Unsupervised Assessment of Balance and Falls Risk Using a Smartphone and Machine Learning
Abstract
:1. Introduction
2. Data
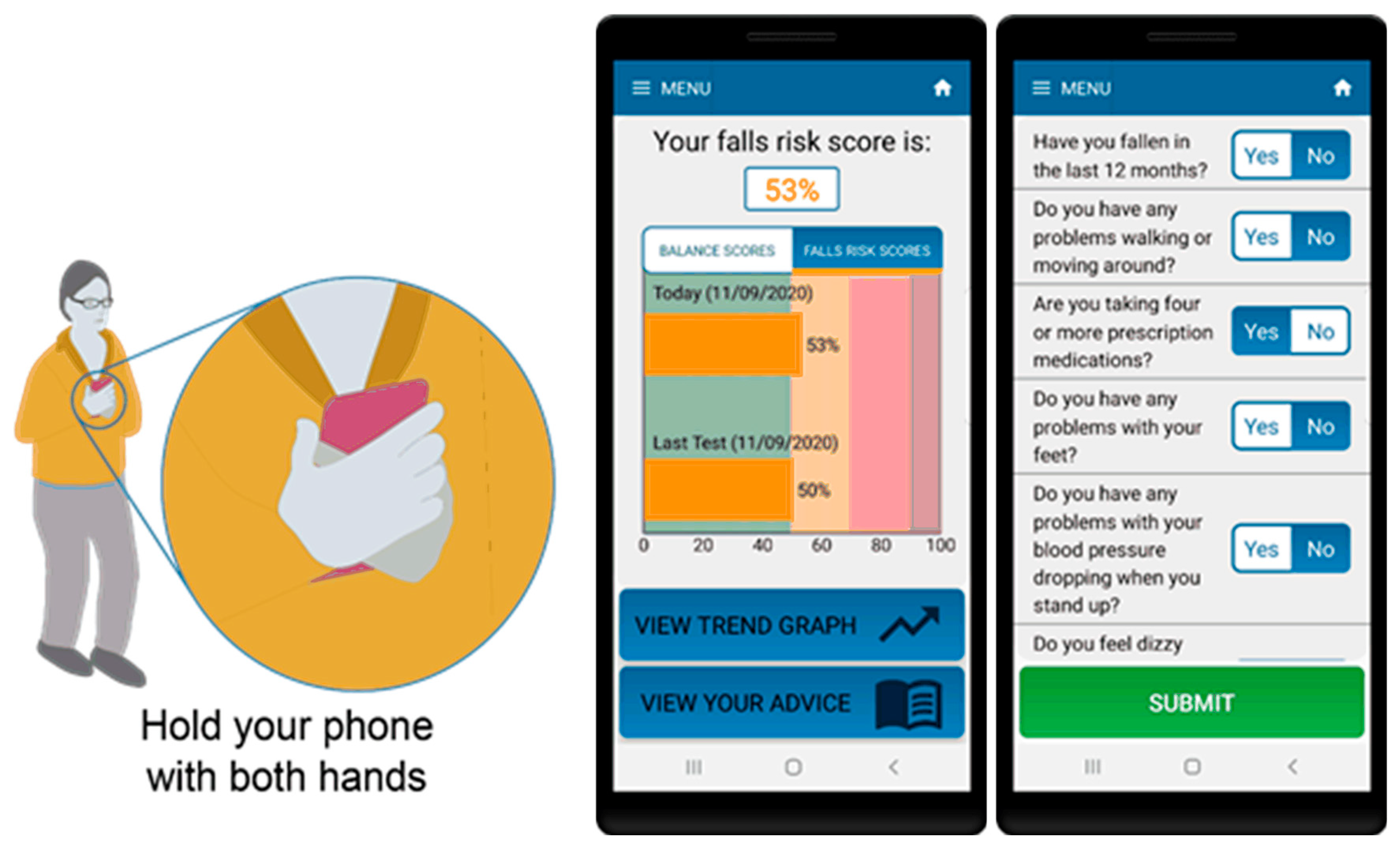
2.1. Smartphone App Data
2.2. Reference Data Set
3. Methods
3.1. Questionnaire
3.2. Standing Balance Test
3.3. Statistical Analysis
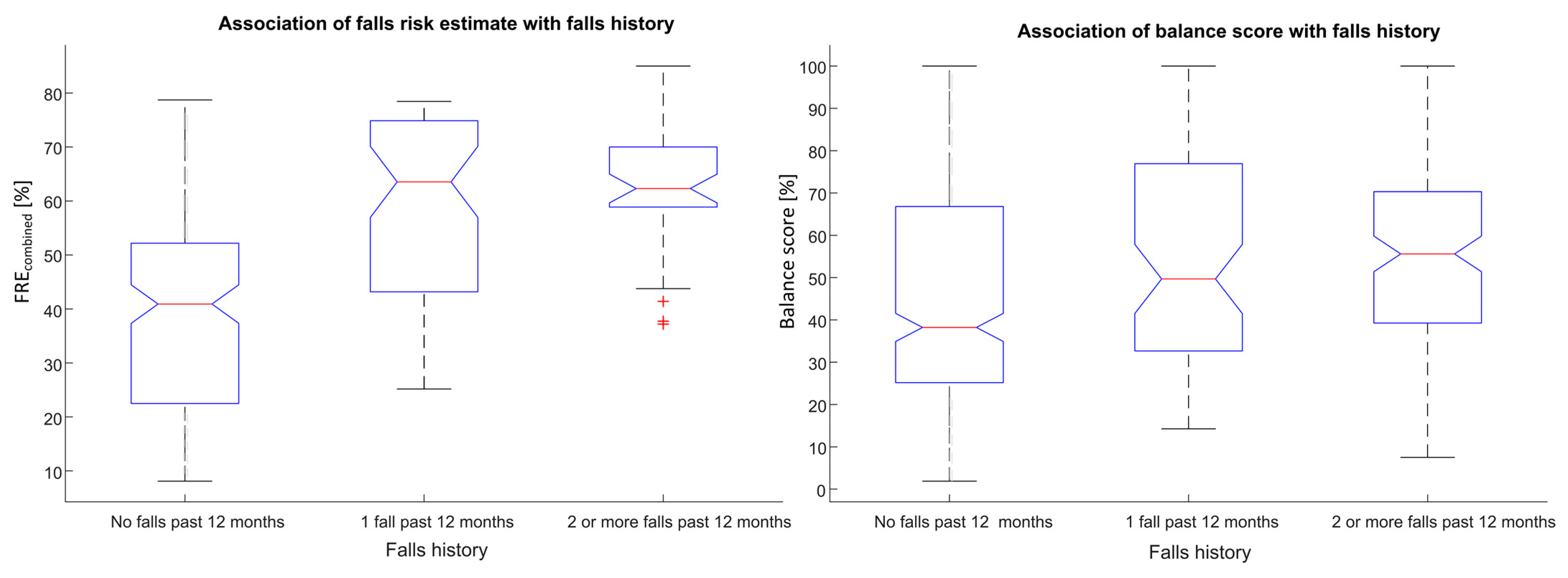
4. Results
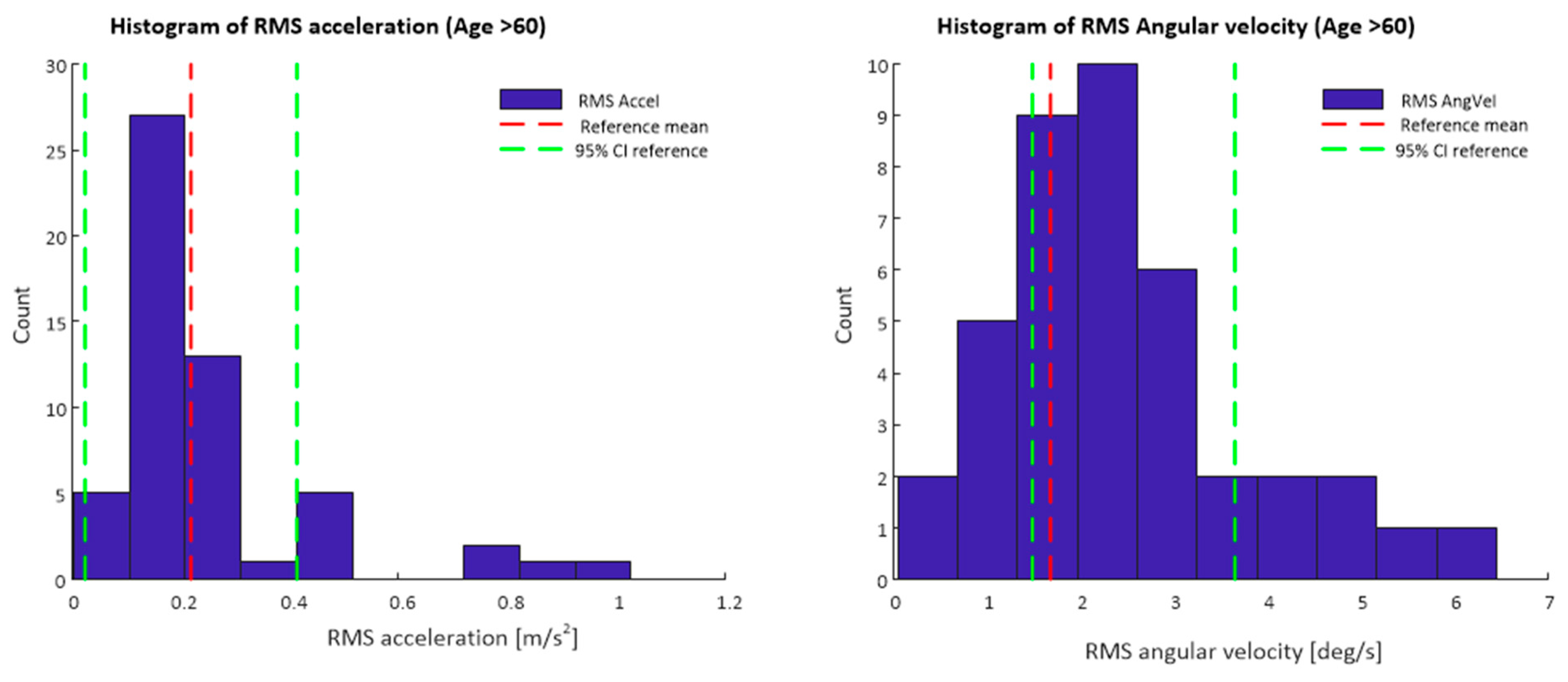
4.1. Inertial Sensor Features
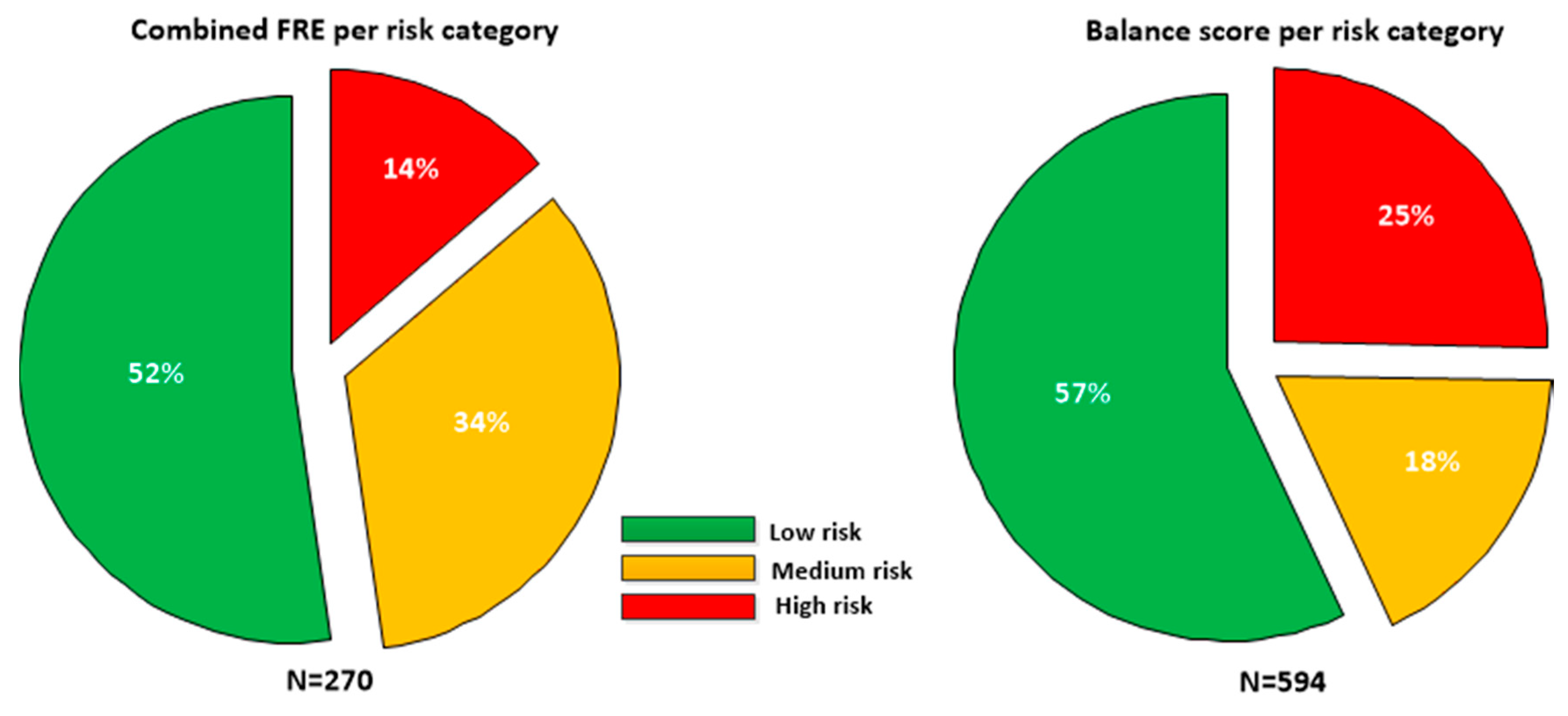
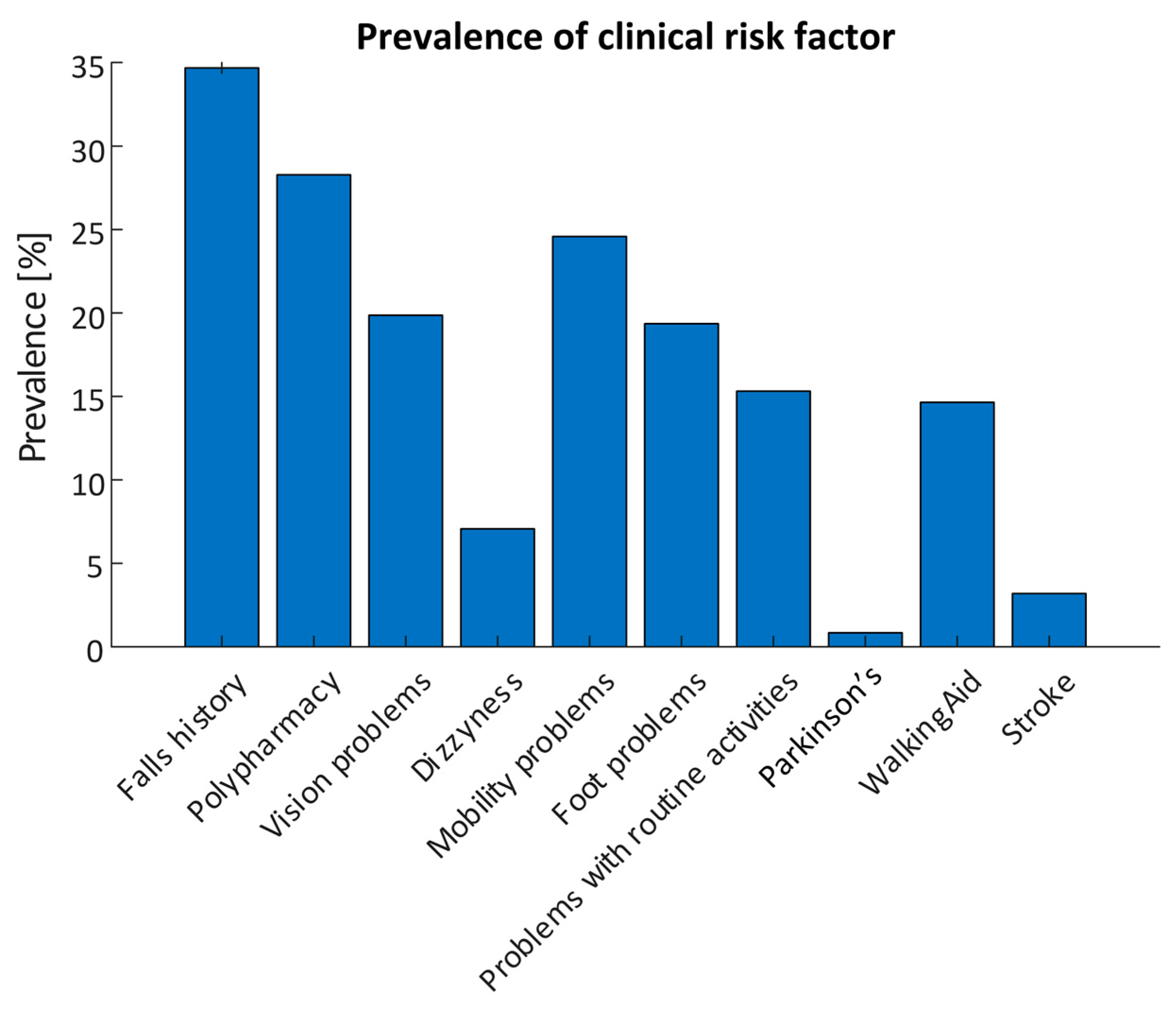
4.2. Questionnaire Results
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). WHO global report on falls prevention in older age. In WHO Department of Ageing and Life Course; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Burns, E.R.; Stevens, J.A.; Lee, R. The direct costs of fatal and non-fatal falls among older adults—United States. J. Saf. Res. 2016, 58, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Hartholt, K.A.; van Beeck, E.F.; Polinder, S.; van der Velde, N.; van Lieshout, E.M.M.; Panneman, M.J.M.; van der Cammen, T.J.M.; Patka, P. Societal Consequences of Falls in the Older Population: Injuries, Healthcare Costs, and Long-Term Reduced Quality of Life. J. Trauma Inj. Infect. Crit. Care 2011, 71, 748–753. [Google Scholar] [CrossRef] [Green Version]
- Sherrington, C.; Fairhall, N.J.; Wallbank, G.K.; Tiedemann, A.; Michaleff, Z.A.; Howard, K.; Clemson, L.; Hopewell, S.; Lamb, S.E. Exercise For Preventing Falls in Older People Living in the Community. Cochrane Database Syst. Rev. 2019, 1. [Google Scholar] [CrossRef]
- Gillespie, L.D.; Robertson, M.C.; Gillespie, W.J.; Sherrington, C.; Gates, S.; Clemson, L.M.; E Lamb, S. Interventions for preventing falls in older people living in the community. Cochrane Database Syst. Rev. 2012, 2021, CD007146. [Google Scholar] [CrossRef]
- Greene, B.R.; McManus, K.; Redmond, S.J.; Caulfield, B.; Quinn, C.C. Digital assessment of falls risk, frailty, and mobility impairment using wearable sensors. NPJ Digit. Med. 2019, 2, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Cheng, W.-Y.; Bourke, A.K.; Lipsmeier, F.; Bernasconi, C.; Belachew, S.; Gossens, C.; Graves, J.S.; Montalban, X.; Lindemann, M. U-turn speed is a valid and reliable smartphone-based measure of multiple sclerosis-related gait and balance impairment. Gait Posture 2021, 84, 120–126. [Google Scholar] [CrossRef]
- Boudreaux, E.D.; Waring, M.E.; Hayes, R.B.; Sadasivam, R.S.; Mullen, S.; Pagoto, S.L. Evaluating and selecting mobile health apps: Strategies for healthcare providers and healthcare organizations. Transl. Behav. Med. 2014, 4, 363–371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akbar, S.; Coiera, E.; Magrabi, F. Safety concerns with consumer-facing mobile health applications and their consequences: A scoping review. J. Am. Med. Inform. Assoc. 2020, 27, 330–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pew-Research Center. Survey of U.S. Adults Conducted Jan. 25-Feb. 8, 2021; Pew Research Center: Washington, DC, USA, 2021. [Google Scholar]
- Hughes, K.J.; Salmon, N.; Galvin, R.; Casey, B.; Clifford, A.M. Interventions to improve adherence to exercise therapy for falls prevention in community-dwelling older adults: Systematic review and meta-analysis. Age Ageing 2018, 48, 185–195. [Google Scholar] [CrossRef] [PubMed]
- McGarrigle, L.; Boulton, E.; Todd, C. Map the apps: A rapid review of digital approaches to support the engagement of older adults in strength and balance exercises. BMC Geriatr. 2020, 20, 1–11. [Google Scholar] [CrossRef]
- Palestra, G.; Mohamed Rebiai, M.; Courtial, E.; Koutsouris, D. Evaluation of a Rehabilitation System for the Elderly in a Day Care Center. Information 2019, 10, 3. [Google Scholar] [CrossRef] [Green Version]
- Uzor, S.; Baillie, L. Recov-R: Evaluation of a Home-Based Tailored Exergame System to Reduce Fall Risk in Seniors. ACM Trans. Comput. Hum. Interact. 2019, 26, 23. [Google Scholar] [CrossRef]
- Roeing, K.L.; Hsieh, K.L.; Sosnoff, J.J. A systematic review of balance and fall risk assessments with mobile phone technology. Arch. Gerontol. Geriatr. 2017, 73, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Rasche, P.; Nitsch, V.; Rentemeister, L.; Coburn, M.; Buecking, B.; Bliemel, C.; Bollheimer, L.C.; Pape, H.-C.; Knobe, M.; Rossi, A.; et al. The Aachen Falls Prevention Scale: Multi-Study Evaluation and Comparison. JMIR Aging 2019, 2, e12114. [Google Scholar] [CrossRef] [PubMed]
- Ghislieri, M.; Gastaldi, L.; Pastorelli, S.; Tadano, S.; Agostini, V. Wearable Inertial Sensors to Assess Standing Balance: A Systematic Review. Sensors 2019, 19, 4075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moral-Munoz, J.A.; Esteban-Moreno, B.; Herrera-Viedma, E.; Cobo, M.J.; Pérez, I.J. Smartphone Applications to Perform Body Balance Assessment: A Standardized Review. J. Med. Syst. 2018, 42, 119. [Google Scholar] [CrossRef] [PubMed]
- Shany, T.; Redmond, S.; Marschollek, M.; Lovell, N. Assessing fall risk using wearable sensors: A practical discussion. Zeitschrift für Gerontologie und Geriatrie 2012, 45, 694–706. [Google Scholar] [CrossRef]
- Doheny, E.P.; Greene, B.R.; Foran, T.; Cunningham, C.; Fan, C.W.; Kenny, R.A. Diurnal variations in the outcomes of instrumented gait and quiet standing balance assessments and their association with falls history. Physiol. Meas. 2012, 33, 361–373. [Google Scholar] [CrossRef]
- Hsieh, K.L.; Roach, K.L.; Wajda, D.A.; Sosnoff, J.J. Smartphone technology can measure postural stability and discriminate fall risk in older adults. Gait Posture 2019, 67, 160–165. [Google Scholar] [CrossRef]
- Fiems, C.L.; Miller, S.A.; Buchanan, N.; Knowles, E.; Larson, E.; Snow, R.; Moore, E.S. Does a Sway-Based Mobile Application Predict Future Falls in People With Parkinson Disease? Arch. Phys. Med. Rehabil. 2020, 101, 472–478. [Google Scholar] [CrossRef]
- McManus, K.; Greene, B.R.; Ader, L.G.M.; Caulfield, B. Single IMU assessment of postural sway and falls risk in older adults. In Proceeding of the IEEE Conference on Biomedical and Health Informatics (BHI) 2021 and the IEEE Conference on Body Sensor Networks (BSN) 2021 (BHI-BSN 2021), Athens, Greece, 21 September 2021. [Google Scholar]
- Greene, B.R.; McManus, K.; Caulfield, B. Automatic fusion of inertial sensors and clinical risk factors for accurate fall risk assessment during balance assessment. In Proceedings of the IEEE Biomedical Health Informatics Conference, Las Vegas, NV, USA, 4–7 March 2018. [Google Scholar]
- Greene, B.R.; Doheny, E.P.; Kenny, R.A.; Caulfield, B. Classification of frailty and falls history using a combination of sensor-based mobility assessments. Physiol. Meas. 2014, 35, 2053–2066. [Google Scholar] [CrossRef]
- Campbell, A.J.; Robertson, M.C.; Gardner, M.M.; Norton, R.N.; Tilyard, M.W.; Buchner, D.M. Randomised controlled trial of a general practice programme of home based exercise to prevent falls in elderly women. BMJ 1997, 315, 1065–1069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skelton, D.; Dinan, S.; Campbell, M.; Rutherford, O. Tailored group exercise (Falls Management Exercise—FaME) reduces falls in community-dwelling older frequent fallers (an RCT). Age Ageing 2005, 34, 636–639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Geriatrics Society; British Geriatrics Society; American Academy of Orthopaedic Surgeons Panel on Falls Prevention. Guideline for the prevention of falls in older persons. J. Am. Geriatr. Soc. 2001, 49, 664–672. [Google Scholar] [CrossRef]
- Ferraris, F.; Grimaldi, U.; Parvis, M. Procedure for effortless in-field calibration of three-axis rate gyros and accelerometers. Sens. Mater. 1995, 7, 311–330. [Google Scholar]
- Greene, B.R.; Redmond, S.J.; Caulfield, B. Fall Risk Assessment Through Automatic Combination of Clinical Fall Risk Factors and Body-Worn Sensor Data. IEEE J. Biomed. Health Inform. 2017, 21, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Viertio-Oja, H.; Maja, V.; Sarkela, M.; Talja, P.; Tenkanen, N.; Tolvanenlaakso, H.; Paloheimo, M.P.J.; Vakkuri, A.P.; Ylihankala, A.M.; Merilainen, P. Description of the Entropytm algorithm as applied in the Datex-Ohmeda S/5tm Entropy Module. Acta Anaesthesiol. Scand. 2004, 48, 154–161. [Google Scholar] [CrossRef]
- Rasche, P.W.V.; Mertens, A.W.; Bröhl, C.; Theis, S.; Seinsch, T.; Wille, M.; Pape, H.-C.; Knobe, M. The “Aachen fall prevention App”—A Smartphone application app for the self-assessment of elderly patients at risk for ground level falls. Patient Saf. Surg. 2017, 11, 1–4. [Google Scholar] [CrossRef]
- Hsieh, K.L.; Fanning, J.T.; A Rogers, W.; A Wood, T.; Sosnoff, J.J. A Fall Risk mHealth App for Older Adults: Development and Usability Study. JMIR Aging 2018, 1, e11569. [Google Scholar] [CrossRef] [Green Version]
- Papi, E.; Chiou, S.-Y.; McGregor, A.H. Feasibility and acceptability study on the use of a smartphone application to facilitate balance training in the ageing population. BMJ Open 2020, 10, e039054. [Google Scholar] [CrossRef]
- Hoffman, G.J.; Ha, J.; Alexander, N.B.; Langa, K.M.; Tinetti, M.; Min, L.C. Underreporting of Fall Injuries of Older Adults: Implications for Wellness Visit Fall Risk Screening. J. Am. Geriatr. Soc. 2018, 66, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Cattelani, L.; Cattelani, L.; Palumbo, P.; Palmerini, L.; Bandinelli, S.; Becker, C.; Chesani, F.; Chiari, L. FRAT-up, a Web-based Fall-Risk Assessment Tool for Elderly People Living in the Community. J. Med. Internet Res. 2015, 17, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thorbahn, L.D.B.; A Newton, R. Use of the Berg Balance Test to Predict Falls in Elderly Persons. Phys. Ther. 1996, 76, 576–583. [Google Scholar] [CrossRef]
- Barry, E.; Galvin, R.; Keogh, C.; Horgan, F.; Fahey, T. Is the Timed Up and Go test a useful predictor of risk of falls in community dwelling older adults: A systematic review and meta- analysis. BMC Geriatr. 2014, 14, 14. [Google Scholar] [CrossRef]
- Greene, B.R.; O’Donovan, A.; Romero-Ortuno, R.; Cogan, L.; Ni Scanaill, C.; Kenny, R.A. Quantitative Falls Risk Assessment Using the Timed Up and Go Test. IEEE Trans. Biomed. Eng. 2010, 57, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
| Inertial Sensor Feature | Smartphone | Lumbar |
|---|---|---|
| RMS acceleration (m/s2) * | 0.24 ± 0.18 | 0.22 ± 0.10 |
| RMS acceleration-X-axis | 0.10 ± 0.07 | 0.10 ± 0.05 |
| RMS acceleration-Y-axis (m/s2) * | 0.07 ± 0.06 | 0.09 ± 0.04 |
| RMS acceleration-Z-axis (m/s2) * | 0.17 ± 0.16 | 0.10 ± 0.04 |
| RMS angular velocity (°/s) * | 2.20 ± 1.20 | 1.68 ± 1.01 |
| Median frequency acceleration (Hz) * | 2.23 ± 1.47 | 5.32 ± 1.72 |
| RMS angular velocity-X-axis (°/s) | 1.29 ± 0.75 | 0.91 ± 0.56 |
| RMS angular velocity-Y-axis (°/s)* | 1.17 ± 0.79 | 0.72 ± 0.43 |
| Spectral edge frequency acceleration (Hz) * | 2.79 ± 0.95 | 4.14 ± 0.46 |
| Spectral entropy acceleration * | 0.47 ± 0.13 | 0.76 ± 0.07 |
| Median frequency angular velocity (Hz) * | 2.23 ± 1.40 | 2.87 ± 1.17 |
| Spectral edge frequency angular velocity (Hz) * | 3.45 ± 0.87 | 3.64 ± 0.50 |
| Spectral entropy angular velocity * | 0.5 ± 0.12 | 0.75 ± 0.06 |
| Age (years) * | 52.57 ± 16.89 | 74.45 ± 6.70 |
| Height (cm) * | 170.45 ± 10.81 | 164.35 ± 9.22 |
| Weight (kg) * | 82.56 ± 30.52 | 74.57 ± 13.69 |
| Class | FREcombined | Balance Score | Class | FREcombined | Balance Score | ||
|---|---|---|---|---|---|---|---|
| Acc (%) | 61.78 | 54.88 | Acc (%) | 69.87 | 59.93 | ||
| Sens (%) | Non-faller | 86.86 | 71.65 | Sens (%) | Non-faller | 86.86 | 62.89 |
| 1-fall | 25.00 | 13.89 | Faller | 37.86 | 54.37 | ||
| Recurrent faller | 8.96 | 28.36 | Pred (%) | Non-faller | 72.47 | 72.19 | |
| Pred (%) | Non-faller | 72.47 | 67.80 | Faller | 60.47 | 43.75 | |
| 1-fall | 19.57 | 29.41 | |||||
| Recurrent faller | 32.43 | 25.33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greene, B.R.; McManus, K.; Ader, L.G.M.; Caulfield, B. Unsupervised Assessment of Balance and Falls Risk Using a Smartphone and Machine Learning. Sensors 2021, 21, 4770. https://doi.org/10.3390/s21144770
Greene BR, McManus K, Ader LGM, Caulfield B. Unsupervised Assessment of Balance and Falls Risk Using a Smartphone and Machine Learning. Sensors. 2021; 21(14):4770. https://doi.org/10.3390/s21144770
Chicago/Turabian StyleGreene, Barry R., Killian McManus, Lilian Genaro Motti Ader, and Brian Caulfield. 2021. "Unsupervised Assessment of Balance and Falls Risk Using a Smartphone and Machine Learning" Sensors 21, no. 14: 4770. https://doi.org/10.3390/s21144770
APA StyleGreene, B. R., McManus, K., Ader, L. G. M., & Caulfield, B. (2021). Unsupervised Assessment of Balance and Falls Risk Using a Smartphone and Machine Learning. Sensors, 21(14), 4770. https://doi.org/10.3390/s21144770






