An Ethanol Vapor Sensor Based on a Microfiber with a Quantum-Dot Gel Coating
Abstract
1. Introduction
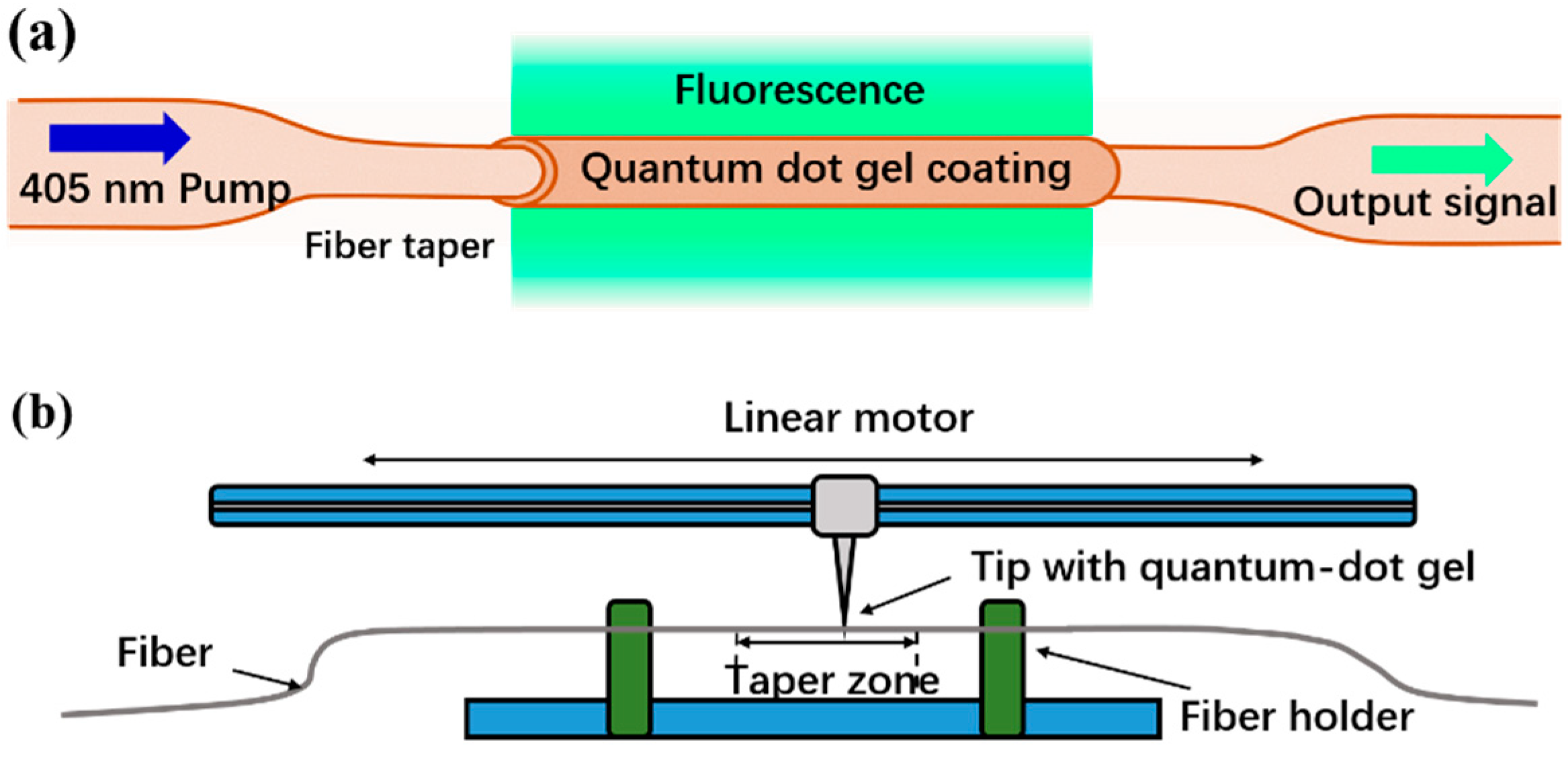
2. Sensor Fabrication
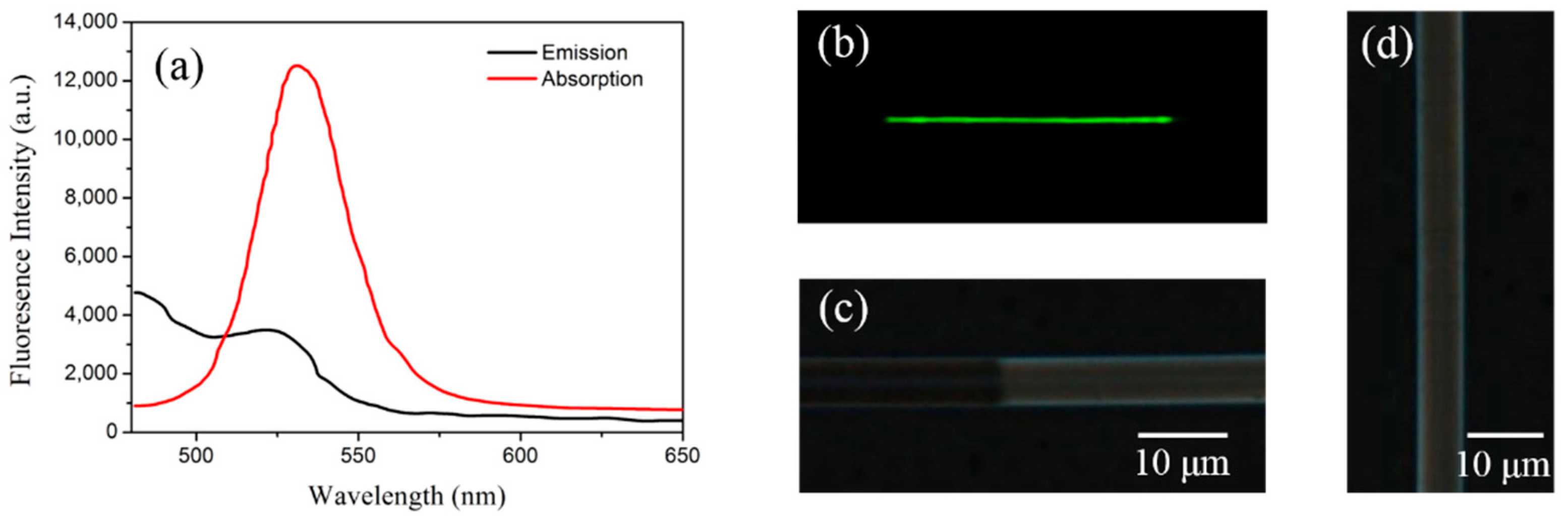
3. Results
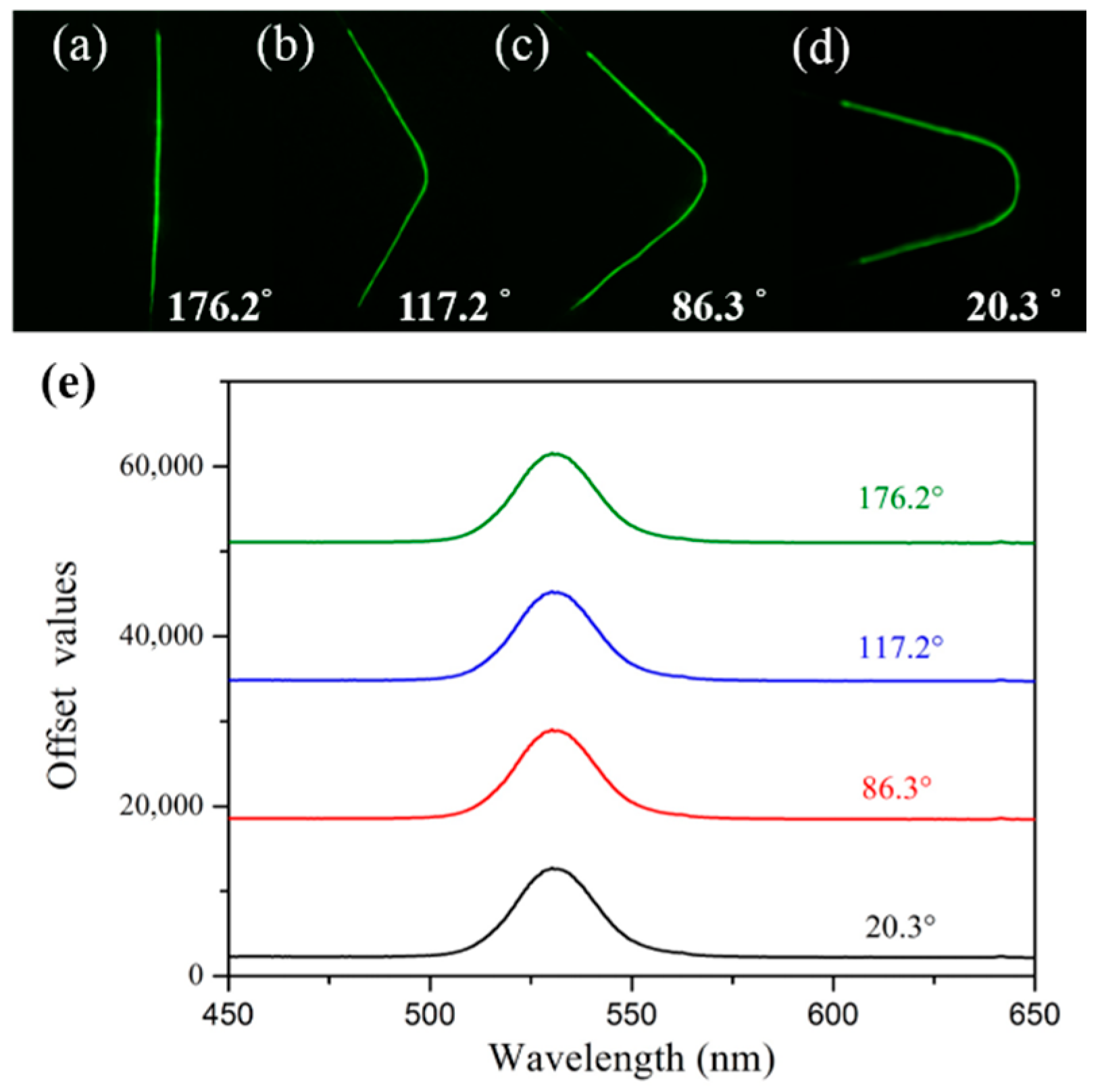
3.1. Bending Test
3.2. Sensing Performance
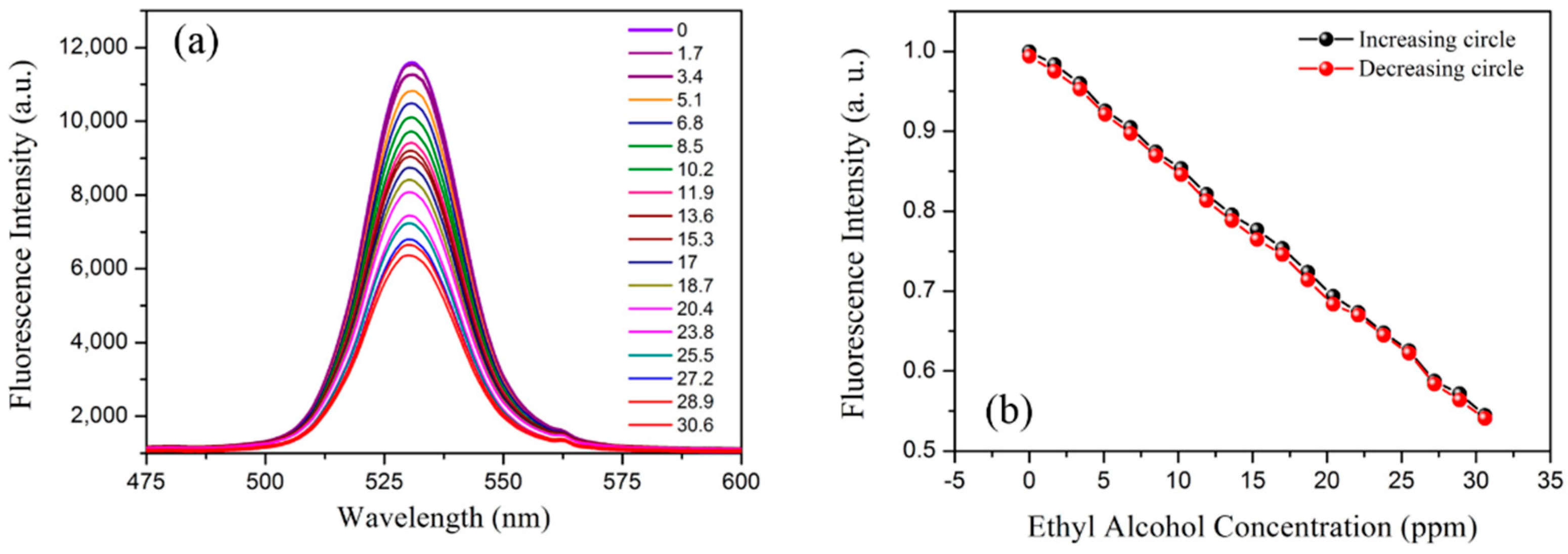
3.2.1. Ethanol Vapor Sensing
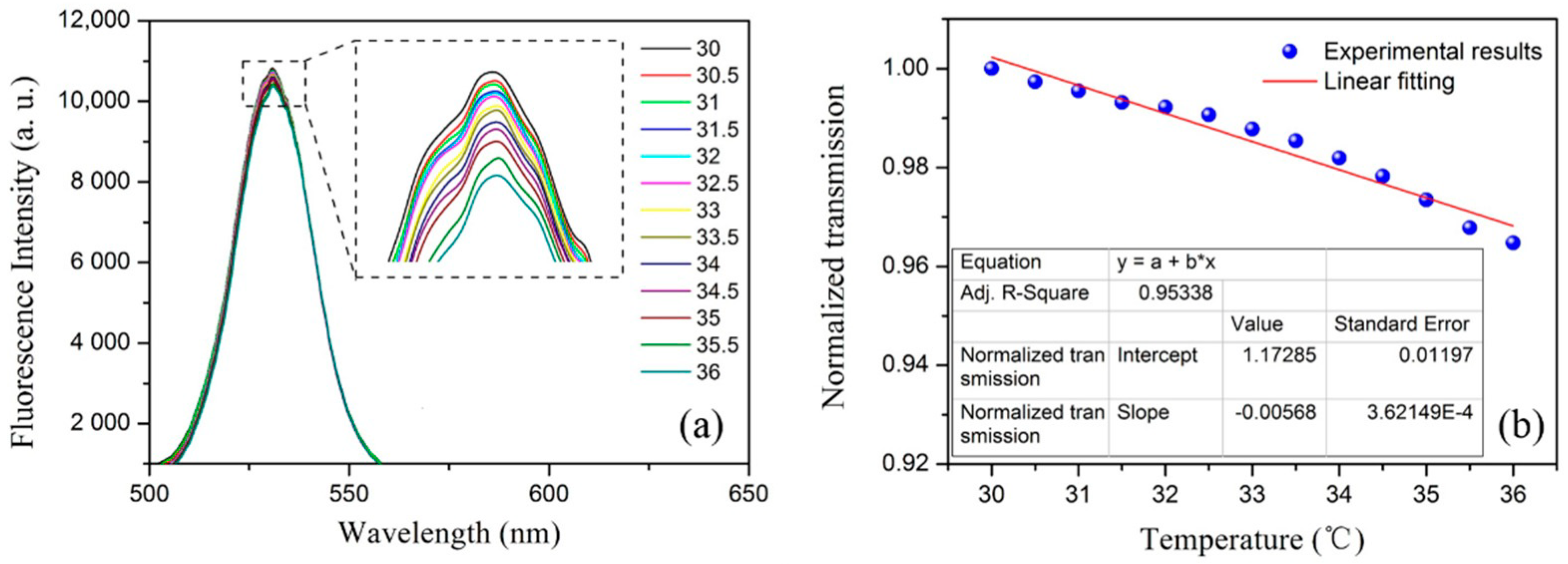
3.2.2. Temperature Response
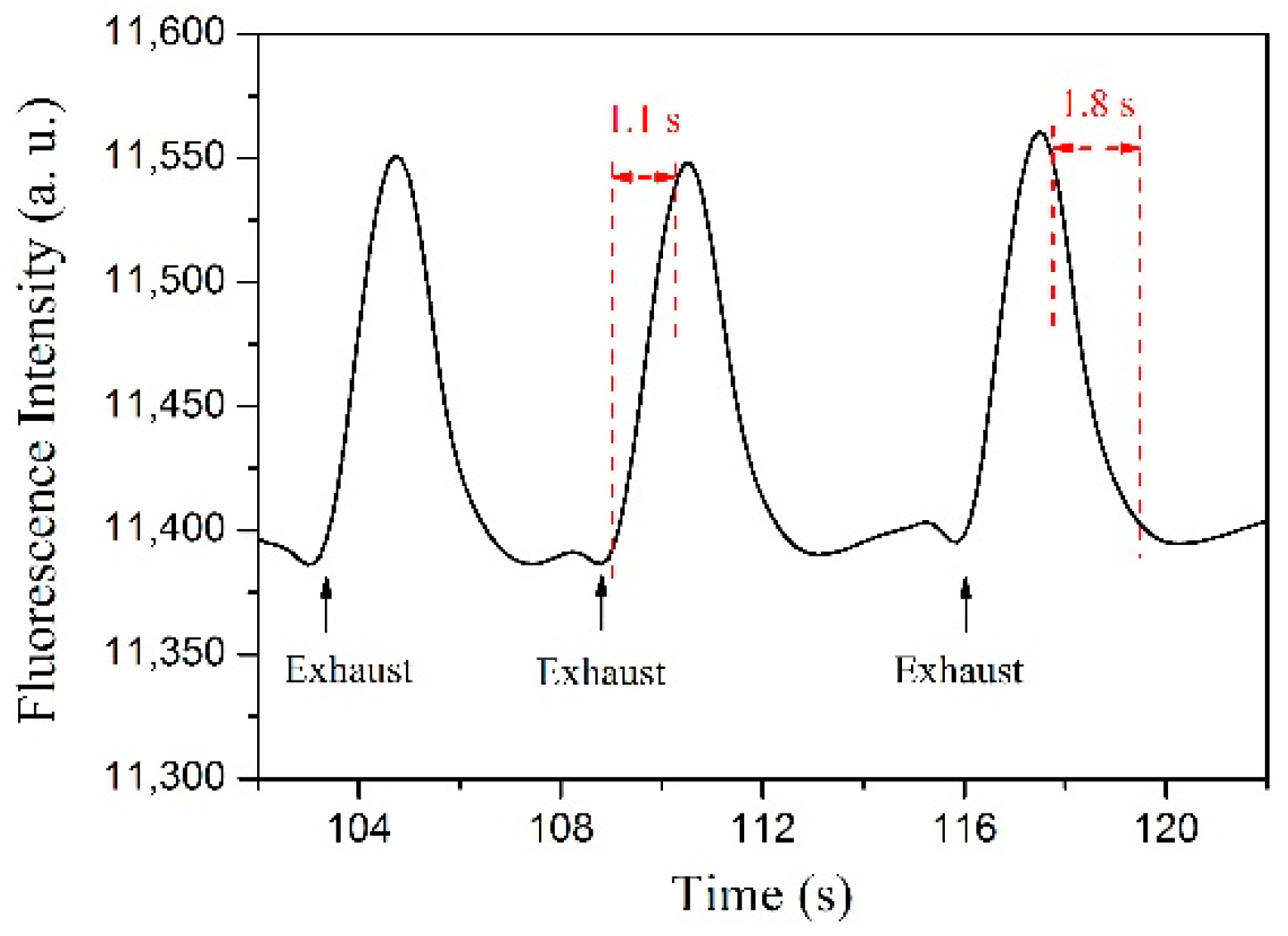
3.2.3. Time Response
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Neri, G.; Bonavita, A.; Micali, G.; Donato, N.; Deorsola, F.A.; Mossino, P.; Amato, I.; de Benedetti, B. Ethanol sensors based on Pt-doped tin oxide nanopowders synthesised by gel-combustion. Sens. Actuators B 2006, 117, 196–204. [Google Scholar] [CrossRef]
- Liu, H.; Li, M.; Voznyy, O.; Hu, L.; Fu, Q.; Zhou, D.; Xia, Z.; Sargent, E.H.; Tang, J. Physically Flexible, Rapid-Response Gas Sensor Based on Colloidal Quantum Dot Solids. Adv. Mater. 2014, 26, 2718–2724. [Google Scholar] [CrossRef] [PubMed]
- Ippolito, S.J.; Ponzoni, A.; Kalantar-Zadeh, K.; Wlodarski, W.; Comini, E.; Faglia, G.; Sberveglieri, G. Layered WO3/ZnO/360 LiTaO3 SAW gas sensor sensitive towards ethanol vapour and humidity. Sens. Actuators B 2006, 117, 442–450. [Google Scholar] [CrossRef]
- Latinoa, M.; Montaninia, R.; Donatoc, N.; Neria, G. Ethanol Sensing Properties of PMMA-Coated Fiber Bragg Grating. Procedia Eng. 2012, 47, 1263–1266. [Google Scholar] [CrossRef]
- Konstantaki, M.; Klini, A.; Anglos, D.; Pissadakis, S. An ethanol vapor detection probe based on a ZnO nanorod coated optical fiber long period grating. Opt. Express 2012, 20, 8472–8484. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wu, G.; Chen, J.; Yan, S.; Liu, Z.; Xu, F.; Lu, Y. Ethanol Gas Sensor Based on a Hybrid Polymethyl Methacrylate–Silica Microfiber Coupler. J. Lightw. Technol. 2018, 36, 2031–2036. [Google Scholar] [CrossRef]
- Tung, T.T.; Nine, M.J.; Krebsz, M.; Pasinszki, T.; Coghlan, C.J.; Tran, D.N.H.; Losic, D. Recent advances in sensing applications of graphene assemblies and their composites. Adv. Funct. Mater. 2017, 27, 1702891. [Google Scholar] [CrossRef]
- Hernaez, M.; Mayes, A.G.; Melendi-Espina, S. Graphene oxide in lossy mode resonance-based optical fiber sensors for ethanol detection. Sensors 2018, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Kumar, R.; Wei, F.; Han, W.; Mallik, A.K.; Yuan, J.; Wan, S.; He, X.; Kang, Z.; Li, F.; et al. High sensitivity optical fiber sensors for simultaneous measurement of methanol and ethanol. Sens. Actuators B 2018, 271, 1–8. [Google Scholar]
- Kudo, H.; Sawai, M.; Suzuki, Y.; Wang, X.; Gessei, T.; Takahashi, D.; Arakawa, T.; Mitsubayashi, K. Fiber-optic bio-sniffer (biochemical gas sensor) for high-selective monitoring of ethanol vapor using 335 nm UV-LED. Sens. Actuators B 2010, 147, 676–680. [Google Scholar] [CrossRef]
- Walker, G.W.; Sundar, V.C.; Rudzinski, C.M.; Wun, A.W.; Bawendi, M.G. Quantum-dot optical temperature probes. Appl. Phys. Lett. 2003, 83, 3555–3557. [Google Scholar] [CrossRef]
- Larrion, B.; Hernaez, M.; Arregui, F.J.; Goicoechea, J.; Bravo, J.; Matıas, I.R. Photonic Crystal Fiber Temperature Sensor Based on Quantum Dot Nanocoatings. J. Sens. 2009, 2009, 1687–7268. [Google Scholar] [CrossRef]
- De Bastida, G.; Arregui, F.J.; Goicoechea, J.; Matias, I.R. Quantum Dots-Based Optical Fiber Temperature Sensors Fabricated by Layer-by-Layer. IEEE Sens. J. 2006, 6, 1378–1379. [Google Scholar] [CrossRef]
- Kalathimekkad, S.; Missinne, J.; Schaubroeck, D.; Mandamparambil, R.; van Steenberge, G. Alcohol Vapor Sensor Based on Fluorescent Dye-Doped Optical Waveguides. IEEE Sens. J. 2015, 15, 76–81. [Google Scholar] [CrossRef]
- Meng, C.; Xiao, Y.; Wang, P.; Zhang, L.; Liu, Y.; Tong, L. Quantum-Dot-Doped Polymer Nanofibers for Optical Sensing. Adv. Mater. 2011, 23, 3770–3774. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, F.; Ooi, P.C.; Ye, Y.; Kim, T.W.; Guo, T. Room temperature pH-dependent ammonia gas sensors using graphene quantum dots. Sens. Actuators B 2016, 222, 763–768. [Google Scholar] [CrossRef]
- Chu, C.; Chuang, C. Optical fiber sensor for dual sensing of dissolved oxygen and Cu2+ ions based on PdTFPP/CdSe embedded in sol–gel matrix. Sens. Actuators B 2015, 209, 94–99. [Google Scholar] [CrossRef]
- Zhou, M.; Guo, J.; Yang, C. Ratiometric fluorescence sensor for Fe3+ ions detection based on quantum dot-doped hydrogel optical fiber. Sens. Actuators B 2018, 264, 52–58. [Google Scholar] [CrossRef]
- Brus, L.E. Electron-electron and electron-hole interactions in small semiconductor crystallites: The size dependence of the lowest excited electronic state. J. Chem. Phys. 1984, 80, 4403–4409. [Google Scholar] [CrossRef]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, S.; Yan, G.; Wu, C.; He, S. An Ethanol Vapor Sensor Based on a Microfiber with a Quantum-Dot Gel Coating. Sensors 2019, 19, 300. https://doi.org/10.3390/s19020300
Hu S, Yan G, Wu C, He S. An Ethanol Vapor Sensor Based on a Microfiber with a Quantum-Dot Gel Coating. Sensors. 2019; 19(2):300. https://doi.org/10.3390/s19020300
Chicago/Turabian StyleHu, Siqi, Guofeng Yan, Chunzhou Wu, and Sailing He. 2019. "An Ethanol Vapor Sensor Based on a Microfiber with a Quantum-Dot Gel Coating" Sensors 19, no. 2: 300. https://doi.org/10.3390/s19020300
APA StyleHu, S., Yan, G., Wu, C., & He, S. (2019). An Ethanol Vapor Sensor Based on a Microfiber with a Quantum-Dot Gel Coating. Sensors, 19(2), 300. https://doi.org/10.3390/s19020300






