Screening Method for Anti-Colon Cancer Drugs Using Two Sensor Cell Lines with Human β4-Galactosyltransferase 4 Gene Promoters
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Culture
2.3. Reporter Plasmid Construction
2.4. Establishment of Sensor Cell Lines
2.5. Treatment with Compounds
2.6. Luciferase Assay
2.7. Immunoblot Analysis
2.8. Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
2.9. Statistical Analysis
3. Results and Discussion
3.1. Establishment of Two Sensor Cell Lines with β4GalT4 Gene Promoter Regions
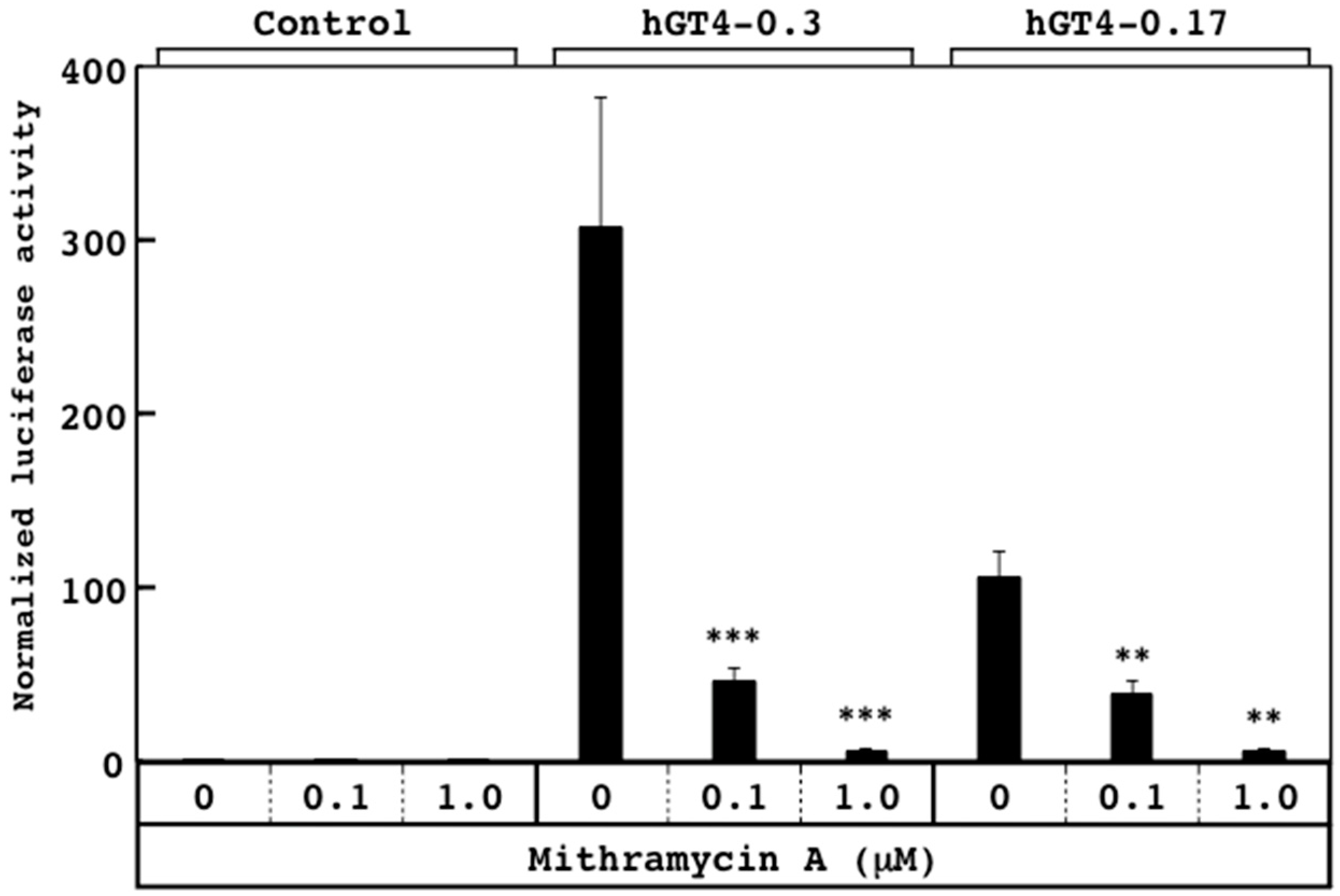
3.2. Responsiveness of Sensor Cells to Mithramycin A
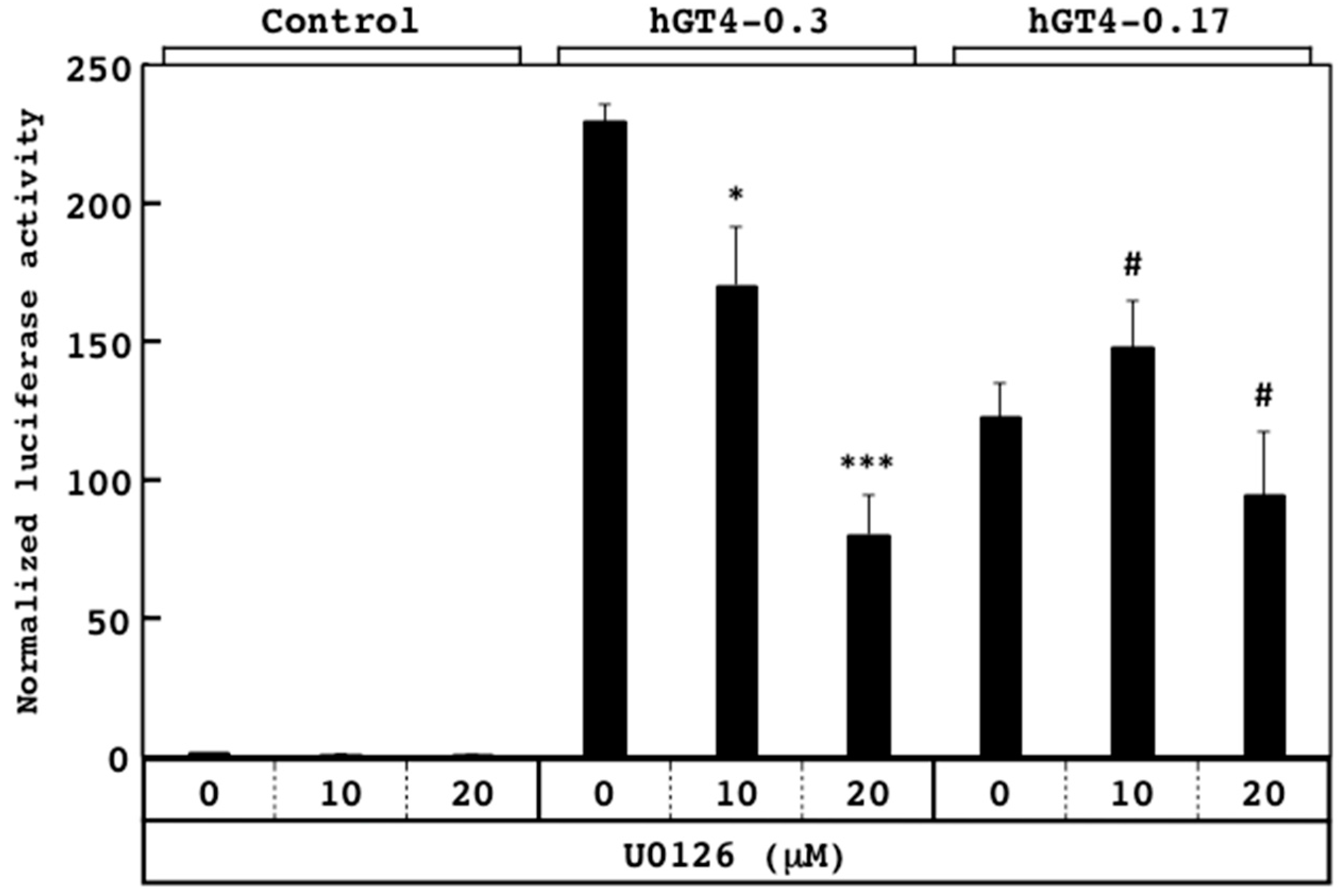
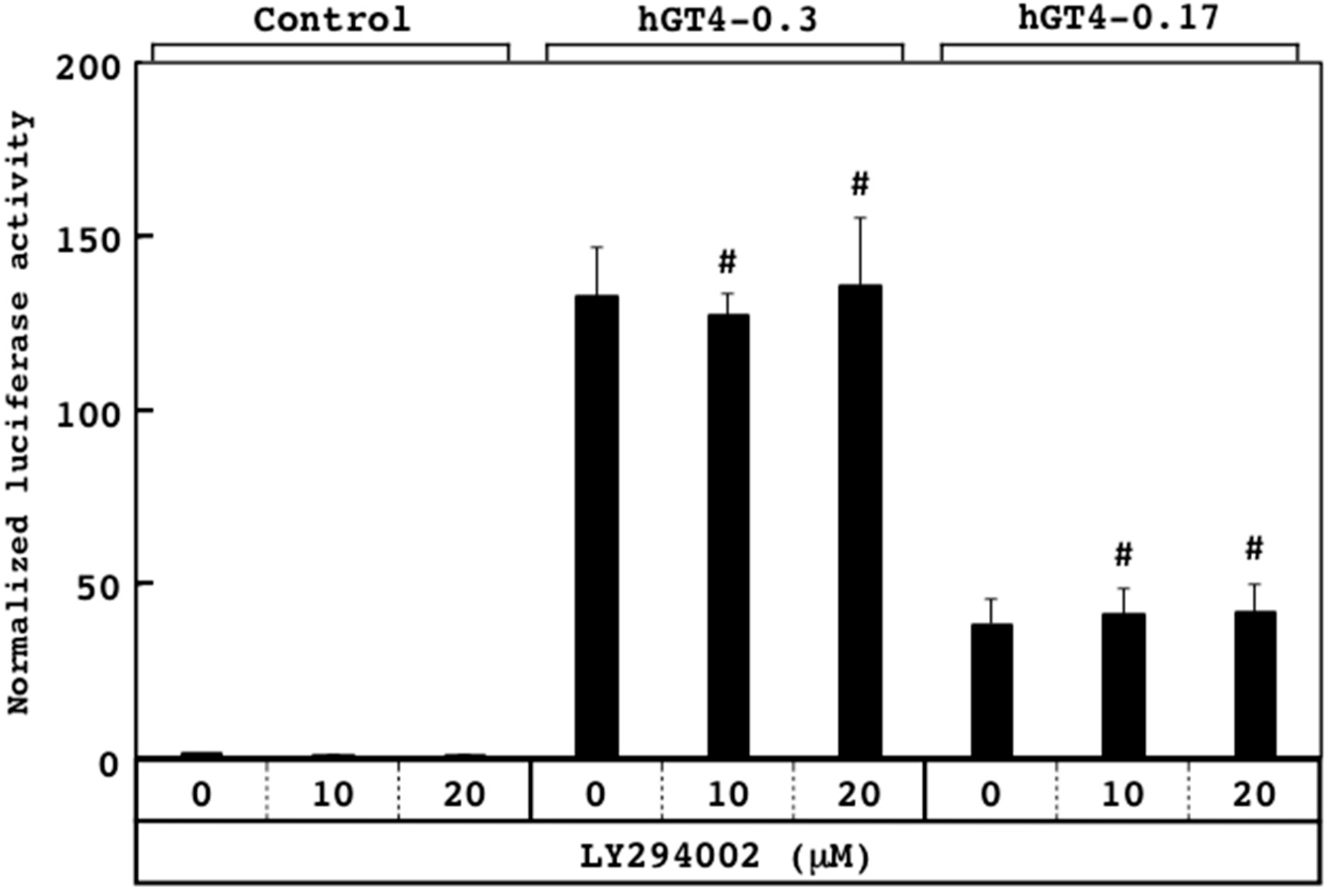
3.3. Responsiveness of Sensor Cells to U0126 and LY294002
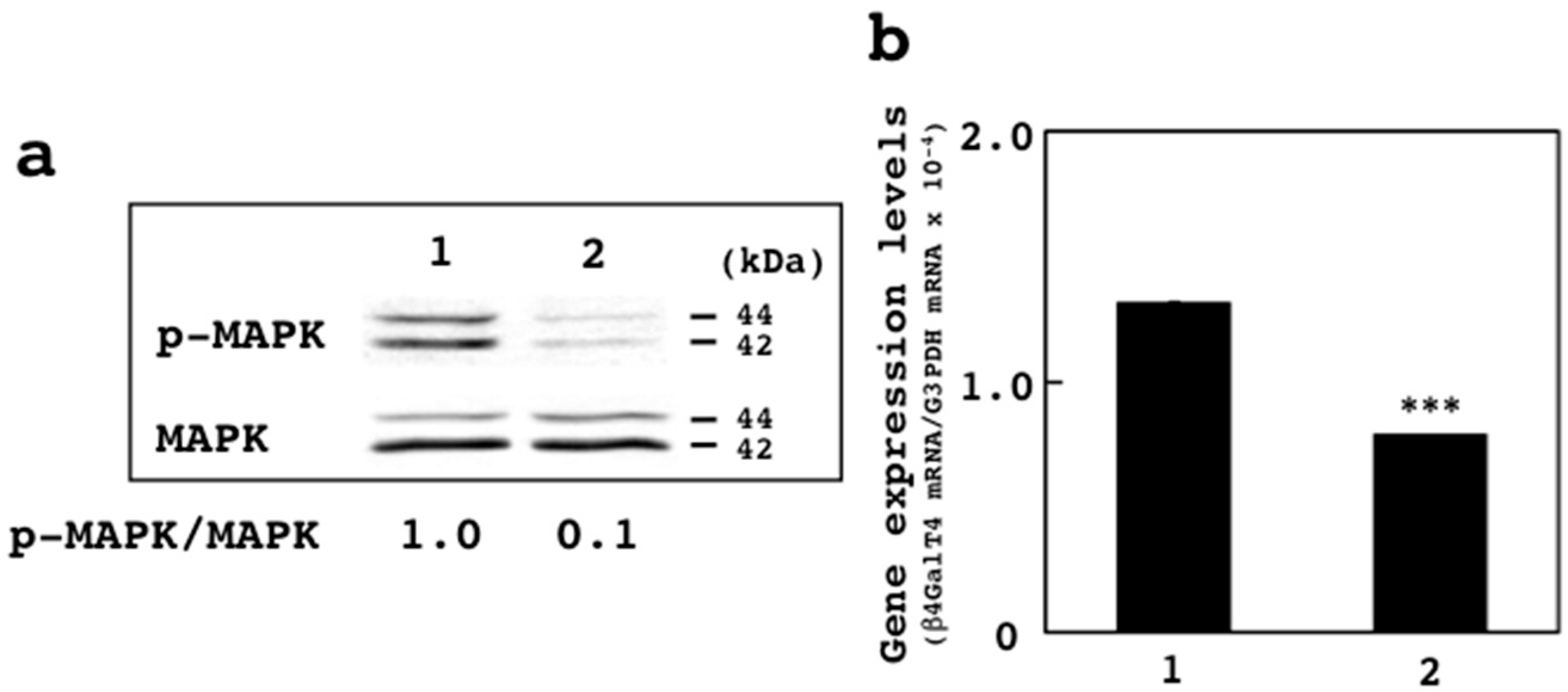
3.4. Effects of U0126 on Expression of β4GalT4 Gene
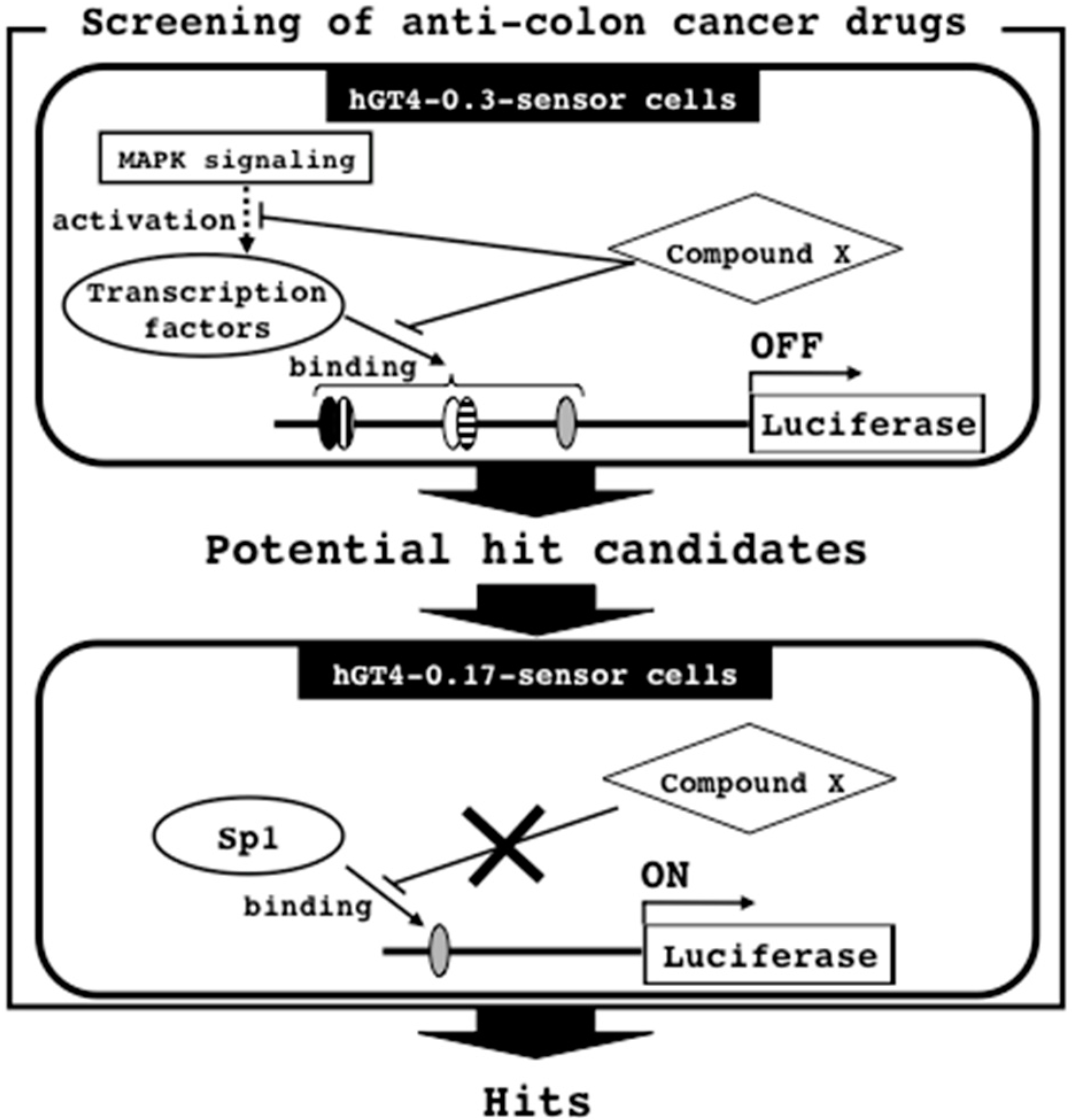
3.5. Screening Strategy for Anti-Colon Cancer Drugs
3.6. MAPK Signaling and β4GalT4 Gene Expression
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kobata, A. Altered glycosylation of surface glycoproteins in tumor cells and its clinical application. Pigment Cell Res. 1989, 2, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Hakomori, S. Glycosylation defining cancer malignancy: New wine in an old bottle. Proc. Natl. Acad. Sci. USA 2002, 81, 10231–10233. [Google Scholar] [CrossRef] [PubMed]
- Asada, M.; Furukawa, K.; Segawa, K.; Endo, T.; Kobata, A. Increased expression of highly branched N-glycans at cell surface is correlated with the malignant phenotypes of mouse tumor cells. Cancer Res. 1997, 57, 1073–1080. [Google Scholar] [PubMed]
- Guo, H.B.; Lee, I.; Kamar, M.; Akiyama, S.K.; Pierce, M. Aberrant N-glycosylation of β1 integrin causes reduced α5β1 integrin clustering and stimulates cell migration. Cancer Res. 2002, 62, 6837–6845. [Google Scholar] [PubMed]
- Dennis, J.W. Effects of swainsonine and polyinosinic:polycytidylic acid on murine tumor cell growth and metastasis. Cancer Res. 1986, 46, 5131–5136. [Google Scholar] [PubMed]
- Humphries, M.J.; Matsumoto, K.; White, S.L.; Olden, K. Inhibition of experimental metastasis by castanospermine in mice: Blockage of two distinct stages of tumor colonization by oligosaccharide processing inhibitors. Cancer Res. 1986, 46, 5215–5222. [Google Scholar] [PubMed]
- Furukawa, K.; Clausen, H.; Sato, T. UDP-Gal: βGlcNAc β1,4-galactosyltransferase, polypeptide 2-6; xylosylprotein β1, 4-galactosyltransferase, polypeptide 7 (galactosyltransferase I) (B4GALT2-7). In Handbook of Glycosyltransferases and Related Genes; Taniguchi, N., Honke, K., Fukuda, M., Narimatsu, H., Yamaguchi, Y., Angata, T., Eds.; Springer: Tokyo, Japan, 2014; pp. 63–72. ISBN 978-4-431-54241-4. [Google Scholar]
- Shirane, K.; Kuji, R.; Tareyanagi, C.; Sato, T.; Kobayashi, Y.; Furukawa, S.; Murata, T.; Kubota, S.; Ishikawa, Y.; Segawa, K.; et al. Gene expression levels of β4-galactosyltransferase 5 correlate with the tumorigenic potentials of B16-F10 mouse melanoma cells. Glycobiology 2014, 24, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.S.; Chang, H.Y.; Li, C.P.; Liu, J.M.; Huang, T.S. Tumor β-1,4-galactosyltransferase IV overexpression is closely associated with colorectal cancer metastasis and poor prognosis. Clin. Cancer Res. 2005, 11, 8615–8622. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, A.; Fukushima, N.; Sato, T. Transcriptional mechanism of the β4-galactosyltransferase 4 gene in SW480 human colon cancer cell line. Biol. Pharm. Bull. 2017, 40, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Mulloy, B.; Dell, A.; Stanley, P.; Prestegard, J.H. Structural analysis of glycans. In Essentials of Glycobiology, 3rd ed.; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2017; pp. 639–652. ISBN 978-1-621821-32-8. [Google Scholar]
- Lynch, R.A.; Etchin, J.; Battle, T.E.; Frank, D.A. A small-molecule enhancer of signal transducer and activator of transcription 1 transcriptional activity accentuates the antiproliferative effects of IFN-gamma in human cancer cells. Cancer Res. 2007, 67, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Migita, S.; Kanehira, K.; Sonezaki, S.; Taniguchi, A. Development of sensor cells using NF-κB pathway activation for detection of nanoparticle-induced inflammation. Sensors 2011, 11, 7219–7230. [Google Scholar] [CrossRef] [PubMed]
- Cooksey, R.C.; Crawford, J.T.; Jacobs, W.R., Jr.; Shinnick, T.M. A rapid method for screening antimicrobial agents for activities against a strain of Mycobacterium tuberculosis expressing firefly luciferase. Antimicrob. Agents Chemother. 1993, 37, 1348–1352. [Google Scholar] [CrossRef] [PubMed]
- Baldari, C.T.; Di Somma, M.M.; Majolini, M.B.; Ulivieri, C.; Milia, E.; Telford, J.L. NF-AT-luciferase reporter T cell lines as tools to screen immunosuppressive drugs. Biologicals 1998, 26, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Egawa, K.; Nose, K.; Kunimoto, S.; Takeuchi, T. HIF-1-dependent VEGF reporter gene assay by a stable transformant of CHO cells. Biol. Pharm. Bull. 2003, 26, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Chiba-Mizutani, T.; Miura, H.; Matsuda, M.; Matsuda, Z.; Yokomaku, Y.; Miyauchi, K.; Nishizawa, M.; Yamamoto, N.; Sugiura, W. Use of new T-cell-based cell lines expressing two luciferase reporters for accurately evaluating susceptibility to anti-human immunodeficiency virus type 1 drugs. J. Clin. Microbiol. 2007, 45, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Miao, J.; Wang, J.; Li, Q.; Cui, L. Plasmodium falciparum: Development of a transgenic line for screening antimalarials using firefly luciferase as the reporter. Exp. Parasitol. 2008, 120, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Furukawa, K. Transcriptional regulation of the human β-1,4-galactosyltransferase V gene in cancer cells: Essential role of transcription factor Sp1. J. Biol. Chem. 2004, 279, 39574–39583. [Google Scholar] [CrossRef] [PubMed]
- Anders, M.; Christian, C.; McMahon, M.; McCormick, F.; Korn, W.M. Inhibition of the Raf/MEK/ERK pathway up-regulates expression of the coxsackievirus and adenovirus receptor in cancer cells. Cancer Res. 2003, 63, 2088–2095. [Google Scholar] [PubMed]
- Wang, H.; Duan, L.; Zou, Z.; Li, H.; Yuan, S.; Chen, X.; Zhang, Y.; Li, X.; Sun, H.; Zha, H.; et al. Activation of the PI3K/Akt/mTOR/p70S6K pathway is involved in S100A4-induced viability and migration in colorectal cancer cells. Int. J. Med. Sci. 2014, 11, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Muramoto, K.; Tange, R.; Ishii, T.; Miyauchi, K.; Sato, T. Downregulation of transcription factor Sp1 suppresses malignant properties of A549 human lung cancer cell line with decreased β4-galactosylation of highly branched N-glycans. Biol. Pharm. Bull. 2017, 40, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- Beishline, K.; Azizkhan-Clifford, J. Sp1 and the ‘hallmarks of cancer’. FEBS J. 2015, 282, 224–258. [Google Scholar] [CrossRef] [PubMed]
- Tsunoda, T.; Takagi, T. Estimating transcription factor bindability on DNA. Bioinformatics 1999, 15, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Ropponen, K.M.; Kellokoski, J.K.; Pirinen, R.T.; Moisio, K.I.; Eskelinen, M.J.; Alhava, E.M.; Kosma, V.M. Expression of transcription factor AP-2 in colorectal adenomas and adenocarcinomas; comparison of immunohistochemistry and in situ hybridisation. J. Clin. Pathol. 2001, 54, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y.; Nosho, K.; Shima, K.; Freed, E.; Irahara, N.; Philips, J.; Meyerhardt, J.A.; Hornick, J.L.; Shivdasani, R.A.; Fuchs, C.S.; et al. Relationship of CDX2 loss with molecular features and prognosis in colorectal cancer. Clin. Cancer Res. 2009, 15, 4665–4673. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Gorlick, R.; Liefshitz, A.; Danenberg, K.; Danenberg, P.C.; Danenberg, P.V.; Klimstra, D.; Jhanwar, S.; Cordon-Cardo, C.; Fong, Y.; et al. Levels of E2F-1 expression are higher in lung metastasis of colon cancer as compared with hepatic metastasis and correlate with levels of thymidylate synthase. Cancer Res. 2000, 60, 2365–2367. [Google Scholar] [PubMed]
- Sase, T.; Suzuki, T.; Miura, K.; Shiiba, K.; Sato, I.; Nakamura, Y.; Takagi, K.; Onodera, Y.; Miki, Y.; Watanabe, M.; et al. Runt-related transcription factor 2 in human colon carcinoma: A potent prognostic factor associated with estrogen receptor. Int. J. Cancer 2012, 131, 2284–2293. [Google Scholar] [CrossRef] [PubMed]
- Ray, R.; Snyder, R.C.; Thomas, S.; Koller, C.A.; Miller, D.M. Mithramycin blocks protein binding and function of the SV40 early promoter. J. Clin. Investig. 1989, 83, 2003–2007. [Google Scholar] [CrossRef] [PubMed]
- Favata, M.F.; Horiuchi, K.Y.; Manos, E.J.; Daulerio, A.J.; Stradley, D.A.; Feeser, W.S.; Van Dyk, D.E.; Pitts, W.J.; Earl, R.A.; Hobbs, F.; et al. Identification of a novel inhibitor of mitogen-activated protein kinase kinase. J. Biol. Chem. 1998, 273, 18623–18632. [Google Scholar] [CrossRef] [PubMed]
- Vlahos, C.J.; Matter, W.F.; Hui, K.Y.; Brown, R.F. A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002). J. Biol. Chem. 1994, 269, 5241–5248. [Google Scholar] [PubMed]
- Roberts, P.J.; Der, C.J. Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 2007, 26, 3291–3310. [Google Scholar] [CrossRef] [PubMed]
- Berkovich, E.; Ginsberg, D. Ras induces elevation of E2F-1 mRNA levels. J. Biol. Chem. 2001, 276, 42851–42856. [Google Scholar] [CrossRef] [PubMed]
- Niu, D.-F.; Kondo, T.; Nakazawa, T.; Oishi, N.; Kawasaki, T.; Mochizuki, K.; Yamane, T.; Katoh, R. Transcription factor Runx2 is a regulator of epithelial–mesenchymal transition and invasion in thyroid carcinomas. Lab. Investig. 2012, 92, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.Y.; Black, A.R.; Kostic, D.; Pajovic, S.; Hoover, C.N.; Azizkhan, J.C. Cell cycle-regulated association of E2F1 and Sp1 is related to their functional interaction. Mol. Cell. Biol. 1996, 16, 1668–1675. [Google Scholar] [CrossRef] [PubMed]
- Tsantoulis, P.K.; Gorgoulis, V.G. Involvement of E2F transcription factor family in cancer. Eur. J. Cancer 2005, 41, 2403–2414. [Google Scholar] [CrossRef] [PubMed]
- Kayed, H.; Jiang, X.; Keleg, S.; Jesnowski, R.; Giese, T.; Berger, M.R.; Esposito, I.; Löhr, M.; Friess, H.; Kleeff, J. Regulation and functional role of the Runt-related transcription factor-2 in pancreatic cancer. Br. J. Cancer 2007, 97, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Onodera, Y.; Miki, Y.; Suzuki, T.; Takagi, K.; Akahira, J.; Sakyu, T.; Watanabe, M.; Inoue, S.; Ishida, T.; Ohuchi, N.; et al. Runx2 in human breast carcinoma: Its potential roles in cancer progression. Cancer Sci. 2010, 101, 2670–2675. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.A.; Pollett, A.; Gallinger, S.; Dick, J.E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 2007, 445, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Ricci-Vitiani, L.; Lombardi, D.G.; Pilozzi, E.; Biffoni, M.; Todaro, M.; Peschle, C.; De Maria, R. Identification and expansion of human colon-cancer-initiating cells. Nature 2007, 445, 111–115. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukushima, N.; Sugiyama, A.; Sato, T. Screening Method for Anti-Colon Cancer Drugs Using Two Sensor Cell Lines with Human β4-Galactosyltransferase 4 Gene Promoters. Sensors 2018, 18, 2573. https://doi.org/10.3390/s18082573
Fukushima N, Sugiyama A, Sato T. Screening Method for Anti-Colon Cancer Drugs Using Two Sensor Cell Lines with Human β4-Galactosyltransferase 4 Gene Promoters. Sensors. 2018; 18(8):2573. https://doi.org/10.3390/s18082573
Chicago/Turabian StyleFukushima, Naomichi, Atena Sugiyama, and Takeshi Sato. 2018. "Screening Method for Anti-Colon Cancer Drugs Using Two Sensor Cell Lines with Human β4-Galactosyltransferase 4 Gene Promoters" Sensors 18, no. 8: 2573. https://doi.org/10.3390/s18082573
APA StyleFukushima, N., Sugiyama, A., & Sato, T. (2018). Screening Method for Anti-Colon Cancer Drugs Using Two Sensor Cell Lines with Human β4-Galactosyltransferase 4 Gene Promoters. Sensors, 18(8), 2573. https://doi.org/10.3390/s18082573



