Additive Effect of a Combination of Artocarpus lakoocha and Glycyrrhiza glabra Extracts on Tyrosinase Inhibition in Melanoma B16 Cells
Abstract
1. Introduction
2. Results
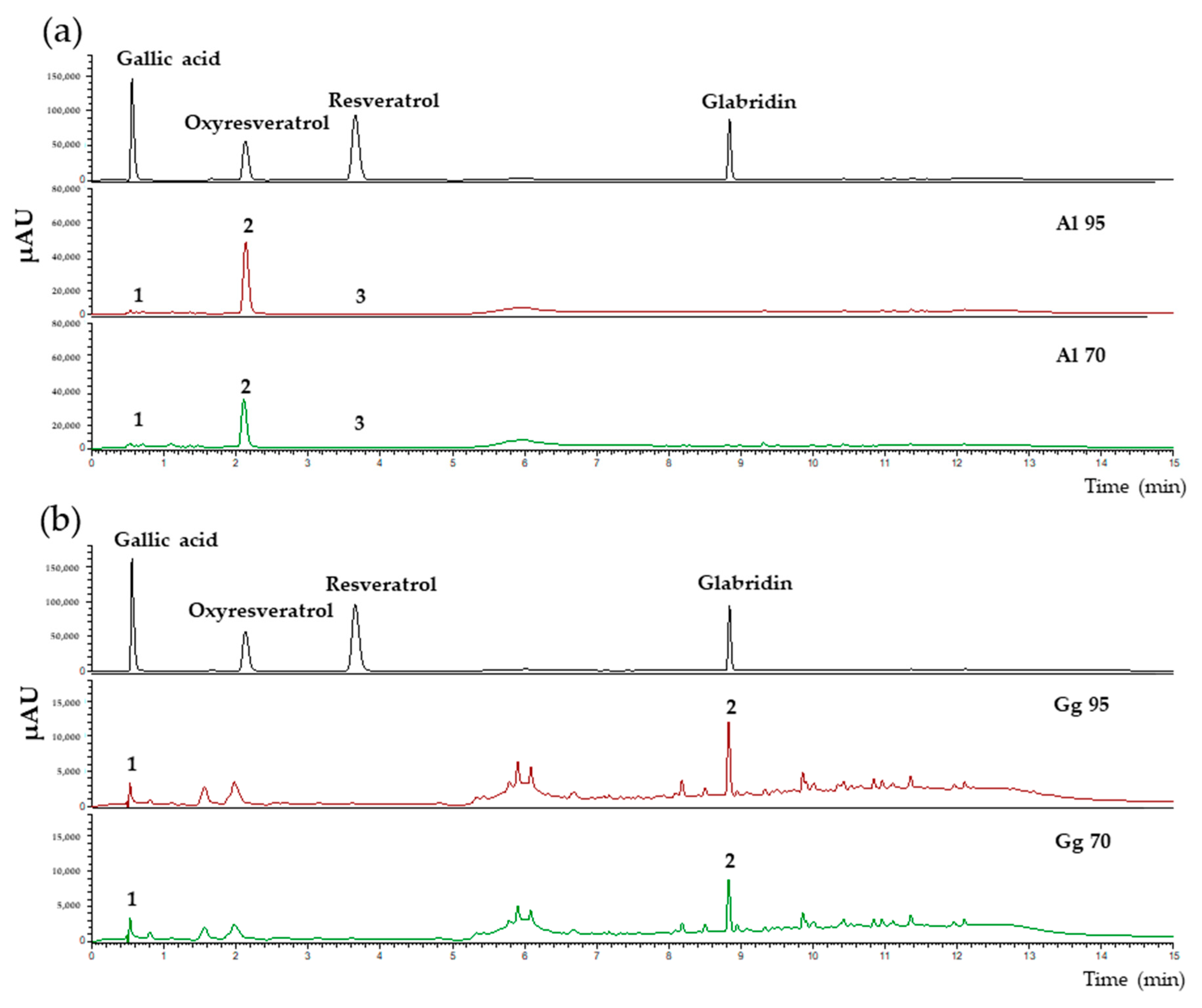
2.1. Chemical Profiles, Total Phenolic, Flavonoid Contents Antioxidant and Tyrosinase Inhibitory Activities in Extracts of A. lakoocha and G. glabra
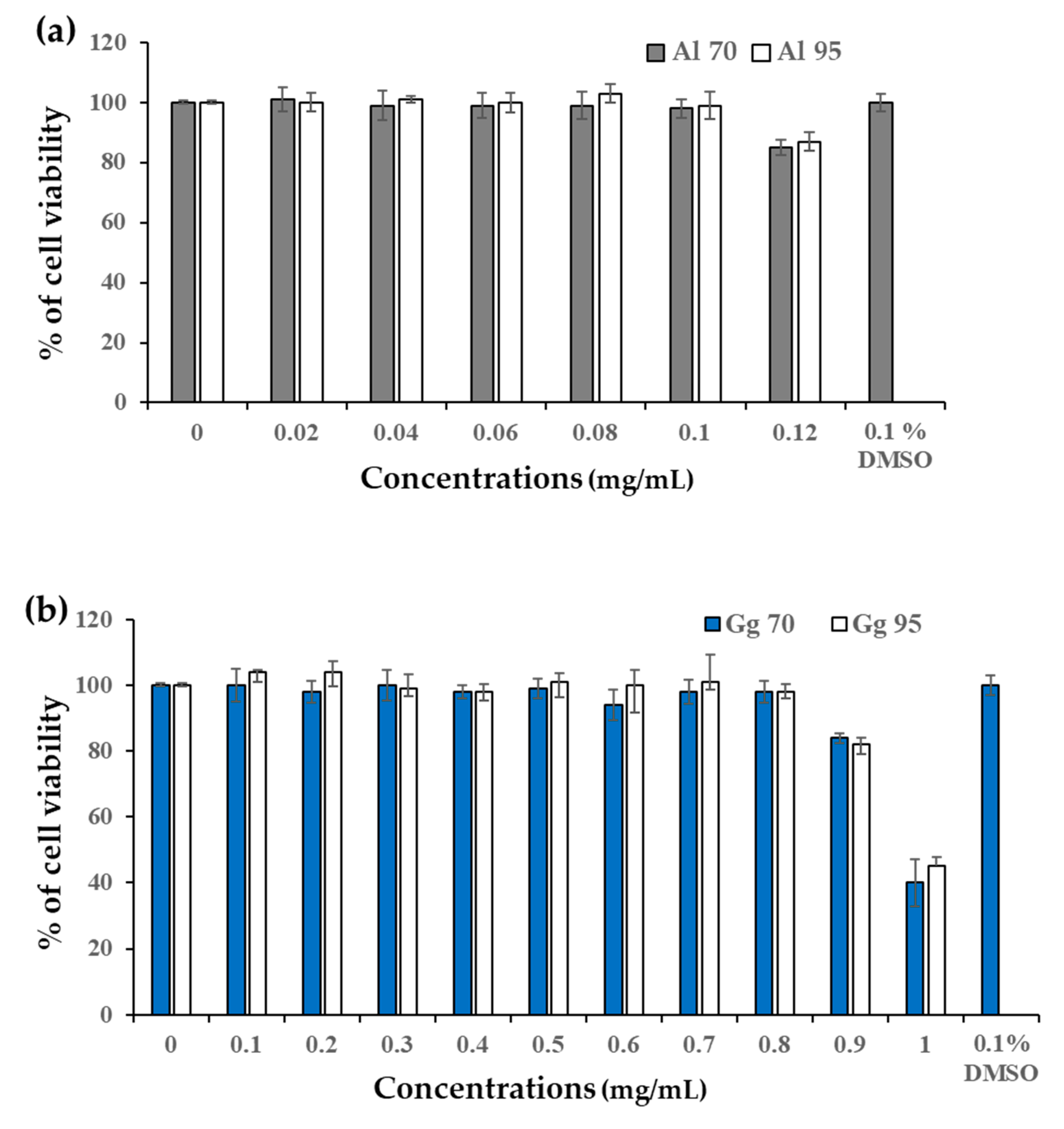
2.2. Cytotoxicity of A. lakoocha and G. glabra Extracts in B16 Cells
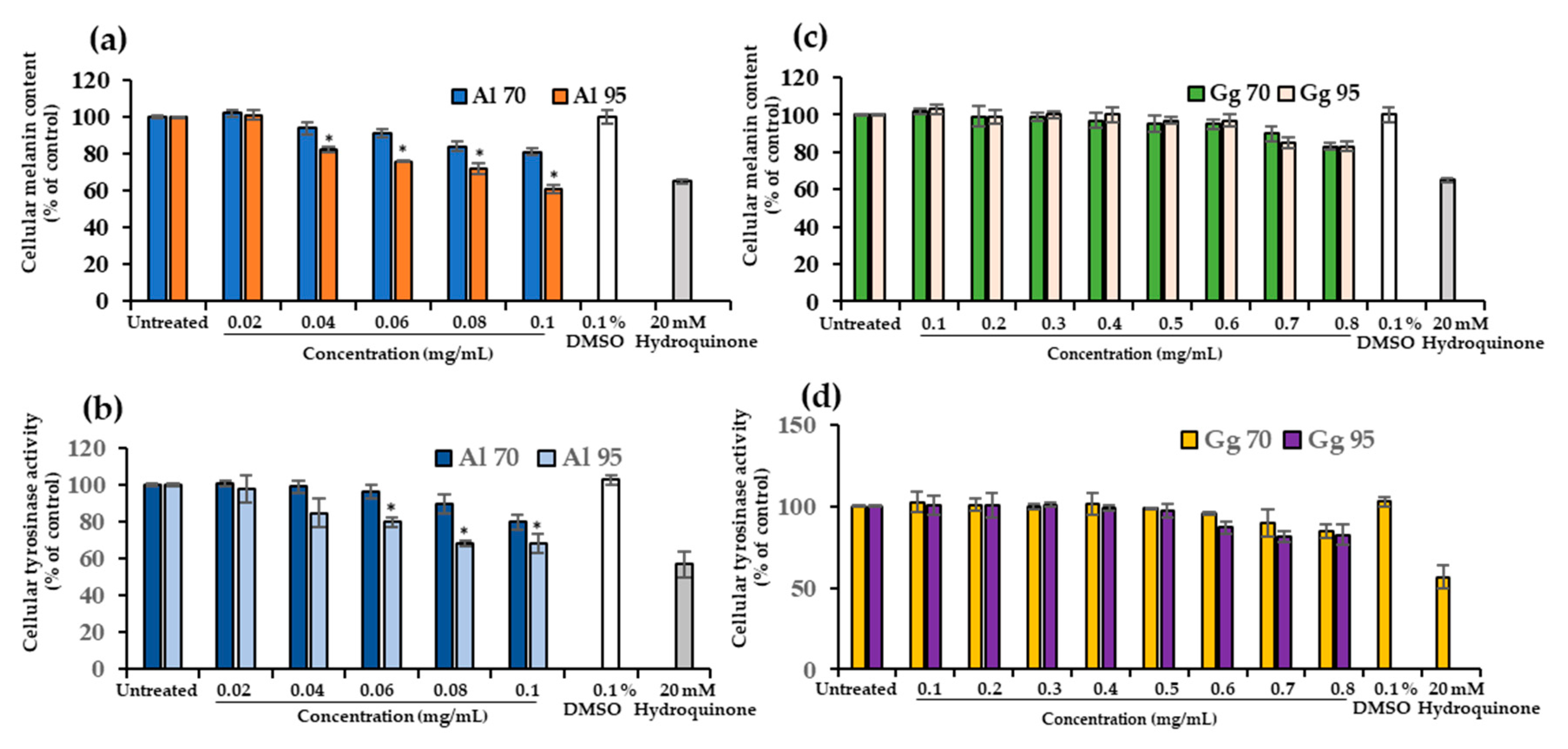
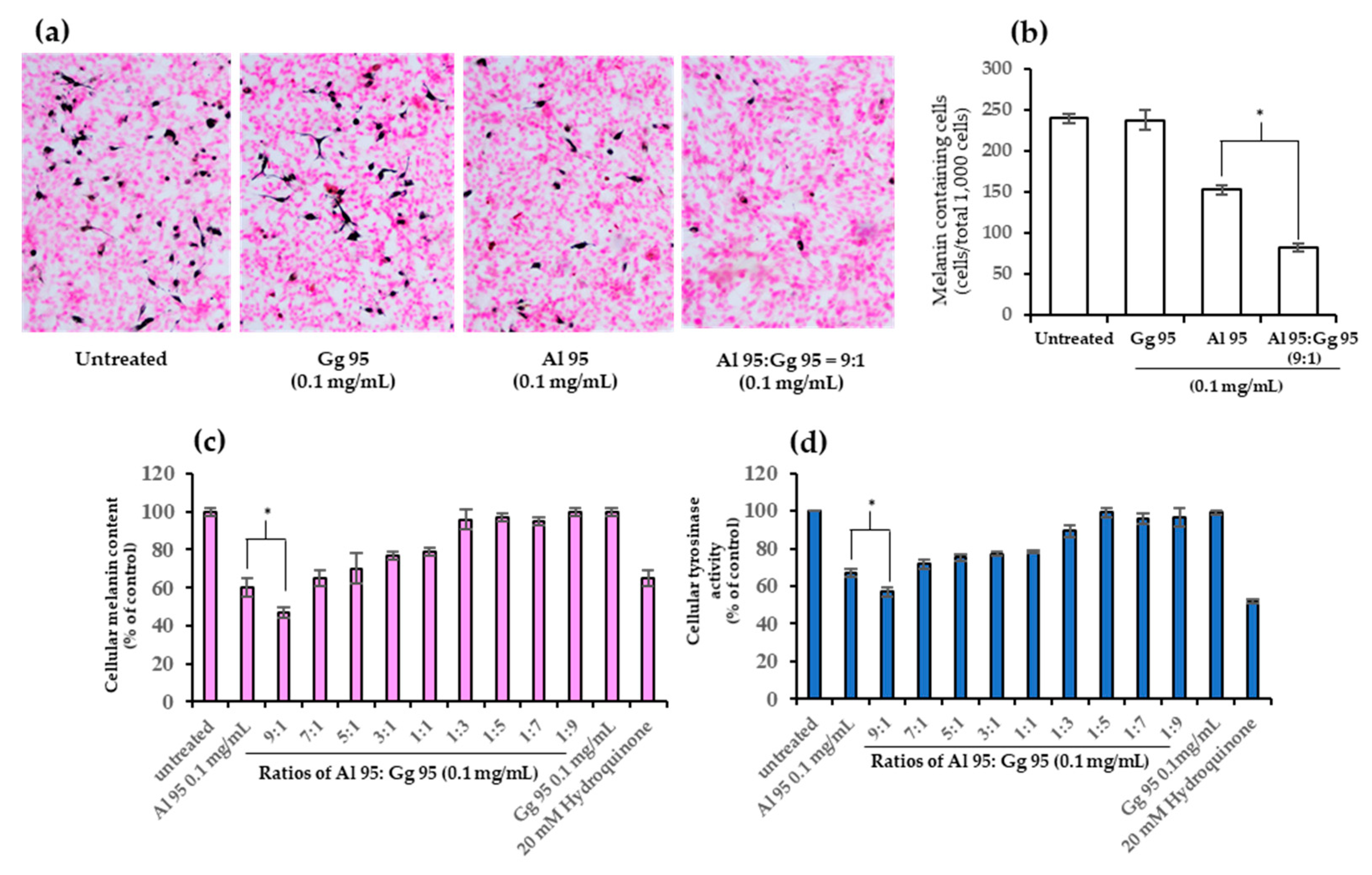
2.3. A. lakoocha and G. glabra Extracts Decreased Cellular Melanin Content and Tyrosinase Activity
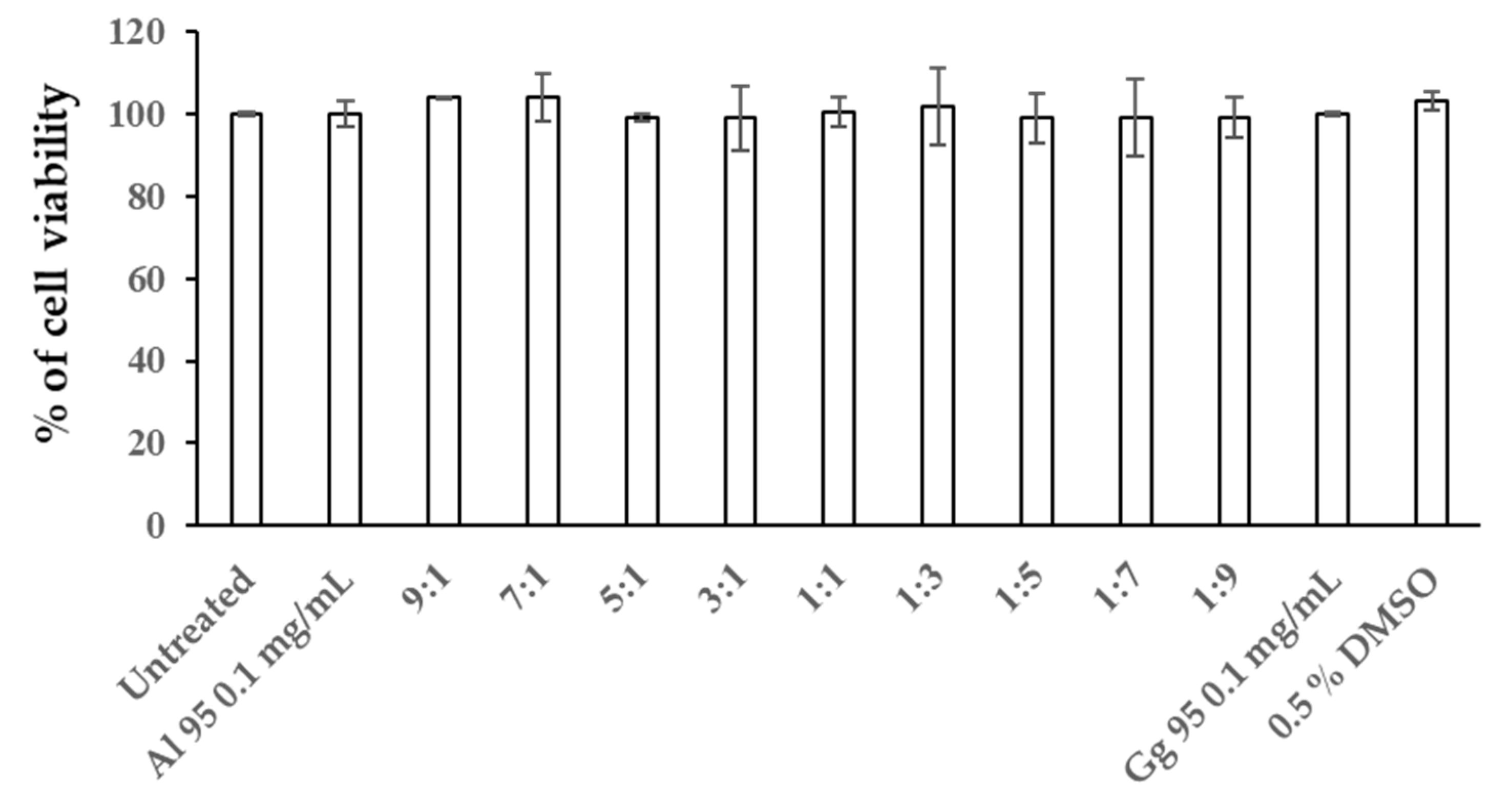
2.4. Combined Effects of A. lakoocha and G. glabra Extracts on Cellular Melanin Content and Tyrosinase Activity
2.5. Effect of Combined A. lakoocha and G. glabra Extracts on Proteins Related to Melanogenesis in B16 Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Materials and Extraction
4.3. Determination of Total Phenolic Content
4.4. Determination of Flavonoid Content
4.5. Phytochemical Screening Assay
4.6. 2,2-di-phenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Assay
4.7. Mushroom Tyrosinase Assays
4.8. Cell Viability Assay
4.9. Melanin Content Assay
4.10. Cellular Tyrosinase Activity Assay
4.11. Evaluation of Additive Effects of Al and Gg Extracts on Cellular Melanin Content and Tyrosinase Activity
4.12. Western Blot Analysis
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Meyskens, F.L., Jr.; Farmer, P.; Fruehauf, J.P. Redox regulation in human melanocytes and melanoma. Pigment Cell Res. 2001, 14, 148–154. [Google Scholar] [CrossRef]
- Kobayashi, T.; Vieira, W.D.; Potterf, B.; Sakai, C.; Imokawa, G.; Hearing, V.J. Modulation of melanogenic protein expression during the switch from eu- to pheomelanogenesis. J. Cell Sci. 1995, 108, 2301–2309. [Google Scholar]
- Kim, S.S.; Kim, M.J.; Choi, Y.H.; Kim, B.K.; Kim, K.S.; Park, K.J.; Park, S.M.; Lee, N.; Hyun, C.G. Down-regulation of tyrosinase, TRP-1, TRP-2 and MITF expressions by citrus press-cakes in murine B16 F10 melanoma. Asian Pac. J. Trop. Biomed. 2013, 3, 617–622. [Google Scholar] [CrossRef]
- Land, E.J.; Ramsden, C.A.; Riley, P.A. Tyrosinase autoactivation and the chemistry of ortho-quinone amines. Acc. Chem. Res. 2003, 36, 300–308. [Google Scholar] [CrossRef]
- Tachibana, M. Cochlear melanocytes and MITF signaling. J. Investig. Dermatol. Symp. Proc. 2001, 6, 95–98. [Google Scholar] [CrossRef]
- Yen, F.L.; Wang, M.C.; Liang, C.J.; Ko, H.H.; Lee, C.W. Melanogenesis Inhibitor(s) from Phyla nodiflora Extract. Evid. Based Complement. Alternat. Med. 2012, 2012, 867494. [Google Scholar] [CrossRef]
- Kobayashi, T.; Urabe, K.; Winder, A.; Jiménez-Cervantes, C.; Imokawa, G.; Brewington, T.; Solano, F.; García-Borrón, J.C.; Hearing, V.J. Tyrosinase related protein 1 (TRP1) functions as a DHICA oxidase in melanin biosynthesis. EMBO J. 1994, 13, 5818–5825. [Google Scholar] [CrossRef]
- Ye, Y.; Chu, J.H.; Wang, H.; Xu, H.; Chou, G.X.; Leung, A.K.; Fong, W.F.; Yu, Z.L. Involvement of p38 MAPK signaling pathway in the anti-melanogenic effect of San-bai-tang, a Chinese herbal formula, in B16 cells. J. Ethnopharmacol. 2010, 132, 533–535. [Google Scholar] [CrossRef]
- Levy, C.; Khaled, M.; Fisher, D.E. MITF: Master regulator of melanocyte development and melanoma oncogene. Trends Mol. Med. 2006, 12, 406–414. [Google Scholar] [CrossRef]
- Tsang, T.F.; Ye, Y.; Tai, W.C.; Chou, G.X.; Leung, A.K.; Yu, Z.L.; Hsiao, W.L.J. Inhibition of the p38 and PKA signaling pathways is associated with the anti-melanogenic activity of Qian-wang-hong-bai-san, a Chinese herbal formula, in B16 cells. Ethnopharmacology 2012, 141, 622–628. [Google Scholar] [CrossRef]
- Buscà, R.; Ballotti, R. Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res. 2000, 13, 60–69. [Google Scholar] [CrossRef]
- Chang, T.S. An updated review on tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2400–2475. [Google Scholar] [CrossRef]
- Huang, W.Y.; Lee, P.C.; Huang, L.K.; Lu, L.P.; Liao, W.C. Stability studies of ascorbic acid 2-glucoside in cosmetic lotion using surface response methodology. Bioorg. Med. Chem. Lett. 2013, 23, 1583–1587. [Google Scholar] [CrossRef]
- Shivhare, A.; Ambrose, S.J.; Zhang, H.; Purves, R.W.; Scott, R.W. Stable and recyclable Au25 clusters for the reduction of 4-nitrophenol. Chem. Commun. 2013, 49, 276–278. [Google Scholar] [CrossRef]
- Yao, C.; Oh, J.H.; Oh, I.G.; Park, C.H.; Chung, J.H. [6]-Shogaol inhibits melanogenesis in B16 mouse melanoma cells through activation of the ERK pathway. Acta Pharmacol. Sin. 2013, 34, 289–294. [Google Scholar] [CrossRef]
- Gonçalez, M.L.; Correa, M.A.; Chorilli, M. Skin delivery of kojic acid-loaded nanotechnology-based drug delivery systems for the treatment of skin aging. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Kumar, D.; Ghosh, R.; Pal, B.C. Glucosidase inhibitory terpenoids from Potentilla fulgens and their quantitative estimation by validated HPLC method. J. Funct. Foods 2013, 5, 1135–1141. [Google Scholar] [CrossRef]
- Son, K.H.; Heo, M.Y. The evaluation of depigmenting efficacy in the skin for the development of new whitening agents in Korea. Int. J. Cosmet. Sci. 2013, 35, 9–18. [Google Scholar] [CrossRef]
- Eimpunth, S.; Wanitphadeedecha, R.; Manuskiatti, W. A focused review on acne-induced and aesthetic procedure-related post-inflammatory hyperpigmentation in Asians. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 7–18. [Google Scholar] [CrossRef]
- Hsieh, P.W.; Chen, W.Y.; Aljuffali, I.A.; Chen, C.C.; Fang, J.Y. Co-drug strategy for promoting skin targeting and minimizing the transdermal diffusion of hydroquinone and tranexamic acid. Curr. Med. Chem. 2013, 20, 4080–4092. [Google Scholar] [CrossRef]
- Tse, T.W.; Hui, E. Tranexamic acid: An important adjuvant in the treatment of melasma. J. Cosmet. Dermatol. 2013, 12, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Carmen, P.; Laurian, V.; Mircea, T. Natural Resources Containing Arbutin. Determination of Arbutin in the Leaves of Bergenia crassifolia (L.) Fritsch. acclimated in Romania. Not. Bot. Horti Agrobot. Cluj Napoca 2009, 37, 129–132. [Google Scholar]
- Amer, M.; Metwalli, M. Topical hydroquinone in the treatment of some hyperpigmentary disorders. Int. J. Dermatol. 1998, 37, 449–450. [Google Scholar] [CrossRef]
- Haddad, A.L.; Matos, L.F.; Brunstein, F.; Ferreira, L.M.; Silva, A.; Costa, D., Jr. A clinical, prospective, randomized, double-blind trial comparing skin whitening complex with hydroquinone vs. placebo in the treatment of melasma. Int. J. Dermatol. 2003, 42, 153–156. [Google Scholar] [CrossRef]
- Nerya, O.; Vaya, J.; Musa, R.; Izrael, S.; Arie, B.R.; Tamir, S. Glabrene and Isoliquiritigenin as Tyrosinase Inhibitors from Licorice Roots. J. Agric. Food Chem. 2003, 51, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Ookubo, N.; Michiue, H.; Kitamatsu, M.; Kamamura, M.; Nishiki, T.; Ohmori, I.; Matsui, H. The transdermal inhibition of melanogenesis by a cell-membrane-permeable peptide delivery system based on poly-arginine. Biomaterials 2014, 35, 4508–4516. [Google Scholar] [CrossRef]
- Fujimoto, N.; Watanabe, H.; Nakatani, T.; Roy, G.; Ito, A. Induction of thyroid tumours in (C57BL/6N × C3H/N)F1 mice by oral administration of kojic acid. Food Chem. Toxicol. 1998, 36, 697–703. [Google Scholar] [CrossRef]
- Takizawa, T.; Mitsumori, K.; Tamura, T.; Nasu, M.; Ueda, M.; Imai, T.; Hirose, M. Hepatocellular tumor induction in heterozygous p53-deficient CBA mice by a 26-week dietary administration of kojic acid. Toxicol. Sci. 2003, 73, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Teeranachaideekul, V.; Nithitanakool, S.; Junhunkit, T.; Ponpanich, L.; Nopporn, N.; Detamornrat, U.; Chulasiri, M. Liposomes: A novel carrier system for Artocarpus lakoocha extract to improve skin whitening. J. Asian Assoc. Sch. Pharm. 2013, 2, 243–253. [Google Scholar]
- Salguero, C.P. The tradition of Thai medicine. In A Thai Herbal: Traditional Recipes for Health and Harmony; Barton, L., Ed.; Findhorn Press Scotland: Forres, UK, 2003; pp. 1–19. [Google Scholar]
- Maneechai, S.; Likhitwitayawuid, K.; Sritularak, B.; Palanuvej, C.; Ruangrungsi, N.; Sirisa-Ard, P. Quantitative analysis of oxyresveratrol content in Artocarpus lakoocha and Puag-Haad. Med. Princ. Pract. Int. J. Kuwait Univ. Health Sci. Cent. 2009, 18, 223–227. [Google Scholar] [CrossRef]
- Sritularak, B.; De-Eknamkul, W.; Likhitwitayawuid, K. Tyrosinase inhibitors from Artocarpus lackoocha. Thai J. Pharm. Sci. 1998, 22, 149–155. [Google Scholar]
- Singhatong, S.; Leelarungrayub, D.; Chaiyasut, C. Antioxidant and toxicity activites of Artocarpus lakoocha Roxb. Heartwood extract. J. Med. Plants Res. 2010, 4, 947–953. [Google Scholar]
- Tengamnuay, P.; Pengrungruangwong, K.; Pheansri, I.; Likhitwitayawuid, K. Artocarpus lakoocha heartwood extract as a novel cosmetic ingredient: Evaluation of the in vitro anti-tyrosinase and in vivo skin whitening activities. Int. J. Cosmet. Sci. 2006, 28, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Likhitwitayawuid, K.; Sornsute, A.; Sritularak, B.; Ploypradith, P. Chemical transformations of oxyresveratrol (trans-2,4,3′,5′-tetrahydroxystilbene) into a potent tyrosinase inhibitor and a strong cytotoxic agent. Bioorg. Med. Chem. Lett. 2006, 16, 5650–5653. [Google Scholar] [CrossRef] [PubMed]
- Hiroaki, H.; Hiroshi, S. Economic importance of licorice. Plant. Biotechnol. 2009, 26, 101–104. [Google Scholar]
- Russo, M.; Serra, D.; Suraci, F.; Di Sanzo, R.; Fuda, S.; Postorino, S. The potential of e-nose aroma profiling for identifying the geographical origin of licorice (Glycyrrhiza glabra L.) roots. Food Chem. 2014, 165, 467–474. [Google Scholar] [CrossRef]
- Marjan, N.H.; Hossein, H. Review of pharmacological effects of Glycyrrhiza sp. And its bioactive compounds. Phytother. Res. 2008, 22, 709–724. [Google Scholar]
- Kawaguchi, Y.; Gou, K.; Kawa, Y.; Kashima, M.; Mizoguchi, M. The inhibitory effects of licorice extracts on melanogenesis: In vivo studies. Jpn. J. Dermatol. 1992, 102, 689–694. [Google Scholar]
- Kameyama, K.; Sakai, C.; Tagawa, M. Effect of oil-soluble licorice extract on melanogenesis. Pigment Cell Res. 1994, 7, 372. [Google Scholar]
- Fu, B.; Li, H.; Wang, X.; Lee, F.S.; Cui, S. Isolation and identification of flavonoids in licorice and a study of their inhibitory effects on tyrosinase. J. Agric. Food Chem. 2005, 53, 7408–7414. [Google Scholar] [CrossRef]
- Yokota, T.; Nishio, H.; Kubota, Y.; Mizoguchi, M. The inhibitory effect of glabridin from licorice extracts on melanogenesis and inflammation. Pigment Cell Res. 1998, 11, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Povichit, N.; Phrutivorapongkul, A.; Suttajit, M.; Leelapornpisid, P. Antiglycation and antioxidant activities of oxyresveratrol extracted from the heartwood of Artocarpus lakoocha Roxb. Maejo Int. J. Sci. Technol. 2010, 4, 454–461. [Google Scholar]
- Borah, H.J.; Singhal, R.; Hazarika, S. Artocarpus lakoocha roxb: An untapped bioresource of resveratrol from North East India, its extractive separation and antioxidant activity. Ind. Crops Prod. 2017, 950, 75–82. [Google Scholar] [CrossRef]
- Mohammed, S.K. Antioxidative activities of aqueous and ethanolic extracts of licorice roots. Pak. J. Nutr. 2014, 13, 267–270. [Google Scholar] [CrossRef]
- Tian, M.; Yan, H.; Row, K.H. Extraction of Glycyrrhizic acid and Glabridin from licorice. Int. J. Mol. Sci. 2008, 9, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Espin, J.C.; García-Ruiz, P.A.; Tudela, J.; García-Cánovas, F. Study of stereospecificity in mushroom tyrosinase. Biochem. J. 1998, 331, 547–551. [Google Scholar] [CrossRef]
- Bae-Harboe, Y.S.; Park, H.Y. Tyrosinase: A central regulatory protein for cutaneous pigmentation. J. Investig. Dermatol. 2012, 132, 2678–2680. [Google Scholar] [CrossRef]
- Videira, I.F.; Moura, D.F.; Magina, S. Mechanisms regulating melanogenesis. An. Bras. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Wu, C.; Yaar, M.; Stachur, C.M.; Kosmadaki, M.; Gilchrest, B.A. Role of BMP-4 and Its Signaling Pathways in Cultured Human Melanocytes. Int. J. Cell Biol. 2009, 2009. [Google Scholar] [CrossRef]
- Funayama, M.; Arakawa, H.; Yamamoto, R.; Nishino, T.; Shin, T.; Murao, S. Effects of alpha- and beta-arbutin on activity of tyrosinases from mushroom and mouse melanoma. Biosci. Biotechnol. Biochem. 1995, 59, 143–144. [Google Scholar] [CrossRef]
- Teanpaisan, R.; Ruangkiatkul, P.; Thammasitboon, K.; Puripattanavong, J.; Faroongsarng, D. Effectiveness of Artocarpus lakoocha extract, poloxamer 407, on Enterococcus faecalis in vitro. J. Investig. Clin. Dent. 2013, 4, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Suhartati, T.; Yandri, H.; Hadi, S. The bioactivity test of artonin E from the bark of Artocarpus rigida Blume. Eur. J. Sci. Res. 2008, 23, 330–337. [Google Scholar]
- Heinrich, M.; Heneka, B.; Ankli, A.; Rimpler, H.; Sticher, O.; Kostiza, T. Spasmolytic and antidiarrhoeal properties of the Yucatec Mayan medicinal plant Casimiroa tetrameria. J. Pharm. Pharmacol. 2005, 57, 1081–1085. [Google Scholar] [CrossRef]
- Verpoorte, R.; Choi, Y.H.; Kim, H.K. Ethnopharmacology and systems biology: A perfect holistic match. J. Ethnopharmacol. 2005, 100, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Diwakar, G.; Rana, J.; Scholten, J.D. Inhibition of melanin production by a combination of Siberian larch and pomegranate fruit extracts. Fitoterapia 2012, 83, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Diwakar, G.; Rana, J.; Saito, L.; Vredeveld, D.; Zemaitis, D.; Scholten, J. Inhibitory effect of a novel combination of Salvia hispanica (chia) seed and Punica granatum (pomegranate) fruit extracts on melanin production. Fitoterapia 2014, 97, 164–171. [Google Scholar] [CrossRef]
- Han, S.M.; Yeo, J.H.; Cho, Y.H.; Pak, S.C. Royal jelly reduces melanin synthesis through down-regulation of tyrosinase expression. Am. J. Chin. Med. 2011, 39, 1253–1260. [Google Scholar] [CrossRef]
- Gilchrest, B.A.; Eller, M.S. DNA photodamage stimulates melanogenesis and other photoprotective responses. J. Investig. Dermatol. Symp. Proc. 1999, 4, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Couteau, C.; Coiffard, L. Overview of skin whitening agents: Drugs and cosmetic products. Cosmetics 2016, 3, 27. [Google Scholar] [CrossRef]
- Katsambas, A.D.; Stratigos, A.J. Depigmenting and bleaching agents: Coping with hyperpigmentation. Clin. Dermatol. 2001, 19, 483–488. [Google Scholar] [CrossRef]
- Arung, E.T.; Shimizu, K.; Kondo, R. Artocarpus plants as a potential source of skin whitening agents. Nat. Prod. Commun. 2011, 6, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Javanmardi, J.; Stushnoff, C.; Locke, E.; Vivanco, J.M. Antioxidant activity and total phenolic content of Iranian Ocimum accessions. Food Chem. 2003, 83, 547–550. [Google Scholar] [CrossRef]
- Joubert, E.; Manley, M.; Botha, M. Evaluation of spectrophotometric methods for screening of green rooibos (Aspalathus linearis) and green honeybush (Cyclopia genistoides) extracts for high levels of bio-active compounds. Phytochem. Anal. 2008, 19, 169–178. [Google Scholar] [CrossRef]
- Itsarasook, K.; Ingkaninan, K.; Viyoch, J. Artocarpin-enriched extract reverses collagen metabolism in UV-exposed fibroblasts. Biologia 2014, 69, 943–951. [Google Scholar] [CrossRef]
- Ko, H.H.; Tsai, Y.T.; Yen, M.H.; Lin, C.C.; Liang, C.J.; Yang, T.H.; Lee, C.W.; Yen, F.L. Norartocarpetin from a folk medicine Artocarpus communis plays a melanogenesis inhibitor without cytotoxicity in B16F10 cell and skin irritation in mice. BMC Complement. Altern. Med. 2013, 13, 348. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Katsambas, A.D. Optimal management of recalcitrant disorders of hyperpigmentation in dark-skinned patients. Am. J. Clin. Dermatol. 2004, 5, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Bilodeau, M.L.; Greulich, J.D.; Hullinger, R.L.; Bertolotto, C.; Ballotti, R.; Andrisani, O.M. BMP-2 stimulates tyrosinase gene expression and melanogenesis in differentiated melanocytes. Pigment Cell Res. 2001, 14, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Rodboon, T.; Puchadapirom, P.; Okada, S.; Suwannalert, P. Oxyresveratrol from Artocarpus lakoocha Roxb. Inhibit Melanogenesis in B16 Melanoma Cells through the Role of Cellular Oxidants. Walailak J. Sci. Technol. 2015, 13, 261–270. [Google Scholar]
- Lin, V.C.; Ding, H.Y.; Kuo, S.Y.; Chin, L.W.; Wu, J.Y.; Chang, T.S. Evaluation of in vitro and in vivo depigmenting activity of raspberry ketone from rheum officinale. Int. J. Mol. 2011, 12, 4819–4835. [Google Scholar] [CrossRef]
- Zaidi, K.U.; Ali, S.A.; Ali, A.S. Purified Mushroom Tyrosinase Induced Melanogenic Protein Expression in B16F10 Melanocytes: A Quantitative Densitometric Analysis. Open Med. Chem. J. 2018, 12, 36–47. [Google Scholar] [CrossRef][Green Version]

| Extracts | Tyrosinase Inhibitory Activity IC50 (Mean ± SD) (mg/mL) | Radical Scavenging Activity IC50 (Mean ± SD) (mg/mL) |
|---|---|---|
| Al70 | 0.028 ± 0.0029 | 0.081 ± 0.027 |
| Al95 | 0.017 ± 0.0024 1 | 0.074 ± 0.024 |
| Gg70 | 0.114 ± 0.0057 | 0.365 ± 0.171 |
| Gg95 | 0.074 ± 0.0011 | 0.223 ± 0.072 |
| Kojic acid | 0.495 ± 0.029 | - |
| Ascorbic acid | - | 0.007 ± 0.0008 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panichakul, T.; Rodboon, T.; Suwannalert, P.; Tripetch, C.; Rungruang, R.; Boohuad, N.; Youdee, P. Additive Effect of a Combination of Artocarpus lakoocha and Glycyrrhiza glabra Extracts on Tyrosinase Inhibition in Melanoma B16 Cells. Pharmaceuticals 2020, 13, 310. https://doi.org/10.3390/ph13100310
Panichakul T, Rodboon T, Suwannalert P, Tripetch C, Rungruang R, Boohuad N, Youdee P. Additive Effect of a Combination of Artocarpus lakoocha and Glycyrrhiza glabra Extracts on Tyrosinase Inhibition in Melanoma B16 Cells. Pharmaceuticals. 2020; 13(10):310. https://doi.org/10.3390/ph13100310
Chicago/Turabian StylePanichakul, Tasanee, Teerapat Rodboon, Prasit Suwannalert, Chanchai Tripetch, Rittipun Rungruang, Nattaporn Boohuad, and Piyawan Youdee. 2020. "Additive Effect of a Combination of Artocarpus lakoocha and Glycyrrhiza glabra Extracts on Tyrosinase Inhibition in Melanoma B16 Cells" Pharmaceuticals 13, no. 10: 310. https://doi.org/10.3390/ph13100310
APA StylePanichakul, T., Rodboon, T., Suwannalert, P., Tripetch, C., Rungruang, R., Boohuad, N., & Youdee, P. (2020). Additive Effect of a Combination of Artocarpus lakoocha and Glycyrrhiza glabra Extracts on Tyrosinase Inhibition in Melanoma B16 Cells. Pharmaceuticals, 13(10), 310. https://doi.org/10.3390/ph13100310




