The Therapeutic Potential of Naringenin: A Review of Clinical Trials
Abstract
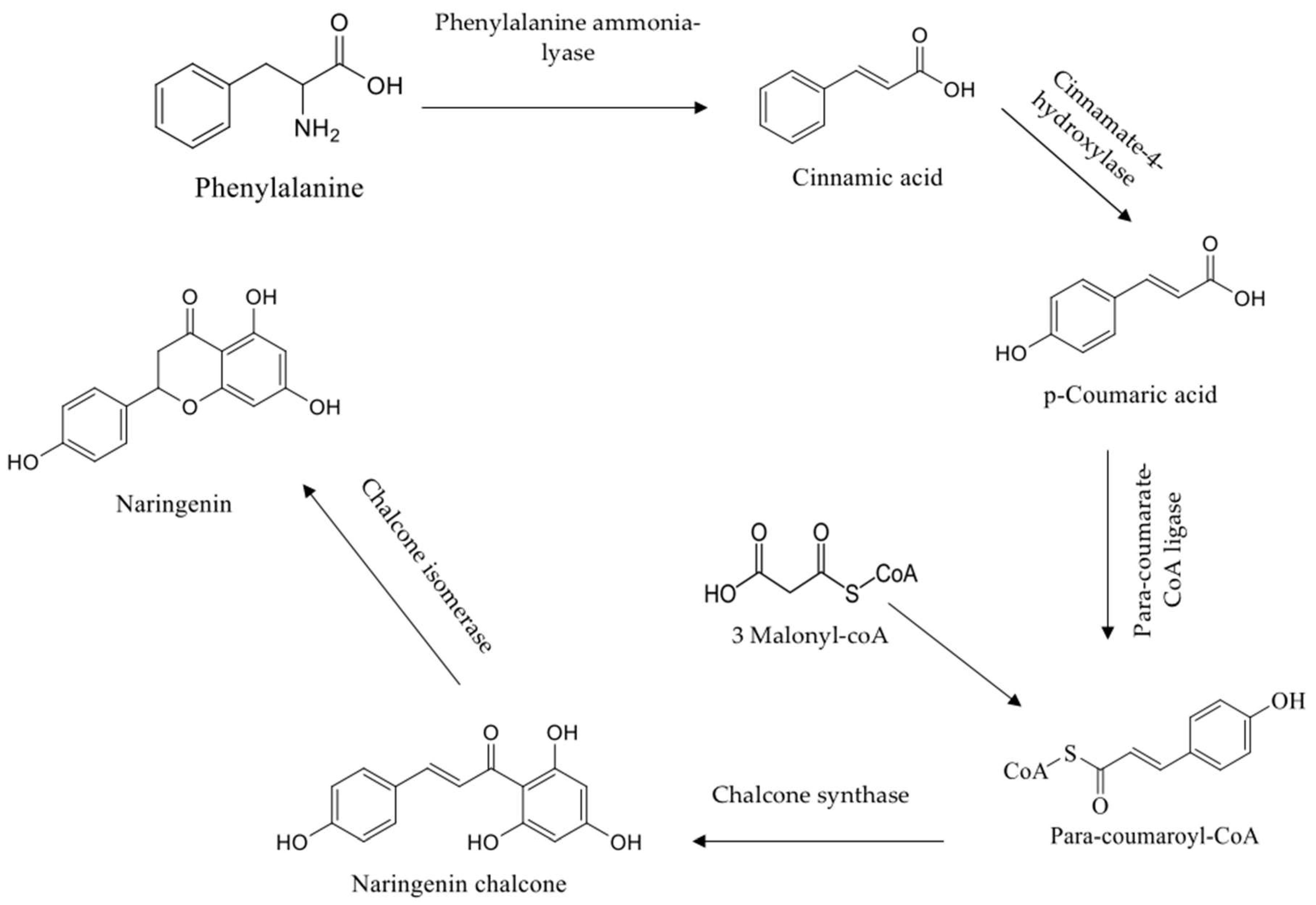
1. Introduction
2. Preclinical Pharmacological Activities of Naringenin
3. Bioavailability of Naringenin
4. Naringenin in Clinical Trials
4.1. Role of Naringenin in Cardiovascular Diseases
4.2. Role of Naringenin in Endothelial Function
4.3. Role of Naringenin in Weight Control
4.4. Role of Naringenin as Anti-HCV Activity
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mbaveng, A.T.; Zhao, Q.; Kuete, V. Chapter 20—Harmful and protective effects of phenolic compounds from African medicinal plants. In Toxicological Survey of African Medicinal Plants; Elsevier: New York, NY, USA, 2014; pp. 577–609. [Google Scholar]
- Jadeja, R.N.; Devkar, R.V. Polyphenols and flavonoids in controlling non-alcoholic steatohepatitis. In Polyphenols in Human Health and Disease; Academic Press: San Diego, CA, USA, 2014; pp. 615–623. [Google Scholar]
- Zobeiri, M.; Belwal, T.; Parvizi, F.; Naseri, R.; Farzaei, M.H.; Nabavi, S.F.; Sureda, A.; Nabavi, S.M. Naringenin and its nano-formulations for fatty liver: Cellular modes of action and clinical perspective. Curr. Pharm. Biotechnol. 2018, 19, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Soltana, H.; De Rosso, M.; Lazreg, H.; Vedova, A.D.; Hammami, M.; Flamini, R. LC-QTOF characterization of non-anthocyanic flavonoids in four Tunisian fig varieties. J. Mass Spectrom. JMS 2018, 53, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, L.J.; Borradaile, N.M.; Huff, M.W. Antiatherogenic properties of naringenin, a citrus flavonoid. Cardiovasc. Drug Rev. 1999, 17, 160–178. [Google Scholar] [CrossRef]
- De Souza Bido, G.; de Lourdes Lucio Ferrarese, M.; Marchiosi, R.; Ferrarese-Filho, O. Naringenin inhibits the growth and stimulates the lignification of soybean root. Braz. Arch. Biol. Technol. 2010, 53, 533–542. [Google Scholar] [CrossRef]
- Koopman, F.; Beekwilder, J.; Crimi, B.; van Houwelingen, A.; Hall, R.D.; Bosch, D.; van Maris, A.J.; Pronk, J.T.; Daran, J.M. De novo production of the flavonoid naringenin in engineered saccharomyces cerevisiae. Microb. Cell Fact. 2012, 11, 155. [Google Scholar] [CrossRef]
- Jeandet, P.; Sobarzo-Sánchez, E.; Clément, C.; Nabavi, S.; Habtemariam, S.; Nabavi, S.; Cordelier, S. Engineering stilbene metabolic pathways in microbial cells. Biotechnol. Adv. 2018, 36, 2264. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Alvarez, R.; Botas, A.; Albillos, S.M.; Rumbero, A.; Martin, J.F.; Liras, P. Molecular genetics of naringenin biosynthesis, a typical plant secondary metabolite produced by Streptomyces clavuligerus. Microb. Cell Fact. 2015, 14, 178. [Google Scholar] [CrossRef]
- Eichenberger, M.; Lehka, B.J.; Folly, C.; Fischer, D.; Martens, S.; Simon, E.; Naesby, M. Metabolic engineering of saccharomyces cerevisiae for de novo production of dihydrochalcones with known antioxidant, antidiabetic, and sweet tasting properties. Metab. Eng. 2017, 39, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhou, T.; Du, G.; Zhou, J.; Chen, J. Modular optimization of heterologous pathways for de novo synthesis of (2S)-naringenin in Escherichia coli. PLoS ONE 2014, 9, e101492. [Google Scholar] [CrossRef]
- Pandey, R.; Parajuli, P.; Koffas, M.; Sohng, J. Microbial production of natural and non-natural flavonoids: Pathway engineering, directed evolution and systems/synthetic biology. Biotechnol. Adv. 2016, 34, 634. [Google Scholar] [CrossRef]
- Trantas, E.A.; Koffas, M.A.; Xu, P.; Ververidis, F. When plants produce not enough or at all: Metabolic engineering of flavonoids in microbial hosts. Front. Plant Sci. 2015, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.M.; Shirooie, S.; Šamec, D.; Tomczyk, M.; Milella, L.; Russo, D.; Habtemariam, S.; Suntar, I.; Rastrelli, L.; Daglia, M.; et al. Flavonoid biosynthetic pathways in plants: Versatile targets for metabolic engineering. Biotechnol. Adv. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Liang, Y.; Wang, D.; Yan, Z.; Yin, H.; Wu, D.; Su, Q. Naringenin induces laxative effects by upregulating the expression levels of c-Kit and SCF, as well as those of aquaporin 3 in mice with loperamide-induced constipation. Int. J. Mol. Med. 2018, 41, 649–658. [Google Scholar] [CrossRef] [PubMed]
- Karim, N.; Jia, Z.; Zheng, X.; Cui, S.; Chen, W. A recent review of citrus flavanone naringenin on metabolic diseases and its potential sources for high yield-production. Trends Food Sci. Technol. 2018, 79, 35–54. [Google Scholar] [CrossRef]
- Ke, J.Y.; Banh, T.; Hsiao, Y.H.; Cole, R.M.; Straka, S.R.; Yee, L.D.; Belury, M.A. Citrus flavonoid naringenin reduces mammary tumor cell viability, adipose mass, and adipose inflammation in obese ovariectomized mice. Mol. Nutr. Food Res. 2017, 61, 1600934. [Google Scholar] [CrossRef] [PubMed]
- Pinho-Ribeiro, F.A.; Zarpelon, A.C.; Fattori, V.; Manchope, M.F.; Mizokami, S.S.; Casagrande, R.; Verri, W.A., Jr. Naringenin reduces inflammatory pain in mice. Neuropharmacology 2016, 105, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, J.; Zhang, X.-M.; Zhou, L.; Liao, X.-L.; Yang, B. Practical synthesis of naringenin. J. Chem. Res. 2015, 39, 455–457. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information, PubChem. Compound Database, cid=439246. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/439246 (accessed on 16 November 2018).
- Jayachitra, J.; Nalini, N. Effect of naringenin (citrus flavanone) on lipid profile in ethanol-induced toxicity in rats. J. Food Biochem. 2012, 36, 502–511. [Google Scholar] [CrossRef]
- Erlund, I.; Meririnne, E.; Alfthan, G.; Aro, A. Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice. J. Nutr. 2001, 131, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Frabasile, S.; Koishi, A.C.; Kuczera, D.; Silveira, G.F.; Verri, W.A., Jr.; Duarte Dos Santos, C.N.; Bordignon, J. The citrus flavanone naringenin impairs dengue virus replication in human cells. Sci. Rep. 2017, 7, 41864. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Hassandarvish, P.; Lani, R.; Yadollahi, P.; Jokar, A.; Bakar, S.A.; Zandi, K. Inhibition of chikungunya virus replication by hesperetin and naringenin. RSC Adv. 2016, 6, 69421–69430. [Google Scholar] [CrossRef]
- Goncalves, D.; Lima, C.; Ferreira, P.; Costa, P.; Costa, A.; Figueiredo, W.; Cesar, T. Orange juice as dietary source of antioxidants for patients with hepatitis c under antiviral therapy. Food Nutr. Res. 2017, 61, 1296675. [Google Scholar] [CrossRef] [PubMed]
- Nahmias, Y.; Goldwasser, J.; Casali, M.; van Poll, D.; Wakita, T.; Chung, R.T.; Yarmush, M.L. Apolipoprotein b-dependent hepatitis c virus secretion is inhibited by the grapefruit flavonoid naringenin. Hepatology 2008, 47, 1437–1445. [Google Scholar] [CrossRef] [PubMed]
- Da Pozzo, E.; Costa, B.; Cavallini, C.; Testai, L.; Martelli, A.; Calderone, V.; Martini, C. The citrus flavanone naringenin protects myocardial cells against age-associated damage. Oxidative Med. Cell. Longev. 2017, 2017, 9536148. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.K.; Ha, S.J.; Jung, C.H.; Kim, Y.T.; Lee, H.K.; Kim, M.O.; Lee, M.H.; Mottamal, M.; Bode, A.M.; Lee, K.W.; et al. Naringenin targets ERK2 and suppresses UVB-induced photoaging. J. Cell. Mol. Med. 2016, 20, 909–919. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, B.; Chen, X.; Zhang, N.; Li, G.; Zhang, L.H.; Tan, L.Y. Naringenin ameliorates behavioral dysfunction and neurological deficits in a d-galactose-induced aging mouse model through activation of PI3K/AKT/NRF2 pathway. Rejuvenation Res. 2017, 20, 462–472. [Google Scholar] [CrossRef]
- Ghofrani, S.; Joghataei, M.T.; Mohseni, S.; Baluchnejadmojarad, T.; Bagheri, M.; Khamse, S.; Roghani, M. Naringenin improves learning and memory in an Alzheimer’s disease rat model: Insights into the underlying mechanisms. Eur. J. Pharmacol. 2015, 764, 195–201. [Google Scholar] [CrossRef]
- Seyedrezazadeh, E.; Kolahian, S.; Shahbazfar, A.A.; Ansarin, K.; Pour Moghaddam, M.; Sakhinia, M.; Sakhinia, E.; Vafa, M. Effects of the flavanone combination hesperetin-naringenin, and orange and grapefruit juices, on airway inflammation and remodeling in a murine asthma model. Phytother. Res. 2015, 29, 591–598. [Google Scholar] [CrossRef]
- Chandrika, B.B.; Steephan, M.; Kumar, T.R.S.; Sabu, A.; Haridas, M. Hesperetin and naringenin sensitize HER2 positive cancer cells to death by serving as HER2 tyrosine kinase inhibitors. Life Sci. 2016, 160, 47–56. [Google Scholar] [CrossRef]
- Hernandez-Aquino, E.; Muriel, P. Beneficial effects of naringenin in liver diseases: Molecular mechanisms. World J. Gastroenterol. 2018, 24, 1679–1707. [Google Scholar] [CrossRef]
- Arul, D.; Subramanian, P. Naringenin (citrus flavonone) induces growth inhibition, cell cycle arrest and apoptosis in human hepatocellular carcinoma cells. Pathol. Oncol. Res. 2013, 19, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Park, S.; Bazer, F.W.; Song, G. Naringenin-induced apoptotic cell death in prostate cancer cells is mediated via the PI3K/AKT and MAPK signaling pathways. J. Cell. Biochem. 2017, 118, 1118–1131. [Google Scholar] [CrossRef] [PubMed]
- Nasr Bouzaiene, N.; Chaabane, F.; Sassi, A.; Chekir-Ghedira, L.; Ghedira, K. Effect of apigenin-7-glucoside, genkwanin and naringenin on tyrosinase activity and melanin synthesis in B16F10 melanoma cells. Life Sci. 2016, 144, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Stompor, M.; Uram, L.; Podgorski, R. In vitro effect of 8-prenylnaringenin and naringenin on fibroblasts and glioblastoma cells-cellular accumulation and cytotoxicity. Molecules 2017, 22, 1092. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Dong, W.; Zeng, W.; Zhang, L.; Zhang, C.; Qiu, Y.; Wang, L.; Yin, X.; Zhang, C.; Liang, W. Naringenin prevents TGF-beta1 secretion from breast cancer and suppresses pulmonary metastasis by inhibiting PKC activation. Breast Cancer Res. 2016, 18, 38. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Jeong, K.H.; Shin, W.H.; Bae, Y.S.; Jung, U.J.; Kim, S.R. Naringenin ameliorates kainic acid-induced morphological alterations in the dentate gyrus in a mouse model of temporal lobe epilepsy. Neuroreport 2016, 27, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Al-Rejaie, S.S.; Aleisa, A.M.; Abuohashish, H.M.; Parmar, M.Y.; Ola, M.S.; Al-Hosaini, A.A.; Ahmed, M.M. Naringenin neutralises oxidative stress and nerve growth factor discrepancy in experimental diabetic neuropathy. Neurol. Res. 2015, 37, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Al-Dosari, D.I.; Ahmed, M.M.; Al-Rejaie, S.S.; Alhomida, A.S.; Ola, M.S. Flavonoid naringenin attenuates oxidative stress, apoptosis and improves neurotrophic effects in the diabetic rat retina. Nutrients 2017, 9, 1161. [Google Scholar] [CrossRef]
- Sandeep, M.S.; Nandini, C.D. Influence of quercetin, naringenin and berberine on glucose transporters and insulin signalling molecules in brain of streptozotocin-induced diabetic rats. Biomed. Pharmacother. 2017, 94, 605–611. [Google Scholar]
- Ren, B.; Qin, W.; Wu, F.; Wang, S.; Pan, C.; Wang, L.; Zeng, B.; Ma, S.; Liang, J. Apigenin and naringenin regulate glucose and lipid metabolism, and ameliorate vascular dysfunction in type 2 diabetic rats. Eur. J. Pharmacol. 2016, 773, 13–23. [Google Scholar] [CrossRef]
- Roy, S.; Ahmed, F.; Banerjee, S.; Saha, U. Naringenin ameliorates streptozotocin-induced diabetic rat renal impairment by downregulation of TGF-beta1 and IL-1 via modulation of oxidative stress correlates with decreased apoptotic events. Pharm. Biol. 2016, 54, 1616–1627. [Google Scholar] [CrossRef] [PubMed]
- Sirovina, D.; Orsolic, N.; Gregorovic, G.; Koncic, M.Z. Naringenin ameliorates pathological changes in liver and kidney of diabetic mice: A preliminary study. Arch. Ind. Hyg. Toxicol. 2016, 67, 19–24. [Google Scholar]
- Shinyoshi, S.; Kamada, Y.; Matsusaki, K.; Chigwechokha, P.K.; Tepparin, S.; Araki, K.; Komatsu, M.; Shiozaki, K. Naringenin suppresses Edwardsiella tarda infection in GAKS cells by NanA sialidase inhibition. Fish Shellfish Immunol. 2017, 61, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.; Pan, T.; Zhu, A.L.; Zhang, M.H. Anti-inflammatory and anti-arthritic properties of naringenin via attenuation of NF-kappab and activation of the heme oxygenase HO-1/related factor 2 pathway. Pharmacol. Rep. 2017, 69, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Hua, F.Z.; Ying, J.; Zhang, J.; Wang, X.F.; Hu, Y.H.; Liang, Y.P.; Liu, Q.; Xu, G.H. Naringenin pre-treatment inhibits neuroapoptosis and ameliorates cognitive impairment in rats exposed to isoflurane anesthesia by regulating the PI3/AKT/PTEN signalling pathway and suppressing NF-kappab-mediated inflammation. Int. J. Mol. Med. 2016, 38, 1271–1280. [Google Scholar] [CrossRef]
- Park, S.; Lim, W.; Bazer, F.W.; Song, G. Naringenin induces mitochondria-mediated apoptosis and endoplasmic reticulum stress by regulating MAPK and AKT signal transduction pathways in endometriosis cells. Mol. Hum. Reprod. 2017, 23, 842–854. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, N.; Fan, S.; Zheng, X.; Yang, Y.; Zhu, Y.; Lu, Y.; Chen, Q.; Zhou, H.; Zheng, J. The citrus flavonoid naringenin confers protection in a murine endotoxaemia model through AMPK-ATF3-dependent negative regulation of the TLR4 signalling pathway. Sci. Rep. 2016, 6, 39735. [Google Scholar] [CrossRef]
- Shan, S.; Zhang, Y.; Wu, M.; Yi, B.; Wang, J.; Li, Q. Naringenin attenuates fibroblast activation and inflammatory response in a mechanical stretch-induced hypertrophic scar mouse model. Mol. Med. Rep. 2017, 16, 4643–4649. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Zeng, W.; Zhang, F.; Zhang, C.; Liang, W. Naringenin ameliorates acute inflammation by regulating intracellular cytokine degradation. J. Immunol. 2017, 199, 3466–3477. [Google Scholar] [CrossRef]
- Fouad, A.A.; Albuali, W.H.; Jresat, I. Protective effect of naringenin against lipopolysaccharide-induced acute lung injury in rats. Pharmacology 2016, 97, 224–232. [Google Scholar] [CrossRef]
- Shi, L.B.; Tang, P.F.; Zhang, W.; Zhao, Y.P.; Zhang, L.C.; Zhang, H. Naringenin inhibits spinal cord injury-induced activation of neutrophils through miR-223. Gene 2016, 592, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; Guo, L.; Tian, F.D.; An, N.; Luo, L.; Hao, R.H.; Wang, B.; Zhou, Z.H. Naringenin regulates production of matrix metalloproteinases in the knee-joint and primary cultured articular chondrocytes and alleviates pain in rat osteoarthritis model. Braz. J. Med. Biol. Res. 2017, 50, e5714. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.; Shahid, A.; Ali, N.; Hasan, S.K.; Majed, F.; Sultana, S. Amelioration of benzo[a]pyrene-induced oxidative stress and pulmonary toxicity by naringenin in Wistar rats: A plausible role of COX-2 and NF-kappab. Hum. Exp. Toxicol. 2017, 36, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Manchope, M.F.; Calixto-Campos, C.; Coelho-Silva, L.; Zarpelon, A.C.; Pinho-Ribeiro, F.A.; Georgetti, S.R.; Baracat, M.M.; Casagrande, R.; Verri, W.A., Jr. Naringenin inhibits superoxide anion-induced inflammatory pain: Role of oxidative stress, cytokines, Nrf-2 and the NO-cGMP-PKG-KATP channel signaling pathway. PLoS ONE 2016, 11, e0153015. [Google Scholar] [CrossRef] [PubMed]
- Chtourou, Y.; Kamoun, Z.; Zarrouk, W.; Kebieche, M.; Kallel, C.; Gdoura, R.; Fetoui, H. Naringenin ameliorates renal and platelet purinergic signalling alterations in high-cholesterol fed rats through the suppression of ROS and NF-kappab signaling pathways. Food Funct. 2016, 7, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Al-Roujayee, A.S. Naringenin improves the healing process of thermally-induced skin damage in rats. J. Int. Med Res. 2017, 45, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.H.; Wang, M.S.; Zeng, X.A.; Xu, X.M.; Brennan, C.S. Membrane and genomic DNA dual-targeting of citrus flavonoid naringenin against staphylococcus aureus. Integr. Biol. 2017, 9, 820–829. [Google Scholar] [CrossRef]
- Kozlowska, J.; Potaniec, B.; Zarowska, B.; Aniol, M. Synthesis and biological activity of novel o-alkyl derivatives of naringenin and their oximes. Molecules 2017, 22, 1485. [Google Scholar] [CrossRef]
- Liang, J.; Halipu, Y.; Hu, F.; Yakeya, B.; Chen, W.; Zhang, H.; Kang, X. Naringenin protects keratinocytes from oxidative stress injury via inhibition of the NOD2-mediated NF-kappab pathway in pemphigus vulgaris. Biomed. Pharmacother. 2017, 92, 796–801. [Google Scholar] [CrossRef]
- Stylos, E.; Chatziathanasiadou, M.V.; Tsiailanis, A.; Kellici, T.F.; Tsoumani, M.; Kostagianni, A.D.; Deligianni, M.; Tselepis, A.D.; Tzakos, A.G. Tailoring naringenin conjugates with amplified and triple antiplatelet activity profile: Rational design, synthesis, human plasma stability and in vitro evaluation. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2609–2618. [Google Scholar] [CrossRef]
- Wang, K.; Chen, Z.; Huang, J.; Huang, L.; Luo, N.; Liang, X.; Liang, M.; Xie, W. Naringenin prevents ischaemic stroke damage via anti-apoptotic and anti-oxidant effects. Clin. Exp. Pharmacol. Physiol. 2017, 44, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; An, W.; Gao, A. Protective effects of naringenin in cardiorenal syndrome. J. Surg. Res. 2016, 203, 416–423. [Google Scholar] [CrossRef]
- Tang, J.Y.; Jin, P.; He, Q.; Lu, L.H.; Ma, J.P.; Gao, W.L.; Bai, H.P.; Yang, J. Naringenin ameliorates hypoxia/reoxygenation-induced endoplasmic reticulum stress-mediated apoptosis in H9c2 myocardial cells: Involvement in ATF6, IRE1alpha and perk signaling activation. Mol. Cell. Biochem. 2017, 424, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Habauzit, V.; Verny, M.A.; Milenkovic, D.; Barber-Chamoux, N.; Mazur, A.; Dubray, C.; Morand, C. Flavanones protect from arterial stiffness in postmenopausal women consuming grapefruit juice for 6 mo: A randomized, controlled, crossover trial. Am. J. Clin. Nutr. 2015, 102, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Bawazeer, N.A.; Choudary, H.; Zamzami, M.A.; Abdulaal, W.H.; Zeyadi, M.; ALbukhari, A.; Middleton, B.; Moselhy, S.S. Possible regulation of LDL-receptor by naringenin in HepG2 hepatoma cell line. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 278–287. [Google Scholar] [CrossRef]
- Meng, X.M.; Zhang, Y.; Huang, X.R.; Ren, G.L.; Li, J.; Lan, H.Y. Treatment of renal fibrosis by rebalancing TGF-beta/Smad signaling with the combination of asiatic acid and naringenin. Oncotarget 2015, 6, 36984–36997. [Google Scholar] [CrossRef]
- Shi, R.; Xiao, Z.T.; Zheng, Y.J.; Zhang, Y.L.; Xu, J.W.; Huang, J.H.; Zhou, W.L.; Li, P.B.; Su, W.W. Naringenin regulates CFTR activation and expression in airway epithelial cells. Cell. Physiol. Biochem. 2017, 44, 1146–1160. [Google Scholar] [CrossRef] [PubMed]
- Oguido, A.; Hohmann, M.S.N.; Pinho-Ribeiro, F.A.; Crespigio, J.; Domiciano, T.P.; Verri, W.A., Jr.; Casella, A.M.B. Naringenin eye drops inhibit corneal neovascularization by anti-inflammatory and antioxidant mechanisms. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5764–5776. [Google Scholar] [CrossRef] [PubMed]
- Adana, M.Y.; Akang, E.N.; Peter, A.I.; Jegede, A.I.; Naidu, E.C.S.; Tiloke, C.; Chuturgoon, A.A.; Azu, O.O. Naringenin attenuates highly active antiretroviral therapy-induced sperm DNA fragmentations and testicular toxicity in sprague-dawley rats. Andrology 2018, 6, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Maatouk, M.; Elgueder, D.; Mustapha, N.; Chaaban, H.; Bzeouich, I.M.; Loannou, I.; Kilani, S.; Ghoul, M.; Ghedira, K.; Chekir-Ghedira, L. Effect of heated naringenin on immunomodulatory properties and cellular antioxidant activity. Cell Stress Chaperones 2016, 21, 1101–1109. [Google Scholar] [CrossRef]
- Lin, H.; Zhou, Z.; Zhong, W.; Huang, P.; Ma, N.; Zhang, Y.; Zhou, C.; Lai, Y.; Huang, S.; An, H.; et al. Naringenin inhibits alcoholic injury by improving lipid metabolism and reducing apoptosis in zebrafish larvae. Oncol. Rep. 2017, 38, 2877–2884. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.J.; Ku, K.L.; Lin, I.H.; Yeh, C.C. Naringenin attenuates hepatitis b virus x protein-induced hepatic steatosis. BMC Complement. Altern. Med. 2017, 17, 505. [Google Scholar] [CrossRef]
- Lim, W.; Song, G. Naringenin-induced migration of embrynoic trophectoderm cells is mediated via PI3K/AKT and ERK1/2 MAPK signaling cascades. Mol. Cell. Endocrinol. 2016, 428, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Tiku, A.B. Biochemical and molecular mechanisms of radioprotective effects of naringenin, a phytochemical from citrus fruits. J. Agric. Food Chem. 2016, 64, 1676–1685. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.Y.; Cole, R.M.; Hamad, E.M.; Hsiao, Y.H.; Cotten, B.M.; Powell, K.A.; Belury, M.A. Citrus flavonoid, naringenin, increases locomotor activity and reduces diacylglycerol accumulation in skeletal muscle of obese ovariectomized mice. Mol. Nutr. Food Res. 2016, 60, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Borges, G.; van der Hooft, J.; Clifford, M.N.; Del Rio, D.; Lean, M.E.; Roberts, S.A.; Kellerhals, M.B.; Crozier, A. Orange juice (poly)phenols are highly bioavailable in humans. Am. J. Clin. Nutr. 2014, 100, 1378–1384. [Google Scholar] [CrossRef]
- Kanaze, F.I.; Bounartzi, M.I.; Georgarakis, M.; Niopas, I. Pharmacokinetics of the citrus flavanone aglycones hesperetin and naringenin after single oral administration in human subjects. Eur. J. Clin. Nutr. 2007, 61, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Su, W.; Bai, Y.; Chen, T.; Yan, Z.; Wang, J.; Su, M.; Zheng, Y.; Peng, W.; Yao, H. Urinary metabolite profiling of flavonoids in Chinese volunteers after consumption of orange juice by UFLC-Q-TOF-MS/MS. J. Chromatogr. B 2017, 1061–1062, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Polyviou, T.; Ludwig, I.A.; Nastase, A.M.; Moreno-Rojas, J.M.; Garcia, A.L.; Malkova, D.; Crozier, A. Bioavailability of orange juice (poly)phenols: The impact of short-term cessation of training by male endurance athletes. Am. J. Clin. Nutr. 2017, 106, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Aschoff, J.K.; Riedl, K.M.; Cooperstone, J.L.; Hogel, J.; Bosy-Westphal, A.; Schwartz, S.J.; Carle, R.; Schweiggert, R.M. Urinary excretion of citrus flavanones and their major catabolites after consumption of fresh oranges and pasteurized orange juice: A randomized cross-over study. Mol. Nutr. Food Res. 2016, 60, 2602–2610. [Google Scholar] [CrossRef]
- Duque, A.L.R.F.; Monteiro, M.; Adorno, M.A.T.; Sakamoto, I.K.; Sivieri, K. An exploratory study on the influence of orange juice on gut microbiota using a dynamic colonic model. Food Res. Int. 2016, 84, 160–169. [Google Scholar] [CrossRef]
- Zaidun, N.H.; Thent, Z.C.; Latiff, A.A. Combating oxidative stress disorders with citrus flavonoid: Naringenin. Life Sci. 2018, 208, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Amawi, H.; Ashby, C.R., Jr.; Tiwari, A.K. Cancer chemoprevention through dietary flavonoids: What’s limiting? Chin. J. Cancer 2017, 36, 50. [Google Scholar] [CrossRef] [PubMed]
- Testai, L.; Calderone, V. Nutraceutical value of citrus flavanones and their implications in cardiovascular disease. Nutrients 2017, 9, 502. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hayasaka, S.; Shibata, Y.; Ojima, T.; Saegusa, T.; Gotoh, T.; Ishikawa, S.; Nakamura, Y.; Kayaba, K. Frequency of citrus fruit intake is associated with the incidence of cardiovascular disease: The Jichi Medical School cohort study. J. Epidemiol. 2011, 21, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Kumpulainen, J.; Jarvinen, R.; Rissanen, H.; Heliovaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef]
- Cassidy, A.; Rimm, E.B.; O’Reilly, E.J.; Logroscino, G.; Kay, C.; Chiuve, S.E.; Rexrode, K.M. Dietary flavonoids and risk of stroke in women. Stroke 2012, 43, 946–951. [Google Scholar] [CrossRef]
- Onakpoya, I.; O’Sullivan, J.; Heneghan, C.; Thompson, M. The effect of grapefruits (Citrus paradisi) on body weight and cardiovascular risk factors: A systematic review and meta-analysis of randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2017, 57, 602–612. [Google Scholar] [CrossRef]
- Reshef, N.; Hayari, Y.; Goren, C.; Boaz, M.; Madar, Z.; Knobler, H. Antihypertensive effect of sweetie fruit in patients with stage i hypertension. Am. J. Hypertens. 2005, 18, 1360–1363. [Google Scholar] [CrossRef]
- Toth, P.P.; Patti, A.M.; Nikolic, D.; Giglio, R.V.; Castellino, G.; Biancucci, T.; Geraci, F.; David, S.; Montalto, G.; Rizvi, A.; et al. Bergamot reduces plasma lipids, atherogenic small dense LDL, and subclinical atherosclerosis in subjects with moderate hypercholesterolemia: A 6 months prospective study. Front. Pharmacol. 2015, 6, 299. [Google Scholar] [CrossRef]
- Jung, U.J.; Kim, H.J.; Lee, J.S.; Lee, M.K.; Kim, H.O.; Park, E.J.; Kim, H.K.; Jeong, T.S.; Choi, M.S. Naringin supplementation lowers plasma lipids and enhances erythrocyte antioxidant enzyme activities in hypercholesterolemic subjects. Clin. Nutr. 2003, 22, 561–568. [Google Scholar] [CrossRef]
- Mollace, V.; Sacco, I.; Janda, E.; Malara, C.; Ventrice, D.; Colica, C.; Visalli, V.; Muscoli, S.; Ragusa, S.; Muscoli, C.; et al. Hypolipemic and hypoglycaemic activity of bergamot polyphenols: From animal models to human studies. Fitoterapia 2011, 82, 309–316. [Google Scholar] [CrossRef]
- Demonty, I.; Lin, Y.; Zebregs, Y.E.; Vermeer, M.A.; van der Knaap, H.C.; Jakel, M.; Trautwein, E.A. The citrus flavonoids hesperidin and naringin do not affect serum cholesterol in moderately hypercholesterolemic men and women. J. Nutr. 2010, 140, 1615–1620. [Google Scholar] [CrossRef] [PubMed]
- Constans, J.; Bennetau-Pelissero, C.; Martin, J.F.; Rock, E.; Mazur, A.; Bedel, A.; Morand, C.; Berard, A.M. Marked antioxidant effect of orange juice intake and its phytomicronutrients in a preliminary randomized cross-over trial on mild hypercholesterolemic men. Clin. Nutr. 2015, 34, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Silveira, J.Q.; Dourado, G.K.; Cesar, T.B. Red-fleshed sweet orange juice improves the risk factors for metabolic syndrome. Int. J. Food Sci. Nutr. 2015, 66, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Rendeiro, C.; Dong, H.; Saunders, C.; Harkness, L.; Blaze, M.; Hou, Y.; Belanger, R.L.; Corona, G.; Lovegrove, J.A.; Spencer, J.P. Flavanone-rich citrus beverages counteract the transient decline in postprandial endothelial function in humans: A randomised, controlled, double-masked, cross-over intervention study. Br. J. Nutr. 2016, 116, 1999–2010. [Google Scholar] [CrossRef]
- Dallas, C.; Gerbi, A.; Elbez, Y.; Caillard, P.; Zamaria, N.; Cloarec, M. Clinical study to assess the efficacy and safety of a citrus polyphenolic extract of red orange, grapefruit, and orange (Sinetrol-XPur) on weight management and metabolic parameters in healthy overweight individuals. Phytother. Res. 2014, 28, 212–218. [Google Scholar] [CrossRef]
- Stohs, S.J.; Preuss, H.G.; Keith, S.C.; Keith, P.L.; Miller, H.; Kaats, G.R. Effects of p-synephrine alone and in combination with selected bioflavonoids on resting metabolism, blood pressure, heart rate and self-reported mood changes. Int. J. Med Sci. 2011, 8, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Aldrich, C.; Bertozzi, C.; Georg, G.I.; Kiessling, L.; Lindsley, C.; Liotta, D.; Merz, K.M.; Schepartz, A.; Wang, S. The ecstasy and agony of assay interference compounds. ACS Cent. Sci. 2017, 3, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Baell, J.B. Feeling nature’s pains: Natural products, natural product drugs, and pan assay interference compounds (pains). J. Nat. Prod. 2016, 79, 616–628. [Google Scholar] [CrossRef] [PubMed]
- Jardim, A.C.G.; Shimizu, J.F.; Rahal, P.; Harris, M. Plant-derived antivirals against hepatitis c virus infection. Virol. J. 2018, 15, 34. [Google Scholar] [CrossRef] [PubMed]
| Therapeutics | Diseases | Treatment | Targets and Effects | Route | Experimental Model | Ref. |
|---|---|---|---|---|---|---|
| Anti-Hepatitis C virus | Hepatitis C | 2.7 mg/500 mL | Lipid profile and liver enzyme AST (decreased) | p.o. | Adult patients | [25] |
| 200 µM | Inhibition of apolipoprotein B secretion | - | In vitro, Huh7.5.1 human hepatoma cell | [26] | ||
| Antiaging | Aging-associated damage | 4–40 μM | Reduction of senescence markers (X-gal, cell cycle regulator), oxidative stress (radical oxidative species, mitochondrial metabolic activity, mitochondrial calcium buffer capacity, estrogenic signaling functions) | - | In vitro, H9c2 embryonic rat cells | [27] |
| Photoaging | 1–4 MED (45 mJ/cm2) | Anti-photoaging effects by suppression of ERK2 activity and decrease of FRA1 stability, AP-1 transactivation and MMP-1 expression | - | In vitro, HaCaT keratinocyte cell line and the BJ human fibroblast cell | [28] | |
| Senescence process | 50 mg/kg | Promotion of PI3K/Akt signaling, nuclear factor-erythroid 2-related factor 2, heme oxygenase 1, NAD(P)H-quinone oxidoreductase 1 | p.o. | In vivo, mice | [29] | |
| Anti-Alzheimer | Alzheimer | 100 mg/kg | Mitigation of lipid peroxidation and apoptosis, attenuation of impairment of learning and memory | p.o. | In vivo, Wistar rats | [30] |
| Antiasthma | Asthma | 9 mg/100 mL of the prepared fluid | Lowered subepithelial fibrosis, smooth muscle hypertrophy, and lung atelectasis | p.o. | In vivo, BALB/c mice | [31] |
| Anticancer | Breast cancer | 250 µM | Inhibition of HER2-TK activity, anti-proliferative, pro-apoptotic and anti-cancerous activity | - | In vitro, SKBR3 and MDA-MB-231 breast tumor cells | [32] |
| Liver cancer | 100–200 μM | Block in G0/G1 and G2/M phase, accumulation of p53, apoptosis induction by nuclei damage, increased ratio of Bax/Bcl-2, release of cytochrome C, and sequential activation of caspase-3 | p.o. | In vitro, human hepatocellular carcinoma HepG2 cells | [33,34] | |
| Postmenopausal breast cancer | High-fat (HF), high-fat diet with low naringenin (LN; 1% naringenin) or high-fat diet with high naringenin (HN; 3% naringenin) | Inhibition of cell growth, increases phosphorylation of AMP-activated protein kinase, down-regulation of CyclinD1 expression, and induction cell death. In vivo, delay of tumor growth (whereas no alteration of final tumor weight was observed) | p.o. | In vitro, E0771 mammary tumor cells. In vivo, ovariectomized C57BL/6 mice injected with E0771 cells | [17] | |
| Prostate cancer | 5–50 μM | Inhibition of proliferation and migration, induction of apoptosis and ROS production. Loss of mitochondrial membrane potential and increased ratio of Bax/Bcl-2 | - | In vitro, PC3 and LNCaP prostate cancer cells | [35] | |
| Melanoma | 25–100 μM | Antiproliferative activity, increase of subG0/G1, S and G2/M phase cell proportion, decrease of cell proportion in G0/G1 phases | - | In vitro, B16F10 melanoma cells | [36] | |
| Gliomas-brain cancer | 211 µM | Cytotoxicity | - | In vitro, human glioblastoma U-118 MG cells | [37] | |
| Breast cancer | 200 mg/kg | Decreased secretion of TGF-β1 and accumulation of intracellular TGF-β1. Inhibition of TGF-β1 transport from the trans-Golgi network, and PKC activity | - | In vivo, Balb/c mice inoculated with breast carcinoma 4T1-Luc2 cells | [38] | |
| Anti-Chikungunya virus | Chikungunya infection | 6.818 µM | Inhibition of CHIKV intracellular replication | - | In vitro, CHIKV infected hamster kidney cells (BHK-21) | [24] |
| Anticonvulsant | Epilepsy | 50–100 mg/kg | Inhibited production of TNFα and IL-1β, delaying the onset of seizures, and inhibiting activation of the mammalian target of rapamycin complex 1 | p.o. | In vivo, male C57BL/6 mice injected with kainic acid | [39] |
| Anti-dengue virus | Dengue | 250 μM | Prevention of infection | - | In vitro, dengue virus infected human-derived Huh7.5 hepatoma cell | [23] |
| Antidiabetic | Diabetic neuropathy | 25–50 mg/kg | Attenuation of diabetic-induced changes in serum glucose, insulin and pro-inflammatory cytokines (TNF-alpha, IL-1beta, and IL-6). Attenuation of oxidative stress biomarkers. Decrease of insulin growth factor and nerve growth factor | p.o. | In vivo, streptozotocin-induced diabetic rats | [40] |
| Diabetic retinopathy | 50 mg/kg | Amelioration of oxidative stress, neurotrophic factors (brain derived neurotrophic factor (BDNF)), tropomyosin related kinase B (TrkB) and synaptophysin), and apoptosis regulatory proteins (Bcl-2, Bax, and caspase-3) | p.o. | In vivo, streptozotocin-induced diabetic rats | [41] | |
| Diabetes | 0.05% | Improved glucose transporters (GLUTs 1, 3), and insulin receptor substrate 1 (IRS 1) levels | p.o. | In vivo, streptozotocin-induced diabetic rats | [42] | |
| Vascular endothelial dysfunction | 50–100 mg/kg | Lowered levels of blood glucose, serum lipid, malonaldehyde, ICAM-1 and insulin resistance index, increased SOD activity and improved impaired glucose tolerance | p.o. | In vivo, streptozotocin-induced diabetic rats | [43] | |
| Diabetic renal impairment | 5–10 mg/kg | Decrease in malondialdehyde levels, and affected superoxide dismutase, catalase and glutathione enzyme activities. Reduction in apoptosis activity, TGF-β1, and IL-1 expression | p.o. | In vivo, streptozotocin-induced diabetic rats | [44] | |
| Diabetes complications | 50 mg/kg | Decreased lipid peroxidation level in liver and kidney tissue | p.o. | In vivo, alloxan-induced diabetic mice | [45] | |
| Anti-Edwardsiellosis | Edwardsiellosis | 200–400 µM | Down-regulation of Edwardsiella tarda infections | - | In vitro, Goldfish scale fibroblast (GAKS) cells | [46] |
| Anti-hyperlipidemic | Alcohol abuse, alcohol intolerance, alcohol dependence and other alcohol related disabilities | 50 mg/kg | Decreased levels of plasma and tissue total cholesterol, triglycerides, free fatty acids, HMG CoA reductase and collagen content | p.o. | In vivo, male Wistar rats | [21] |
| Anti-inflammatory | Arthritic inflammation | 5–20 mg/kg | Down-regulation of TNF-α, and NF-κB mRNA. Increased Nrf-2/HO-1s | p.o. | In vivo, Wistar rats | [47] |
| Cognitive effect-memory impairment | 25–100 mg/kg | Decreased expression of caspase-3, Bad, Bax, NF-κB, tumor necrosis factor-α, interleukin (IL)-6 and IL-1β | p.o. | In vivo, newborn Sprague-Dawley rats | [48] | |
| Endometriosis | 5–100 µM | Antiproliferative and proapoptotic effect (Bax and Bak increased, activated MAPK and inactivated PI3K). Depolarization of mitochondrial membrane potential Activation of eIF2α and IRE1α, GADD153 and GRP78 proteins | - | In vitro, VK2/E6E7, vaginal mucosa derived epithelial endometriosis cells, and End1/E6E7, endocervix epithelial derived endometriotic cells | [49] | |
| Endotoxaemia | 10 mg/kg | Suppression of TNF-α, IL-6, TLR4, inducible NO synthase (iNOS), cyclo-oxygenase-2 (COX2) and NADPH oxidase-2 (NOX2), NF-κB and mitogen-activated protein kinase (MAPK) | p.o. | In vivo, BALB/c mice In vitro, peritoneal macrophages obtained from the rats | [50] | |
| Hypertrophic scars (HS) | 25–50 µM | Inhibition of hypertrophic scars. Downregulation of TNF-α, IL-1β, IL-6 and TGF-β1 | p.o. | In vivo, female KM mice | [51] | |
| Liver diseases | 50 mg/kg | Inhibition of oxidative stress, through TGF-β pathway and prevention of the trans-differentiation of hepatic stellate cells (HSC). Pro-apoptotic effect, inhibition of MAPK, TLR, VEGF, and TGF-β, Modulation of lipids and cholesterol synthesis. | p.o. | In vivo | [33] | |
| LPS-induced endotoxemia and Con A–induced hepatitis | 100 μM 50 mg/kg 10 mg/kg | Post-translational inhibition of TNF-α and IL-6 (no interfering with TLR signaling cascade, cytokine mRNA stability, or protein translation) | - p.o. i.p. | In vitro, murine macrophage cell line RAW264.7 In vivo, female C57BL/6 mice In vivo, female BALB/c mice | [52] | |
| Lung injury | 50–100 mg/kg | Down-regulation of nuclear factor-x03BA;B, inducible NO synthase, tumor necrosis factor-α, caspase-3; increased heat shock protein 70 | p.o. | In vivo, rats | [53] | |
| Neuroinflammation-spinal cord injury | 50–100 mg/kg | Repression of miR-223 | p.o. | In vivo, female Wistar rats | [54] | |
| Osteoarthritis | 40 mg/kg | Reduction in pain behavior and improvement in the tissue morphology. Inhibition of MMP-3 expression and NF-κB pathway | p.o. | In vivo, male Wistar rats | [55] | |
| Oxidative stress and lung damage | 100 mg/kg | Reduction of oxidative stress, increase of antioxidant enzymes. Down-regulation of NF-κB, and COX-2 | p.o. | In vivo, Wistar rats | [56] | |
| Pain | 16.7–150 mg/kg | Analgesic effect, through activation of NO−cGMP−PKG−ATP-sensitive potassium channel pathway. Reduction of neutrophil recruitment, tissue oxidative stress, and cytokine production (IL-33, TNF-α, and IL-1β). Downregulation of mRNA expression of gp91phox, cyclooxygenase (COX)-2, and preproendothelin-1. Upregulation of nuclear factor (erythroid-derived 2)-like 2 (Nrf2) mRNA, and heme oxygenase (HO-1) mRNA expression, and NF-κB | p.o. | In vivo, male Swiss mice | [18,57] | |
| Protective effect on renal failure | 50 mg/Kg | Improvement of renal markers. Decreased lipid profile and inhibition of pro-oxidant and inflammation markers | - | In vivo, rats | [58] | |
| Skin damage-burns | 25–100 mg/kg | Inhibition of TNF-α, IL-1β, IL-6, NO, PGE2, caspase-3, LTB4 and NF-κB levels. Increased SOD, catalase, GPx and GST activities | p.o. | In vivo, male Wistar albino rats | [59] | |
| Antimicrobial | Food-borne Staphylococcus aureus | 0.92–3.68 mM) | Increased bacterial membrane permeability and changed cell morphology | - | In vitro, Staphylococcus aureus ATCC 6538 | [60] |
| Escherichia coli, Staphylococcus aureus, Candida albicans, Alternaria alternata, Fusarium linii, Aspergillus niger | OD in the range of 0–0.49 vs. 1.87 for controls | Antibacterial activity | - | In vitro, Escherichia coli ATCC10536, Staphylococcus aureus DSM799, Candida albicans DSM1386, Alternaria alternata CBS1526, Fusarium linii KB-F1, and Aspergillus niger DSM1957 | [61] | |
| Antioxidant | Skin injury | Pemphigus vulgaris (PV) serum treated HaCaT cell | Down-regulation of Dsg1, Dsg3, E-cadherin, ROS production, amelioration of the drop of mitochondrial membrane potential. Increase of the activity of SOD, GSH-Px and TAC. Decreased of NOD2, RIPK2 and NF-κB p-p65, | - | In vitro, human keratinocyte cell line HaCaT | [62] |
| Antiplatelet | Cardiovascular diseases | - | Antiplatelet activity targeting PAR-1, P2Y12 and COX-1 platelet activation pathways | - | In silico | [63] |
| Anti-stroke damage | Ischaemic stroke | 20–80 µM | Inhibition of apoptosis and oxidative stress, and regulation of the localization of Nrf2 protein | p.o. | In vivo/in vitro, cortical neuron cells isolated from neonatal Sprague-Dawley rats | [64] |
| Cardioprotective | Cardiorenal syndrome | 50 mM; 25–50 mg/kg | Attenuation of cardiac remodeling and cardiac dysfunction, decrease of left ventricle weight (LVW), increase of body weight (BW), decrease of LVW/BW, blood urea, type-B natriuretic peptide, aldosterone, angiotensin (Ang) II, C-reactive protein | p.o. | In vivo, male Sprague Dawley rats In vitro, cardiac fibroblasts | [65] |
| Hypoxia/reoxygenation (H/R) injury | 80 µM | Overexpression of Bcl-2, glucose-regulated protein 78, cleaved activating transcription factor 6 (ATF6) and phosphorylation levels of phospho-extracellular regulated protein kinases (PERK). Decrease of caspase-3, and Bax | - | In vitro, rat cardiomyocyte H9c2 cells | [66] | |
| Arterial stiffness in postmenopausal | 210 mg/day | Decreased carotid-femoral pulse wave velocity | p.o. | Patients, healthy postmenopausal women | [67] | |
| Atherosclerosis and coronary heart diseases | 200 µM | Upregulation of SREBP-1a promoter activity | - | In vitro, human hepatoma HepG2 cells | [68] | |
| Chronic kidney disease | Renal fibrosis/ obstructive nephropathy | 50 mg/kg | Inhibition of Smad3 phosphorylation and transcription | p.o. | In vivo, C57BL6 male mice | [69] |
| Expectorant | Sputum symptoms | 100 µM | Increase of CFTR expression, stimulation of chloride anion secretion | apical | In vivo, Sprague-Dawley rats | [70] |
| Eye-protective | Corneal neovascularization | 0.08–80 µg; 8 µL of 0.01–10 g/L solution | Inhibition of alkali burn-induced neutrophil (myeloperoxidase activity and recruitment of Lysm-GFP+ cells) and macrophage (N-acetyl-β-D glycosaminidase activity) recruitment. Inhibition of IL-1β., IL-6 production, Vegf, Pdgf, and Mmp14 mRNA expression | Eye drop | In vivo, male Swiss mice | [71] |
| Fertility | Infertility | 40–80 mg/kg | Attenuation of DNA fragmentation and sperm count during antiretroviral therapy | p.o. | In vivo, male Sprague-Dawley rats | [72] |
| Immunomodulatory | Immunodepression | 5.4–21.6 μg/mL | Increase of B cell proliferation, and NK activity | - | In vitro, spleen mice lymphocytes and peritoneal macrophages obtained from pathogen-free male BALB/c mice | [73] |
| Laxative | Constipation | 75–300 mg/kg | Amelioration of constipation, increased c-Kit, SCF, and aquaporin 3 | p.o. | In vivo, ICR mice | [15] |
| Hepatoprotective | Alcoholic liver disease/steatosis | 2.5–10 mg/kg | Reduction of alcohol-related gene expression (cyp2y3, cyp3a65, hmgcra, hmgcrb, fasn, fabp10α, fads2 and echs1) | - | In vivo, zebrafish larvae | [74] |
| Hepatitis B virus protein X (HBx)-induced hepatic steatosis | 30 mg/kg | Down-regulation of SREBP1c, LXRα, and PPARγ genes | p.o. | In vivo, HBx-transgenic C57BL/6 mice In vitro, HBx-transfected human hepatoma HepG2 cells | [75] | |
| Pregnancy | Migration mechanism(s) of peri-implantation conceptuses | 20 µM | Stimulation of pTr cells migration, through PI3K/AKT and ERK1/2 MAPK signaling pathways | - | In vitro, porcine trophectoderm (pTr) cells | [76] |
| Radioprotective | Radiation-induced DNA, chromosomal and membrane damage. | 50 mg/kg | Inhibition of NF-kB pathway, apoptotic proteins: p53, Bax, Bcl-2 | p.o. | In vivo, Swiss albino mice | [77] |
| Weight loss | Obesity: Muscle loss and metabolic syndrome in postmenopausal women. | 3% naringenin diet | Down-regulation of genes involved in de novo lipogenesis, lipolysis and triglyceride synthesis/storage | p.o. | In vivo, C57BL/6J mice | [78] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals 2019, 12, 11. https://doi.org/10.3390/ph12010011
Salehi B, Fokou PVT, Sharifi-Rad M, Zucca P, Pezzani R, Martins N, Sharifi-Rad J. The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals. 2019; 12(1):11. https://doi.org/10.3390/ph12010011
Chicago/Turabian StyleSalehi, Bahare, Patrick Valere Tsouh Fokou, Mehdi Sharifi-Rad, Paolo Zucca, Raffaele Pezzani, Natália Martins, and Javad Sharifi-Rad. 2019. "The Therapeutic Potential of Naringenin: A Review of Clinical Trials" Pharmaceuticals 12, no. 1: 11. https://doi.org/10.3390/ph12010011
APA StyleSalehi, B., Fokou, P. V. T., Sharifi-Rad, M., Zucca, P., Pezzani, R., Martins, N., & Sharifi-Rad, J. (2019). The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals, 12(1), 11. https://doi.org/10.3390/ph12010011









