Towards Naples Ecological REsearch for Augmented Observatories (NEREA): The NEREA-Fix Module, a Stand-Alone Platform for Long-Term Deep-Sea Ecosystem Monitoring †
Abstract
1. Introduction
1.1. Ocean Observatories
1.2. The Strategic Need for a New Ecological Monitoring of the Deep Sea
1.3. Objectives
2. Deployment Characteristics
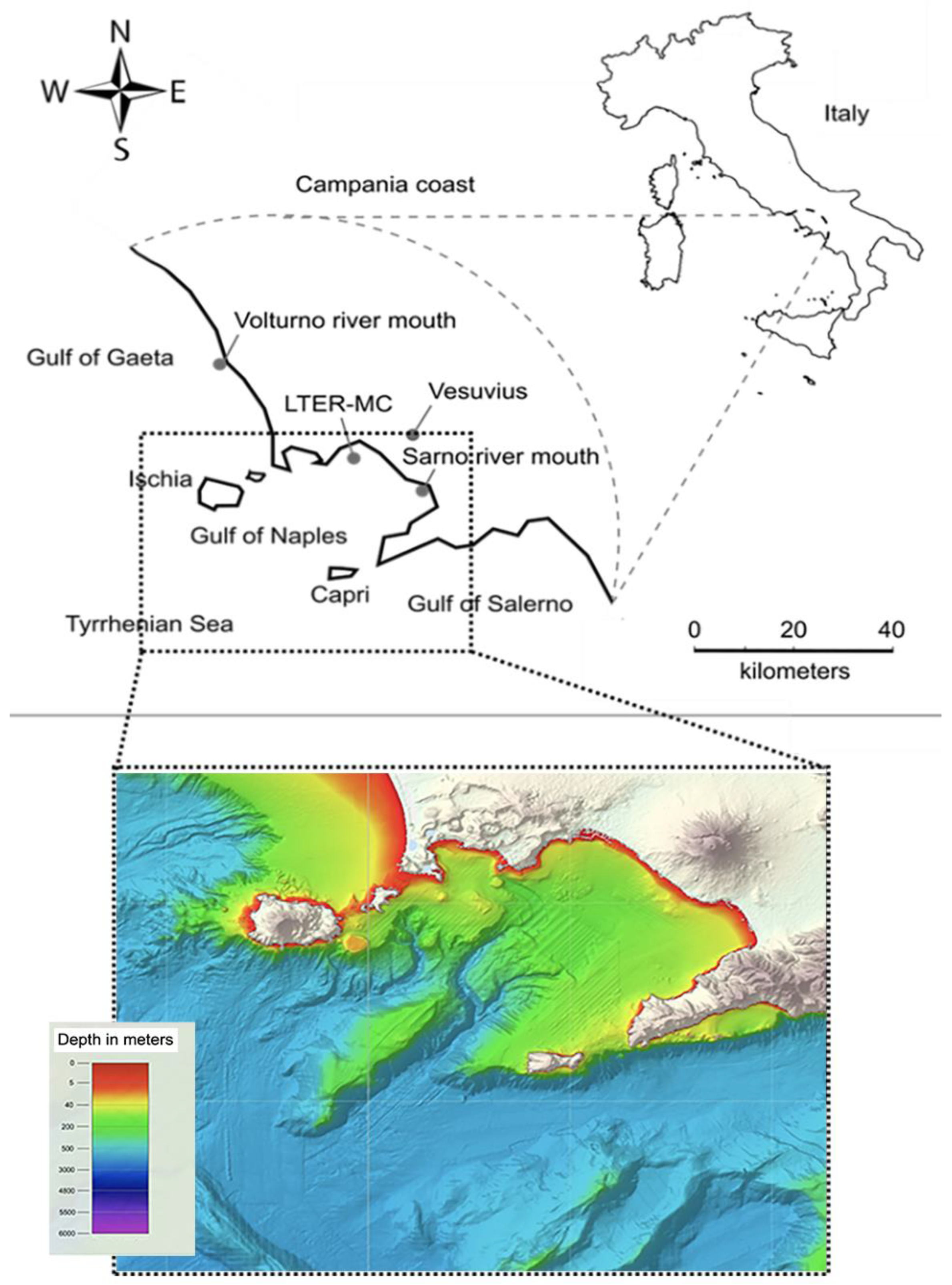
2.1. The Deployment Area
2.2. NEREA Research Infrastructure
2.3. The Seabed Platform (SP), the Mooring Line (ML) and the Surface Buoy
2.4. Instruments and Sensors
2.5. The Coupled Data Acquisition by NEREA-Fix and NEREA-Mob and Omics Expansion
3. Discussion
4. Towards a Network of Deep-Sea Augmented Observatories
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Falkowsky, P.G.; Barber, R.T.; Smetacek, V.V. Biogeochemical Controls and Feedbacks on Ocean Primary Production. Science 1998, 281, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Aguzzi, J.; Chatzievangelou, D.; Marini, S.; Fanelli, E.; Danovaro, R.; Flögel, S.; Lebris, N.; Juanes, F.; De Leo, F.; Del Rio, J.; et al. New high-tech interactive and flexible networks for the future monitoring of deep-sea ecosystems. Environ. Sci. Technol. 2019, 53, 6616–6631. [Google Scholar] [CrossRef] [PubMed]
- Dañobeitia, J.J.; Pouliquen, S.; Johannessen, T.; Basset, A.; Cannat, M.; Pfeil, B.G.; Fredella, M.I.; Materia, P.; Gourcuff, C.; Magnifico, G.; et al. Toward a comprehensive and integrated strategy of the European marine research infrastructures for ocean observations. Front. Mar. Sci. 2020, 7, 180. [Google Scholar] [CrossRef]
- Rountree, R.; Aguzzi, J.; Marini, S.; Fanelli, E.; De Leo, C.F.; Del Rio, J.; Juanes, F. Towards an optimal design for ecosystem-level ocean observatories. Oceanogr. Mar. Biol. 2020, 58, 177975. [Google Scholar]
- Aguzzi, J.; Chatzievangelou, D.; Francescangeli, M.; Marini, S.; Bonofiglio, F.; del Río, J.; Danovaro, R. The hierarchic treatment of marine ecological information from spatial networks of benthic platforms. Sensors 2020, 20, 1751. [Google Scholar] [CrossRef]
- Chirayath, V.; Li, A. Next-Generation Optical Sensing Technologies for Exploring Ocean Worlds—NASA FluidCam, MiDAR, and NeMO-Net. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- D’Alelio, D.; Eveillard, D.; Coles, V.J.; Caputi, L.; D’Alcalá, M.R.; Iudicone, D. Modelling the complexity of plankton communities exploiting omics potential: From present challenges to an integrative pipeline. Curr. Opin. Syst. Boil. 2019, 13, 68–74. [Google Scholar] [CrossRef]
- Stec, K.F.; Caputi, L.; Buttigieg, P.L.; D’Alelio, D.; Ibarbalz, F.M.; Sullivan, M.B.; Chaffron, S.; Bowler, C.; D’Alcalá, M.R.; Iudicone, D. Modelling plankton ecosystems in the meta-omics era. Are we ready? Mar. Genom. 2017, 32, 1–17. [Google Scholar] [CrossRef]
- Danovaro, R.; Fanelli, E.; Aguzzi, J.; Billett, D.; Carugati, L.; Corinaldesi, C.; Dell’Anno, A.; Gjerde, K.; Jamieson, A.J.; Kark, S.; et al. Ecological indicators for a global deep-ocean monitoring and conservation strategy. Nat. Ecol. Evol. 2020, 4, 181–192. [Google Scholar] [CrossRef]
- Danovaro, R.; Aguzzi, J.; Fanelli, E.; Billet, D.; Gjerde, K.; Jamieson, A.; Ramirez-Llodra, E.; Smith, C.R.; Snelgrove, P.V.R.; Thomsen, L.; et al. A new international ecosystem-based strategy for the global deep ocean. Science 2017, 355, 452–454. [Google Scholar] [CrossRef]
- Aguzzi, J.; Doya, C.; Tecchio, S.; De Leo, F.; Azzurro, E.; Costa, C.; Sbragaglia, V.; Del Río, J.; Navarro, J.; Ruhl, H.A.; et al. Coastal observatories for monitoring of fish behaviour and their responses to environmental changes. Rev. Fish Biol. Fish. 2015, 25, 463–483. [Google Scholar] [CrossRef]
- Bicknell, A.W.J.; Godley, B.J.; Sheenhan, E.V.; Votier, S.C.; Witt, M.J. Camera technology for monitoring marine biodiversity and human impact. Front. Ecol. Environ. 2016, 8, 424–432. [Google Scholar] [CrossRef]
- Chauvet, P.; Metaxas, A.; Hay, A.E.; Matabos, M. Annual and seasonal dynamics of deep-sea megafaunal epibenthic communities in Barkley Canyon (British Columbia, Canada): A response to climatology, surface productivity and benthic boundary layer variation. Prog. Oceanogr. 2018, 169, 89–105. [Google Scholar] [CrossRef]
- De Leo, F.C.; Ogata, B.; Sastri, A.R.; Heesemann, M.; Mihály, S.; Galbraith, M.; Morley, M.G. High-frequency observations from a deep-sea cabled observatory reveal seasonal overwintering of Neocalanus spp. in Barkley Canyon, NE Pacific: Insights into particulate organic carbon flux. Progress Oceanogr. 2018, 169, 120–137. [Google Scholar] [CrossRef]
- Del Río, J.; Nogueras, M.; Toma, D.; Martínez, E.; Artero, C.; Bghiel, I.; Martinez, M.; Cadena, J.; Garcia-Benadi, A.; Sarria, D.; et al. A decadal balance for a cabled observatory deployment. IEEE Access 2020, 8, 33163–33177. [Google Scholar] [CrossRef]
- Painting, S.J.; Collingridge, K.A.; Durand, D.; Grémare, A.; Créach, V.; Bernard, G. Marine monitoring in Europe: Is it adequate to address environmental threats and pressures? Ocean Sci. 2020, 16, 235–252. [Google Scholar] [CrossRef]
- Gawarkiewicz, G.; Plueddemann, A.J. Scientific rationale and conceptual design of a process-oriented shelfbreak observatory: The OOI Pioneer Array. J. Oper. Oceanogr. 2020, 13, 19–36. [Google Scholar] [CrossRef]
- Ruhl, H.A.; André, M.; Beranzoli, L.; Çağatay, M.N.; Colaço, A.; Cannat, M.; Dañobeitia, J.J.; Favali, P.; Géli, L.; Gillooly, M.; et al. Societal need for improved understanding of climate change, anthropogenic impacts, and geo-hazard warning drive development of ocean observatories in European Seas. Prog. Oceanogr. 2011, 91, 1–33. [Google Scholar] [CrossRef]
- Smith, L.M.; Barth, J.A.; Kelley, D.S.; Plueddemann, A.; Rodero, I.; Ulses, G.A.; Vardaro, M.F.; Weller, R. The Ocean Observatories Initiative. Oceanography 2018, 31, 16–35. [Google Scholar] [CrossRef]
- Levin, L.A.; Bett, B.J.; Gates, A.R.; Heimbach, P.; Howe, B.M.; Janssen, F.; McCurdy, A.; Ruhl, H.A.; Snelgrove, P.; Stocks, K.I.; et al. Global Observing Needs in the Deep Ocean. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Canals, M.; Puig, P.; De Madron, X.D.; Heussner, S.; Palanques, A.; Fabres, J. Flushing submarine canyons. Nature 2006, 444, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Thurber, A.R.; Sweetman, A.K.; Narayanaswamy, B.E.; Jones, D.O.B.; Ingels, J.; Hansman, R.L. Ecosystem function and services provided by the deep sea. Biogeosciences 2014, 11, 3941–3963. [Google Scholar] [CrossRef]
- Danovaro, R.; Snelgrove, P.V.R.; Tyler, P. Challenging the paradigms of deep-sea ecology. Trends Ecol. Evol. 2014, 29, 465–475. [Google Scholar] [CrossRef]
- Puig, P.; Palanques, A.; Martín, J. Contemporary Sediment-Transport Processes in Submarine Canyons. Ann. Rev. Mar. Sci. 2014, 6, 53–77. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Arcaya, U.; Ramirez-Llodra, E.; Aguzzi, J.; Allcock, A.L.; Davies, J.S.; Dissanayake, A.; Harris, P.; Howell, K.; Huvenne, V.A.I.; Ismail, K.; et al. Ecological role of submarine canyons and need for canyon conservation: A review. Front. Mar. Sci. 2017, 31, 53–77. [Google Scholar] [CrossRef]
- Fanelli, E.; Bianchelli, S.; Danovaro, R. Deep-sea mobile megafauna of Mediterranean submarine canyons and open slopes: Analysis of spatial and bathymetric gradients. Prog. Oceanogr. 2018, 168, 23–24. [Google Scholar] [CrossRef]
- Afonso, P.; McGinty, N.; Graça, G.; Fontes, J.; Inácio, M.; Totland, A.; Menezes, G. Vertical Migrations of a Deep-Sea Fish and Its Prey. PLoS ONE 2014, 9, e97884. [Google Scholar] [CrossRef]
- Cartes, J.E.; Fanelli, E.; Papiol, V.; Maynou, F. Trophic relationships at meso-spatial and seasonal scales of macro and megafauna in a submarine canyon and the adjacent slope off the Catalonian coast (western Mediterranean). J. Sea Res. 2010, 63, 180–190. [Google Scholar] [CrossRef]
- Rogers, A.D. Environmental change in the deep ocean. Annu. Rev. Environ. Resour. 2015, 40, 1–38. [Google Scholar] [CrossRef]
- Doya, C.; Aguzzi, J.; Pardo, M.; Matabos, M.; Company, J.; Costa, C.; Mihály, S.; Canals, M. Diel behavioral rhythms in sablefish (Anoplopoma fimbria) and other benthic species, as recorded by the Deep-sea cabled observatories in Barkley canyon (NEPTUNE-Canada). J. Mar. Syst. 2014, 130, 69–78. [Google Scholar] [CrossRef]
- Aguzzi, J.; Fanelli, E.; Ciuffardi, T.; Schirone, A.; Craig, G.; The NEMO Consortium. Inertial bioluminescence rhythms at the Central Mediterranean KM3NeT deep-sea neutrino telescope. Sci. Rep. 2017, 7, 44938. [Google Scholar] [CrossRef] [PubMed]
- Aguzzi, J.; Fanelli, E.; Ciuffardi, T.; Schirone, A.; De Leo, F.C.; Doya, C.; Kawato, M.; Miyazaki, M.; Furushima, Y.; Costa, C.; et al. Faunal activity rhythms influencing early community succession of an implanted whale carcass offshore in Sagami Bay, Japan. Sci. Rep. 2018, 8, 11163. [Google Scholar] [CrossRef] [PubMed]
- Graeme, C.H.; Ferreira, L.C.; Sequeira, A.M.M.; Meekan, M.G.; Duarte, C.M.; Bailey, H.; Bailleul, F.; Bowen, W.D.; Caley, M.J.; Costa, D.P.; et al. Key questions in marine megafauna movement ecology. Trends Ecol. Evol. 2010, 31, 463–475. [Google Scholar]
- Milligan, R.J.; Scott, E.M.; Jones, D.O.B.; Bett, B.J.; Jamieson, A.J.; O’Brien, R.; Pereira Costa, S.; Rowe, G.T.; Ruhl, H.A.; Smith, K.L., Jr.; et al. Evidence for seasonal cycles in deep-sea fish abundances: A great migration in the deep SE Atlantic? J. Animal. Ecol. 2020. [Google Scholar] [CrossRef]
- Beale, A.D.; Whitmore, D.; Moran, D. Life in a dark biosphere: A review of circadian physiology in “arrhythmic” environments. J. Comp. Physiol. B 2016, 186, 947–968. [Google Scholar] [CrossRef] [PubMed]
- Suetsugu, K.; Ohta, S. Day-night changes in species composition of deep-sea demersal fishes. J. Oceanog. 2005, 61, 187–196. [Google Scholar] [CrossRef]
- Benoit-Bird, K.J.; Au, W.W. Extreme diel horizontal migrations by a tropical nearshore resident micronekton community. Mar. Ecol. Prog. Ser. 2006, 319, 1–14. [Google Scholar] [CrossRef]
- Mendia, L.; d’Elia, E.; Evison, L.; Tosti, E. Pollution Control and Quality Levels of the Bay of Naples. In Mediterranean Coastal Pollution; Jenkins, S.H., Ed.; Pergamon Press Ltd.: Oxford, UK, 1980; pp. 615–631. [Google Scholar]
- Volterra, L.; Tosti, E.; Vero, A.; Izzo, G. Microbiological pollution of marine sediments in the southern stretch of the Gulf of Naples. Water Air Soil Pollut. 1985, 26, 175–184. [Google Scholar] [CrossRef]
- Naso, B.; Perrone, D.; Ferrante, M.C.; Bilancione, M.; Lucisano, A. Persistent organic pollutants in edible marine species from the Gulf of Naples, Southern Italy. Sci. Total Environ. 2005, 343, 83–95. [Google Scholar] [CrossRef]
- Tornero, V.; Ribera d’Alcalà, M. Contamination by hazardous substances in the Gulf of Naples and nearby coastal areas: A review of sources, environmental levels and potential impacts in the MSFD perspective. Sci. Total Environ. 2014, 466–467, 820–840. [Google Scholar] [CrossRef]
- Taviani, M.; Angeletti, L.; Cardone, F.; Montagna, P.; Danovaro, R. A unique and threatened deep water coral-bivalve biotope new to the Mediterranean Sea offshore the Naples megalopolis. Sci. Rep. 2019, 3411, 9. [Google Scholar] [CrossRef]
- De Ruggiero, P.; Napolitano, E.; Iacono, R.; Pierini, S. A high-resolution modelling study of the circulation along the Campania coastal system, with a special focus on the Gulf of Naples. Cont. Shelf Res. 2016, 122, 85–101. [Google Scholar] [CrossRef]
- De Ruggiero, P.; Napolitano, E.; Iacono, R.; Pierini, S.; Spezie, G. A baroclinic coastal trapped wave event in the Gulf of Naples (Tyrrhenian Sea). Ocean Dyn. 2018, 68, 1683–1694. [Google Scholar] [CrossRef]
- Hopkins, T.S. Recent observations on the intermediate and deep-water circulation in the southern Tyrrhenian Sea. Oceanol. Acta 1988, 9, 41–50. [Google Scholar]
- Milia, A. The Dohrn canyon: A response to the eustatic fall and tectonic uplift of the outer shelf along the eastern Tyrrhenian Sea margin, Italy. Geo-Mar. Lett. 2000, 20, 101–108. [Google Scholar] [CrossRef]
- Colloca, F.; Garofalo, G.; Bitetto, I.; Facchini, M.T.; Grati, F.; Martiradonna, A.; Mastrantonio, G.; Nikolioudakis, N.; Ordinas, F.; Scarcella, G.; et al. The seascape of demersal fish nursery areas in the North Mediterranean Sea, a first step towards the implementation of spatial planning for trawl fisheries. PLoS ONE 2015, 10, e0119590. [Google Scholar] [CrossRef]
- Zingone, A.; Dubroca, L.; Iudicone, D.; Margiotta, F.; Corato, F.; Ribera d’Alcalà, M.; Saggiomo, V.; Sarno, D. Coastal Phytoplankton Do Not Rest in Winter. Estuaries Coasts 2010, 33, 342–361. [Google Scholar] [CrossRef]
- Zingone, A.; D’Alelio, D.; Mazzocchi, M.G.; Montresor, M.; Sarno, D. Time series and beyond: Multifaceted plankton research at a marine Mediterranean LTER site. Nat. Conserv. 2019, 34, 273–310. [Google Scholar] [CrossRef]
- Graf, G. Benthic-pelagic coupling in a deep-sea benthic community. Nature 1989, 341, 437–439. [Google Scholar] [CrossRef]
- Conese, I.; Fanelli, E.; Miserocchi, S.; Langone, L. Food web structure and trophodynamics of deep-sea plankton from the Bari Canyon and adjacent slope (Southern Adriatic, central Mediterranean Sea). Prog. Ocean. 2019, 175, 92–104. [Google Scholar] [CrossRef]
- Bozzano, R.; Fanelli, E.; Pensieri, S.; Picco, P.; Schiano, M.E. Temporal variations in zooplankton biomass in the Ligurian Sea inferred from long time series of ADCP data. Ocean Sci. 2014, 10, 93–105. [Google Scholar] [CrossRef]
- Herring, P.J.; Gaten, E.; Shelton, P.M. Are vent shrimps blinded by science? Nature 1999, 398, 116. [Google Scholar] [CrossRef]
- Martignac, F.; Daroux, A.; Bagliniere, J.-L.; Ombredane, D.; Guillard, J. The use of acoustic cameras in shallow waters: New hydroacoustic tools for monitoring migratory fish population. A review of DIDSON technology. Fish Fish. 2015, 16, 486–510. [Google Scholar] [CrossRef]
- Matabos, M.; Bui, A.O.; Mihály, S.; Aguzzi, J.; Juniper, D.S.K.; Ajayamohan, R.S. High-frequency study of benthic megafaunal community dynamics in Barkley canyon: A multidisciplinary approach using the NEPTUNE Canada network. J. Mar. Syst. 2014, 130, 56–68. [Google Scholar] [CrossRef]
- Sbragaglia, V.; Dominoni, D.; Nuñez, J.D.; Coco, S.; Fanelli, E.; Azzurro, E.; Marini, S.; Nogueras, M.; Ponti, M.; Del Río, J.; et al. Annual rhythms of temporal niche partitioning in the Sparidae family are driven by different environmental cues. Sci. Rep. 2018, 9, 1708. [Google Scholar] [CrossRef]
- Marini, S.; Fanelli, E.; Sbragaglia, V.; Azzurro, E.; del Rio-Fernandez, J.; Aguzzi, J. Tracking Fish Abundance by Underwater Image Recognition. Sci. Rep. 2018, 8, 13748. [Google Scholar] [CrossRef]
- Del Río, J.; Toma, D.M.; Martínez, E.; O’Reilly, T.C.; Delory, E.; Pearlman, J.S.; Waldmann, C.; Jirka, S. A Sensor Web Architecture for integrating smart oceanographic sensors into the semantic sensor Web. IEEE J. Ocean. Eng. 2018, 43, 830–842. [Google Scholar] [CrossRef]
- Chaturvedi, K.; Kolbe, T.H. Towards establishing cross-platform interoperability for sensors in smart cities. Sensors 2019, 19, 562. [Google Scholar] [CrossRef]
- Steele, B.; Chandler, J.; Reddy, S. Algorithms for Data Science; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Corgnati, L.; Marini, S.; Mazzei, L.; Ottaviani, E.; Aliani, S.; Conversi, A.; Griffa, A. Looking inside the Ocean: Toward an Autonomous Imaging System for Monitoring Gelatinous Zooplankton. Sensors 2016, 16, 2124. [Google Scholar] [CrossRef]
- Marini, S.; Corgnati, L.; Mantovani, C.; Bastianini, M.; Ottaviani, E.; Fanelli, E.; Aguzzi, J.; Griffa, A.; Poulain, P.-M. Automated estimate of fish abundance through the autonomous imaging device GUARD1. Measurement 2018, 126, 72–75. [Google Scholar] [CrossRef]
- Papworth, D.J.; Marini, S.; Conversi, A. A Novel, Unbiased Analysis Approach for Investigating Population Dynamics: A Case Study on Calanus finmarchicus and Its Decline in the North Sea. PLoS ONE 2016, 11, e0158230. [Google Scholar] [CrossRef] [PubMed]
- Cordier, T.; Esling, P.; Lejzerowicz, F.; Visco, J.; Ouadahi, A.; Martins, C.; Cedhagen, T.; Pawlowski, J. Predicting the ecological quality status of marine environments from eDNA metabarcoding data using supervised machine learning. Environ. Sci. Technol. 2017, 51, 9118–9126. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R.; Fanelli, E.; Canals, M.; Ciuffardi, T.; Fabri, M.-C.; Taviani, M.; Argyrou, M.; Azzurro, E.; Bianchelli, S.; Cantafaro, A.; et al. Towards a marine strategy for the deep Mediterranean Sea. Mar. Pol. 2020, 112, 103781. [Google Scholar] [CrossRef]
- Di Piazza, A.; Conti, F.L.; Noto, L.V.; Viola, F.; La Loggia, G. Comparative analysis of different techniques for spatial interpolation of rainfall data to create a serially complete monthly time series of precipitation for Sicily, Italy. Int. J. App. Earth Obs. Geoinformat. 2011, 13, 396–408. [Google Scholar] [CrossRef]
- Hengl, T. A Practical Guide to Geostatistical Mapping, 2nd ed.; Office for Official Publications of the European Communitie: Brussels, Belgium, 2009; ISBN 978-92-79-06904-8. [Google Scholar]
- Li, J.; Heap, A.D. A review of comparative studies of spatial interpolation methods in environmental sciences: Performance and impact factors. Ecol. Informat. 2011, 6, 228–241. [Google Scholar] [CrossRef]
- Pace, D.S.; Miragliuolo, A.; Mussi, B. The case study of the marine Canyon of Cuma (Tyrrhenian Sea, Italy): Implication for cetacean conservation off Ischia Island. In Mediterranean Submarine Canyon, Ecology and Governance; Würtz, M., Ed.; IUCN: Gland, Switzerland, 2012; pp. 89–97. [Google Scholar]
- Taberlet, P.; Coissac, E.; Hajibabaei, M.; Rieseberg, L.H. Environmental DNA. Mol. Ecol. 2012, 21, 1789–1793. [Google Scholar] [CrossRef]
- Deiner, K.; Bik, H.M.; Mächler, E.; Seymour, M.; Lacoursière-Roussel, A.; Altermatt, F.; Creer, S.; Bista, I.; Lodge, D.M.; de Vere, N.; et al. Environmental DNA metabarcoding: Transforming how we survey animal and plant communities. Mol. Ecol. 2017, 26, 5872–5895. [Google Scholar] [CrossRef]
- Bohmann, K.; Evans, A.; Gilbert, M.T.P.; Carvalho, G.R.; Creer, S.; Knapp, M.; Yu, U.W.; De Bruyn, M. Environmental DNA for wildlife biology and biodiversity monitoring. Trends Ecol. Evol. 2014, 29, 358–367. [Google Scholar] [CrossRef]
- Kelly, R.P.; Port, J.A.; Yamahara, K.M.; Martone, R.G.; Lowell, N.; Thomsen, P.F.; Mach, M.; Bennett, M.; Prahler, E.; Caldwell, M.R.; et al. Harnessing DNA to improve environmental management. Science 2014, 344, 1455–1456. [Google Scholar] [CrossRef]
- Handley, L.L. How will the ‘molecular revolution’ contribute to biological recording? Boil. J. Linn. Soc. 2015, 115, 750–766. [Google Scholar] [CrossRef]
- Creer, S.; Deiner, K.; Frey, S.; Porazinska, D.; Taberlet, P.; Thomas, W.K.; Potter, C.; Bik, H.M. The ecologist’s field guide to sequence-based identification of biodiversity. Methods Ecol. Evol. 2016, 7, 1008–1018. [Google Scholar] [CrossRef]
- Stefanni, S.; Stanković, D.; Borme, D.; de Olazabal, A.; Juretić, T.; Pallavicini, A.; Tirelli, V. Multi-marker metabarcoding approach to study mesozooplankton at basin scale. Sci. Rep. 2018, 8, 12085. [Google Scholar] [CrossRef] [PubMed]
- Valentini, A.; Taberlet, P.; Miaud, C.; Civade, R.; Herder, J.; Thomsen, P.F.; Bellemain, E.; Besnard, A.; Coissac, E.; Boyer, F.; et al. Next-generation monitoring of aquatic biodiversity using environmental DNA metabarcoding. Mol. Ecol. 2016, 25, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.P.; Closek, C.J.; O’Donnell, J.L.; Kralj, J.E.; Shelton, A.O.; Samhouri, J.F. Genetic and manual survey methods yield different and complementary views of an ecosystem. Front. Mar. Sci. 2017, 3, 283. [Google Scholar] [CrossRef]
- Ushio, M.; Murakami, H.; Masuda, R.; Sado, T.; Miya, M.; Sakurai, S.; Yamanaka, H.; Minamoto, T.; Kondoh, M. Quantitative monitoring of multispecies fish environmental DNA using high-throughput sequencing. Metabarcoding Metagenom. 2018, 2, e23297. [Google Scholar]
- Pont, D.; Rocle, M.; Valentini, A.; Civade, R.; Jean, P.; Maire, A.; Roset, N.; Schabuss, M.; Zornig, H.; Dejean, T. Environmental DNA reveals quantitative patterns of fish biodiversity in large rivers despite its downstream transportation. Sci. Rep. 2018, 8, 10361. [Google Scholar] [CrossRef]
- Trenkel, V.M.; Vaz, S.; Albouy, C.; Brind’Amour, A.; Laffargue, P.; Romagnan, J.B.; Lorance, P. We can reduce the impact of monitoring on marine living resources. Mar. Ecol. Prog. Ser. 2019, 609, 277–282. [Google Scholar] [CrossRef]
- Manea, E.; Bianchelli, S.; Fanelli, E.; Danovaro, R.; Gissi, E. Towards an Ecosystem-Based Marine Spatial Planning in the deep Mediterranean Sea. Sci. Total Environ. 2020, 715, 136884. [Google Scholar] [CrossRef]
- Fanelli, E.; Bianchelli, S.; Foglini, F.; Canals, M.; Castellan, G.; Güell-Bujons, Q.; Galil, B.; Goren, M.; Evans, J.; Fabri, M.-C.; et al. Guidelines for the protection of the deep Mediterranean Sea. Conserv. Lett. in press.
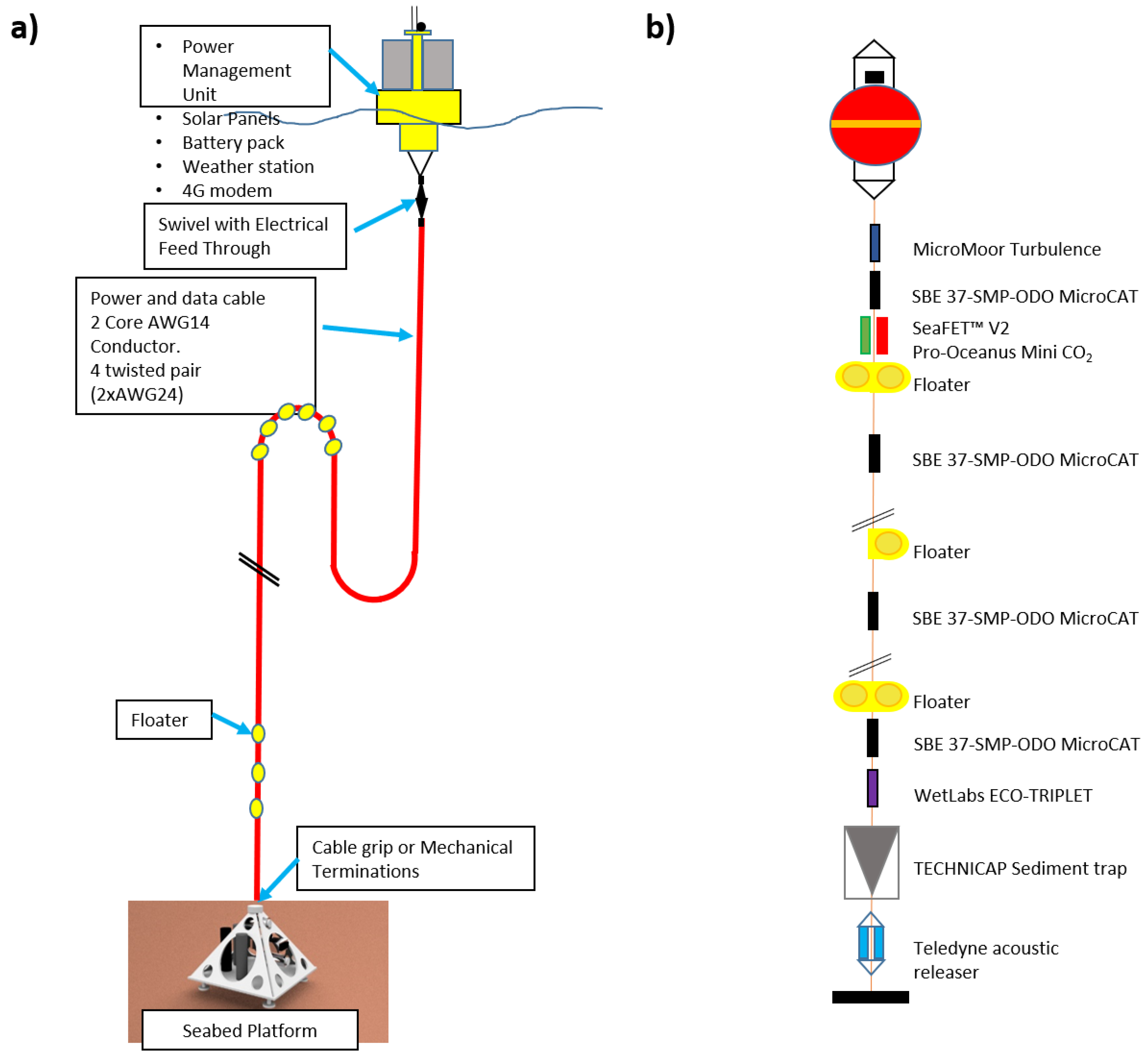

| Mooring Line (ML) | ||
| Num. of Units | Sensor/Probe/Tool | Description |
| 4 | SBE 37-SMP-ODO MicroCAT (positioned at 4 different depths along the vertical) | Integrated measurements of depth, temperature, salinity and dissolved oxygen |
| 1 | MicroMoor Turbulence Measurement Mooring | Measurement of turbulent flow in the marine environment |
| 1 | Pro-Oceanus Mini CO2 | CO2 estimate |
| 1 | SeaFET™ V2 | pH estimate |
| 1 | WetLabs ECO-TRIPLET | Integrated measurements of fluorescence, CDOM (Colored Dissolved Organic Matter) and turbidity (above the sediment trap) |
| 1 | TECHNICAP Sediment trap | Determination of downward mass fluxes of particles and associate elements |
| 2 | Teledyne acoustic releaser | Recovery of mooring |
| Seabed Platform (SP) | ||
| Num. of Units | Sensor/Probe/Tool | Description |
| 1 | Deep SeapHOx™ V2 | Integrated measurements of depth, temperature, salinity, pH and dissolved oxygen |
| 1 | WHLS75-Long Ranger 75 kHz Self-Contained ADCP (Sentinel Configuration) | For gathering detailed data on seasonal and annual current structure fluctuations |
| 1 | WetLabs ECO-TRIPLET | Integrated measurements of fluorescence, CDOM and turbidity |
| 1 | Pro-Oceanus Mini CO2 | CO2 estimate |
| 1 | ARIS Voyager 3000 System | Dual-frequency identification sonar (DIDSON) technology with the release of high-resolution and high definition imaging sonars |
| 1 | LUXUS Colour Zoom Camera II and LUXUS Power LED | 720p video camera |
| 1 | TRIOS RAMSES PAR sensor | Hyperspectral radiometer |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fanelli, E.; Aguzzi, J.; Marini, S.; del Rio, J.; Nogueras, M.; Canese, S.; Stefanni, S.; Danovaro, R.; Conversano, F. Towards Naples Ecological REsearch for Augmented Observatories (NEREA): The NEREA-Fix Module, a Stand-Alone Platform for Long-Term Deep-Sea Ecosystem Monitoring . Sensors 2020, 20, 2911. https://doi.org/10.3390/s20102911
Fanelli E, Aguzzi J, Marini S, del Rio J, Nogueras M, Canese S, Stefanni S, Danovaro R, Conversano F. Towards Naples Ecological REsearch for Augmented Observatories (NEREA): The NEREA-Fix Module, a Stand-Alone Platform for Long-Term Deep-Sea Ecosystem Monitoring . Sensors. 2020; 20(10):2911. https://doi.org/10.3390/s20102911
Chicago/Turabian StyleFanelli, Emanuela, Jacopo Aguzzi, Simone Marini, Joaquin del Rio, Marc Nogueras, Simonepietro Canese, Sergio Stefanni, Roberto Danovaro, and Fabio Conversano. 2020. "Towards Naples Ecological REsearch for Augmented Observatories (NEREA): The NEREA-Fix Module, a Stand-Alone Platform for Long-Term Deep-Sea Ecosystem Monitoring " Sensors 20, no. 10: 2911. https://doi.org/10.3390/s20102911
APA StyleFanelli, E., Aguzzi, J., Marini, S., del Rio, J., Nogueras, M., Canese, S., Stefanni, S., Danovaro, R., & Conversano, F. (2020). Towards Naples Ecological REsearch for Augmented Observatories (NEREA): The NEREA-Fix Module, a Stand-Alone Platform for Long-Term Deep-Sea Ecosystem Monitoring . Sensors, 20(10), 2911. https://doi.org/10.3390/s20102911












