The current demand of technology for PD appraisal, intervention, and rehabilitation varied in its requirements and can be counted from cost, usability, working, efficiency, design, and continuous quantitative and qualitative information [
12]. In the beginning, clinical based scales were set to check motor symptoms severity which resulted in an uneven ratings and wrong measurements. The introduction of a smart environment such as body attached sensors, ubiquitous networking, and embedded sensors facilitates healthcare allied assistants to automatically monitor PD patients in real world environments. For instance, the researchers in [
23] monitored full body tremor, which is one of the dominant symptoms among PD patients, using an inertial measurement unit (IMU) based motion capture system and detecting tremor against non-tremor dominant individuals among a group of 40 PD as well as from 20 healthy controls. In [
24], the authors focused on another PD debilitating symptom that is freezing and discussed the variety of lightweight and wearable inertial sensors that may help in monitoring FOG (freezing of gate) in PD patients which uses dopaminergic medication. Similarly, in [
25], the authors proposed a method for finding gait freezing events amid normal walking using skin conductance (SC) features and multivariate Gaussians.
The research study in [
26] brings the key challenges in using wearable sensors i.e., data management, scalability, interoperability, standardization, security, and privacy and also proposed a smart glove in which flex sensors are attached to detect motor symptoms such as tremor, rigidity, and slowness of movement. The different endowed e-health wearables that assimilate contextual data are: DynaPort MiniMod Hybrid (worn on the lower back), Parkinson’s Kinetigraph (a wrist worn logger), a KinetiSense motion system (for dyskinesia measurements), ActivPAL, Stepwatch 3 (step activity monitor), Shimmer (records walking and turning), Mobi8Senior mobility monitor (SMM, Philips), SENSE-PARK system (for gait, hypokinesia, dyskinesia, sleeping), GAITrite (gait analysis systems), Opal (to asses quality of turning), Actigraphs (to monitor sleep), and also cueing devices such as auditory cueing devices, visual cueing devices, and somatosensory stimuli devices [
27,
28,
29,
30,
31,
32,
33]. All of these devices help in feature classifying of PDs determining the disease severity, motor impairment, and also the improvements after the exercises.
3.2.1. Wearable Device(s)
Most of the wearable devices are developed based on inertial sensors that are comprised of an accelerometer and gyroscope. Accelerometers are used to measure accelerations but are unable to measure the rotations or angles. Therefore, these can not help with detecting the turns during walking activities. On the other side, gyroscopes serve in detection of angular velocity of body and also there are less chances of mechanical noise than in an accelerometer’s case, hence turning is better assessed during motion. Being critical with gyroscopes, it can be mentioned that their drawback relates to the high power consumption during long-term recording. Battery life, type, and number of inertial sensors, sampling rate, recording and processing time, and, most importantly, the learning algorithm is the key factor that makes the difference between the accuracy and precision of wearable devices. In this section, we highlight the ones that assess motor symptoms of PD patients and offer insights in diagnosis, cueing, and testing.
In [
27], the authors proposed a device named Opal, with a weight of 22 g. It is built using inertial sensors, a battery, and includes 8 GB of memory storage. The device is tested in a research study in which users wore three Opals, one on the belt and the remaining two inside shoes. Data were recorded at 128 Hz and later uploaded to a laptop. The data from this device help in assessing the quality of turning. The researchers in [
29] introduced a Kinesia system that consists of a software application, a hybrid sensor worn on the finger, and an automated web-based symptoms assessment system. The patients wore the sensors on the index finger of the most affected hand. The assessment is based on five motor tasks each of 15 s to predict finger tapping, dyskinesia, hand opening and closing, and also the postural tremor. The Parkinson’s Kinetigraph from [
30,
32] is used to measure the wrist movements and is worn like a wrist bracelet. It weighs around 35 g, and it has a three-axis iMEMS accelerometer (ADXL345 analog device) that records acceleration with a value of
g at sampling frequency of 50 samples/s. The device is developed using a digital microcontroller with flash memory along with a rechargeable battery. The sensor apprehends the Bradykinesia and dyskinesia values in a two-minute span for 10 days using a fuzzy logic algorithm. The device is preferred to be worn at the most affected limb of PD patients. The GAITRite in [
30] is a walkway with a length of 4.6 m connected with Windows XP through the serial port. The thickness of passageway is 1/800 and has 16,128 sensors attached between two layers of vinyl and a rubber. It helps with demonstrating Bradykinesia and can be used in the replacement of conducting a traditional timed test such as a TUG test or filling questionnaires from PD patients [
28].
Actigraphs are movement detectors, which are constructed with accelerometers and a memory for recording the movements for few weeks. The programs are developed to determine the levels of rest/movement, rhythmic parameters, and running/sitting parameters. In [
27], Actigraphs, in the form of wrist worn activity sensor, are used for sleep monitoring. The KinetiSense motion system in [
30] served for accurate measurements of tremors, Bradykinesia, and dyskinesia. It is built using accelerometers and gyroscopes that are attached on three areas of the body. This system is considered to be beneficial in developing new therapies. Stepwatch 3 is called an ankle acceleromater, fixed on the leg for counting stride rate. It is one of the devices with most valid and reliable results in monitoring ambulatory activity as discussed in [
27,
30]. ActivPAL™ in [
31] is a small, lightweight activity monitor device that has a uni-axial accelerometer fixed on the upper thigh, at 10 Hz sampling frequency. The raw data in form of spreadsheet are exported for further analysis in MATLAB. The SENSE-PARK System in [
31] has a set of inertial sensors (three are used during daytime and one in night phase) that helps in detecting movements of PD subjects i.e., FOG, dyskinesia, tremor, and sleep using an algorithm. The system also has a Wii balance board for collecting information such as body weight and sway.
In the study [
31], SHIMMER sensors are introduced. These are kinematic sensors developed with gyroscope and an accelerometer that performs sampling at 102.4 Hz. These are attached in the form of elastic bands. The recorded data are transferred to computer wireless using Bluetooth. A total of 21 features are selected for reliability analysis from the recorded data. Another ambulatory assessing device is Mobi8 proposed in [
31], which is a multichannel data logger with a dimension of 11.4 × 9.8 × 3.7 cm
, weighs up to 165 g, has a 3D sensor (Analog Devices ADXL330), and is worn on the lower back. It records anterior-posterior, vertical, and mediolateral, respectively. For finding daily life activities such as walking, the Senior Mobility Monitor (SMM) [
31] was implemented. It is comprised of an accelerometer and barometer. The data are sampled at 50 Hz for the accelerometer and 25 Hz for the barometer. SMM is required to be worn at sternum height. The data are analyzed using a wavelet-based decision tree algorithm in MATLAB
®, version 2013a. DynaPort MiniMod Hybrid in [
32] weighs 74 g and has dimensions of 87 × 45 × 14 mm. It is attached on a belt on the back to show lower body movements in performing DLA. The device consists of accelerometer with a limitation of
g, a resolution of
mg, and a triaxial gyroscope. The readings are stored on an SD card at a frequency of 100 Hz and transferred in MATLAB Software for further analysis of gait features. The system aids in monitoring and classifying the quality and quantity of gait in PD faller and non-faller groups.
The study [
33] provides a technological review on available wearable cueing devices, highlighting the current auditory, visual, and somatosensory cueing devices. The auditory cueing devices include Android applications based on Google Glass, GaitAssist (equipped with two inertial sensors and a smartphone with android application and wired headphones), FoG detection devices with wireless ear sets, Metronome Peterson bodyBeat and Metronome SDM300 SAMICK (Peterson Electro-Musical Products, Inc., Alsip, IL, USA), and devices with a movement sensor enabled with Bluetooth and wired headphones. These devices produce a typical and distinctive sounding tones (i.e., tap, tick, click, and beat) in beats/minute. In this way, it generates temporal information such as step interval, through the rhythmical beat. Some visual cueing devices are: Laser shoes, Smart Gait-Aid (Android app on binocular smart glasses), and Visual-auditory walker. These devices demonstrated that visual stimuli can diminish the FOG occurrence during walking. Parallel patterns aid in conveying spatial parameters’ information, such as step duration. Some somatography cueing devices are: CueStim (two channel electrical stimulator), Vibrating waistband, and a Vibrating system named VibroGait.
3.2.2. Insoles Models and Technical Features
According to primary research studies, there are two ways to evaluate motion activities of PD patients: subjective and objective. The subjective methods are based on questionnaires, UPDRS criteria, or Hoehn and Yahr scales in which there are more chances of getting an incorrect evaluation and error in scaling. The objective assessment is based on a huge variety of body worn sensors such as accelerometers, gyroscopes, magnetometers, force sensors, etc. that detect each fine movement and angles of a person performing (daily life activities) DLA, but the criterion validity of these wearable e-health devices vary from one to another. Not all the aforementioned devices in [
27,
28,
29,
30,
31,
32,
33] are appropriate for daily routines in people with Parkinson’s disorder, and it is hard to find a single wearable device for diagnosis, monitoring, and rehabilitation of PD. A systematic review in [
34] provided a potential solution for continuous and unobstructed appraisal of Parkinson’s patients that resides in smart insoles.
There is no doubt that humans wear shoes continuously, and the insoles are much cheaper than other wearable devices commercially available in the market.
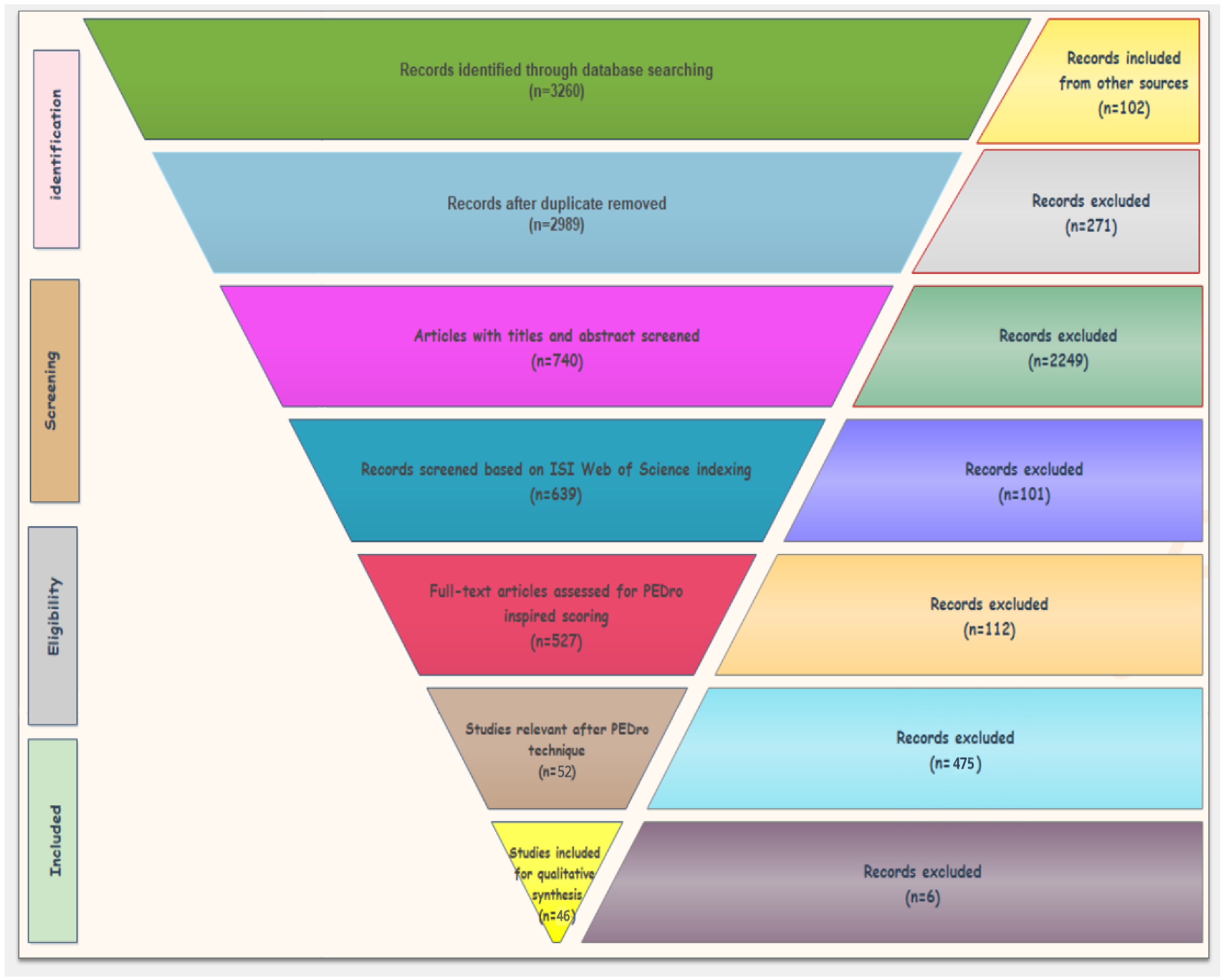
Table 7 presents an overview of the insole models discussed in papers [
35,
36,
37,
38,
39]. These insoles can extract gait features and help in classification of PD stages and in daily monitoring for rehabilitation purposes. The results of the comparative study [
34] emphasize that the data validity of smart insoles is 75% to 100% accurate, 75% to 100% precise, and the specificity lies between 73% to 100%. In this way, we can say that smart wearables [
40] allow quantitative, objective, and reliable evaluation of motor activities.
3.2.3. Algorithms for Analysis of Gait Pattern
However, besides the acquisition of a robust data set from wearable insoles, another noteworthy aspect regards the algorithms that accurately learn from the data and also accurately predict [
41]. Machine learning algorithms are currently considered as the pervasive part of the smart environment, but PD data are different in a more sophisticated way from the traditional clinical data as it is comprised of high-frequency continuous digital sensors readings of around tens of thousands per second.
ML algorithms such as random forest (RF), decision trees (DTs), logistic regression (LR), support vector machine (SVM), hidden Markov models (HMMs), naive Bayes, clustering algorithms, and neural networks (NNs) have been implemented successfully in medicine [
32] and recently expanded to use them for sensor based PD motor assessment. A systematic study [
42] revealed that ML algorithms depend on the data and on the features that are needed to be extracted as it is discussed in [
43], where a TRIS (treatment response objective index) algorithm is discussed that examines clinical effectiveness with regard to dose response.
The outcome of studying these insoles models proved that smart insoles can identify either the subjects are sitting, standing, walking, or lying and can also differentiate between normal gait and Parkinsonism gait features. Last but not least, these can be useful to find the improvements in movement after exercises.