Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies
Abstract
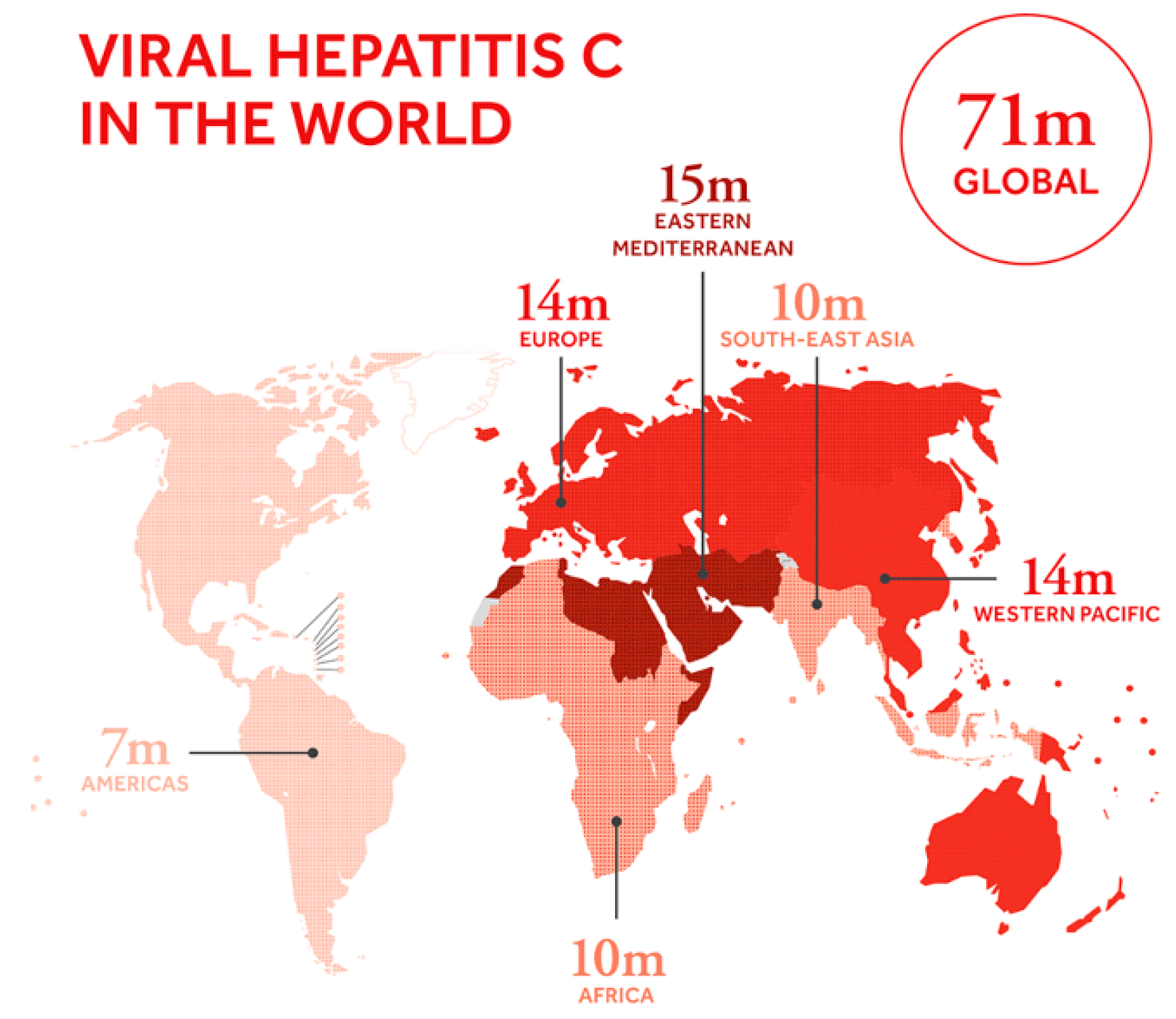
1. Introduction
2. HCV Proteins and Their Functions
3. Enzyme Immunoassay (EIA)
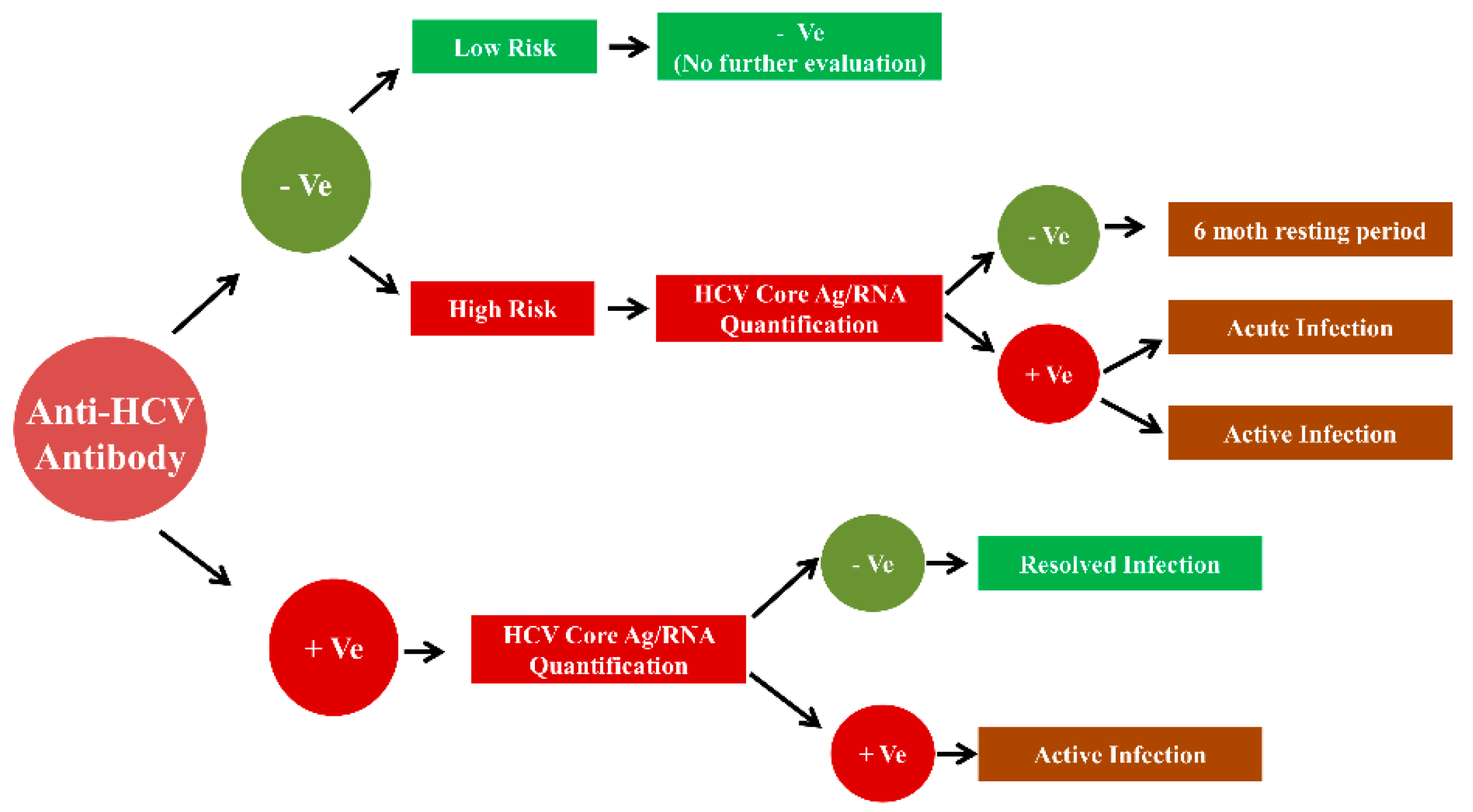
3.1. Anti-HCV Antibody Detection
3.2. Core Antigen Detection
3.3. Multiple Antigen Detection
4. Chemiluminescence Immunoassay (CIA)
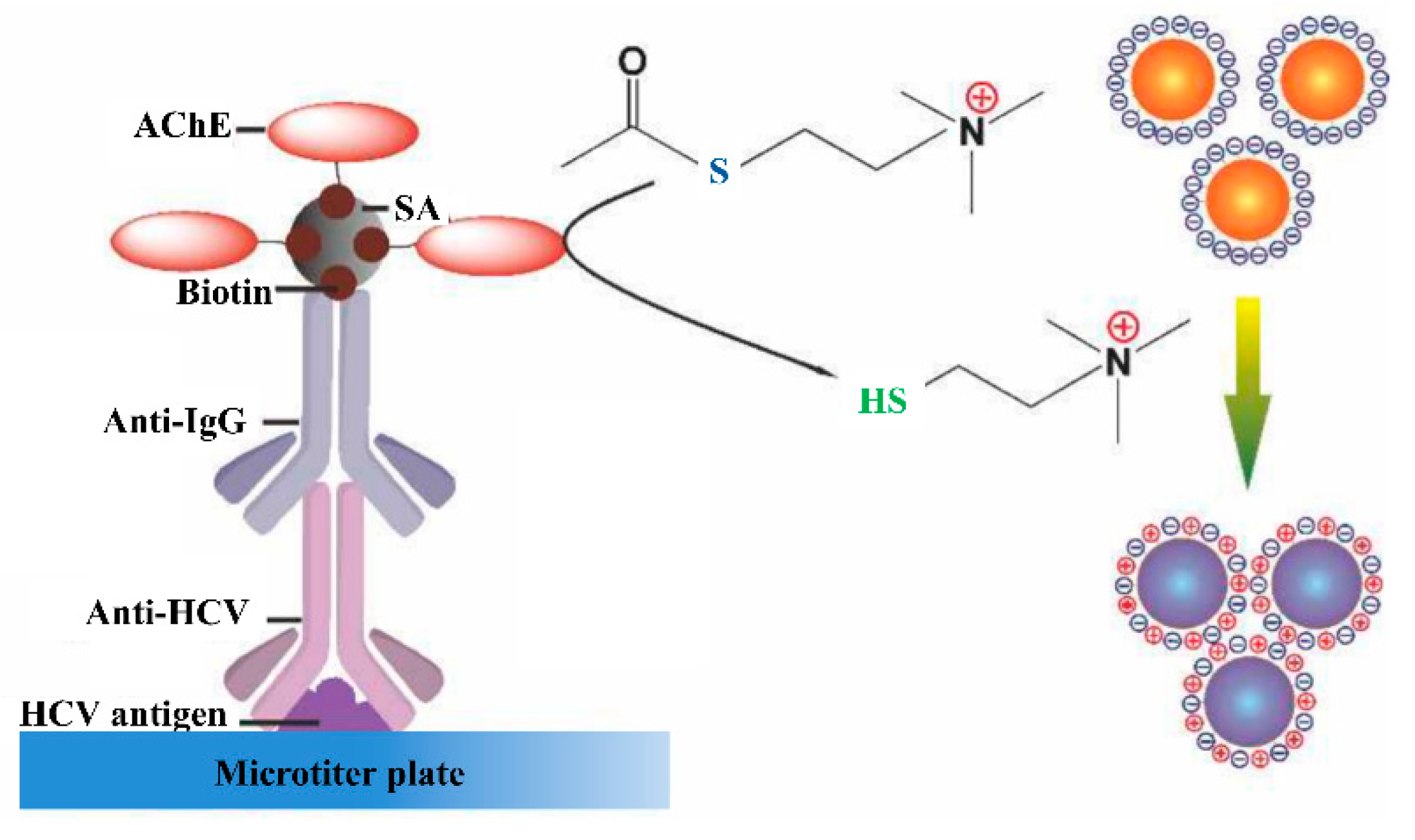
4.1. Anti-Hcv Antibody Detection
4.2. Core Antigen Detection
5. Rapid Immunoassays (RIA)
5.1. Anti-HCV Antibody Detection
5.2. Core Antigen Detection
6. Recombinant Immunoblot Assay (RIBA)
7. Electrochemical Immunosensors (EI)
7.1. Anti-HCV Antibody Detection
7.2. Core Antigen Detection
7.3. Detection of Other Antigens
8. Nanotechnology
8.1. Anti-HCV Antibody Detection
8.2. Core Antigen Detection
8.3. Detection of Other Antigens
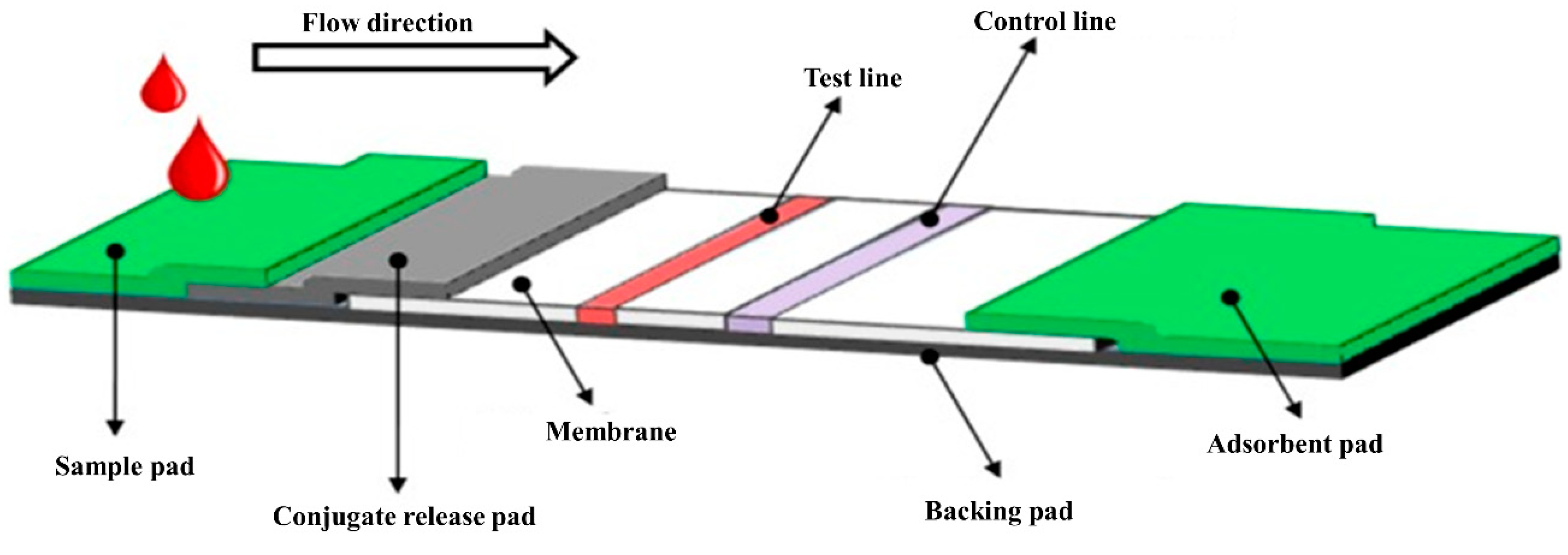
9. Lateral Flow Assay (LFA)
9.1. Anti-HCV Antibody Detection
9.2. Core Antigen Detection
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- WHO. Global Hepatitis Report. 2017. Available online: https://afro.who.int/sites/default/files/2017-06/9789241565455-eng.pdf (accessed on 14 August 2019).
- Li, H.-C.; Lo, S.-Y. Hepatitis C virus: Virology, diagnosis and treatment. World J. Hepatol. 2015, 7, 1377–1389. [Google Scholar] [CrossRef]
- Gravitz, L. Introduction: A smouldering public-health crisis. Nature 2011, 474, S2–S4. [Google Scholar] [CrossRef] [PubMed]
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age–sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar] [CrossRef]
- Alter, H.J.; Seeff, L.B. Recovery, Persistence, and Sequelae in Hepatitis C Virus Infection: A Perspective on Long-Term Outcome. Semin. Liver Dis. 2000, 20, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Al Olaby, R.R.; Azzazy, H.M. Hepatitis C virus RNA assays: Current and emerging technologies and their clinical applications. Expert Rev. Mol. Diagn. 2011, 11, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, M.; Gupta, E.; Choudhary, A. Hepatitis C virus: Screening, diagnosis, and interpretation of laboratory assays. Asian J. Transfus. Sci. 2014, 8, 19–25. [Google Scholar] [CrossRef]
- Douglas, L.; Nguyen, M.D.; Ke, Q. Clinical Monitoring of Chronic Hepatitis C Based on its Natural History and Therapy. Hum. Endocr. Pract. 2013, 19, 292–300. [Google Scholar]
- Smith, B.D.; Morgan, R.L.; A Beckett, G.; Falck-Ytter, Y.; Holtzman, D.; Teo, C.-G.; Jewett, A.; Baack, B.; Rein, D.B.; Patel, N.; et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945–1965. MMWR. Recomm. Rep. 2012, 61, 1–32. [Google Scholar] [PubMed]
- Schnuriger, A.; Dominguez, S.; Valantin, M.-A.; Tubiana, R.; Duvivier, C.; Ghosn, J.; Simon, A.; Katlama, C.; Thibault, V. Early Detection of Hepatitis C Virus Infection by Use of a New Combined Antigen-Antibody Detection Assay: Potential Use for High-Risk Individuals. J. Clin. Microbiol. 2006, 44, 1561–1563. [Google Scholar] [CrossRef]
- Richter, S.S. Laboratory Assays for Diagnosis and Management of Hepatitis C Virus Infection. J. Clin. Microbiol. 2002, 40, 4407–4412. [Google Scholar] [CrossRef]
- Zeuzem, S.; Berg, T.; Gane, E.; Ferenci, P.; Foster, G.R.; Fried, M.W.; Hezode, C.; Hirschfield, G.M.; Jacobson, I.; Nikitin, I.; et al. Simeprevir Increases Rate of Sustained Virologic Response Among Treatment-Experienced Patients with HCV Genotype-1 Infection: A Phase IIb Trial. Gastroenterology 2014, 146, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.N. Hepatitis C Screening. Ochsner J. 2014, 14, 664–668. [Google Scholar] [PubMed]
- Backus, L.I.; Boothroyd, D.B.; Phillips, B.R.; Belperio, P.; Halloran, J.; Mole, L.A. A Sustained Virologic Response Reduces Risk of All-Cause Mortality in Patients with Hepatitis C. Clin. Gastroenterol. Hepatol. 2011, 9, 509–516.e1. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for the Screening, Care and Treatment of Persons with Hepatitis C Infection; WHO Library Cataloguing-in-Publication Data: Geneva, Switzerland, 2014. [Google Scholar]
- Gavin, C.; Andrew, T.; Kelly, C.; Corklin, S.; John, H.; George, D.; Juergen, R.; Jordan, F. Role of Serologic and Molecular Diagnostic Assays in Identification and Management of Hepatitis C Virus Infection. J. Clin. Microbiol. 2016, 54, 265–273. [Google Scholar]
- Amjad, M.; Moudgal, V.; Faisal, M. Laboratory Methods for Diagnosis and Management of Hepatitis C Virus Infection. Lab. Med. 2013, 44, 292–299. [Google Scholar] [CrossRef][Green Version]
- Clerc, O.; Greub, G. Routine use of point-of-care tests: Usefulness and application in clinical microbiology. Clin. Microbiol. Infect. 2010, 16, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Loggi, E.; Galli, S.; Vitale, G.; Di Donato, R.; Vukotic, R.; Grandini, E.; Margotti, M.; Guarneri, V.; Furlini, G.; Galli, C.; et al. Monitoring the treatment of hepatitis C with directly acting antivirals by serological and molecular methods. PLoS ONE 2017, 12, e0187755. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Guidelines for laboratory testing and result reporting of antibody to hepatitis C virus. MMWR Recomm. Rep. 2013, 52, 1–13. [Google Scholar]
- Khuroo, M.S.; Khuroo, N.S.; Khuroo, M.S. Diagnostic Accuracy of Point-of-Care Tests for Hepatitis C Virus Infection: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0121450. [Google Scholar] [CrossRef]
- Freiman, J.M.; Tran, T.M.; Schumacher, S.G.; White, L.F.; Ongarello, S.; Cohn, J.; Philippa, J.E.; Benjamin, P.L.; Claudia, M.D. HCV Core Antigen Testing for Diagnosis of HCV Infection: A systematic review and meta-analysis. Ann. Intern. Med. 2016, 165, 345–355. [Google Scholar] [CrossRef]
- Penin, F.; Dubuisson, J.; Rey, F.A.; Moradpour, D.; Pawlotsky, J.M. Structural biology of hepatitis C virus. Hepatology 2004, 39, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Dubuisson, J. Hepatitis C virus proteins. World J. Gastroenterol. 2007, 13, 2406–2415. [Google Scholar] [CrossRef] [PubMed]
- Reed, K.E.; Rice, C.M. Overview of Hepatitis C Virus Genome Structure, Polyprotein Processing, and Protein Properties. Curr. Top. Microbiol. Immunol. 2000, 242, 55–84. [Google Scholar] [PubMed]
- Nielsen, S.U.; Bassendine, M.F.; Burt, A.D.; Bevitt, D.J.; Toms, G.L. Characterization of the genome and structural proteins of hepatitis C virus resolved from infected human liver. J. Gen. Virol. 2004, 85, 1497–1507. [Google Scholar] [CrossRef] [PubMed]
- El Awady, M.K.; El Abd, Y.S.; Shoeb, H.A.; Tabll, A.A.; Hosny, A.E.D.M.S.; El Shenawy, R.M.; Atef, K.; El Din, N.G.B.; Bahgat, M.M. Circulating viral core and E1 antigen levels as supplemental markers for HCV Chronic hepatitis. Virol. J. 2006, 3, 67. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Delphine, D.; Parissa, V.; Jeanine, S.; Elisabeth, D.; Anne-Marie, R.A. Sensitivity of a rapid immuno-chromatographic test for Hepatitis C antibodies detection. J. Clin. Virol. 2008, 41, 129–133. [Google Scholar]
- Park, J.H.; Jee, M.H.; Kwon, O.S.; Keum, S.J.; Jang, S.K. Infectivity of hepatitis C virus correlates with the amount of envelope protein E2: Development of a new aptamer-based assay system suitable for measuring the infectious titer of HCV. Virology 2013, 439, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Zaaijer, H.L.; Vallari, D.S.; Cunningham, M.; Lesniewski, R.; Reesink, H.W.; Der Van Poel, C.L.; Lelie, P.N. E2 and NS5: New antigens for detection of hepatitis C virus antibodies. J. Med. Virol. 1994, 44, 395–397. [Google Scholar] [CrossRef]
- Xie, L.; Wu, X.-D.; Huang, D.-Z.; Chen, H.-L.; He, L.-X.; Wang, J.; Han, D.-K. Clinical application and analysis of hepatitis C virus NS3 antigen detection by ELISA in human serum. Chin. Med. J. 2007, 120, 294–299. [Google Scholar] [CrossRef]
- Barth, S.; Blohm, L.; Holz, S.; Piechotta, G.; Dammers, C.; Kleines, M.; Melmer, G.; Nebling, E.; Puttman, C.; Albers, J.; et al. Rapid detection of different human anti-HCV immunoglobulins on electrical biochips. Antib. Technol. J. 2014, 4, 23. [Google Scholar] [CrossRef]
- Chaudhary, R.; Burres, E. Detection of Antibody to Envelope (E2) Antigen of Hepatitis C Virus. Can. J. Infect. Dis. 1997, 8, 229–231. [Google Scholar] [CrossRef] [PubMed]
- Santolini, E.; Migliaccio, G.; La Monica, N. Biosynthesis and biochemical properties of the hepatitis C virus core protein. J. Virol. 1994, 68, 3631–3641. [Google Scholar] [PubMed]
- Muerhoff, A.S.; Jiang, L.; Shah, D.O.; Gutierrez, R.A.; Patel, J.; Garolis, C.; Kyrk, C.R.; Leckie, G.; Frank, A.; Stewart, J.L.; et al. Detection of HCV core antigen in human serum and plasma with an automated chemiluminescent immunoassay. Transfusion 2002, 42, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.O.; Chang, C.D.; Jiang, L.X.; Cheng, K.Y.; Muerhoff, A.S.; Gutierrez, R.A.; Leary, T.P.; Desai, S.M.; Batac-Herman, I.V.; Salbilla, V.A.; et al. Combination HCV core antigen and antibody assay on a fully automated chemiluminescence analyzer. Transfusion 2003, 43, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, P.L.; Franchini, M.; Gandini, G.; Antonello, O.; Olzer, D.; Turrini, A.; Aprili, G. HCV core antigen assay. Transfusion 2001, 41, 1172. [Google Scholar] [CrossRef] [PubMed]
- Laperche, S.; Elghouzzi, M.-H.; Morel, P.; Le Marrec, N.; Girault, A.; Bouchardeau, F.; Deschaseaux, M.; Piquet, Y.; Asso-Bonnet, M.; Servant-Delmas, A.; et al. Is an assay for simultaneous detection of hepatitis C virus core antigen and antibody a valuable alternative to nucleic acid testing? Transfusion 2005, 45, 1965–1972. [Google Scholar] [CrossRef] [PubMed]
- Dubuisson, J.; Penin, F.; Moradpour, D. Interaction of hepatitis C virus proteins with host cell membranes and lipids. Trends Cell Boil. 2002, 12, 517–523. [Google Scholar] [CrossRef]
- Deleersnyder, V.; Pillez, A.; Wychowski, C.; Blight, K.; Xu, J.; Hahn, Y.S.; Rice, C.M.; Dubuisson, J. Formation of native hepatitis C virus glycoprotein complexes. J. Virol. 1997, 71, 697–704. [Google Scholar]
- Lin, C.; Lindenbach, B.D.; Prágai, B.M.; McCourt, D.W.; Rice, C.M. Processing in the hepatitis C virus E2-NS2 region: Identification of p7 and two distinct E2-specific products with different C termini. J. Virol. 1994, 68, 5063–5073. [Google Scholar]
- Blight, K.J.; Kolykhalov, A.A.; Rice, C.M. Efficient initiation of HCV RNA replication in cell culture. Science 2000, 290, 1972–1974. [Google Scholar] [CrossRef]
- Lohmann, V.; Körner, F.; Koch, J.; Herian, U.; Theilmann, L.; Bartenschlager, R. Replication of subgenomic hepatitis C virus RNAs in a hepatoma cell line. Science 1999, 285, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, I.C.; Marcotrigiano, J.; Dentzer, T.G.; Rice, C.M. Structure of the catalytic domain of the hepatitis C virus NS2-3 protease. Nature 2006, 442, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Sansonno, D.E.; Dammacco, F.; Rice, C.M.; Blum, H.E.; Moradpour, D.; Wölk, B.; Kräusslich, H.-G. Subcellular Localization, Stability, and trans-Cleavage Competence of the Hepatitis C Virus NS3-NS4A Complex Expressed in Tetracycline-Regulated Cell Lines. J. Virol. 2000, 74, 2293–2304. [Google Scholar]
- Kim, J.; Morgenstern, K.; Lin, C.; Fox, T.; Dwyer, M.; Landro, J.; Chambers, S.; Markland, W.; Lepre, C.; O’Malley, E.; et al. Crystal Structure of the Hepatitis C Virus NS3 Protease Domain Complexed with a Synthetic NS4A Cofactor Peptide. Cell 1996, 87, 343–355. [Google Scholar] [CrossRef]
- Sillanpää, M.; Melén, K.; Porkka, P.; Fagerlund, R.; Nevalainen, K.; Lappalainen, M.; Julkunen, I. Hepatitis C virus core, NS3, NS4B and NS5A are the major immunogenic proteins in humoral immunity in chronic HCV infection. Virol. J. 2009, 6, 84. [Google Scholar] [CrossRef] [PubMed]
- Dufour, D.R.; Talastas, M.; Fernandez, M.D.; Harris, B.; Strader, D.B.; Seeff, L.B. Low-positive anti-hepatitis C virus enzyme immunoassay results: An important predictor of low likelihood of hepatitis C infection. Clin. Chem. 2003, 49, 479–486. [Google Scholar] [CrossRef][Green Version]
- Saludes, V.; González, V.; Planas, R.; Matas, L.; Ausina, V.; Martró, E. Tools for the diagnosis of hepatitis C virus infection and hepatic fibrosis staging. World J. Gastroenterol. 2014, 20, 3431–3442. [Google Scholar] [CrossRef]
- Morota, K.; Fujinami, R.; Kinukawa, H.; Machida, T.; Ohno, K.; Saegusa, H.; Takeda, K. A new sensitive and automated chemiluminescent microparticle immunoassay for quantitative determination of hepatitis C virus core antigen. J. Virol. Methods 2009, 157, 8–14. [Google Scholar] [CrossRef]
- Fondjo, C.L.K.; Ngoupo, P.A.T.; Ngono, L.; Plantier, J.C.; Njouom, R. Performace evaluation of three rapid screening assays for detection of antibodies to hepatitis C virus in Cameroon. BMC Res. Notes 2018, 11(1), 352. [Google Scholar] [CrossRef]
- Martin, P.; Fabrizi, F.; Dixit, V.; Quan, S.; Brezina, M.; Kaufman, E.; Dinello, R.; Polito, A.; Gitnick, G. Automated RIBA Hepatitis C Virus (HCV) Strip Immunoblot Assay for Reproducible HCV Diagnosis. J. Clin. Microbiol. 1998, 36, 387–390. [Google Scholar]
- Duan, L.; Wang, Y.; Li, S.S.; Wan, Z.; Zhai, J. Rapid and simultaneous detection of human hepatitis B virus and hepatitis C virus antibodies based on a protein chip assay using nano-gold immunological amplifi cation and silver staining method. BMC Infect. Dis. 2005, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.-Q.; Ji, C.-F.; Yang, X.-Q.; Wang, R.; Yang, S.; Zhang, H.-Q.; Zhang, J.-G. An improved gold nanoparticle probe-based assay for HCV core antigen ultrasensitive detection. J. Virol. Methods 2017, 243, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Changhyun, R.; Ho-Young, L.; Sang-Eun, K.; Sung-Kee, J. Quantum-dots-based detection of hepatitis C virus (HCV) NS3 using RNA aptamer on chip. J. Chem. Technol. Biotechnol. 2010, 85, 1130–1134. [Google Scholar]
- Roh, C.; Lee, H.Y.; Kim, S.E.; Jo, S.K. A highly sensitive and selective viral protein detection method based on RNA oligonucleotide nanoparticle. Int. J. Nanomed. 2010, 5, 323. [Google Scholar] [CrossRef]
- Mikawa, A.Y.; Santos, S.A.T.; Kenfe, F.R.; Da Silva, F.H.; Da Costa, P.I. Development of a rapid one-step immunochromatographic assay for HCV core antigen detection. J. Virol. Methods 2009, 158, 160–164. [Google Scholar] [CrossRef]
- Xiang, T.; Jiang, Z.; Zheng, J.; Lo, C.; Tsou, H.; Ren, G.; Zhang, J.; Huang, A.; Lai, G. A novel double antibody sandwich-lateral flow immunoassay for the rapid and simple detection of hepatitis C virus. Int. J. Mol. Med. 2012, 30, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, Y.S.; Jo, M.; Jin, M.; Lee, D.-K.; Kim, S. Chip-based detection of hepatitis C virus using RNA aptamers that specifically bind to HCV core antigen. Biochem. Biophys. Res. Commun. 2007, 358, 47–52. [Google Scholar] [CrossRef]
- Pleshakova, T.O.; Kaysheva, A.L.; Shumov, I.D.; Ziborov, V.S.; Bayzyanova, J.M.; Konev, V.A.; Uchaikin, V.F.; Archakov, A.I.; Ivanov, Y.D. Detection of Hepatitis C Virus Core Protein in Serum Using Aptamer-Functionalized AFM Chips. Micromachines 2019, 10, 129. [Google Scholar] [CrossRef]
- Hügle, T.; Fehrmann, F.; Bieck, E.; Kohara, M.; Kräusslich, H.-G.; Rice, C.M.; Blum, H.E.; Moradpour, D. The Hepatitis C Virus Nonstructural Protein 4B Is an Integral Endoplasmic Reticulum Membrane Protein. Virology 2001, 284, 70–81. [Google Scholar] [CrossRef]
- Lundin, M.; Monné, M.; Widell, A.; Von Heijne, G.; Persson, M.A.A. Topology of the Membrane-Associated Hepatitis C Virus Protein NS4B. J. Virol. 2003, 77, 5428–5438. [Google Scholar] [CrossRef]
- Brass, V.; Bieck, E.; Montserret, R.; Wölk, B.; Hellings, J.A.; Blum, H.E.; Penin, F.; Moradpour, D. An amino-terminal amphipathic alpha-helix mediates membrane association of the hepatitis C virus nonstructural protein 5A. J. Biol. Chem. 2002, 277, 8130–8139. [Google Scholar] [CrossRef]
- Penin, F.; Ramboarina, S.; Montserret, R.; Ficheux, D.; Moradpour, D.; Brass, V.; Appel, N.; Blum, H.E.; Bartenschlager, R. Structure and Function of the Membrane Anchor Domain of Hepatitis C Virus Nonstructural Protein 5A. J. Boil. Chem. 2004, 279, 40835–40843. [Google Scholar] [CrossRef] [PubMed]
- Ivashkina, N.; Wölk, B.; Lohmann, V.; Bartenschlager, R.; Blum, H.E.; Penin, F.; Moradpour, D. The Hepatitis C Virus RNA-Dependent RNA Polymerase Membrane Insertion Sequence Is a Transmembrane Segment. J. Virol. 2002, 76, 13088–13093. [Google Scholar] [CrossRef] [PubMed]
- Moradpour, D.; Brass, V.; Bieck, E.; Friebe, P.; Gosert, R.; Blum, H.E.; Bartenschlager, R.; Penin, F.; Lohmann, V. Membrane Association of the RNA-Dependent RNA Polymerase Is Essential for Hepatitis C Virus RNA Replication†. J. Virol. 2004, 78, 13278–13284. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schmidt-Mende, J.; Bieck, E.; Hügle, T.; Penin, F.; Rice, C.M.; Blum, H.E.; Moradpour, D. Determinants for Membrane Association of the Hepatitis C Virus RNA-dependent RNA Polymerase. J. Boil. Chem. 2001, 276, 44052–44063. [Google Scholar] [CrossRef] [PubMed]
- Barrera, J.M.; Franscis, B.; Ercilla, G.; Nelles, M.; Archod, D.; Darner, J.; Lee, S.R. Improved detection of Anti-HCV in Post transfuson Hepatitis by third generation ELISA. Vox Sang. 1995, 68, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Morishima, C.; Gretch, D.R. Clinical use of Hepatitis C virus test for the diagnosis and monitoring during therapy. Clin. Liver Dis. 1999, 3, 717–740. [Google Scholar] [CrossRef]
- Lanoir, D.; Touzet, S.; Meyaud-Kraemer, L.; Trepo, C.; Colin, C.; Bailly, F. The HEPATIS Group. Sensitivity and specificity of third-generation hepatitis C virus antibody detection assays: An analysis of the literature. J. Viral Hepat. 2001, 8, 87–95. [Google Scholar]
- Hamid, M.S.; Mitra, R.; Alizadeh, A.H.M. Recommendations for Prevention and Control of Hepatitis C Virus (HCV) Infection and HCV-Related Chronic Disease. J. Liver 2013, 3, 147. [Google Scholar]
- Alter, H.J. New kit on the block: Evaluation of second-generation assays for detection of antibody to the hepatitis C virus. Hepatology 1992, 15, 350–353. [Google Scholar] [CrossRef]
- Araujo, A.C.; Hayden, T.M.; Kamili, S.; Drobeniuc, J. Laboratory Diagnostics for Hepatitis C Virus Infection. Clin. Infect. Dis. 2012, 55, S43–S48. [Google Scholar]
- Chen, S.L. The Natural History of Hepatitis C Virus (HCV) Infection. Int. J. Med. Sci. 2006, 3, 47. [Google Scholar] [CrossRef] [PubMed]
- Griffin, J.F.T.; Spittle, E.; Rodgers, C.R.; Liggett, S.; Cooper, M.; Bakker, D.; Bannantine, J.P. Immunoglobulin G1 Enzyme-Linked Immunosorbent Assay for Diagnosis of Johne’s Disease in Red Deer (Cervus elaphus). Clin. Diagn. Lab. Immunol. 2005, 12, 1401–1409. [Google Scholar] [CrossRef] [PubMed]
- Swellam, M.; Mahmoud, M.S.; Ali, A.A.-F. Diagnosis of hepatitis C virus infection by enzyme-linked immunosorbent assay and reverse transcriptase-nested polymerase chain reaction: A comparative evaluation. IUBMB Life 2011, 63, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, B.; Ferretti, F.; Vindigni, C.; Terzuoli, L. Assessment of a Test for the Screening and Diagnosis of Celiac Disease. J. Clin. Lab. Anal. 2014, 30, 65–70. [Google Scholar] [CrossRef]
- Cano, H.; Candela, M.J.; Lozano, M.L.; Vicente, V. Application of a new enzyme-linked immunosorbent assay for detection of total hepatitis C virus core antigen in blood donors. Transfus. Med. 2003, 13, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Lopes, E.; Granato, C.; Lanzoni, V.; Granero, L.; Paranhos-Baccalà, G.; Tomiyama, H.; Silva, A.; Ferraz, M. Evaluation of an enzyme immunoassay for hepatitis C virus antibody detection using a recombinant protein derived from the core region of hepatitis C virus genome. Memórias do Instituto Oswaldo Cruz 2000, 95, 717–720. [Google Scholar] [CrossRef]
- Laperche, S.; Le Marrec, N.; Simon, N.; Bouchardeau, F.; Defer, C.; Maniez-Montreuil, M.; Levayer, T.; Zappitelli, J.-P.; Lefrère, J.-J.; Maniez-Montreuil, M.; et al. A new HCV core antigen assay based on disassociation of immune complexes: An alternative to molecular biology in the diagnosis of early HCV infection. Transfusion 2003, 43, 958–962. [Google Scholar] [CrossRef]
- Icardi, G.; Ansaldi, F.; Bruzzone, B.M.; Durando, P.; Lee, S.; De Luigi, C.; Crovari, P. Novel Approach to Reduce the Hepatitis C Virus (HCV) Window Period: Clinical Evaluation of a New Enzyme-Linked Immunosorbent Assay for HCV Core Antigen. J. Clin. Microbiol. 2001, 39, 3110–3114. [Google Scholar] [CrossRef]
- Massaguer, A.; Forns, X.; Costa, J.; Feliu, A.; García-Retortillo, M.; Navasa, M.; Rimola, A.; García-Valdecasas, J.C.; Sánchez-Tapias, J.M. Performance of Hepatitis C Virus Core Antigen Immunoassay in Monitoring Viral Load after Liver Transplantation. Transplantation 2005, 79, 1441–1444. [Google Scholar] [CrossRef]
- Alzahrani, A.J.; Obeid, O.E. Detection of hepatitis c virus core antigen in blood donors using a new enzyme immunoassay. J. Fam. Community Med. 2004, 11, 103–107. [Google Scholar]
- Garbuglia, A.R.; Monachetti, A.; Galli, C.; Sabatini, R.; Ferreri, M.L.; Capobianchi, M.R.; Bagnarelli, P. HCV core antigen and HCV-RNA in HIV/HCV co-infected patients with different HCV genotypes. BMC Infect. Dis. 2014, 14, 222. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Chang, K.C.; Wang, J.H.; Tsai, P.S.; Hung, S.F.; Hung, C.H.; Chen, C.H.; Lu, S.N. Is hepatitis C virus core antigen an adequate marker forcommunity screening? J. Clin. Microbiol. 2012, 50, 1989–1993. [Google Scholar] [CrossRef] [PubMed]
- Ottiger, C.; Gygli, N.; Huber, A.R. Detection limit of architect hep-atitis C core antigen assay in correlation with HCV RNA, andrenewed confirmation algorithm for reactive anti-HCV samples. J. Clin. Virol. 2013, 58, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Hu, K.-Q.; Hu, K. A highly specific and sensitive hepatitis C virus antigen enzyme immunoassay for One-step diagnosis of viremic hepatitis C virus infection. Hepatology 2016, 64, 415–424. [Google Scholar]
- Hu, K.-Q.; Cui, W.; Rouster, S.D.; Sherman, K.E. Hepatitis C virus antigens enzyme immunoassay for one-step diagnosis of hepatitis C virus coinfection in human immunodeficiency virus infected individuals. World J. Hepatol. 2019, 11, 442–449. [Google Scholar] [PubMed]
- Dufour, D.R.; Talastas, M.; Fernandez, M.D.; Harris, B. Chemiluminescence Assay Improves Specificity of Hepatitis C Antibody Detection. Clin. Chem. 2003, 49, 940–944. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cinquanta, L.; Fontana, D.E.; Bizzaro, N. Chemiluminescent immunoassay technology: What does it change in autoantibody detection? Auto Immun. Highlights 2017, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.; Fish, G.E.; Smith, M.B. Laboratory Evaluation of a Fully Automated Chemiluminescence Immunoassay for Rapid Detection of HBsAg, Antibodies to HBsAg, and Antibodies to Hepatitis C Virus. J. Clin. Microbiol. 2004, 42, 610–617. [Google Scholar] [CrossRef]
- Sinyoung, K.; Jeong-Ho, K.; Seoyoung, Y.; Youn-Hee, P.; Hyon-Suk, K. Clinical Performance Evaluation of Four Automated Chemiluminescence Immunoassays for Hepatitis C Virus Antibody Detection. J. Clin. Microbiol. 2008, 46, 3919–3923. [Google Scholar]
- Tang, W.; Chen, W.; Amini, A.; Boeras, D.; Falconer, J.; Kelly, H.; Peeling, R.; Varsaneux, O.; Tucker, J.D.; Easterbrook, P. Diagnostic accuracy of tests to detect Hepatitis C antibody: A meta-analysis and review of the literature. BMC Infect. Dis. 2017, 17, 695. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Wei, B.; Liu, Q.; Wang, T.; Li, D.; Rao, C.; Tao, C.; Wang, L. Evaluation of the Novel HISCL Chemiluminescence Enzyme Immunoassay for Laboratory Screening of Hepatitis C Virus. Clin. Vaccine Immunol. 2016, 23, 652–654. [Google Scholar] [CrossRef] [PubMed]
- Rockstroh, J.K.; Feld, J.J.; Chevaliez, S.; Cheng, K.; Wedemeyer, H.; Sarrazin, C.; Maasoumy, B.; Herman, C.; Hackett, J.; Cohen, D.E.; et al. HCV core antigen as an alternate test to HCV RNA for assessment of virologic responses to all-oral, interferon-free treatment in HCV genotype 1 infected patients. J. Virol. Methods 2017, 245, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, L.; Fu, C.; Wang, Y.; Sun, S. Employment of 4-(1,2,4-triazol-1-yl)phenol as a signal enhancer of the chemiluminescent luminol-H2O2-horseradish peroxidase reaction for detection of hepatitis C virus in real samples. Luminescence 2015, 30, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, R.C. Laboratory Turnaround Time. Clin. Biochem. Rev. 2007, 28, 179–194. [Google Scholar] [PubMed]
- Smith, B.D.; Jewett, A.; Drobeniuc, J.; Kamili, S. Rapid diagnostic HCV antibody assays. Antivir. Ther. 2012, 17, 1409–1413. [Google Scholar] [CrossRef]
- Vercauteren, G. Hepatitis C Assays: Operational Characteristics (Phase i); Report 1, 2001, WHO/BCT/BTS/01.2; Blood Safety and Clinical Technology, World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Chevaliez, S.; Poiteau, L.; Rosa, I.; Soulier, A.; Roudot-Thoraval, F.; Laperche, S.; Hezode, C.; Pawlotsky, J.-M. Prospective assessment of rapid diagnostic tests for the detection of antibodies to hepatitis C virus, a tool for improving access to care. Clin. Microbiol. Infect. 2016, 22, 459.e1–459.e6. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.P.; Khan, H.I.; Cubitt, W.D. Detection of Antibodies to Hepatitis C Virus in Dried Blood Spot Samples from Mothers and Their Offspring in Lahore, Pakistan. J. Clin. Microbiol. 1999, 37, 2061–2063. [Google Scholar]
- Daniel, H.D.J.; Abraham, P.; Raghuraman, S.; Vivekanandan, P.; Subramaniam, T.; Sridharan, G. Evaluation of a Rapid Assay as an Alternative to Conventional Enzyme Immunoassays for Detection of Hepatitis C Virus-Specific Antibodies. J. Clin. Microbiol. 2005, 43, 1977–1978. [Google Scholar] [CrossRef]
- Firdaus, R.; Saha, K.; Sadhukhan, P.C. Rapid immunoassay alone is insufficient for the detection of hepatitis C virus infection among high-risk population. J. Viral. Hepat. 2013, 20, 290–293. [Google Scholar] [CrossRef]
- Skidmore, S. Recombinant immunoblot assay for hepatitis C antibody. Lancet 1990, 335, 1346. [Google Scholar] [CrossRef]
- Van Der Poel, C.; Reesink, H.; Van Boven, J.; Cuypers, H.; Winkel, I.; Exel-Oehlers, P.; Schaasberg, W.; Lelie, P.; Weiner, A.; Polito, A.; et al. Confirmation of hepatitis C virus infection by new four-antigen recombinant immunoblot assay. Lancet 1991, 337, 317–319. [Google Scholar] [CrossRef]
- Contreras, A.M.; Toribio, J.G.; Celis, A.; Rivera, P.K.; Méndez, C.; Olivares, L.; Alvarado, M.A.; Tornero-Romo, C.M.; Orozco-Hernández, A.; Hernández-Lugo, M.I.; et al. Very low hepatitis C antibody levels predict false-positive results and avoid supplemental testing. Transfusion 2008, 48, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Feucht, H.H.; Zöllner, B.; Polywka, S.; Laufs, R. Study on Reliability of Commercially Available Hepatitis C Virus Antibody Tests. J. Clin. Microbiol. 1995, 33, 620–624. [Google Scholar]
- Rafik, M.; Bakr, S.; Soliman, D.; Mohammed, N.; Ragab, D.; Elhady, W.A.; Samir, N. Characterization of differential antibody production against hepatitis C virus in different HCV infection status. Virol. J. 2016, 13, 116. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.M.; Zarife, M.A.S.; Reis, E.A.G.; Reis, M.G. Indeterminate RIBA results were associated with the absence of hepatitis C virus RNA (HCV-RNA) in blood donors. Rev. Soc. Bras. Med. Trop. 2014, 47, 12–17. [Google Scholar] [CrossRef][Green Version]
- Moorman, A.C.; Drobenuic, J.; Kamili, S. Prevalence of false-positive hepatitis C antibody results, National Health and Nutrition Examination Study (NHANES) 2007–2012. J. Clin. Virol. 2017, 89, 1–4. [Google Scholar] [CrossRef]
- Tang, D.; Yuan, R.; Chai, Y. Ultrasensitive Electrochemical Immunosensor for Clinical Immunoassay Using Thionine-Doped Magnetic Gold Nanospheres as Labels and Horseradish Peroxidase as Enhancer. Anal. Chem. 2008, 80, 1582–1588. [Google Scholar] [CrossRef]
- Mani, V.; Chikkaveeraiah, B.V.; Patel, V.; Gutkind, J.S.; Rusling, J.F. Ultrasensitive immunosensor for cancer biomarker proteins using gold nanoparticle film electrodes and multienzyme-particle amplification. ACS Nano 2009, 3, 585–594. [Google Scholar] [CrossRef]
- Lu, L.; Liu, B.; Li, S.; Zhang, W.; Xie, G. Improved electrochemical immunosensor for myeloperoxidase in human serum based on nanogold/cerium dioxide-BMIMPF6/l-Cysteine composite film. Colloids Surfaces B Biointerfaces 2011, 86, 339–344. [Google Scholar] [CrossRef]
- Moraes, M.L.; Lima, L.R.; Silva, R.R.; Cavicchioli, M.; Ribeiro, S.J.L. Immunosensor Based on Immobilization of Antigenic Peptide NS5A-1 from HCV and Silk Fibroin in Nanostructured Films. Langmuir 2013, 29, 3829–3834. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Xie, G.; Zhang, W.; Liang, M.; Liu, B.; Xiang, H. Label-free sandwich type of immunosensor for hepatitis C viruscore antigen based on the use of gold nanoparticles on a nanostructured metal oxide surface. Microchim. Acta 2012, 178, 331–340. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, X. A portable paper-based microfluidic platform for multiplexed electrochemical detection of human immunodeficiency virus and hepatitis C virus antibodies in serum. Biomicrofluidics 2016, 10, 024119. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Liang, M.; Wang, L.; Xiang, H.; Jiang, Y.; Li, Y.; Xie, G. MultisHRP-DNA-coated CMWNTs as signal labels for an ultrasensitive hepatitis C virus core antigen electrochemical immunosensor. Biosens. Bioelectron. 2013, 47, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Valipour, A.; Roushani, M. Using Boehmite Nanoparticles as an Undercoat, and Riboflavin as a Redox Probe for Immunosensor Designing: Ultrasensitive Detection of Hepatitis C Virus Core Antigen. Anal. Bioanal. Chem. Res. 2018, 5, 353–361. [Google Scholar]
- Liang, M.; Wang, L.; Ma, C.; Zhang, M.; Xie, G. Sandwich Immunoassay for Hepatitis C Virus Non-Structural 5A Protein Using a Glassy Carbon Electrode Modified with an Au-MoO 3 /Chitosan Nanocomposite. Anal. Lett. 2013, 46, 1241–1254. [Google Scholar] [CrossRef]
- Abraham, A.M.; Kannangai, R.; Sridharan, G. Nanotechnology: A new frontier in virus detection in clinical practice. Indian J. Med Microbiol. 2008, 26, 297. [Google Scholar]
- Suni, I.I. Impedance methods for electrochemical sensors using nanomaterials. TrAC Trends Anal. Chem. 2008, 27, 604–611. [Google Scholar] [CrossRef]
- Wang, J.; Lin, Y. Functionalized carbon nanotubes and nanofibers for biosensing applications. TrAC Trends Anal. Chem. 2008, 27, 619–626. [Google Scholar] [CrossRef]
- Cui, R.; Huang, H.; Yin, Z.; Gao, D.; Zhu, J.-J. Horseradish peroxidase-functionalized gold nanoparticle label for amplified immunoanalysis based on gold nanoparticles/carbon nanotubes hybrids modified biosensor. Biosens. Bioelectron. 2008, 23, 1666–1673. [Google Scholar] [CrossRef]
- Jie, G.; Zhang, J.; Wang, D.; Cheng, C.; Chen, H.-Y.; Zhu, J.-J. Electrochemiluminescence Immunosensor Based on CdSe Nanocomposites. Anal. Chem. 2008, 80, 4033–4039. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, K.; Zhou, J.; Xuan, J.; Yan, W.; Jiang, L.-P.; Zhu, J.-J. Electrochemical immunosensor based on colloidal carbon sphere array. Biosens. Bioelectron. 2010, 25, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-H.; Tang, H.; Jiang, J.-H. Enzyme mediated assembly of gold nanoparticles for ultrasensitive colorimetric detection of hepatitis C virus antibody. Anal. Methods 2017, 9, 3777–3781. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, G.-X. [A protein array based on quantum dots (QDs) encoded microbeads for detection of hepatitis C virus]. Zhonghua shi yan he lin chuang bing du xue za zhi Zhonghua shiyan he linchuang bingduxue zazhi Chin. J. Exp. Clin. Virol. 2013, 27, 67–69. [Google Scholar]
- Roh, C. A Facile Inhibitor Screening of Hepatitis C Virus NS3 Protein Using Nanoparticle-Based RNA. Biosensors 2012, 2, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Koczula, K.M.; Gallotta, A. Lateral flow assays. Essays Biochem. 2016, 30, 111–120. [Google Scholar] [CrossRef]
- Kosack, C.S.; Nick, S.; Shanks, L. Diagnostic accuracy evaluation of the ImmunoFlow HCV rapid immunochromatographic test for the detection of hepatitis C antibodies. J. Virol. Methods 2014, 204, 6–10. [Google Scholar] [CrossRef]
- Lee, S.R.; Kardos, K.W.; Schiff, E.; Berne, C.A.; Mounzer, K.; Banks, A.T.; Tatum, H.A.; Friel, T.J.; Demicco, M.P.; Lee, W.M.; et al. Evaluation of a new, rapid test for detecting HCV infection, suitable for use with blood or oral fluid. J. Virol. Methods 2011, 172, 27–31. [Google Scholar] [CrossRef]
- Cha, Y.J.; Park, Q.; Kang, E.-S.; Yoo, B.C.; Park, K.U.; Kim, J.-W.; Hwang, Y.-S.; Kim, M.H. Performance Evaluation of the OraQuick Hepatitis C Virus Rapid Antibody Test. Ann. Lab. Med. 2013, 33, 184–189. [Google Scholar] [CrossRef]
- Gao, F.; Talbot, E.A.; Loring, C.H.; Power, J.J.; Dionne-Odom, J.; Alroy-Preis, S.; Jackson, P.; Bean, C.L. Performance of the OraQuick HCV Rapid Antibody Test for Screening Exposed Patients in a Hepatitis C Outbreak Investigation. J. Clin. Microbiol. 2014, 52, 2650–2652. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, L.; Shen, X. Development of a Nucleic Acid Lateral Flow Strip for Detection of Hepatitis C Virus (Hcv) Core Antigen. Nucleotides Nucleic Acids 2013, 32, 59–68. [Google Scholar] [CrossRef] [PubMed]
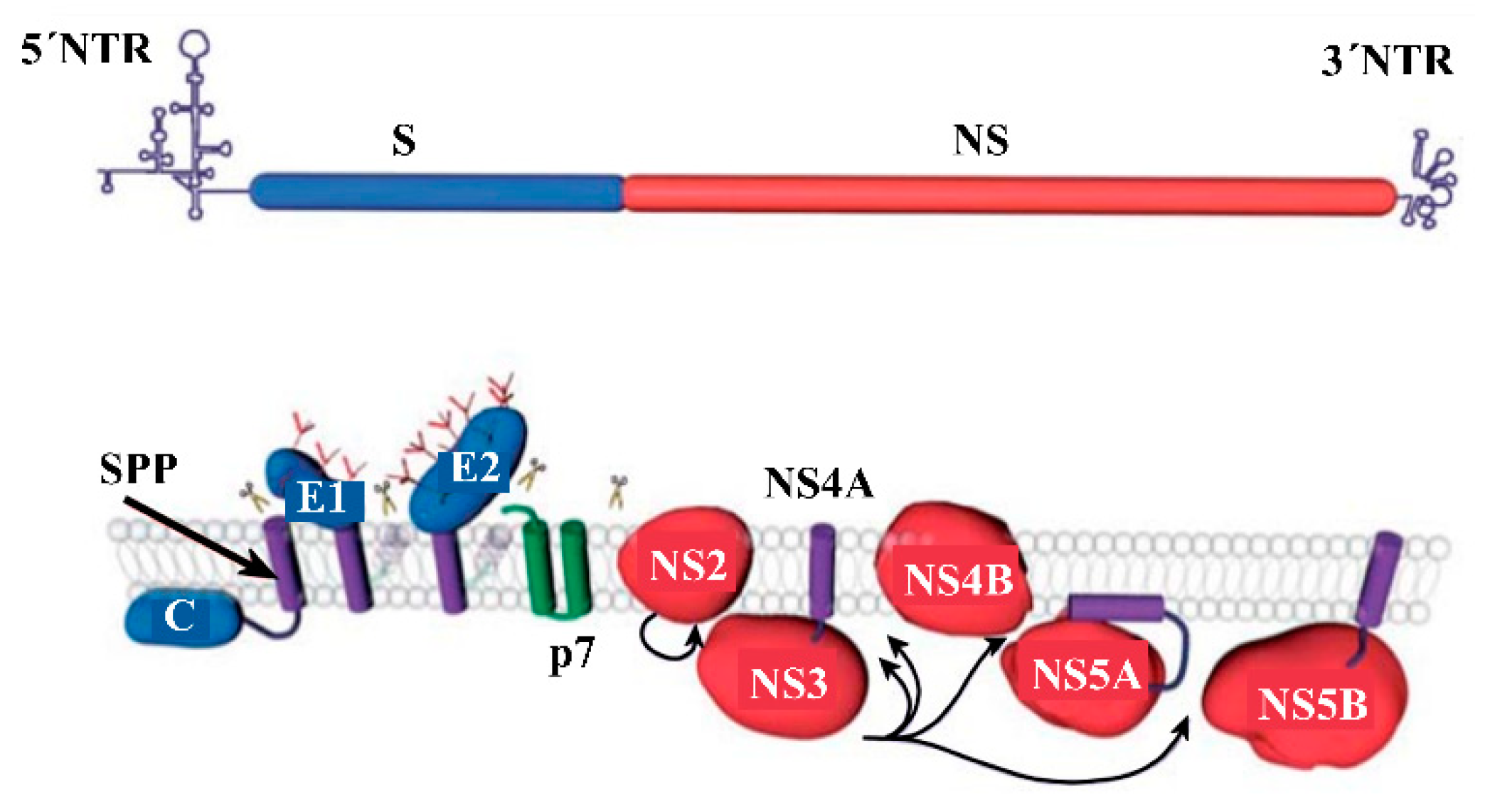
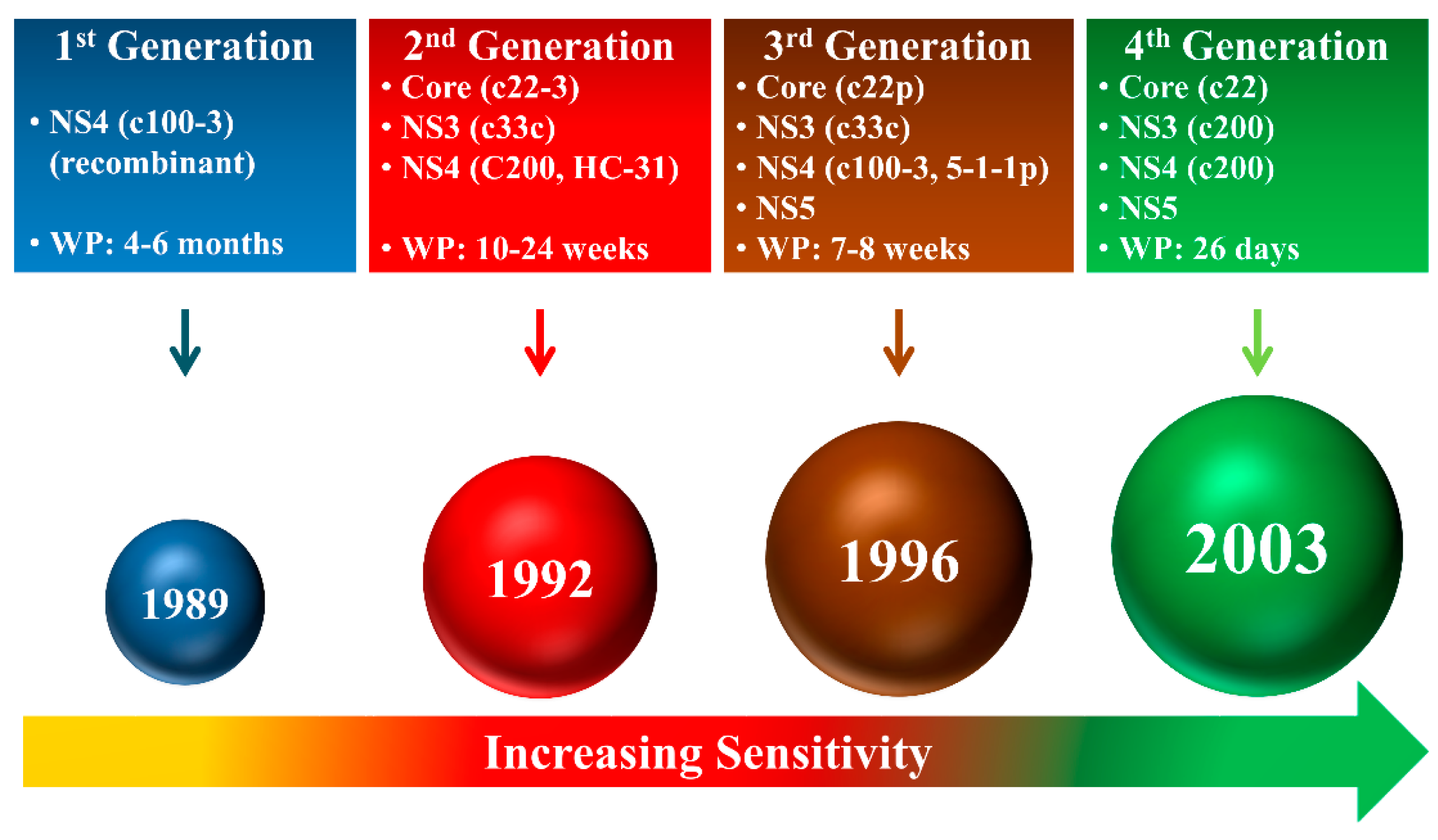
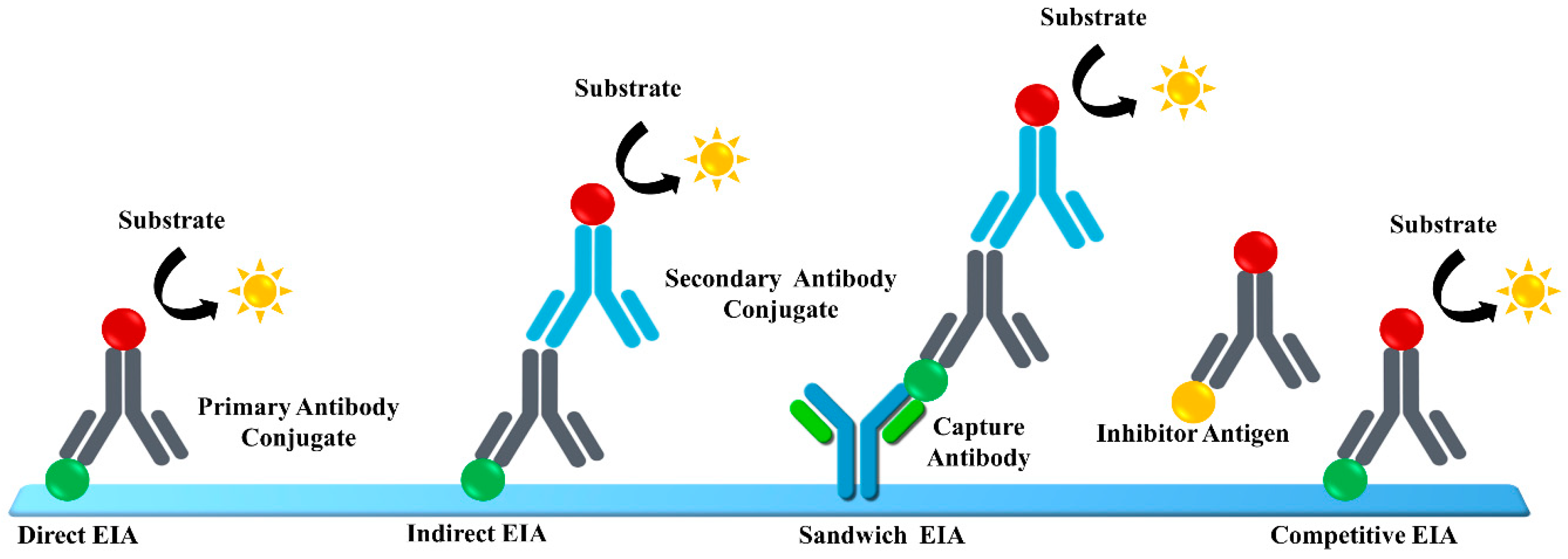


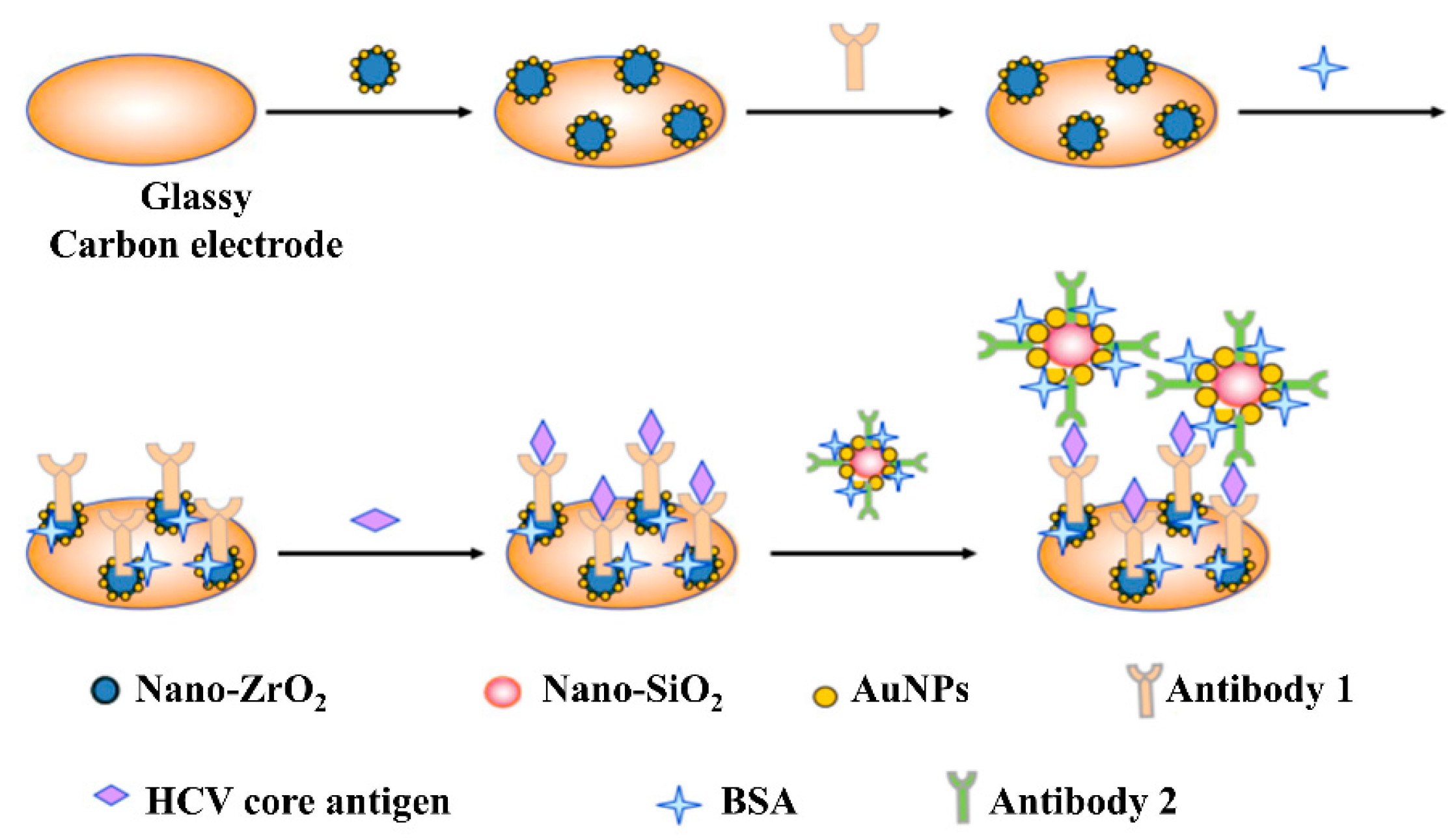
| HCV Proteins | Genetic Stability | Function | Molar Mass | HCV Antigen Detection? | Anti-HCV Antibody Detection? |
|---|---|---|---|---|---|
| Structural Proteins | |||||
| Core (C): P22 | Stable | A significant component of viral nucleocapsid, Binds viral RNA during assembly | 20 KDa [26] | Yes [27] | Yes [28] |
| E: gp 35 envelope glycoproteins | A high degree of genetic diversity | Receptor binding and HCV entry into target cells | 31 kDa [26] | Yes [27] | - |
| E2: gp 70 envelope glycoproteins | 62 kDa [26] | Yes [29,30] | - | ||
| Non-Structural Proteins | |||||
| NS1: p7 small polypeptide | Stable | Ion channel localized to plasma membrane | 7 KDa [24] | - | - |
| NS2: p23 | - | Component of NS2-3 proteinase | 21 KDa [24] | - | - |
| NS3: p70 | - | Serine protease and RNA helicase | 69 KDa [24] | Yes [31] | Yes [7] |
| NS4A: p8 | Stable | Protease cofactor | 6 KDa [24] | - | Yes [32] |
| NS4B: p27 | Stable | Proteins | 27 KDa [24] | - | Yes [7] |
| Components of the viral replicase complex | |||||
| NS5A:p56/58 | Stable | Cofactor for NS5B | 56 KDa [24] | Yes [33] | Yes [7] |
| Regulate response to INF-α treatment | |||||
| NS5B: p68 | Stable | RNA dependent polymerase | 68 KDa [24] | - | Yes [7] |
| Technology | HCV Proteins | Core | E1 | E2 | NS1 | NS2 | NS3 | NS4A | NS4 | NS5A | NS5B | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EIA | Ab | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [31,47,48] | ||||
| Ag | ✓ | ✓ | ||||||||||
| CIA | Ab | ✓ | ✓ | ✓ | ✓ | ✓ | [28,35,49,50] | |||||
| Ag | ✓ | |||||||||||
| RA | Ab | ✓ | ✓ | ✓ | ✓ | [21,51] | ||||||
| Ag | ||||||||||||
| RIBA | Ab | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [30,52] | |||
| Ag | ||||||||||||
| EI | Ab | [52] | ||||||||||
| Ag | ✓ | ✓ | ||||||||||
| NT | Ab | ✓ | ✓ | ✓ | [53,54,55,56] | |||||||
| Ag | ✓ | ✓ | ✓ | |||||||||
| LFA | Ab | ✓ | ✓ | ✓ | [57,58] | |||||||
| Ag | ✓ | |||||||||||
| ABA | Ab | [29,59,60] | ||||||||||
| Ag | ✓ | ✓ |
| Assay | Assay Principle | Solid Phase | HCV Antigen | Reaction Time (min) |
|---|---|---|---|---|
| Architect Anti-HCV, Abbott Laboratories | ECIA | Paramagnetic Particles | Core, NS3, NS4 | 29 |
| The LIAISON® XL murex HCV Ab, DiaSorin | CIA | Paramagnetic Particles | Core, NS3, NS4 | 46 |
| Vitros Anti-HCV, Ortho Clinical Diagnostics | CIA | Microwell | Core, NS3, NS4, NS5 | 55 |
| Elecsys Anti-HCV, Roche Diagnostics | ECA | Paramagnetic Particles | Core, NS3, NS4 | 18 |
| ADVIA Centaur HCV Assay, Siemens | CIA | Magnetic Particles | C22-3 (core), NS3, c200, NS5 | 58 |
| Access HCV Ab PLUS, Bio-Rad Laboratories | CIA | Paramagnetic Particles | Core, NS3, NS4, NS5 | 55 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warkad, S.D.; Song, K.-S.; Pal, D.; Nimse, S.B. Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies. Sensors 2019, 19, 4257. https://doi.org/10.3390/s19194257
Warkad SD, Song K-S, Pal D, Nimse SB. Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies. Sensors. 2019; 19(19):4257. https://doi.org/10.3390/s19194257
Chicago/Turabian StyleWarkad, Shrikant Dashrath, Keum-Soo Song, Dilipkumar Pal, and Satish Balasaheb Nimse. 2019. "Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies" Sensors 19, no. 19: 4257. https://doi.org/10.3390/s19194257
APA StyleWarkad, S. D., Song, K.-S., Pal, D., & Nimse, S. B. (2019). Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies. Sensors, 19(19), 4257. https://doi.org/10.3390/s19194257