Unobtrusive Vital Sign Monitoring in Automotive Environments—A Review
Abstract
1. Introduction
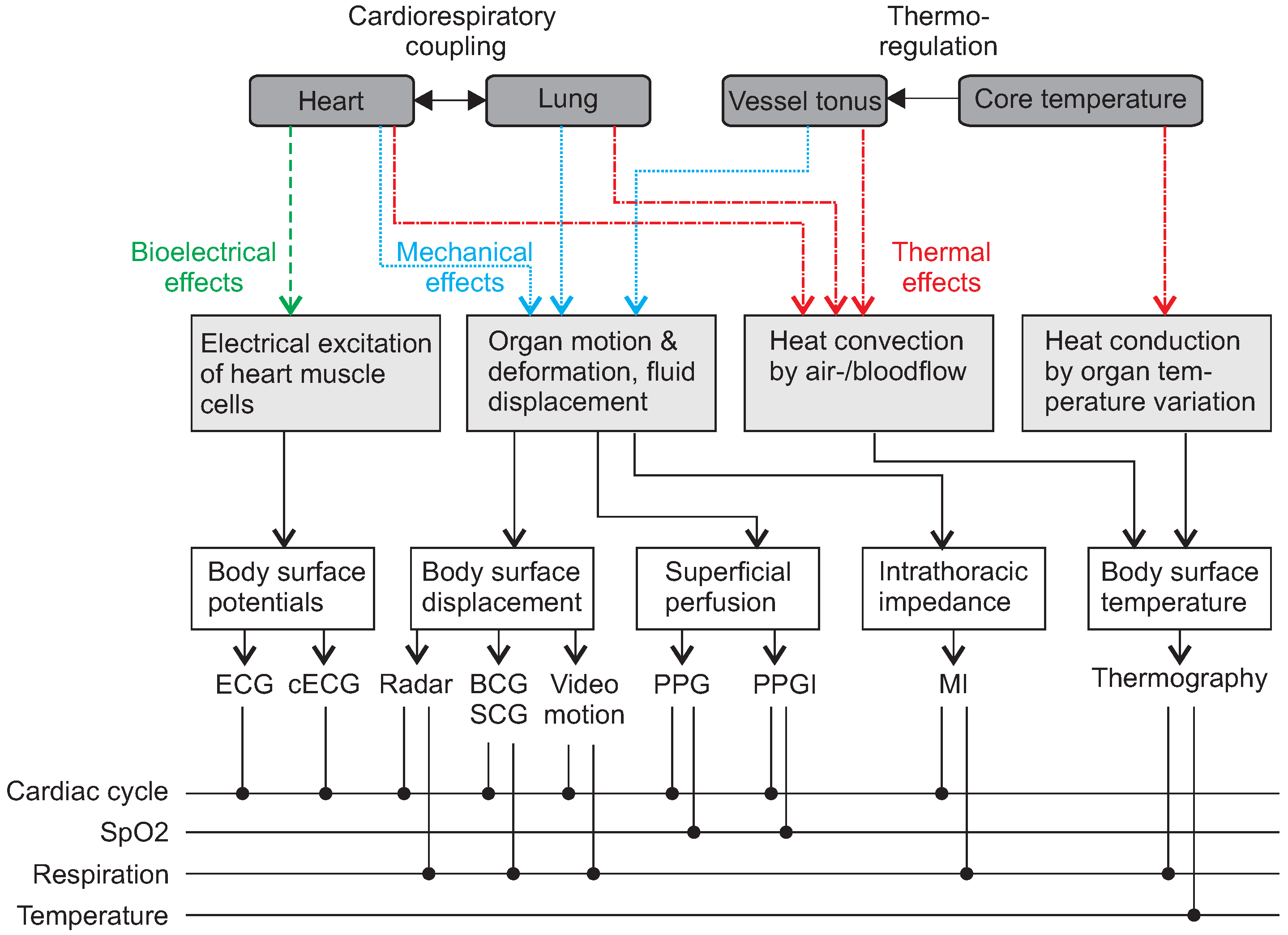
2. Unobtrusive Sensing of Physiological Parameters and Features
3. ECG Monitoring in a Car
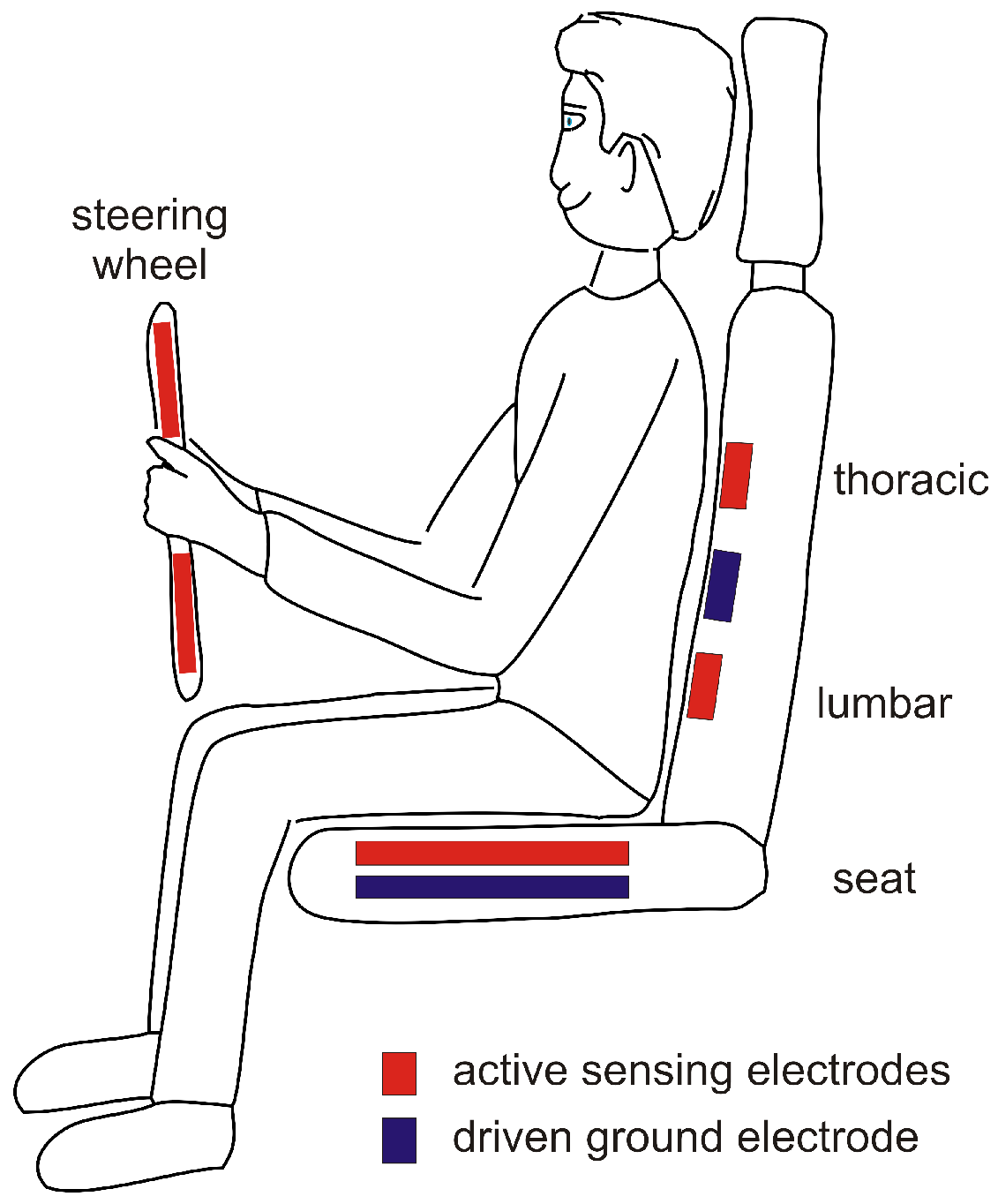
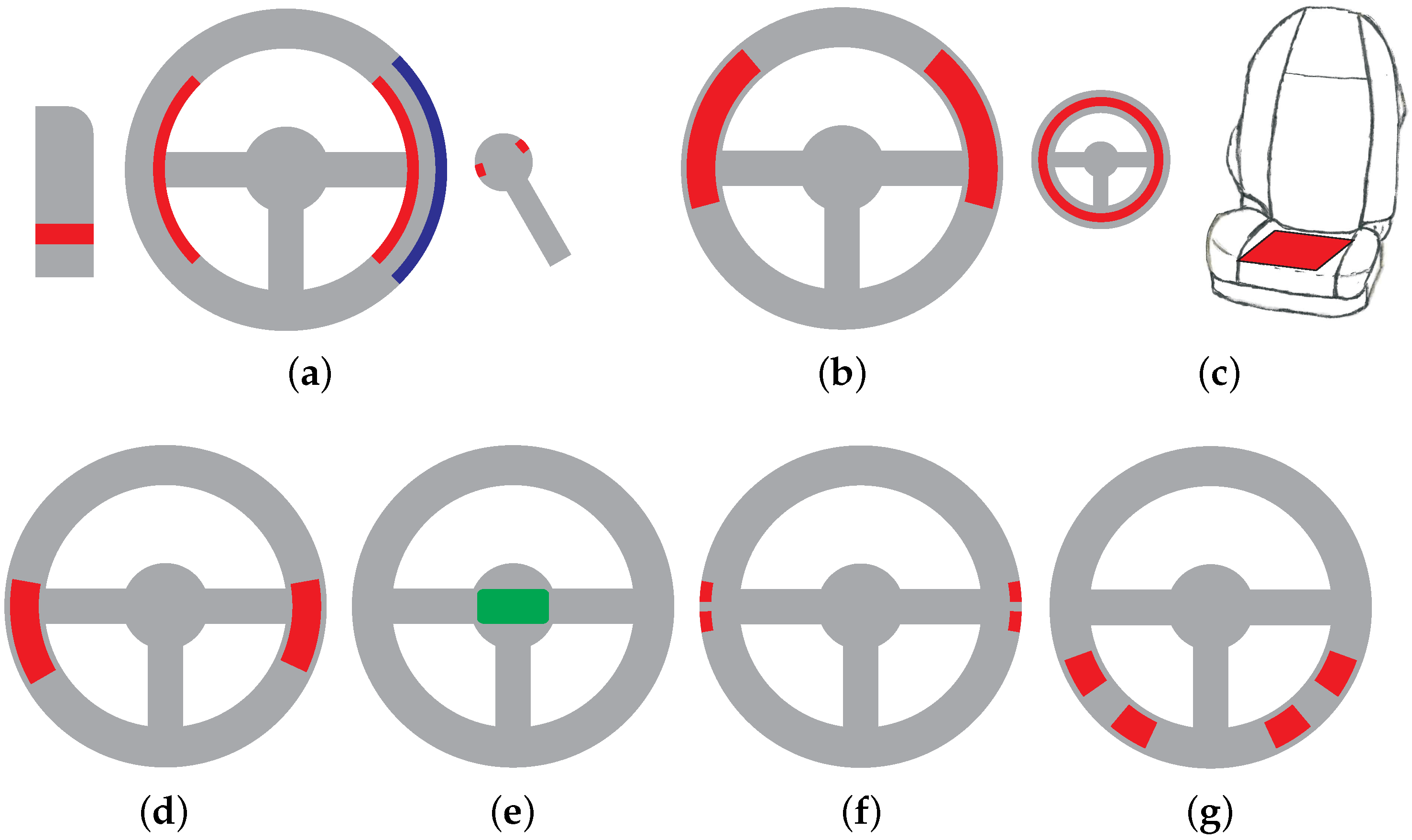
3.1. Conductive ECG Monitoring
Hybrid ECG Monitoring
3.2. Capacitive ECG Monitoring
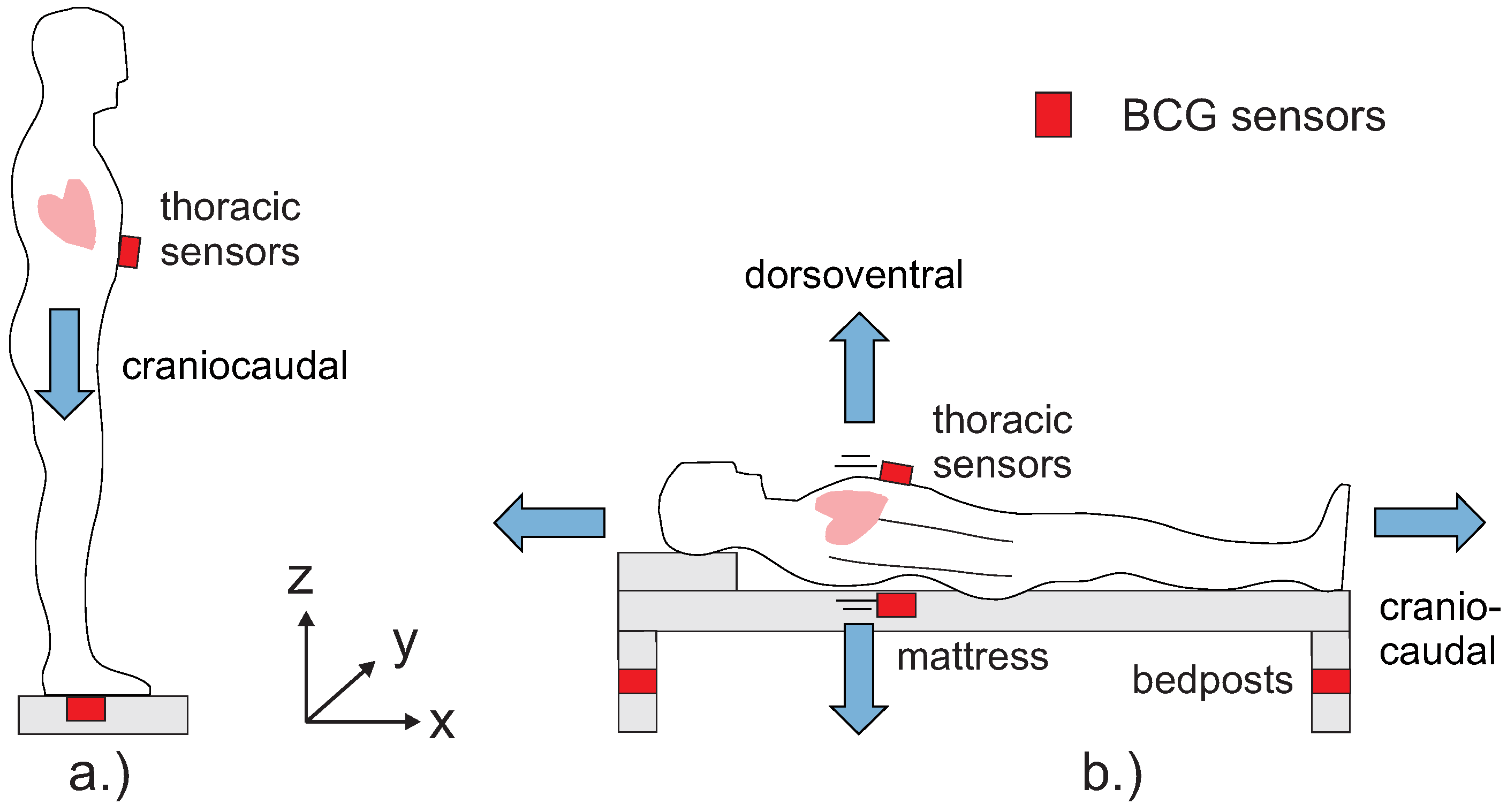
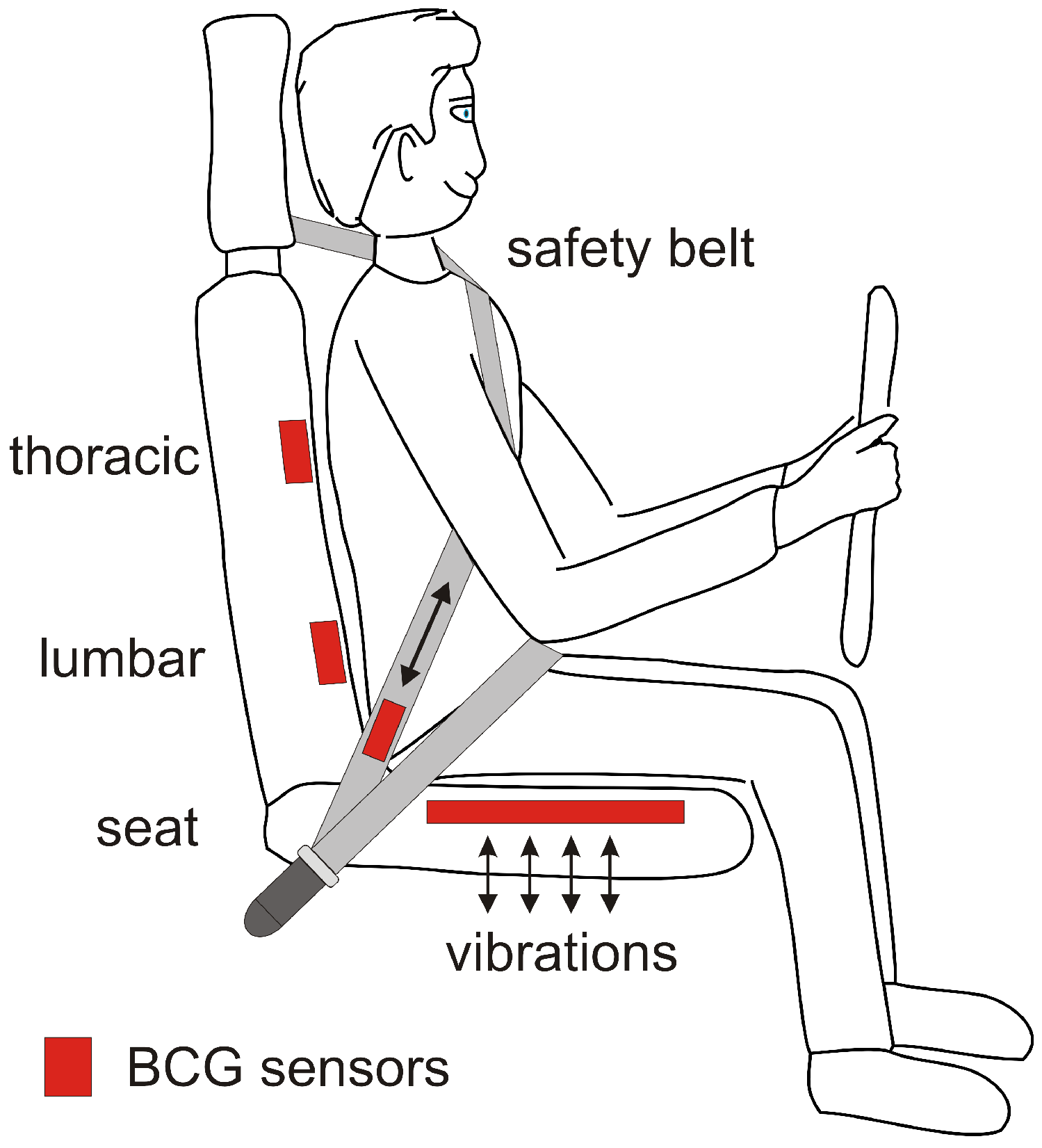
4. Ballistocardiography
- Phonocardiogram, or
- Apexcardiogram, or
- Kinetocardiogram, or
- Seismocardiogram.
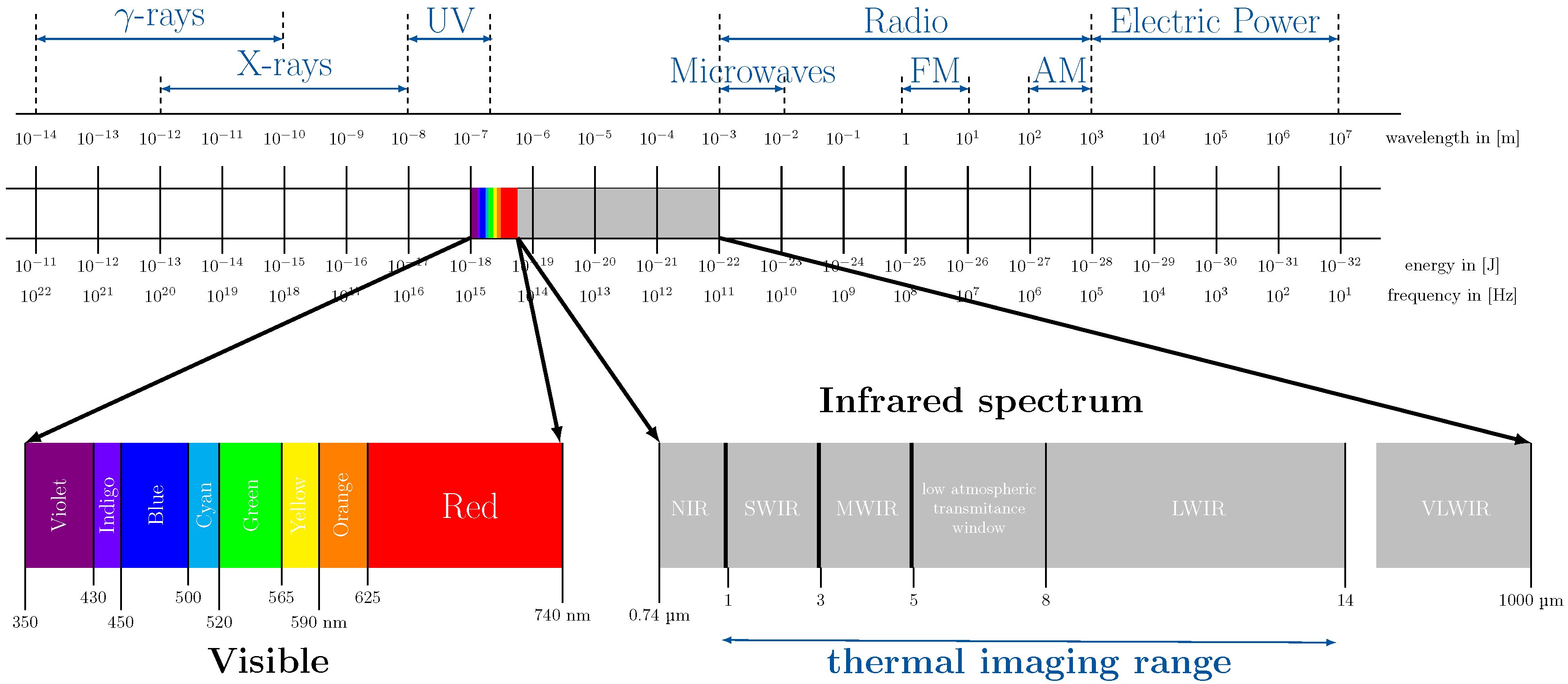
5. Optical Methods
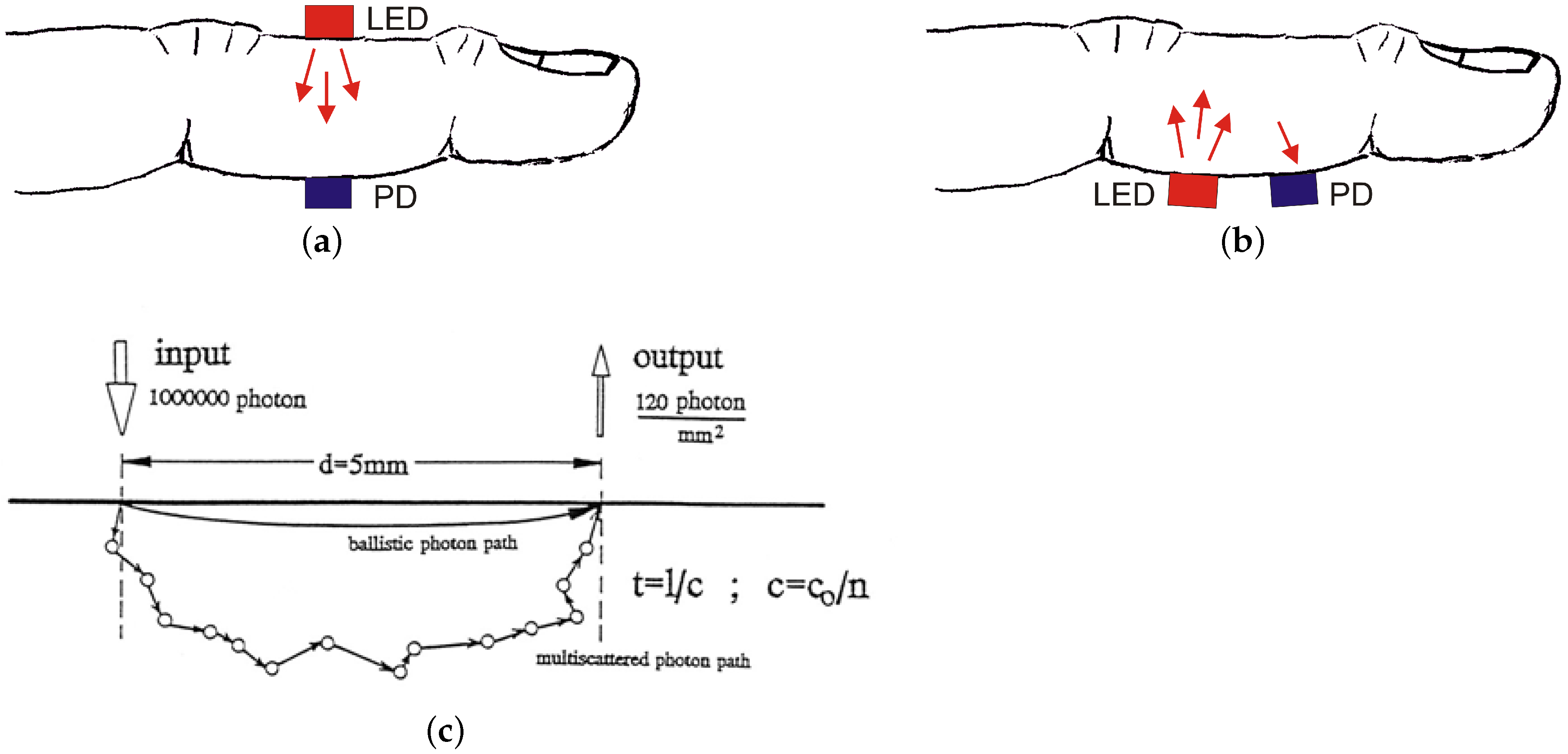
5.1. Photoplethysmography
PPG Monitoring in the Car
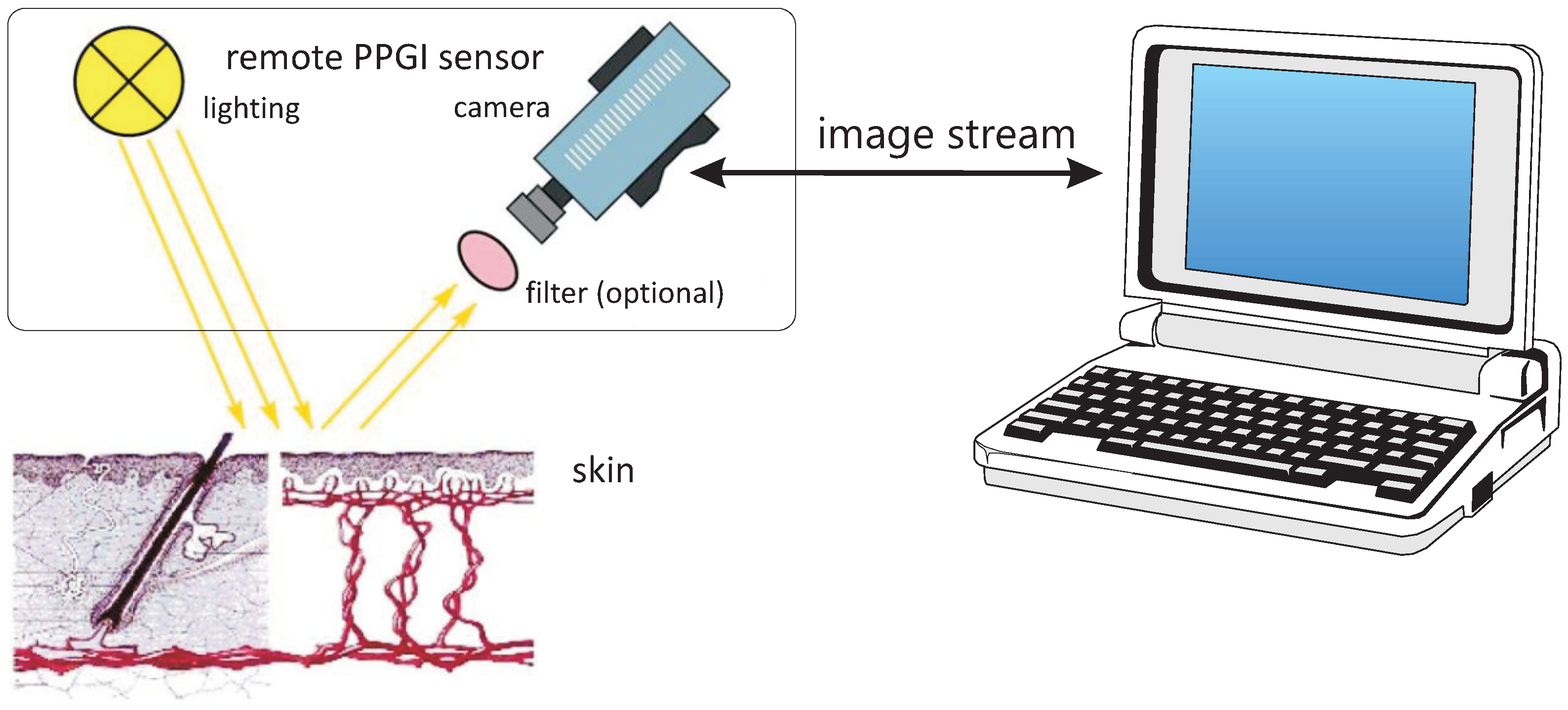
5.2. PPG Imaging
PPGI Vital Sign Monitoring in the Car
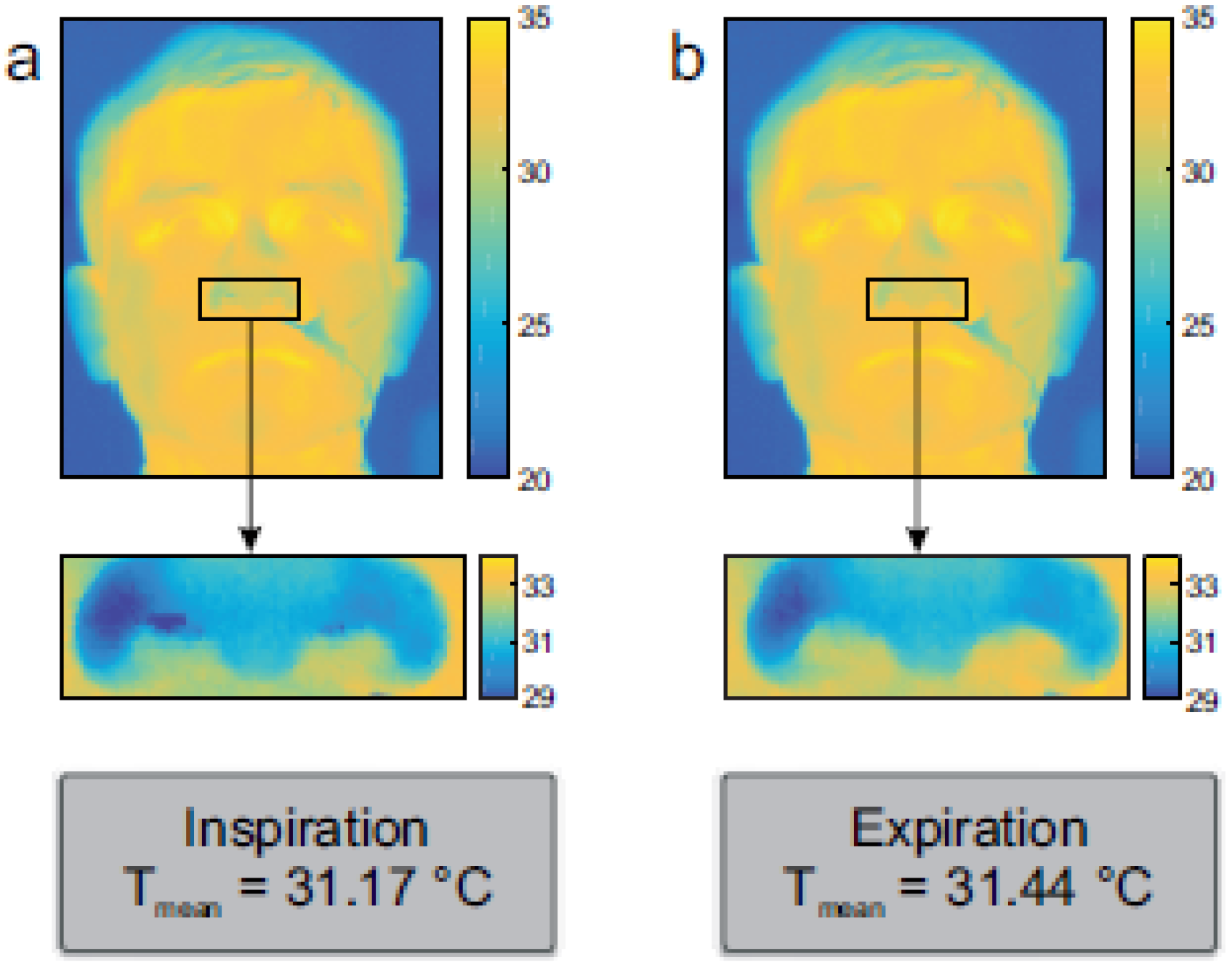
5.3. Far Infrared Imaging (Thermography)
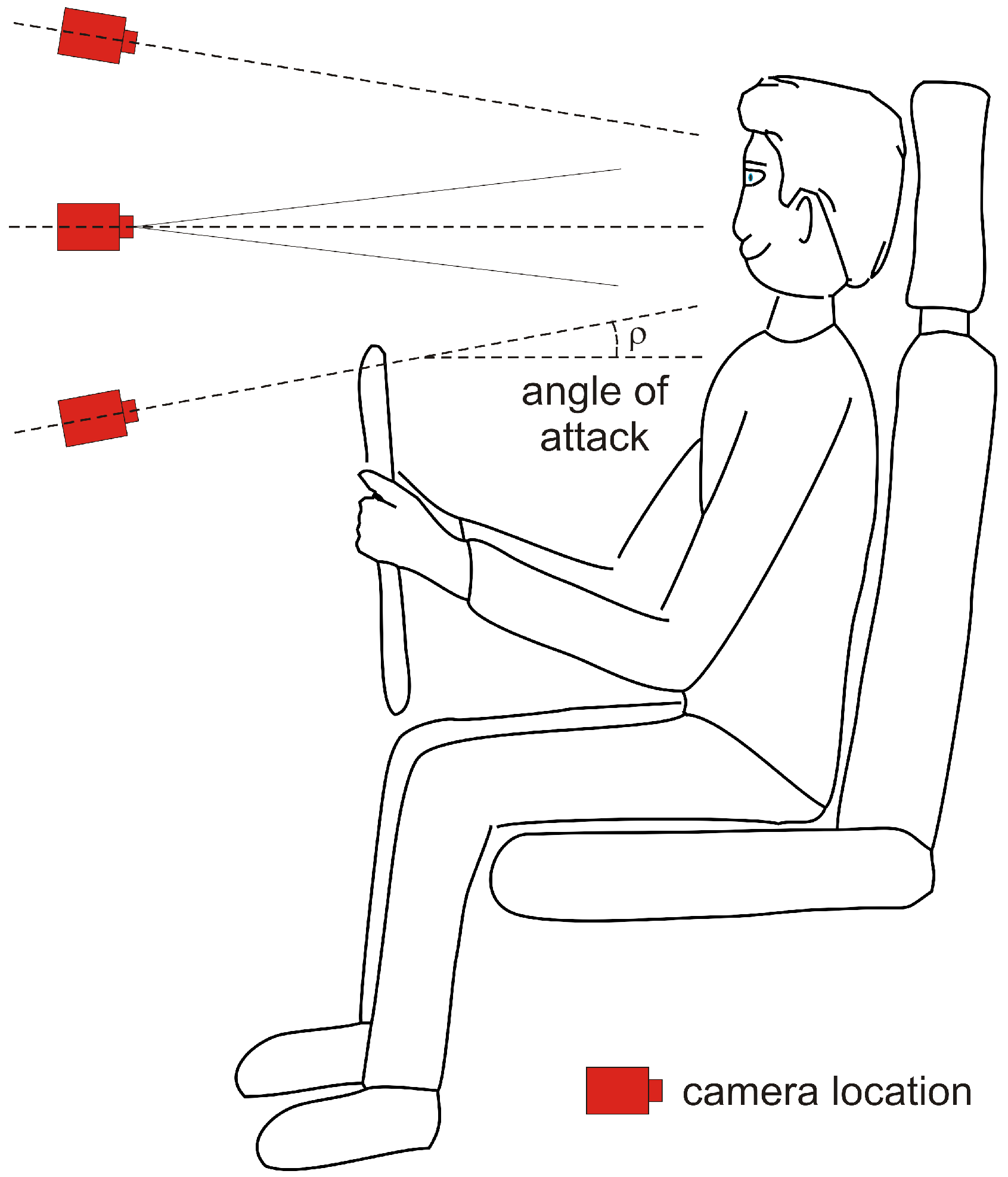
5.4. Other Camera-Based Automotive Monitoring
5.4.1. Automotive Monitoring Using Visible Light (VIS)
5.4.2. Automotive Monitoring Using Near Infrared Light (NIR)
5.4.3. Automotive Monitoring Using Far Infrared Light (FIR)
5.5. Image Fusion
5.6. Video Motion
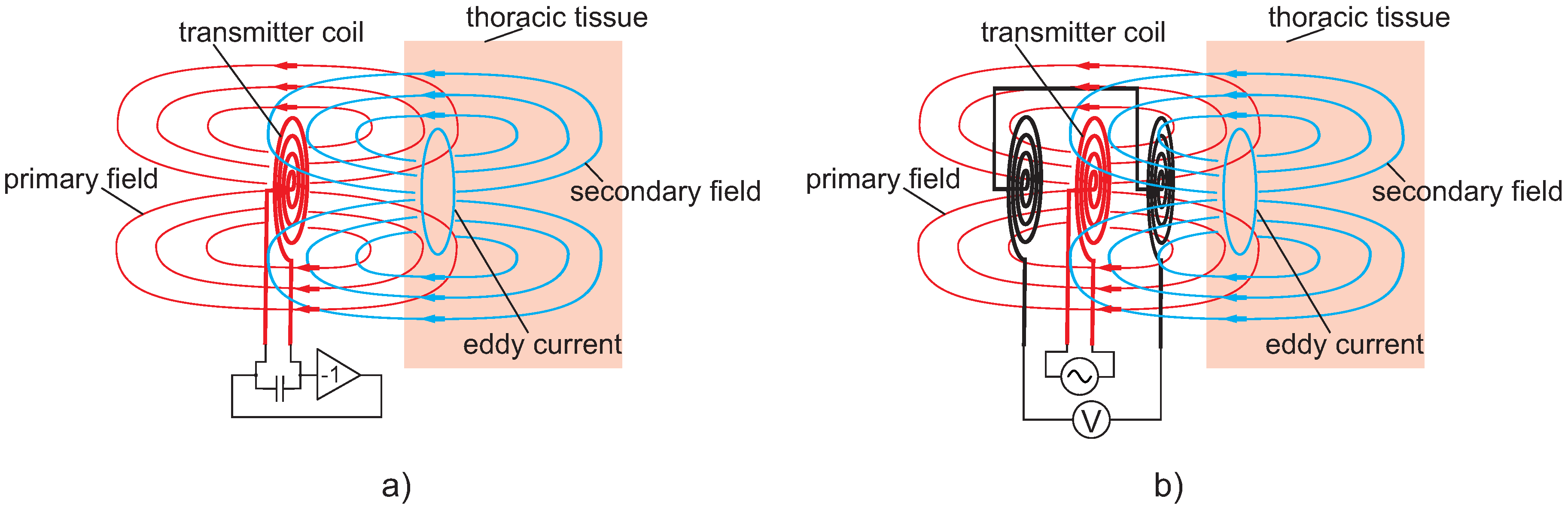
6. Magnetic Induction
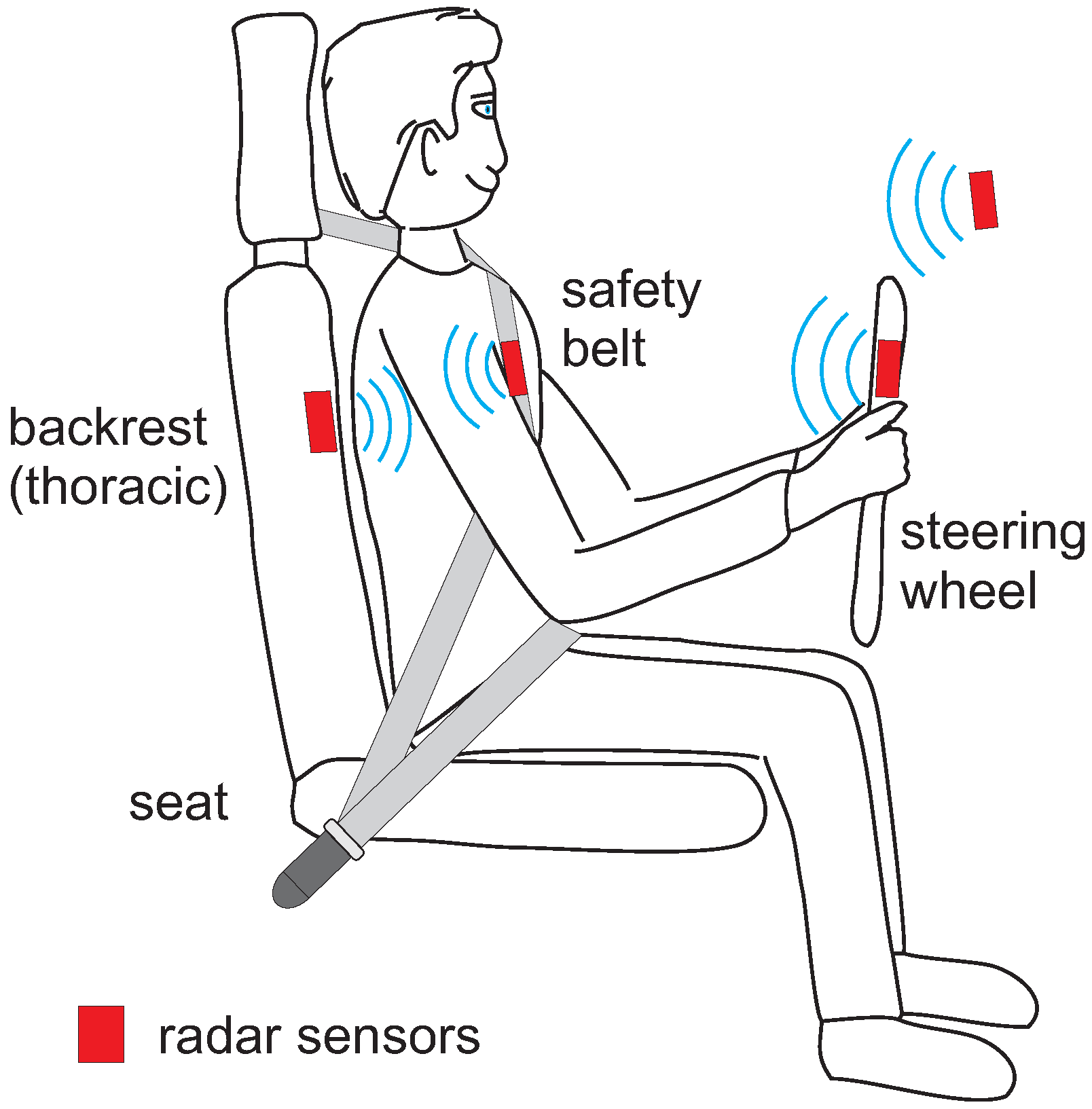
7. Radar-Based Methods
8. Challenges of In-Vehicle Measurements
8.1. Motion Artifacts
8.2. Variable Light Conditions
9. Sensor Fusion
- artifact compensation, e.g., by utilizing adaptive filtering with one sensor serving as the noise signal.
- source separation, for example by applying algorithms based on statistical dependencies between the signals (like independent component analysis).
- coverage rate enhancement, e.g., by utilizing multiple sensors measuring the same vital sign and, therefore, increasing the probability that this vital sign is obtainable at a point in time.
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BCG | ballistocardiography |
| cbPPG | camera-based photoplethysmography |
| cECG | capacitive electrocardiogram |
| CW | continuous wave |
| ECG | electrocardiogram |
| EMD | empirical mode decomposition |
| EMFi | electromechanical film |
| EPIC | electric potential integrated circuit |
| FCC | Federal Communications Commission |
| FIR | far infrared light |
| HRV | heart rate variability |
| ICA | independent component analysis |
| iPPG | imaging photoplethysmography |
| IRT | infrared thermography |
| ISM | industrial, scientific and medical |
| KIT | Karlsruhe Institute of Technology |
| LED | light-emitting diode |
| LWIR | long wave infrared light |
| MI | magnetic impedance |
| MWIR | medium wave infrared light |
| ncPPG | non-contact photoplethysmography |
| NIR | near infrared light |
| PAT | pulse arrival time |
| PD | photodetector |
| PERCLOS | percentage eye closure |
| PPG | photoplethysmogram |
| PPGI | photoplethysmography imaging |
| PTT | pulse transit time |
| QL | quality label |
| RADAR | radio detecting and ranging |
| rPPG | reflective or remote photoplethysmography |
| RSA | respiratory sinus arrhythmia |
| Rx | receiver |
| SpO2 | oxygen saturation |
| SVM | support vector machine |
| tPPG | transmissive photoplethysmography |
| Tx | transmitter |
| UWB | ultra-wide band |
| VIS | visible light |
References
- Anonymous. Vital Signs (Wikipedia Article). Available online: https://en.wikipedia.org/wiki/Vital_signs (accessed on 14 May 2018).
- Elliott, M.; Coventry, A. Critical care: The eight vital signs of patient monitoring. Br. J. Nurs. 2012, 21, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Brueser, C.; Hoog Antink, C.; Wartzek, T.; Walter, M.; Leonhardt, S. Ambient and Unobtrusive Cardiorespiratory Monitoring Techniques. IEEE Rev. Biomed. Eng. 2015, 8, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Naziyok, T.P.; Zeleke, A.A.; Röhrig, R. Contactless Patient Monitoring for General Wards: A Systematic Technology Review. Stud. Health Technol. Inform. 2016, 228, 707–711. [Google Scholar] [PubMed]
- Zaunseder, S.; Henning, A.; Wedekind, D.; Trumpp, A.; Malberg, H. Unobtrusive acquisition of cardiorespiratory signals—Available techniques and perspectives for sleep medicine. Somnologie 2017, 21, 93–100. [Google Scholar] [CrossRef]
- Healey, J.; Seger, J.; Picard, R. Quantifying driver stress: Developing a system for collecting and processing bio-metric signals in natural situations. Biomed. Sci. Instrum. 1999, 35, 193–198. [Google Scholar] [PubMed]
- Healey, J.A.; Picard, R.W. Detecting stress during real-world driving tasks using physiological sensors. IEEE Trans. Intell. Transp. Syst. 2005, 6, 156–166. [Google Scholar] [CrossRef]
- Liang, S.F.; Lin, C.T.; Wu, R.C.; Chen, Y.C.; Huang, T.Y.; Jung, T.P. Monitoring Driver’s Alertness Based on the Driving Performance Estimation and the EEG Power Spectrum Analysis. In Proceedings of the 27th Annual Conference of the IEEE Engineering in Medicine and Biology Society, Shanghai, China, 1–4 September 2005. [Google Scholar]
- Knipling, R.R.; Wierwille, W.W. Vehicle-Based Drowsy Driver Detection: Current Status and Future Prospects. In Proceedings of the IVHS America Fourth Annual Meeting, Atlanta, GA, USA, 17–20 April 1994. [Google Scholar]
- Ueno, H.; Kaneda, M.; Tsukino, M. Development of Drowsiness Detection System. In Proceedings of the Vehicle Navigation and Information Systems Conference, Yokohama, Japan, 31 August–2 September 1994. [Google Scholar]
- Begum, S. Intelligent Driver Monitoring Systems based on Physiological Sensor Signals: A Review. In Proceedings of the 16th International IEEE Annual Conference on Intelligent Transportation Systems (ITSC 2013), The Hague, The Netherlands, 6–9 October 2013. [Google Scholar]
- Rhiu, I.; Kwon, S.; Bahn, S.; Yun, M.H.; Yu, W. Research Issues in Smart Vehicles and Elderly Drivers: A Literature Review. Int. J. Hum. Comput. Interact. 2015, 31, 635–666. [Google Scholar] [CrossRef]
- Choi, Y.J.; Han, S.I.; Kong, S.-H.; Ko, H. Driver Status Monitoring Systems for Smart Vehicles using Physiological Sensors: A safety enhancement system from automobile manufacturers. IEEE Signal Process. Mag. 2016, 33, 22–34. [Google Scholar] [CrossRef]
- Scalise, L.; Millis, R.M. Non contact heart monitoring. Adv. Electrocardiogr. Methods Anal. 2012, 81–106. [Google Scholar] [CrossRef]
- Morbiducci, U.; Scalise, L.; de Melis, M.; Grigioni, M. Optical Vibrocardiography: A Novel Tool for the Optical Monitoring of Cardiac Activity. Ann. Biomed. Eng. 2007, 35, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Kranjec, J.; Begus, S.; Gersak, G.; Drnovsek, J. Non-contact heart rate and heart rate variability measurements: A review. Biomed. Signal Process. Control 2014, 13, 102–112. [Google Scholar] [CrossRef]
- Kranjec, J.; Beguš, S.; Drnovšek, J.; Geršak, G. Novel Methods for Noncontact Heart Rate Measurement: A Feasibility Study. IEEE Trans. Instrum. Meas. 2014, 63, 838–847. [Google Scholar] [CrossRef]
- Wagner, G.S.; Strauss, D.G. Marriott’s Practical Electrocardiography, 12th ed.; LWW: Philadelphia, PA, USA, 2013. [Google Scholar]
- Goldberger, A.L.; Goldberger, Z.D.; Shvilkin, A. Goldberger’s Clinical Electrocardiography: A Simplified Approach, 9th ed.; Elsevier: Philadelphia, PA, USA, 2017. [Google Scholar]
- Jeong, I.C.; Lee, D.H.; Park, S.W.; Ko, J.I.; Yoon, H.R. Automobile driver’s stress index provision system that utilizes electrocardiogram. In Proceedings of the 2007 IEEE Intelligent Vehicles Symposium, Istanbul, Turkey, 13–15 June 2007. [Google Scholar]
- Lee, H.B.; Choi, J.M.; Kim, J.S.; Kim, Y.S.; Baek, H.J.; Ryu, M.S.; Sohn, R.H.; Park, K.S. Nonintrusive biosignal measurement system in a vehicle. In Proceedings of the 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 23–26 August 2007; pp. 2303–2306. [Google Scholar]
- Hu, S.; Bowlds, R.; Yu, X. Non-Intrusive ECG Measurement on Vehicle Steering Wheel and Driver Seat. J. Med. Devices 2008, 2, 027520. [Google Scholar] [CrossRef]
- Shin, H.-S.; Jung, S.-J.; Kim, J.-J.; Chung, W.-Y. Real time car driver’s condition monitoring system. In Proceedings of the IEEE Sensors 2010 Conference, Waikoloa, HI, USA, 1–4 November 2010; pp. 951–954. [Google Scholar]
- Jung, S.J.; Shin, H.S.; Chung, W.Y. Driver fatigue and drowsiness monitoring system with embedded electrocardiogram sensor on steering wheel. IET Intell. Transp. Syst. 2014, 8, 43–50. [Google Scholar] [CrossRef]
- Heuer, S.; Chamadiyaz, B.; Gharbiy, A.; Kunze, C.; Wagner, M. Unobtrusive In-Vehicle Biosignal Instrumentation for Advanced Driver Assistance and Active Safety. In Proceedings of the IEEE EMBS Conference on Biomedical Engineering & Sciences (IECBES 2010), Kuala Lumpur, Malaysia, 30 November–2 December 2010. [Google Scholar]
- D’Angelo, L.T.; Parlow, J.; Spiessl, W.; Hoch, S.; Lueth, T.C. A system for unobtrusive in-car vital parameter acquisition and processing. In Proceedings of the 4th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth 2010), Munich, Germany, 22–25 March 2010; pp. 1–7. [Google Scholar]
- D’Angelo, L.T.; Lueth, T.C. Integrated Systems for Distraction—Free Vital Signs Measurement in Vehicles. Automobiltech. Z. ATZ-Worldw. 2011, 113, 52–56. [Google Scholar] [CrossRef]
- Gomez-Clapers, J.; Casanella, R. A Fast and Easy-to-Use ECG Acquisition and Heart Rate Monitoring System using a Wireless Steering Wheel. IEEE Sens. J. 2012, 12, 610–616. [Google Scholar] [CrossRef]
- Silva, H.; Lourenco, A.; Fred, A. In-Vehicle Driver Recognition based on Hands ECG Signals. In Proceedings of the 17th International Conference on Intelligent User Interfaces (IUI’12), Lisbon, Portugal, 14–17 February 2012. [Google Scholar]
- Vavrinský, E.; Tvarožek, V.; Stopjaková, V.; Soláriková, P.; Brezina, I. Monitoring of car driver physiological parameters. In Proceedings of the Eighth International Conference on Advanced Semiconductor Devices and Microsystems, Smolenice, Slovakia, 25–27 October 2010; pp. 227–230. [Google Scholar]
- Vavrinsky, E.; Tvarozek, V.; Gaspierik, P.; Stopjakova, V.; Donoval, M.; Teleka, P. Design of non-invasive setup for car driver biomonitoring. Procedia Chem. 2012, 6, 203–210. [Google Scholar] [CrossRef]
- Matsuda, T.; Makikawa, M. ECG monitoring of a car driver using capacitively-coupled electrodes. In Proceedings of the 30th Annual International IEEE EMBS Conference, Vancouver, BC, Canada, 20–24 August 2008; pp. 1315–1318. [Google Scholar]
- Xu, X.; Ta, L. A Novel Driver-friendly ECG Monitoring System Based on Capacitive-coupled Electrode. Inf. Technol. J. 2013, 12, 4730–4734. [Google Scholar] [CrossRef]
- Baek, H.J.; Lee, H.B.; Kim, J.S.; Choi, J.M.; Kim, K.K.; Park, K.S. Nonintrusive biological signal monitoring in a car to evaluate a driver’s stress and health state. Telemed. J. E-Health 2009, 15, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.C. The Insulated Electrode. In Proceedings of the 20th Annual Conference on Engineering in Medicine and Biology, Boston, MA, USA, 5–6 September 1967; p. 157. [Google Scholar]
- Ishijima, M. Monitoring of electrocardiograms in bed without utilizing body surface electrodes. IEEE Trans. Biomed. Eng. 1993, 40, 593–594. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.K.; Kim, K.K.; Park, K.S. The ECG measurement in the bathtub using the insulated electrodes. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–5 September 2004; Volume 4, pp. 2383–2385. [Google Scholar]
- Kim, K K.; Lim, Y.K.; Park, K.S. The electrically non-contacting ECG measurement on the toilet seat using the capacitively-coupled insulated electrodes. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–5 September 2004; Volume 4, pp. 2375–2378. [Google Scholar]
- Lim, Y.G.; Kim, K.K.; Park, K.S. ECG measurement on a chair without conductive contact. IEEE Trans. Biomed. Eng. 2006, 53, 956–959. [Google Scholar] [PubMed]
- Lim, Y.G.; Kim, K.K.; Park, K.S. ECG recording on a bed during sleep without direct skin-contact. IEEE Trans. Biomed. Eng. 2007, 54, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Zhang, Y. Contactless and continuous monitoring of heart electric activities through clothes on a sleeping bed. In Proceedings of the 5th International Conference on Information Technology and Application in Biomedicine, in conjunction with the 2nd International Symposium & Summer School on Biomedical and Health Engineering, Shenzhen, China, 30–31 May 2008; pp. 282–285. [Google Scholar]
- Leonhardt, S.; Aleksandrowicz, A. Non-contact ECG monitoring for automotive application. In Proceedings of the 5th International Workshop on Wearable and Implantable Body Sensor Networks (BSN 2008), Hong Kong, China, 1–3 June 2008. [Google Scholar]
- Chamadiya, B.; Heuer, S.; Hofmann, U.G.; Wagner, M. Towards a capacitively coupled electrocardiography system for car seat integration. In Proceedings of the 4th European Conference of the International Federation for Medical and Biological Engineering, Antwerpen, Belgium, 23–27 November 2008; pp. 1217–1221. [Google Scholar]
- Eilebrecht, B.; Wartzek, T.; Lem, J.; Vogt, R.; Leonhardt, S. Capacitive electrocardiogram measurement system in the driver seat. Automobiltech. Z. ATZ-Worldw. 2011, 113, 50–55. [Google Scholar] [CrossRef]
- Wartzek, T.; Eilebrecht, B.; Lem, J.; Lindner, H.J.; Leonhardt, S.; Walter, M. ECG on the road: Robust and unobtrusive estimation of heart rate. IEEE Trans. Biomed. Eng. 2011, 58, 3112–3120. [Google Scholar] [CrossRef] [PubMed]
- Wartzek, T.; Lammersen, T.; Eilebrecht, B.; Walter, M.; Leonhardt, S. Triboelectricity in capacitive biopotential measurements. IEEE Trans. Biomed. Eng. 2011, 58, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Schumm, J.; Arnrich, B.; Troester, G. ECG Monitoring in an Airplane Seat: Appraising the Signal Quality. IEEE Pervasive Comput. 2012, 11, 28–34. [Google Scholar] [CrossRef]
- Schneider, J.; Koellner, C.; Heuer, S. An Approach to Automotive ECG Measurement Validation using a Car-Integrated Test Framework. In Proceedings of the 2012 Intelligent Vehicles Symposium, Alcala de Henares, Spain, 3–7 June 2012. [Google Scholar]
- Jung, S.J.; Shin, H.S.; Yoo, J.H.; Chung, W.Y. Highly Sensitive Driver Condition Monitoring System using Nonintrusive Active Electrodes. In Proceedings of the 2012 IEEE International Conference on Consumer Electronics (ICCE), Las Vegas, NV, USA, 13–16 January 2012; pp. 305–306. [Google Scholar]
- Fong, E.M.; Chung, W.Y. A Hygroscopic Sensor Electrode for Fast Stabilized Non-Contact ECG Signal Acquisition. Sensors 2015, 15, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Weder, M.; Hegemann, D.; Amberg, M.; Hess, M.; Boesel, L.F.; Abächerli, R.; Meyer, V.R.; Rossi, R.M. Embroidered Electrode with Silver/Titanium Coating for Long-Term ECG Monitoring. Sensors 2015, 15, 1750–1759. [Google Scholar] [CrossRef] [PubMed]
- Plessey WARDEN System. Available online: http://www.plesseysemiconductors.com/products/warden/ (accessed on 3 June 2018).
- Leicht, L.; Eilebrecht, B.; Weyer, S.; Leonhardt, S.; Teichmann, D. Closed-Loop Control of Humidification for Artifact Reduction in Capacitive ECG Measurements. IEEE Trans. Biomed. Circuits Syst. 2017, 11, 300–313. [Google Scholar] [CrossRef] [PubMed]
- Van Hoof, C.; Torfs, T. Get Your Daily Health Check in the Car. IMEC Magazine. September 2017. Available online: https://www.imec-int.com/en/imec-magazine/imec-magazine-september-2017/get-your-daily-health-check-in-the-car (accessed on 23 April 2018).
- Leicht, L.; Skobel, E.; Knackstedt, C.; Mathissen, M.; Sitter, A.; Wartzek, T.; Möhler, W.; Reith, S.; Leonhardt, S.; Teichmann, D. Capacitive ECG Monitoring in Cardiac Patients during Simulated Driving. IEEE Trans. Biomed. Eng. 2018. [Google Scholar] [CrossRef] [PubMed]
- Leonhardt, S.; Aleksandrowicz, A.; Steffen, M. Magnetic and Capacitive Monitoring of Heart and Lung Activity as an Example for Personal Healthcare. In Proceedings of the 2006 3rd IEEE/EMBS International Summer School on Medical Devices and Biosensors, Cambridge, MA, USA, 4–6 September 2006; pp. 57–60. [Google Scholar]
- Aleksandrowicz, A.; Leonhardt, S. Wireless and Non-contact ECG Measurement System—The “Aachen SmartChair”. Acta Polytech. 2007, 47, 68–71. [Google Scholar]
- Chamadiya, B.; Heuer, S.; Wagner, M.; Hofmann, U.G. Textile Capacitive Electrocardiography for an automotive environment. In Proceedings of the International Conference on Biomedical Electronics and Devices (BIODEVICES 2011), Rome, Italy, 26–29 January 2011. [Google Scholar]
- EPIC Seatback Sensor. Available online: http://www.plesseysemiconductors.com/wp-content/uploads/EPIC-Seatback-Sensor-Promotion-Sheet.pdf (accessed on 23 April 2018).
- Leicht, L.; Eilebrecht, B.; Weyer, S.; Wartzek, T.; Leonhardt, S. Active humidification for capacitive-resistive ECG-systems. In Proceedings of the 48. DGBMT Jahrestagung (BMT 2014), Hannover, Germany, 8 October 2014; pp. 818–821. [Google Scholar]
- Leicht, L.; Skobel, E.; Mathissen, M.; Leonhardt, S.; Weyer, S.; Wartzek, T.; Reith, S.; Moehler, W.; Teichmann, D. Capacitive ECG recording and beat-to-beat interval estimation after major cardiac event. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC 2015), Milano, Italy, 25–29 August 2015; pp. 7614–7617. [Google Scholar]
- Arnrich, B.; Kappeler-Setz, C.; Schumm, J.; Troester, G. Design, Implementation and Evaluation of a Multimodal Sensor System Integrated into an Airplane Seat. Sensor Fusion—Foundation and Applications. Thomas, C., Ed.; Chapter 9. pp. 159–170. Available online: https://www.intechopen.com/books/sensor-fusion-foundation-and-applications (accessed on 13 June 2011).
- Magno, M.; Spagnol, C.; Benini, L.; Popovici, E. A low power wireless node for contact and contactless heart monitoring. Microelectr. J. 2014, 45, 1656–1664. [Google Scholar] [CrossRef]
- Leicht, L.; Eilebrecht, B.; Mathissen, M.; Lem, J.; Lindner, A.; Vogt, R.; Leonhardt, S.; Walter, M. Contactless Electrocardiographic Sensor with Moisture Generator. U.S. Patent US2015088317 (A1), 23 September 2013. [Google Scholar]
- Van Hoof, C.; Torfs, T. How Your Car Can Check Your Daily Health. eeNews Automotive. 22 November 2017. Available online: http://www.eenewsautomotive.com/design-center/how-your-car-can-check-your-daily-health/page/0/2 (accessed on 6 June 2018).
- Gordon, J.W. Certain molar movements of the human body produced by the circulation of the blood. J. Anatomy Physiol. 1877, 11, 533–536. [Google Scholar]
- Starr, I.; Rawson, A.J.; Schroeder, H.A.; Joseph, N.R. Studies on the estimation of cardiac output in man, and of abnormalities in cardiac function, from the heart’s recoil and the blood’s impacts; the ballistocardiogram. Am. J. Physiol. 1939, 127, 1–28. [Google Scholar]
- Scarborough, W.R.; Talbot, S.A.; Braunstein, J.R.; Rappaport, M.B.; Dock, W.; Hamilton, W.F.; Smith, J.E.; Nickerson, J.L.; Starr, I. Proposals for ballistocardiographic nomenclature and conventions: Revised and extended: Report of committee on ballistocardiographic terminology. Circulation 1956, 14, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Inan, O.T.; Etemadi, M.; Wiard, R.M.; Giovangrandi, L.; Kovacs, G.T.A. Robust ballistocardiogram acquisition for home monitoring. Physiol. Meas. 2009, 30, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Mack, D.C.; Patrie, J.T.; Suratt, P.M.; Felder, R.A.; Alwan, M.A. Development and preliminary validation of heart rate and breathing rate detection using a passive, ballistocardiography-based sleep monitoring system. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Jansen, B.H.; Larson, B.H.; Shankar, K. Monitoring of the ballistocardiogram with the static charge sensitive bed. IEEE Trans. Biomed. Eng. 1991, 38, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Inan, O.T.; Migeotte, P.F.; Park, K.S.; Etemadi, M.; Tavakolian, K.; Casanella, R.; Zanetti, J.; Tank, J.; Funtova, I.; Prisk, G.K.; et al. Ballistocardiography and Seismocardiography: A Review of Recent Advances. IEEE J. Biomed. Health Inform. 2015, 19, 1414–1427. [Google Scholar] [CrossRef] [PubMed]
- Brueser, C.; Stadlthanner, K.; de Waele, S.; Leonhardt, S. Adaptive beat-to-beat heart rate estimation in ballistocardiograms. IEEE Trans. Inf. Technol. Biomed. 2011, 15, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.; Eilebrecht, B.; Wartzek, T.; Leonhardt, S. The smart car seat: Personalized monitoring of vital signs in automotive applications. Pers. Ubiquitous Comput. 2011, 15, 707–715. [Google Scholar] [CrossRef]
- HARKEN—Heart and Respiration in-Car Embedded Non Intrusive Sensors. Available online: http://cordis.europa.eu/result/rcn/156259_en.html (accessed on 3 September 2016).
- Faurecia Press Release Kit. Available online: www.faurecia.com/files/corporate/publication/file/faurecia_active_wellness_140915_en.pdf (accessed on 5 September 2018).
- Faurecia PRESS KIT AUTO SHANGHAI 2015. Available online: www.faurecia.com/files/corporate/publication/file/presskit_final_web.pdf (accessed on 5 September 2018).
- Wusk, G.; Gabler, H. Non-Invasive Detection of Respiration and Heart Rate with a Vehicle Seat Sensor. Sensors 2018, 18, 1463. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Thakor, N. Photoplethysmography Revisited: From Contact to Noncontact, From Point to Imaging. IEEE Trans. Biomed. Eng. 2016, 63, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Daly, J. Video Camera Monitoring to Detect Changes in Haemodynamics. Ph.D. Thesis, University of Oxford, Oxford, UK, 2016. [Google Scholar]
- Blazek, V.; Wu, T.; Hoelscher, D. Near-infrared CCD imaging: Possibilities for noninvasive and contactless 2D mapping of dermal venous hemodynamics. In SPIE 3923, Optical Diagnostics of Biological Fluids V; The International Society for Optics and Photonics: San Diego, CA, USA, 2000. [Google Scholar]
- Wu, T.; Blazek, V.; Schmitt, H.J. Photoplethysmography imaging: A new noninvasive and noncontact method for mapping of the dermal perfusion changes. In EBIOS 2000: European Conference on Biomedical Optics, Amsterdam, Proceedings of the SPIE 4163, Optical Techniques and Instrumentation for the Measurement of Blood Composition, Structure, and Dynamics; The International Society for Optics and Photonics: San Diego, CA, USA, 2000. [Google Scholar]
- Huelsbusch, M.; Blazek, V. Contactless mapping of rhythmical phenomena in tissue perfusion using PPGI. In SPIE 4683, Medical Imaging 2002: Physiology and Function from Multidimensional Images; The International Society for Optics and Photonics: San Diego, CA, USA, 2002. [Google Scholar]
- Wieringa, F.P.; Mastik, F.; van der Steen, A.F. Contactless multiple wavelength photoplethysmographic imaging: A first step toward “SpO2 camera” technology. Ann. Biomed. Eng. 2005, 33, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Takano, C.; Ohta, Y. Heart rate measurement based on a time-lapse image. Med. Eng. Phys. 2007, 29, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Hu, S. The preliminary investigation of imaging photoplethysmographic system. In Proceedings of the Third International Conference on Optical and Laser Diagnostics, London, UK, 22–25 May 2007. Journal of Physics: Conference Series 85, 012031. [Google Scholar]
- Verkruysse, W.; Svaasand, L.O.; Nelson, J.S. Remote plethysmographic imaging using ambient light. Opt. Express 2008, 16, 21434–21445. [Google Scholar] [CrossRef] [PubMed]
- Wang, W. Robust and Automatic Remote Photoplethysmography. Ph.D. Thesis, Technische Universiteit Eindhoven, Eindhoven, The Netherlands, 2017. [Google Scholar]
- Tarassenko, L.; Villarroel, M.; Guazzi, A.; Jorge, J.; Clifton, D.A.; Pugh, C. Non-contact video-based vital sign monitoring using ambient light and auto-regressive models. Physiol. Meas. 2014, 35, 807–831. [Google Scholar] [CrossRef] [PubMed]
- Villarroel, M.; Guazzi, A.; Jorge, J.; Davis, S.; Watkinson, P.; Green, G.; Shenvi, A.; McCormick, K.; Tarassenko, L. Continuous non-contact vital sign monitoring in neonatal intensive care unit. Healthc. Technol. Lett. 2014, 1, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Guazzi, A.R.; Villarroel, M.; Jorge, J.; Daly, J.; Frise, M.C.; Robbins, P.A.; Tarassenko, L. Non-contact measurement of oxygen saturation with an RGB camera. Biomed. Opt. Express 2015, 6, 3320–3338. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.Y.; Rubinstein, M.; Shih, E.; Guttag, J.; Durand, F.; Freeman, W. Eulerian video magnification for revealing subtle changes in the world. ACM Trans. Graph. 2012, 31, 1–8. [Google Scholar] [CrossRef]
- Blazek, C.R.; Merk, H.F.; Schmid-Schoenbein, H.; Huelsbusch, M.; Blazek, V. Assessment of Allergic Skin Reactions and their Inhibition by Antihistamines using Photoplethysmography Imaging (PPGI). J. Allergy Clin. Immunol. 2006, 117, S226. [Google Scholar] [CrossRef]
- Karlen, W.; Garde, A.; Myers, D.; Scheffer, C.; Ansermino, J.M.; Dumont, G.A. Estimation of Respiratory Rate from Photoplethysmographic Imaging Videos Compared to Pulse Oximetry. IEEE J. Biomed. Health Inform. 2015, 19, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Moco, A.V.; Stuijk, S.; de Haan, G. Ballistocardiographic Artifacts in PPG Imaging. IEEE Trans. Biomed. Eng. 2016, 63, 1804–1811. [Google Scholar] [CrossRef] [PubMed]
- Blazek, V.; Blanik, N.; Blazek, C.R.; Paul, M.; Pereira, C.; Koeny, M.; Venema, B.; Leonhardt, S. Active and Passive Optical Imaging Modality for Unobtrusive Cardiorespiratory Monitoring and Facial Expression Assessment. Anesth. Analg. 2017, 124, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Bloecher, T.; Schneider, J.; Schinle, M.; Stork, W. An online PPGI approach for camera-based heart rate monitoring using beat-to-beat detection. In Proceedings of the 2017 IEEE Sensors Applications Symposium (SAS), Glassboro, NJ, USA, 13–15 March 2017. [Google Scholar]
- Philips IP&S. Vital Signs Camera. Available online: www.vitalsignscamera.com (accessed on 10 June 2018).
- Hu, S.; Zheng, J.; Chouliaras, V.; Summers, R. Feasibility of imaging photoplethysmography. In Proceedings of the 2008 International Conference on BioMedical Engineering and Informatics, Sanya, China, 28–30 May 2008. [Google Scholar]
- Sun, Y.; Yu, X.B. An innovative nonintrusive driver assistance system for vital signal monitoring. IEEE J. Biomed. Health Inform. 2014, 18, 1932–1939. [Google Scholar] [CrossRef] [PubMed]
- Kamshilin, A.A.; Nippolainen, E.; Sidorov, I.S.; Vasilev, P.V.; Erofeev, N.P.; Podolian, N.P.; Romashko, R.V. A new look at the essence of the imaging photoplethysmography. Sci. Rep. 2015, 5, 10494. [Google Scholar] [CrossRef] [PubMed]
- Kuo, J.; Koppel, S.; Charlton, J.L.; Rudin-Brown, C.M. Evaluation of a video-based measure of driver heart rate. J. Saf. Res. 2015, 54, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Blackford, E.B.; Piasecki, A.M.; Estepp, J.R. Measuring pulse rate variability using long-range, non-contact imaging photoplethysmography. In Proceedings of the 2016 IEEE 38th Annual International Conference of the Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 17–20 August 2016; pp. 3930–3936. [Google Scholar]
- McDuff, D.J.; Estepp, J.R.; Piasecki, A.M.; Blackford, E.B. A survey of remote optical photoplethysmographic imaging methods. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC 2015), Milano, Italy, 25–29 August 2015; pp. 6398–6404. [Google Scholar]
- Kwon, S.; Kim, J.; Lee, D.; Park, K. ROI analysis for remote photoplethysmography on facial video. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milano, Italy, 25–29 August 2015; pp. 4938–4941. [Google Scholar]
- Van Gastel, M.; Stuijk, S.; de Haan, G. Motion Robust Remote-PPG in Infrared. IEEE Trans. Biomed. Eng. 2015, 62, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.-F.; Chu, Y.W.; Huang, P.-W.; Chung, M.-L.; Lin, T.-M. A Motion Robust Remote-PPG Approach to Driver’s Health State Monitoring. In Proceedings of the ACCV 2016 International Workshops Computer Vision (ACCV 2016), Taipei, Taiwan, 20–24 November 2016; pp. 463–476. [Google Scholar]
- Van Gastel, M.; Stuijk, S.; de Haan, G. Robust respiration detection from remote photoplethysmography. Biomed. Opt. Express 2016, 7, 4941–4957. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Veeraraghavan, A.; Sabharwal, A. DistancePPG: Robust non-contact vital signs monitoring using a camera. Biomed. Opt. Express 2015, 6, 1565–1588. [Google Scholar] [CrossRef] [PubMed]
- Choe, J.; Chung, D.; Schwichtenberg, A.J.; Delp, E.J. Improving video-based resting heart rate estimation: A comparison of two methods. In Proceedings of the IEEE 58th International Midwest Symposium on Circuits and Systems (MWSCAS), Fort Collins, CO, USA, 2–5 August 2015; pp. 1–4. [Google Scholar]
- Wedekind, D.; Malberg, H.; Zaunseder, S.; Gaetjen, F.; Matschke, K.; Rasche, S. Automated identification of cardiac signals after blind source separation for camera-based photoplethysmography. In Proceedings of the IEEE 35th International Conference on Electronics and Nanotechnology (ELNANO), KJiev, Ukraine, 24–26 April 2015; pp. 422–427. [Google Scholar]
- Rasche, S.; Trumpp, A.; Waldow, T.; Gaetjen, F.; Plötze, K.; Wedekind, D.; Schmidt, M.; Malberg, H.; Matschke, K.; Zaunseder, S. Camera-based photoplethysmography in critical care patients. Clin. Hemorheol. Microcirc. 2016, 64, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Kevat, A.C.; Bullen, D.V.; Davis, P.G.; Kamlin, C.O.F. A systematic review of novel technology for monitoring infant and newborn heart rate. Acta Paediatr. 2017, 106, 710–720. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.J.; Crowe, J.A.; Hayes-Gill, B.R.; Rodmell, P.I. Motion limitations of non-contact photoplethysmography due to the optical and topological properties of skin. Physiol. Meas. 2016, 37, N27–N37. [Google Scholar] [CrossRef] [PubMed]
- Tayibnapis, I.R.; Koo, D.Y.; Choi, M.K.; Kwon, S. A novel driver fatigue monitoring using optical imaging of face on safe driving system. In Proceedings of the International Conference on Control, Electronics, Renewable Energy and Communications (ICCEREC), Bandung, Indonesia, 13–15 September 2016; pp. 115–120. [Google Scholar]
- Iozzia, L.; Cerina, L.; Mainardi, L. Relationships between heart-rate variability and pulse-rate variability obtained from video-PPG signal using ZCA. Physiol. Meas. 2016, 37, 1934–1944. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Xu, G.-Q.; Wang, M.; Zhou, Y.; Feng, W. Webcam based non-contact real-time monitoring for the physiological parameters of drivers. In Proceedings of the IEEE 4th Annual International Conference on Cyber Technology in Automation, Control, and Intelligent Systems (CYBER 2014), Hong Kong, China, 4–7 June 2014. [Google Scholar]
- Zhang, Q.; Xu, G.-Q.; Wang, M.; Zhou, Y.; Feng, W. Webcam-based, non-contact, real-time measurement for the physiological parameters of drivers. Measurement 2017, 100, 311–321. [Google Scholar] [CrossRef]
- Rahman, H.; Barua, S.; Shahina, B. Intelligent driver monitoring based on physiological sensor signals: Application using camera. In Proceedings of the IEEE 18th International Conference on Intelligent Transportation Systems, Las Palmas, Spain, 15–18 September 2015; pp. 2637–2642. [Google Scholar]
- Rahman, H.; Uddin Ahmed, M.; Begum, S.; Funk, P. Real Time Heart Rate Monitoring from Facial RGB Color Video using Webcam. In Proceedings of the 9th Annual Workshop of the Swedish Artificial Intelligence Society (SAIS) 2016, Malmö, Sweden, 2–3 June 2016. [Google Scholar]
- Gücüyener, I. A novel design of heartbeat monitoring system for the motor vehicle. Int. J. Inj. Contr. Saf. Promot. 2015, 16, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Pavlidis, I.; Levine, J.; Baukol, P. Thermal imaging for anxiety detection. In Proceedings of the IEEE workshop on Computer Vision beyond the Visible Spectrum: Methods and Applications, Hilton Head, SC, USA, 16 June 2000. [Google Scholar]
- Pavlidis, I.; Levine, J.; Baukol, P. Thermal image analysis for anxiety detection. In Proceedings of the 2001 International Conference on Image Processing, Thessaloniki, Greece, 7–10 October 2001. [Google Scholar]
- Murthy, R.; Pavlidis, I.; Tsiamyrtzis, P. Touchless Monitoring of Breathing Function. In Proceedings of the 26th Annual International Conference of the IEEE EMBS, San Francisco, CA, USA, 1–5 September 2004. [Google Scholar]
- Fei, J.; Pavlidis, I. Virtual Thermistor. In Proceedings of the 29th Annual International Conference of the IEEE EMBS, Cité Internationale, Lyon, France, 23–26 August 2007. [Google Scholar]
- Fei, J.; Pavlidis, I. Thermistor at a distance: Unobtrusive measurement of breathing. IEEE Trans. Biomed. Eng. 2010, 57, 988–998. [Google Scholar] [PubMed]
- Pereira, C.B.; Yu, X.; Czaplik, M.; Rossaint, R.; Blazek, V.; Leonhardt, S. Remote monitoring of breathing dynamics using infrared thermography. Biomed. Opt. Express 2015, 6, 4378–4394. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.; Shah, M.; da Vitoria Lobo, N. Determining driver visual attention with one camera. IEEE Trans. Intell. Transp. Syst. 2003, 4, 205–218. [Google Scholar] [CrossRef]
- Rongben, W.; Lie, G.; Bingliang, T.; Lisheng, J. Monitoring mouth movement for driver fatigue or distraction with one camera. In Proceedings of the 7th IEEE International Conference on Intelligent Transportation Systems, Washington, DC, USA, 3–6 October 2004; pp. 314–319. [Google Scholar]
- Trivedi, M.M.; Gandhi, T.; McCall, J. Looking-In and Looking-Out of a Vehicle: Computer-Vision-Based Enhanced Vehicle Safety. IEEE Trans. Intell. Transp. Syst. 2007, 8, 108–120. [Google Scholar] [CrossRef]
- Bergasa, L.M.; Buenaposada, J.M.; Nuevo, J.; Jimenez, P.; Baumela, L. Analysing driver’s attention level using computer vision. In Proceedings of the 11th International IEEE Conference on Intelligent Transportation Systems, Beijing, China, 12–15 October 2008; pp. 1149–1154. [Google Scholar]
- Viola, P.; Jones, M.J. Robust real-time face detection. Int. J. Comput. Vis. 2004, 57, 137–154. [Google Scholar] [CrossRef]
- Manoharan, R.; Chandrakala, S.; Khan, W. Drive Safe: An Intelligent System for Monitoring Stress and Pain from Drivers’ Facial Expressions. Int. J. Latest Technol. Eng. Manag. Appl. Sci. 2016, 46–51. [Google Scholar]
- Grace, R.; Byrne, V.E.; Bierman, D.M.; Legrand, J.-M.; Gricourt, D.; Davis, B.K.; Staszewski, J.J.; Carnahan, B. A drowsy driver detection system for heavy vehicles. In Proceedings of the 17th AIAA/IEEE/SAE Digital Avionics Systems Conference (DASC), Bellevue, WA, USA, 31 October–7 November 1998. [Google Scholar]
- Wang, Q.; Yang, J.; Ren, M.; Zheng, Y. Driver Fatigue Detection: A Survey. In Proceedings of the 6th World Congress on Intelligent Control and Automation, Dalian, China, 21–23 June 2006. [Google Scholar]
- Gao, H.; Yüce, A.; Thiran, J.-P. Detecting emotional stress from facial expressions for driving safety. In Proceedings of the IEEE International Conference on Image Processing (ICIP 2014), Paris, France, 27–30 October 2014; pp. 5961–5965. [Google Scholar]
- Levine, J.A.; Pavlidis, I.; Cooper, M. The face of fear. Lancet 2001, 357, 1757. [Google Scholar] [CrossRef]
- Pavlidis, I.; Levine, J. Thermal image analysis for polygraph testing. IEEE Eng. Med. Biol. Mag. 2002, 21, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Puri, C.; Olson, L.; Pavlidis, I.; Levine, J.; Starren, J. Stresscam: Non-contact measurement of users’ emotional states through thermal imaging. In Proceedings of the Conference on Human Factors in Computing Systems (CHI 2005), Portland, OR, USA, 2–7 April 2005. [Google Scholar]
- Khan, M.M.; Ward, R.D.; Ingleby, M. Infrared Thermal Sensing of Positive and Negative Affective States. In Proceedings of the IEEE Conference on Robotics, Automation and Mechatronics, Bangkok, Thailand, 1–3 June 2006. [Google Scholar]
- Hernandez, B.; Olague, G.; Hammoud, R.; Trujillo, L.; Romero, E. Visual learning of texture descriptors for facial expression recognition in thermal imagery. Comput. Vis. Image Underst. 2007, 106, 258–269. [Google Scholar] [CrossRef]
- Nhan, B.R.; Chau, T. Classifying Affective States Using Thermal Infrared Imaging of the Human Face. IEEE Trans. Biomed. Eng. 2010, 57, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Kotani, K.; Chen, F.; Le, B. Estimation of Human Emotions using Thermal Facial Information. In Proceedings of the Fifth International Conference on Graphic and Image Processing (ICGIP 2013), Hong Kong, China, 26–27 October 2013. [Google Scholar]
- Hong, K.; Liu, G. Facial Thermal Image Analysis for Stress Detection. Int. J. Eng. Res. Technol. 2017, 6, 94–98. [Google Scholar]
- Reyes, M.L.; Lee, J.D.; Liang, Y.; Hoffman, J.D.; Huang, R.W. Capturing driver response to in-vehicle human-machine interface technologies using Facial Thermography. In Proceedings of the 5th International Driving Symposium on Human Factors in Driver Assessment, Training and Vehicle Design, Big Sky, MT, USA, 22–25 June 2009. [Google Scholar]
- Zomotor, Z.; Franke, U. Sensor fusion for improved vision based lane recognition and object tracking with range-finders. In Proceedings of the Conference on Intelligent Transportation Systems, Boston, MA, USA, 9–12 November 1997; pp. 595–600. [Google Scholar]
- Fang, Y.; Masaki, I.; Horn, B. Depth-based target segmentation for intelligent vehicles: Fusion of radar and binocular stereo. IEEE Trans. Intell. Transp. Syst. 2002, 3, 196–202. [Google Scholar] [CrossRef]
- She, K.; Bebis, G.; Gu, H.; Miller, R. Vehicle tracking using on-line fusion of color and shape features. In Proceedings of the 7th International IEEE Conference on Intelligent Transportation Systems, Washington, DC, USA, 3–6 October 2004; pp. 731–736. [Google Scholar]
- Krotosky, S.J.; Trivedi, M.M. A Comparison of Color and Infrared Stereo Approaches to Pedestrian Detection. In Proceedings of the 2007 IEEE Intelligent Vehicles Symposium, Istanbul, Turkey, 13–15 June 2007; pp. 81–86. [Google Scholar]
- Pavlidis, I.; Symosek, P.; Fritz, B.; Papanikopoulos, N.; Schwartz, K. Automatic detection of vehicle passengers through near-infrared fusion. In Proceedings of the IEEE Conference on Intelligent Transportation Systems (ITSC), Tokyo, Japan, 5–8 October 1999. [Google Scholar]
- Pavlidis, I.; Morellas, V.; Papanikolopoulos, N. A vehicle occupant counting system based on near-infrared phenomenology and fuzzy neural classification. IEEE Trans. Intell. Transp. Syst. 2000, 1, 72–85. [Google Scholar] [CrossRef]
- Owechko, Y.; Srinivasa, N.; Medasani, S.; Boscolo, R. Vision-based fusion system for smart airbag applications. IEEE Intell. Veh. Symp. 2002, 1, 245–250. [Google Scholar] [CrossRef]
- Trivedi, M.M.; Cheng, S.Y.; Childers, E.M.C.; Krotosky, S.J. Occupant posture analysis with stereo and thermal infrared video: Algorithms and experimental evaluation. IEEE Trans. Veh. Technol. 2004, 53, 1698–1712. [Google Scholar] [CrossRef]
- Gyaourova, A.; Bebis, G.; Pavlidis, I. Fusion of infrared and visible images for face recognition. In Proceedings of the 8th European Conference on Computer Vision (ECCV 2004), Prague, Czech Republic, 11–14 May 2004; pp. 456–468. [Google Scholar]
- Li, L.; Chen, Y.; Li, Z. Yawning detection for monitoring driver fatigue based on two cameras. In Proceedings of the 12th International IEEE Conference on Intelligent Transportation Systems, St Louis, MO, USA, 4–7 October 2009; pp. 1–6. [Google Scholar]
- Corneanu, C.A.; Simron, M.O.; Cohn, J.F.; Escalera Guerrero, S. Survey on RGB, 3D, Thermal, and Multimodal Approaches for Facial Expression Recognition: History, Trends, and Affect-Related Applications. IEEE Trans. Pattern Anal. Mach. Intell. 2016, 38, 1548–1568. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Pan, B.; Chen, H.; Ji, Q. Thermal Augmented Expression Recognition. IEEE Trans. Cybern. 2018, 48, 2203–2214. [Google Scholar] [CrossRef] [PubMed]
- Blanik, N.; Blazek, C.; Pereira, C.; Blazek, V.; Leonhardt, S. Frequency-selective quantification of skin perfusion behavior during allergic testing using photoplethysmography imaging. In Proceedings of the SPIE Medical Imaging 2014: Image Processing, San Diego, CA, USA, 15–20 February 2014. [Google Scholar]
- Balakrishnan, G.; Durand, F.; Guttag, J. Detecting Pulse from Head Motions in Video. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition (CVPR 2013), Portland, OR, USA, 23–28 June 2013; pp. 3430–3437. [Google Scholar]
- Viola, P.; Jones, M. Rapid object detection using a boosted cascade of simple features. In Proceedings of the 2001 International Conference on Computer Vision and Pattern Recognition (CVPR 2001), Kauai Island, HI, USA, 8–14 December 2001; pp. 511–518. [Google Scholar]
- Hoog Antink, C.; Gao, H.; Brueser, C.; Leonhardt, S. Beat-to-beat heart rate estimation fusing multimodal video and sensor data. Biomed. Opt. Express 2015, 6, 2895–2907. [Google Scholar] [CrossRef] [PubMed]
- Teichmann, D.; Foussier, J.; Jia, J.; Leonhardt, S.; Walter, M. Noncontact monitoring of cardiorespiratory activity by electromagnetic coupling. IEEE Trans. Biomed. Eng. 2013, 60, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Cordes, A. Monitoring von Atmung und Herzaktivität Mittels Magnetischer Induktionsmessung. Ph.D. Thesis, RWTH Aachen University, Aachen, Germany, 2013. [Google Scholar]
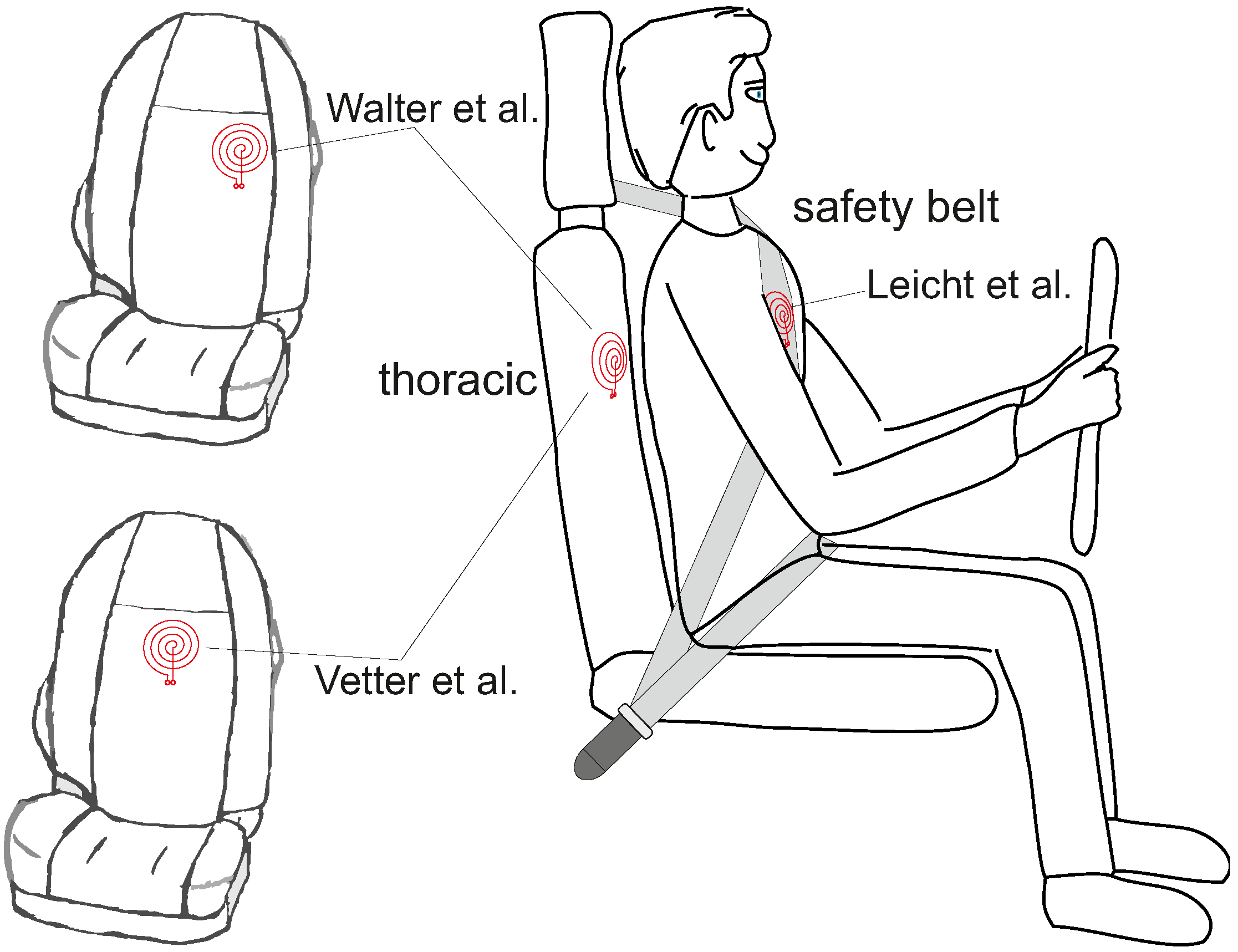
- Leicht, L.; Vetter, P.; Leonhardt, S.; Teichmann, D. The PhysioBelt: A Safety Belt Integrated Sensor System for Heart Activity and Respiration. In Proceedings of the IEEE International Conference on Vehicular Electronics and Safety (IEEE ICVES 2017), Vienna, Austria, 27–28 June 2017. [Google Scholar]
- Vetter, P.; Leicht, L.; Leonhardt, S.; Teichmann, D. Integration of an Electromagnetic Coupled Sensor into a Driver Seat for Vital Sign Monitoring: Initial Insight. In Proceedings of the IEEE International Conference on Vehicular Electronics and Safety (IEEE ICVES 2017), Vienna, Austria, 27–28 June 2017. [Google Scholar]
- Lazaro, A.; Girbau, D.; Villarino, R. Analysis of Vital Signs Monitoring using an IR-UWB Radar. Prog. Electromag. Res. 2010, 100, 265–284. [Google Scholar] [CrossRef]
- Singh, M.; Ramachandran, G. Reconstruction of sequential cardiac in-plane displacement patterns on the chest wall by laser speckle interferometry. IEEE Trans. Biomed. Eng. 1991, 38, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.; Brown, T.; Schwieger, K.; Zimmermann, E.; Malik, W.; Edwards, D.; Ouvry, L.; Oppermann, I. Ultra Wideband: Applications, Technology and Future perspectives. In Proceedings of the 1st International Workshop on Convergent Technologies (IWCT 2005), Oulu, Finland, 6–10 June 2005. [Google Scholar]
- Immoreev, I.Y.; Samkov, S.V. Ultra-Wideband Radar for remote Detection and Measurement of Parameters of the moving Objects on a small Range. In Proceedings of the Conference on Ultrawideband and Ultrashort Impulse Signals, Sevastopol, Ukraine, 19–22 September 2004; pp. 214–216. [Google Scholar]
- TES Electronic Solutions: Whitepaper on Vital Sign Detection Using 60 GHz Radar Technology. Available online: http://www.tes-dst.com/tes-dst/download/White_Paper_Vital_Sign_Detection_60GHz_ENG.pdf (accessed on 5 September 2018).
- Kao, T.-Y.J.; Lin, J. Vital sign detection using 60-GHz Doppler radar system. In Proceedings of the 2013 IEEE International Wireless Symposium (IWS), Bejing, China, 14–18 April 2013. [Google Scholar]
- Caro, C.G.; Bloice, J.A. Contactless apnoea detector based on radar. Lancet 1971, 298, 959–961. [Google Scholar] [CrossRef]
- Ichapurapu, R.; Jain, S.; John, G.; Monday, T.; Lie, D.Y.C.; Banister, R.; Griswold, J. A 2.4 GHz non-contact biosensor system for continuous vital-signs monitoring on a single PCB. In Proceedings of the IEEE 8th International Conference on ASIC (ASICON ’09), Changsha, China, 20–23 October 2009. [Google Scholar]
- Massagram, W.; Lubecke, V.M.; Host-Madsen, A.; Boric-Lubecke, O. Assessment of Heart Rate Variability and Respiratory Sinus Arrhythmia via Doppler Radar. IEEE Trans. Microw. Theory Tech. 2009, 57, 2542–2549. [Google Scholar] [CrossRef]
- Scalise, L.; De Leo, A.; Mariani Primiani, V.; Russo, P.; Shahu, D.; Cerri, G. Non contact monitoring of the respiration activity by electromagnetic sensing. In Proceedings of the 2011 IEEE International Symposium on Medical Measurements and Applications, Bari, Italy, 30–31 May 2011; pp. 418–422. [Google Scholar]
- Bechet, P.; Mitran, R.; Munteanu, M. A non-contact method based on multiple signal classification algorithm to reduce the measurement time for accurately heart rate detection. Rev. Sci. Instrum. 2013, 84, 084707. [Google Scholar] [CrossRef] [PubMed]
- Vinci, G.; Lenhard, T.; Will, C.; Koelpin, A. Microwave Interferometer Radar-Based Vital Sign Detection for Driver Monitoring Systems. In Proceedings of the 2015 IEEE MTT-S International Conference on Microwaves for Intelligent Mobility (ICMIM), Heidelberg, Germany, 27–29 April 2015. [Google Scholar]
- Lee, K.J.; Park, C.; Lee, B. Tracking Driver’s Heart Rate by Continuous-Wave Doppler Radar. In Proceedings of the 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC’16), Orlando, FL, USA, 16–20 August 2016. [Google Scholar]
- Izumi, S.; Matsunaga, D.; Nakamura, R.; Kawaguchi, H.; Yoshimoto, M. A Contact-Less Heart Rate Sensor System for Driver Health Monitoring. Available online: https://pdfs.semanticscholar.org/9059/6a41f8642c5854f88e02a3e121a151747434.pdf (accessed on 31 August 2018).
- Schires, E.; Georgiou, P.; Lande, T.S. Vital Sign Monitoring through the Back using an UWB Impulse Radar with Body Coupled Antennas. IEEE Trans. Biomed. Circuits Syst. 2018, 12, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Serteyn, A.; Vullings, R.; Meftah, M.; Bergmans, J.W. Motion artifacts in capacitive ECG measurements: Reducing the combined effect of DC voltages and capacitance changes using an injection signal. IEEE Trans. Biomed. Eng. 2015, 62, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Wang, F.; Horng, T. Single Self-Injection-Locked Radar With Two Antennas for Monitoring Vital Signs With Large Body Movement Cancellation. IEEE Trans. Microw. Theory Tech. 2017, 65, 5324–5333. [Google Scholar] [CrossRef]
- Ottenbacher, J.; Heuer, S. Motion artefacts in capacitively coupled ECG electrodes. Proc. World Congr. Med. Phys. Biomed. Eng. 2010, 25, 1059–1062. [Google Scholar]
- Eilebrecht, B.; Wartzek, T.; Willkomm, J.; Schommartz, A.; Walter, M.; Leonhardt, S. Motion Artifact Removal from Capacitive ECG Measurements by Means of Adaptive Filtering. In 5th European Conference of the International Federation for Medical and Biological Engineering; Springer: Heidelberg, Germany, 2011; Volume 37, pp. 902–905. [Google Scholar]
- Eilebrecht, B.; Willkomm, J.; Pohl, A.; Wartzek, T.; Leonhardt, S. Impedance measurement system for determination of capacitive electrode coupling. IEEE Trans. Biomed. Circuits Syst. 2013, 7, 682–689. [Google Scholar] [CrossRef] [PubMed]
- Devereux, H.; Smalley, M. Are infra red illuminators eye safe? In Proceedings of the IEEE 29th Annual 1995 International Carnahan Conference on Security Technology, Surrey, UK, 18–20 October 1995. [Google Scholar]
- Jeanne, V.; Asselman, M.; den Brinker, B.; Bulut, M. Camera-based heart rate monitoring in highly dynamic light conditions. In Proceedings of the International Conference on Connected Vehicles and Expo (ICCVE 2013), Las Vegas, NV, USA, 2–6 December 2013. [Google Scholar]
- Lee, D.; Kim, J.; Kwon, S.; Park, K. Heart rate estimation from facial photoplethysmography during dynamic illuminance changes. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milano, Italy, 25–29 August 2015. [Google Scholar]
- Hoog Antink, C.; Schulz, F.; Leonhardt, S.; Walter, M. Motion Artifact Quantification and Sensor Fusion for Unobtrusive Health Monitoring. Sensors 2017, 18, 38. [Google Scholar] [CrossRef] [PubMed]
- Ivanic, P.; Novotny, I.; Rehacek, V.; Tvarozek, V.; Weis, M. Thin film non-symmetric microelectrode array for impedance monitoring of human skin. Thin Solid Films 2003, 433, 332–336. [Google Scholar] [CrossRef]



| 1967 | First description of insulated electrodes (Richardson, [35]) |
| 1993 | cECG in objects of daily life (Ishijima, [36]) |
| 2004 | cECG in a bathtub and toilet seat (Lim, [37], Kim [38]) |
| 2006 | Integration of cECG into a chair (Lim, [39]) |
| 2007 | cECG integrated into beds (Lim, [40]) |
| 2008 | First integration of cECG into an automotive seat (Leonhardt, [42]) |
| 2008 | Textile cECG electrodes in an automotive seat (Chamadiya, [43]) |
| 2011 | Evaluation of cECG during test drives (Eilebrecht, [44]) |
| 2011 | Measurement performance and coverage rate tested during highway and city traffic (Wartzek, [45]) |
| 2011 | Triboelectricity and motion artifacts identified as factor for robustness (Wartzek, [46]) |
| 2011 | cECG evaluated as a technology for passenger monitoring in airplanes (Schumm, [47]) |
| 2012 | Introduction of a quality label to describe cECG signal quality (Schumm, [47]) |
| 2012 | Textile seat cover for retrofitting seats with cECG systems (Schneider, [48]) |
| 2012 | cECG used for heart rate variability analysis (HRV) (Jung, [49]) |
| 2012 | First cECG sensor system commercially available: EPIC system by Plessey Inc. |
| 2015 | Active humidification further evaluated (Fong [50], Weder [51]) |
| 2016 | WARDEN seat cover based system for retrofitting (Plessey, [52]) |
| 2017 | Closed-loop humidification for artifact suppression (Leicht, [53]) |
| 2017 | IMEC sensor system fusing cECG and radar (IMEC, [54]) |
| 2018 | Clinical trial to evalute cECG as a diagnostic tool for heart attack survivors (Leicht, [55]) |
| System | Year | References | Electrode Properties | Special Properties | ||
|---|---|---|---|---|---|---|
| # | Shape | Ground | ||||
| SMART Test Vehicle | 2008 | [42] | 2 | rectangular | in backrest | isolated (coated) metal electrodes |
| Daimler S-class seat | 2008 | [43] | 2 | round | in seat plane | |
| Ford S-Max seat | 2011 | [44] | 6 | rectangular | in seat plane | multi-electrode system |
| Daimler Test Vehicle | 2011 | [58] | 2 | rectangular | textile electrodes | |
| Audi Q5 | 2012 | [48] | 2 | rectangular | in backrest | textile electrodes, removable seat cover |
| Car seat | 2012 | [49] | 2 | rectangular | in seat plane | |
| Ford S-Max seat | 2014/2017 | [53,60] | 2 | rectangular | in seat plane | textile electrodes, release of humidity |
| Plessey EPICTM seat | 2014 | [59] | 3 | round | ||
| Ford S-Max Test Vehicle | 2015 | [61] | 6 | round | in seat plane | deep-drawn electrodes |
| Plessey WARDENTM | 2016 | [52] | 6 | rectangular | backrest, built-into main unit | removable seat cover |
| IMEC car seat | 2017 | [54,65] | 6 | round | up to 64 electrodes possible | |
| Name | Freq. Range | Abb. | Price | Energy | Special Properties |
|---|---|---|---|---|---|
| Visible Light (Vis) | 350 nm–740 nm | VIS | cheap | ambient light (passive) or | ambient light may fluctuate, |
| active illumination | passive mode does not work at night | ||||
| Near Infrared (NIR) | 740 nm–1 μm | NIR | cheap | active illumination | works at night, long exposure may exhaust the retina |
| Far Infrared (FIR) | 3 μm–5 μm | MWIR | expensive | passive | works at night, but glasses are not transparent |
| Far Infrared (FIR) | 8 μm–14 μm | LWIR | expensive | passive | works at night, but glasses are not transparent |
| Name | Abbreviation | First Mentioned | References (First Author, Year) |
|---|---|---|---|
| PPG imaging | PPGi/PPGI | 2000 | Blazek 2000 [81], Wu 2000 [82], Hulsbusch 2002 [83], Blazek 2006 [93], |
| Karlen 2015 [94], Moco 2016 [95], Blazek 2017 [96], Blöcher 2017 [97] | |||
| Vital Signs Camera | VSC | 2004 | Philips Research 2004 [98] |
| imaging PPG | iPPG/IPPG | 2007 | Zheng 2007 [86], Hu 2008 [99], Sun 2014 [100], Kamshilin 2015 [101], |
| Karlen 2015 [94], Kuo 2015 [102], Blackford 2016 [103], Sun 2016 [79] | |||
| remote PPG (imaging) | n.a. | 2008 | Verkruysse 2008 [87], McDuff 2015 [104] |
| remote PPG | rPPG | 2015 | Kwon 2015 [105], Gastel 2015 [106], Wu 2016 [107], Gastel 2016 [108] |
| DistancePPG | n.a. | 2015 | Kumar 2015 [109] |
| video-based HR | n.a. | 2015 | Choe 2015 [110] |
| camera-based PPG | cbPPG | 2015 | Wedekind 2015 [111], Rasche 2016 [112], Kevat 2017 [113] |
| non-contact PPG | ncPPG | 2016 | Butler 2016 [114], Tayibnapis 2016 [115] |
| video-PPG | vPPG | 2016 | Iozzia 2016 [116] |
| Sensor Technique | Type of Contact | Measured Quantity | Energy Injection | Distance | Sensitivity to Positioning | Costs | RL | In-Car Use |
|---|---|---|---|---|---|---|---|---|
| ECG (steering wheel) | galvanic | electric biopotential | no | 0 | + | o | + | [20,21,24,25,27,28,29,31,32,33] |
| cECG | capacitive | electric biopotential | no | mm | + | o | o | [42,43,44,48,49,52,58,59,60,61] |
| BCG | mechanical | displacement, force | no | 0 | – | o | o | [42,74,75,76,78] |
| Video motion | optical | displacement | no | m | – | + | + | not yet |
| PPG (steering wheel) | optical | photon absorption | yes | mm | o | – | + | [23,25,26] |
| PPGi | optical | photon absorption | yes | m | – | + | o | [115,121] |
| Thermography | optical | radiation, temperature | no | m | – | ++ | + | [124,126] |
| MI | electromagnetic | electric bioimpedance | yes | cm | o | o | – | [74,164,165] |
| Radar | electromagnetic | displacement, velocity | yes | m | – | o | o | [177,178,179,180] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonhardt, S.; Leicht, L.; Teichmann, D. Unobtrusive Vital Sign Monitoring in Automotive Environments—A Review. Sensors 2018, 18, 3080. https://doi.org/10.3390/s18093080
Leonhardt S, Leicht L, Teichmann D. Unobtrusive Vital Sign Monitoring in Automotive Environments—A Review. Sensors. 2018; 18(9):3080. https://doi.org/10.3390/s18093080
Chicago/Turabian StyleLeonhardt, Steffen, Lennart Leicht, and Daniel Teichmann. 2018. "Unobtrusive Vital Sign Monitoring in Automotive Environments—A Review" Sensors 18, no. 9: 3080. https://doi.org/10.3390/s18093080
APA StyleLeonhardt, S., Leicht, L., & Teichmann, D. (2018). Unobtrusive Vital Sign Monitoring in Automotive Environments—A Review. Sensors, 18(9), 3080. https://doi.org/10.3390/s18093080






