Sensor-Based Detection of the Severity of Hyperkeratosis in the Teats of Dairy Cows
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Study Design
2.2. Data Collection
2.2.1. Classification of Hyperkeratosis
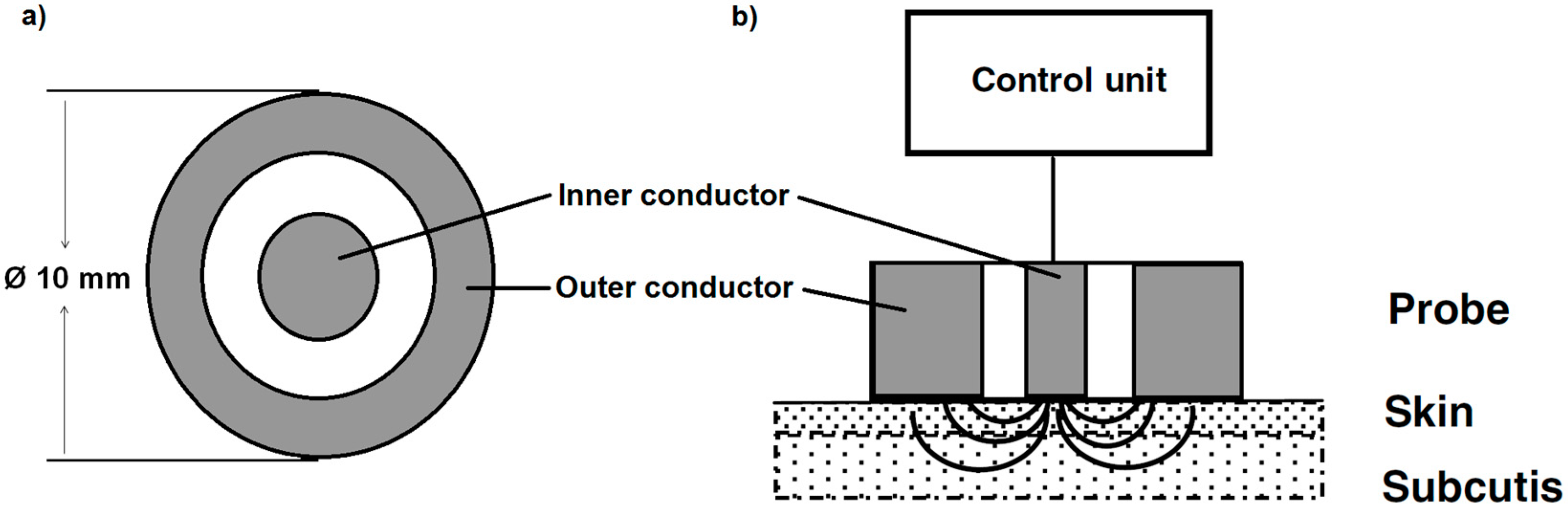
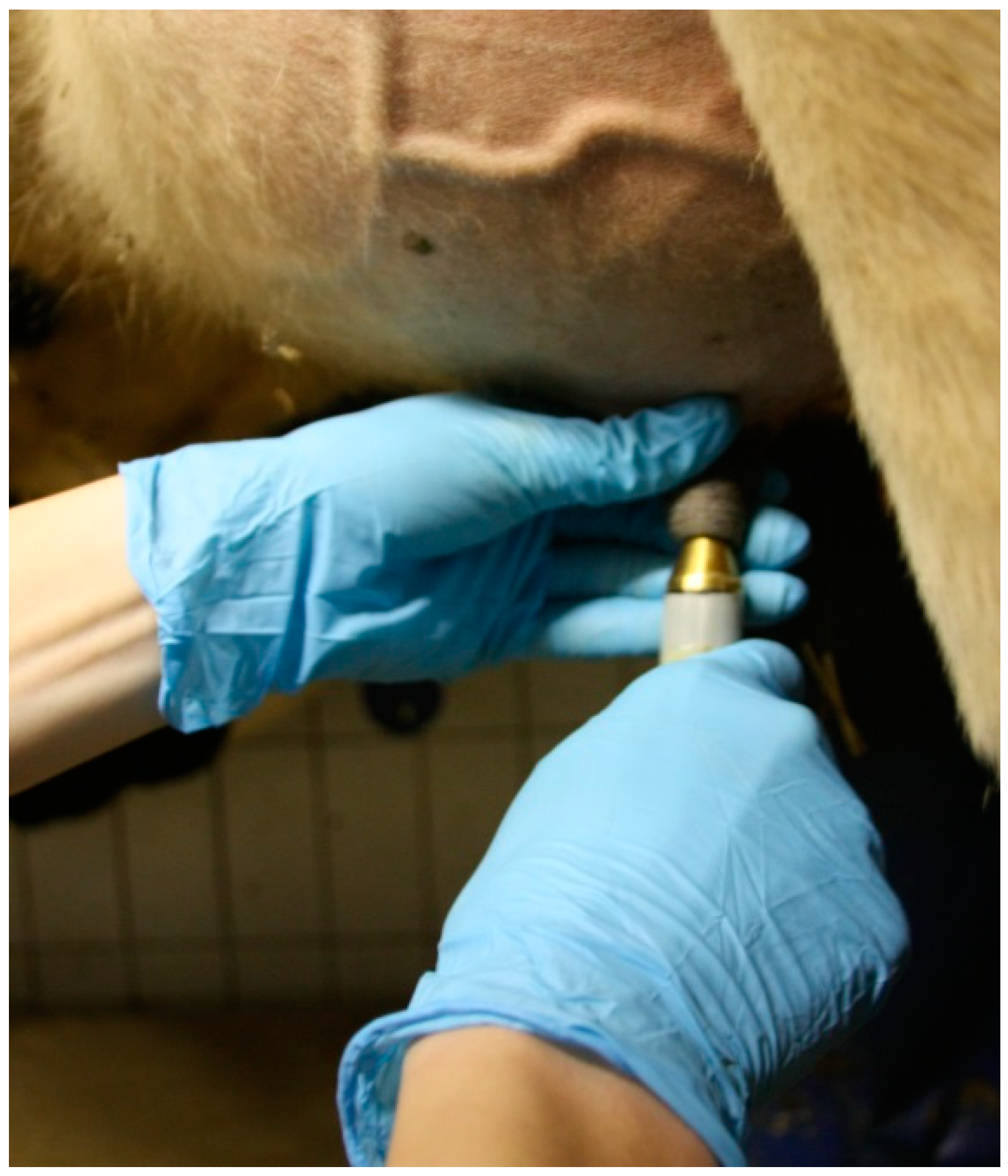
2.2.2. Dielectric Measurement
2.3. Statistical Analysis
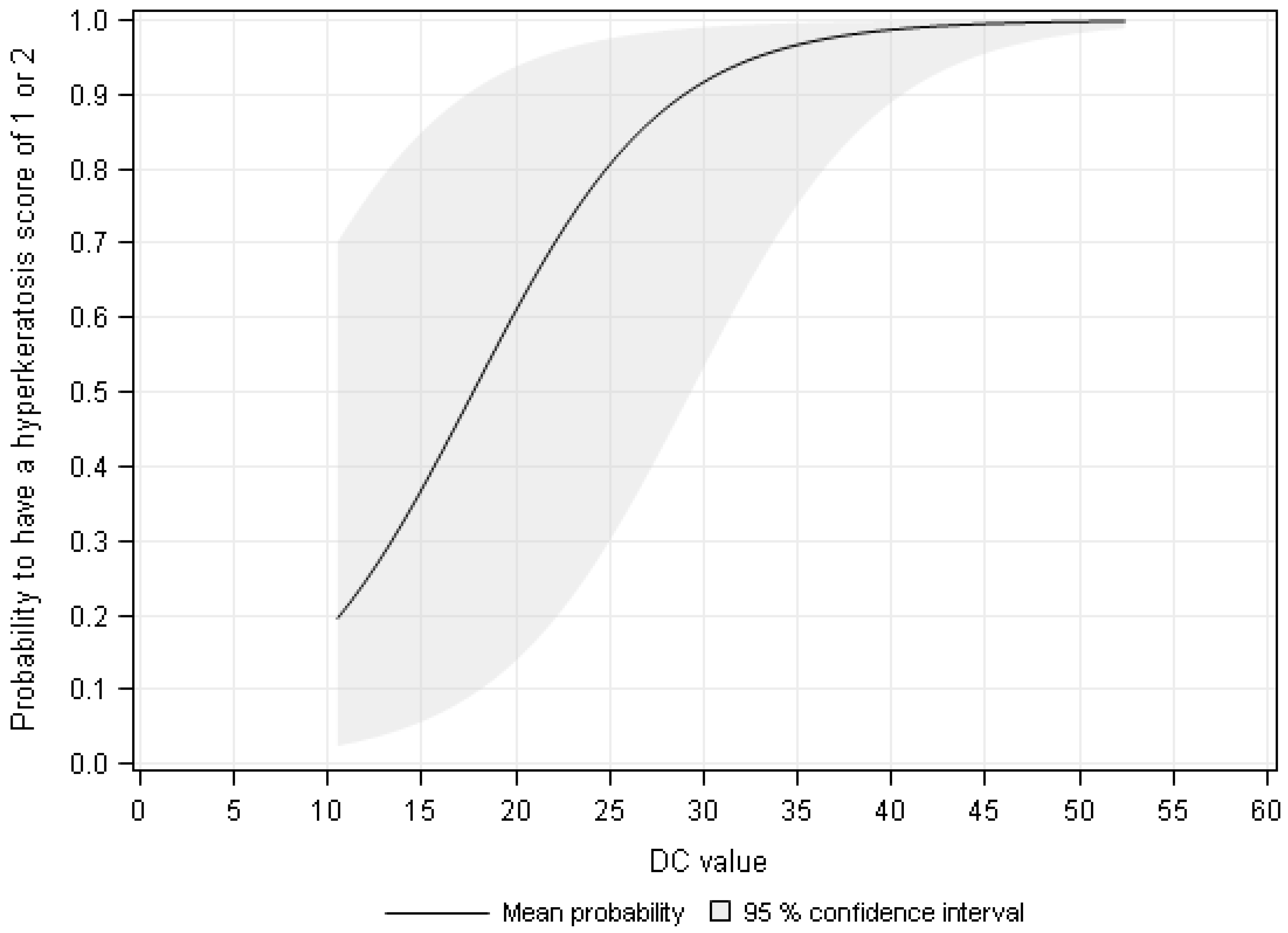
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gleeson, D.E.; O’Callaghan, E.J.; Rath, M. The effects of genotype, milking time and teat-end vacuum pattern on the severity of teat-end hyperkeratosis. Irish J. Agric. Food Res. 2003, 42, 195–203. [Google Scholar]
- Sordillo, L.M.; Streicher, K.L. Mammary gland immunity and mastitis susceptibility. J. Mammary Gland Biol. 2002, 7, 135–146. [Google Scholar] [CrossRef]
- Neijenhuis, F.; Barkema, H.W.; Hogeveen, H.; Noordhuizen, J.P.T.M. Classification and longitudinal examination of callused teat ends in dairy cows. J. Dairy Sci. 2000, 83, 2795–2804. [Google Scholar] [CrossRef]
- Neijenhuis, F.; Mein, G.A.; Reinemann, D.J. Evaluation of bovine teat condition in commercial dairy herds: 4. Relationship between teat-end callosity or hyperkeratosis and mastitis. In Proceedings of the AABP-NMC International Symposium on Mastitis and Milk Quality, Vancouver, BC, Canada, 13–15 September 2001; pp. 362–366. [Google Scholar]
- Singh, V.; Singh, V.K.; Doley, P.; Singh, A. Association between teat condition score and udder health status in dairy bovines. J. Agric. Vet. Sci. 2014, 7, 9–11. [Google Scholar] [CrossRef]
- Gleeson, D.E.; Meaney, W.J.; O’Callaghan, E.J.; Rath, M.V. Effect of teat hyperkeratosis on somatic cell counts of dairy cows. Int. J. Appl. Res. Vet. Med. 2004, 2, 115–122. [Google Scholar]
- Rudovsky, H.J.; Pache, S.; Schulz, J. Hyperkeratoses of the teat: What are the causes? ART-Schriftenreihe 15 2011, 15, 75–78. [Google Scholar]
- Sandrucci, A.; Bava, L.; Zucali, M.; Tamburini, A. Management factors and cow traits influencing milk somatic cell counts and teat hyperkeratosis during different seasons. Revista Brasileira de Zootecnia 2014, 43, 505–511. [Google Scholar] [CrossRef]
- Graff, K. Investigation of connections between morphological characteristics of the udder, the health of the udder and milk-technical parameters in animals of the German Holstein race. Züchtungskunde 2006, 78, 245–247. [Google Scholar]
- Ryšánek, D.; Olejník, P.; Babák, V. Vacuum Fluctuation in Short Milk Tube during Peak Milk Flow; International Committee for Animal Recording (ICAR): Roma, Italy, 2001; pp. 125–130. [Google Scholar]
- Neijenhuis, F.; Klungel, G.H.; Hogeveen, H.; Noordhuizen, J.P.T.M. Machine Milking RISK factors for Teat End Callosity in Dairy Cows on Herd Level; Wageningen Academic Publishers: Wageningen, The Netherlands, 2005; pp. 376–382. [Google Scholar]
- Haeussermann, A.; Britten, J.; Britten, A.; Pahl, C.; Älveby, N.; Hartung, E. Effect of a multi-sided concave liner barrel design on thickness and roughness of teat-end hyperkeratosis. J. Dairy Res. 2016, 83, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Mein, G.A.; Neijenhuis, F.; Morgan, W.F. Evaluation of bovine teat condition in commercial dairy herds: 1. Non-infectious factors. In Proceedings of the AABP-NMC International Symposium on Mastitis and Milk Quality, Vancouver, BC, Canada, 13–15 September 2001; pp. 347–351. [Google Scholar]
- Nuutinen, J.; Ikäheimo, R.; Lahtinen, T. Validation of a new dielectric device to assess changes of tissue water in skin and subcutaneous fat. Physiol. Meas. 2004, 25, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.; Mönkkönen, J.; Lahtinen, M.R.; Nuutinen, J.; Lahtinen, T. Measurement of oedema in irritant-exposed skin by a dielectric technique. Skin Res. Technol. 2006, 12, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Mayrovitz, H.N.; Grammenos, A.; Corbitt, K.; Bartos, S. Young adult gender differences in forearm skin-to-fat tissue dielectric constant values measured at 300 mhz. Skin Res. Technol. 2016, 22, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Birkballe, S.; Jensen, M.R.; Noerregaard, S.; Gottrup, F.; Karlsmark, T. Can tissue dielectric constant measurement aid in differentiating lymphoedema from lipoedema in women with swollen legs? Br. J. Dermatol. 2014, 170, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.S.; Yeshanehe, W.E.; Matts, P.J.; Davey, G.; Mortimer, P.S.; Fuller, L.C. Assessment of skin barrier function in podoconiosis: Measurement of stratum corneum hydration and transepidermal water loss. Br. J. Dermatol. 2013, 168, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Greenhowe, J.; Stephen, C.; McClymont, L.; Munnoch, D.A. Breast oedema following free flap breast reconstruction. Breast 2017, 34, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Palma, M.L.; Tavares, L.; Fluhr, J.W.; Bujan, M.J.; Rodrigues, L.M. Positive impact of dietary water on in vivo epidermal water physiology. Skin Res. Technol. 2015, 21, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Mayrovitz, H.N.; Grammenos, A.; Corbitt, K.; Bartos, S. Age-related changes in male forearm skin-to-fat tissue dielectric constant at 300 mhz. Clin. Physiol. Funct. Imag. 2017, 37, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Nuutinen, J.; Lahtinen, T.; Turunen, M.; Alanen, E.; Tenhunen, M.; Usenius, T.; Kolle, R. A dielectric method for measuring early and late reactions in irradiated human skin. Radiother. Oncol. 1998, 47, 249–254. [Google Scholar] [CrossRef]
- Papp, A.; Lahtinen, T.; Härmä, M.; Nuutinen, J.; Uusaro, A.; Alhava, E. Dielectric measurement in experimental burns: A new tool for burn depth determination? Plast. Reconstr. Surg. 2006, 117, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Guihan, M.; Bates-Jenson, B.M.; Chun, S.; Parachuri, R.; Chin, A.S.; McCreath, H. Assessing the feasibility of subepidermal moisture to predict erythema and stage 1 pressure ulcers in persons with spinal cord injury: A pilot study. J. Spinal. Cord. Med. 2012, 35, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Forcheron, F.; Agay, D.; Scherthan, H.; Riccobono, D.; Herodin, F.; Meineke, V.; Drouet, M. Autologous adipocyte derived stem cells favour healing in a minipig model of cutaneous radiation syndrome. PLoS ONE 2012, 7, e31694. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, D.E.; Nuutinen, J.; Lahtinen, T.; Rissanen, A.; Niskanen, L.K. Changes in abdominal subcutaneous fat water content with rapid weight loss and long-term weight maintenance in abdominally obese men and women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, G.; Ammon, C.; Volkamer, L.; Sürie, C.; Radko, D. Sensor-based monitoring of the prevalence and severity of foot pad dermatitis in broiler chickens. Br. Poult. Sci. 2013, 54, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Aimoto, A.; Matsumoto, T. Noninvasive method for measuring the electrical properties of deep tissues using an open-ended coaxial probe. Med. Eng. Phys. 1996, 18, 641–646. [Google Scholar] [CrossRef]
- Mayrovitz, H.N.; Luis, M. Spatial variations in forearm skin tissue dielectric constant. Skin Res. Technol. 2010, 16, 438–443. [Google Scholar] [CrossRef] [PubMed]
- McCullagh, P.; Nelder, J.A. Generalized Linear Models; CRC Press: Boca Raton, FL, USA, 1989; pp. 164–170. [Google Scholar]
- Mayne, R.K.; Else, R.W.; Hocking, P.M. High dietary concentrations of biotin did not prevent foot pad dermatitis in growing turkeys and external scores were poor indicators of histopathological lesions. Br. Poult. Sci. 2007, 48, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Lahtinen, T.; Nuutinen, J.; Alanen, E. Dielectric properties of skin. In Radio Frequency Radiation Dosimetry; Klauenberg, B.J., Miklavcic, D., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2000; pp. 97–101. [Google Scholar]
- Hashmi, F.; Nester, C.; Wright, C.; Newton, V.; Lam, S. Characterising the biophysical properties of normal and hyperkeratotic foot skin. J. Foot Ankle Res. 2015, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Alanen, E.; Lahtinen, T.; Nuutinen, J. Penetration of electromagnetic fields of an open-ended coaxial probe between 1 Mhz and 1 Ghz in dielectric skin measurements. Phys. Med. Boil. 1999, 44, N169–N176. [Google Scholar] [CrossRef]
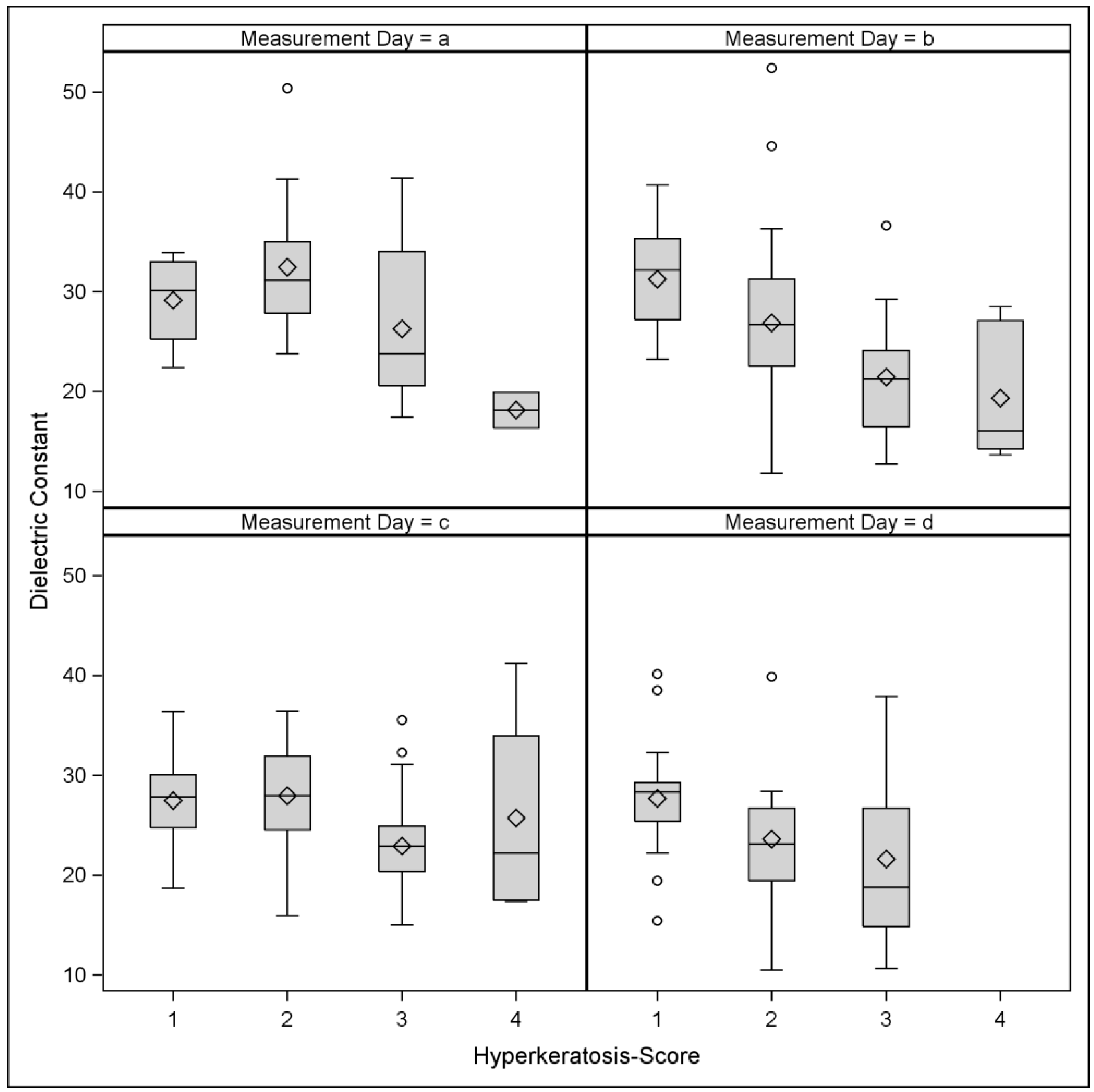
| HK Score | N | Mean | STD | Min | Q1 | Median | Q3 | Max |
|---|---|---|---|---|---|---|---|---|
| 1 | 52 | 28.98 | 5.47 | 15.40 | 25.60 | 28.80 | 32.90 | 40.70 |
| 2 | 110 | 27.91 | 6.95 | 10.50 | 23.75 | 27.85 | 31.90 | 52.43 |
| 3 | 66 | 23.09 | 6.43 | 10.65 | 19.20 | 22.30 | 26.15 | 41.40 |
| 4 | 13 | 21.00 | 7.75 | 13.65 | 16.10 | 17.65 | 25.43 | 41.25 |
| Model | Effect | Estimate | Standard Error | DF | t-Value | p-Value |
|---|---|---|---|---|---|---|
| a | Intercept | 7.64 | 1.97 | 97 | 3.88 | 0.0002 |
| DC 1 value | 0.01 | 0.07 | 138 | 0.17 | 0.8638 | |
| b | Intercept | −3.47 | 1.16 | 97 | −3.00 | 0.0035 |
| DC 1 value | 0.19 | 0.05 | 138 | 4.16 | <0.0001 | |
| c | Intercept | 5.74 | 2.92 | 97 | 1.97 | 0.0520 |
| DC 1 value | 0.17 | 0.11 | 138 | 1.53 | 0.1277 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demba, S.; Hoffmann, G.; Ammon, C.; Rose-Meierhöfer, S. Sensor-Based Detection of the Severity of Hyperkeratosis in the Teats of Dairy Cows. Sensors 2018, 18, 3925. https://doi.org/10.3390/s18113925
Demba S, Hoffmann G, Ammon C, Rose-Meierhöfer S. Sensor-Based Detection of the Severity of Hyperkeratosis in the Teats of Dairy Cows. Sensors. 2018; 18(11):3925. https://doi.org/10.3390/s18113925
Chicago/Turabian StyleDemba, Susanne, Gundula Hoffmann, Christian Ammon, and Sandra Rose-Meierhöfer. 2018. "Sensor-Based Detection of the Severity of Hyperkeratosis in the Teats of Dairy Cows" Sensors 18, no. 11: 3925. https://doi.org/10.3390/s18113925
APA StyleDemba, S., Hoffmann, G., Ammon, C., & Rose-Meierhöfer, S. (2018). Sensor-Based Detection of the Severity of Hyperkeratosis in the Teats of Dairy Cows. Sensors, 18(11), 3925. https://doi.org/10.3390/s18113925






