The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors
Abstract
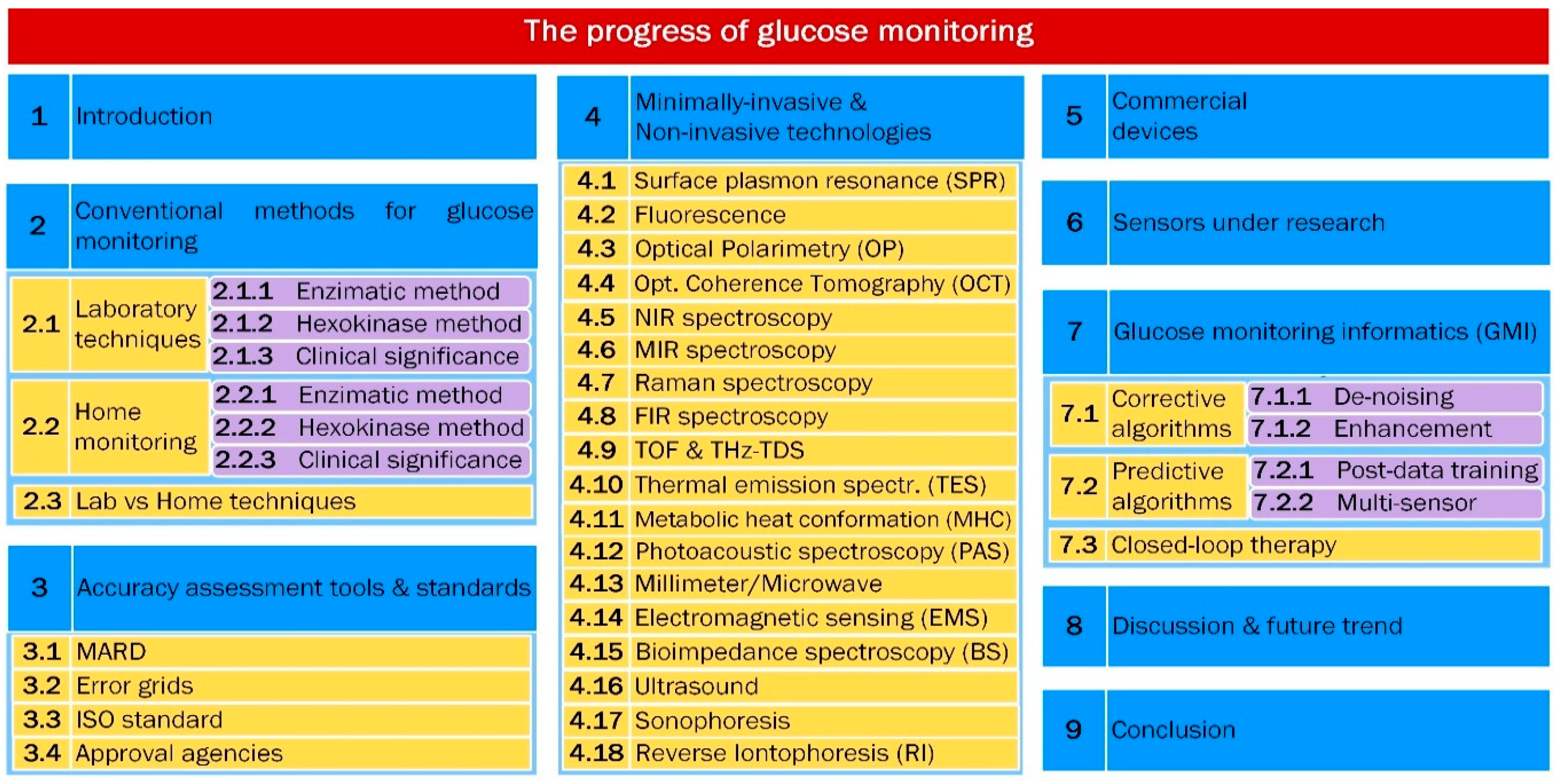
1. Introduction
2. Conventional Methods for Glucose Monitoring
2.1. Laboratory Techniques
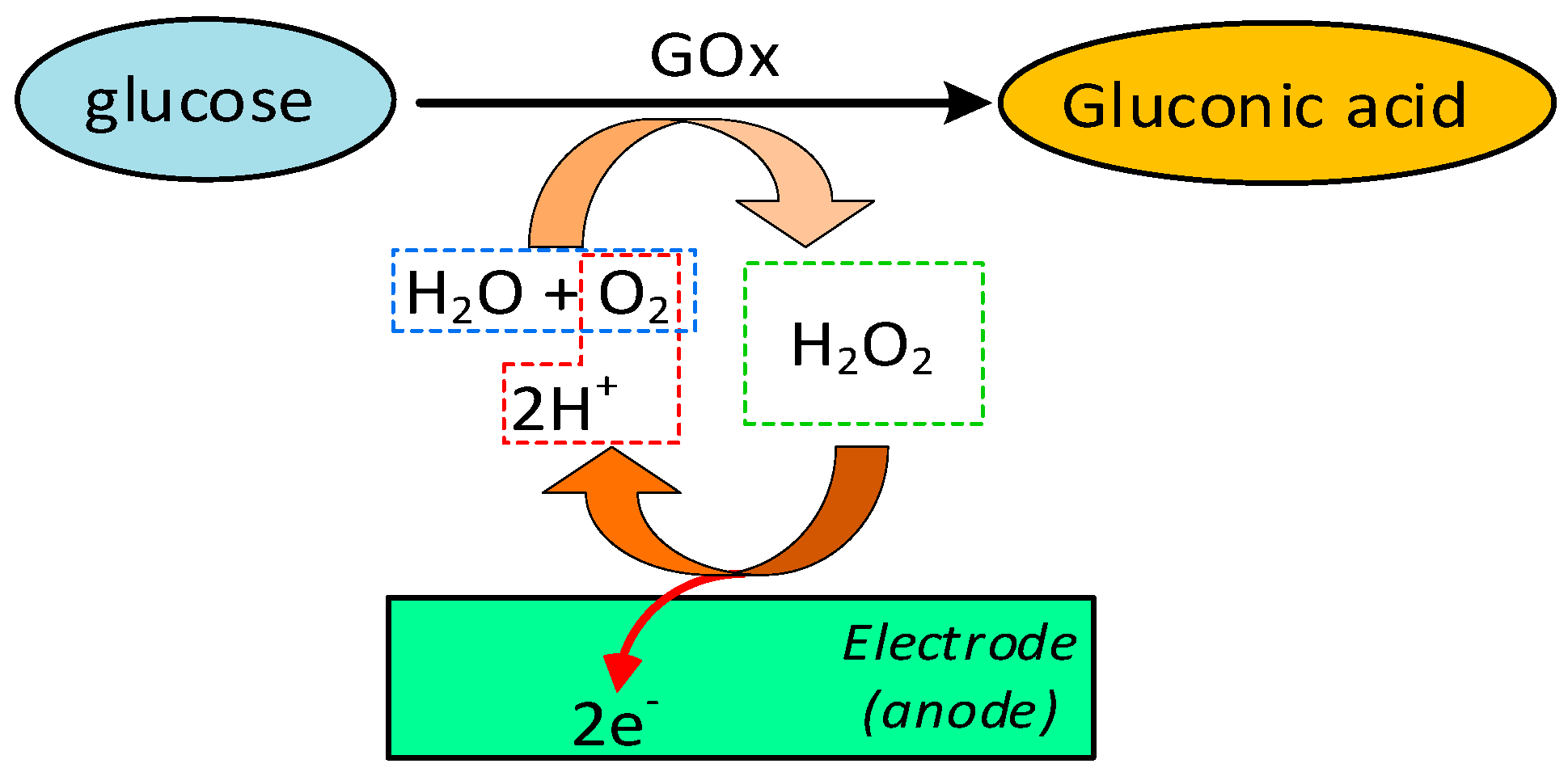
2.1.1. Enzymatic-Amperometric Method
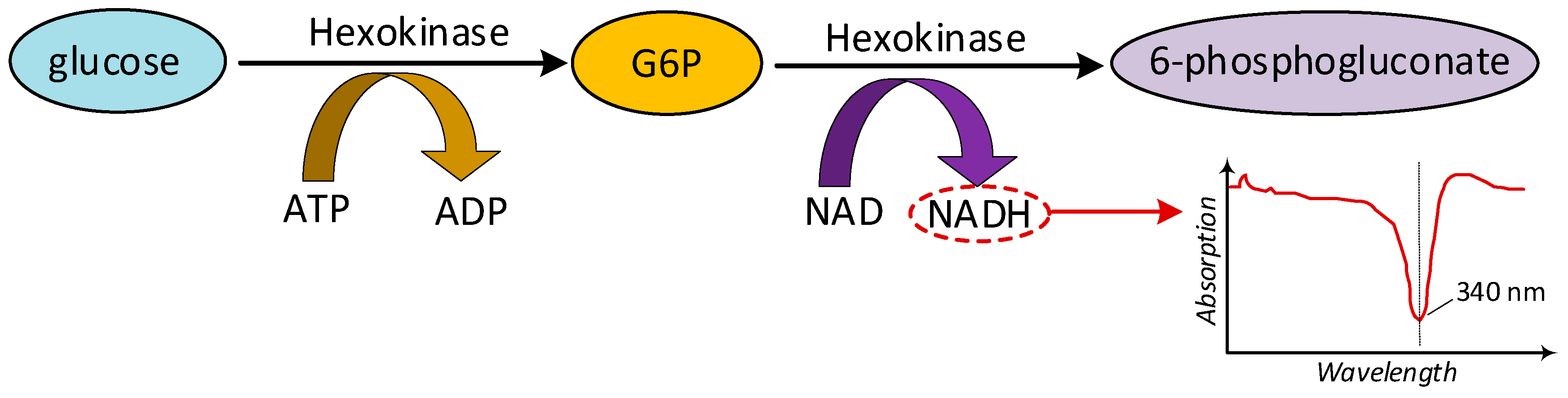
2.1.2. Hexokinase Method
2.1.3. Clinical Significance
2.2. Home-Monitoring Techniques
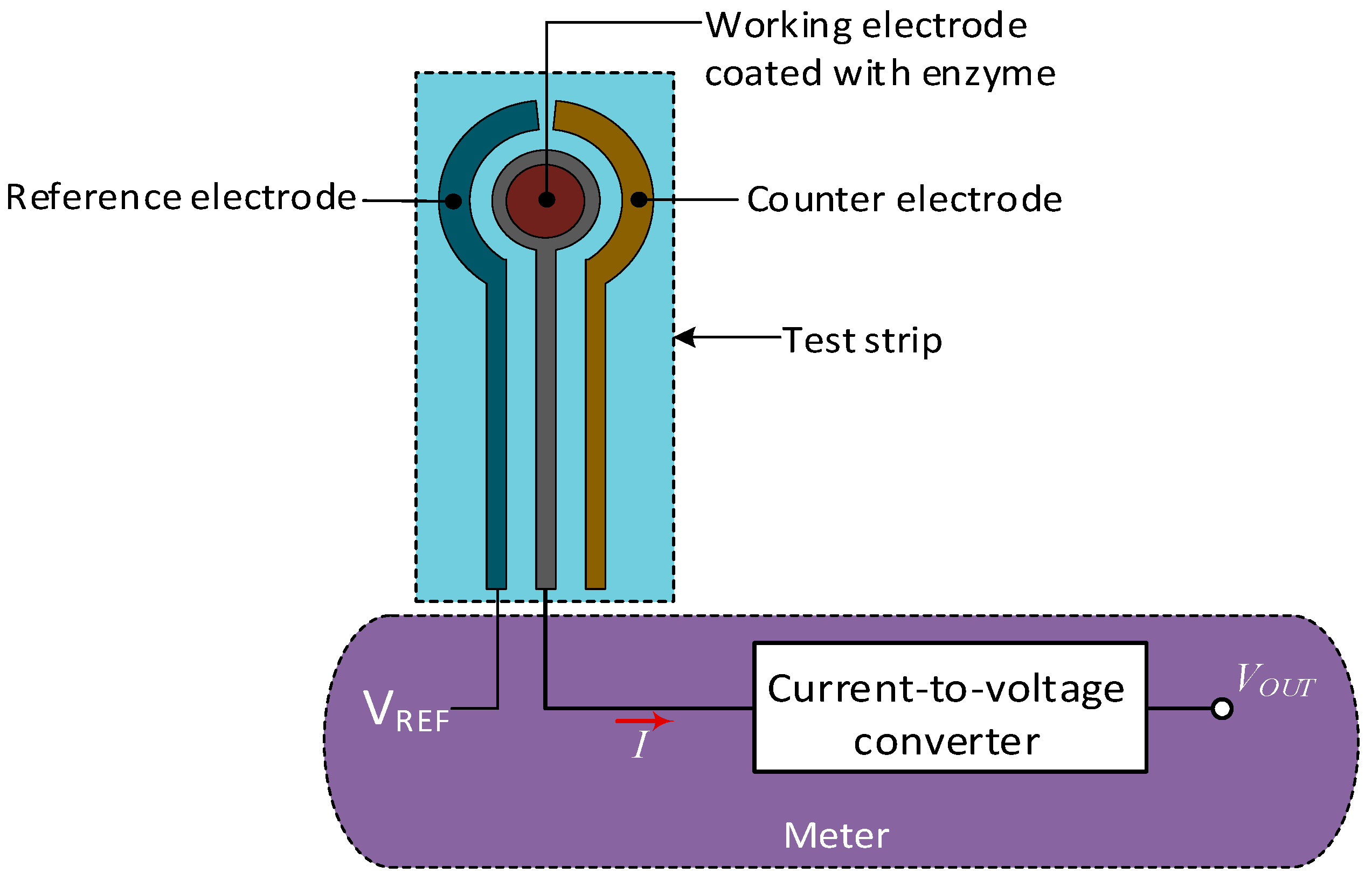
2.2.1. Self-Monitoring Blood Glucose—SMBG
2.2.2. Continuous Glucose Monitoring—CGM
2.2.3. Clinical Significance
2.3. Laboratory Techniques vs Home-Monitoring Techniques
3. Accuracy Assessment Tools and Standards
3.1. Mean Absolute Relative Difference—MARD
3.2. Error Grids–Clarke, Parkes and Surveillance
3.3. ISO 15197 Standard
3.4. Approval Agencies
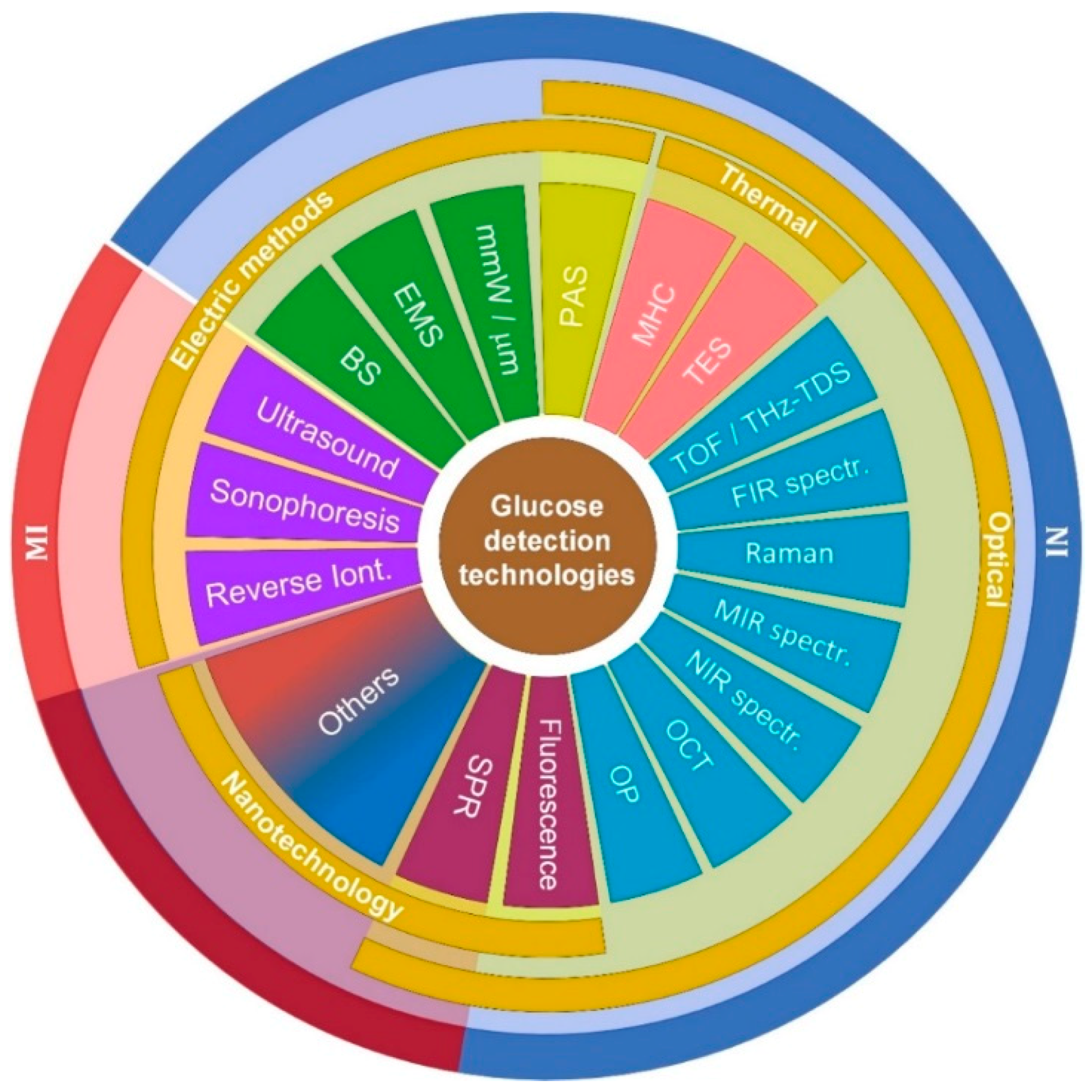
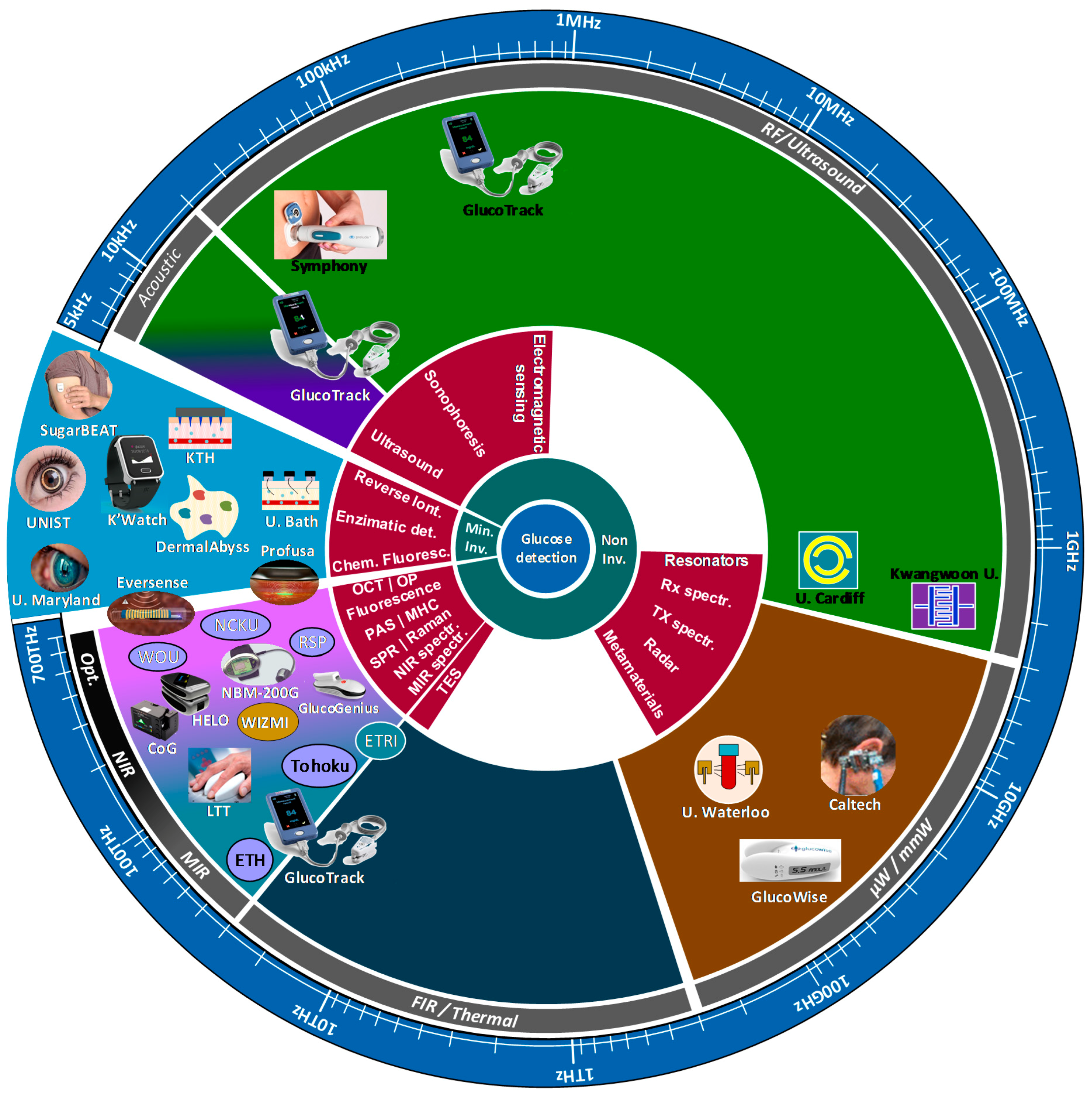
4. Minimally-Invasive and Non-Invasive Technologies
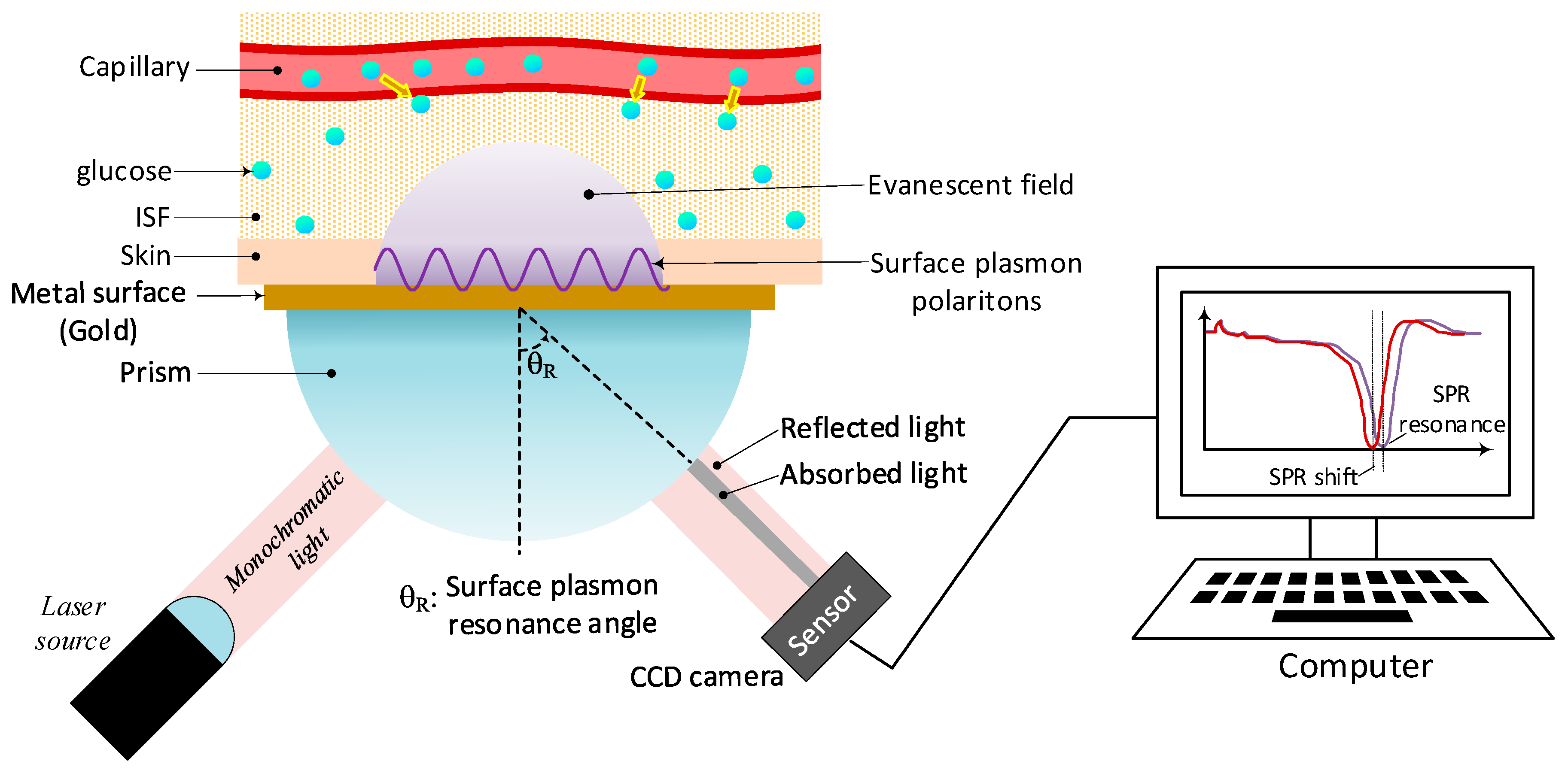
4.1. Surface Plasmon Resonance (SPR)
4.2. Fluorescence
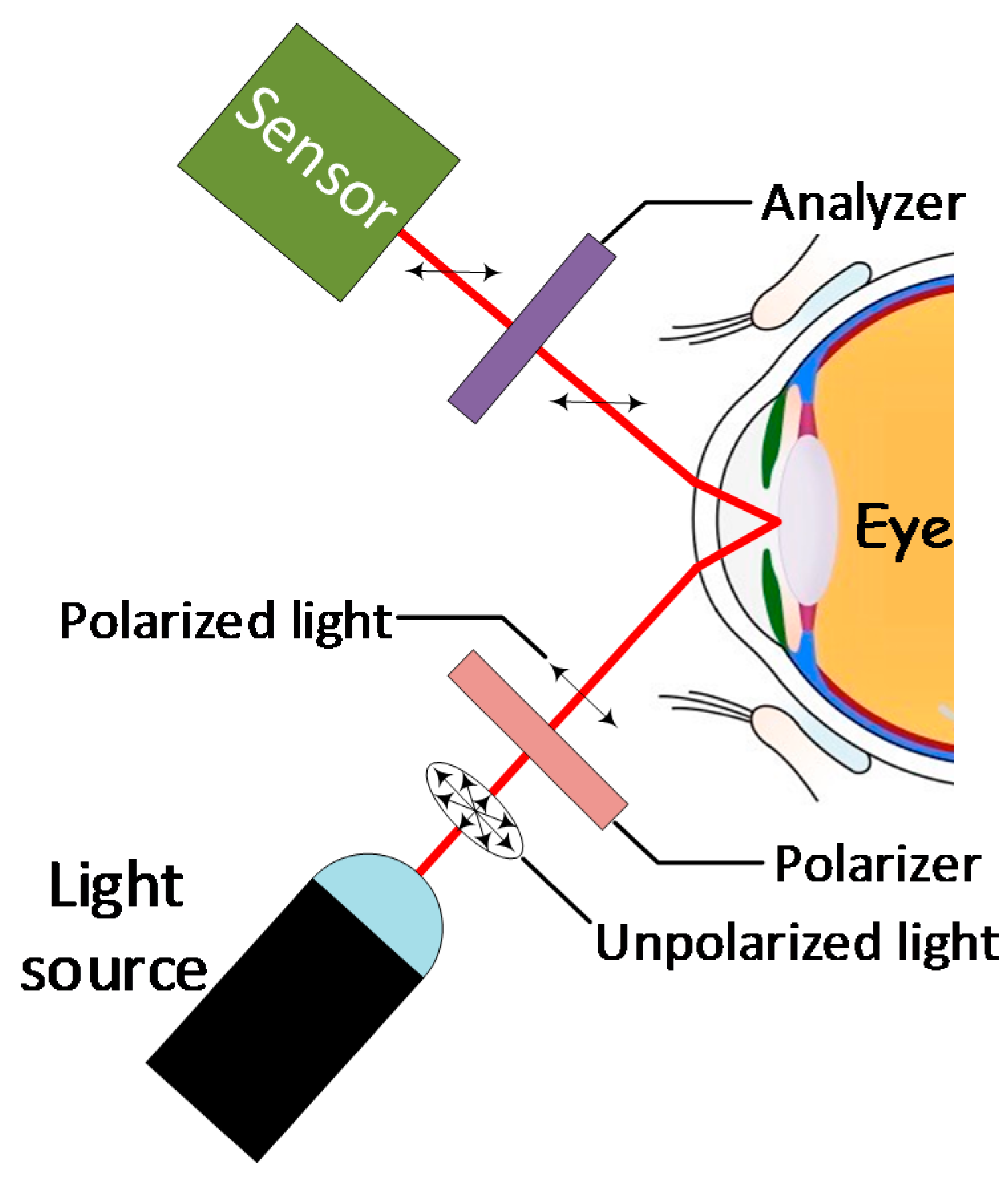
4.3. Optical Polarimetry (OP)
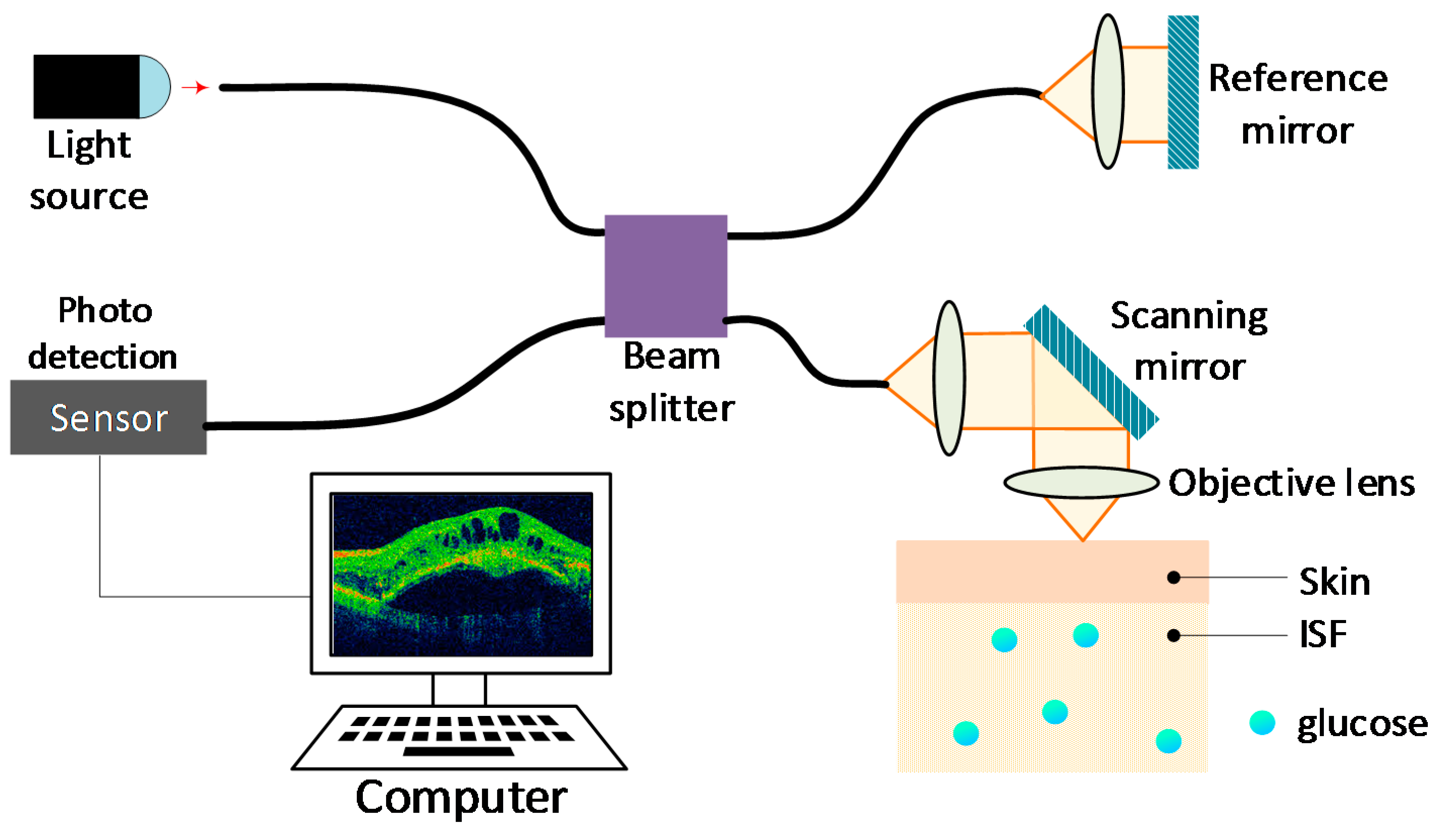
4.4. Optical Coherence Tomography (OCT)
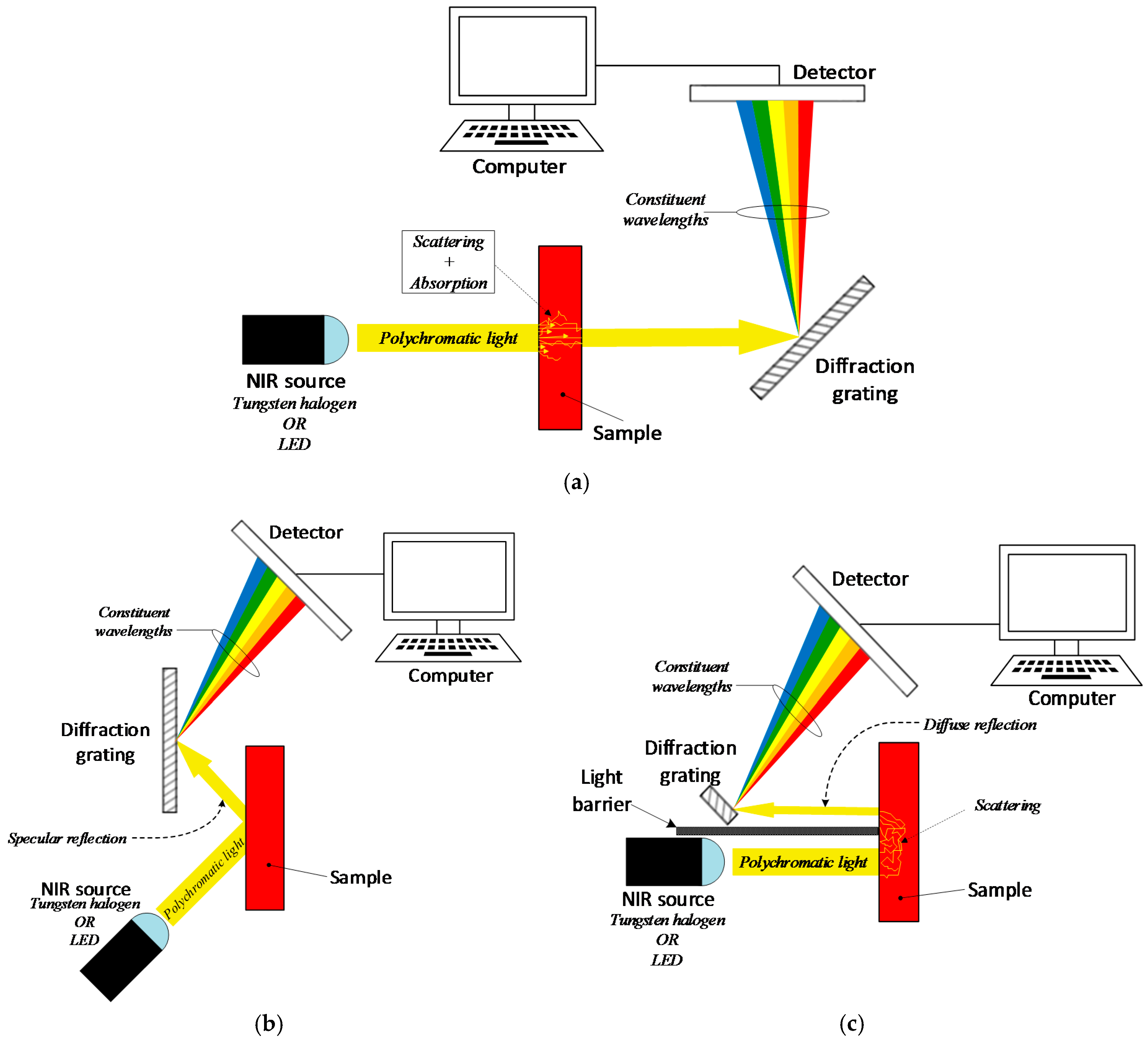
4.5. Near-Infrared Spectroscopy
4.6. Mid-Infrared Spectroscopy
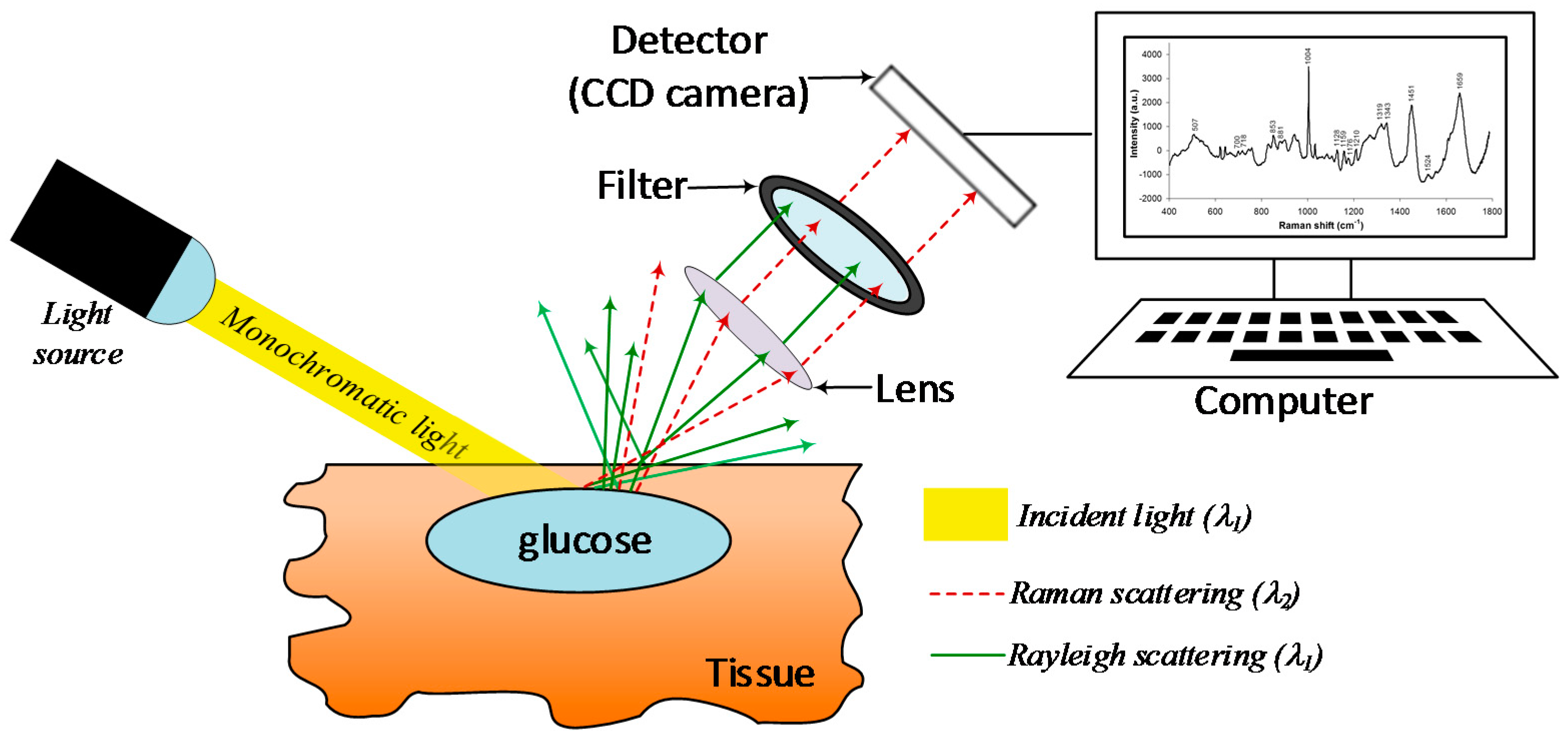
4.7. Raman Spectroscopy
4.8. Far-Infrared Spectroscopy
4.9. Time of Flight (TOF) and Terahertz Time-Domain Spectroscopy (THz-TDS)
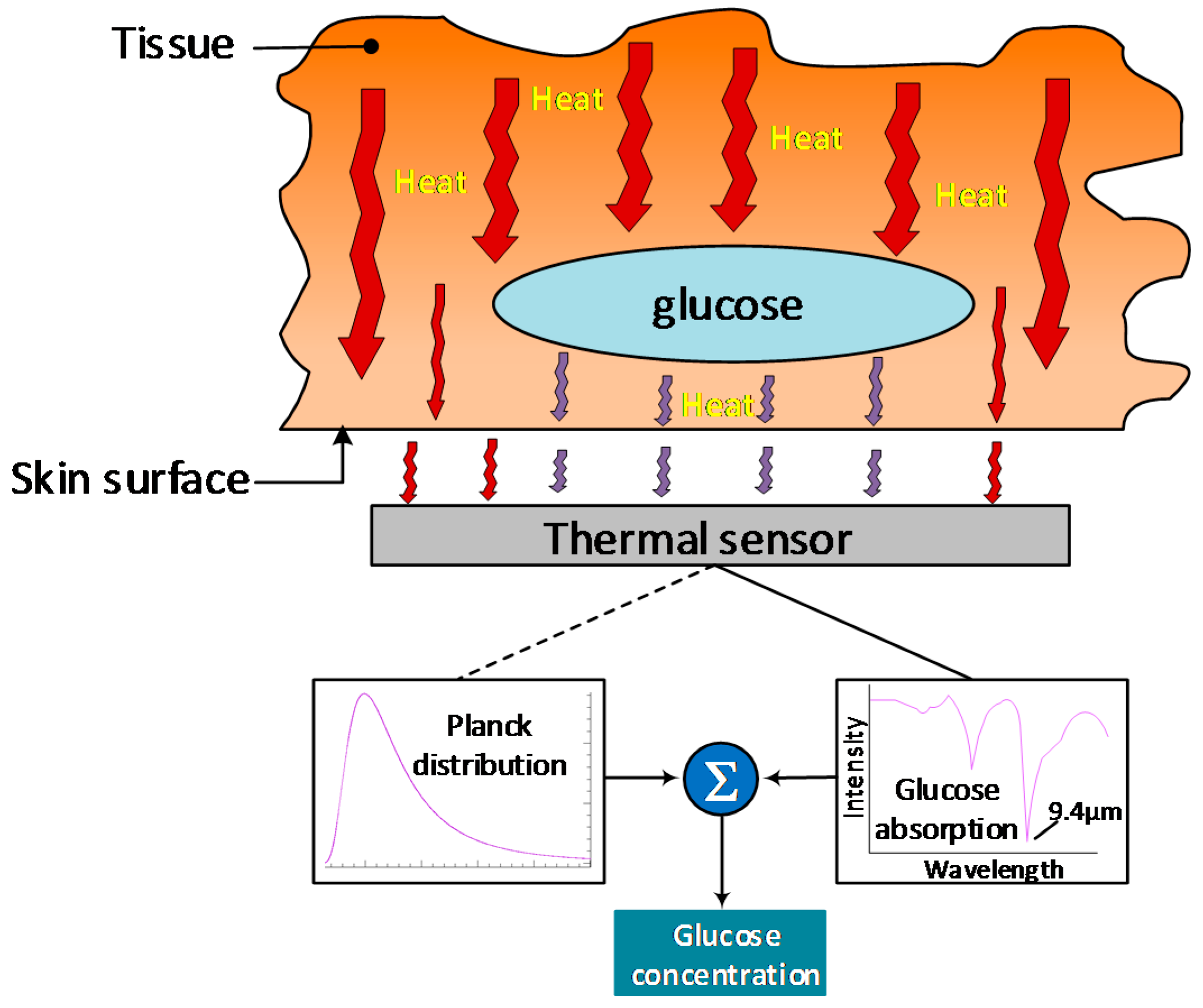
4.10. Thermal Emission Spectroscopy (TES)
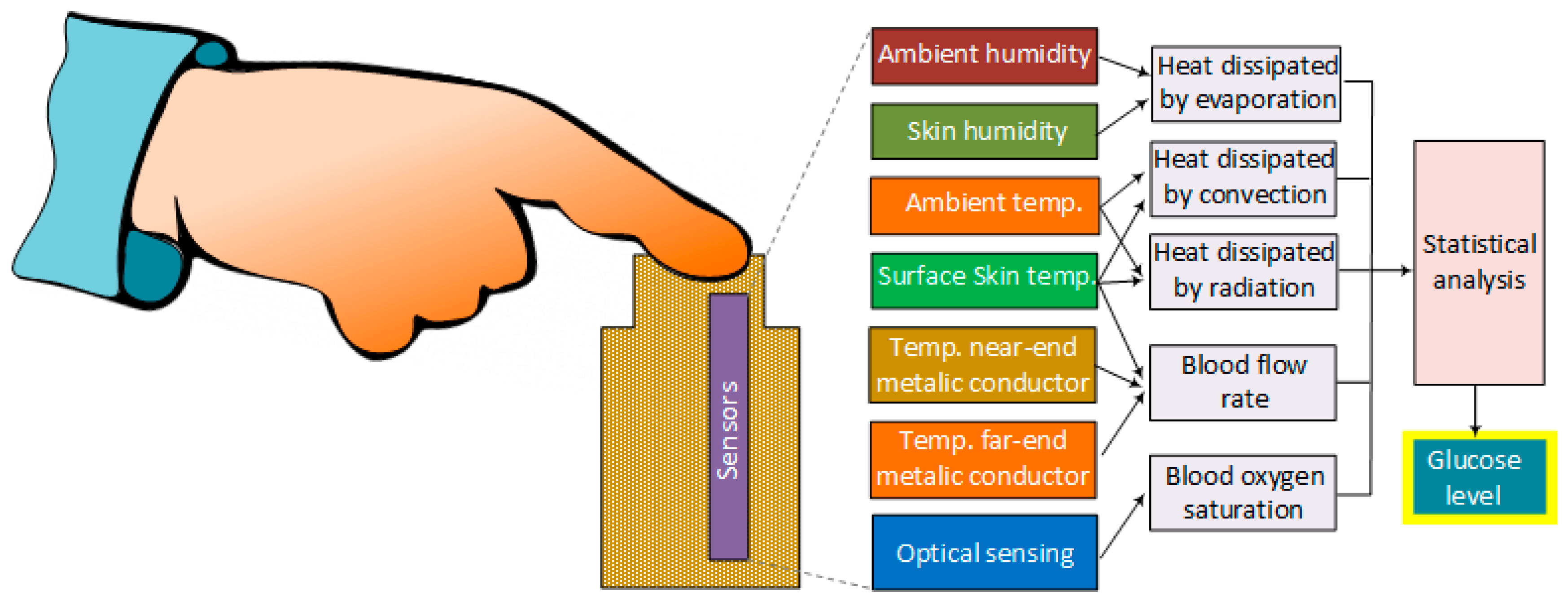
4.11. Metabolic Heat Conformation (MHC)
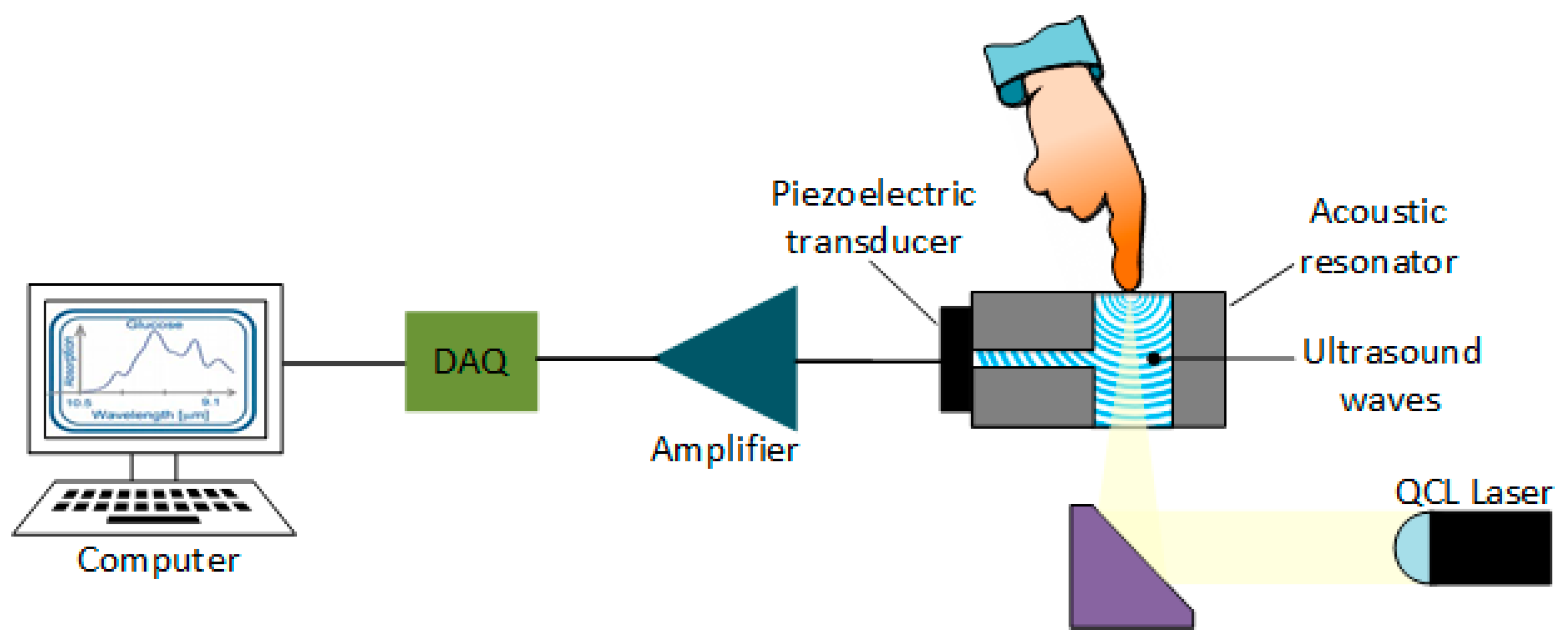
4.12. Photoacoustic Spectroscopy (PAS)
4.13. Millimeter and Microwave Sensing
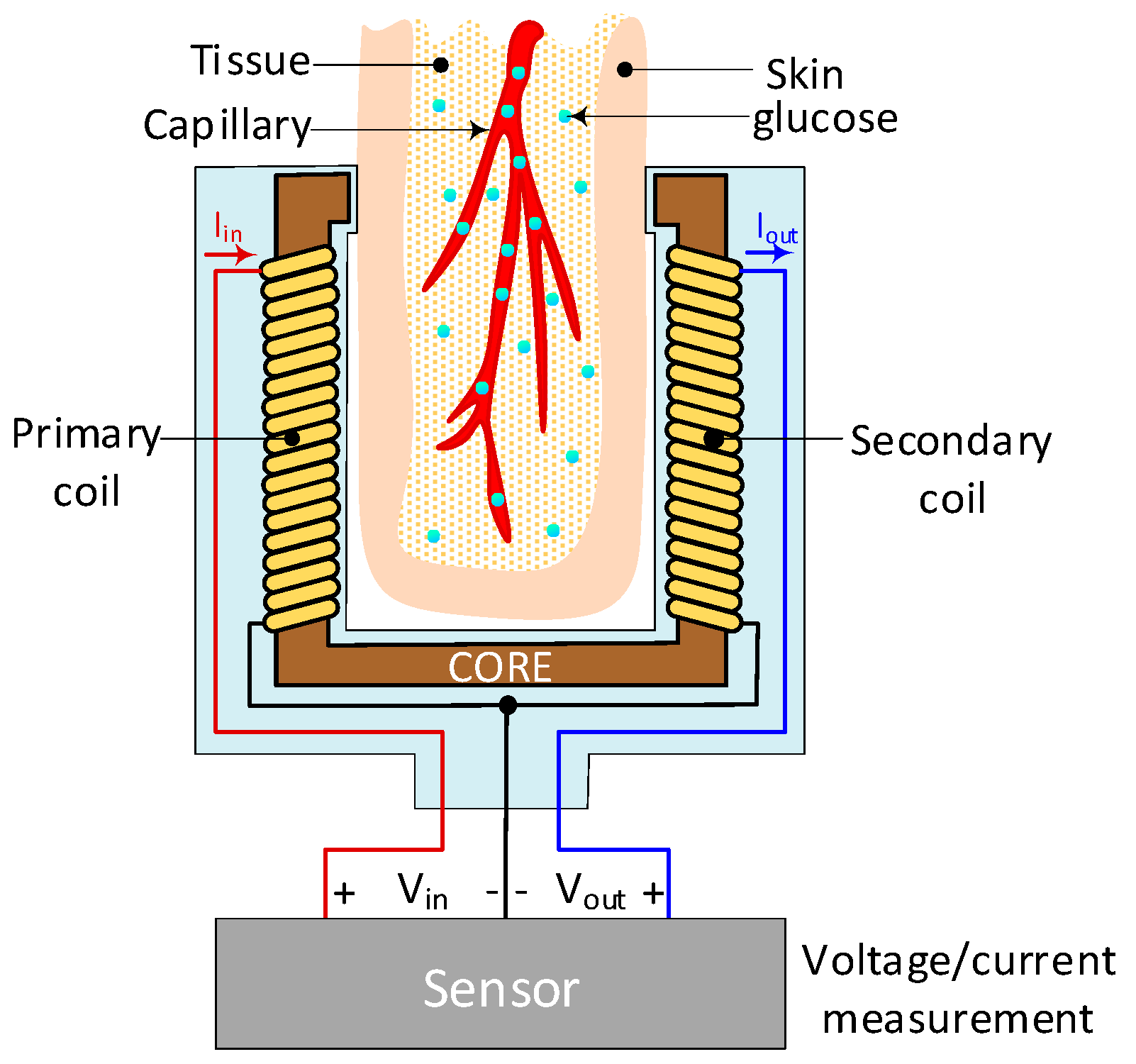
4.14. Electromagnetic Sensing
4.15. Bioimpedance Spectroscopy (BS)
4.16. Ultrasound
4.17. Sonophoresis
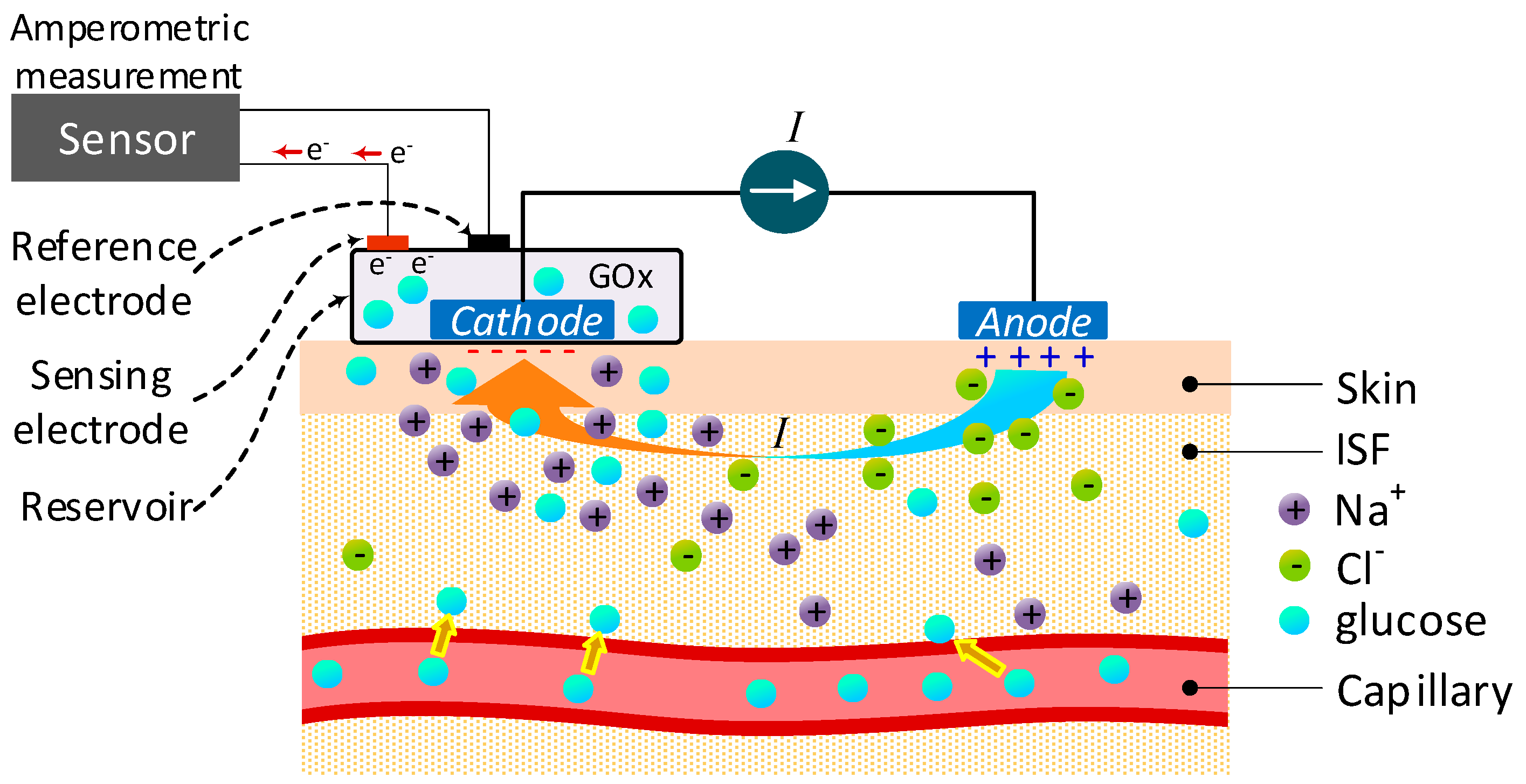
4.18. Reverse Iontophoresis (RI)
5. Commercial Devices
6. Sensors under Research
7. Glucose Monitoring Informatics (GMI)
7.1. Corrective Algorithms
7.1.1. De-Noising
7.1.2. Enhancement
7.2. Predictive Algorithms
7.2.1. Past-Data Training Approach
7.2.2. Multi-Sensor Approach
7.3. Closed-Loop Therapy
8. Discussion and Future Trend
8.1. Past and Present of Non-Invasive Glucose Detection
8.2. Current Research on Non-Invasive (NI) Glucose Monitoring
8.3. Current Research on Minimally-Invasive (MI) Glucose Monitoring
8.4. Considerations for Future Developments in NI and MI Glucose Monitoring
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Diabetes. Available online: http://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 3 October 2018).
- Healthline. The Effects of Low Blood Sugar on Your Body. Available online: https://www.healthline.com/health/low-blood-sugar-effects-on-body#6 (accessed on 2 October 2018).
- Clark, L.C.; Lyons, C. Electrode systems for continuous monitoring in cardiovascular surgery. Ann. N. Y. Acad. Sci. 1962, 102, 29–45. [Google Scholar] [CrossRef] [PubMed]
- So, C.-F.; Choi, K.-S.; Wong, T.K.S.; Chung, J.W.Y. Recent advances in noninvasive glucose monitoring. Med. Dev. (Auckl.) 2012, 5, 45–52. [Google Scholar] [CrossRef]
- Uwadaira, Y.; Ikehata, A. Noninvasive Blood Glucose Measurement. In Nutritional and Therapeutic Interventions for Diabetes and Metabolic Syndrome, 2nd ed.; Bagchi, D., Nair, S., Eds.; Academic Press: San Diego, CA, USA, 2018; pp. 489–504. [Google Scholar]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Thompson, T.J.; Cheng, Y.J.; Zhuo, X.; Zhang, P.; Gregg, E.; Rolka, D.B. Projection of the future diabetes burden in the United States through 2060. Popul. Health Metr. 2018, 16, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tura, A.; Sbrignadello, S.; Cianciavicchia, D.; Pacini, G.; Ravazzani, P. A Low Frequency Electromagnetic Sensor for Indirect Measurement of Glucose Concentration: In Vitro Experiments in Different Conductive Solutions. Sensors 2010, 10, 5346–5358. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhao, X.-L.; Li, Z.-H.; Zhu, Z.-G.; Qian, S.-H.; Flewitt, A.J. Current and Emerging Technology for Continuous Glucose Monitoring. Sensors 2017, 17, 182. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Gal, A.; Mayzel, Y.; Horman, K.; Bahartan, K. Non-invasive Glucose Monitoring: A Review of Challenges and Recent Advances. Curr. Trends Biomed. Eng. Biosci. 2017, 6, 1–8. [Google Scholar] [CrossRef]
- Van Enter, B.J.; von Hauff, E. Challenges and perspectives in continuous glucose monitoring. Chem. Commun. 2018, 54, 5032–5045. [Google Scholar] [CrossRef]
- Khalil, O.S. Spectroscopic and Clinical Aspects of Noninvasive Glucose Measurements. Clin. Chem. 1999, 45, 165. [Google Scholar]
- McMillin, J.M. Blood Glucose. In Clinical Methods: The History, Physical and Laboratory Examinations, 3rd ed.; Walker, H.K., Hall, W.D., Hurst, J.W., Eds.; Butterworth-Heinemann: Boston, MA, USA, 1990. [Google Scholar]
- YSI Incorporated. YSI 2700 SELECT Biochemistry Analyzer User’s Manual; YSI Incorporated: Yellow Springs, OH, USA, 2000. [Google Scholar]
- YSI Incorporated. 2950D Biochemistry Analyzer. Available online: https://www.ysi.com/ysi-2950-biochemistry-analyzer (accessed on 4 October 2018).
- EKF-Diagnostic GmbH. Biosen C-Line & Biosen S-Line. Available online: https://www.ekfdiagnostics.com/res/BS%20Data%20EN%20EU%205.1-02.17.pdf (accessed on 4 October 2018).
- Beckman Coulter. Chemistry Information Sheet. Available online: https://www.beckmancoulter.com/wsrportal/techdocs?docname=/cis/B31851/%25%25/EN_GLUH.pdf (accessed on 4 October 2018).
- U.S. Food & Drug Administration—FDA. Review Memorandum—Quantitative Enzymatic Assay Based on Hexokinase/G-6-PDH Methodology. Available online: https://www.accessdata.fda.gov/cdrh_docs/reviews/K060383.pdf (accessed on 4 October 2018).
- Roche Diagnostics USA. Glucose HK Gen.3. Available online: https://usdiagnostics.roche.com/products/05168791190/PARAM49/overlay.html (accessed on 4 October 2018).
- Delost, M.E. Blood Gas and Critical Care Analyte Analysis. In Equipment for Respiratory Care; Volsko, T.A., Chatburn, R.L., El-Khatib, M.F., Eds.; Jones and Bartlett Publishers, Inc.: Sudbury, MA, USA, 2014. [Google Scholar]
- Burrin, J.M.; Price, C.P. Measurement of Blood Glucose. Ann. Clin. Biochem. 1985, 22, 327–342. [Google Scholar] [CrossRef]
- Liang, Y.M.D.; Wanderer, J.M.D.M.; Nichols, J.H.P.; Klonoff, D.M.D.F.; Rice, M.J.M.D. Blood Gas Analyzer Accuracy of Glucose Measurements. Mayo Clin. Proc. 2017, 92, 1030–1041. [Google Scholar] [CrossRef] [PubMed]
- Inoue, S.; Egi, M.; Kotani, J.; Morita, K. Accuracy of blood-glucose measurements using glucose meters and arterial blood gas analyzers in critically ill adult patients: Systematic review. Crit. Care 2013, 17, R48. [Google Scholar] [CrossRef] [PubMed]
- Dalvi, N. Glucose meter reference design. In Application Note Nr. 1560; Microchip Technology Inc.: Chandler, AZ, USA, 2013. [Google Scholar]
- Rebel, A.; Rice, M.A.; Fahy, B.G. The Accuracy of Point-of-Care Glucose Measurements. J. Diabetes Sci. Technol. 2012, 6, 396–411. [Google Scholar] [CrossRef]
- Ekhlaspour, L.; Mondesir, D.; Lautsch, N.; Balliro, C.; Hillard, M.; Magyar, K.; Radocchia, L.G.; Esmaeili, A.; Sinha, M.; Russell, S.J. Comparative Accuracy of 17 Point-of-Care Glucose Meters. J. Diabetes Sci. Technol. 2016, 11, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.P.; Patra, S.; Bhattacharjee, R.; Chowdhury, S. Erroneously elevated glucose values due to maltose interference in mutant glucose dehydrogenase pyrroloquinolinequinone (mutant GDH-PQQ) based glucometer. BMJ Case Rep. 2017. [Google Scholar] [CrossRef] [PubMed]
- Schultz, D.G. FDA Public Health Notification: Potentially Fatal Errors with GDH-PQQ* Glucose Monitoring Technology. Available online: http://labmed.ucsf.edu/labmanual/db/resource/FDA_glucometer_warning_Aug_2009.pdf (accessed on 24 January 2019).
- Diabetes Australia. Continuous Glucose Monitoring. Available online: https://static.diabetesaustralia.com.au/s/fileassets/diabetes-australia/e2feb45e-ebc4-4133-85e1-b514e67d24de.pdf (accessed on 3 October 2018).
- Wadwa, R.P.; Fiallo-Scharer, R.; VanderWel, B.; Messer, L.H.; Cobry, E.; Chase, H.P. Continuous Glucose Monitoring in Youth with Type 1 Diabetes. Diabetes Technol. Ther. 2009, 11, S83–S91. [Google Scholar] [CrossRef] [PubMed]
- Patton, S.R.; Clements, M.A. Continuous Glucose Monitoring Versus Self-monitoring of Blood Glucose in Children with Type 1 Diabetes- Are there Pros and Cons for Both? US Endocrinol. 2012, 8, 27–29. [Google Scholar] [CrossRef]
- Nardacci, E.A.; Bode, B.W.; Hirsch, I.B. Individualizing Care for the Many. Diabetes Educ. 2010, 36, S4–S19. [Google Scholar] [CrossRef]
- Ward, J.E.F.; Stetson, B.A.; Mokshagundam, S.P.L. Patient perspectives on self-monitoring of blood glucose: perceived recommendations, behaviors and barriers in a clinic sample of adults with type 2 diabetes. J. Diabetes Metab. Disord. 2015, 14, 43. [Google Scholar] [CrossRef]
- Danne, T.; Nimri, R.; Battelino, T.; Bergenstal, R.M.; Close, K.L.; DeVries, J.H.; Garg, S.; Heinemann, L.; Hirsch, I.; Amiel, S.A.; et al. International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care 2017, 40, 1631–1640. [Google Scholar] [CrossRef]
- Reiterer, F.; Polterauer, P.; Schoemaker, M.; Schmelzeisen-Redecker, G.; Freckmann, G.; Heinemann, L.; del Re, L. Significance and Reliability of MARD for the Accuracy of CGM Systems. J. Diabetes Sci. Technol. 2017, 11, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.S. Clinical Implications of Accuracy Measurements of Continuous Glucose Sensors. Diabetes Technol. Ther. 2017, 19, S51–S54. [Google Scholar] [CrossRef] [PubMed]
- Boren, S.A.; Clarke, W.L. Analytical and Clinical Performance of Blood Glucose Monitors. J. Diabetes Sci. Technol. 2010, 4, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C.; Lias, C.; Vigersky, R.; Clarke, W.; Parkes, J.L.; Sacks, D.B.; Kirkman, M.S.; Kovatchev, B. The Surveillance Error Grid. J. Diabetes Sci. Technol. 2014, 8, 658–672. [Google Scholar] [CrossRef]
- Klonoff, D.C. The Need for Clinical Accuracy Guidelines for Blood Glucose Monitors. J. Diabetes Sci. Technol. 2012, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Clarke, W.L.; Cox, D.; Gonder-Frederick, L.A.; Carter, W.; Pohl, S.L. Evaluating Clinical Accuracy of Systems for Self-Monitoring of Blood Glucose. Diabetes Care 1987, 10, 622–628. [Google Scholar] [CrossRef]
- Parkes, J.L.; Slatin, S.L.; Pardo, S.; Ginsberg, B.H. A new consensus error grid to evaluate the clinical significance of inaccuracies in the measurement of blood glucose. Diabetes Care 2000, 23, 1143–1148. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Available online: https://www.iso.org (accessed on 26 September 2018).
- International Organization for Standardization (ISO). ISO 15197:2013. In Vitro Diagnostic Test Systems—Requirements for Blood-Glucose Monitoring Systems for Self-Testing in Managing Diabetes Mellitus; International Organization for Standardization (ISO): Geneva, Switzerland, 2013. [Google Scholar]
- Freckmann, G.; Baumstark, A.; Jendrike, N.; Rittmeyer, D.; Pleus, S.; Haug, C. Accuracy Evaluation of Four Blood Glucose Monitoring Systems in the Hands of Intended Users and Trained Personnel Based on ISO 15197 Requirements. Diabetes Technol. Ther. 2017, 19, 246–254. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). International Organization for Standardization (ISO). In vitro diagnostic test systems—Requirements for blood-glucose monitoring systems for self-testing in managing diabetes mellitus (ISO 15197:2013). In EN ISO 15197:2015; International Organization for Standardization (ISO): Geneva, Switzerland, 2015. [Google Scholar]
- Freckmann, G.; Schmid, C.; Baumstark, A.; Rutschmann, M.; Haug, C.; Heinemann, L. Analytical Performance Requirements for Systems for Self-Monitoring of Blood Glucose With Focus on System Accuracy: Relevant Differences among ISO 15197:2003, ISO 15197:2013, and Current FDA Recommendations. J. Diabetes Sci. Technol. 2015, 9, 885–894. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration (FDA). Blood Glucose Monitoring Test Systems for Prescription Point-of-Care Use; U.S. Food & Drug Administration (FDA): Silver Spring, MD, USA, 2016.
- U.S. Food & Drug Administration (FDA). Self-Monitoring Blood Glucose Test Systems for over-the-Counter Use; U.S. Food & Drug Administration (FDA): Silver Spring, MD, USA, 2016.
- European Commission. In vitro Diagnostic Medical Devices. Available online: http://ec.europa.eu/growth/single-market/european-standards/harmonised-standards/iv-diagnostic-medical-devices/#Note%202.1 (accessed on 12 September 2018).
- Government of Canada. New Requirements for Medical Device Licence Applications for Lancing Devices and Blood Glucose Monitoring Systems. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/medical-devices/activities/announcements/notice-new-requirements-medical-device-licence-applications-lancing-devices-blood-glucose-monitoring-systems.html (accessed on 12 September 2018).
- Agência Nacional de Vigilância Sanitária (ANVISA). Instrução Normativa Nº 24; Agência Nacional de Vigilância Sanitária (ANVISA): Brasília, Brazil, 2018.
- China Food & Drug Administration (CFDA). Glucometer Registration Technical Review Guidelines; Chemical Inspection and Regulation Service (CIRS): Beijing, China, 2016.
- Pharmaceuticals and Medical Devices Agency (PMDA). Handling of Self-Testing Blood Glucose Meters. Available online: http://www.std.pmda.go.jp/stdDB/Data/MDStd/CerStd/Notif/K1100009_01_2016_en.pdf (accessed on 2 October 2018).
- Pharmaceuticals and Medical Devices Agency (PMDA). List of Certification Standards; Pharmaceuticals and Medical Devices Agency (PMDA): Tokyo, Japan, 2018.
- Department of Therapeutic Goods Administration (TGA). Australian Regulatory Guidelines for Medical Devices (ARGMD). Available online: https://www.tga.gov.au/publication/australian-regulatory-guidelines-medical-devices-argmd (accessed on 5 October 2018).
- Standards Australia. ISO 15197:2013. Available online: https://www.standards.org.au/standards-catalogue/international/iso-slash-tc--212/iso--15197-colon-2013 (accessed on 5 October 2018).
- Department of Therapeutic Goods Administration (TGA). Medical Devices Regulation: An Introduction. Available online: http://www.tga.gov.au/sme-assist/medical-devices-regulation-introduction (accessed on 5 October 2018).
- MIT. Carbon Nanotube Sensor Detects Glucose in Saliva. Available online: https://www.technologyreview.com/s/514456/carbon-nanotube-sensor-detects-glucose-in-saliva/ (accessed on 23 November 2018).
- Jia, J.; Guan, W.; Sim, M.; Li, Y.; Li, H. Carbon Nanotubes Based Glucose Needle-type Biosensor. Sensors 2008, 8, 1712–1718. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, Z.; Wang, X. Chapter 5–Carbon Nanotubes for Sensing Applications. In Industrial Applications of Carbon Nanotubes; Peng, H., Li, Q., Chen, T., Eds.; Elsevier: Boston, MA, USA, 2017; pp. 129–150. [Google Scholar] [CrossRef]
- Aslan, K.; Lakowicz, J.R.; Geddes, C.D. Plasmonic Glucose Sensing. In Glucose Sensing; Geddes, C.D., Lakowicz, J.R., Eds.; Springer US: Boston, MA, USA, 2006; pp. 259–282. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, M.L. Saliva Glucose Monitoring System. U.S. patent 2013. [Google Scholar]
- Li, D.C.; Wu, J.W.; Wu, P.; Lin, Y.; Sun, Y.J.; Zhu, R.; Yang, J.; Xu, K.X. Glucose measurement using surface plasmon resonance sensor with affinity based surface modification by borate polymer. In Proceedings of the 2015 Transducers-2015 18th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Anchorage, AK, USA, 21–25 June 2015; pp. 1569–1572. [Google Scholar]
- Srivastava, S.K.; Verma, R.; Gupta, B.D. Surface plasmon resonance based fiber optic glucose biosensor. In Proceedings of the Third Asia Pacific Optical Sensors Conference, Sydney, Australia, 30 January 2012; p. 83511Z. [Google Scholar] [CrossRef]
- Li, D.; Su, J.; Yang, J.; Yu, S.; Zhang, J.; Xu, K.; Yu, H. Optical surface plasmon resonance sensor modified by mutant glucose/galactose-binding protein for affinity detection of glucose molecules. Biomed. Opt. Express 2017, 8, 5206–5217. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Baillargeat, D.; Ho, H.-P.; Yong, K.-T. Nanomaterials enhanced surface plasmon resonance for biological and chemical sensing applications. Chem. Soc. Rev. 2014, 43, 3426–3452. [Google Scholar] [CrossRef] [PubMed]
- McShane, M.; Stein, E. Fluorescence-Based Glucose Sensors. In In Vivo Glucose Sensing; Cunningham, D.D., Stenken, J.A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; Volume 174, pp. 269–316. [Google Scholar]
- Barone, P.W.; Parker, R.S.; Strano, M.S. In Vivo Fluorescence Detection of Glucose Using a Single-Walled Carbon Nanotube Optical Sensor: Design, Fluorophore Properties, Advantages, and Disadvantages. Anal. Chem. 2005, 77, 7556–7562. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C. Overview of Fluorescence Glucose Sensing: A Technology with a Bright Future. J. Diabetes Sci. Technol. 2012, 6, 1242–1250. [Google Scholar] [CrossRef]
- Barone, P.W.; Strano, M.S. Single Walled Carbon Nanotubes as Reporters for the Optical Detection of Glucose. J. Diabetes Sci. Technol. 2009, 3, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hwang, E.; Zhang, J. Fluorescent Nanobiosensors for Sensing Glucose. Sensors 2018, 18, 1440. [Google Scholar] [CrossRef] [PubMed]
- Szmacinski, H.; Lakowicz, J.R. Fluorescence lifetime-based sensing and imaging. Sens. Actuators B Chem. 1995, 29, 16–24. [Google Scholar] [CrossRef]
- Bambot, S.B.; Rao, G.; Romauld, M.; Carter, G.M.; Sipior, J.; Terpetchnig, E.; Lakowicz, J.R. Sensing oxygen through skin using a red diode laser and fluorescence lifetimes. Biosens. Bioelectron. 1995, 10, 643–652. [Google Scholar] [CrossRef]
- DiCesare, N.; Lakowicz, J.R. Evaluation of two synthetic glucose probes for fluorescence-lifetime-based sensing. Anal. Biochem. 2001, 294, 154–160. [Google Scholar] [CrossRef]
- Malik, B.H.; Coté, G.L. Real-time, closed-loop dual-wavelength optical polarimetry for glucose monitoring. J. Biomed. Opt. 2010, 15, 017002. [Google Scholar] [CrossRef] [PubMed]
- Rawer, R.; Stork, W.; Kreiner, C.F. Non-invasive polarimetric measurement of glucose concentration in the anterior chamber of the eye. Graefe’s Arch. Clin. Exp. Ophthalmol. 2004, 242, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Fercher, A.F.; Drexler, W.; Hitzenberger, C.K.; Lasser, T. Optical coherence tomography - principles and applications. Rep. Prog. Phys. 2003, 66, 239. [Google Scholar] [CrossRef]
- Lan, Y.T.; Kuang, Y.P.; Zhou, L.P.; Wu, G.Y.; Gu, P.C.; Wei, H.J.; Chen, K. Noninvasive monitoring of blood glucose concentration in diabetic patients with optical coherence tomography. Laser Phys. Lett. 2017, 14, 035603. [Google Scholar] [CrossRef]
- Agelet, L.E.; Hurburgh, C.R. A Tutorial on Near Infrared Spectroscopy and Its Calibration. Crit. Rev. Anal. Chem. 2010, 40, 246–260. [Google Scholar] [CrossRef]
- Schaare, P.N.; Fraser, D.G. Comparison of reflectance, interactance and transmission modes of visible-near infrared spectroscopy for measuring internal properties of kiwifruit (Actinidia chinensis). Postharvest Biol. Tech. 2000, 20, 175–184. [Google Scholar] [CrossRef]
- Nicolaï, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive measurement of fruit and vegetable quality by means of NIR spectroscopy: A review. Postharvest Biol. Tech. 2007, 46, 99–118. [Google Scholar] [CrossRef]
- Oliver, N.S.; Toumazou, C.; Cass, A.E.G.; Johnston, D.G. Glucose sensors: a review of current and emerging technology. Diabet. Med. 2009, 26, 197–210. [Google Scholar] [CrossRef]
- Maruo, K.; Oota, T.; Tsurugi, M.; Nakagawa, T.; Arimoto, H.; Tamura, M.; Ozaki, Y.; Yamada, Y. New Methodology to Obtain a Calibration Model for Noninvasive Near-Infrared Blood Glucose Monitoring. Appl. Spectr. 2006, 60, 441–449. [Google Scholar] [CrossRef]
- Khalil, O.S. Non-Invasive Glucose Measurement Technologies: An Update from 1999 to the Dawn of the New Millennium. Diabetes Technol. Ther. 2004, 6, 660–697. [Google Scholar] [CrossRef]
- Coates, J. Vibrational Spectroscopy: Instrumentation for Infrared and Raman Spectroscopy. Appl. Spectr. Rev. 1998, 33, 267–425. [Google Scholar] [CrossRef]
- Tura, A.; Maran, A.; Pacini, G. Non-invasive glucose monitoring: Assessment of technologies and devices according to quantitative criteria. Diabetes Res. Clin. Pract. 2007, 77, 16–40. [Google Scholar] [CrossRef] [PubMed]
- Liakat, S.; Bors, K.A.; Huang, T.-Y.; Michel, A.P.M.; Zanghi, E.; Gmachl, C.F. In vitro measurements of physiological glucose concentrations in biological fluids using mid-infrared light. Biomed. Opt. Exp. 2013, 4, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, H.A.; Ashton, H.S.; Spiers, S.; Shen, Y.; Freeborn, S.S.; Hannigan, J.; Lindberg, J.; Rae, P. Advances in Photoacoustic Noninvasive Glucose Testing. Clin. Chem. 1999, 45, 1587. [Google Scholar] [PubMed]
- Liakat, S.; Bors, K.A.; Xu, L.; Woods, C.M.; Doyle, J.; Gmachl, C.F. Noninvasive in vivo glucose sensing on human subjects using mid-infrared light. Biomed. Opt. Exp. 2014, 5, 2397–2404. [Google Scholar] [CrossRef] [PubMed]
- von Lilienfeld-Toal, H.; Weidenmüller, M.; Xhelaj, A.; Mäntele, W. A novel approach to non-invasive glucose measurement by mid-infrared spectroscopy: The combination of quantum cascade lasers (QCL) and photoacoustic detection. Vib. Spectr. 2005, 38, 209–215. [Google Scholar] [CrossRef]
- Bumbrah, G.S.; Sharma, R.M. Raman spectroscopy–Basic principle, instrumentation and selected applications for the characterization of drugs of abuse. Eg. J. Forensic Sci. 2016, 6, 209–215. [Google Scholar] [CrossRef]
- Wiercigroch, E.; Szafraniec, E.; Czamara, K.; Pacia, M.Z.; Majzner, K.; Kochan, K.; Kaczor, A.; Baranska, M.; Malek, K. Raman and infrared spectroscopy of carbohydrates: A review. Spectrochim. Acta Part A Mol. Biomol. Spectr. 2017, 185, 317–335. [Google Scholar] [CrossRef]
- Xu, Y.; Ford, J.F.; Mann, C.K.; Vickers, T.J.; Brackett, J.M.; Cousineau, K.L.; Robey, W.G. Raman measurement of glucose in bioreactor materials. Proc. SPIE 1997, 2976. [Google Scholar] [CrossRef]
- Pandey, R.; Paidi, S.K.; Valdez, T.A.; Zhang, C.; Spegazzini, N.; Dasari, R.R.; Barman, I. Noninvasive Monitoring of Blood Glucose with Raman Spectroscopy. Acc. Chem. Res. 2017, 50, 264–272. [Google Scholar] [CrossRef]
- Chaiken, J.; Deng, B.; Bussjager, R.J.; Shaheen, G.; Rice, D.; Stehlik, D.; Fayos, J. Instrument for near infrared emission spectroscopic probing of human fingertips in vivo. Rev. Sci. Instrum. 2010, 81, 034301. [Google Scholar] [CrossRef] [PubMed]
- Koplik, R. Infrared spectroscopy. Available online: https://web.vscht.cz/~poustkaj/EN%20ASFA%20AU%20Koplik_Infrared_spectroscopy.pdf (accessed on 3 October 2018).
- Alarousu, E.; Hast, J.T.; Kinnunen, M.T.; Kirillin, M.Y.; Myllyla, R.A.; Plucinski, J.; Popov, A.P.; Priezzhev, A.V.; Prykari, T.; Saarela, J.; et al. Noninvasive glucose sensing in scattering media using OCT, PAS, and TOF techniques. In Proceedings of the Saratov Fall Meeting 2003: Optical Technologies Biophysics and Medicine V, Saratov, Russia, 7–10 October 2003; pp. 33–41. [Google Scholar]
- Withayachumnankul, W.; Naftaly, M. Fundamentals of Measurement in Terahertz Time-Domain Spectroscopy. J. IR Millim. THz Waves 2014, 35, 610–637. [Google Scholar] [CrossRef]
- Cherkasova, O.P.; Nazarov, M.M.; Shkurinov, A.P.; Fedorov, V.I. Terahertz spectroscopy of biological molecules. Radiophys. Quantum Electron. 2009, 52, 518. [Google Scholar] [CrossRef]
- Cherkasova, O.; Nazarov, M.; Shkurinov, A. Noninvasive blood glucose monitoring in the terahertz frequency range. Opt. Quantum Electron. 2016, 48, 217. [Google Scholar] [CrossRef]
- Gusev, S.I.; Guseva, V.A.; Simonova, A.A.; Demchenko, P.S.; Sedykh, E.A.; Cherkasova, O.P.; Khodzitsky, M.K. Application of terahertz pulsed spectroscopy for the development of non-invasive glucose measuring method. In Proceedings of the 2017 Progress In Electromagnetics Research Symposium-Spring (PIERS), St Petersburg, Russia, 22–25 May 2017; pp. 3229–3232. [Google Scholar]
- Torii, T.; Chiba, H.; Tanabe, T.; Oyama, Y. Measurements of glucose concentration in aqueous solutions using reflected THz radiation for applications to a novel sub-THz radiation non-invasive blood sugar measurement method. Digit. Health 2017, 3, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Malchoff, C.D.; Shoukri, K.; Landau, J.I.; Buchert, J.M. A novel noninvasive blood glucose monitor. Diabetes Care 2002, 25, 2268–2275. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C. Noninvasive Blood Glucose Monitoring. Diabetes Care 1997, 20, 433. [Google Scholar] [CrossRef]
- Buchert, J.M. Thermal emission spectroscopy as a tool for noninvasive blood glucose measurements. Proc. SPIE 2004, 5566, 100–111. [Google Scholar]
- Cho, O.K.; Kim, Y.O.; Mitsumaki, H.; Kuwa, K. Noninvasive Measurement of Glucose by Metabolic Heat Conformation Method. Clin. Chem. 2004, 50, 1894. [Google Scholar] [CrossRef]
- Tang, F.; Wang, X.; Wang, D.; Li, J. Non-Invasive Glucose Measurement by Use of Metabolic Heat Conformation Method. Sensors 2008, 8, 3335–3344. [Google Scholar] [CrossRef]
- Sandeep, K.V.; Luong, J.H.T. Point-of-Care Glucose Detection for Diabetic Monitoring and Management, 1st ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 19–73. [Google Scholar]
- Patel, P.; Hardik, M.; Patel, P. A Review on Photoacoustic Spectroscopy. Int. J. Pharm. Erud. 2013, 3, 41–56. [Google Scholar]
- Tanaka, Y.; Tajima, T.; Seyama, M. Differential photoacoustic spectroscopy with continuous wave lasers for non-invasive blood glucose monitoring. In Proceedings of the Photons Plus Ultrasound: Imaging and Sensing 2018, San Francisco, CA, USA, 19 February 2018; p. 104945A. [Google Scholar] [CrossRef]
- Kottmann, J.; Rey, J.M.; Sigrist, M.W. Mid-Infrared Photoacoustic Detection of Glucose in Human Skin: Towards Non-Invasive Diagnostics. Sensors 2016, 16, 1663. [Google Scholar] [CrossRef]
- Sim, J.Y.; Ahn, C.-G.; Jeong, E.-J.; Kim, B.K. In vivo Microscopic Photoacoustic Spectroscopy for Non-Invasive Glucose Monitoring Invulnerable to Skin Secretion Products. Sci. Rep. 2018, 8, 1059. [Google Scholar] [CrossRef] [PubMed]
- Pleitez, M.A.; Lieblein, T.; Bauer, A.; Hertzberg, O.; von Lilienfeld-Toal, H.; Mäntele, W. Windowless ultrasound photoacoustic cell for in vivo mid-IR spectroscopy of human epidermis: Low interference by changes of air pressure, temperature, and humidity caused by skin contact opens the possibility for a non-invasive monitoring of glucose in the interstitial fluid. Rev. Sci. Instrum. 2013, 84, 084901. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Tajima, T.; Ajito, K.; Koizumi, H. Selectivity-enhanced glucose measurement in multicomponent aqueous solution by broadband dielectric spectroscopy. In Proceedings of the 2016 IEEE MTT-S International Microwave Symposium (IMS), San Francisco, CA, USA, 22–27 May 2016; pp. 1–3. [Google Scholar]
- Bahar, A.A.M.; Zakaria, Z.; Isa, A.A.M.; Alahnomi, R.A.; Rahman, N.A. Complex Permittivity Measurement Based on Planar Microfluidic Resonator Sensor. In Proceedings of the 2018 18th International Symposium on Antenna Technology and Applied Electromagnetics (ANTEM), Waterloo, ON, Canada, 19–22 August 2018; pp. 1–5. [Google Scholar]
- Shaker, G.; Smith, K.; Omer, A.E.; Liu, S.; Csech, C.; Wadhwa, U.; Safavi-Naeini, S.; Hughson, R. Non-invasive monitoring of glucose level changes utilizing a mm-wave radar system. Int. J. Mob. Hum. Comput. Interact. 2018, 10, 10–29. [Google Scholar] [CrossRef]
- Siegel, P.H.; Tang, A.; Virbila, G.; Kim, Y.; Chang, M.C.F.; Pikov, V. Compact non-invasive millimeter-wave glucose sensor. In Proceedings of the 2015 40th International Conference on Infrared, Millimeter, and Terahertz Waves (IRMMW-THz), Hong Kong, China, 23–28 August 2015; pp. 1–3. [Google Scholar]
- Saha, S.; Cano-Garcia, H.; Sotiriou, I.; Lipscombe, O.; Gouzouasis, I.; Koutsoupidou, M.; Palikaras, G.; Mackenzie, R.; Reeve, T.; Kosmas, P.; et al. A Glucose Sensing System Based on Transmission Measurements at Millimetre Waves using Micro strip Patch Antennas. Sci. Rep. 2017, 7, 6855. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.; Fersch, T.; Weigel, R.; Fischer, G.; Kissinger, D. A novel approach to non-invasive blood glucose measurement based on RF transmission. In Proceedings of the 2011 IEEE International Symposium on Medical Measurements and Applications, Bari, Italy, 30–31 May 2011; pp. 39–42. [Google Scholar]
- Choi, H.; Naylon, J.; Luzio, S.; Beutler, J.; Birchall, J.; Martin, C.; Porch, A. Design and In Vitro Interference Test of Microwave Noninvasive Blood Glucose Monitoring Sensor. IEEE Trans. Microw. Theory Tech. 2015, 63, 3016–3025. [Google Scholar] [CrossRef]
- Zhang, R.; Qu, Z.; Jin, H.; Liu, S.; Luo, Y.; Zheng, Y. Noninvasive Glucose Measurement by Microwave Biosensor with Accuracy Enhancement. In Proceedings of the 2018 IEEE International Symposium on Circuits and Systems (ISCAS), Florence, Italy, 27–30 May 2018; pp. 1–4. [Google Scholar]
- Kim, J.; Babajanyan, A.; Hovsepyan, A.; Lee, K.; Friedman, B. Microwave dielectric resonator biosensor for aqueous glucose solution. Rev. Sci. Instrum. 2008, 79, 086107. [Google Scholar] [CrossRef]
- Yilmaz, T.; Brizzi, A.; Foster, R.; Munoz, M.; Hao, Y. A patch resonator for sensing blood glucose changes. In Proceedings of the 2014 XXXIth URSI General Assembly and Scientific Symposium (URSI GASS), Beijing, China, 16–23 August 2014; pp. 1–4. [Google Scholar]
- Gourzi, M.; Rouane, A.; Guelaz, R.; Alavi, M.S.; McHugh, M.B.; Nadi, M.; Roth, P. Non-invasive glycaemia blood measurements by electromagnetic sensor: Study in static and dynamic blood circulation. J. Med. Eng. Tech. 2005, 29, 22–26. [Google Scholar] [CrossRef]
- Melikyan, H.; Danielyan, E.; Kim, S.; Kim, J.; Babajanyan, A.; Lee, J.; Friedman, B.; Lee, K. Non-invasive in vitro sensing of d-glucose in pig blood. Med. Eng. Phys. 2012, 34, 299–304. [Google Scholar] [CrossRef]
- Weinzimer, S.A. Analysis: PENDRA: The Once and Future Noninvasive Continuous Glucose Monitoring Device? Diabetes Technol. Ther. 2004, 6, 442–444. [Google Scholar] [CrossRef]
- Harman-Boehm, I.; Gal, A.; Raykhman, A.M.; Zahn, J.D.; Naidis, E.; Mayzel, Y. Noninvasive Glucose Monitoring: A Novel Approach. J. Diabetes Sci. Technol. 2009, 3, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Kost, J. Ultrasound-Assisted Insulin Delivery and Noninvasive Glucose Sensing. Diabetes Technol. Ther. 2002, 4, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Dubinsky, T.J.; Cuevas, C.; Dighe, M.K.; Kolokythas, O.; Hwang, J.H. High-Intensity Focused Ultrasound: Current Potential and Oncologic Applications. Am. J. Roentgenol. 2008, 190, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.K.; Potts, R.O.; Ackerman, N.R.; Fermi, S.J.; Tamada, J.A.; Chase, H.P. Correlation of fingerstick blood glucose measurements with GlucoWatch biographer glucose results in young subjects with type 1 diabetes. Diabetes Care 1999, 22, 1708. [Google Scholar] [CrossRef] [PubMed]
- Potts, R.O.; Tamada, J.A.; Tierney, M.J. Glucose monitoring by reverse iontophoresis. Diabetes/Metab. Res. Rev. 2002, 18, S49–S53. [Google Scholar] [CrossRef]
- Cooke, D.; Hurel, S.J.; Casbard, A.; Steed, L.; Walker, S.; Meredith, S.; Nunn, A.J.; Manca, A.; Sculpher, M.; Barnard, M.; et al. Randomized controlled trial to assess the impact of continuous glucose monitoring on HbA1c in insulin-treated diabetes (MITRE Study). Diabetic Med. 2009, 26, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Gandrud, L.M.; Paguntalan, H.U.; Van Wyhe, M.M.; Kunselman, B.L.; Leptien, A.D.; Wilson, D.M.; Eastman, R.C.; Buckingham, B.A. Use of the Cygnus GlucoWatch biographer at a diabetes camp. Pediatrics 2004, 113, 108–111. [Google Scholar] [CrossRef]
- Wentholt, I.M.E.; Hoekstra, J.B.L.; Zwart, A.; DeVries, J.H. Pendra goes Dutch: lessons for the CE mark in Europe. Diabetologia 2005, 48, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- MIT Technology Review. Blood Sugar Crash. Available online: https://www.technologyreview.com/s/529026/blood-sugar-crash/ (accessed on 11 October 2018).
- Segman, Y. Device and Method for Noninvasive Glucose Assessment. J. Diabetes Sci. Technol. 2018, 1–10. [Google Scholar] [CrossRef]
- Pfützner, A.; Strobl, S.; Demircik, F.; Redert, L.; Pfützner, J.; Pfützner, A.H.; Lier, A. Evaluation of a New Noninvasive Glucose Monitoring Device by Means of Standardized Meal Experiments. J. Diabetes Sci. Technol. 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pfützner, A. Evaluation of the CNOGA COMBO GLUCOMETER and the MTX Non-Invasive Body Signaling Device During a Standardized Meal Test in Patients with Diabetes Mellitus and in Healthy Subjects; Pfützner Science & Health Institute: Mainz, Germany, 2017. [Google Scholar]
- Segman, Y. Optical sensor device and image processing unit for measuring chemical concentrations, chemical saturations and biophysical parameters. U.S. Patent US8792948B2, 25 March 2005. [Google Scholar]
- Amir, O.; Weinstein, D.; Zilberman, S.; Less, M.; Perl-Treves, D.; Primack, H.; Weinstein, A.; Gabis, E.; Fikhte, B.; Karasik, A. Continuous Noninvasive Glucose Monitoring Technology Based on “Occlusion Spectroscopy”. J. Diabetes Sci. Technol. 2007, 1, 463–469. [Google Scholar] [CrossRef]
- Fine, I.; Ma’Ayan, L. Glucose level control method and system. U.S. Patent US7266400B2, 6 May 2003. [Google Scholar]
- Eisen, L.; Fine, I.; Goldinov, L. Wearable pulse oximetry device. U.S. Patent US9730622B2, 30 August 2011. [Google Scholar]
- Offord, C. Will the Noninvasive Glucose Monitoring Revolution Ever Arrive? Available online: https://www.the-scientist.com/news-analysis/will-the-noninvasive-glucose-monitoring-revolution-ever-arrive-30754 (accessed on 6 October 2018).
- World Global Network. Science behind the HELO. Available online: https://www.wearablelifestyles.net/science-behind-the-helo/ (accessed on 6 October 2018).
- Pfützner, A.; Sachsenheimer, D.; Mills, L.; Deakin, S.; Moore, K.; Saini, S.; MacRury, S. Evaluation of the Non-Invasive Glucose Monitoring Device GlucoTrack in Patients with Type 2 Diabetes and Subjects with Prediabetes; Pfützner Science & Health Institute: Mainz, Germany, 2012. [Google Scholar]
- Harman-Boehm, I.; Gal, A.; Raykhman, A.M.; Naidis, E.; Mayzel, Y. Noninvasive Glucose Monitoring: Increasing Accuracy by Combination of Multi-Technology and Multi-Sensors. J. Diabetes Sci. Technol. 2010, 4, 583–595. [Google Scholar] [CrossRef] [PubMed]
- MediWise. GlucoWise. Available online: http://www.gluco-wise.com/ (accessed on 26 September 2018).
- Nemaura Medical. Nemaura Announces Positive Results for Its SugarBEAT® European Clinical Program. Available online: http://nemauramedical.com/nemaura-announces-positive-results-sugarbeat-european-clinical-program/ (accessed on 26 September 2018).
- Saur, N.M.; England, M.R.; Menzie, W.; Melanson, A.M.; Trieu, M.-Q.; Berlin, J.; Hurley, J.; Krystyniak, K.; Kongable, G.L.; Nasraway, S.A. Accuracy of a Novel Noninvasive Transdermal Continuous Glucose Monitor in Critically Ill Patients. J. Diabetes Sci. Technol. 2014, 8, 945–950. [Google Scholar] [CrossRef]
- Hadar, E.; Chen, R.; Toledano, Y.; Tenenbaum-Gavish, K.; Atzmon, Y.; Hod, M. Noninvasive, continuous, real-time glucose measurements compared to reference laboratory venous plasma glucose values. J. Matern. Fetal Neonatal Med. 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Optronics Online. QST Developed a Non-Invasive Blood Glucose Measurement Technique with a Mid-Infrared Laser. Available online: http://www.optronics-media.com/news/20170822/47807/ (accessed on 8 October 2018).
- PKvitality. K’Watch Continuous Glucose Monitoring (CGM) Device. Available online: https://www.pkvitality.com/wp-content/uploads/2018/11/PKVITALITY-KWatch-CGM-21112018-EN.pdf (accessed on 12 December 2018).
- PKvitality. K’Watch Glucose CGM Reinvented. Available online: https://www.pkvitality.com/ktrack-glucose/ (accessed on 11 January 2018).
- DeHennis, A.; Tankiewicz, S.; Whitehurst, T. Analyte sensor. U.S. Patent US 9,901,293 B2, 24 February 2015. [Google Scholar]
- Jafri, R.Z.; Balliro, C.A.; El-Khatib, F.; Maheno, M.; Hillard, M.A.; Donovan, A.J.; Selagamsetty, R.; Zheng, H.U.I.; Damiano, E.; Russell, S.J. A Three-Way Accuracy Comparison of the Dexcom G5, Abbott Freestyle Libre Pro, and Senseonics Eversense CGM Devices in an Outpatient Study of Subjects with Type 1 Diabetes. Diabetes 2018, 67, 14-OR. [Google Scholar] [CrossRef]
- Senseonics. Eversense User Guide. Available online: https://www.eversensediabetes.com/wp-content/uploads/2018/08/LBL-1602-01-001-Rev-D_Eversense-User-Guide_mgdL_R1-2.pdf (accessed on 18 January 2019).
- Tang, F.; You, Z.; Wang, X.; Li, Y.; Yan, Y.; Fan, Z. Non-Invasive Blood Glucose Detector Based on Metabolic Heat-Optical Method. Chinese Patent CN102293654B, 17 June 2011. [Google Scholar]
- Siegel, P.H.; Lee, Y.; Pikov, V. Millimeter-wave non-invasive monitoring of glucose in anesthetized rats. In Proceedings of the 2014 39th International Conference on Infrared, Millimeter, and Terahertz waves (IRMMW-THz), Tucson, AZ, USA, 14–19 September 2014; pp. 1–2. [Google Scholar]
- Siegel, P.H.; Tang, A.; Kim, R.; Virbila, G.; Chang, F.; Pikov, V. Noninvasive in vivo millimeter-wave measurements of glucose: First results in human subjects. In Proceedings of the 2017 42nd International Conference on Infrared, Millimeter, and Terahertz Waves (IRMMW-THz), Cancun, Mexico, 27 August–1 September 2017; pp. 1–2. [Google Scholar]
- Chen, L.; Tse, W.H.; Chen, Y.; McDonald, M.W.; Melling, J.; Zhang, J. Nanostructured biosensor for detecting glucose in tear by applying fluorescence resonance energy transfer quenching mechanism. Biosens. Bioelectr. 2017, 91, 393–399. [Google Scholar] [CrossRef]
- Monte-Moreno, E. Non-invasive estimate of blood glucose and blood pressure from a photoplethysmograph by means of machine learning techniques. Artif. Intell. Med. 2011, 53, 127–138. [Google Scholar] [CrossRef]
- Philip, L.A.; Rajasekaran, K.; Jothi, E.S.J. Continous monitoring of blood glucose using photophlythesmograph signal. In Proceedings of the 2017 International Conference on Innovations in Electrical, Electronics, Instrumentation and Media Technology (ICEEIMT), Coimbatore, India, 3–4 February 2017; pp. 187–191. [Google Scholar]
- Kino, S.; Omori, S.; Katagiri, T.; Matsuura, Y. Hollow optical-fiber based infrared spectroscopy for measurement of blood glucose level by using multi-reflection prism. Biomed. Opt. Exp. 2016, 7, 701–708. [Google Scholar] [CrossRef]
- Lundsgaard-Nielsen, S.M.; Pors, A.; Banke, S.O.; Henriksen, J.E.; Hepp, D.K.; Weber, A. Critical-depth Raman spectroscopy enables home-use non-invasive glucose monitoring. PLoS ONE 2018, 13, e0197134. [Google Scholar] [CrossRef]
- Chen, T.-L.; Lo, Y.-L.; Liao, C.-C.; Phan, Q.-H. Noninvasive measurement of glucose concentration on human fingertip by optical coherence tomography. J. Biomed. Opt. 2018, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.; Fischer, G.; Weigel, R.; Kissinger, D. Microwave-Based Noninvasive Concentration Measurements for Biomedical Applications. IEEE Trans. Microw. Theory Tech. 2013, 61, 2195–2204. [Google Scholar] [CrossRef]
- Choi, H.; Luzio, S.; Beutler, J.; Porch, A. Microwave noninvasive blood glucose monitoring sensor: Human clinical trial results. In Proceedings of the 2017 IEEE MTT-S International Microwave Symposium (IMS), Honolulu, HI, USA, 4–9 June 2017; pp. 876–879. [Google Scholar]
- Lipani, L.; Dupont, B.G.R.; Doungmene, F.; Marken, F.; Tyrrell, R.M.; Guy, R.H.; Ilie, A. Non-invasive, transdermal, path-selective and specific glucose monitoring via a graphene-based platform. Nat. Nanotechnol. 2018, 13, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Vega, K.; Jiang, N.; Liu, X.; Kan, V.; Barry, N.; Maes, P.; Yetisen, A.; Paradiso, J. The dermal abyss: interfacing with the skin by tattooing biosensors. In Proceedings of the 2017 ACM International Symposium on Wearable Computers (ISWC’17), Maui, HI, USA, 11–15 September 2017; pp. 138–145. [Google Scholar]
- Park, J.; Kim, J.; Kim, S.-Y.; Cheong, W.H.; Jang, J.; Park, Y.-G.; Na, K.; Kim, Y.-T.; Heo, J.H.; Lee, C.Y.; et al. Soft, smart contact lenses with integrations of wireless circuits, glucose sensors, and displays. Sci. Adv. 2018, 4. [Google Scholar] [CrossRef]
- Badugu, R.; Reece, E.A.; Lakowicz, J.R. Glucose-sensitive silicone hydrogel contact lens toward tear glucose monitoring. J. Biomed. Opt. 2018, 23, 057005. [Google Scholar] [CrossRef] [PubMed]
- Ribet, F.; Stemme, G.; Roxhed, N. Real-time intradermal continuous glucose monitoring using a minimally invasive microneedle-based system. Biomed. Microdevices 2018, 20, 101. [Google Scholar] [CrossRef] [PubMed]
- Gamsey, S.; Suri, J.T.; Wessling, R.A.; Singaram, B. Continuous Glucose Detection Using Boronic Acid-Substituted Viologens in Fluorescent Hydrogels: Linker Effects and Extension to Fiber Optics. Langmuir 2006, 22, 9067–9074. [Google Scholar] [CrossRef]
- Gamsey, S.; Bernat, V.; Kutyavin, A.; Clary, J.W.; Pradhan, S. Near-IR glucose sensors. U.S. Patent US20180179233A1, 27 December 2016. [Google Scholar]
- Larin, K.V.; Eledrisi, M.S.; Motamedi, M.; Esenaliev, R.O. Noninvasive Blood Glucose Monitoring With Optical Coherence Tomography. Diabetes Care 2002, 25, 2263–2267. [Google Scholar] [CrossRef]
- Palerm, C.C.; Bequette, B.W. Hypoglycemia Detection and Prediction Using Continuous Glucose Monitoring—A Study on Hypoglycemic Clamp Data. J. Diabetes Sci. Techol. 2007, 1, 624–629. [Google Scholar] [CrossRef]
- Mahmoudi, Z.; Dencker Johansen, M.; Christiansen, J.S.; Hejlesen, O.K. A Multistep Algorithm for Processing and Calibration of Microdialysis Continuous Glucose Monitoring Data. Diabetes Technol. Ther. 2013, 15, 825–835. [Google Scholar] [CrossRef]
- Facchinetti, A. Continuous Glucose Monitoring Sensors: Past, Present and Future Algorithmic Challenges. Sensors 2016, 16, 2093. [Google Scholar] [CrossRef] [PubMed]
- Barceló-Rico, F.; Bondia, J.; Díez, J.L.; Rossetti, P. A Multiple Local Models Approach to Accuracy Improvement in Continuous Glucose Monitoring. Diabetes Technol. Ther. 2011, 14, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Kirchsteiger, H.; Zaccarian, L.; Renard, E.; Re, L.D. LMI-Based Approaches for the Calibration of Continuous Glucose Measurement Sensors. IEEE J. Biomed. Health Inf. 2015, 19, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Sparacino, G.; Zanderigo, F.; Maran, A.; Cobelli, C. Continuous glucose monitoring and hypo/hyperglycaemia prediction. Diabetes Res. Clin. Pract. 2006, 74, S160–S163. [Google Scholar] [CrossRef]
- Reifman, J.; Rajaraman, S.; Gribok, A.; Ward, W.K. Predictive Monitoring for Improved Management of Glucose Levels. J. Diabetes Sci. Technol. 2007, 1, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Gandía, C.; Facchinetti, A.; Sparacino, G.; Cobelli, C.; Gómez, E.J.; Rigla, M.; de Leiva, A.; Hernando, M.E. Artificial Neural Network Algorithm for Online Glucose Prediction from Continuous Glucose Monitoring. Diabetes Technol. Ther. 2010, 12, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Zanon, M.; Sparacino, G.; Facchinetti, A.; Talary, S.M.; Mueller, M.; Caduff, A.; Cobelli, C. Non-Invasive Continuous Glucose Monitoring with Multi-Sensor Systems: A Monte Carlo-Based Methodology for Assessing Calibration Robustness. Sensors 2013, 13. [Google Scholar] [CrossRef]
- Eadie, M.; Steele, R. Non-invasive Blood Glucose Monitoring and Data Analytics. In Proceedings of the ICCDA’17, Lakeland, FL, USA, 19–23 May 2017; pp. 138–142. [Google Scholar]
- Sandham, W.; Nikoletou, D.; Hamilton, D.; Paterson, K.; Japp, A.; Macgregor, C. Blood glucose prediction for diabetes therapy using a recurrent artificial neural network. In Proceedings of the EUSIPCO, Rhodes, Greece, 8–11 September 1998. [Google Scholar]
- Tresp, V.; Briegel, T.; Moody, J. Neural-network models for the blood glucose metabolism of a diabetic. IEEE Trans. Neural Netw. 1999, 10, 1204–1213. [Google Scholar] [CrossRef]
- Mougiakakou, S.G.; Prountzou, K.; Nikita, K.S. A Real Time Simulation Model of Glucose-Insulin Metabolism for Type 1 Diabetes Patients. In Proceedings of the 2005 IEEE Engineering in Medicine and Biology 27th Annual Conference, Shanghai, China, 17–18 January 2006; pp. 298–301. [Google Scholar]
- Smith, J. The Pursuit of Noninvasive Glucose: Hunting the Deceitful Turkey, 5th ed. Available online: https://www.researchgate.net/publication/317267760_The_Pursuit_of_Noninvasive_Glucose_5th_Edition (accessed on 14 December 2018).
- Purvinis, G.; Cameron, B.D.; Altrogge, D.M. Noninvasive polarimetric-based glucose monitoring: an in vivo study. J. Diabetes Sci. Techol. 2011, 5, 380–387. [Google Scholar] [CrossRef]
- Winkler, A.M.; Bonnema, G.T.; Barton, J.K. Optical polarimetry for noninvasive glucose sensing enabled by Sagnac interferometry. Appl. Opt. 2011, 50, 2719–2731. [Google Scholar] [CrossRef]
- Yao, H.; Shum, A.J.; Cowan, M.; Lähdesmäki, I.; Parviz, B.A. A contact lens with embedded sensor for monitoring tear glucose level. Biosens. Bioelectron. 2011, 26, 3290–3296. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.X.; Miyajima, K.; Takahashi, D.; Arakawa, T.; Sano, K.; Sawada, S.-i.; Kudo, H.; Iwasaki, Y.; Akiyoshi, K.; Mochizuki, M.; et al. Soft contact lens biosensor for in situ monitoring of tear glucose as non-invasive blood sugar assessment. Talanta 2011, 83, 960–965. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hodge, W.G. Contact lens integrated with a biosensor for the detection of glucose and other components in tears. U.S. Patent US8385998B2, 24 October 2008. [Google Scholar]
- Ruan, J.-L.; Chen, C.; Shen, J.-H.; Zhao, X.-L.; Qian, S.-H.; Zhu, Z.-G. A Gelated Colloidal Crystal Attached Lens for Noninvasive Continuous Monitoring of Tear Glucose. Polymers 2017, 9. [Google Scholar] [CrossRef]
- Elsherif, M.; Hassan, M.U.; Yetisen, A.K.; Butt, H. Wearable Contact Lens Biosensors for Continuous Glucose Monitoring Using Smartphones. ACS Nano 2018, 12, 5452–5462. [Google Scholar] [CrossRef] [PubMed]
- Tseng, R.C.; Chen, C.-C.; Hsu, S.-M.; Chuang, H.-S. Contact-Lens Biosensors. Sensors 2018, 18, 2651. [Google Scholar] [CrossRef] [PubMed]
| Method | Equipment | Intended Use | Sample Type | Range | Ref. |
|---|---|---|---|---|---|
| Enzymatic | YSI 2700 | Laboratory Point-of-care |
| 0–2500 mg/dL | [14] |
| YSI 2950D | Laboratory | 5–2500 mg/dL | [15] | ||
| Biosen C-Line/S-Line | Laboratory Point-of-care |
| 9–900 mg/dL | [16] | |
| Hexokinase | Beckman Coulter DxC 800 | Laboratory |
| 5–700 mg/dL | [17] |
| Abbott ARCHITECT c8000/c16000 | 1–800 mg/dL | [18] | |||
| Hitachi 917 | 2–750 mg/dL | [18] | |||
| Cobas c 701/702 | 2–750 mg/dL | [19] |
| Characteristics | Laboratory | Self-Monitoring |
|---|---|---|
| Accuracy | Very good | Good |
| Sensitivity | Very good | Good |
| Measurement time | Long | Quick |
| Trained laboratory personnel | Yes | No |
| Sample type | Blood, serum, plasma, urine | Blood, ISF |
| Blood extraction method | Invasive | Invasive |
| Clarke Error Grid [40] | Parkes Error Grid Type 1 Diabetes [41] | Parkes Error Grid Type 2 Diabetes [41] | Surveillance Error Grid [38] | |
|---|---|---|---|---|
| Risk zones | A to E | A to E | Green to Dark-red | |
A Green | Clinically correct decisions | No effect on clinical action | No risk | |
B G/Y | Clinically uncritical decisions | Altered clinical action or little or no effect on clinical outcome | Mild risk | |
C Y/R | Overcorrections that could lead to a poor outcome | Altered clinical action: likely to affect clinical outcome | Moderate risk | |
D Red | Dangerous failure to detect and treat | Altered clinical action: potential significant medical risk | High risk | |
E Dark Red | Erroneous treatment | Altered clinical action: potential dangerous consequences. | Extreme risk | |
| Agency | Country | Guideline/Standard | Release Year | Device Type | Glucose Concentration | Criteria |
|---|---|---|---|---|---|---|
| Food & Drug Administration (FDA) | USA [47,48] | UCM 380325 | 2016 | BGMS | ≥75 mg/dL | 95% within ±12% 98% within ±15% |
| <75 md/dL | 95% within ±12 mg/dL 98% within ±15 mg/dL | |||||
| UCM 380327 | SMBG | Entire range | 95% within ±15% 99% within ±20% | |||
| European Medicines Agency (EMA) | EU [49] | EN ISO 15197 | 2015 | BGMS/SMBG | ≥100 mg/dL | 95% within ±15% |
| Health Canada | Canada [50] | ISO 15197 | 2013 | <100 mg/dL | 95% within ±15 mg/dL | |
| Agência Nacional de Vigilância Sanitária (ANVISA) | Brazil [51] | |||||
| China Food & Drug Administration (CFDA) | China [52] | |||||
| Pharmaceuticals and Medical Devices Agency (PMDA) | Japan [53,54] | Entire range (Type 1 Diabetes) | 99% within Zones A & B of Parkes Error Grid | |||
| Therapeutic Goods Administration TGA | Australia [55,56,57] |
| Bioimpedance Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| Fluorescence | |
|---|---|
| Advantages | Disadvantages |
|
|
| Optical Polarimetry | |
|---|---|
| Advantages | Disadvantages |
|
|
| Optical Coherence Tomography | |
|---|---|
| Advantages | Disadvantages |
|
|
| NIR Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| MIR Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| Raman Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| FIR Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
| Less scattering than NIR and MIR | Strong water absorption makes extremely difficult the identification of other molecules in the sample. |
| TOF/THz-TDS | |
|---|---|
| Advantages | Disadvantages |
|
|
| Sonophoresis | |
|---|---|
| Advantages | Disadvantages |
|
|
| Metabolic Heat Conformation | |
|---|---|
| Advantages | Disadvantages |
|
|
| Photoacoustic Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| Millimeter and Microwave Sensing | |
|---|---|
| Advantages | Disadvantages |
|
|
| Electromagnetic Sensing | |
|---|---|
| Advantages | Disadvantages |
|
|
| Bioimpedance Spectroscopy | |
|---|---|
| Advantages | Disadvantages |
|
|
| Ultrasound | |
|---|---|
| Advantages | Disadvantages |
|
|
| Sonophoresis | |
|---|---|
| Advantages | Disadvantages |
|
|
| Reverse Iontophoresis | |
|---|---|
| Advantages | Disadvantages |
|
|
| Device | Technology |
|---|---|
| GlucoWatch | Reverse iontophoresis |
| GluCall | Reverse iontophoresis |
| Pendra | Impedance spectroscopy |
| Glucoband | Impedance spectroscopy |
| Hitachi Ltd. | Metabolic heat conformation |
| Aprise | Photoacoustic spectroscopy |
| C8 Medisensors | Raman spectroscopy |
| Diasensor 1000 | NIR spectroscopy |
| TouchTrack Pro | NIR spectroscopy |
| GluControl | NIR spectroscopy |
| Device | Technology | Target | Type | Accuracy | Status | Ref. |
|---|---|---|---|---|---|---|
| Combo Glucometer (Cnoga Medical) | NIR spectroscopy (combination of four LEDs and four sensors to analyse absorption and scattering pattern) λ: 625 nm, 740 nm, 850 nm, 940 nm | Finger | NI NCGM | PEG Zone A: 96.6% Zone B: 3.4% MARD: 14.4% | Available | [136,137,138,139] |
| NBM-200G* (OrSense) | NIR spectroscopy (Occlusion spectroscopy) λ: 610 nm, 810 nm | Finger | NI Point-of-care | CEG Zone A: 69.7% Zone B: 25.7% | Dropped | [140,141,142,143] |
| HELO Extense (World Global Network) | NIR spectroscopy | Finger | NI NCGM | N/A | Available | [144] |
| GlucoTrack (Integrity Applications) | Combination of:
| Ear lobe | NI NCGM | PEG Zone A: 62.4% Zone B: 37.6% MARD: 19.7% | Available | [127,145,146] |
| GlucoWise (MediWise) | mm-Wave Transmission spectroscopy f: 60 GHz | Hand | NI NCGM | N/A | Under develop-ment | [118,147] |
| SugarBEAT (Nemaura Medical) | Reverse iontophoresis | Upper arm | MI CGM | MARD: 13.76% | Waiting for CE approval | [148] |
| Symphony (Echo Therapeutics) | Sonophoresis | Skin | MI CGM | CEG Zone A: 81.7% Zone B: 18.3% MARD: 12.3% | Unknown | [149] |
| WizmiTM (Wear2b Ltd) | NIR spectroscopy | Arm wrist | NI CGM | CEG Zone A: 93% Zone B: 7% MARD: 7.23% | Proof of concept | [150] |
| LTT (Light Touch technology) | MIR spectroscopy/Optical Parametric Oscillation λ: 6–9 μm | Finger | NI NCGM | N/A | Under develop-ment | [151] |
| K’Watch (PKvitality) | Enzymatic detection/microneedles | Arm wrist | MI CGM | N/A | Pre-clinical tests | [11,152,153] |
| Eversense® (Senseonics) | Fluorescence | Upper arm | MI CGM | MARD: 14.8% | Available | [154,155,156] |
| Health-Care Computer | Metabolic heat conformation λ: 660 nm, 760 nm, 850 nm, 940 nm | Finger | NI NCGM | 87% | Available | [107,157] |
| GlucoGenius | N/A | Unknown | ||||
| GlucoDiary | ||||||
| G2 Mobile (Eser Digital) |
| Institution | Technology | Comments | Target | Ref. |
|---|---|---|---|---|
| Polytechnic University of Catalunya | NIR spectroscopy Photoplethysmo-graphy |
| Finger | [161] |
| Karunya University | NIR spectroscopy Photoplethysmo-graphy |
| Forearm & finger | [162] |
| Tohoku University | MIR spectroscopy Trapezoidal multireflection |
| Oral mucosa Inner lips | [163] |
| ETH Zurich | MIR spectroscopy Photoacoustic detection |
| Forearm | [111] |
| RSP Systems | Raman spectroscopy |
| Hand palm | [164] |
| University Western Ontario (WOU) | Fluorescence Resonance Energy Transfer (FRET) | Spectral determination based on competition reaction between fluorophore’s donor and acceptor | - - - | [160] |
| Electronics and Telecomm. Research Inst. of Korea (ETRI) | Photoacoustic spectroscopy |
| Fingertip | [112] |
| National Cheng Kung University (NCKU) | Optical Coherence Tomography |
| Fingertip | [165] |
| Caltech | Millimeter-wave Transmission |
| Ear lobe | [117,159] |
| University of Waterloo | Millimeter-wave transmission & reflection |
| - - - | [116] |
| University of Erlangen-Nuremberg | Millimeter-wave transmission & reflection |
| - - - | [119,166] |
| Cardiff University | Microwave Split-ring resonance |
| Abdomen | [120,167] |
| University of Bath | Reverse iontophoresis |
| Skin | [168] |
| MIT–DermalAbyss | Chemical fluorescence |
| Skin | [169] |
| Ulsan National Inst. of Science and Technology (UNIST) | Contact lenses–Enzymatic detection |
| Tears | [170] |
| University of Maryland | Contact lenses-fluorescence |
| Tears | [171] |
| KTH Royal Inst. of Technology | Microneedle-Enzymatic detection |
| Forearm | [172] |
| Profusa, Inc. | Fluorescence |
| - - - | [173,174] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villena Gonzales, W.; Mobashsher, A.T.; Abbosh, A. The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors. Sensors 2019, 19, 800. https://doi.org/10.3390/s19040800
Villena Gonzales W, Mobashsher AT, Abbosh A. The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors. Sensors. 2019; 19(4):800. https://doi.org/10.3390/s19040800
Chicago/Turabian StyleVillena Gonzales, Wilbert, Ahmed Toaha Mobashsher, and Amin Abbosh. 2019. "The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors" Sensors 19, no. 4: 800. https://doi.org/10.3390/s19040800
APA StyleVillena Gonzales, W., Mobashsher, A. T., & Abbosh, A. (2019). The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors. Sensors, 19(4), 800. https://doi.org/10.3390/s19040800






