Variability of Muscle Synergies in Hand Grasps: Analysis of Intra- and Inter-Session Data
Abstract
1. Introduction
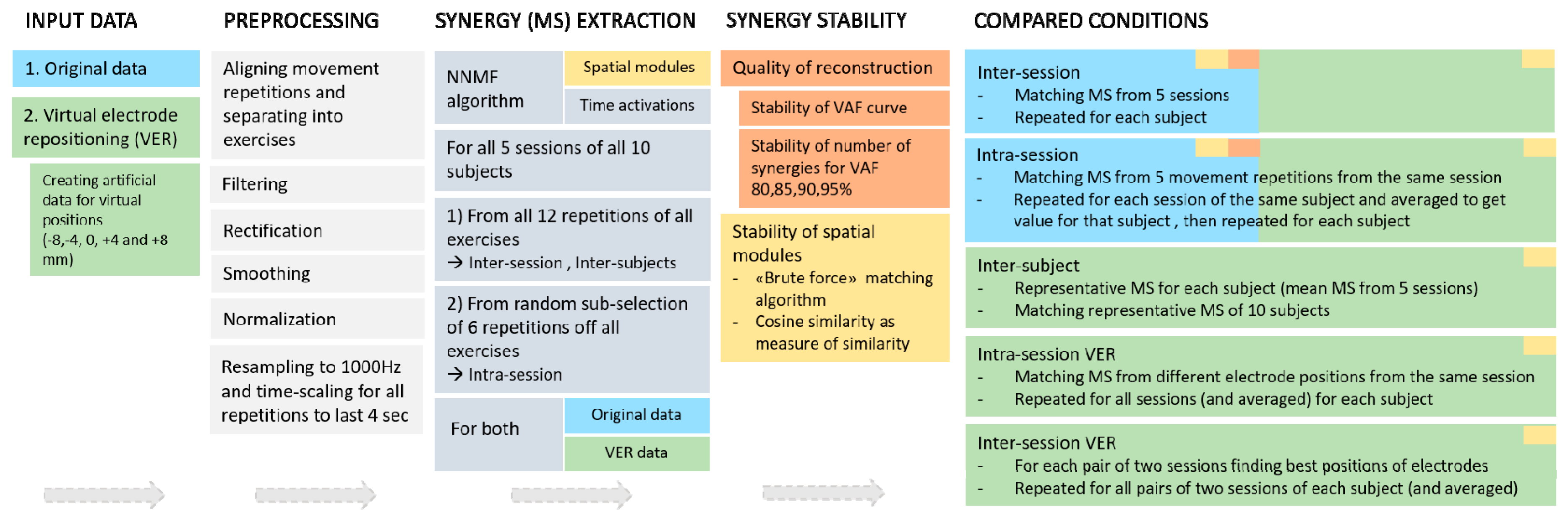
2. Materials and Methods
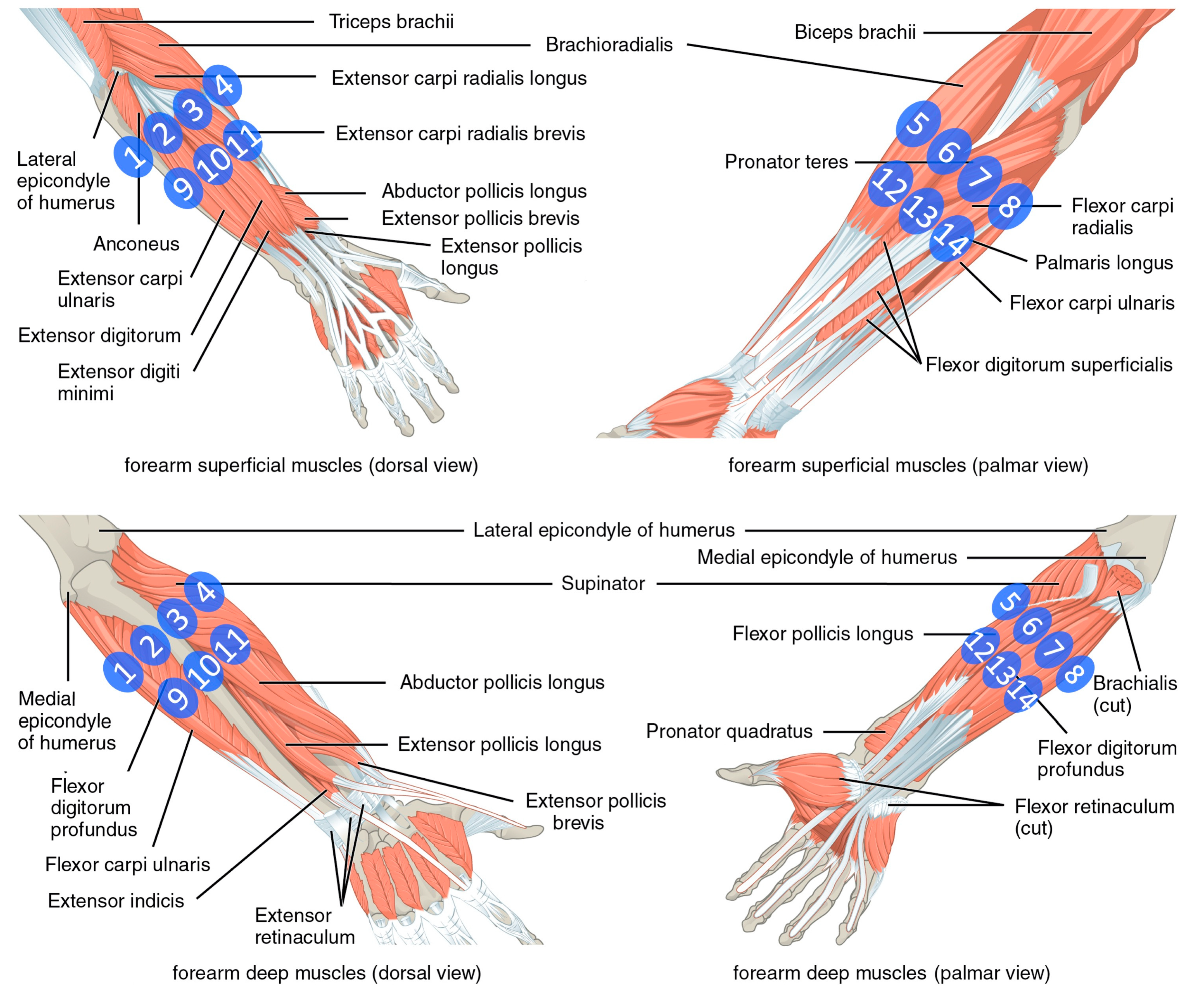
2.1. Data
2.2. Data Analysis: EMG Processing and Synergy Extraction and Matching
2.2.1. Preprocessing
2.2.2. Synergy Extraction
2.2.3. Synergy Matching
2.3. Data Analysis: Considered Cases
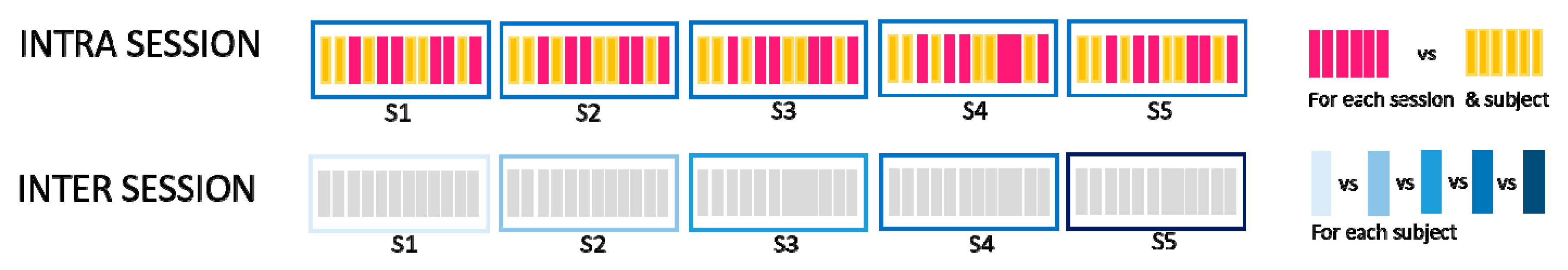
2.3.1. Intra-Session and Inter-Session
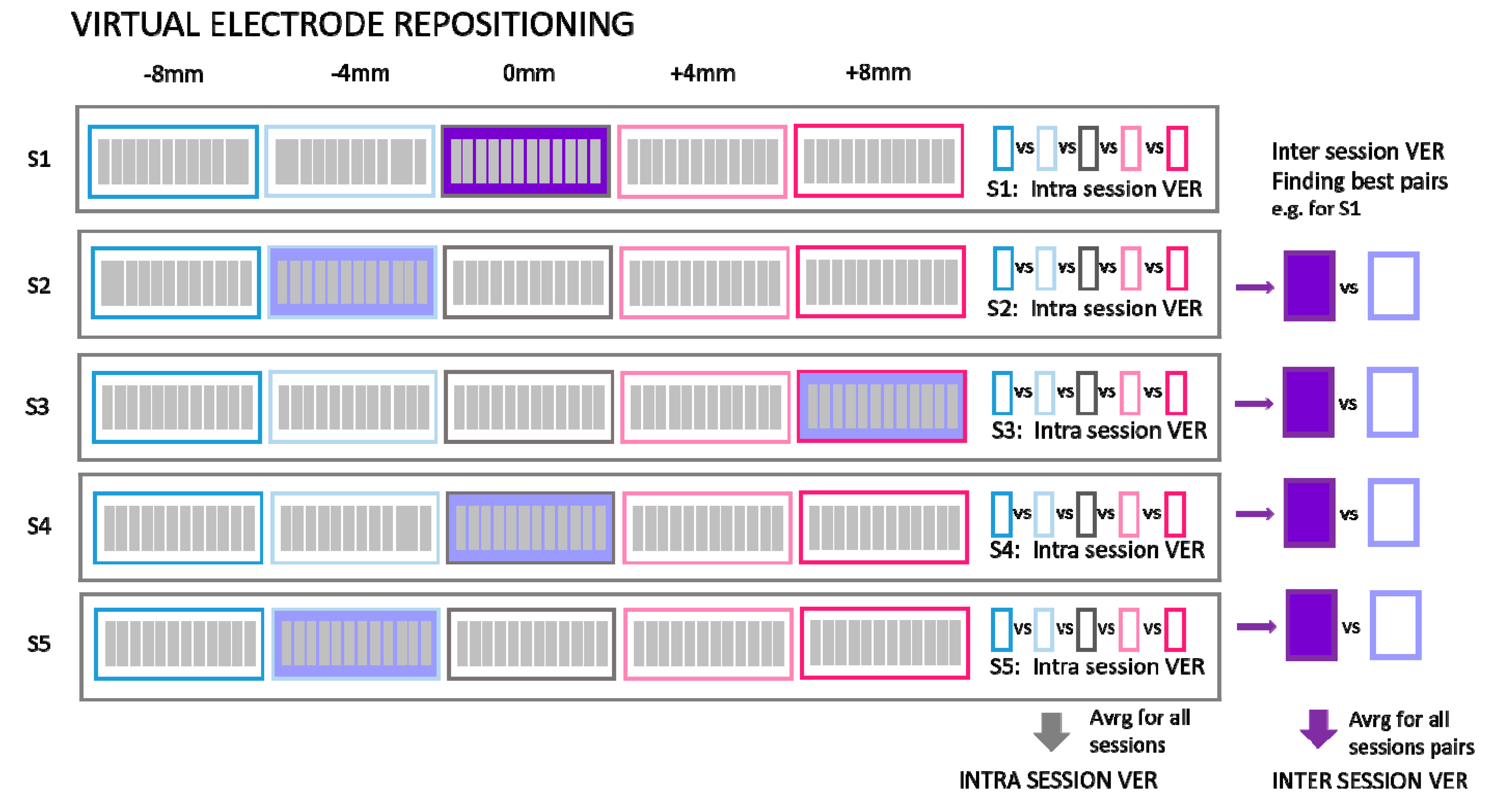
2.3.2. Comparison of Synergies from Virtual Electrode Repositioning
2.4. Data Analysis: Outcome Measures
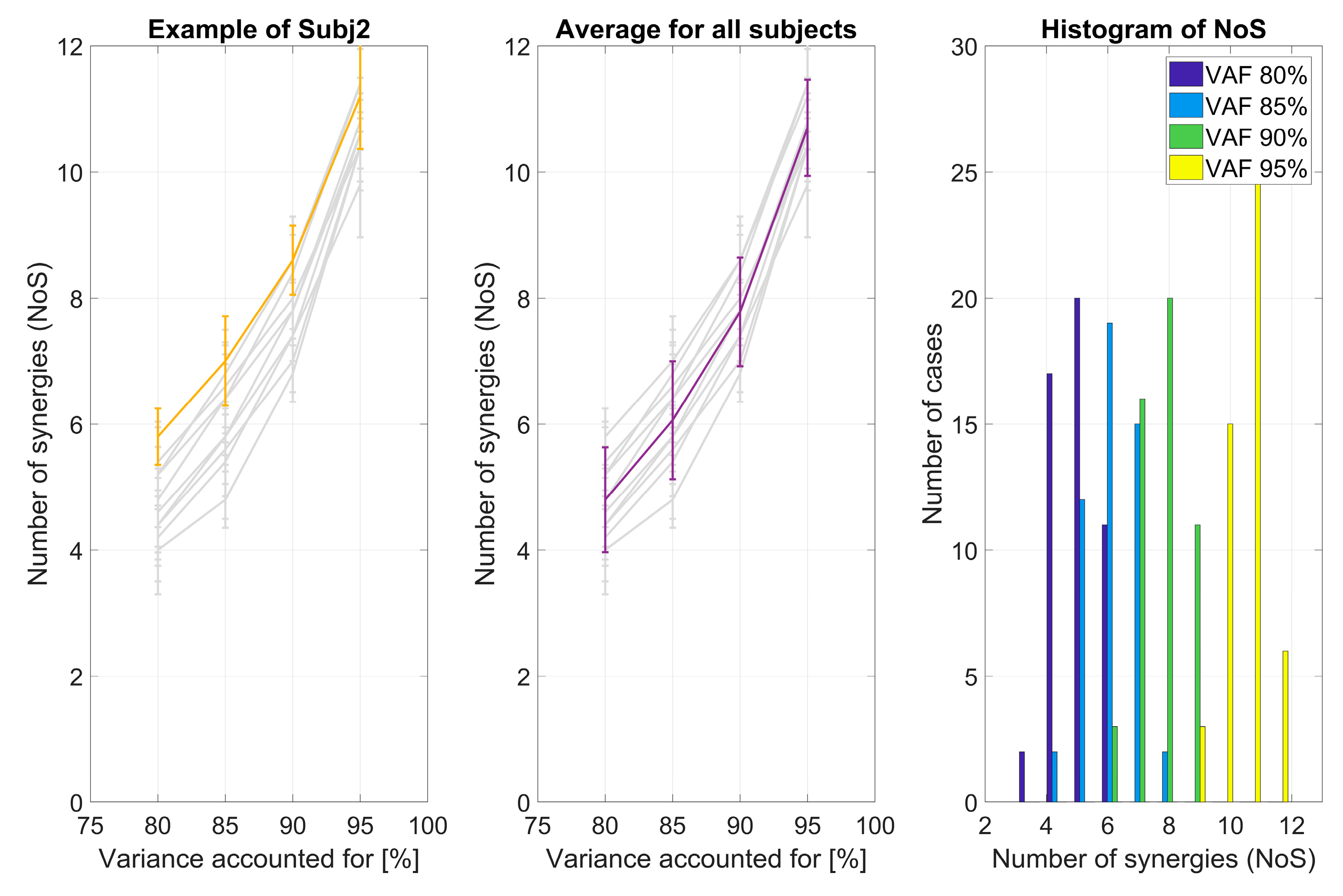
2.4.1. VAF and Number of Extracted Synergies
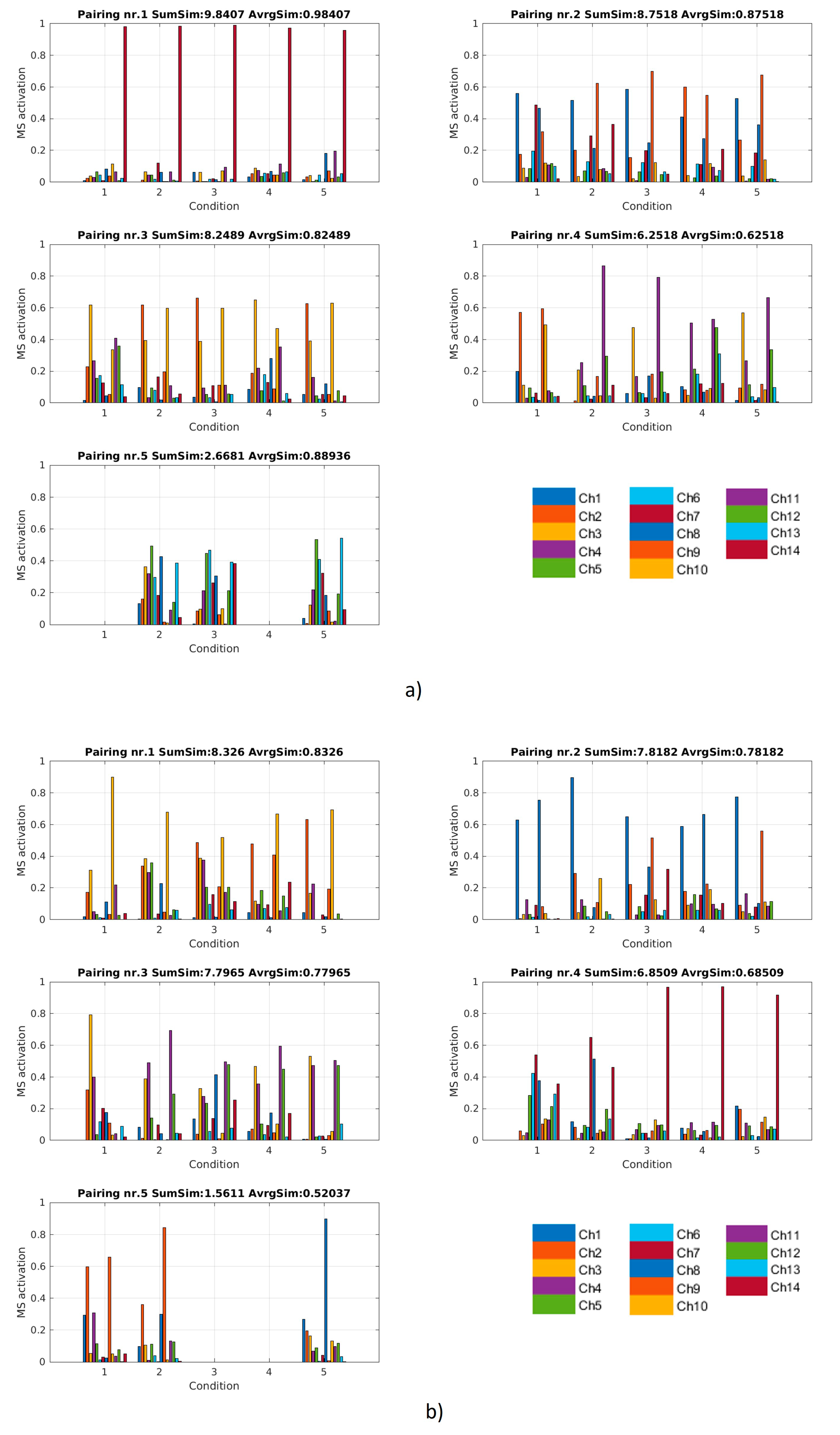
2.4.2. Similarity of Synergy Spatial Composition
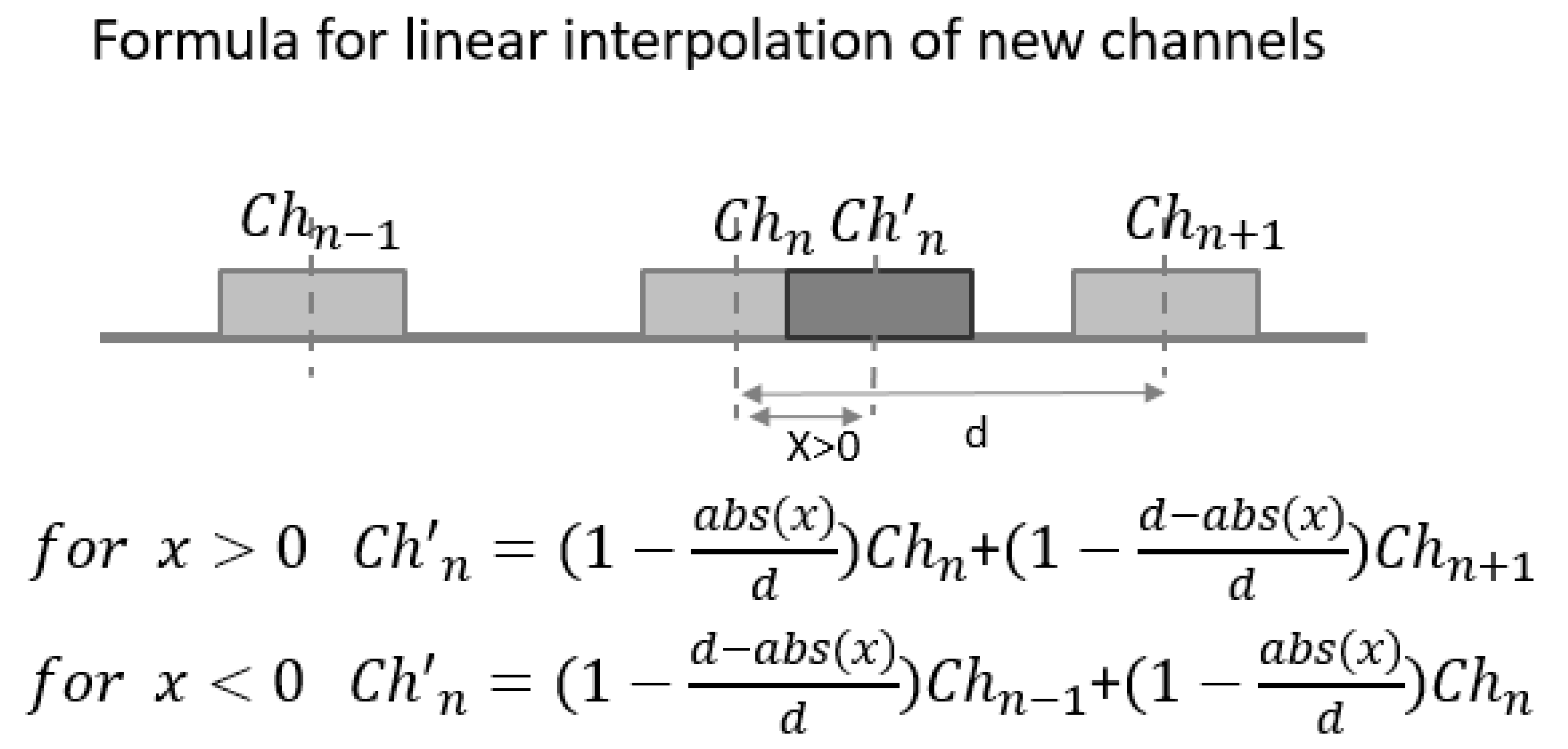
2.4.3. Virtual Electrode Repositioning
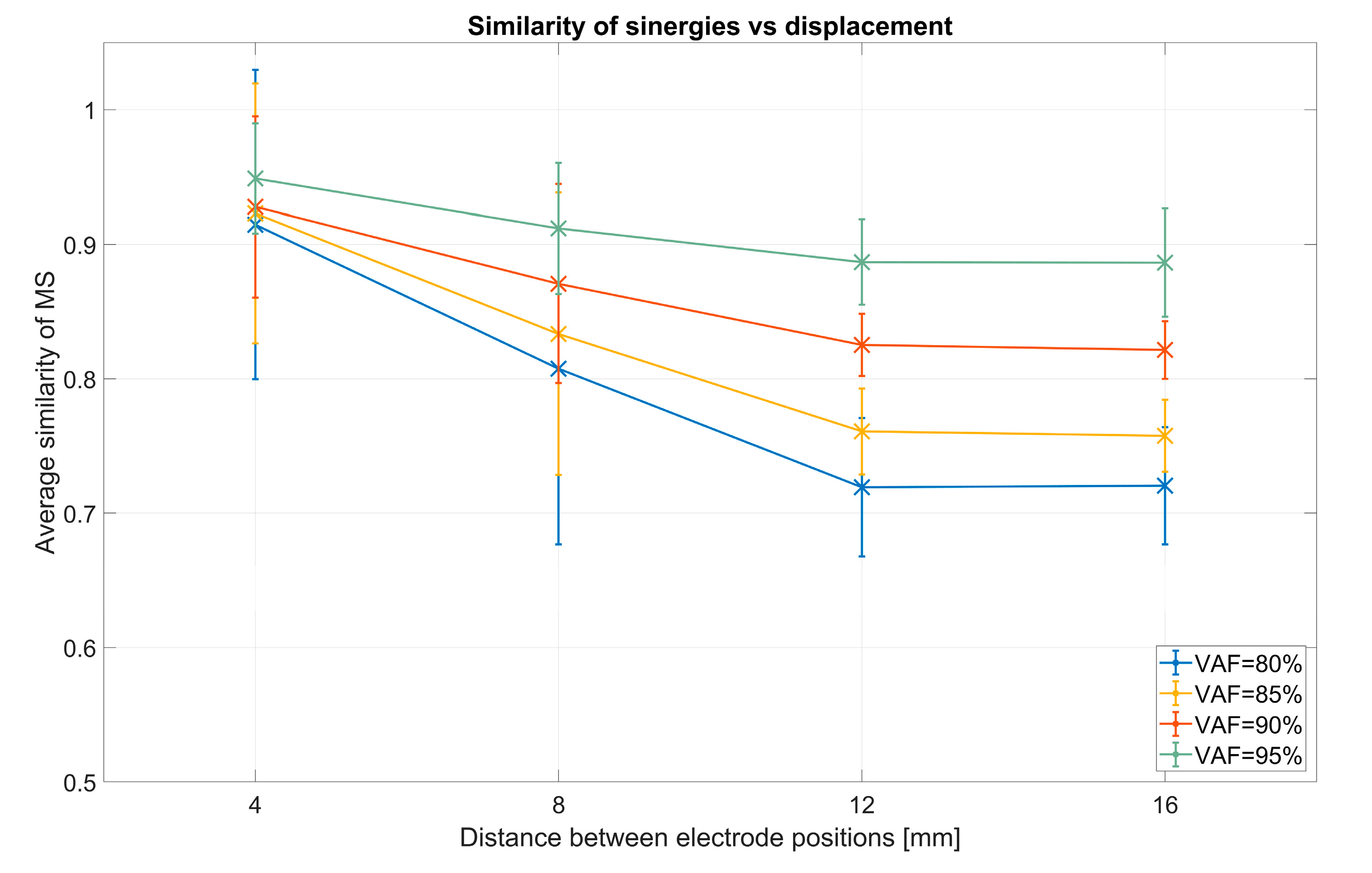
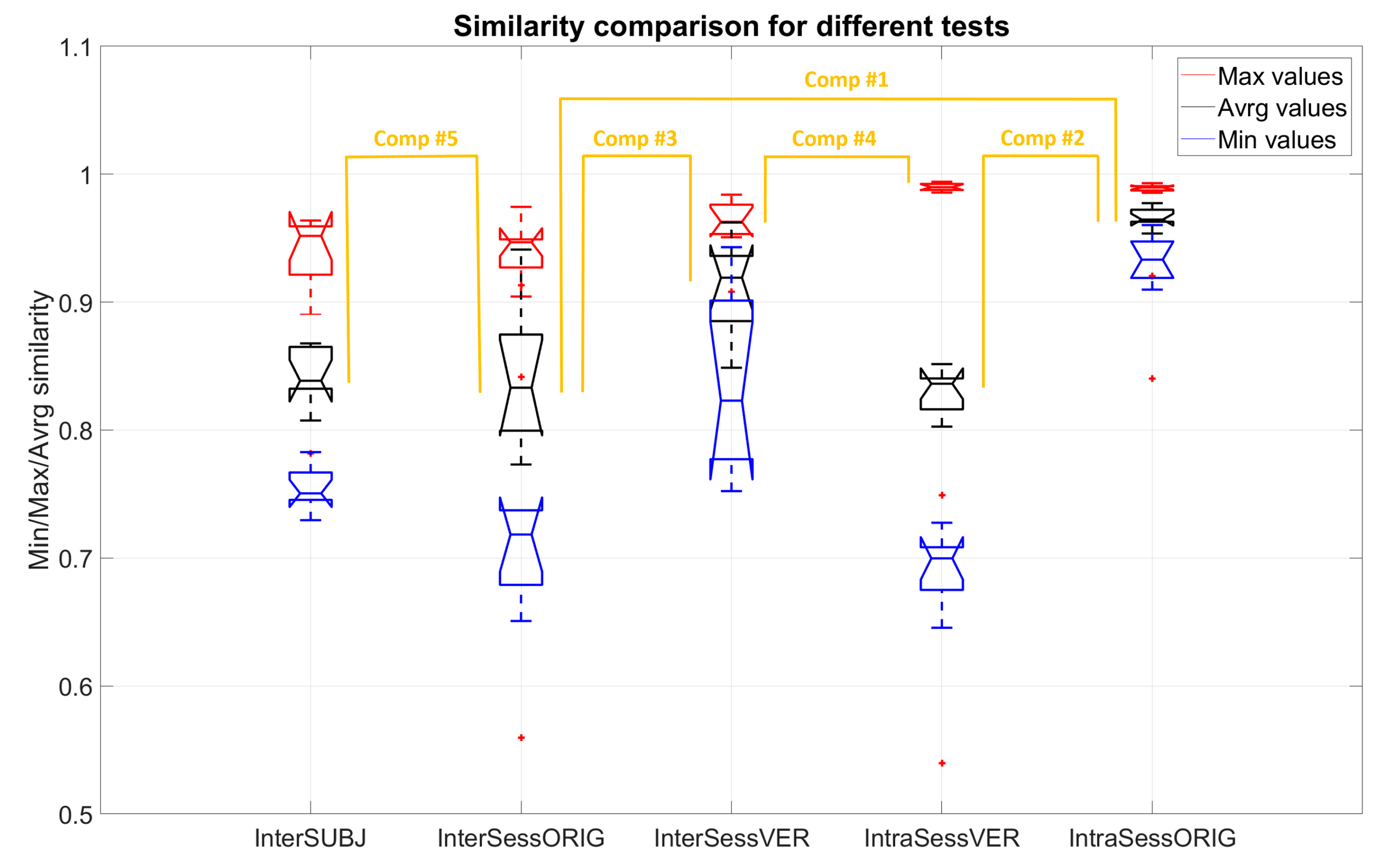
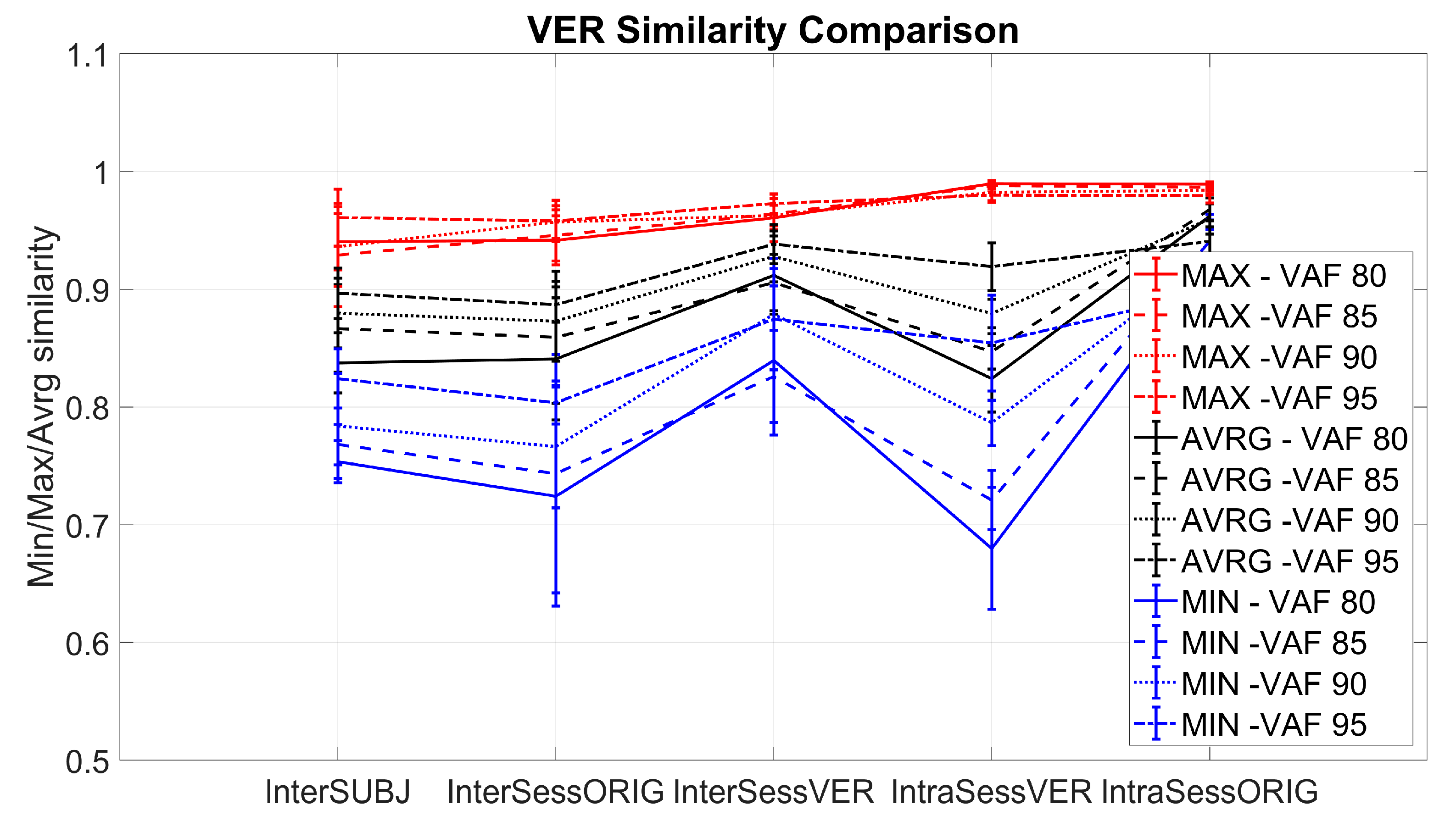
- Intra-session virtual electrode repositioning (IntraSessVER)The first step for our analysis was to investigate if a different synergy similarity is detected when slightly repositioning electrodes within the same session. This finding can confirm that electrode repositioning might influence synergy similarity and serves as motivation for the following steps of the analyses.To achieve this, we created a simulated dataset to compare muscle synergy similarity from different virtual electrode positions for the same session. We compared average similarities for all possible repositioning distances (4 mm, 8 mm, 12 mm, and 16 mm). This was repeated for all VAF values. Maximal similarity of synergies (MAX) between any two steps of rotations for each session (and subjects), average similarities (AVRG) between all step pairs, and minimal similarities (MIN) were calculated. MAX values represented, for each subject, the average (computed across 5 sessions) of maximum similarities detected between different electrode positions within each session. To achieve AVRG value similarity, values of all step pairs in each of the sessions were averaged (for each subject). Finally, MIN values represented the average (computed across 5 sessions) of minimum similarities for each subject detected for any combination of electrode positions within same session. In this way, our IntraSessVER dataset was composed of 10 AVRG, 10 MIN, and 10 MAX matching values (one for each subject representing an average of 5 sessions) for each VAF value.
- Inter-session virtual electrode repositioning (InterSessVER)Here, we aimed to assess if repositioning electrodes virtually between sessions can lead to higher similarity between sessions. If this condition was met, a part of the variability in the muscle synergy similarity that existed between sessions can be due to slightly different positions of electrodes. For each possible combination of two sessions, we created a simulated dataset computing the maximum muscle synergy similarity by calculating all combinations of steps for those two sessions. Maximum, minimum, and average similarities between different step pairs (25 combinations) and session pairs (10 combinations) were calculated for all the subjects. MAX values represented the highest similarity that each subject achieved between any two sessions having the option to also shift electrodes between sessions. MIN values reported the minimum value that each subject had between any two sessions. AVRG values were the average of MAX values found for each session pair, calculated for each subject. In this way, our InterSessVER dataset was composed of 10 ARVG, 10 MIN, and 10 MAX matching values (one for each subject) for each VAF value.
- Intra-session without electrode repositioning (IntraSessORIG)The intra-session dataset was created by computing the similarity of the synergies extracted from a different selection of movement repetitions but from the same session (no electrode repositioning). This was the same analysis as already described in Section 2.4.2, but it was repeated using only 8 channels instead of 14 in order to be able to compare the results with that of intra-session using virtual electrode repositioning. Minimum, maximum, and average values of 5 sub-selections of movements from each session were calculated. Then, the average of minimum, maximum, and average values over 5 sessions for each subject were compared with values of the other conditions. MAX values represented for each subject the average (computed across 5 sessions) of maximum similarities detected between movement sub-selections, AVRG values represented the average of average similarities between sub-selections for all sessions, and the MIN average represented minimum similarities of its sub-selections. In this way, our IntraSessORIG dataset was composed of 10 AVRG, 10 MIN, and 10 MAX matching values (one for each subject representing average of 5 sessions) for each VAF value.
- Inter-session without electrode repositioning (InterSessORIG)The intersession dataset was created by computing the similarity of synergies between sessions. The dataset was analogous to the one described in Section 2.4.2 but for only 8 channels instead of 14 and with the original positions of electrodes (no electrode repositioning). MAX values represent the maximum similarity that each subject achieved between any two sessions (with original positions of electrodes), while MIN values report the minimum value that each subject had between any two sessions. AVRG values are the average of all possible session pairs (10 combinations) for each subject. In this way, our InterSessORIG dataset is composed of 10 AVRG, 10 MIN, and 10 MAX matching values (one for each subject) for each VAF value.
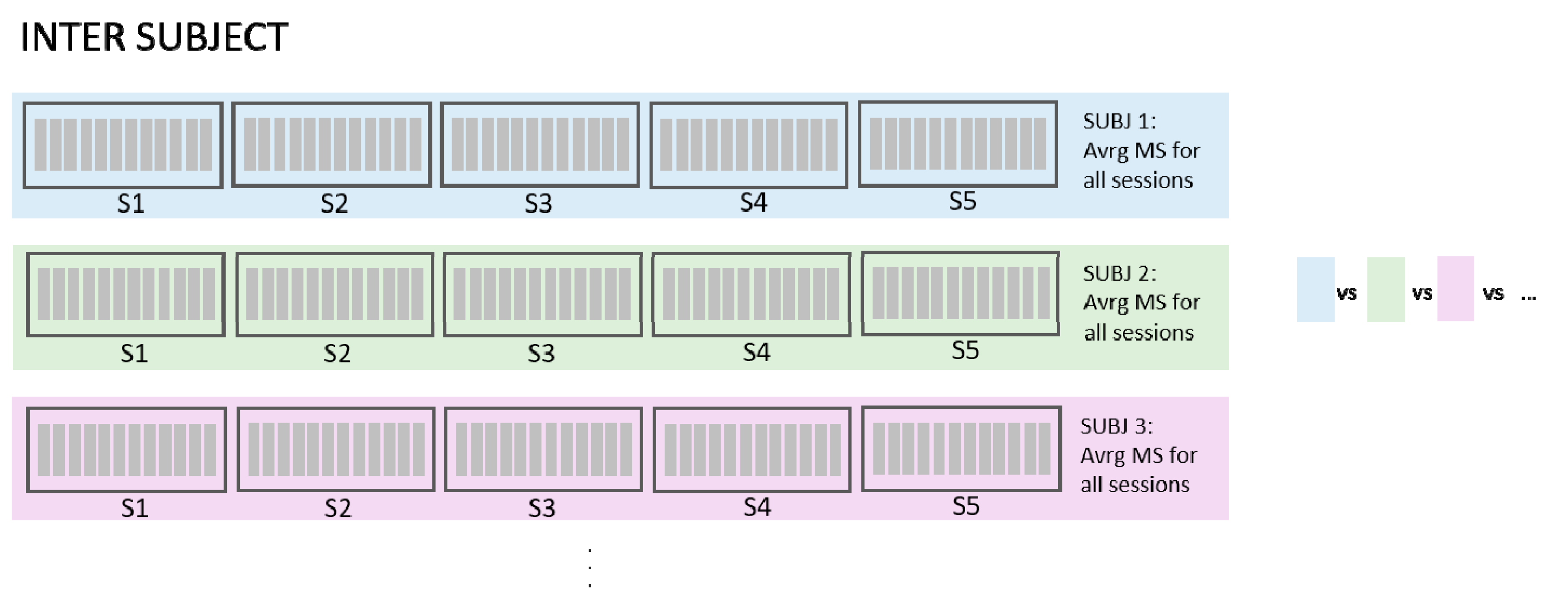
- Inter-subject (InterSUBJ)Finally, we considered a fifth condition that we labelled inter-subject (portrayed graphically in Figure 8). The assessment of similarity of spatial synergies between people can enhance the understanding of the generalization of the synergies. The average synergies computed across 5 sessions for each person were calculated (using groups from inter-session matching described in Section 2.4.2) and averaged in the view that they represent individual synergies better than if we chose synergies from one session randomly. These average synergies, representing individual synergies of 10 participants, were then compared in a pairwise manner (45 combinations). MAX values represent the maximum similarity of synergies that each subject had with one of the other subjects (9 combinations), AVRG values are average similarities that each subject had with all other subjects, and MIN values represent the minimum value of similarities each subject had with any of all other subjects. In this way, our InterSUBJ dataset is composed of 10 AVRG, 10 MIN, and 10 MAX matching values (one for each subject representing the average of comparison with 9 other people) for each VAF value.
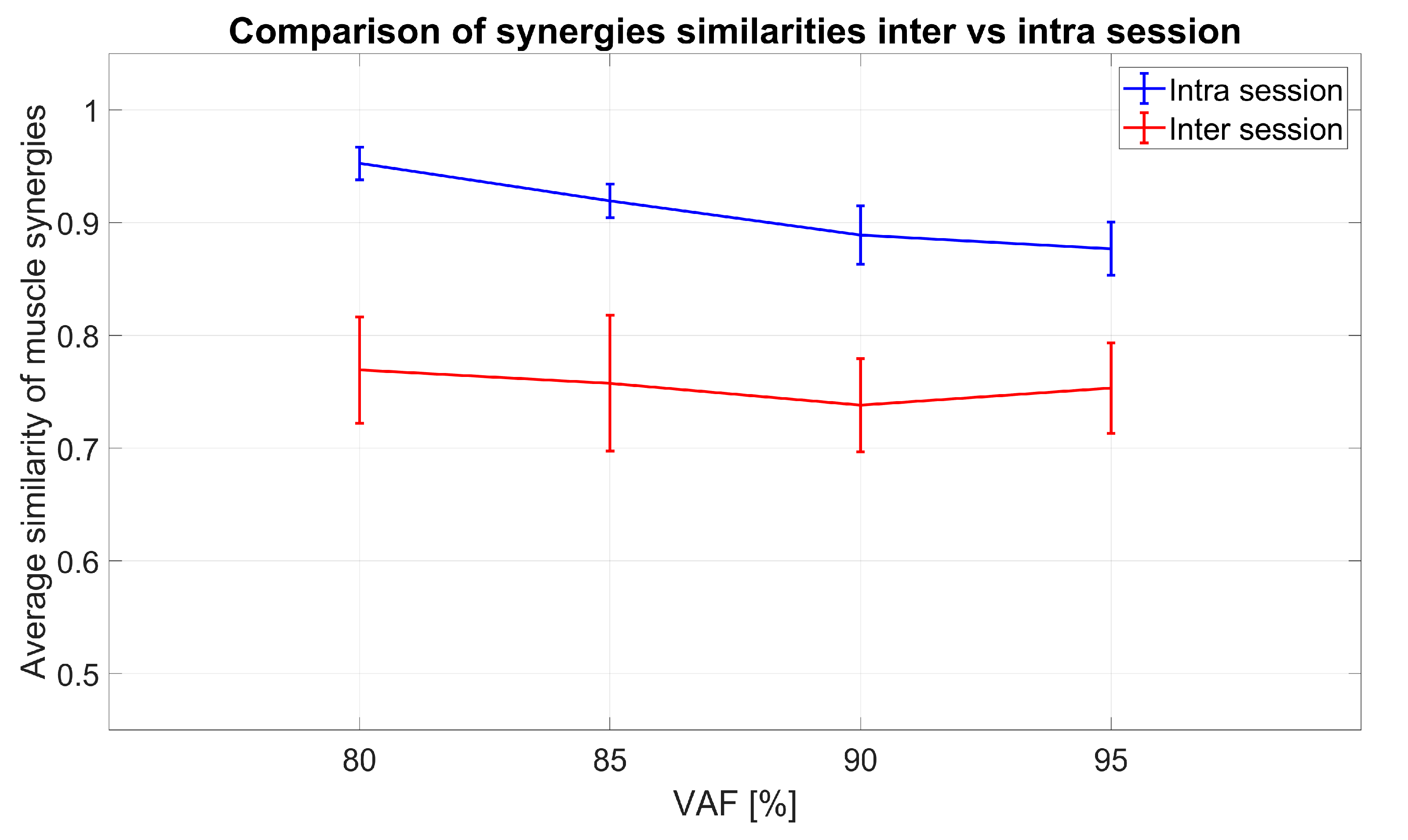
- Comp#1: to test whether significant differences existed between similarities of synergies extracted from the same sessions (IntraSessORIG) and synergies from different sessions (InterSessORIG) both without virtually repositioning of electrodes. This was analogous to the comparison in Section 2.4.2 but with 8 electrodes instead of 14.
- Comp#2: to test whether a significant difference exists in average values of similarities between intra-session (IntraSessORIG) and intra-session with different steps in virtual electrode repositioning (IntraSessVER). This allowed to show whether higher, lower, or comparable intra-session synergy similarity was achievable when electrodes are virtually repositioned.
- Comp#3: to test whether a significant difference exists between the average value of similarities of sessions with original positions (InterSessORIG) and sessions with the best electrode positioning combination (InterSessVER). This allows us to test whether on average it is possible to get higher inter-session similarity when allowing electrode displacement between sessions. In other words, is it possible that slight electrode repositioning between sessions is one of the factors influencing lower synergy similarity?
- Comp#4: to test whether a significant difference exists between maximal similarities that can be achieved for inter (InterSessVER) and intra-session (IntraSessVER) conditions while allowing electrode repositioning. This allows to test if, by moving electrodes between sessions, for any combination of sessions, it is possible to get similarity as high as with intra-session data.
- Comp#5: to test whether a significant difference exists between synergies of the same subject extracted in different sessions (InterSessORIG) when compared to synergies of other subjects (InterSUBJ). This investigation tests how well muscle synergies generalize between individuals with respect to the average inter-session similarity. The analysis was done in order to assess whether lower inter-session (with respect to intra-session) similarities impose serious concern for multi-session or longitudinal assessments. If inter-subject similarities were lower than inter-session ones, synergies contained subject specific information, but generalization of synergies between subjects may not be very high. On the other hand, if inter-subject similarities were on the same level (or higher) than inter-session, then the synergies generalize well between subjects but it is not possible to distinguish whether synergies are from the same subject but recorded during different sessions or from a completely different subject. This can be a serious point to consider for multi-session assessments.
2.5. Statistical Analysis
3. Results
3.1. Intra-Session and Inter-Session Variability
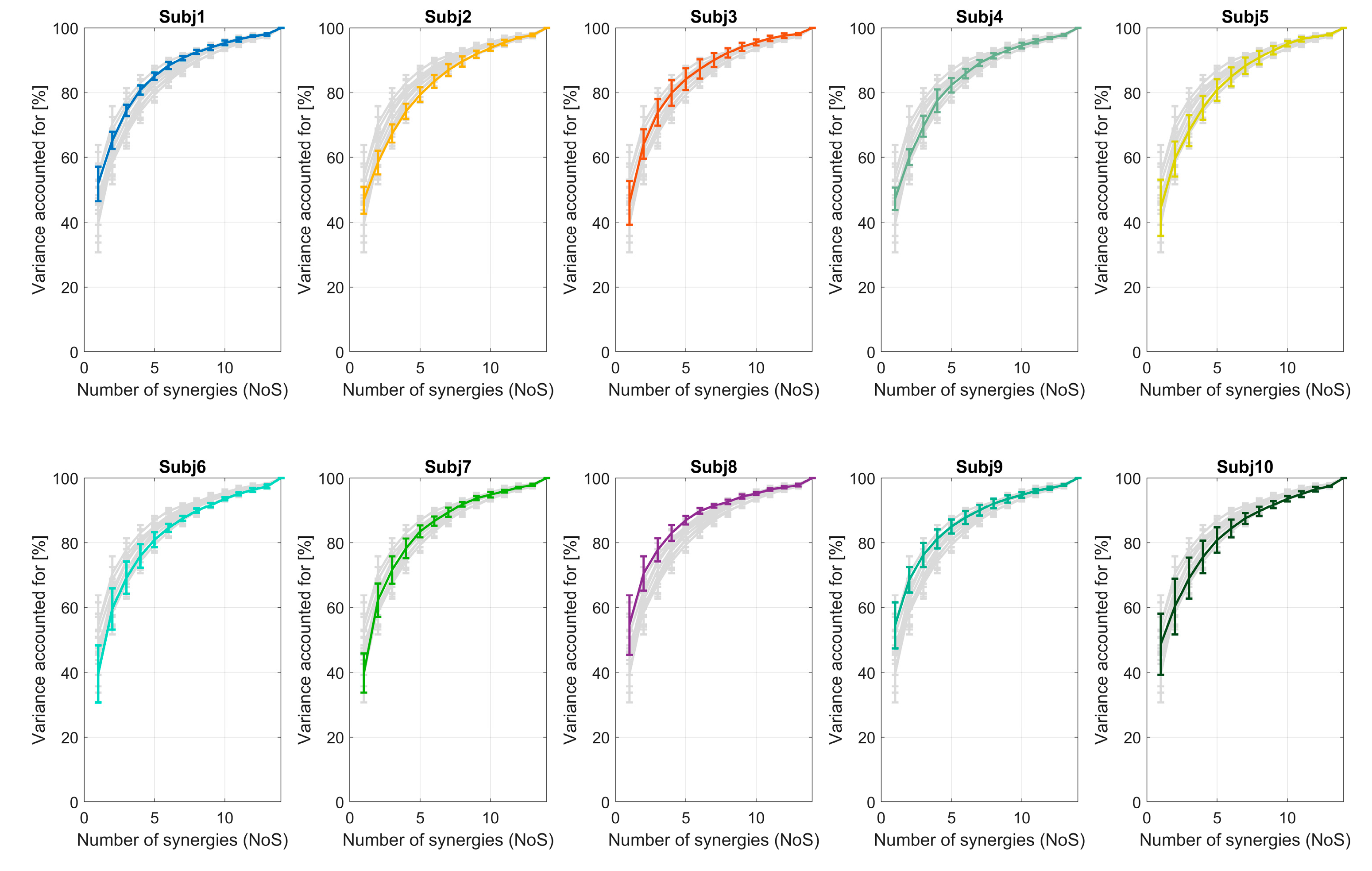
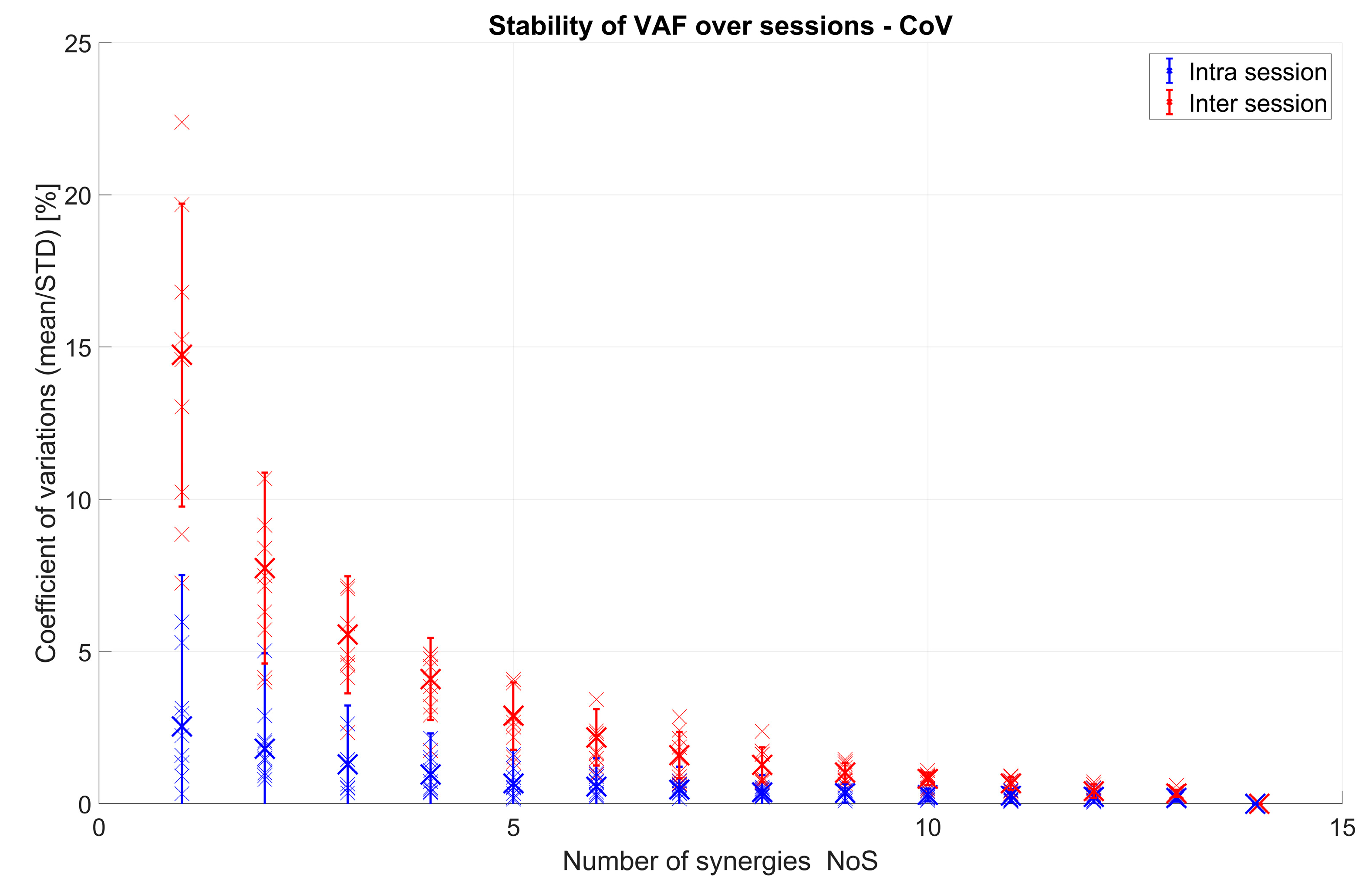
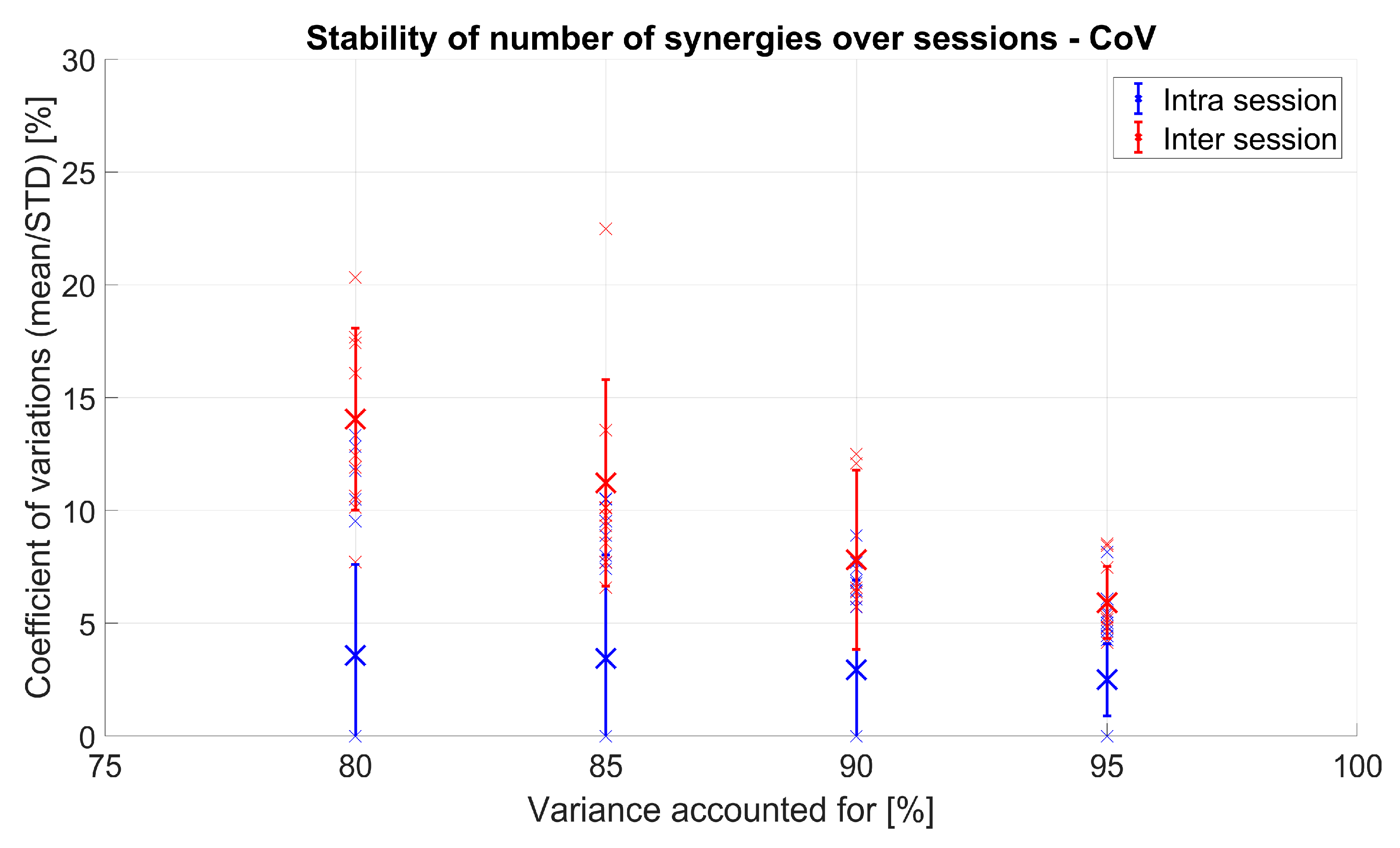
3.1.1. VAF and Number of Extracted Synergies
3.1.2. Spatial Muscle Synergy Similarity
3.2. Virtual Electrode Repositioning
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bernstein, N. The Co-Ordination and Regulation of Movement; Pergamon Press: Oxford, UK, 1967. [Google Scholar]
- D’Avella, A.; Bizzi, E. Shared and specific muscle synergies in natural motor behaviors. Proc. Natl. Acad. Sci. USA 2005, 102, 3076–3081. [Google Scholar] [CrossRef]
- D’Avella, A.; Saltiel, P.; Bizzi, E. Combinations of muscle synergies in the construction of a natural motor behavior. Nat. Neurosci. 2003, 6, 300–308. [Google Scholar] [CrossRef]
- Tresch, M.C.; Cheung, V.C.K.; d’Avella, A. Matrix Factorization Algorithms for the Identification of Muscle Synergies: Evaluation on Simulated and Experimental Data Sets. J. Neurophysiol. 2006, 95, 2199–2212. [Google Scholar] [CrossRef] [PubMed]
- Hilt, P.M.; Delis, I.; Pozzo, T.; Berret, B. Space-by-Time Modular Decomposition Effectively Describes Whole-Body Muscle Activity during Upright Reaching in Various Directions. Front. Comput. Neurosci. 2018, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Chvatal, S.A.P.D.; Ting, L.H.P.D. Common muscle synergies for balance and walking. Front. Comput. Neurosci. 2013, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Tomita, A.; Ando, R.; Watanabe, K.; Akima, H. Muscle synergies are consistent across level and uphill treadmill running. Sci. Rep. 2018, 8, 5979. [Google Scholar] [CrossRef]
- Turpin, N.A.; Guével, A.; Durand, S.; Hug, F. No evidence of expertise-related changes in muscle synergies during rowing. J. Electromyogr. Kinesiol. 2011, 21, 1030–1040. [Google Scholar] [CrossRef]
- Hug, F.; Turpin, N.A.; Guével, A.; Dorel, S. Is interindividual variability of EMG patterns in trained cyclists related to different muscle synergies? J. Appl. Physiol. 2010, 108, 1727–1736. [Google Scholar] [CrossRef]
- Alibeji, N.A.; Kirsch, N.A.; Sharma, N. A Muscle Synergy-Inspired Adaptive Control Scheme for a Hybrid Walking Neuroprosthesis. Front. Bioeng. Biotechnol. 2015, 3, 203. [Google Scholar] [CrossRef]
- Steele, K.M.; Rozumalski, A.; Schwartz, M.H. Muscle synergies and complexity of neuromuscular control during gait in cerebral palsy. Dev. Med. Child. Neurol. 2015, 57, 1176–1182. [Google Scholar] [CrossRef]
- Cheung, V.C.K.; Turolla, A.; Agostini, M.; Silvoni, S.; Bennis, C.; Kasi, P.; Paganoni, S.; Bonato, P.; Bizzi, E. Muscle synergy patterns as physiological markers of motor cortical damage. Proc. Natl. Acad. Sci. USA 2012, 109, 14652–14656. [Google Scholar] [CrossRef] [PubMed]
- Dipietro, L.; Krebs, H.I.; Fasoli, S.E.; Volpe, B.T.; Stein, J.; Bever, C.; Hogan, N. Changing Motor Synergies in Chronic Stroke. J. Neurophysiol. 2007, 98, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.M.; Bao, Y.; Zhuang, C.; Li, S.; Wang, T.; Cui, L.; Xie, Q.; Lan, N. Synergy-Based FES for Post-Stroke Rehabilitation of Upper-Limb Motor Functions. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Pirondini, E.; Coscia, M.; Marcheschi, S.; Roas, G.; Salsedo, F.; Frisoli, A.; Bergamasco, M.; Micera, S. Evaluation of the effects of the Arm Light Exoskeleton on movement execution and muscle activities: A pilot study on healthy subjects. J. Neuroeng. Rehabil. 2016, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- García-Cossio, E.; Broetz, D.; Birbaumer, N.; Ramos-Murguialday, A. Cortex Integrity Relevance in Muscle Synergies in Severe Chronic Stroke. Front. Hum. Neurosci. 2014, 8, 744. [Google Scholar] [CrossRef]
- Roh, J.; Rymer, W.Z.; Beer, R.F. Evidence for altered upper extremity muscle synergies in chronic stroke survivors with mild and moderate impairment. Front. Hum. Neurosci. 2015, 9, 6. [Google Scholar] [CrossRef]
- Pan, B.; Sun, Y.; Xie, B.; Huang, Z.; Wu, J.; Hou, J.; Liu, Y.; Huang, Z.; Zhang, Z. Alterations of Muscle Synergies during Voluntary Arm Reaching Movement in Subacute Stroke Survivors at Different Levels of Impairment. Front. Comput. Neurosci. 2018, 12, 69. [Google Scholar] [CrossRef]
- Tropea, P.; Monaco, V.; Coscia, M.; Posteraro, F.; Micera, S. Effects of early and intensive neuro-rehabilitative treatment on muscle synergies in acute post-stroke patients: A pilot study. J. Neuroeng. Rehabil. 2013, 10, 103. [Google Scholar] [CrossRef]
- Kristiansen, M.; Samani, A.; Madeleine, P.; Hansen, E.A. Muscle synergies during bench press are reliable across days. J. Electromyogr. Kinesiol. 2016, 30, 81–88. [Google Scholar] [CrossRef]
- D’Avella, A.; Portone, A.; Fernandez, L.; Lacquaniti, F. Control of Fast-Reaching Movements by Muscle Synergy Combinations. J. Neurosci. 2006, 26, 7791–7810. [Google Scholar] [CrossRef]
- Scano, A.; Dardari, L.; Molteni, F.; Giberti, H.; Tosatti, L.M.; d’Avella, A. A Comprehensive Spatial Mapping of Muscle Synergies in Highly Variable Upper-Limb Movements of Healthy Subjects. Front. Physiol. 2019, 10, 1231. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Gizzi, L.; Farina, D.; Kersting, U.G. Motor modules of human locomotion: Influence of EMG averaging, concatenation, and number of step cycles. Front. Hum. Neurosci. 2014, 8, 335. [Google Scholar] [CrossRef] [PubMed]
- Taborri, J.; Palermo, E.; Masiello, D.; Rossi, S. Factorization of EMG via muscle synergies in walking task: Evaluation of intra-subject and inter-subject variability. In Proceedings of the 2017 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), Turin, Italy, 22–25 May 2017; pp. 1–6. [Google Scholar]
- Frère, J.; Hug, F. Between-subject variability of muscle synergies during a complex motor skill. Front. Comput. Neurosci. 2012, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Steinwender, G.; Saraph, V.; Scheiber, S.; Zwick, E.B.; Uitz, C.; Hackl, K. Intrasubject repeatability of gait analysis data in normal and spastic children. Clin. Biomech. 2000, 15, 134–139. [Google Scholar] [CrossRef]
- Zahedi, M.S.; Spence, W.D.; Solomonidis, S.E.; Paul, J.P. Repeatability of kinetic and kinematic measurements in gait studies of the lower limb amputee. Prosthet. Orthot. Int. 1987, 11, 55–64. [Google Scholar] [CrossRef]
- Atzori, M.; Gijsberts, A.; Castellini, C.; Caputo, B.; Hager, A.-G.M.; Elsig, S.; Giatsidis, G.; Bassetto, F.; Müller, H. Electromyography data for non-invasive naturally-controlled robotic hand prostheses. Sci. Data 2014, 1, 140053. [Google Scholar] [CrossRef]
- Palermo, F.; Cognolato, M.; Gijsberts, A.; Müller, H.; Caputo, B.; Atzori, M. Repeatability of grasp recognition for robotic hand prosthesis control based on sEMG data. In Proceedings of the 2017 International Conference on Rehabilitation Robotics (ICORR), London, UK, 17–21 July 2017; pp. 1154–1159. [Google Scholar]
- Van der Krogt, M.M.; Oudenhoven, L.; Buizer, A.I.; Dallmeijer, A.; Dominici, N.; Harlaar, J. The effect of EMG processing choices on muscle synergies before and after BoNT-A treatment in cerebral palsy. In Proceedings of the European Society of Movement Analysis for Adults and Children, Seville, Spain, 26 September–1 October 2016. [Google Scholar]
- Kim, Y.; Bulea, T.C.; Damiano, D.L. Novel Methods to Enhance Precision and Reliability in Muscle Synergy Identification during Walking. Front. Hum. Neurosci. 2016, 10, 455. [Google Scholar] [CrossRef]
- Atzori, M.; Castellini, C.; Müller, H. Spatial Registration of Hand Muscle Electromyography Signals. In Proceedings of the 7th International Workshop on Biosignal Interpretation, Como, Italy, 2–4 July 2012. [Google Scholar]
- Steinhardt, C.R.; Bettthauser, J.; Hunt, C.; Thakor, N. Registration of EMG Electrodes to Reduce Classification Errors due to Electrode Shift. In Proceedings of the 2018 IEEE Biomedical Circuits and Systems Conference (BioCAS), Cleveland, OH, USA, 17–19 October 2018; pp. 1–4. [Google Scholar]
- Gijsberts, A.; Atzori, M.; Castellini, C.; Müller, H.; Caputo, B. Movement Error Rate for Evaluation of Machine Learning Methods for sEMG-Based Hand Movement Classification. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 735–744. [Google Scholar] [CrossRef]
- Feix, T.; Romero, J.; Schmiedmayer, H.-B.; Dollar, A.M.; Kragic, D. The GRASP Taxonomy of Human Grasp Types. IEEE Trans. Hum.-Mach. Syst. 2016, 46, 66–77. [Google Scholar] [CrossRef]
- Kuzborskij, I.; Gijsberts, A.; Caputo, B. On the challenge of classifying 52 hand movements from surface electromyography. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 4931–4937. [Google Scholar]
- Hargrove, L.; Englehart, K.; Hudgins, B. A training strategy to reduce classification degradation due to electrode displacements in pattern recognition based myoelectric control. Biomedical Signal. Proc. Control 2008, 3, 175–180. [Google Scholar] [CrossRef]
- Scano, A.; Chiavenna, A.; Molinari Tosatti, L.; Müller, H.; Atzori, M. Muscle Synergy Analysis of a Hand-Grasp Dataset: A Limited Subset of Motor Modules May Underlie a Large Variety of Grasps. Front. Neurorobot. 2018, 12, 57. [Google Scholar] [CrossRef] [PubMed]
- Castellini, C.; van der Smagt, P. Evidence of muscle synergies during human grasping. Biol. Cybern. 2013, 107, 233–245. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Castellini, C.; Gruppioni, E.; Davalli, A.; Sandini, G. Fine detection of grasp force and posture by amputees via surface electromyography. J. Physiol. Paris 2009, 103, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.; Rymer, W.Z.; Perreault, E.J.; Yoo, S.B.; Beer, R.F. Alterations in upper limb muscle synergy structure in chronic stroke survivors. J. Neurophysiol. 2012, 109, 768–781. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.D.; Seung, H.S. Learning the parts of objects by non-negative matrix factorization. Nature 1999, 401, 788–791. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.D.; Seung, H.S. Algorithms for Non-negative Matrix Factorization. In Advances in Neural Information Processing Systems; MIT Press: Cambridge, MA, USA, 2000; pp. 556–562. [Google Scholar]
- Shuman, B.; Goudriaan, M.; Bar-On, L.; Schwartz, M.H.; Desloovere, K.; Steele, K.M. Repeatability of muscle synergies within and between days for typically developing children and children with cerebral palsy. Gait Posture 2016, 45, 127–132. [Google Scholar] [CrossRef]
- Hagio, S.; Kouzaki, M. The flexible recruitment of muscle synergies depends on the required force-generating capability. J. Neurophysiol. 2014, 112, 316–327. [Google Scholar] [CrossRef]
- Cheung, V.C.K.; Piron, L.; Agostini, M.; Silvoni, S.; Turolla, A.; Bizzi, E. Stability of muscle synergies for voluntary actions after cortical stroke in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 19563–19568. [Google Scholar] [CrossRef]
- Banks, C.L.; Pai, M.M.; McGuirk, T.E.; Fregly, B.J.; Patten, C. Methodological Choices in Muscle Synergy Analysis Impact Differentiation of Physiological Characteristics Following Stroke. Front. Comput. Neurosci. 2017, 11, 78. [Google Scholar] [CrossRef]
- Barradas, V.R.; Kutch, J.J.; Kawase, T.; Koike, Y.; Schweighofer, N. When 90% of the variance is not enough: Residual EMG from muscle synergy extraction influences task performance. J. Neurophysiol. 2020, 123, 2180–2190. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2013. [Google Scholar]
- Latash, M.; Scholz, J.; Schöner, G. Motor Control Strategies Revealed in the Structure of Motor Variability. Exerc. Sport Sci. Rev. 2002, 30, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Jarque-Bou, N.J.; Scano, A.; Atzori, M.; Müller, H. Kinematic synergies of hand grasps: A comprehensive study on a large publicly available dataset. J. NeuroEng. Rehabil. 2019, 16, 63. [Google Scholar] [CrossRef] [PubMed]
- Stival, F.; Michieletto, S.; Cognolato, M.; Pagello, E.; Müller, H.; Atzori, M. A quantitative taxonomy of human hand grasps. J. Neuroeng. Rehabil. 2019, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Jarque-Bou, N.J.; Vergara, M.; Sancho-Bru, J.L.; Gracia-Ibáñez, V.; Roda-Sales, A. A calibrated database of kinematics and EMG of the forearm and hand during activities of daily living. Sci. Data 2019, 6, 270. [Google Scholar] [CrossRef] [PubMed]
- Tortora, S.; Moro, M.; Menegatti, E. Dual-Myo Real-Time Control of a Humanoid Arm for Teleoperation. In Proceedings of the 2019 14th ACM/IEEE International Conference on Human-Robot Interaction (HRI), Daegu, Korea, 11–13 March 2019; pp. 624–625. [Google Scholar]
- Santello, M.; Bianchi, M.; Gabiccini, M.; Ricciardi, E.; Salvietti, G.; Prattichizzo, D.; Ernst, M.; Moscatelli, A.; Jörntell, H.; Kappers, A.M.L.; et al. Hand synergies: Integration of robotics and neuroscience for understanding the control of biological and artificial hands. Phys. Life Rev. 2016, 17, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Buongiorno, D.; Barsotti, M.; Barone, F.; Bevilacqua, V.; Frisoli, A. A Linear Approach to Optimize an EMG-Driven Neuromusculoskeletal Model for Movement Intention Detection in Myo-Control: A Case Study on Shoulder and Elbow Joints. Front. Neurorobot. 2018, 12, 74. [Google Scholar] [CrossRef]
- Schaffelhofer, S.; Sartori, M.; Scherberger, H.; Farina, D. Musculoskeletal Representation of a Large Repertoire of Hand Grasping Actions in Primates. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 210–220. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pale, U.; Atzori, M.; Müller, H.; Scano, A. Variability of Muscle Synergies in Hand Grasps: Analysis of Intra- and Inter-Session Data. Sensors 2020, 20, 4297. https://doi.org/10.3390/s20154297
Pale U, Atzori M, Müller H, Scano A. Variability of Muscle Synergies in Hand Grasps: Analysis of Intra- and Inter-Session Data. Sensors. 2020; 20(15):4297. https://doi.org/10.3390/s20154297
Chicago/Turabian StylePale, Una, Manfredo Atzori, Henning Müller, and Alessandro Scano. 2020. "Variability of Muscle Synergies in Hand Grasps: Analysis of Intra- and Inter-Session Data" Sensors 20, no. 15: 4297. https://doi.org/10.3390/s20154297
APA StylePale, U., Atzori, M., Müller, H., & Scano, A. (2020). Variability of Muscle Synergies in Hand Grasps: Analysis of Intra- and Inter-Session Data. Sensors, 20(15), 4297. https://doi.org/10.3390/s20154297






