In-Vitro Evaluation of Antioxidant, Antiproliferative and Photo-Protective Activities of Benzimidazolehydrazone Derivatives
Abstract
1. Introduction
2. Results and Discussion
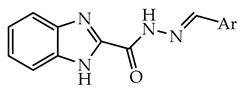
2.1. Chemistry
2.2. Antioxidant Activity
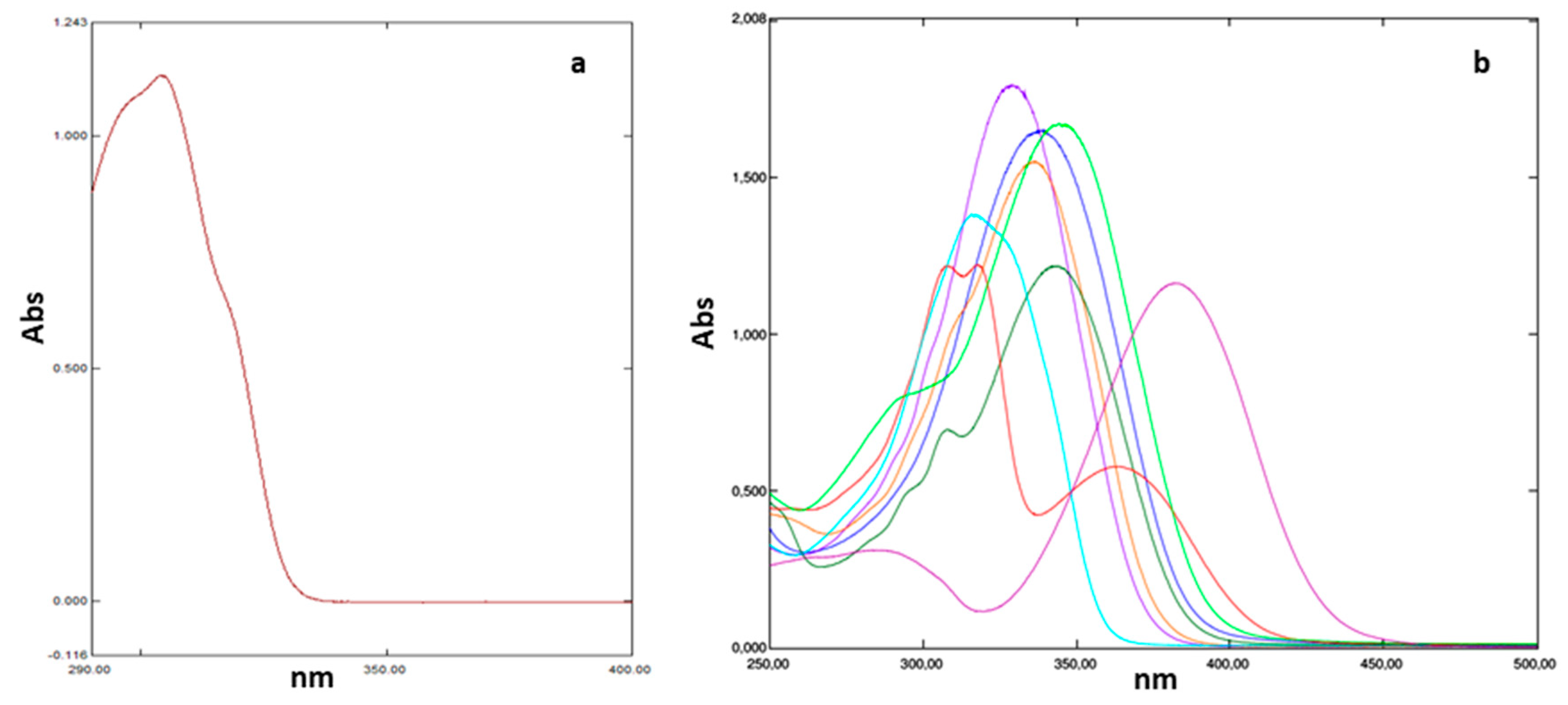
2.3. Evaluation of UV-Filtering Parameters
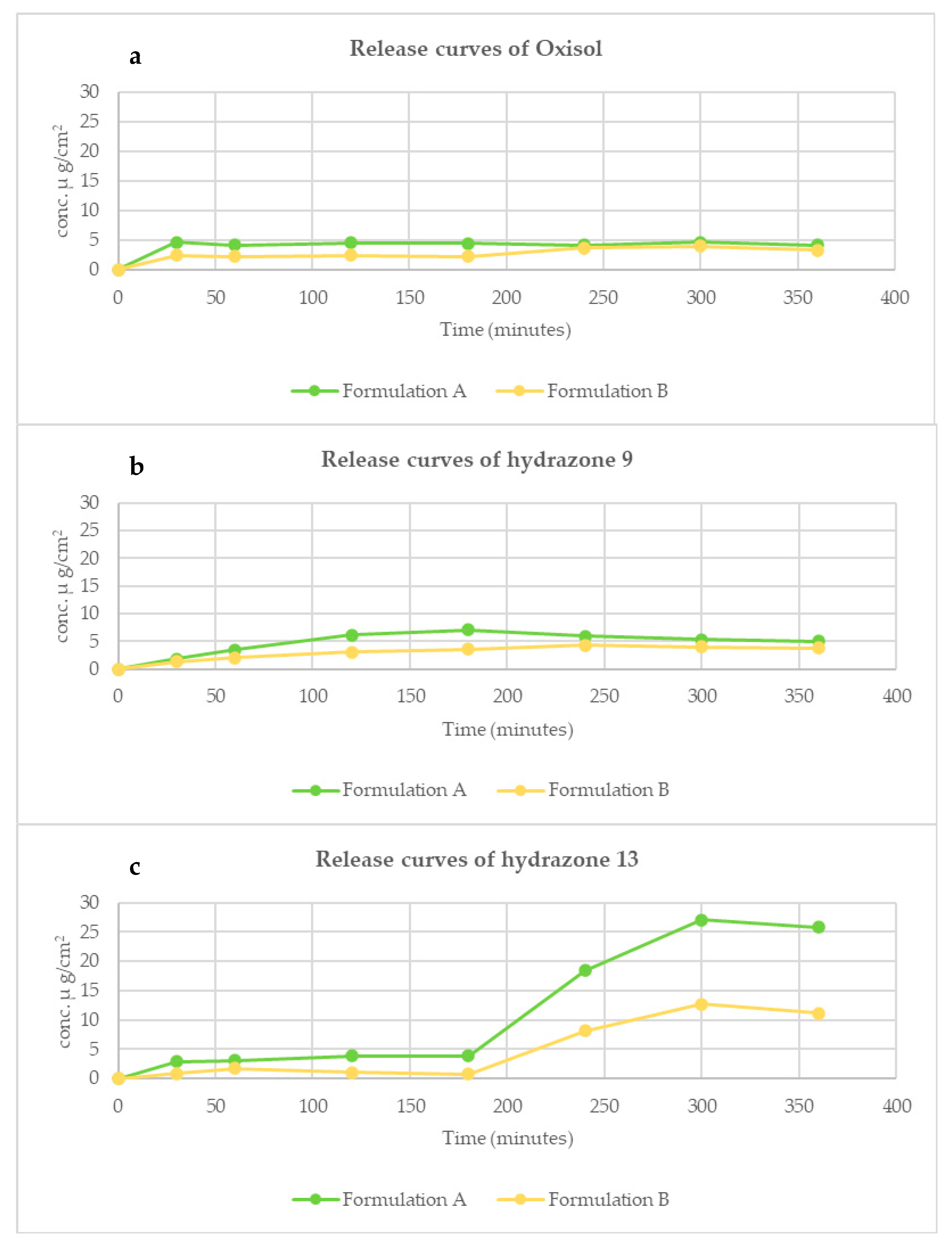
2.4. Evaluation of Release In Vitro
2.5. Antiproliferative Activity
3. Materials and Methods
3.1. General Methods
3.2. Antioxidant Activity Evaluation
3.2.1. DPPH Test
3.2.2. FRAP Test
3.2.3. Oxygen Radical Absorbance Capacity (ORAC)
3.3. Evaluation of Filtering Parameters
3.4. Preparation of Formulations
3.5. Franz Cells Apparatus
3.6. Growth Inhibition Assays
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gaba, M.; Mohan, C. Development of drugs based on imidazole and benzimidazole bioactive heterocycles: Recent advances and future directions. Med. Chem. Res. 2015, 25, 173–210. [Google Scholar] [CrossRef]
- Tahlan, S.; Kumar, S.; Narasimhan, B. Pharmacological significance of heterocyclic 1H-benzimidazole scaffolds: A review. BMC Chem. 2019, 13, 101–121. [Google Scholar] [CrossRef] [PubMed]
- Boggu, P.R.; Kim, Y.; Jung, S.-H. Discovery of benzimidazole analogs as a novel interleukin-5 inhibitors. Eur. J. Med. Chem. 2019, 181, 111574. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.D.; Bagal, S.; Blackwell, P.; Blakemore, D.C.; Brown, B.; Bungay, P.J.; Corless, M.; Crawforth, J.; Fengas, D.; Fenwick, D.R.; et al. The discovery and optimization of benzimidazoles as selective NaV1.8 blockers for the treatment of pain. Bioorg. Med. Chem. 2019, 27, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Huang, M.; Li, A.; Zhao, F.; Li, D.; Liu, D.; Zhao, L. Design, synthesis and biological evaluation of benzimidazole derivatives as novel human Pin1 inhibitors. Bioorg. Med. Chem. Lett. 2018, 29, 1859–1863. [Google Scholar] [CrossRef] [PubMed]
- Zaman, K.; Rahim, F.; Taha, M.; Ullah, H.; Wadood, A.; Nawaz, M.; Khan, F.; Wahab, Z.; Shah, S.A.A.; Rehman, A.U.; et al. Synthesis, in vitro urease inhibitory potential and molecular docking study of Benzimidazole analogues. Bioorg. Chem. 2019, 89, 103024. [Google Scholar] [CrossRef]
- Zawawi, N.K.N.A.; Taha, M.; Ahmat, N.; Wadood, A.; Ismail, N.H.; Rahim, F.; Azam, S.S.; Abdullah, N. Benzimidazole derivatives as new α-glucosidase inhibitors and in silico studies. Bioorg. Chem. 2016, 64, 29–36. [Google Scholar] [CrossRef]
- Bansal, Y.; Silakari, O. The therapeutic journey of benzimidazoles: A review. Bioorg. Med. Chem. 2012, 20, 6208–6236. [Google Scholar] [CrossRef]
- Mavrova, A.T.; Yancheva, D.; Anastassova, N.; Anichina, K.; Zvezdanović, J.; Đorđević, A.; Markovic, D.; Šmelcerović, A. Synthesis, electronic properties, antioxidant and antibacterial activity of some new benzimidazoles. Bioorg. Med. Chem. 2015, 23, 6317–6326. [Google Scholar] [CrossRef]
- Özil, M.; Parlak, C.; Baltaş, N. A simple and efficient synthesis of benzimidazoles containing piperazine or morpholine skeleton at C-6 position as glucosidase inhibitors with antioxidant activity. Bioorg. Chem. 2018, 76, 468–477. [Google Scholar] [CrossRef]
- Bino, A.; Baldisserotto, A.; Scalambra, E.; Dissette, V.; Vedaldi, D.E.; Salvador, A.; Durini, E.; Manfredini, S.; Vertuani, S. Design, synthesis and biological evaluation of novel hydroxy-phenyl-1H-benzimidazoles as radical scavengers and UV-protective agents. J. Enzym. Inhib. Med. Chem. 2017, 32, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Cindrić, M.; Sović, I.; Mioč, M.; Hok, L.; Boček, I.; Roškarić, P.; Butković, K.; Martin-Kleiner, I.; Starčević, K.; Vianello, R.; et al. Experimental and Computational Study of the Antioxidative Potential of Novel Nitro and Amino Substituted Benzimidazole/Benzothiazole-2-Carboxamides with Antiproliferative Activity. Antioxidants 2019, 8, 477. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazúr, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Karaaslan, C.; Kadri, H.; Coban, T.; Suzen, S.; Westwell, A.D. Synthesis and antioxidant properties of substituted 2-phenyl-1H-indoles. Bioorg. Med. Chem. Lett. 2013, 23, 2671–2674. [Google Scholar] [CrossRef] [PubMed]
- Dröge, W. Free Radicals in the Physiological Control of Cell Function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Halliwell, B. Free Radicals and Antioxidants: A Personal View. Nutr. Rev. 2009, 52, 253–265. [Google Scholar] [CrossRef]
- Halliwell, B. Antioxidants in Human Health and Disease. Annu. Rev. Nutr. 1996, 16, 33–50. [Google Scholar] [CrossRef]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free Radicals, Antioxidants in Disease and Health. Int. J. Biomed. Sci. IJBS 2008, 4, 89–96. [Google Scholar]
- Valko, M.; Rhodes, C.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- Khan, K.M.; Shah, Z.; Ahmad, V.U.; Khan, M.; Taha, M.; Rahim, F.; Ali, S.; Ambreen, N.; Perveen, S.; Choudhary, M.I.; et al. 2,4,6-Trichlorophenylhydrazine Schiff bases as DPPH radical and super oxide anion scavengers. Med. Chem. 2012, 8, 452–461. [Google Scholar] [CrossRef]
- Kareem, H.S.; Ariffin, A.; Nordin, N.; Heidelberg, T.; Abdul-Aziz, A.; Kong, K.W.; Yehye, W.A. Correlation of antioxidant activities with theoretical studies for new hydrazone compounds bearing a 3,4,5-trimethoxy benzyl moiety. Eur. J. Med. Chem. 2015, 103, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Emami, S.; Esmaili, Z.; Dehghan, G.; Bahmani, M.; Hashemi, S.M.; Mirzaei, H.; Shokrzadeh, M.; Moradi, S.E. Acetophenone benzoylhydrazones as antioxidant agents: Synthesis, in vitro evaluation and structure-activity relationship studies. Food Chem. 2018, 268, 292–299. [Google Scholar] [CrossRef]
- Maltarollo, V.G.; De Resende, M.F.; Kronenberger, T.; Lino, C.I.; Sampaio, M.C.P.D.; Pitta, M.G.D.R.; Rêgo, M.J.B.D.M.; Labanca, R.A.; Oliveira, R.B. In vitro and in silico studies of antioxidant activity of 2-thiazolylhydrazone derivatives. J. Mol. Graph. Model. 2019, 86, 106–112. [Google Scholar] [CrossRef]
- Baldisserotto, A.; Demurtas, M.; Lampronti, I.; Moi, D.; Balboni, G.; Vertuani, S.; Manfredini, S.; Onnis, V. Benzofuran hydrazones as potential scaffold in the development of multifunctional drugs: Synthesis and evaluation of antioxidant, photoprotective and antiproliferative activity. Eur. J. Med. Chem. 2018, 156, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Demurtas, M.; Baldisserotto, A.; Lampronti, I.; Moi, D.; Balboni, G.; Pacifico, S.; Vertuani, S.; Manfredini, S.; Onnis, V. Indole derivatives as multifunctional drugs: Synthesis and evaluation of antioxidant, photoprotective and antiproliferative activity of indole hydrazones. Bioorg. Chem. 2019, 85, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Baldisserotto, A.; Demurtas, M.; Lampronti, I.; Tacchini, M.; Moi, D.; Balboni, G.; Pacifico, S.; Vertuani, S.; Manfredini, S.; Onnis, V. Synthesis and evaluation of antioxidant and antiproliferative activity of 2-arylbenzimidazoles. Bioorg. Chem. 2020, 94, 103396. [Google Scholar] [CrossRef] [PubMed]
- Onnis, V.; Demurtas, M.; Deplano, A.; Balboni, G.; Baldisserotto, A.; Manfredini, S.; Pacifico, S.; Liekens, S.; Balzarini, J. Design, Synthesis and Evaluation of Antiproliferative Activity of New Benzimidazolehydrazones. Molecules 2016, 21, 579. [Google Scholar] [CrossRef] [PubMed]
- Belkheiri, N.; Bouguerne, B.; Bedos-Belval, F.; Duran, H.; Bernis, C.; Salvayre, R.; Negre-Salvayre, A.; Baltas, M. Synthesis and antioxidant activity evaluation of a syringic hydrazones family. Eur. J. Med. Chem. 2010, 45, 3019–3026. [Google Scholar] [CrossRef] [PubMed]
- Sambandan, D.R.; Ratner, D. Sunscreens: An overview and update. J. Am. Acad. Dermatol. 2011, 64, 748–758. [Google Scholar] [CrossRef]
- Diffey, B.L.; Robson, J. A new substrate to measure sunscreen protection factors throughout the ultraviolet spectrum. J. Soc. Cosmet. Chem. 1989, 40, 127–133. [Google Scholar]
- Manfredini, S.; Vertuani, S.; Scalambra, E. New Dualistic Molecules Having UV Radiation Filtering Ability at Wide Spectrum and Potent Damping Activity of the Reactivity of Free Radicals (Radicals Scavenging). WO Patent 2013/102843,A2, 1 November 2013. [Google Scholar]
- Chinembiri, T.N.; Du Plessis, L.; Gerber, M.; Hamman, J.H.; Du Plessis, J. Review of Natural Compounds for Potential Skin Cancer Treatment. Molecules 2014, 19, 11679–11721. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, J.; Rangarajan, M.; Shao, Y.; Lavoie, E.J.; Huang, T.-C.; Ho, C.-T. Antioxidative Phenolic Compounds from Sage (Salvia officinalis). J. Agric. Food Chem. 1998, 46, 4869–4873. [Google Scholar] [CrossRef]
- Xu, G.; Ye, X.; Chen, J.; Liu, D. Effect of Heat Treatment on the Phenolic Compounds and Antioxidant Capacity of Citrus Peel Extract. J. Agric. Food Chem. 2007, 55, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Pessina, F.; Marazova, K.; Ninfali, P.; Avanzi, L.; Manfredini, S.; Sgaragli, G. In vitro neuroprotection by novel antioxidants in guinea-pig urinary bladder subjected to anoxia-glucopenia/reperfusion damage. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2004, 370, 521–528. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free. Radic. Boil. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Matts, P.J.; Alard, V.; Brown, M.W.; Ferrero, L.; Gers-Barlag, H.; Issachar, N.; Moyal, D.; Wolber, R. The COLIPAin vitroUVA method: A standard and reproducible measure of sunscreen UVA protection. Int. J. Cosmet. Sci. 2010, 32, 35–46. [Google Scholar] [CrossRef]
- Lampronti, I.; Martello, D.; Gambari, R.; Borgatti, M.; Lambertini, E.; Piva, R.; Jabbar, S.; Choudhuri, M.S.K.; Khan, M.T.H.; Gambari, R. In vitro antiproliferative effects on human tumor cell lines of extracts from the Bangladeshi medicinal plant Aegle marmelos Correa. Phytomedicine 2003, 10, 300–308. [Google Scholar] [CrossRef]
- Spagnoletti, A.; Guerrini, A.; Tacchini, M.; Vinciguerra, V.; Leone, C.; Maresca, I.; Simonetti, G.; Sacchetti, G.; Angiolella, L. Chemical Composition and Bio-efficacy of Essential Oils from Italian Aromatic Plants: Mentha suaveolens, Coridothymus capitatus, Origanum hirtum and Rosmarinus officinalis. Nat. Prod. Commun. 2016, 11, 1517–1520. [Google Scholar] [CrossRef]
 | ||||
|---|---|---|---|---|
| Compound | Aryl | DPPH a µmolTE/g | FRAP a µmolTE/g | ORAC a µmolTE/g |
| Ferulic acid | 4379.0 ± 9.8 | 6872.6 ± 9.7 | 15,906.4 ± 14.2 | |
| 3 | 2-OH-phenyl | <23.2 b | 48.0 ± 2.6 | - |
| 4 | 3-OH-phenyl | <13.0 b | 1023.3 ± 9.4 | 21,808.0 ± 99.1 |
| 5 | 4-OH-phenyl | 29.6 ± 0.1 | 1040.1 ± 11.2 | 30,911.3± 36.4 |
| 6 | 2,4-(OH)2-phenyl | 200.6 ± 1.5 | 286.7 ± 10.3 | 9296.8 ± 72.3 |
| 7 | 2,5-(OH)2-phenyl | 9387.9 ± 13.8 | 5330.6 ± 29.6 | 17,856.9 ± 35.1 |
| 8 | 2,3,4-(OH)3-phenyl | 10,945.2 ± 38.5 | 10,064.6 ± 24.6 | 4221.5 ± 10.5 |
| 9 | 2,4,6-(OH)3-phenyl | 192.6 ± 3.5 | 4071.7 ± 10.0 | 4098.6 ± 39.1 |
| 10 | 2-OH-4-OMe-phenyl | 201.2 ± 4.9 | 231.9 ± 5.5 | - |
| 11 | 2-OH-3-OEt-phenyl | 38.1 ± 1.6 | 104.5 ± 2.1 | - |
| 12 | 3-OH-4-OMe-phenyl | 171.5 ± 6.1 | 3418.8 ± 15.2 | 17,170.7 ± 18.4 |
| 13 | 2-OH-4-N(Et)2-phenyl | 1065.0 ± 5.9 | 3525.4 ± 13.4 | 754.4 ± 19.0 |
| 14 | 2-OH-5-Cl-phenyl | << 13.29 b | 54.8 ± 1.3 | - |
| 15 | 2-OH-5-Br-phenyl | 28.8 ± 1.2 | 81.7 ±2.7 | - |
| Compound | SPF | UVA/UVB | UVAPF0 | λc (nm) |
|---|---|---|---|---|
| PBSA | 3.42 | 0.29 | 1.04 | 322 |
| 4 | 11.54 | 0.27 | 1.60 | 343 |
| 5 | 12.32 | 0.75 | 2.95 | 353 |
| 6 | 4.81 | 1.26 | 4.84 | 366 |
| 7 | 11.23 | 0.32 | 3.24 | 377 |
| 8 | 8.12 | 2.23 | 6.79 | 366 |
| 9 | 8.34 | 2.27 | 10.65 | 370 |
| 12 | 8.95 | 1.13 | 3.60 | 358 |
| 13 | 1.57 | 1.18 | 3.99 | 394 |
| Compound | IC50 (μM) | Compound | IC50 (μM) | |
|---|---|---|---|---|
| Colo-38 | Colo-38 | Ha-Cat | ||
| 4 | 362.63 ± 15.05 | 8 | 46.6 2 ± 3.57 | |
| 5 | 216.16 ± 40.21 | 9 | 459.59 ± 103.87 | |
| 6 | 46.62 ± 0.82 | 12 | 0.84 ± 0.03 | 53.16 ± 4.33 |
| 7 | 35.49 ± 0.42 | 13 | 0.50 ± 0.12 | 5.03 ± 0.76 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldisserotto, A.; Demurtas, M.; Lampronti, I.; Tacchini, M.; Moi, D.; Balboni, G.; Vertuani, S.; Manfredini, S.; Onnis, V. In-Vitro Evaluation of Antioxidant, Antiproliferative and Photo-Protective Activities of Benzimidazolehydrazone Derivatives. Pharmaceuticals 2020, 13, 68. https://doi.org/10.3390/ph13040068
Baldisserotto A, Demurtas M, Lampronti I, Tacchini M, Moi D, Balboni G, Vertuani S, Manfredini S, Onnis V. In-Vitro Evaluation of Antioxidant, Antiproliferative and Photo-Protective Activities of Benzimidazolehydrazone Derivatives. Pharmaceuticals. 2020; 13(4):68. https://doi.org/10.3390/ph13040068
Chicago/Turabian StyleBaldisserotto, Anna, Monica Demurtas, Ilaria Lampronti, Massimo Tacchini, Davide Moi, Gianfranco Balboni, Silvia Vertuani, Stefano Manfredini, and Valentina Onnis. 2020. "In-Vitro Evaluation of Antioxidant, Antiproliferative and Photo-Protective Activities of Benzimidazolehydrazone Derivatives" Pharmaceuticals 13, no. 4: 68. https://doi.org/10.3390/ph13040068
APA StyleBaldisserotto, A., Demurtas, M., Lampronti, I., Tacchini, M., Moi, D., Balboni, G., Vertuani, S., Manfredini, S., & Onnis, V. (2020). In-Vitro Evaluation of Antioxidant, Antiproliferative and Photo-Protective Activities of Benzimidazolehydrazone Derivatives. Pharmaceuticals, 13(4), 68. https://doi.org/10.3390/ph13040068









