Maggi F. Phytochemicals and Enzyme Inhibitory Capacities of the Methanolic Extracts from the Italian Apple Cultivar Mela Rosa dei Monti Sibillini. Pharmaceuticals. 2020; 13(6):127. https://doi.org/10.3390/ph13060127
Chicago/Turabian StyleLópez, Víctor, Francisco Les, Serena Mevi, Joice Guileine Nkuimi Wandjou, Guillermo Cásedas, Giovanni Caprioli, and Filippo Maggi. 2020. "Phytochemicals and Enzyme Inhibitory Capacities of the Methanolic Extracts from the Italian Apple Cultivar Mela Rosa dei Monti Sibillini" Pharmaceuticals 13, no. 6: 127. https://doi.org/10.3390/ph13060127
APA StyleLópez, V., Les, F., Mevi, S., Nkuimi Wandjou, J. G., Cásedas, G., Caprioli, G., & Maggi, F. (2020). Phytochemicals and Enzyme Inhibitory Capacities of the Methanolic Extracts from the Italian Apple Cultivar Mela Rosa dei Monti Sibillini. Pharmaceuticals, 13(6), 127. https://doi.org/10.3390/ph13060127










