Antimicrobial Peptides for Topical Treatment of Osteomyelitis and Implant-Related Infections: Study in the Spongy Bone
Abstract
1. Introduction
2. Results
2.1. Design of Peptide Analogues, Their Biological Activities and Structure
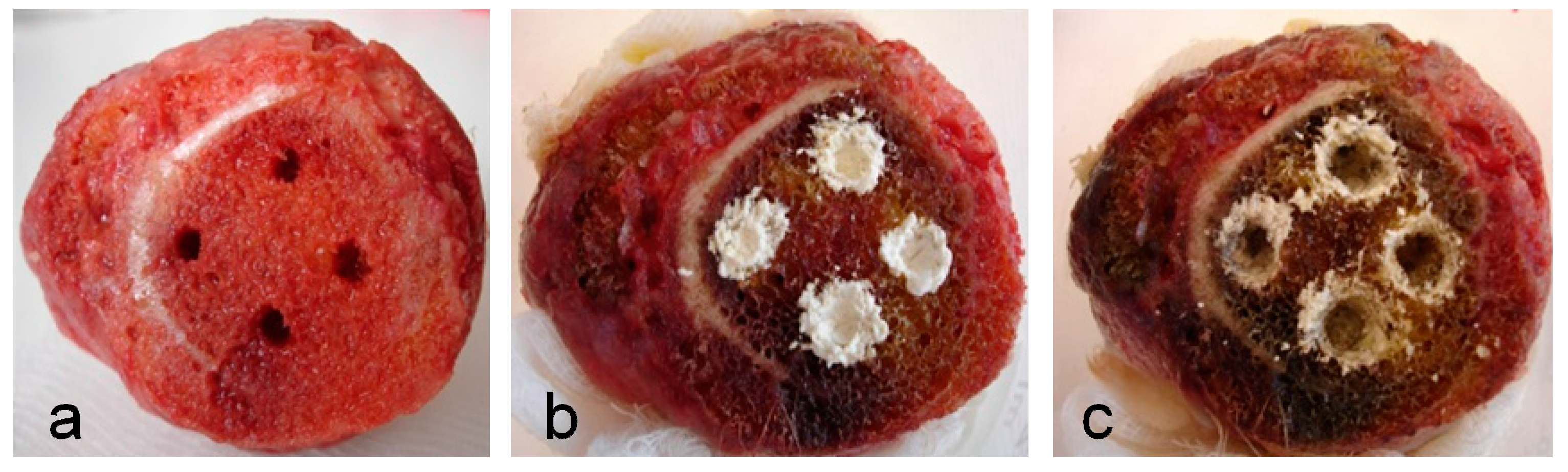
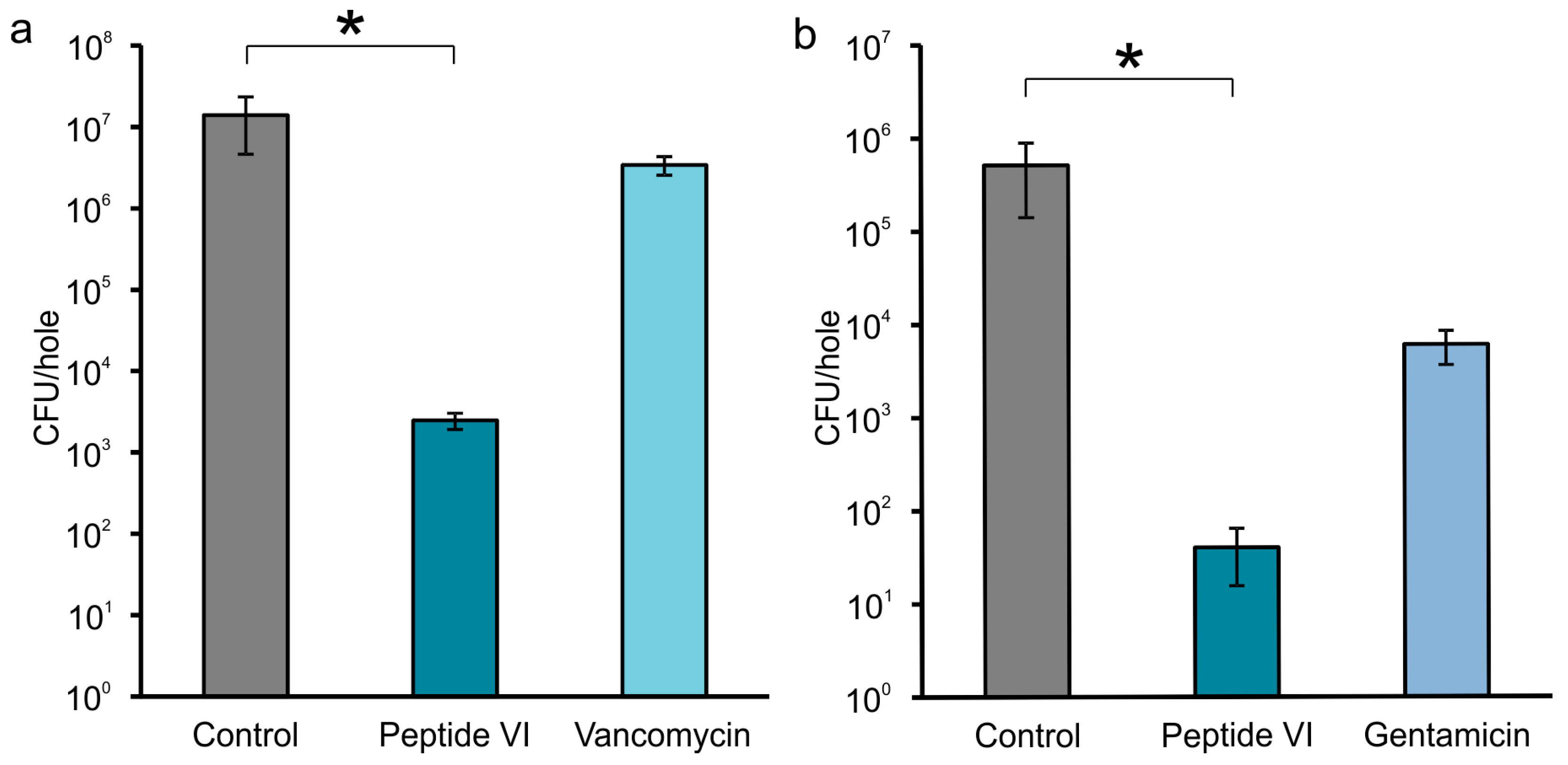
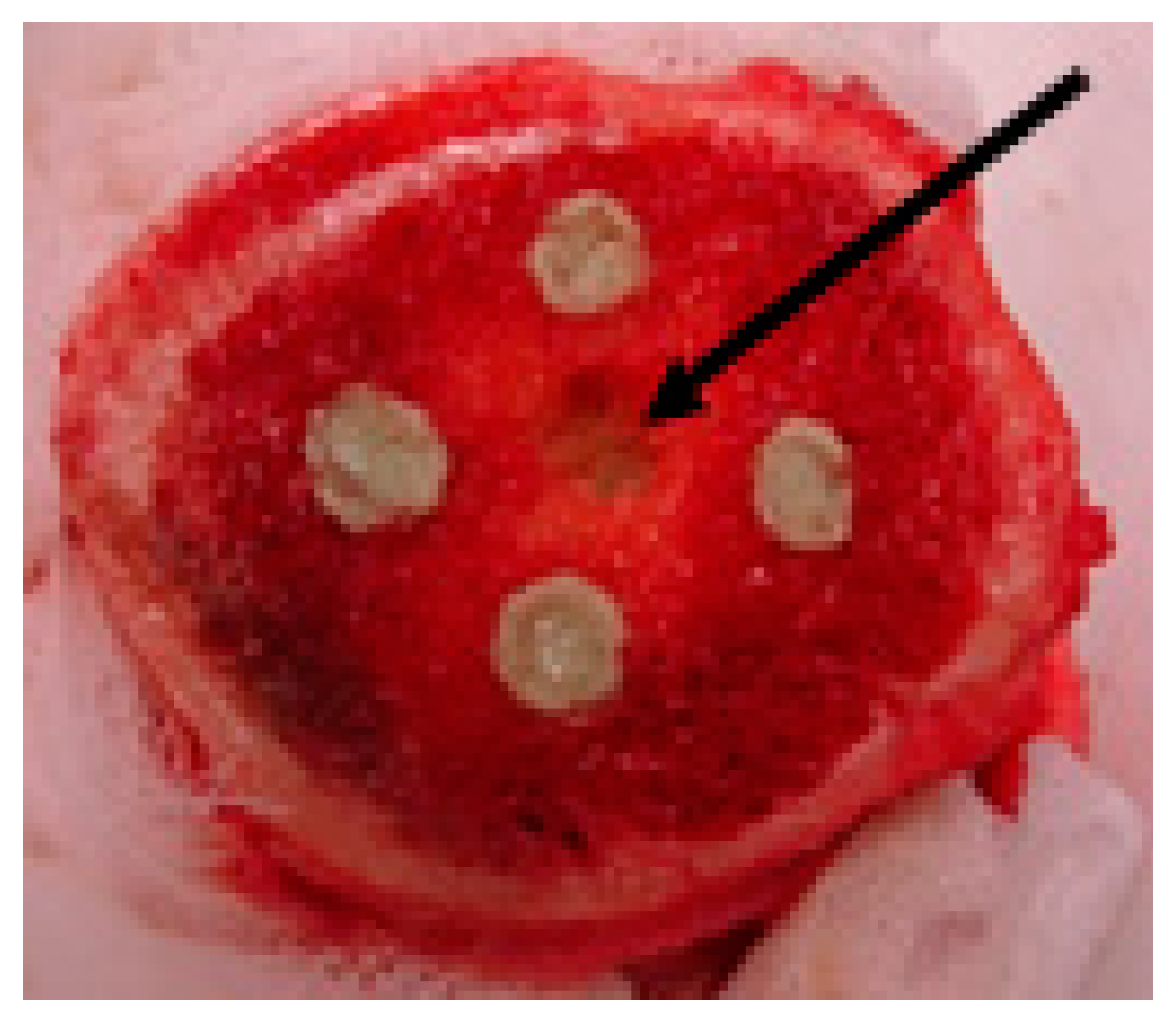
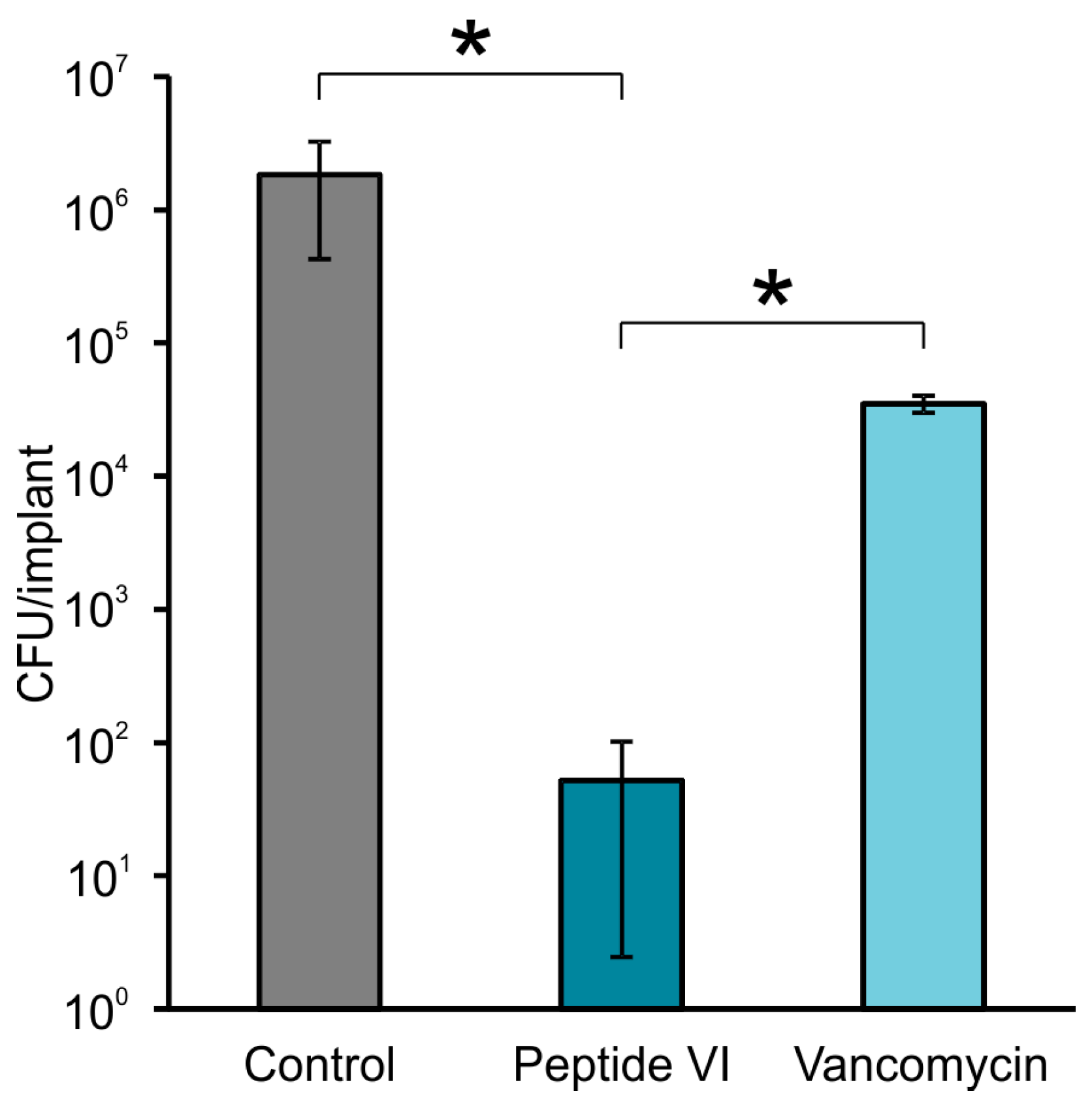
2.2. Efficacy of AMPs in Experimental Model of Bone Infection
2.3. AMPs Preventing Biofilm Formation on the Surface of Model Implants
3. Discussion
4. Materials and Methods
4.1. Bone Samples and Microbial Strains
4.2. Peptide Synthesis
4.3. Antimicrobial Activity Determination and Haemolysis Assay
4.4. Efficacy of AMPs in Experimental Model of Bone Infection
4.5. AMPs Preventing Biofilm Formation on Surface of Model Implants
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Birt, M.C.; Anderson, D.W.; Toby, E.B.; Wang, J. Osteomyelitis: Recent advances in pathophysiology and therapeutic strategies. J. Orthop. 2017, 14, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Nandi, S.K.; Bandyopadhyay, S.; Das, P.; Samanta, I.; Mukherjee, P.; Roy, S.; Kundu, B. Understanding osteomyelitis and its treatment through local drug delivery system. Biotechnol. Adv. 2016, 34, 1305–1317. [Google Scholar] [CrossRef] [PubMed]
- Winkler, H.; Haiden, P. Treatment of chronic bone infection. Oper. Tech. Orthop. 2016, 26, 2–11. [Google Scholar] [CrossRef]
- Hanssen, A.D.; Osmon, D.R.; Patel, R. Local antibiotic delivery systems: Where are we and where are we going? Clin. Orthop. Relat. Res. 2005, 437, 111–114. [Google Scholar] [CrossRef]
- Webb, N.D.; McCanless, J.D.; Courtney, H.S.; Bumgardner, J.D.; Haggard, W.O. Daptomycin eluted from calcium sulfate appears effective against Staphylococcus. Clin. Orthop. Relat. Res. 2008, 466, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Gálvez-López, R.; Peña-Monje, A.; Antelo-Lorenzo, R.; Guardia-Olmedo, J.; Moliz, J.; Hernández-Quero, J.; Parra-Ruiz, J. Elution kinetics, antimicrobial activity, and mechanical properties of 11 different antibiotic loaded acrylic bone cement. Diagn. Microbiol. Infect. Dis. 2014, 78, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.; Leung, F.; Duncan, C.; Mugabe, C.; Burt, H. The use of bone cement for the localized, controlled release of the antibiotics vancomycin, linezolid, or fusidic acid: Effect of additives on drug release rates and mechanical strength. Drug Deliv. Transl. Res. 2011, 1, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.C.J.; Spencer, R.F. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. J. Bone Jt. Surg. Br. 2007, 89, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Rouse, M.S.; Piper, K.E.; Jacobson, M.; Jacofsky, D.J.; Steckelberg, J.M.; Patel, R. Daptomycin treatment of Staphylococcus aureus experimental chronic osteomyelitis. J. Antimicrob. Chemother. 2006, 57, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Montanaro, L.; Speziale, P.; Arciola, C.R. Antibiotic-loaded biomaterials and the risks for the spread of antibiotic resistance following their prophylactic and therapeutic clinical use. Biomaterials 2010, 31, 6363–6377. [Google Scholar] [CrossRef] [PubMed]
- Arciola, C.R.; Campoccia, D.; Ehrlich, G.D.; Montanaro, L. Biofilm-based implant infections in orthopaedics. Adv. Exp. Med. Biol. 2015, 830, 29–46. [Google Scholar] [PubMed]
- Romano, C.L.; Toscano, M.; Romano, D.; Drago, L. Antibiofilm agents and implant-related infections in orthopaedics: Where are we? J. Chemother. 2013, 25, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.; Monteiro, F.J.; Ferraz, M.P. Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomaterials 2012, 2, 176–194. [Google Scholar] [CrossRef] [PubMed]
- Brogden, N.K.; Brogden, K.A. Will new generations of modified antimicrobial peptides improve their potential as pharmaceuticals? Int. J. Antimicrob. Agents 2011, 38, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Baltzer, S.A.; Brown, M.H. Antimicrobial peptides—Promising alternatives to conventional antibiotics. J. Mol. Microbiol. Biotechnol. 2011, 20, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Reiling, S.; Zarena, D.; Wang, G. Host defense antimicrobial peptides as antibiotics: Design and application strategies. Curr. Opin. Chem. Biol. 2017, 38, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, M.R.; Yount, N.Y. Mechanisms of antimicrobial peptide action and resistance. Pharmacol. Rev. 2003, 55, 27–55. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Haney, E.F.; Vogel, H.J. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol. 2011, 29, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gallo, R.L. Antimicrobial peptides. Curr. Biol. 2016, 26, R14–R19. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Wang, G. Individual and combined effects of engineered peptides and antibiotics on Pseudomonas aeruginosa biofilms. Pharmaceuticals 2017, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Faber, C.; Stallmann, H.P.; Lyaruu, D.M.; de Blieck, J.M.A.; Bervoets, T.J.M.; van Nieuw Amerongen, A.; Wuisman, P.I.J.M. Release of antimicrobial peptide Dhvar-5 from polymethylmethacrylate beads. J. Antimicrob. Chemother. 2003, 51, 1359–1364. [Google Scholar] [CrossRef] [PubMed]
- Stallmann, H.P.; Faber, C.; Bronckers, A.L.J.J.; van Nieuw Amerongen, A.; Wuisman, P.I.J.M. Osteomyelitis prevention in rabbits using antimicrobial peptide hLF1-11- or gentamicin-containing calcium phosphate cement. J. Antimicrob. Chemother. 2004, 54, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Faber, C.; Hoogendoorn, R.J.W.; Stallmann, H.P.; Lyaruu, D.M.; van Nieuw Amerongen, A.; Wuisman, P.I.J.M. In vivo comparison of Dhvar-5 and gentamicin in MRSA osteomyelitis prevention model. J. Antimicrob. Chemother. 2004, 54, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Faber, C.; Stallmann, H.P.; Lyaruu, D.M.; Joosten, U.; von Eiff, C.; van Nieuw Amerongen, A.; Wuisman, P.I.J.M. Comparable efficacies of the antimicrobial peptide human lactoferrin 1-11 and gentamicin in a chronic methicillin-resistant Staphylococcus aureus osteomyelitis model. Antimicrob. Agents Chemother. 2005, 49, 2438–2444. [Google Scholar] [CrossRef] [PubMed]
- Dijkshoorn, L.; Brouwer, C.P.J.M.; Bogaards, S.J.P.; Nemec, A.; van den Broek, P.J.; Nibbering, P.H. The synthetic N-terminal peptide of human lactoferin, hLF(1-11), is highly effective against experimental infection caused by multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2004, 48, 4919–4921. [Google Scholar] [CrossRef] [PubMed]
- De Breij, A.; Riool, M.; Kwakman, P.H.S.; de Boer, L.; Cordfunke, R.A.; Drijfhout, J.W.; Cohen, O.; Emanuel, N.; Zaat, S.A.J.; Nibbering, P.H.; et al. Prevention of Staphylococcus aureus biomaterial-associated infections using a polymer-lipid coating containing the antimicrobial peptide OP-145. J. Control. Release 2016, 222, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Laverty, G.; Gorman, S.P.; Gilmore, B.F. Antimicrobial peptide incorporated poly(2-hydroxyethyl methacrylate) hydrogels for the prevention of Staphylococcus epidermidis-associated biomaterial infections. J. Biomed. Mater. Res. Part A 2012, 100A, 1803–1814. [Google Scholar] [CrossRef] [PubMed]
- Čeřovský, V.; Buděšínský, M.; Hovorka, O.; Cvačka, J.; Voburka, Z.; Slaninová, J.; Borovičková, L.; Fučík, V.; Bednárová, L.; Votruba, I.; et al. Lasioglossins: Three novel antimicrobial peptides from the venom of the eusocial bee Lasioglossum laticeps (Hymenoptera: Halictidae). ChemBioChem 2009, 10, 2089–2099. [Google Scholar] [CrossRef] [PubMed]
- Monincová, L.; Buděšínský, M.; Slaninová, J.; Hovorka, O.; Cvačka, J.; Voburka, Z.; Fučík, V.; Borovičková, L.; Bednárová, L.; Straka, J.; et al. Novel antimicrobial peptides from the venom of the eusocial bee Halictus sexcinctus (Hymenoptera: Halictidae) and their analogs. Amino Acids 2010, 39, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Li, X.; Wang, Z. APD2: The updated antimicrobial peptide database and its application in peptide design. Nucleic Acids Res. 2009, 37, D933–D937. [Google Scholar] [CrossRef] [PubMed]
- Nešuta, O.; Buděšínský, M.; Hadravová, R.; Monincová, L.; Humpoličková, J.; Čeřovský, V. How proteases from Enterococcus faecalis contribute to its resistance to short α-helical antimicrobial peptides. Pathog. Dis. 2017, 75, ftx091. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Ericksen, B.; Tucker, K.; Lubkowski, J.; Lu, W. Synthesis and characterization of human α-defensins 4-6. J. Pept. Res. 2004, 64, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.J.; Groundwater, P.W.; Todd, A.; Worsley, A.J. Antibacterial Agents: Chemistry, Mode of Action, Mechanism of Resistance and Clinical Applications, 1st ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2012; Chapters 5.2.4–5.2.6; pp. 308–313. [Google Scholar]
- Anderson, R.J.; Groundwater, P.W.; Todd, A.; Worsley, A.J. Antibacterial Agents: Chemistry, Mode of Action, Mechanism of Resistance and Clinical Applications, 1st ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2012; Chapters 4.1.4–4.1.6; pp. 158–161. [Google Scholar]
- Melicherčík, P.; Čeřovský, V.; Nešuta, O.; Jahoda, D.; Landor, I.; Ballay, R.; Fulín, P. Testing the efficacy of antimicrobial peptides in the topical treatment of induced osteomyelitis in rats. Folia Microbiol. 2018, 63, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Hardouin, P.; Pansini, V.; Cortet, B. Bone marrow fat. Jt. Bone Spine 2014, 81, 313–319. [Google Scholar] [CrossRef] [PubMed]
| Peptide | Sequence | Molecular Mass (Da) | tR (min) | |
|---|---|---|---|---|
| Calculated | Found | |||
| I | GKWMKLLKKILK-NH2 | 1483.98 | 1484.0 | 31.74 |
| II | GKWMKLLKKILK-NH2 | 1483.98 | 1484.3 | 31.74 |
| III | GKWKKLLKKILK-NH2 | 1481.03 | 1481.0 | 26.59 |
| IV | GKWMKMLKKILK-NH2 | 1501.94 | 1501.9 | 30.23 |
| V | GKWLKLLKKILK-NH2 | 1466.02 | 1466.7 | 32.84 |
| VI | GKWVKLLKKILK-NH2 | 1452.01 | 1452.0 | 30.34 |
| VII | GKWVKLLKKILK-NH2 | 1452.01 | 1452.0 | 30.34 |
| VIII | GKWMKLLKKILK-NH2 | 1483.98 | 1484.0 | 27.59 |
| IX | KWMKLLKKILK-NH2 | 1426.96 | 1427.0 | 27.82 |
| X | KWMKLLKKILK-NH2 | 1426.96 | 1427.0 | 27.82 |
| XI | βAKWMKLLKKILK-NH2 | 1497.99 | 1498.5 | 31.63 |
| XII | βAKWMKLLKKILK-NH2 | 1497.99 | 1498.2 | 31.63 |
| Antimicrobial Activity MIC (μmol/L) | Haemolytic Activity LC50 (μmol/L) c | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Peptide | MRSA 6271 | MRSA a | MRSA b | S. e. b | E. f. b | P. a. 5482 | P. a. a | C. a. | |
| I | 6.3 | 3.6 | 5.7 | 2.3 | 13.3 | 2.8 | 10.0 | 5.7 | 339 |
| II | 11.3 | 5.3 | 9.7 | 2.0 | 12.7 | 4.5 | 12.5 | 5.7 | >400 |
| III | 29.7 | 11.5 | 25.3 | 4.7 | >100 | 2.9 | 9.5 | 9.5 | >400 |
| IV | 20.0 | 10.0 | 20.0 | 3.2 | >100 | 5.3 | 12.7 | 12.7 | >200 |
| V | 10.0 | 8.0 | 10.0 | 2.2 | 50.7 | 4.0 | 10.7 | 16.0 | 196 |
| VI | 9.0 | 5.0 | 10.0 | 2.5 | >100 | 3.0 | 12.7 | 6.3 | >400 |
| VII | 22.0 | 15.0 | 22.0 | 4.2 | 25.3 | 4.5 | 25.3 | 12.5 | >400 |
| VIII | 18.7 | 13.3 | 32.0 | 4.0 | >100 | 5.0 | 32.0 | 8.0 | >400 |
| IX | 13.5 | 8.0 | 17.7 | 3.2 | >100 | 2.9 | 5.8 | 9.5 | >400 |
| X | 19.3 | 14.0 | 14.0 | 3.5 | 36 | 4.5 | 18.0 | 11.3 | >400 |
| XI | 11.3 | 5.7 | 10.0 | 2.8 | 59.0 | 2.8 | 7.0 | 11.3 | 270 |
| XII | 16.0 | 8.0 | 16.0 | 2.5 | 32.0 | 3.2 | 8.0 | 5.3 | >400 |
| Vanc | 0.9 | 1.1 | 1.1 | 1.5 | 1.3 | >200 | >320 | - | - |
| Gent | 325 | 3.9 | 4.5 | 109 | 95.2 | 2.0 | >220 | - | - |
| Bacteria | Colony Forming Units (CFU)/Hole | |||
|---|---|---|---|---|
| ChronOS Alone | ChronOS with Peptide No | ChronOS with Antibiotic | ||
| MRSA 6271 | 7.0 × 107 | (I) 7.6 × 102 | N/A | |
| MRSA 6271 | 4.0 × 105 (3 × 107) | (I + II) 2.0 × 103 | Vancomycin | 2.0 × 105 |
| MRSA 6271 | 2.1 × 106 | (XII) 0 | Vancomycin | 1.3 × 106 |
| MRSA a | 1.2 × 106 | (I + II) 0 | N/A | |
| MRSA a | 1.6 × 106 | (I + II) 4.0 × 102 | N/A | |
| MRSA a | 2.8 × 106 | (I + II) 0 | N/A | |
| MRSA a | 1.2 × 107 (6 × 108) | (I + II) 4.8 × 103 | Vancomycin | 6.0 × 106 |
| MRSA a | 2.0 × 106 | (IV) 2.5 × 103 | Ceftriaxone | 4.1 × 106 |
| MRSA a | 3.4 × 107 | (I) 2.3 × 105/(IX) 2.3 × 105 | N/A | |
| MRSA a | 3.9 × 106 | (VI) 0/(IX) 1.3 × 104 | Gentamicin | 2.9 × 105 |
| MRSA a | 1.4 × 107 | (X) 1.8 × 104 | Vancomycin | 1.4 × 107 |
| MRSA b | 7.0 × 107 | (I + II) 5.4 × 102 | Gentamicin | 9.2 × 105 |
| MRSA b | 4.8 × 106 | (XI) 4.0 | N/A | |
| MRSA b | 5.0 × 105 | (XI + XII) 0 | N/A | |
| S. e. | 1.2 × 105 | (I) 0/(III) 0 | N/A | |
| S. e. | 7.2 × 104 (2.4 × 107) | (V) 1.2 × 102 | Gentamicin | 6.0 × 103 |
| S. e. | 9.5 × 104 | (VI + VII) 0 | N/A | |
| P. a. 5482 | 1.0 × 109 | (I + II) 4.0 × 104/(XI) 2.0 × 105 | N/A | |
| Microbe | CFU/Implant | |||
|---|---|---|---|---|
| Palacos®r without Peptide | Palacos®r with Peptide No | Palacos®r with Antibiotic or Antimycotic | ||
| MRSA 6271 | 6.0 × 104 | (VI) 0 | N/A | |
| MRSA 6271 | 2.0 × 106 | (VI) 8.6 × 101 | N/A | |
| MRSA 6271 | 6.9 × 104 | (IX) 0/(IX) 0 | Vancomycin | 6.9 × 104 |
| *MRSA 6271 | 3.3 × 105/1 × 106 | (VI) 1.4 × 102/(VI) 0 | N/A | |
| MRSA a | 7.0 × 106 | (IV) 0/(IV) 0 | N/A | |
| MRSA a | 8.8 × 103 | (IX) 0/(IX) 0 | N/A | |
| MRSA b | 7.5 × 106/3.5 × 106 | (VI) 2.5 × 102/(VI) 1.0 × 101 | N/A | |
| *MRSA b | 1.2 × 105/0.6 × 105 | (I) 0/(I) 0 | Vancomycin | 4.0 × 106 |
| S. e. | 6.0 × 104/3.2 × 103 | (VI) 4.0 × 102/(VI) 0 | N/A | |
| E. f. | 2.9 × 105 | (I) 0/(I) 0 | N/A | |
| *E. f. | 2.8 × 106 | (I) 0 | Vancomycin | 4.0 × 106 |
| P. a. 5482 | 1.6 × 106/4 × 106 | (I) 5 × 101/(I) 4 | N/A | |
| C. albicans c | 4.4 × 105 | (VIII) 6.0 × 103 | Fluconazole | 3.6 × 104 |
| C. albicans d | 6.0 × 103 | (I) 4.0/(I) 0/(IX) 3.4 × 102 | N/A | |
| C. albicans d | 5.4 × 104/1 × 105 | (VIII) 0/(VIII) 0 | Amphotericin B | 0 |
| * C. tropicalis | 3.7 × 105 | (I) 0/(I) 4.0 | Amphotericin B | 1.3 × 106 |
| C. glabrata | 1.3 × 106 | (VIII) 2.4 × 103 | Clotrimazole | 1.2 × 106 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melicherčík, P.; Nešuta, O.; Čeřovský, V. Antimicrobial Peptides for Topical Treatment of Osteomyelitis and Implant-Related Infections: Study in the Spongy Bone. Pharmaceuticals 2018, 11, 20. https://doi.org/10.3390/ph11010020
Melicherčík P, Nešuta O, Čeřovský V. Antimicrobial Peptides for Topical Treatment of Osteomyelitis and Implant-Related Infections: Study in the Spongy Bone. Pharmaceuticals. 2018; 11(1):20. https://doi.org/10.3390/ph11010020
Chicago/Turabian StyleMelicherčík, Pavel, Ondřej Nešuta, and Václav Čeřovský. 2018. "Antimicrobial Peptides for Topical Treatment of Osteomyelitis and Implant-Related Infections: Study in the Spongy Bone" Pharmaceuticals 11, no. 1: 20. https://doi.org/10.3390/ph11010020
APA StyleMelicherčík, P., Nešuta, O., & Čeřovský, V. (2018). Antimicrobial Peptides for Topical Treatment of Osteomyelitis and Implant-Related Infections: Study in the Spongy Bone. Pharmaceuticals, 11(1), 20. https://doi.org/10.3390/ph11010020





