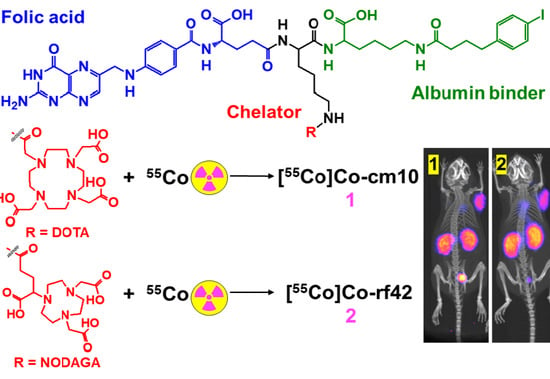
New 55Co-labeled Albumin-Binding Folate Derivatives as Potential PET Agents for Folate Receptor Imaging
Abstract
:1. Introduction
2. Results and Discussion
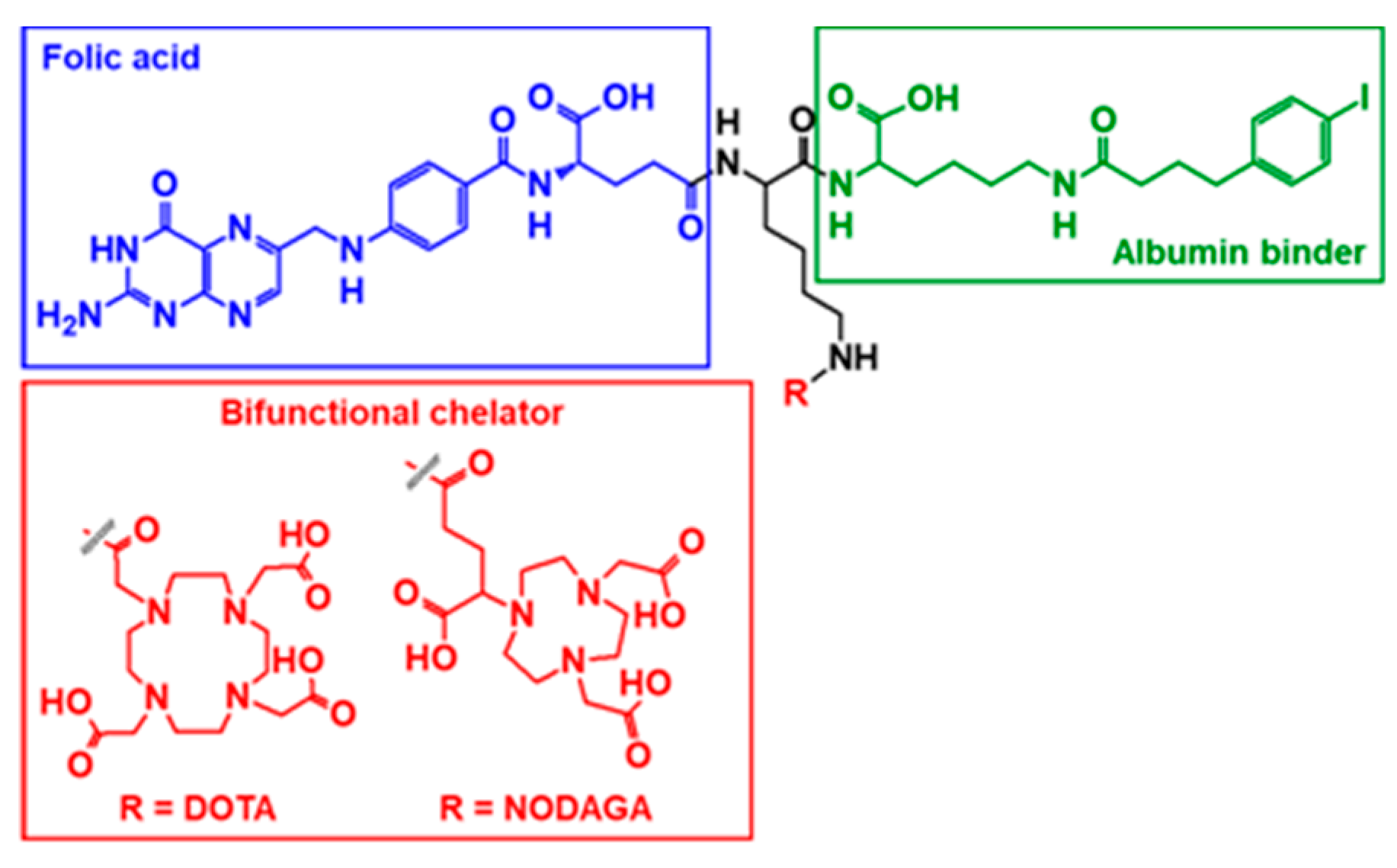
2.1. Radiolabeling
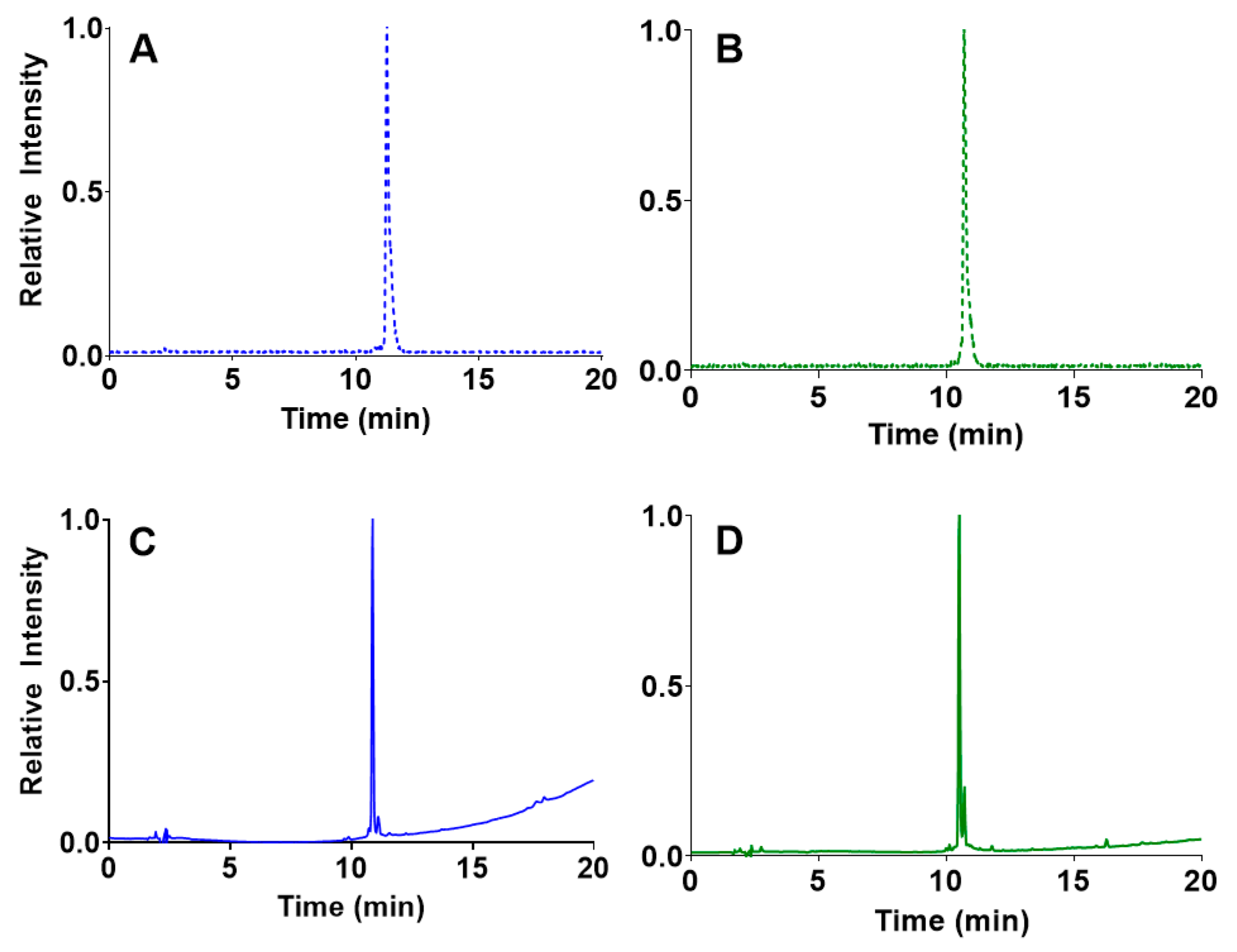
2.2. In Vitro Stability Determination
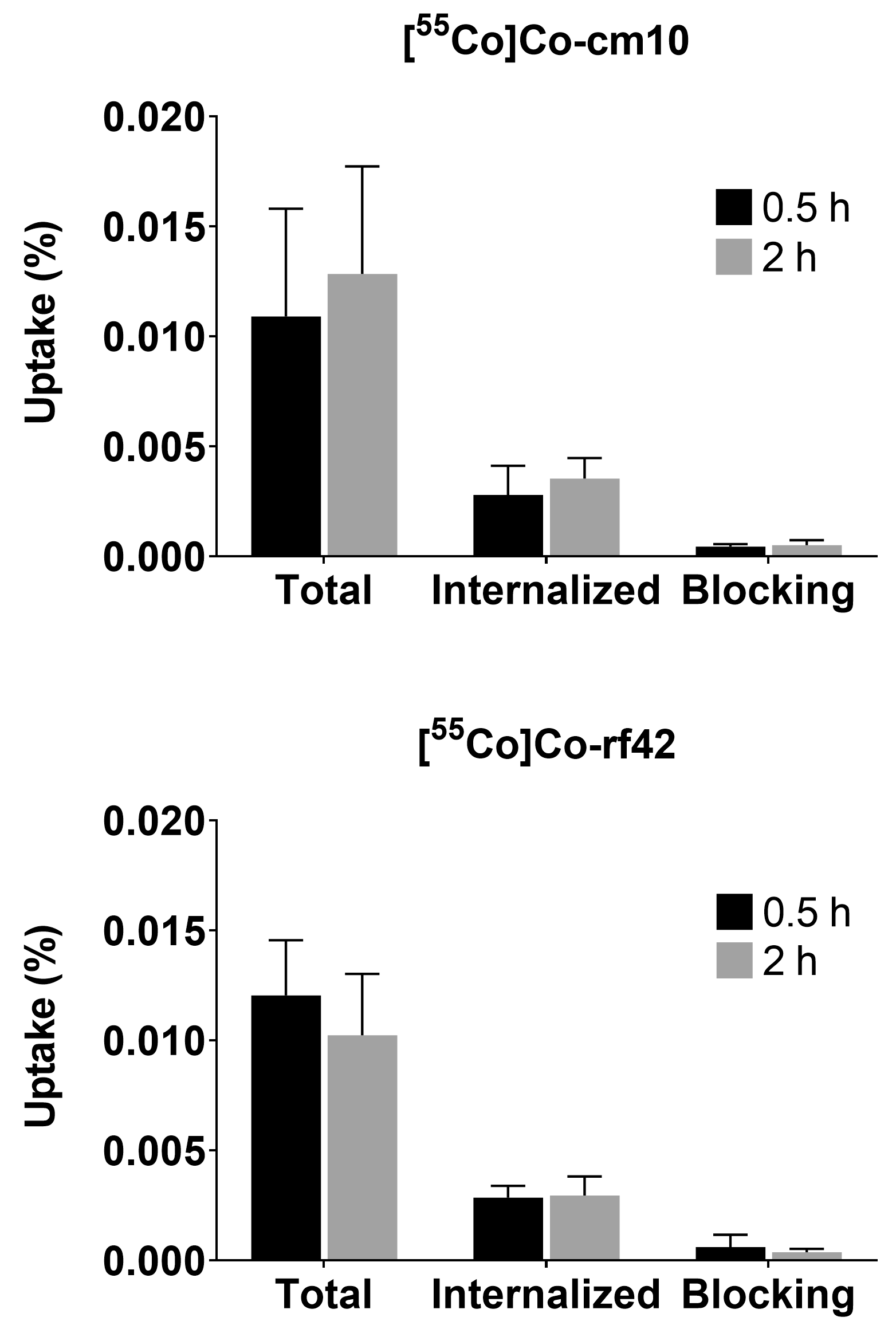
2.3. Cell Binding and Internalization
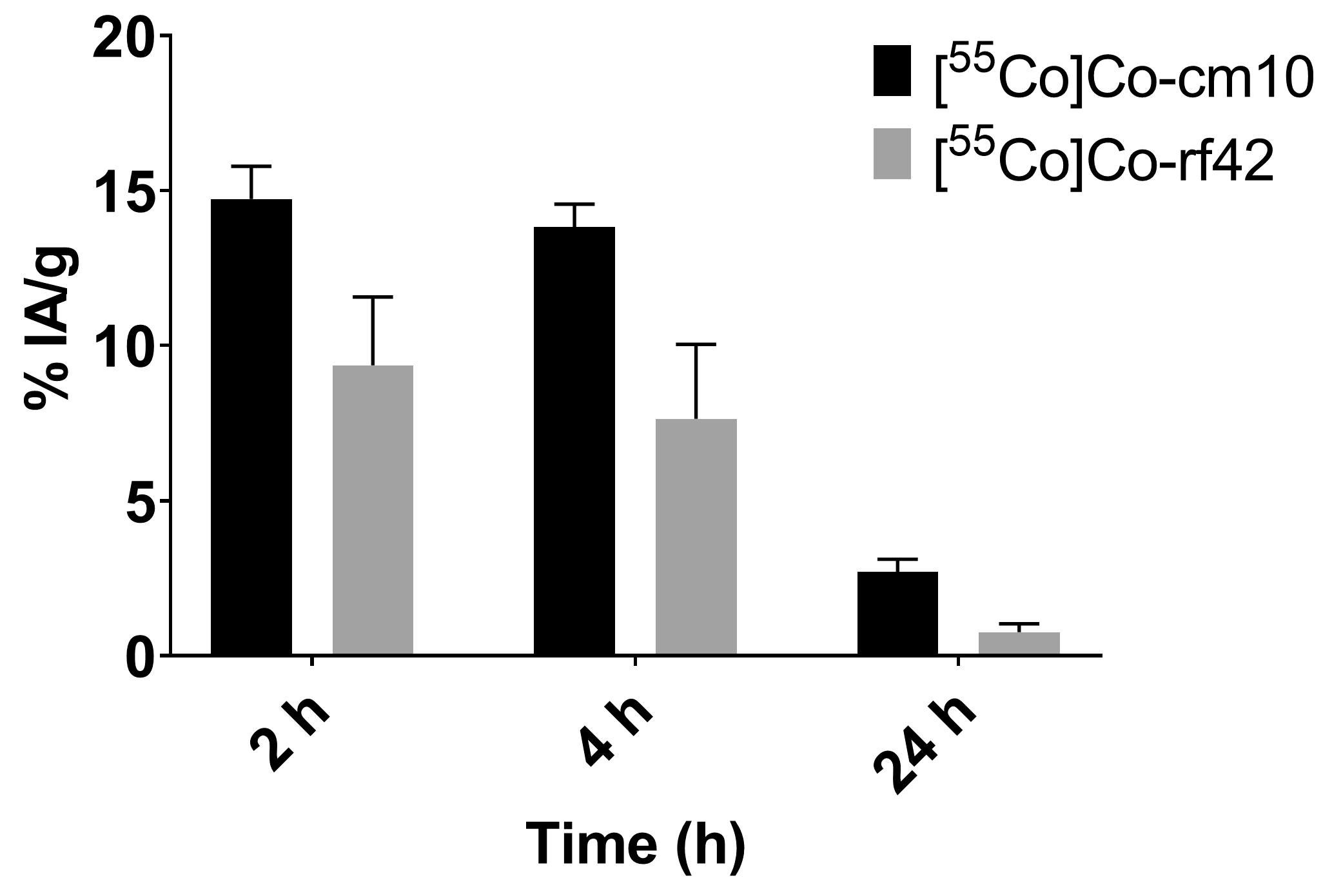
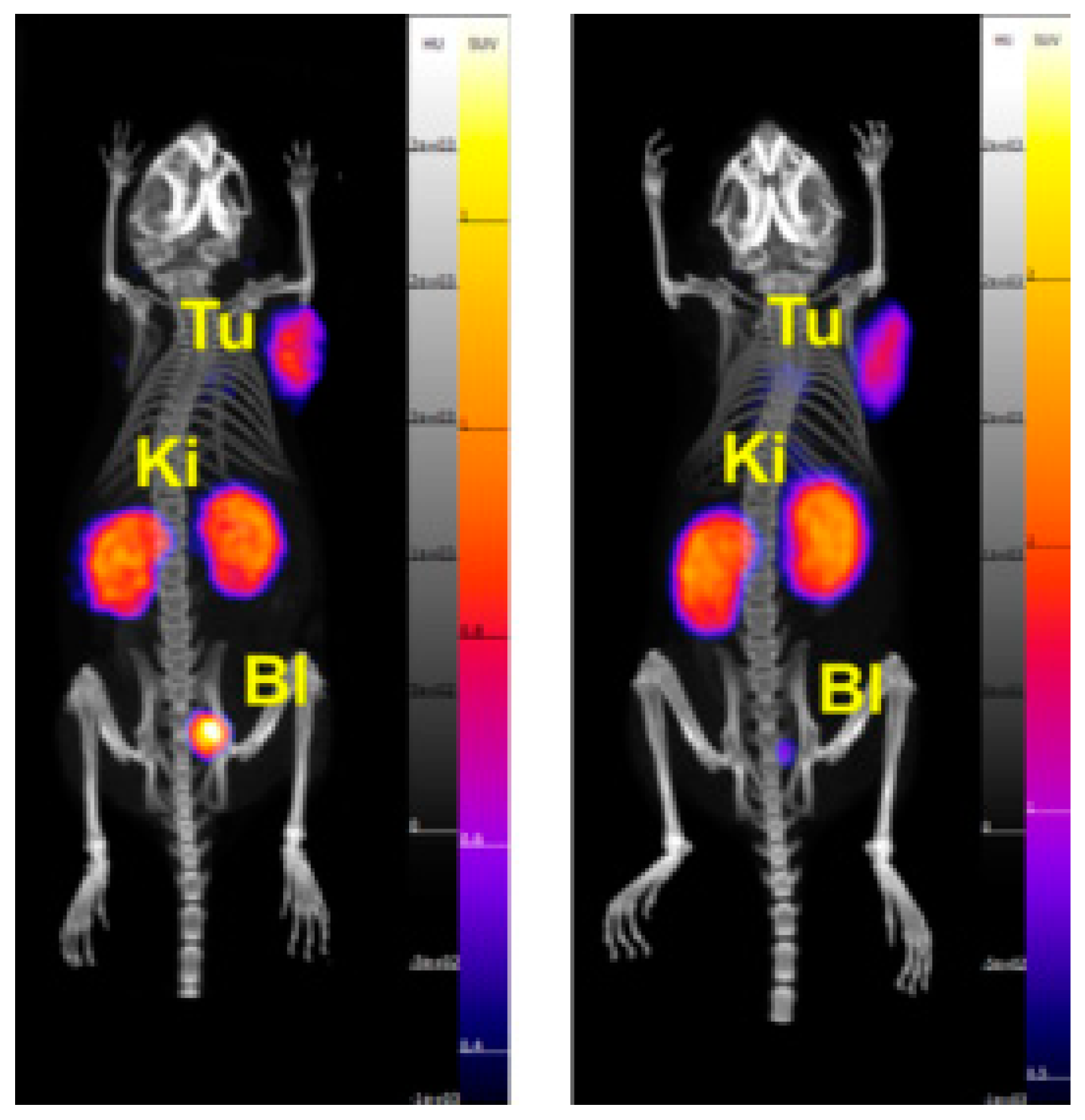
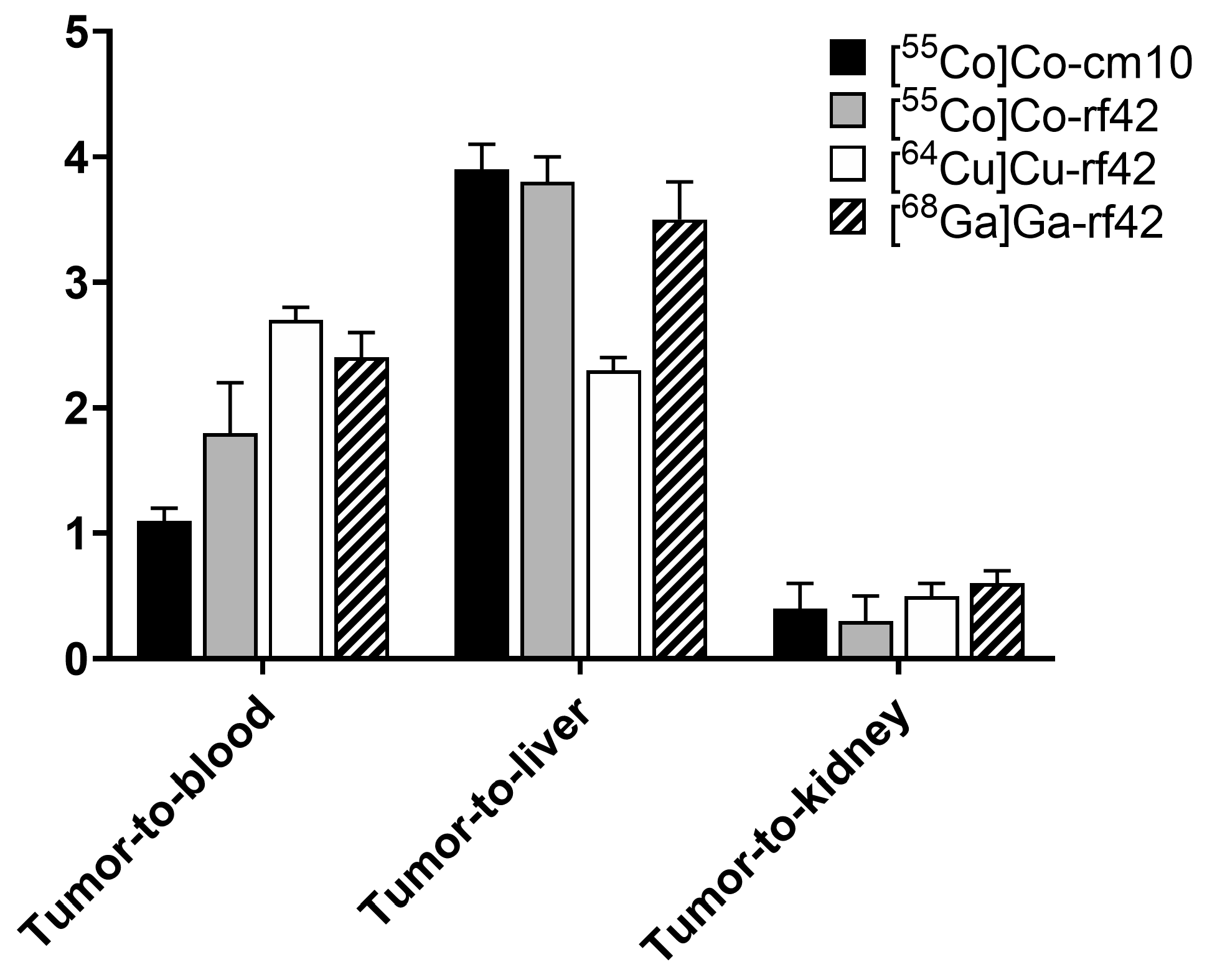
2.4. In Vivo Biodistribution and PET Imaging
3. Materials and Methods
3.1. General Methods and Instrumentation
3.2. Cobalt-55 Production and Purification
3.3. Preparation of 55Co-labeled Radiofolates
3.4. In Vitro Stability Determination
3.5. Cell Culture
3.6. Cell Binding and Internalization
3.7. In Vivo Biodistribution and PET Imaging
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Low, P.S.; Kularatne, S.A. Folate-targeted therapeutic and imaging agents for cancer. Curr. Opin. Chem. Biol. 2009, 13, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.; Bax, H.J.; Josephs, D.H.; Ilieva, K.M.; Pellizzari, G.; Opzoomer, J.; Bloomfield, J.; Fittall, M.; Grigoriadis, A.; Figini, M.; et al. Targeting folate receptor alpha for cancer treatment. Oncotarget 2016, 7, 52553–52574. [Google Scholar] [CrossRef] [PubMed]
- Sudimack, J.; Lee, R.J. Targeted drug delivery via the folate receptor. Adv. Drug Deliv. Rev. 2000, 41, 147–162. [Google Scholar] [CrossRef]
- Siegel, B.A.; Dehdashti, F.; Mutch, D.G.; Podoloff, D.A.; Wendt, R.; Sutton, G.P.; Burt, R.W.; Ellis, P.R.; Mathias, C.J.; Green, M.A.; et al. Evaluation of 111In-DTPA-folate as a receptor-targeted diagnostic agent for ovarian cancer: Initial clinical results. J. Nucl. Med. 2003, 44, 700–707. [Google Scholar]
- Fisher, R.E.; Siegel, B.A.; Edell, S.L.; Oyesiku, N.M.; Morgenstern, D.E.; Messmann, R.A.; Amato, R.J. Exploratory Study of 99mTc-EC20 imaging for identifying patients with folate receptor-positive solid tumors. J. Nucl. Med. 2008, 49, 899–906. [Google Scholar] [CrossRef]
- [18F]-AZAFOL AS POSITRON EMISSION TOMOGRAPHY (PET) TRACER in FR Positive Cancer Imaging. Available online: https://clinicaltrials.gov/ct2/show/NCT03242993 (accessed on 19 June 2019).
- Parker, N.; Turk, M.J.; Westrick, E.; Lewis, J.D.; Low, P.S.; Leamon, C.P. Folate receptor expression in carcinomas and normal tissues determined by a quantitative radioligand binding assay. Anal. Biochem. 2005, 338, 284–293. [Google Scholar] [CrossRef]
- Birn, H.; Selhub, J.; Christensen, E.I. Internalization and intracellular transport of folate-binding protein in rat kidney proximal tubule. Am. J. Physiol. Cell Physiol. 1993, 264, C302–C310. [Google Scholar] [CrossRef]
- Birn, H.; Nielsen, S.; Christensen, E.I. Internalization and apical-to-basolateral transport of folate in rat kidney proximal tubule. Am. J. Physiol.-Ren. Physiol. 1997, 272, F70–F78. [Google Scholar] [CrossRef]
- Müller, C.; Schibli, R.; Krenning, E.P.; de Jong, M. Pemetrexed improves tumor selectivity of 111In-DTPA-folate in mice with folate receptor-positive ovarian cancer. J. Nucl. Med. 2008, 49, 623–629. [Google Scholar] [CrossRef]
- Müller, C.; Mindt, T.L.; de Jong, M.; Schibli, R. Evaluation of a novel radiofolate in tumour-bearing mice: Promising prospects for folate-based radionuclide therapy. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 938–946. [Google Scholar] [CrossRef]
- Müller, C.; Struthers, H.; Winiger, C.; Zhernosekov, K.; Schibli, R. DOTA conjugate with an albumin-binding entity enables the first folic acid-targeted 177Lu-radionuclide tumor therapy in mice. J. Nucl. Med. 2013, 54, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Bunka, M.; Haller, S.; Köster, U.; Groehn, V.; Bernhardt, P.; van der Meulen, N.; Türler, A.; Schibli, R. Promising prospects for 44Sc-/47Sc-based theragnostics: Application of 47Sc for radionuclide tumor therapy in mice. J. Nucl. Med. 2014, 55, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Farkas, R.; Siwowska, K.; Ametamey, S.M.; Schibli, R.; van der Meulen, N.P.; Müller, C. 64Cu- and 68Ga-Based PET imaging of folate receptor-positive tumors: Development and evaluation of an albumin-binding NODAGA-folate. Mol. Pharm. 2016, 13, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Guzik, P.; Siwowska, K.; Cohrs, S.; Schmid, R.M.; Schibli, R. Combining albumin-binding properties and interaction with pemetrexed to improve the tissue distribution of radiofolates. Molecules (Basel Switz.) 2018, 23, 1465. [Google Scholar] [CrossRef] [PubMed]
- Dennis, M.S.; Zhang, M.; Meng, Y.G.; Kadkhodayan, M.; Kirchhofer, D.; Combs, D.; Damico, L.A. Albumin Binding as a general strategy for improving the pharmacokinetics of proteins. J. Biol. Chem. 2002, 277, 35035–35043. [Google Scholar] [CrossRef]
- Antunes, P.; Ginj, M.; Zhang, H.; Waser, B.; Baum, R.P.; Reubi, J.C.; Maecke, H. Are radiogallium-labelled DOTA-conjugated somatostatin analogues superior to those labelled with other radiometals? Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 982–993. [Google Scholar] [CrossRef]
- Lane, S.R.; Nanda, P.; Rold, T.L.; Sieckman, G.L.; Figueroa, S.D.; Hoffman, T.J.; Jurisson, S.S.; Smith, C.J. Optimization, biological evaluation and microPET imaging of copper-64-labeled bombesin agonists, [64Cu-NO2A-(X)-BBN(7–14)NH2], in a prostate tumor xenografted mouse model. Nucl. Med. Biol. 2010, 37, 751–761. [Google Scholar] [CrossRef]
- Wild, D.; Fani, M.; Fischer, R.; Pozzo, L.D.; Kaul, F.; Krebs, S.; Rivier, J.E.F.; Reubi, J.C.; Maecke, H.R.; Weber, W.A. Comparison of somatostatin receptor agonist and antagonist for peptide receptor radionuclide therapy: A pilot study. J. Nucl. Med. 2014, 55, 1248–1252. [Google Scholar] [CrossRef]
- Heppeler, A.; André, J.P.; Buschmann, I.; Wang, X.; Reubi, J.-C.; Hennig, M.; Kaden, T.A.; Maecke, H.R. Metal-ion-dependent biological properties of a chelator-derived somatostatin analogue for tumour Targeting. Chem. Eur. J. 2008, 14, 3026–3034. [Google Scholar] [CrossRef]
- Zhou, Y.; Baidoo, K.E.; Brechbiel, M.W. Mapping Biological Behaviors by Application of Longer-Lived Positron Emitting Radionuclides. Adv. Drug Deliv. Rev. 2013, 65, 1098–1111. [Google Scholar] [CrossRef]
- Andersen, T.L.; Baun, C.; Olsen, B.B.; Dam, J.H.; Thisgaard, H. Improving contrast and detectability—Imaging with [55Co]Co-DOTATATE in comparison with [64Cu]Cu-DOTATATE and [68Ga]Ga-DOTATATE. J. Nucl. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Umbricht, C.A.; Benešová, M.; Hasler, R.; Schibli, R.; van der Meulen, N.P.; Müller, C. Design and preclinical evaluation of an albumin-binding PSMA ligand for 64Cu-based PET imaging. Mol. Pharm. 2018, 15, 5556–5564. [Google Scholar] [CrossRef] [PubMed]
- Fani, M.; Pozzo, L.D.; Abiraj, K.; Mansi, R.; Tamma, M.L.; Cescato, R.; Waser, B.; Weber, W.A.; Reubi, J.C.; Maecke, H.R. PET of somatostatin receptor-positive tumors using 64Cu- and 68Ga-somatostatin antagonists: The chelate makes the difference. J. Nucl. Med. 2011, 52, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.-Y.; Meng, L.-H.; Ding, J.; Xie, Y.-Y.; Huang, X.-Y. New tetraazamacrocyclic ligand with neutral pendent groups 1,4,7,10-tetrakis(2-cyanoethyl)-1,4,7,10-tetraazacyclododecane (L) and its cobalt(II), nickel(II) and copper(II) complexes: Synthesis, structural characterization and antitumor activity. Polyhedron 2000, 19, 217–223. [Google Scholar] [CrossRef]
- Anderegg, G.; Arnaud-Neu, F.; Delgado, R.; Felcman, J.; Popov, K. Critical evaluation of stability constants of metal complexes of complexones for biomedical and environmental applications (IUPAC Technical Report). Pure Appl. Chem. 2005, 77, 1445–1495. [Google Scholar] [CrossRef]
- Low, P.S.; Henne, W.A.; Doorneweerd, D.D. Discovery and development of folic-acid-based receptor targeting for imaging and therapy of cancer and inflammatory diseases. Acc. Chem. Res. 2008, 41, 120–129. [Google Scholar] [CrossRef]
- Maurer, A.H.; Elsinga, P.; Fanti, S.; Nguyen, B.; Oyen, W.J.G.; Weber, W.A. Imaging the folate receptor on cancer cells with 99mTc-Etarfolatide: Properties, clinical use, and future potential of folate receptor imaging. J. Nucl. Med. 2014, 55, 701–704. [Google Scholar] [CrossRef]
- Manrique-Arias, J.C.; Avila-Rodriguez, M.A. A simple and efficient method of nickel electrodeposition for the cyclotron production of 64Cu. Appl. Radiat. Isot. 2014, 89, 37–41. [Google Scholar] [CrossRef]
- McCarthy, D.W.; Shefer, R.E.; Klinkowstein, R.E.; Bass, L.A.; Margeneau, W.H.; Cutler, C.S.; Anderson, C.J.; Welch, M.J. Efficient production of high specific activity 64Cu using a biomedical cyclotron. Nucl. Med. Biol. 1997, 24, 35–43. [Google Scholar] [CrossRef]
- Mastren, T.; Marquez, B.V.; Sultan, D.E.; Bollinger, E.; Eisenbeis, P.; Voller, T.; Lapi, S.E. Cyclotron production of high specific activity 55Co and in vivo evaluation of the stability of 55Co metal-chelate-peptide complexes. Mol. Imaging 2015, 14, 526–533. [Google Scholar] [CrossRef]
- Ladino, C.A.; Chari, R.V.J.; Bourret, L.A.; Kedersha, N.L.; Goldmacher, V.S. Folate-maytansinoids: Target-selective drugs of low molecular weight. Int. J. Cancer 1997, 73, 859–864. [Google Scholar] [CrossRef]
| 1 h | 4 h | 24 h | |
|---|---|---|---|
| PBS | 97 ± 2 | 95 ± 1 | 93.4 ± 0.3 |
| Mouse Serum | 98 ± 1 | 97.8 ± 0.6 | 95 ± 1 |
| 1 h | 4 h | 24 h | |
|---|---|---|---|
| PBS | 98 ± 2 | 95.6 ± 0.9 | 93 ± 3 |
| Mouse Serum | 97.5 ± 0.5 | 97.2 ± 0.4 | 94.6 ± 0.6 |
| Organ | 4 h | 4 h Blocking b | 24 h |
|---|---|---|---|
| Blood | 13.8 ± 0.8 | 17 ± 3 | 2.6 ± 0.3 |
| Heart | 4.7 ± 0.6 | 4.2 ± 0.7 | 1.7 ± 0.2 |
| Lungs | 5.4 ± 0.6 | 7 ± 3 | 1.9 ± 0.3 |
| Pancreas | 2.4 ± 0.7 | 2.4 ± 0.7 | 1.4 ± 0.2 |
| Spleen | 2.0 ± 0.4 | 2.2 ± 0.4 | 1.0 ± 0.1 |
| Stomach | 0.8 ± 0.2 | 1.5 ± 0.1 | 0.34 ± 0.09 |
| Liver | 3.7 ± 0.6 | 3.5 ± 0.5 | 1.9 ± 0.3 |
| Kidney | 36 ± 7 | 16 ± 4 | 30 ± 2 |
| Intestine | 1.9 ± 0.3 | 2.3 ± 0.4 | 0.92 ± 0.08 |
| Fat | 5 ± 1 | 4 ± 1 | 3 ± 2 |
| Skin | 6 ± 2 | 5 ± 1 | 7 ± 3 |
| Muscle | 2.4 ± 0.9 | 2.1 ± 0.7 | 1.3 ± 0.3 |
| Bone | 1.8 ± 0.6 | 2.2 ± 0.8 | 0.8 ± 0.2 |
| Brain | 0.8 ± 0.1 | 0.6 ± 0.2 | 0.45 ± 0.08 |
| Tumor | 17 ± 2 | 8 ± 4 | 13 ± 2 |
| Organ | 4 h | 4 h Blocking b | 24 h |
|---|---|---|---|
| Blood | 8 ± 3 | 15 ± 2 | 1.0 ± 0.4 |
| Heart | 2.9 ± 0.4 | 4 ± 1 | 2.0 ± 0.4 |
| Lungs | 4 ± 1 | 5.6 ± 0.3 | 1.5 ± 0.4 |
| Pancreas | 2.4 ± 0.5 | 2.2 ± 0.2 | 1.9 ± 0.3 |
| Spleen | 1.5 ± 0.3 | 2.1 ± 0.3 | 0.6 ± 0.2 |
| Stomach | 0.8 ± 0.4 | 1.1 ± 0.5 | 0.5 ± 0.2 |
| Liver | 3.8 ± 0.6 | 4 ± 1 | 2.9 ± 0.5 |
| Kidney | 53 ± 12 | 12 ± 4 | 46 ± 14 |
| Intestine | 2.2 ± 0.5 | 2.2 ± 0.3 | 0.8 ± 0.2 |
| Fat | 3.2 ± 0.9 | 2.7 ± 0.6 | 2.7 ± 0.2 |
| Skin | 8 ± 1 | 3.5 ± 0.8 | 6 ± 2 |
| Muscle | 1.6 ± 0.4 | 1.5 ± 0.3 | 1.2 ± 0.6 |
| Bone | 1.3 ± 0.2 | 1.7 ± 0.5 | 0.6 ± 0.2 |
| Brain | 0.7 ± 0.1 | 0.5 ± 0.1 | 0.4 ± 0.1 |
| Tumor | 17 ± 4 | 7 ± 2 | 15 ± 6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radford, L.L.; Fernandez, S.; Beacham, R.; El Sayed, R.; Farkas, R.; Benešová, M.; Müller, C.; Lapi, S.E. New 55Co-labeled Albumin-Binding Folate Derivatives as Potential PET Agents for Folate Receptor Imaging. Pharmaceuticals 2019, 12, 166. https://doi.org/10.3390/ph12040166
Radford LL, Fernandez S, Beacham R, El Sayed R, Farkas R, Benešová M, Müller C, Lapi SE. New 55Co-labeled Albumin-Binding Folate Derivatives as Potential PET Agents for Folate Receptor Imaging. Pharmaceuticals. 2019; 12(4):166. https://doi.org/10.3390/ph12040166
Chicago/Turabian StyleRadford, Lauren L., Solana Fernandez, Rebecca Beacham, Retta El Sayed, Renata Farkas, Martina Benešová, Cristina Müller, and Suzanne E. Lapi. 2019. "New 55Co-labeled Albumin-Binding Folate Derivatives as Potential PET Agents for Folate Receptor Imaging" Pharmaceuticals 12, no. 4: 166. https://doi.org/10.3390/ph12040166
APA StyleRadford, L. L., Fernandez, S., Beacham, R., El Sayed, R., Farkas, R., Benešová, M., Müller, C., & Lapi, S. E. (2019). New 55Co-labeled Albumin-Binding Folate Derivatives as Potential PET Agents for Folate Receptor Imaging. Pharmaceuticals, 12(4), 166. https://doi.org/10.3390/ph12040166