Microchambers with Solid-State Phosphorescent Sensor for Measuring Single Mitochondrial Respiration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mitochondrial Isolation
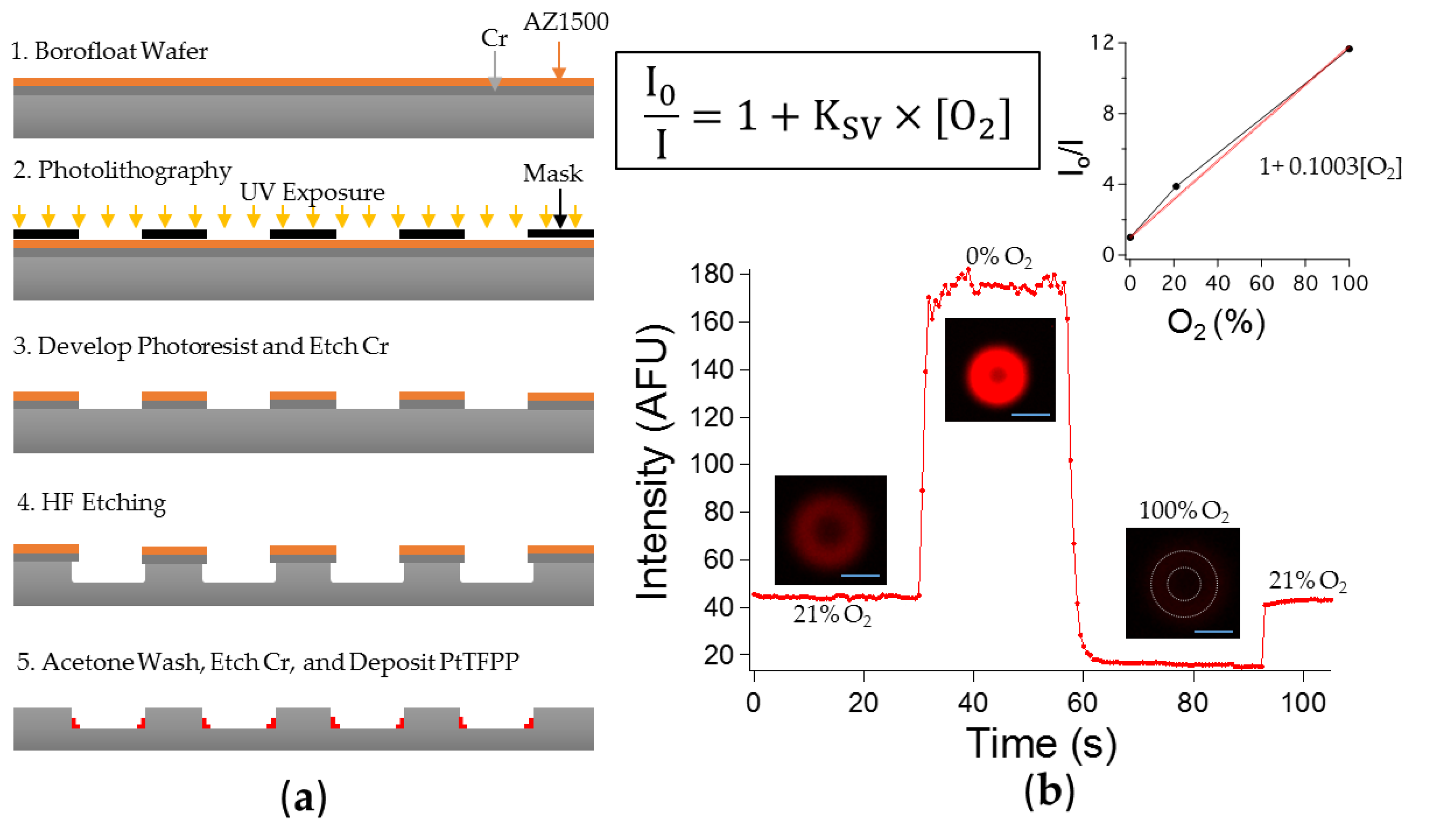
2.2. Microchambers Fabrication
2.3. PDMS and Viton-Coated Top Insulating Layer
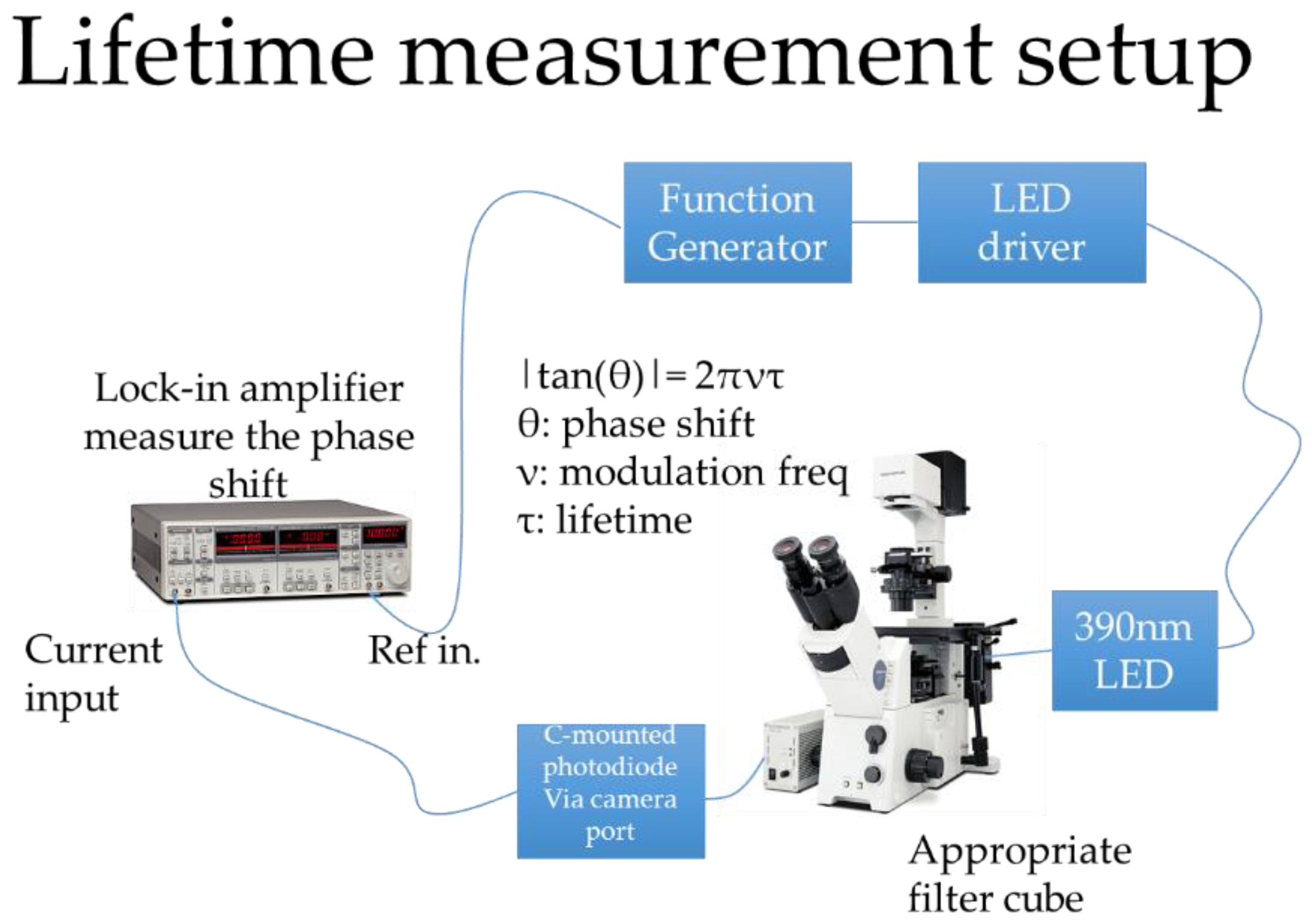
2.4. Oxygen Calibration and Sealing Test
2.5. Experimental Procedure
3. Results
3.1. Device Characterization
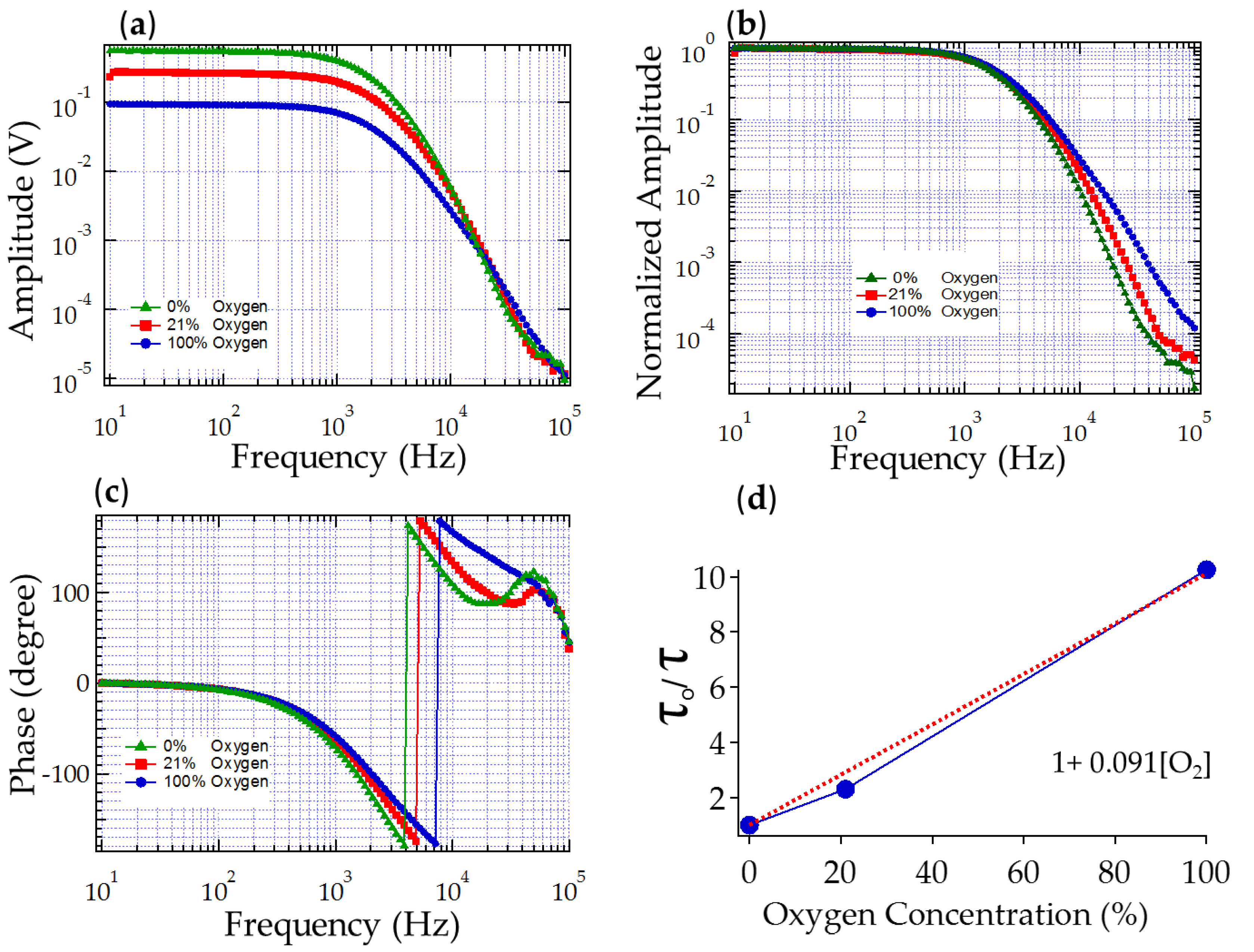
3.1.1. Sensitivity to Oxygen: Oxygen Quenches PtTFPP Phosphorescence
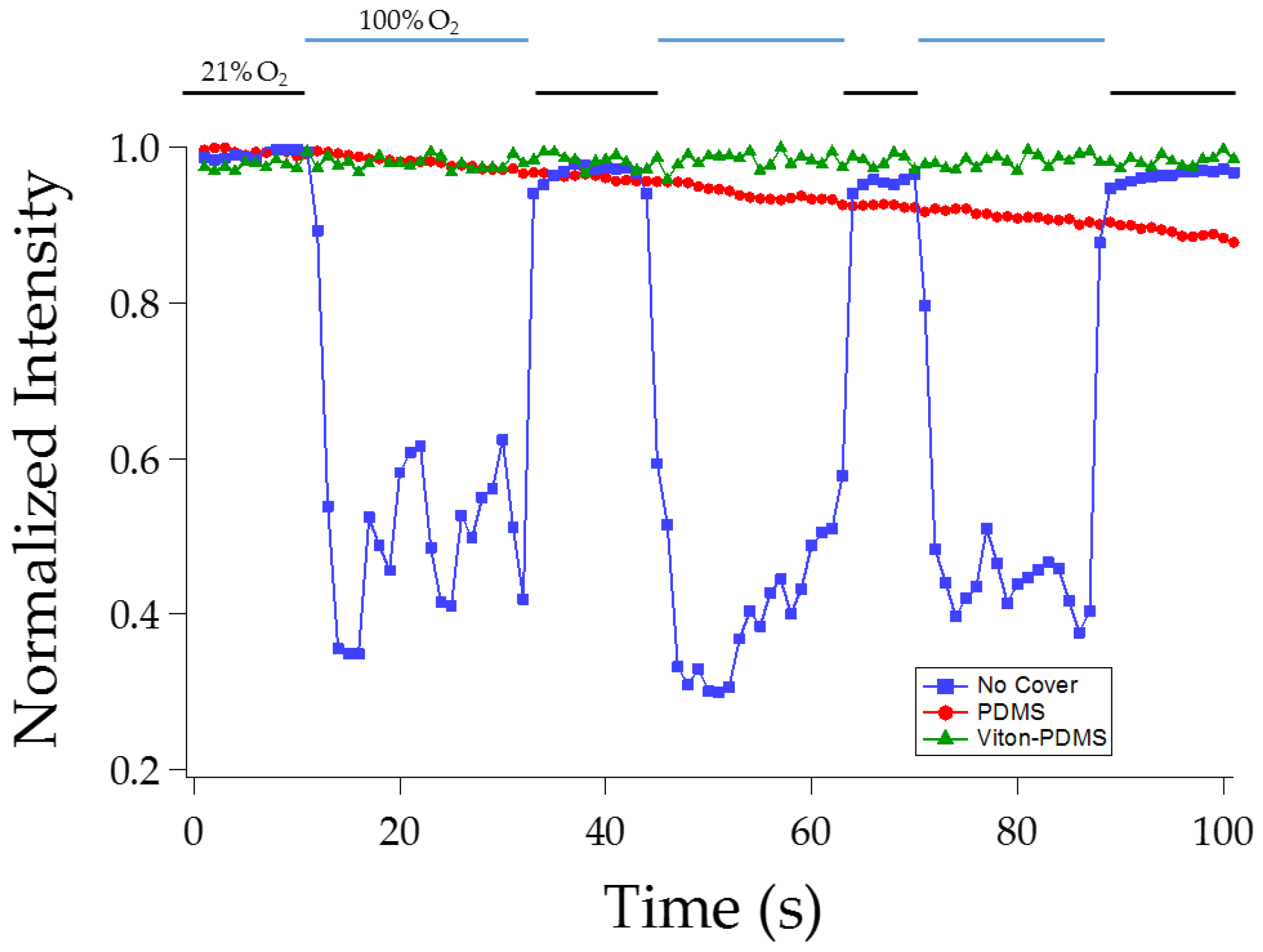
3.1.2. Effect of Viton Coating
3.2. Single Mitochondrial Respiration
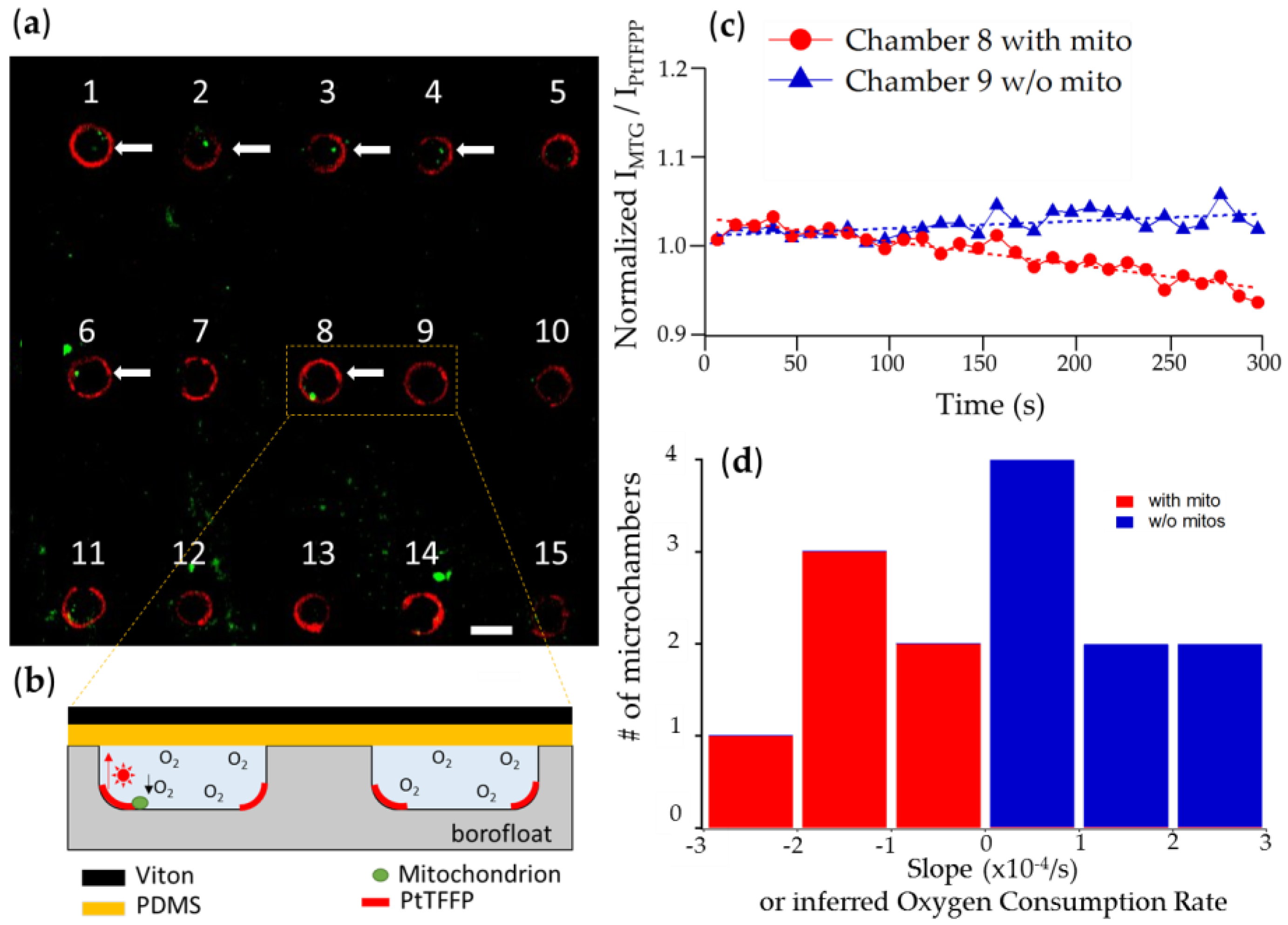
3.2.1. Chambers with Mitochondrion vs. Empty Chambers
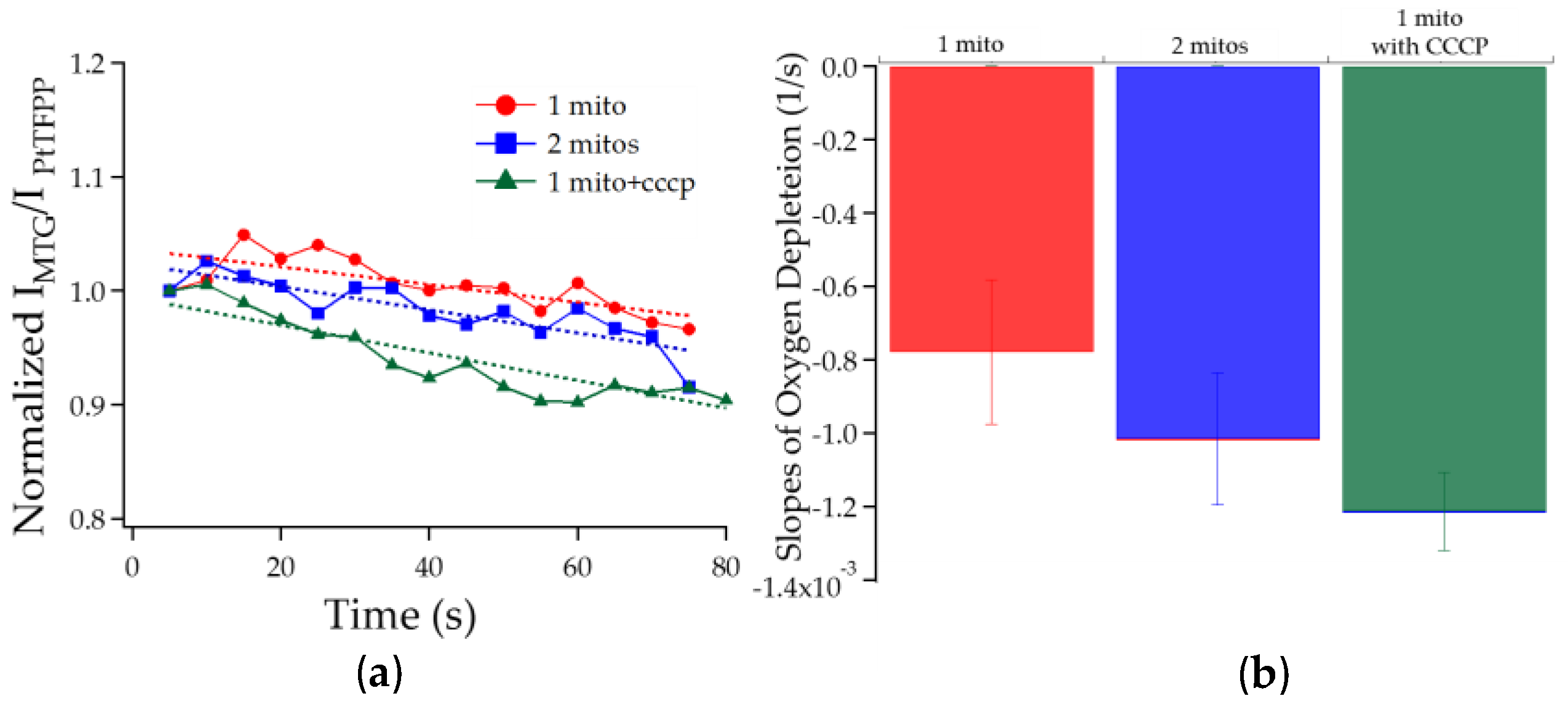
3.2.2. Chambers with One Mitochondrion vs. Two vs. One Treated with CCCP
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PDMS | polydimethylsiloxane |
| PtTFPP | Pt(II)-meso-tetra(pentafluorophenyl)porphine |
| PS | polystyrene |
| OCR | Oxygen consumption rate |
Apendix
A.I. 1: Qualitative vs. Quantitative Measurement of Respiration
A.I. 2. Proper Interpretation of Single Mitochondrial Respiration Data
A.I. 3. Lighting Conditions

References
- Wallace, D.C.; Fan, W.; Procaccio, V. Mitochondrial energetics and therapeutics. Annu. Rev. Pathol. 2010, 5, 297–348. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C. Bioenergetic origins of complexity and disease. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, D.; Ferguson, S. Bioenergetics, 4th ed.; Elsevier: Amsterdam, Netherlands, 2013. [Google Scholar]
- Chipuk, J.E.; Moldoveanu, T.; Llambi, F.; Parsons, M.J.; Green, D.R. The BCL-2 family reunion. Mol. Cell 2010, 37, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C. Mitochondria and cancer. Nat. Rev. Cancer 2012, 12, 685–698. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.W.; Brand, M.D.; Petrosyan, S.; Ashok, D.; Elorza, A.A.; Ferrick, D.A.; Murphy, A.N. High throughput microplate respiratory measurements using minimal quantities of isolated mitochondria. PLoS ONE 2011, 6, e21746. [Google Scholar] [CrossRef] [PubMed]
- Sharpley, M.S.; Marciniak, C.; Eckel-Mahan, K.; McManus, M.; Crimi, M.; Waymire, K.; Lin, C.S.; Masubuchi, S.; Friend, N.; Koike, M.; et al. Heteroplasmy of mouse mtDNA Is genetically unstable and results in altered behavior and cognition. Cell 2012, 151, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Waymire, K.G.; Narula, N.; Li, P.; Rocher, C.; Coskun, P.E.; Vannan, M.A.; Narula, J.; Macgregor, G.R.; Wallace, D.C. A mouse model of mitochondrial disease reveals germline selection against severe mtDNA mutations. Science 2008, 319, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Schönberg, A.; Schaefer, M.; Schroeder, R.; Nasidze, I.; Stoneking, M. Detecting heteroplasmy from high-throughput sequencing of complete human mitochondrial DNA genomes. Am. J. Hum. Genet. 2010, 87, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Payne, B.A.I.; Wilson, I.J.; Yu-Wai-Man, P.; Coxhead, J.; Deehan, D.; Horvath, R.; Taylor, R.W.; Samuels, D.C.; Santibanez-koref, M.; Chinnery, P.F. Universal heteroplasmy of human mitochondrial DNA. Hum. Mol. Genet. 2013, 22, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Galan-Acosta, L.; Norberg, E. Glucose metabolism provide distinct prosurvival benefits to non-small cell lung carcinomas. Biochem. Biophys. Res. Commun. 2015, 460, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Smiley, S.T.; Reers, M.; Mottola-Hartshorn, C.; Lin, M.; Chen, A.; Smith, T.W.; Steele, G.D.; Chen, L.B. Intracellular heterogeneity in mitochondrial membrane potentials revealed by a J-aggregate-forming lipophilic cation JC-1. Proc. Natl. Acad. Sci. USA 1991, 88, 3671–3675. [Google Scholar] [CrossRef] [PubMed]
- Nagrath, S.; Sequist, L.V.; Maheswaran, S.; Bell, D.W.; Ryan, P.; Balis, U.J.; Tompkins, R.G.; Haber, D.A. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 2011, 450, 1235–1239. [Google Scholar] [CrossRef] [PubMed]
- Frezza, C.; Cipolat, S.; Scorrano, L. Organelle isolation: Functional mitochondria from mouse liver, muscle and cultured fibroblasts. Nat. Protoc. 2007, 2, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Deegan, R.D.; Bakajin, O.; Dupont, T.F.; Huber, G.; Nagel, S.R.; Witten, T.A. Capillary flow as the cause of ring stains from dried liquid drops. Nature 1997, 389, 827–829. [Google Scholar] [CrossRef]
- Quaranta, M.; Borisov, S.M.; Klimant, I. Indicators for optical oxygen sensors. Bioanal. Rev. 2012, 4, 115–157. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, R.I.; Papkovsky, D.B. Intracellular probes for imaging oxygen concentration: How good are they? Methods Appl. Fluoresc. 2015, 3, 034001. [Google Scholar] [CrossRef]
- Van Dam, M. Solvent-Resistant Elastomeric Microfluidic Devices and Applications. Ph.D. Thesis, California Institute of Technology, Pasadena, CA, USA, 2006. [Google Scholar]
- Celina, M.; Gillen, K.T. Oxygen permeability measurements on elastomers at temperatures up to 225 °C. Macromolecules 2005, 38, 2754–2763. [Google Scholar] [CrossRef]
- Peacock, R.N. Practical selection of elastomer materials for vacuum seals. J. Vac. Sci. Technol. 1980, 17, 330. [Google Scholar] [CrossRef]
- Drioli, E.; Giorno, G. Comprehensive Membrane Science and Engineering, 1st ed.; Elsevier: Amsterdam, Netherlands, 2010. [Google Scholar]
- Molter, T.W.; McQuaide, S.C.; Suchorolski, M.T.; Strovas, T.J.; Burgess, L.W.; Meldrum, D.R.; Lidstrom, M.E. A microwell array device capable of measuring single-cell oxygen consumption rates. Sens. Actuators B 2009, 135, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.C.; Wolf, R.; Granger, D.; Taylor, Z. Continuous recording of blood oxygen tensions by polarography. J. Appl. Physiol. 1953, 6, 189–193. [Google Scholar] [PubMed]
- Hutter, E.; Unterluggauer, H.; Garedew, A.; Jansen-Durr, P.; Gnaiger, E. High-resolution respirometry—A modern tool in aging research. Exp. Gerontol. 2006, 41, 457. [Google Scholar] [CrossRef]
- Instruments, W. Warner Instruments—Mitocell Miniature Respirometer (MT-200, MT-200A). Available online: http://www.warneronline.com/product_info.cfm?id=386 (accessed on 7 July 2016).
- Vollmer, A.P.; Probstein, R.F.; Gilbert, R.; Thorsen, T. Development of an integrated microfluidic platform for dynamic oxygen sensing and delivery in a flowing medium. Lab Chip 2005, 5, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-C.; Luk, H.-N.; Lin, Y.-T.T.; Yuan, C.-Y. A Clark-type oxygen chip for in situ estimation of the respiratory activity of adhering cells. Talanta 2010, 81, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Papkovsky, D.B. Methods in optical oxygen sensing: Protocols and critical analyses. Methods Enzymol. 2004, 381, 715–735. [Google Scholar] [PubMed]
- Skolimowski, M.; Nielsen, M.W.; Emnéus, J.; Molin, S.; Taboryski, R.; Sternberg, C.; Dufva, M.; Geschke, O. Microfluidic dissolved oxygen gradient generator biochip as a useful tool in bacterial biofilm studies. Lab Chip 2010, 10, 2162–2169. [Google Scholar] [CrossRef] [PubMed]
- Grad, L.I.; Sayles, L.C.; Lemire, B.D. Introduction of an additional pathway for lactate oxidation in the treatment of lactic acidosis and mitochondrial dysfunction in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 2005, 102, 18367–18372. [Google Scholar] [CrossRef] [PubMed]
- Hansatech Instruments Oxygen Electrode & Chlorophyll Fluorescence Measurement Systems. Available online: http://www.hansatech-instruments.com/oxyg1.htm (accessed on 15 January 2016).
- O’Riordan, T.C.; Buckley, D.; Ogurtsov, V.; O’Connor, R.; Papkovsky, D.B. A cell viability assay based on monitoring respiration by optical oxygen sensing. Anal. Biochem. 2000, 278, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Will, Y.; Hynes, J.; Ogurtsov, V.I.; Papkovsky, D.B. Analysis of mitochondrial function using phosphorescent oxygen-sensitive probes. Nat. Protoc. 2006, 1, 2563–2572. [Google Scholar] [CrossRef] [PubMed]
- Nock, V.; Blaikie, R.J.; David, T. Patterning, integration and characterisation of polymer optical oxygen sensors for microfluidic devices. Lab Chip 2008, 8, 1300–1307. [Google Scholar] [CrossRef] [PubMed]
- Nock, V.; Alkaisi, M.; Blaikie, R.J. Photolithographic patterning of polymer-encapsulated optical oxygen sensors. Microelectron. Eng. 2010, 87, 814–816. [Google Scholar] [CrossRef]
- Molter, T.W.; McQuaide, S.C.; Holl, M.R.; Meldrum, D.R.; Dragavon, J.M.; Anderson, J.B.; Young, C.A.; Burgess, L.W.; Lidstrom, M.E. A new approach for measuring single-cell oxygen consumption rates. IEEE Trans. Autom. Sci. Eng. 2008, 5, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Grist, S.; Schmok, J.; Liu, M.-C.; Chrostowski, L.; Cheung, K. Designing a microfluidic device with integrated ratiometric oxygen sensors for the long-term control and monitoring of chronic and cyclic hypoxia. Sensors 2015, 15, 20030–20052. [Google Scholar] [CrossRef] [PubMed]
- Dragavon, J.; Molter, T.; Young, C.; Strovas, T.; McQuaide, S.; Holl, M.; Zhang, M.; Cookson, B.; Jen, A.; Lidstrom, M.; et al. A cellular isolation system for real-time single-cell oxygen consumption monitoring. J. R. Soc. Interface 2008, 5, S151–S159. [Google Scholar] [CrossRef] [PubMed]
- Gear, A.R.; Bednarek, J.M. Direct counting and sizing of mitochondria in solution. J. Cell Biol. 1972, 54, 325–345. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, R.I.; Kondrashina, A.V.; Koren, K.; Klimant, I.; Zhdanov, A.V.; Pakan, J.M.P.; McDermott, K.W.; Papkovsky, D.B. Small molecule phosphorescent probes for O2 imaging in 3D tissue models. Biomater. Sci. 2014, 2, 853. [Google Scholar] [CrossRef]
- Liu, H.; Yang, H.; Hao, X.; Xu, H.; Lv, Y.; Xiao, D.; Wang, H.; Tian, Z. Development of polymeric nanoprobes with improved lifetime dynamic range and stability for intracellular oxygen sensing. Small 2013, 9, 2639–2648. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, H.; Ito, H.; Inoue, M.; Tabata, K.; Sato, Y.; Yamagata, K.; Kizaka-Kondoh, S.; Kadonosono, T.; Yano, S.; Inoue, M.; et al. High resolution imaging of intracellular oxygen concentration by phosphorescence lifetime. Sci. Rep. 2015, 5, 10657. [Google Scholar] [CrossRef] [PubMed]
- Rumsey, W.L.; Vanderkooi, J.M.; Wilson, D.F. Imaging of phosphorescence: A novel method for measuring oxygen distribution in perfused tissue. Science 1988, 241, 1649–1651. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, R.I.; Papkovsky, D.B. Optical probes and techniques for O2 measurement in live cells and tissue. Cell. Mol. Life Sci. 2012, 69, 2025–2039. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Bull, B.; Christensen, K.; McNeill, J. Ratiometric single-nanoparticle oxygen sensors for biological imaging. Angew. Chem. Int. Ed. Engl. 2009, 48, 2741–2745. [Google Scholar] [CrossRef] [PubMed]
- Collins, T.J.; Berridge, M.J.; Lipp, P.; Bootman, M.D. Mitochondria are morphologically and functionally heterogeneous within cells. Eur. Mol. Biol. Organ. J. 2002, 21, 1616–1627. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; White, K.; Turnbull, D.M. Mitochondrial morphology, topology, and membrane interactions in skeletal muscle: A quantitative three-dimensional electron microscopy study. J. Appl. Physiol. (Bethesda, Md. 1985) 2013, 114, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Huser, J.; Blatter, L.A. Fluctuations in mitochondrial membrane potential caused by repetitive gating of the permeability transition pore. Biochem. J. 1999, 343, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Lascaratos, G.; Ji, D.; Wood, J.P.M.; Osborne, N.N. Visible light affects mitochondrial function and induces neuronal death in retinal cell cultures. Vis. Res. 2007, 47, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Fang, H.; Shang, W.; Liu, L.; Xu, Z.; Ye, T.; Wang, X.; Zheng, M.; Chen, Q.; Cheng, H. Superoxide flashes: Early mitochondrial signals for oxidative stress-induced apoptosis. J. Biol. Chem. 2011, 286, 27573–27581. [Google Scholar] [CrossRef] [PubMed]
| Company/Group | O2 Reporter | Detection Method | Chamber Size | Sealed? | Fluid Handling | Adhesion? | # of cells | Amount of mitos | Throughput | Cost |
| Oroboros[24] | Clark | Electrical | 2 mL | X | X | - | 106 | 1 mg | low | high |
| Warner [25,30] | Clark | Electrical | 100 µL | X | - | - | - | 20 µg | low | mid |
| Hansatech [31] | Clark | Electrical | 1 mL | X | - | - | 107 | 5 mg | low | mid |
| Seahorse [6,11] | Luminescence | Intensity | 5 µL | - | X | X | - | 10 µg | high | high |
| MitoXpress [32,33] | Luminescence | Intensity, Lifetime | 200 µL | X | - | - | - | 60 µg | high | mid |
| Canterbury [34,35] | PtOEPK | Intensity | - | X | X | X | - | - | - | low |
| MIT [26] | PtOEPK | Lifetime | 20 nL | - | X | - | - | - | - | low |
| Uwash [22,36] | Pt-porphyrin | Lifetime | 80 pL | X | - | - | 1 | - | mid | low |
| This work | PtTFPP | Intensity | 1.5 pL | X | - | - | - | 1 pg | mid | low |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pham, T.D.; Wallace, D.C.; Burke, P.J. Microchambers with Solid-State Phosphorescent Sensor for Measuring Single Mitochondrial Respiration. Sensors 2016, 16, 1065. https://doi.org/10.3390/s16071065
Pham TD, Wallace DC, Burke PJ. Microchambers with Solid-State Phosphorescent Sensor for Measuring Single Mitochondrial Respiration. Sensors. 2016; 16(7):1065. https://doi.org/10.3390/s16071065
Chicago/Turabian StylePham, Ted D., Douglas C. Wallace, and Peter J. Burke. 2016. "Microchambers with Solid-State Phosphorescent Sensor for Measuring Single Mitochondrial Respiration" Sensors 16, no. 7: 1065. https://doi.org/10.3390/s16071065
APA StylePham, T. D., Wallace, D. C., & Burke, P. J. (2016). Microchambers with Solid-State Phosphorescent Sensor for Measuring Single Mitochondrial Respiration. Sensors, 16(7), 1065. https://doi.org/10.3390/s16071065





