All-Fiber CO2 Sensor Using Hollow Core PCF Operating in the 2 µm Region
Abstract
1. Introduction
2. Sensor Design and FBG Tuned Laser
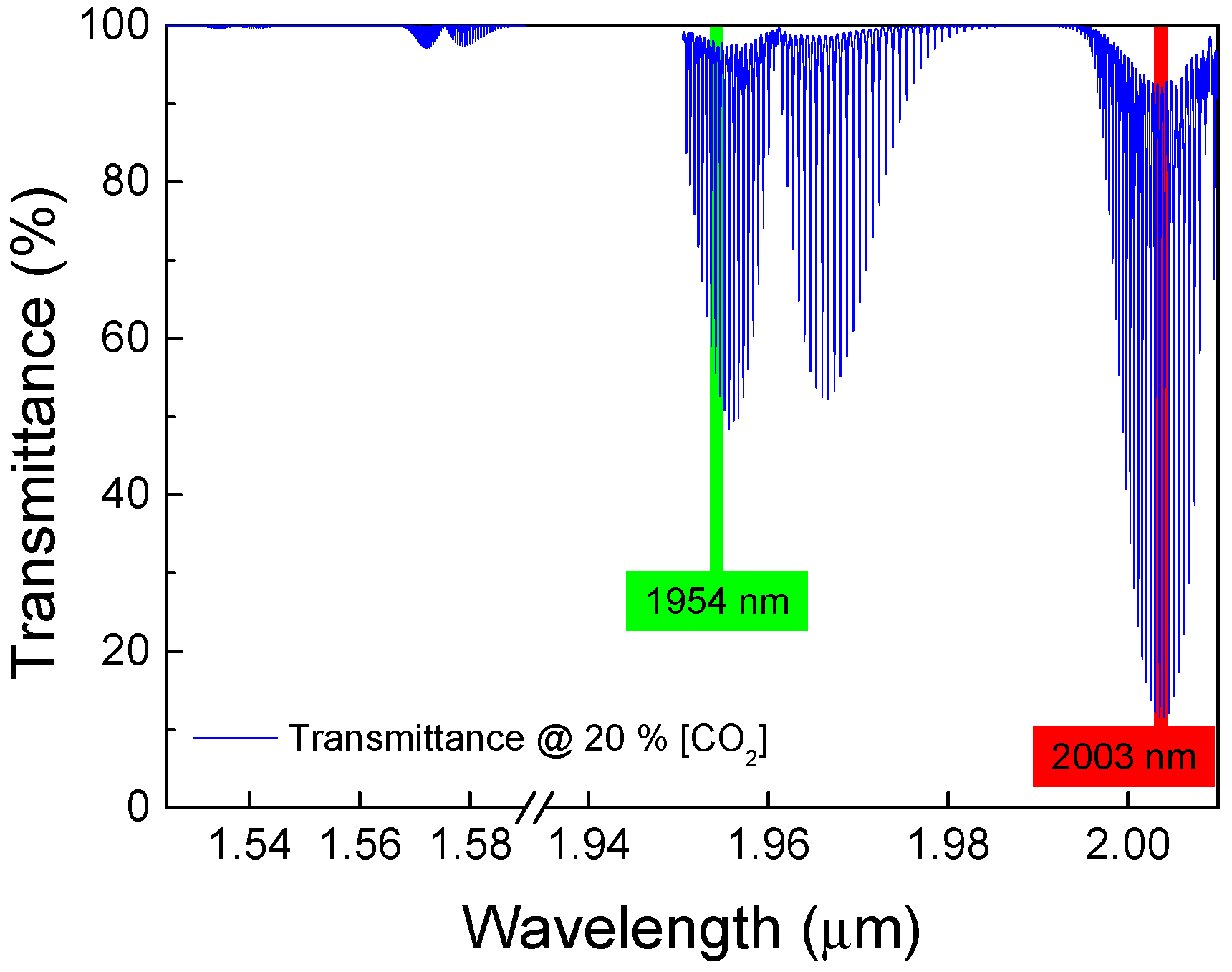
2.1. Working Principle
2.2. Sensor Design
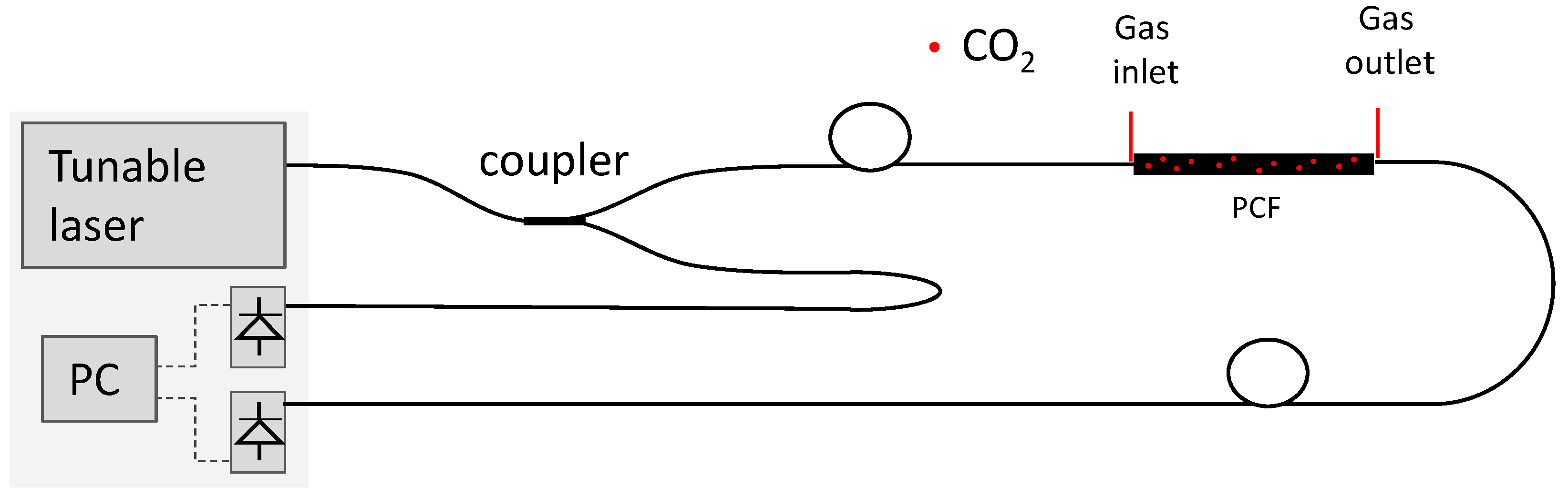
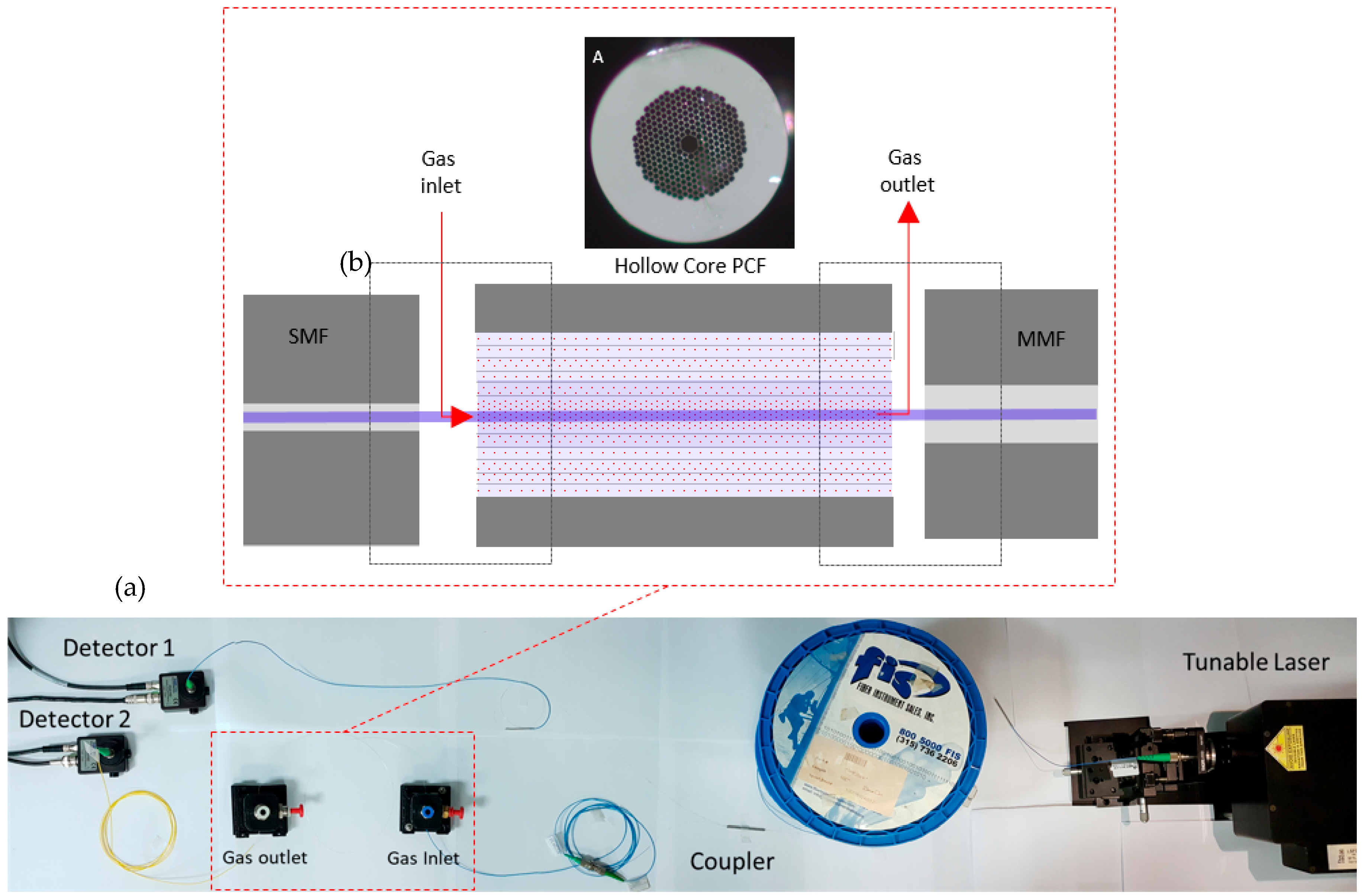
2.3. Implementation
- Optical cavity: The optical cavity is composed of a 74 cm long HC-PCF (NKT HC 2000);
- tunable laser: The laser used to obtain the results presented in the next section was a single-frequency tunable Cr2+:ZnS/Se laser from IPG Photonics Corporation, with an output power up to 2 W. Its spectral linewidth is 0.7 MHz, which is equivalent to 0.01 pm at 2 micron and the tuning range is adjustable from 1933 nm to 2245 nm. The scanning speed can be selected between 0.04 nm/s and 35.70 nm/s, with discrete steps of 0.14 pm;
- fibers and coupler: Light is launched in the HC-PCF from a single mode fiber (SMF 28) and collected with a multi-mode fiber. The fiber coupler is designed to operate as 50/50 at 2 µm; and
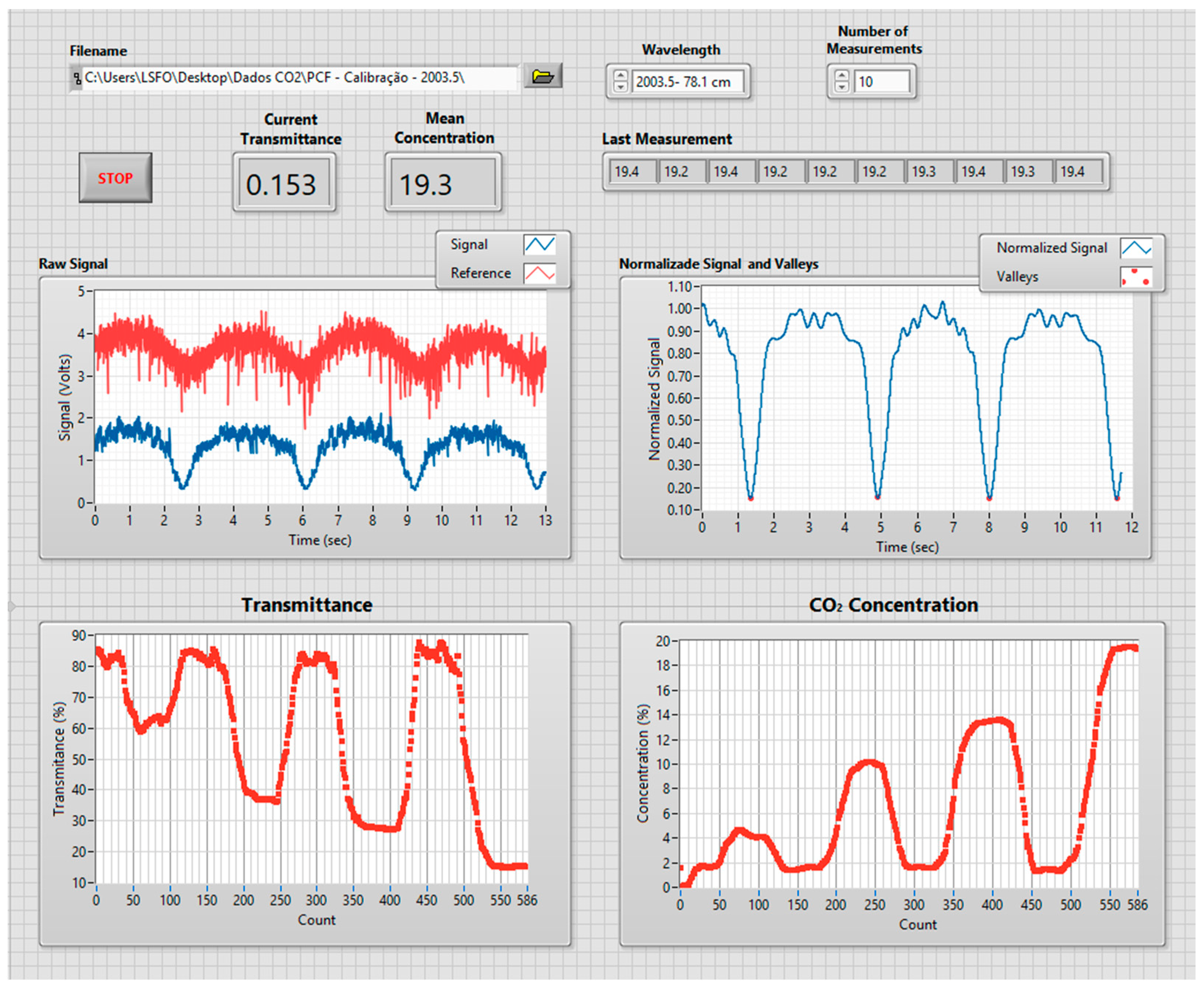
- processing unit: The acquisition and processing unit uses a National Instruments NI 6009 DAC board (National Instruments, Austin, TX, USA) [17], connected to a PC running a program written in Labview. The program is responsible for all the digital control and processing, going from the DAC board settings, all the way to the user interface. Figure 5 shows the end user interface where information on the CO2 concentration history and optical signal can be visualized.
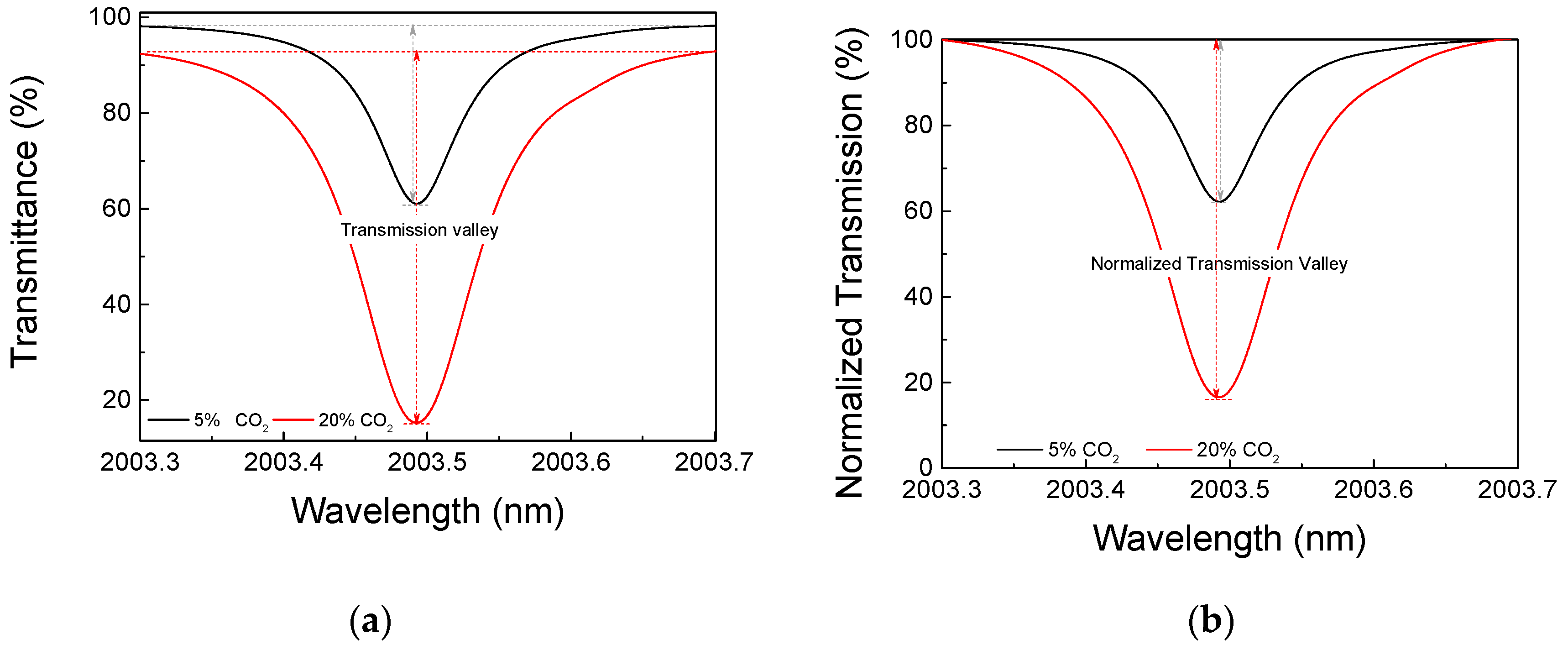
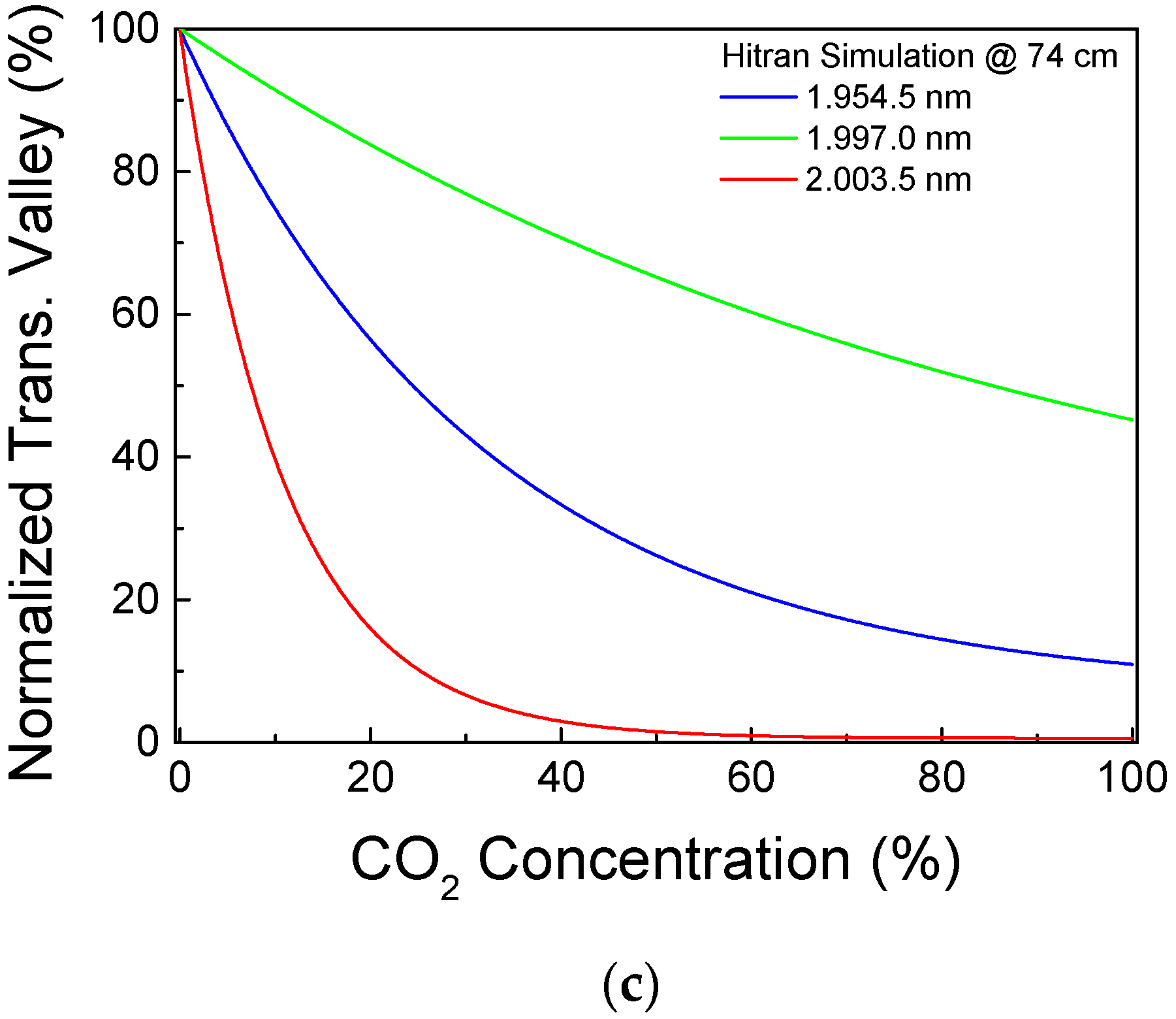
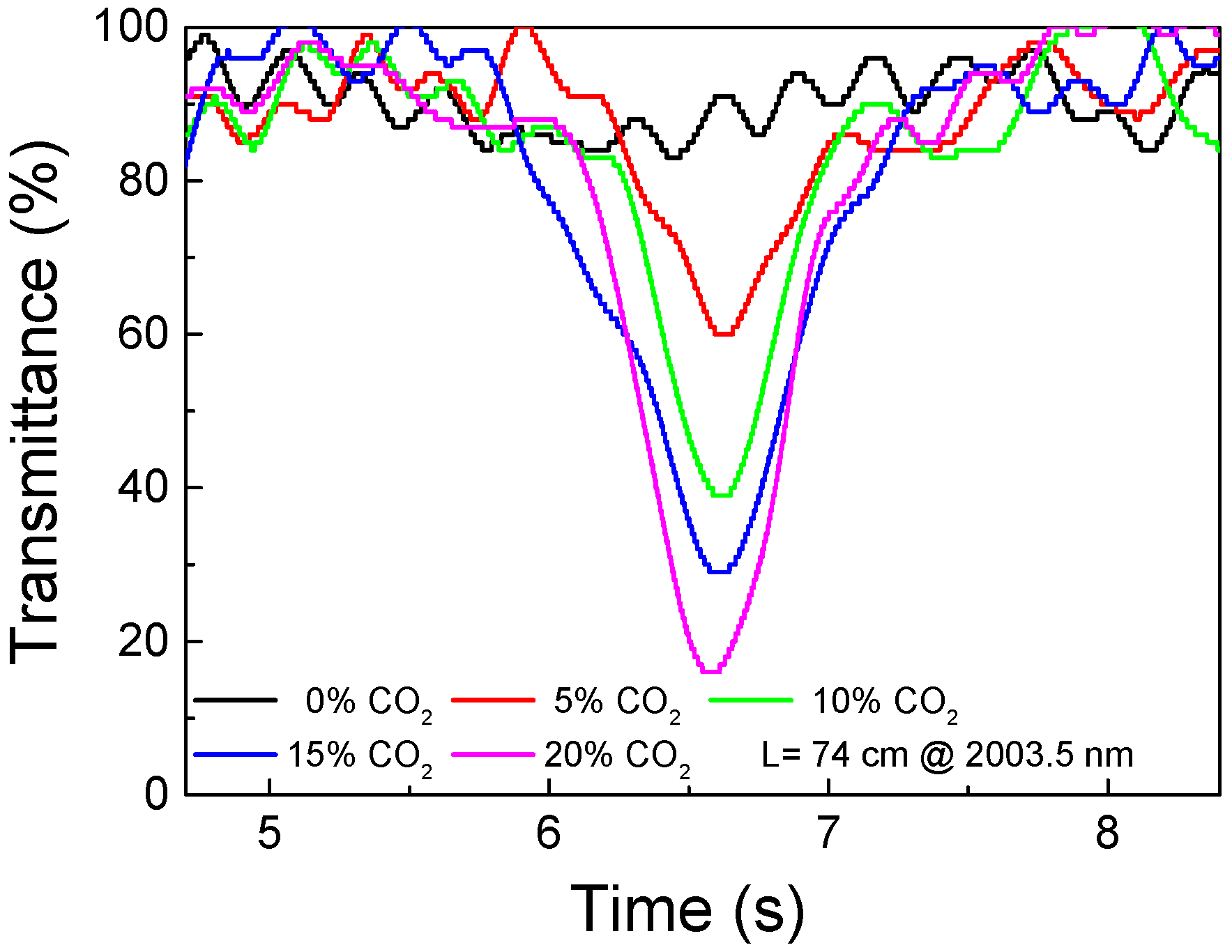
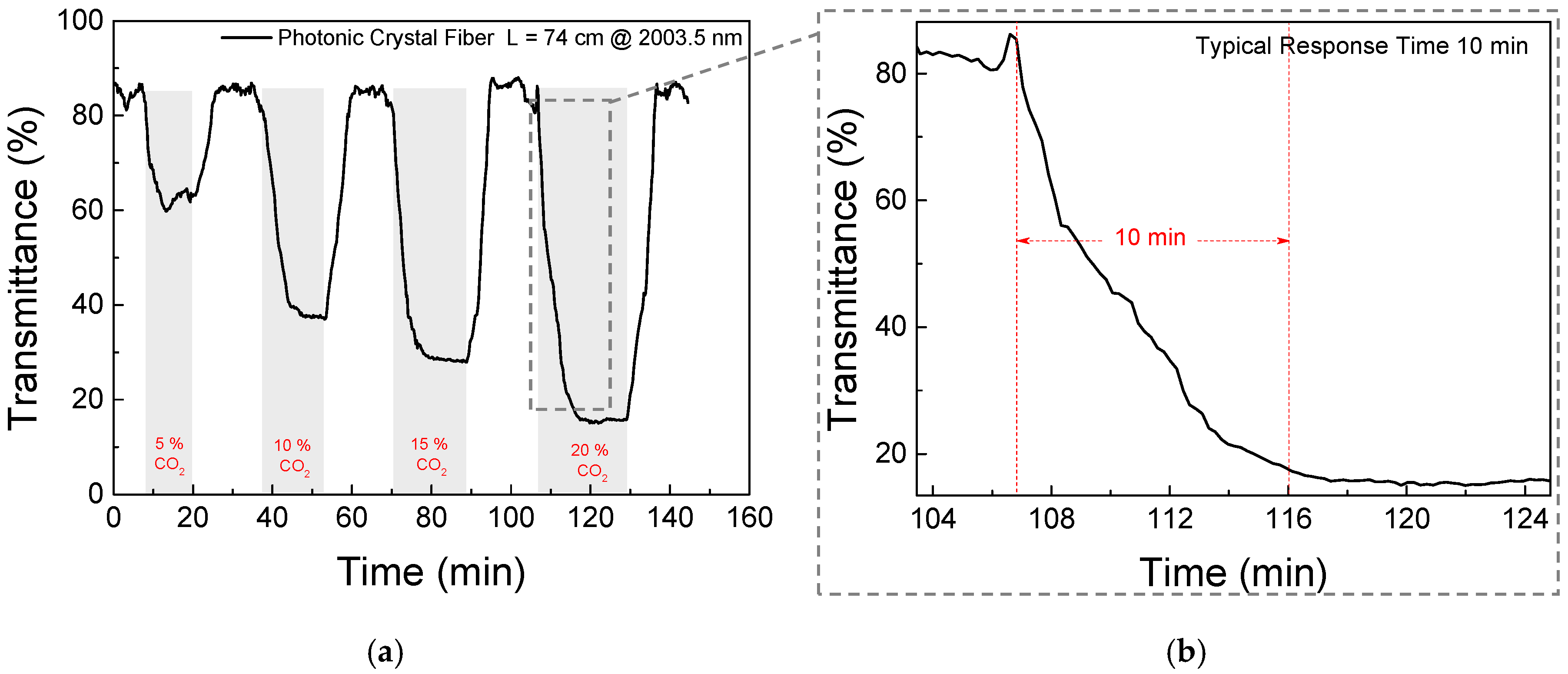
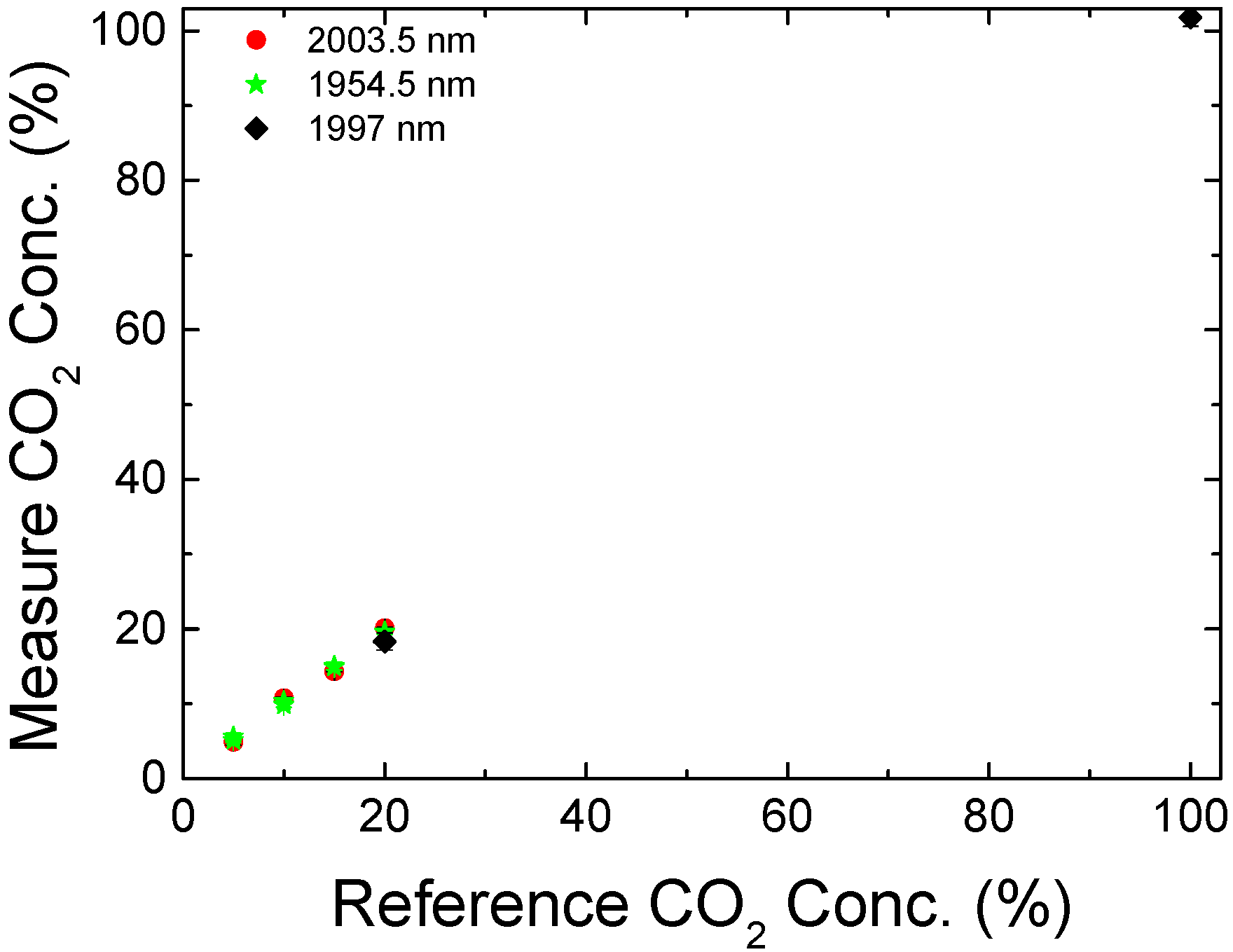
3. Results and Discussion
- Wavelength span: 0.6 nm (2003.1 nm to 2003.7 nm);
- sweep speed: 0.2 nm/s; and
- measurement update interval: 12 s.
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Qiao, X.; Shao, Z.; Bao, W.; Rong, Q. Fiber Bragg Grating Sensors for the Oil Industry. Sensors 2017, 17, 429. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, R. Fiber Bragg Gratings; Academic Press: Cambridge, MA, USA, 1999. [Google Scholar]
- Light, P.S.; Couny, F.; Benabid, F. Low optical insertion-loss and vacuum-pressure all-fiber acetylene cell based on hollow-core photonic crystal fiber. Opt. Lett. 2006, 31, 2538–2540. [Google Scholar] [CrossRef] [PubMed]
- Cubillas, A.M.; Lazaro, J.M.; Silva-Lopez, M.; Conde, O.M.; Petrovich, M.N.; Lopez-Higuera, J.M. Methane sensing at 1300 nm band with hollow-core photonic bandgap fibre as gas cell. Electron. Lett. 2008, 44, 403–404. [Google Scholar] [CrossRef]
- Ritari, T. Gas sensing using air-guiding photonic bandgap fibers. Opt. Express 2004, 12, 4080–4087. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Chang, A.S.P.; Chen, B.; Gu, C.; Bond, T.C. High sensitivity gas sensing by Raman spectroscopy in photonic crystal fiber. Sens. Actuators B Chem. 2013, 176, 64–68. [Google Scholar] [CrossRef]
- Ebnali-Heidari, M.; Koohi-Kamali, F.; Ebnali-Heidari, A.; Moravvej-Farshi, M.K.; Kuhlmey, B.T. Designing Tunable Microstructure Spectroscopic Gas Sensor Using Optofluidic Hollow-Core Photonic Crystal Fiber. IEEE J. Quantum Electron. 2014, 50, 1–8. [Google Scholar] [CrossRef]
- Villatoro, J. Photonic crystal fiber interferometer for chemical vapor detection with high sensitivity. Opt. Express 2009, 17, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Schilt, S.; Matthey, R.; Tow, K.H.; Thévenaz, L.; Südmeyer, T. All-fiber versatile laser frequency reference at 2 μm for CO2 space-borne lidar applications. CEAS J. 2017, 9, 493–505. [Google Scholar] [CrossRef]
- Absorption. In IUPAC Compendium of Chemical Terminology; IUPAC: Research Triagle Park, NC, USA, 1997.
- Beer–Lambert law (or Beer–Lambert–Bouguer law). In IUPAC Compendium of Chemical Terminology; IUPAC: Research Triagle Park, NC, USA, 1996.
- Izawa, T.; Shibata, N.; Takeda, A. Optical attenuation in pure and doped fused silica in the IR wavelength region. Total Opt. Attenuat. Bulk Fused Silic. Appl. Phys. Lett. 1977, 31, 264. [Google Scholar] [CrossRef]
- Jeunhomme, L.B. Single-Mode Fiber Optics: Principles and Applications; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Rothman, L.S. The HITRAN2012 molecular spectroscopic database. J. Quant. Spectrosc. Radiat. Transf. 2013, 130, 4–50. [Google Scholar] [CrossRef]
- Wei, W.; Chang, J.; Huang, Q.; Wang, Q.; Liu, Y.; Qin, Z. Water vapor concentration measurements using TDALS with wavelength modulation spectroscopy at varying pressures. Sens. Rev. 2017, 37, 172–179. [Google Scholar] [CrossRef]
- Hoo, Y.L.; Liu, S.; Ho, H.L.; Jin, W. Fast Response Microstructured Optical Fiber Methane Sensor with Multiple Side-Openings. IEEE Photonics Technol. Lett. 2010, 22, 296–298. [Google Scholar] [CrossRef]
- SPECIFICATIONS USB-6009. Available online: http://www.ni.com/pdf/manuals/375296c.pdf (accessed on 28 October 2018).
- Valiunas, J.K.; Tenuta, M.; Das, G. A Gas Cell Based on Hollow-Core Photonic Crystal Fiber (PCF) and Its Application for the Detection of Greenhouse Gas (GHG): Nitrous Oxide (N2O). J. Sens. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mejia Quintero, S.M.; Guedes Valente, L.C.; De Paula Gomes, M.S.; Gomes da Silva, H.; Caroli de Souza, B.; Morikawa, S.R.K. All-Fiber CO2 Sensor Using Hollow Core PCF Operating in the 2 µm Region. Sensors 2018, 18, 4393. https://doi.org/10.3390/s18124393
Mejia Quintero SM, Guedes Valente LC, De Paula Gomes MS, Gomes da Silva H, Caroli de Souza B, Morikawa SRK. All-Fiber CO2 Sensor Using Hollow Core PCF Operating in the 2 µm Region. Sensors. 2018; 18(12):4393. https://doi.org/10.3390/s18124393
Chicago/Turabian StyleMejia Quintero, Sully Milena, Luiz Carlos Guedes Valente, Marcos Sebastião De Paula Gomes, Hugo Gomes da Silva, Bernardo Caroli de Souza, and Sergio R. K. Morikawa. 2018. "All-Fiber CO2 Sensor Using Hollow Core PCF Operating in the 2 µm Region" Sensors 18, no. 12: 4393. https://doi.org/10.3390/s18124393
APA StyleMejia Quintero, S. M., Guedes Valente, L. C., De Paula Gomes, M. S., Gomes da Silva, H., Caroli de Souza, B., & Morikawa, S. R. K. (2018). All-Fiber CO2 Sensor Using Hollow Core PCF Operating in the 2 µm Region. Sensors, 18(12), 4393. https://doi.org/10.3390/s18124393



