A Portable Laser Photoacoustic Methane Sensor Based on FPGA
Abstract
:1. Introduction
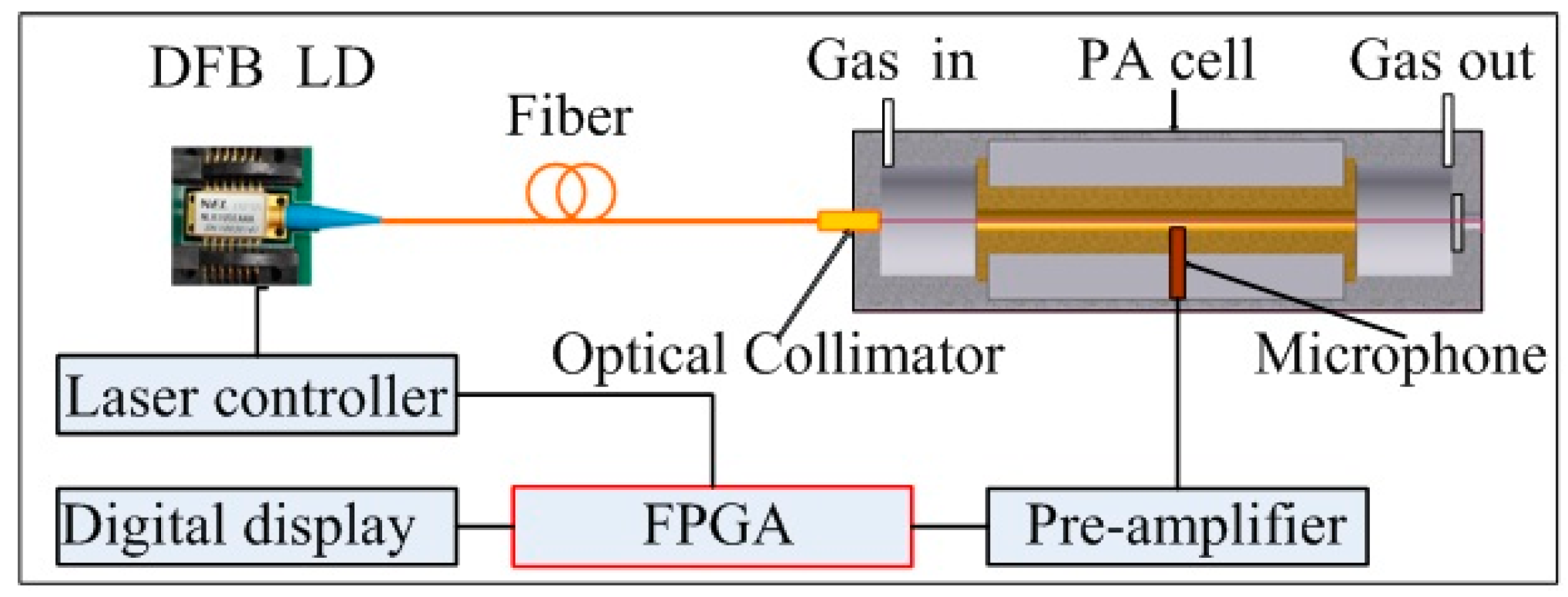
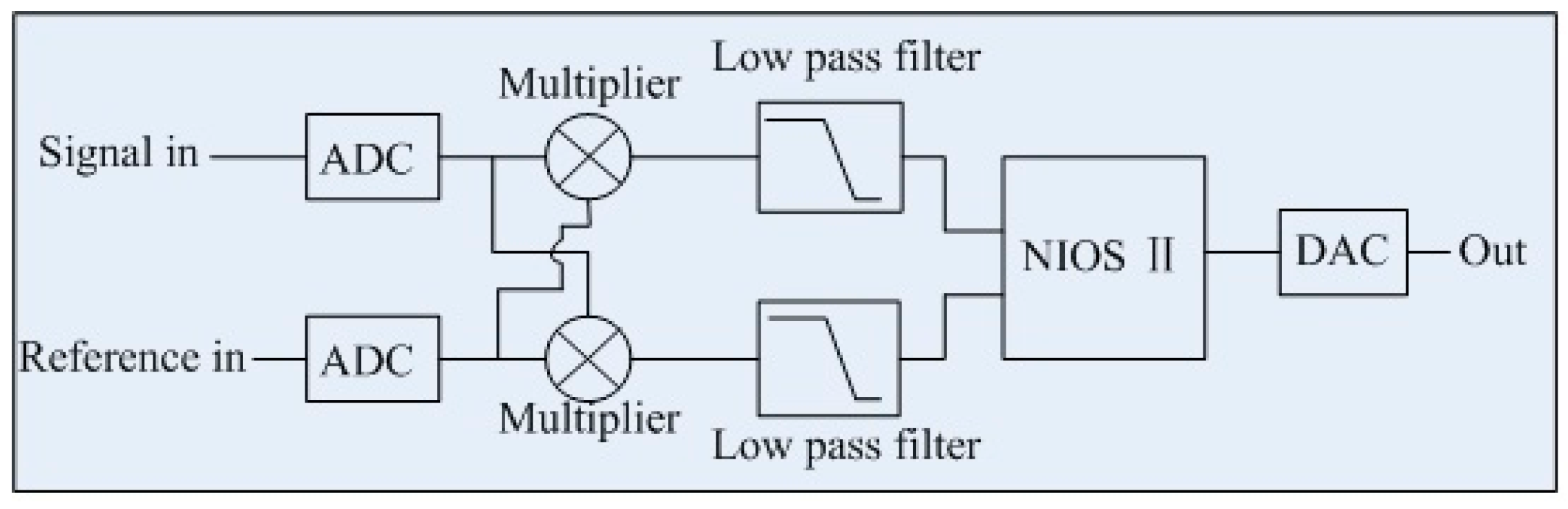
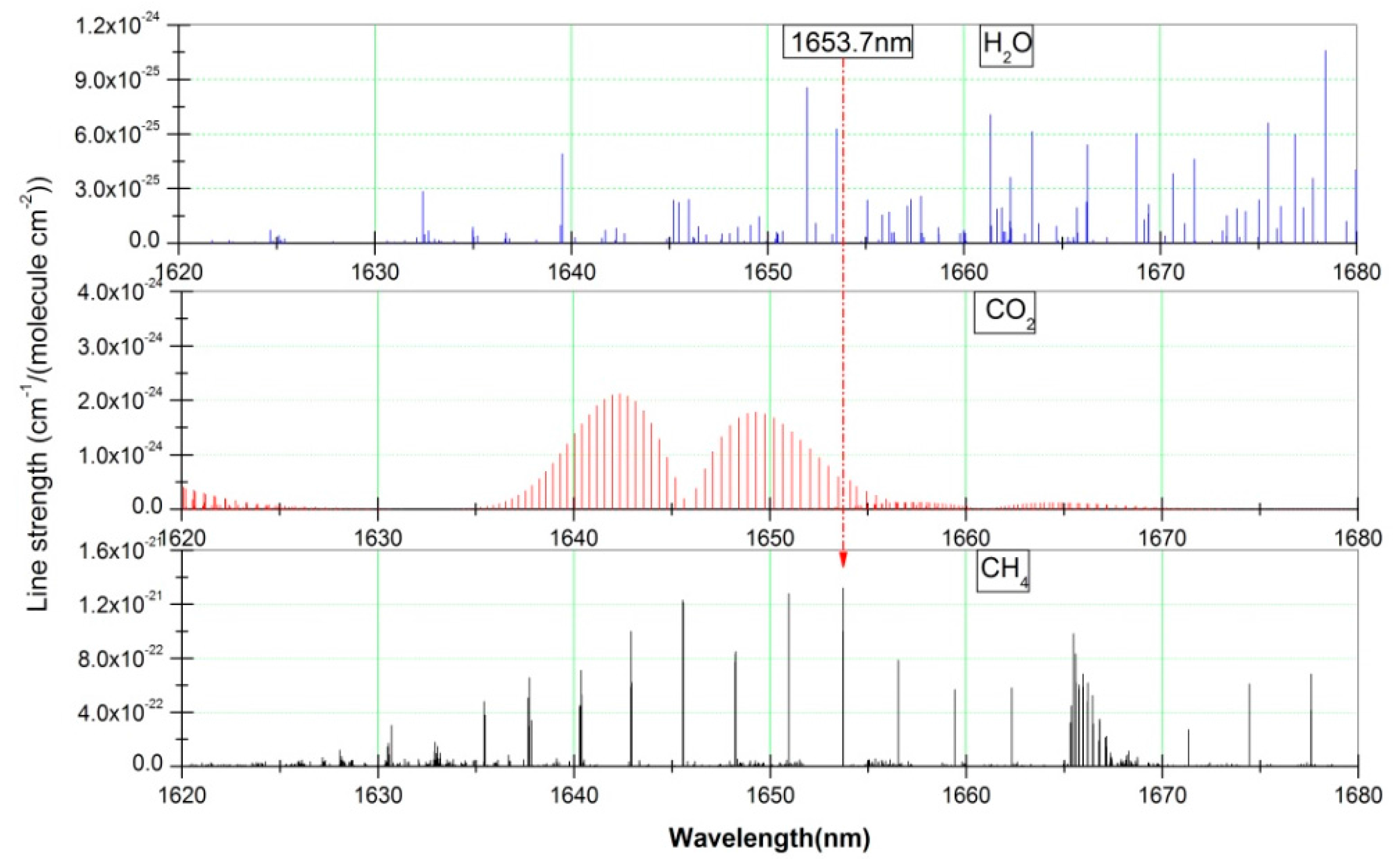
2. Sensor Design
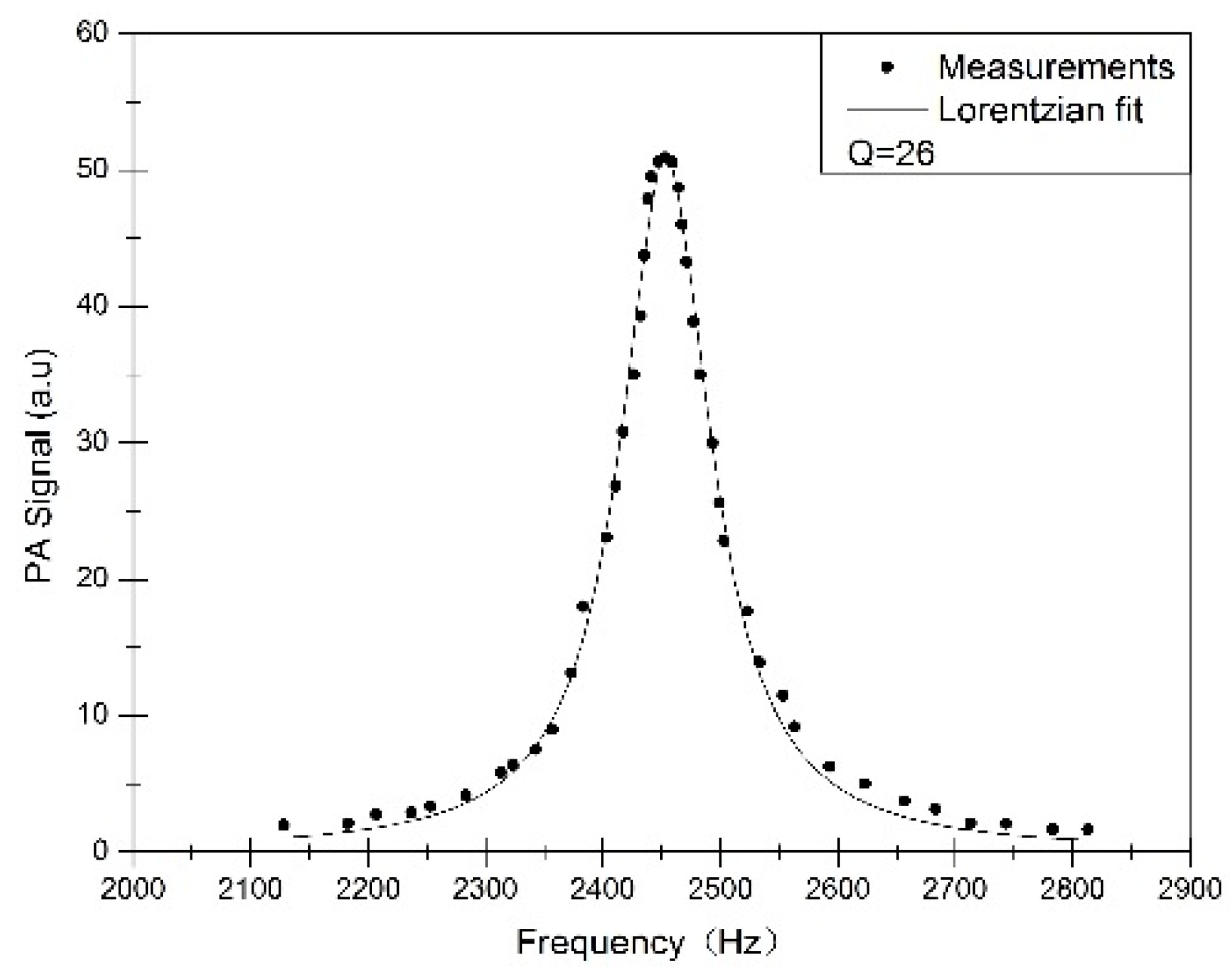
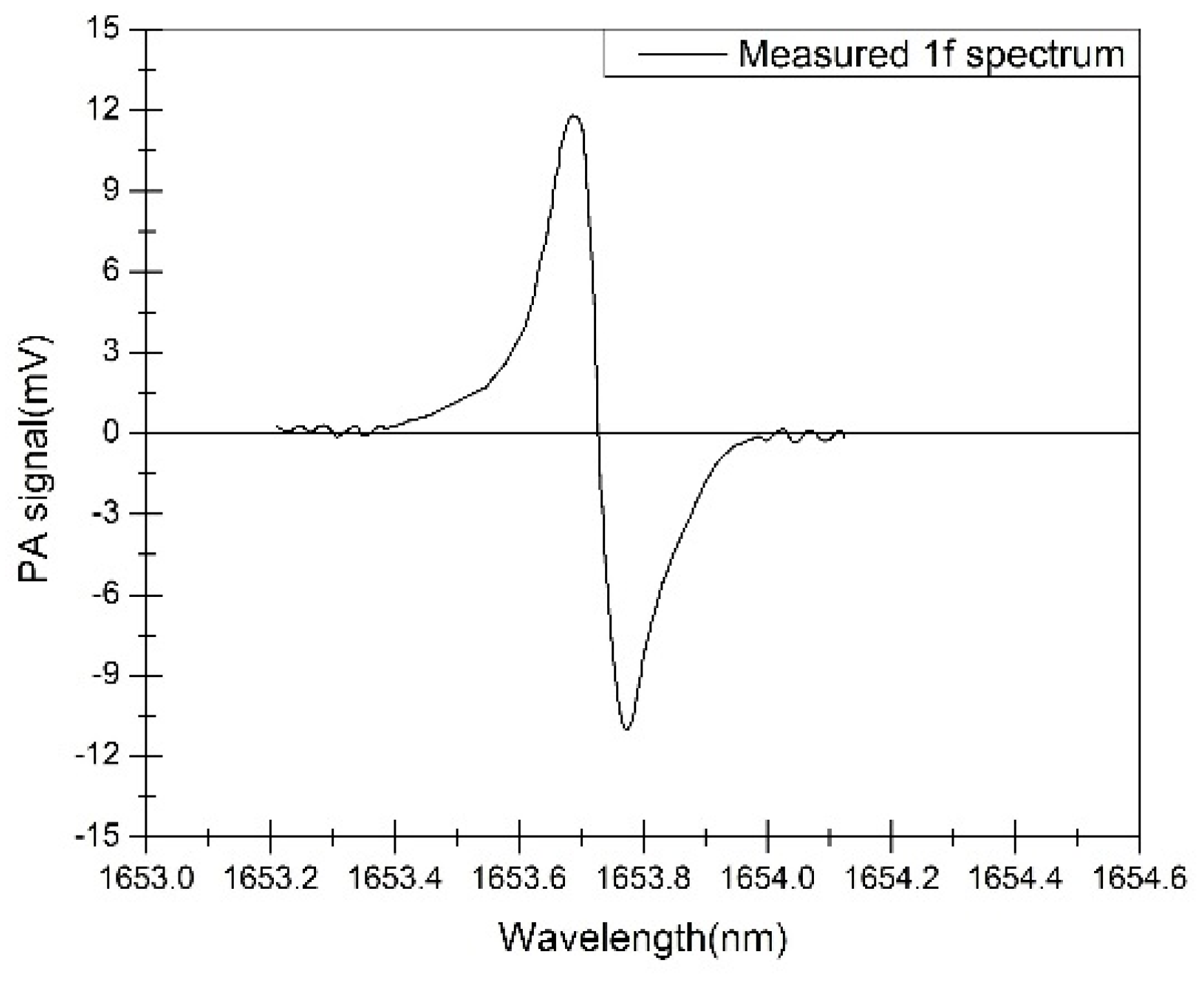
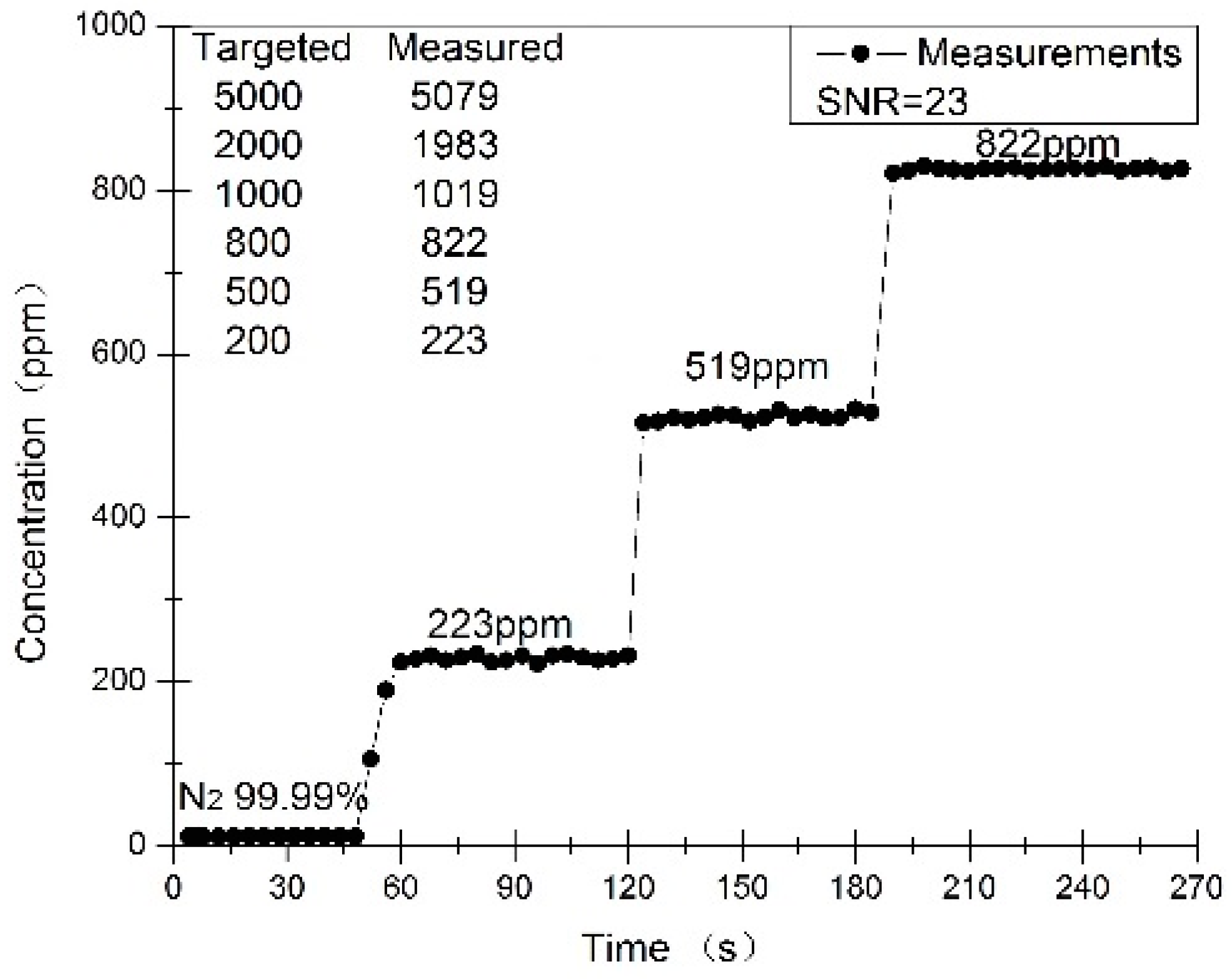
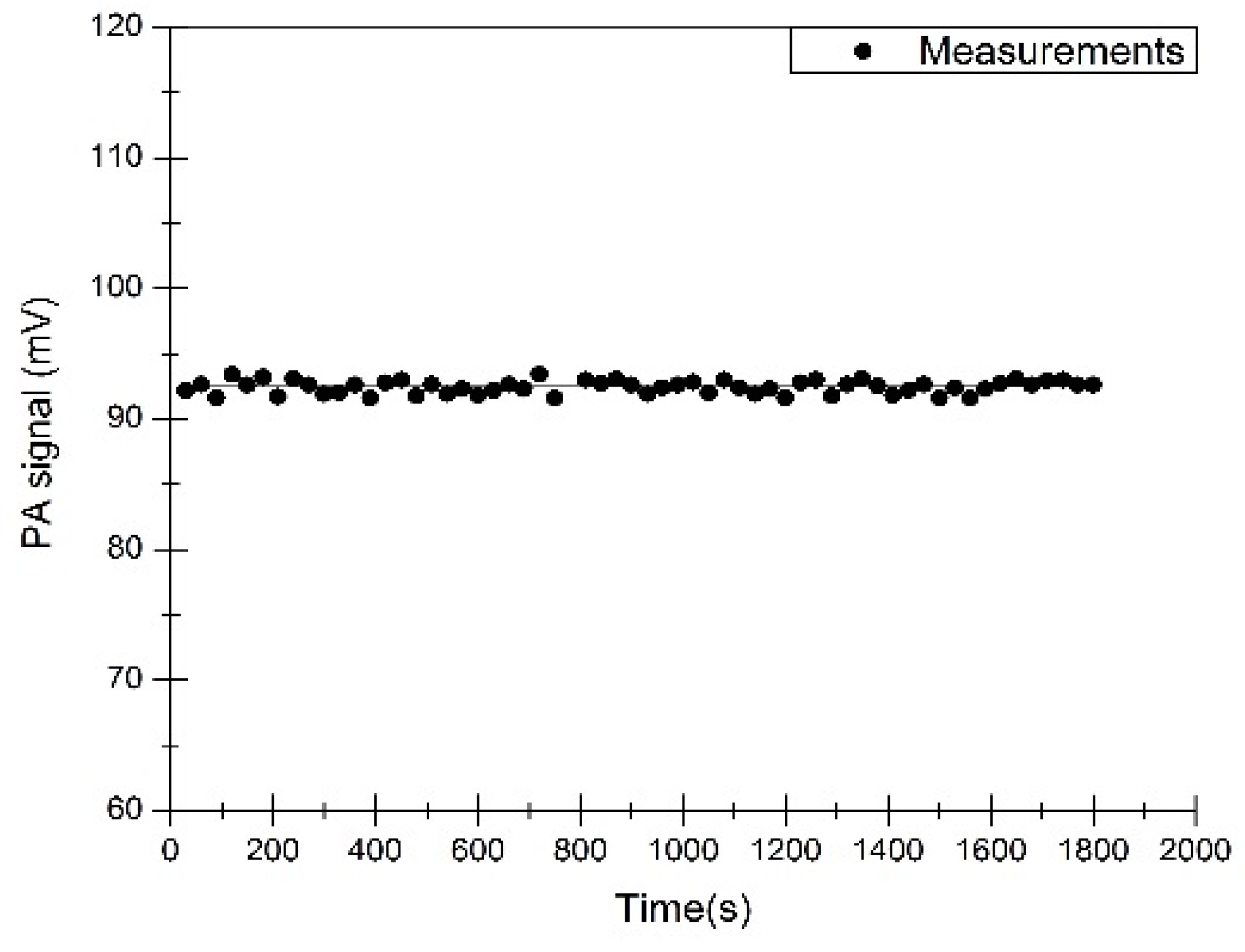
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, J.; Lu, X.T.; Yang, Z.H. Remote system of natural gas leakage based on multi-wavelength characteristics spectrum analysis. Spectrosc. Spect. Anal. 2014, 34, 1249–1252. [Google Scholar]
- Jahjah, M.; Ren, W.; Stefanski, P.; Lewicki, R.; Zhang, J.W.; Jiang, W.Z.; Tarka, J.; Tittel, F.K. A compact QCL based methane and nitrous oxide sensor for environmental and medical applications. Analyst 2014, 139, 2065–2069. [Google Scholar] [CrossRef] [PubMed]
- Karacan, C.Ö.; Ruiz, F.A.; Cotè, M.; Phipps, S. Coal mine methane: A review of capture and utilization practices with benefits to mining safety and to greenhouse gas reduction. Int. J. Coal Geol. 2011, 86, 121–156. [Google Scholar] [CrossRef]
- Cheng, Y.-P.; Wang, L.; Zhang, X.-L. Environmental impact of coal mine methane emissions and responding strategies in China. Int. J. Greenh. Gas. Control 2011, 5, 157–166. [Google Scholar] [CrossRef]
- Costello, B.P.J.D.; Ledochowski, M.; Ratcliffe, N.M. The importance of methane breath testing: A review. J. Breath Res. 2013, 7, 024001. [Google Scholar] [CrossRef] [PubMed]
- Shemshad, J.; Aminossadati, S.M.; Kizil, M.S. A review of developments in near infrared methane detection based on tunable diode laser. Sens. Actuators B Chem. 2012, 171, 77–92. [Google Scholar] [CrossRef]
- Wang, C.J.; Sahay, P. Breath analysis using laser spectroscopic techniques: Breath biomarkers, spectral fingerprints, and detection limits. Sensors 2009, 9, 8230–8262. [Google Scholar] [CrossRef] [PubMed]
- Sigrist, M.W.; Bartlome, R.; Marinov, D.; Rey, J.M.; Vogler, D.E.; Wachter, H. Trace gas monitoring with infrared laser-based detection schemes. Appl. Phys. B 2008, 90, 289–300. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Liang, L.; Yu, Q. Tunable fiber laser based photoacoustic spectrometer for multi-gas analysis. Sens. Actuators B Chem. 2011, 160, 1268–1272. [Google Scholar] [CrossRef]
- Jahjah, M.; Jiang, W.Z.; Sanchez, N.P.; Ren, W.; Patimisco, P.; Spagnolo, V.; Herndon, S.C.; Griffin, R.J.; Tittel, F.K. Atmospheric CH4 and N2O measurements near greater Houston area landfills using a QCL-based qepas sensor system during discover-AQ 2013. Opt. Lett. 2014, 39, 957–960. [Google Scholar] [CrossRef] [PubMed]
- Patel, C.K.N. Laser photoacoustic spectroscopy helps fight terrorism: High sensitivity detection of chemical warfare agent and explosives. Eur. Phys. J. Spec. Top. 2008, 153, 1–18. [Google Scholar] [CrossRef]
- Rocha, M.V.; Sthel, M.S.; Silva, M.G.; Paiva, L.B.; Pinheiro, F.W.; Miklos, A.; Vargas, H. Quantum-cascade laser photoacoustic detection of methane emitted from natural gas powered engines. Appl. Phys. B 2012, 106, 701–706. [Google Scholar] [CrossRef]
- Holthoff, E.L.; Heaps, D.A.; Pellegrino, P.M. Development of a mems-scale photoacoustic chemical sensor using a quantum cascade laser. IEEE Sens. J. 2010, 10, 572–577. [Google Scholar] [CrossRef]
- Sampaolo, A.; Patimisco, P.; Giglio, M.; Vitiello, M.; Beere, H.; Ritchie, D.; Scamarcio, G.; Tittel, F.; Spagnolo, V. Improved tuning fork for terahertz quartz-enhanced photoacoustic spectroscopy. Sensors 2016, 16, 439. [Google Scholar] [CrossRef] [PubMed]
- Sigrist, M.W. Trace gas monitoring by laser photoacoustic spectroscopy and related techniques (plenary). Rev. Sci. Instrum. 2003, 74, 485–490. [Google Scholar] [CrossRef]
- Hanyecz, V.; Mohacsi, A.; Pogany, A.; Varga, A.; Bozoki, Z.; Kovacs, I.; Szabo, G. Multi-component photoacoustic gas analyzer for industrial applications. Vib. Spectrosc. 2010, 52, 63–68. [Google Scholar] [CrossRef]
- Besson, J.P.; Schilt, S.; Thevenaz, L. Sub-ppm multi-gas photoacoustic sensor. Spectrochim. Acta. A 2006, 63, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Webber, M.E.; Pushkarsky, M.; Patel, C.K.N. Fiber-amplifier-enhanced photoacoustic spectroscopy with near-infrared tunable diode lasers. Appl. Opt. 2003, 42, 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Kosterev, A.A.; Bakhirkin, Y.A.; Tittel, F.K. Methane detection by means of quartz enhanced photoacoustic spectroscopy in NIR. In Proceedings of the 2007 Conference on Lasers & Electro-Optics/Quantum Electronics and Laser Science Conference (CLEO/QELS 2007), Baltimore, MD, USA, 6–11 May 2007. [CrossRef]
- Ma, Y.; Yu, G.; Zhang, J.; Yu, X.; Sun, R.; Tittel, F. Quartz enhanced photoacoustic spectroscopy based trace gas sensors using different quartz tuning forks. Sensors 2015, 15, 7596–7604. [Google Scholar] [CrossRef] [PubMed]
- Bernard-Schwarz, M.; Zwick, W.; Wenzel, L.; Klier, J.; Groschl, M. Field programmable gate array-assigned complex-valued computation and its limits. Rev. Sci. Instrum. 2014, 85, 093104. [Google Scholar] [CrossRef] [PubMed]
- Schwettmann, A.; Sedlacek, J.; Shaffer, J.P. Field-programmable gate array based locking circuit for external cavity diode laser frequency stabilization. Rev. Sci. Instrum. 2011, 82, 103103. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K. A highly integrated FPGA-based nuclear magnetic resonance spectrometer. Rev. Sci. Instrum. 2007, 78, 033103. [Google Scholar] [CrossRef] [PubMed]
- Restelli, A.; Abbiati, R.; Geraci, A. Digital field programmable gate array-based lock-in amplifier for high-performance photon counting applications. Rev. Sci. Instrum. 2005, 76, 093112. [Google Scholar] [CrossRef]
- Jinghong, L.; Kai, L.; Peng, Y. Design and Implementation of Flame Combustion State Detection System Based on FPGA. In Proceedings of the 2015 27th Chinese Control and Decision Conference, Qingdao, China, 23–25 May 2015.
- Botella, G.; Martín H., J.A.; Santos, M.; Meyer-Baese, U. FPGA-based multimodal embedded sensor system integrating low- and mid-level vision. Sensors 2011, 11, 8164–8179. [Google Scholar] [CrossRef] [PubMed]
- González, D.; Botella, G.; Meyer-Baese, U.; García, C.; Sanz, C.; Prieto-Matías, M.; Tirado, F. A low cost matching motion estimation sensor based on the NIOS II microprocessor. Sensors 2012, 12, 13126–13149. [Google Scholar] [CrossRef] [PubMed]
- Rodriguezdonate, C.; Botella, G.; Garcia, C.; Cabalyepez, E.; Prietomatias, M. Early experiences with opencl on FPGAs: Convolution case study. In Proceedings of the IEEE International Symposium on Field-Programmable Custom Computing Machines, Vancouver, BC, Canada, 3–5 May 2015; p. 235.
- González, D.; Botella, G.; García, C.; Prieto, M.; Tirado, F. Acceleration of block-matching algorithms using a custom instruction-based paradigm on a NIOS II microprocessor. EURASIP J. Adv. Signal. Process. 2013, 2013, 1–20. [Google Scholar] [CrossRef]
- Windh, S.; Ma, X.; Halstead, R.J.; Budhkar, P.; Luna, Z.; Hussaini, O.; Najjar, W.A. High-level language tools for reconfigurable computing. Proc. IEEE 2015, 103, 390–408. [Google Scholar] [CrossRef]
- Rothman, L.S.; Gordon, I.E.; Barbe, A.; Benner, D.C.; Bernath, P.E.; Birk, M.; Boudon, V.; Brown, L.R.; Campargue, A.; Champion, J.P.; et al. The HITRAN 2008 molecular spectroscopic database. J. Quant. Spectrosc. Radiat. Transfer 2009, 110, 533–572. [Google Scholar] [CrossRef]



© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Wang, H.; Liu, X. A Portable Laser Photoacoustic Methane Sensor Based on FPGA. Sensors 2016, 16, 1551. https://doi.org/10.3390/s16091551
Wang J, Wang H, Liu X. A Portable Laser Photoacoustic Methane Sensor Based on FPGA. Sensors. 2016; 16(9):1551. https://doi.org/10.3390/s16091551
Chicago/Turabian StyleWang, Jianwei, Huili Wang, and Xianyong Liu. 2016. "A Portable Laser Photoacoustic Methane Sensor Based on FPGA" Sensors 16, no. 9: 1551. https://doi.org/10.3390/s16091551
APA StyleWang, J., Wang, H., & Liu, X. (2016). A Portable Laser Photoacoustic Methane Sensor Based on FPGA. Sensors, 16(9), 1551. https://doi.org/10.3390/s16091551




