Zn(II)-Coordinated Quantum Dot-FRET Nanosensors for the Detection of Protein Kinase Activity
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. FRET Measurement
2.3. Protein Kinase Assay
2.4. Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry (MALDI-MS) Analysis
3. Results and Discussion
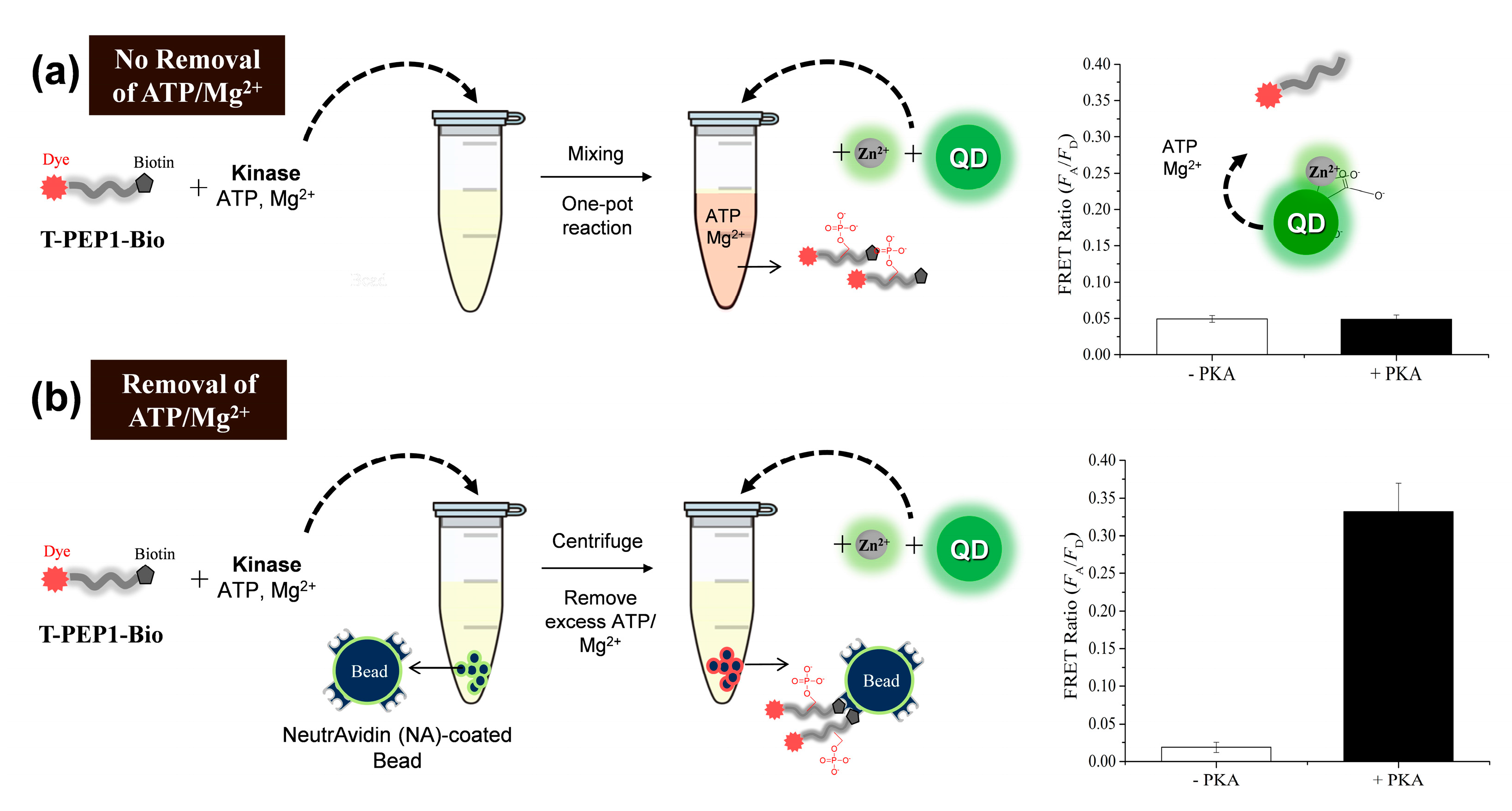
3.1. Detection Principle and Metal Affinity-Based QD-FRET
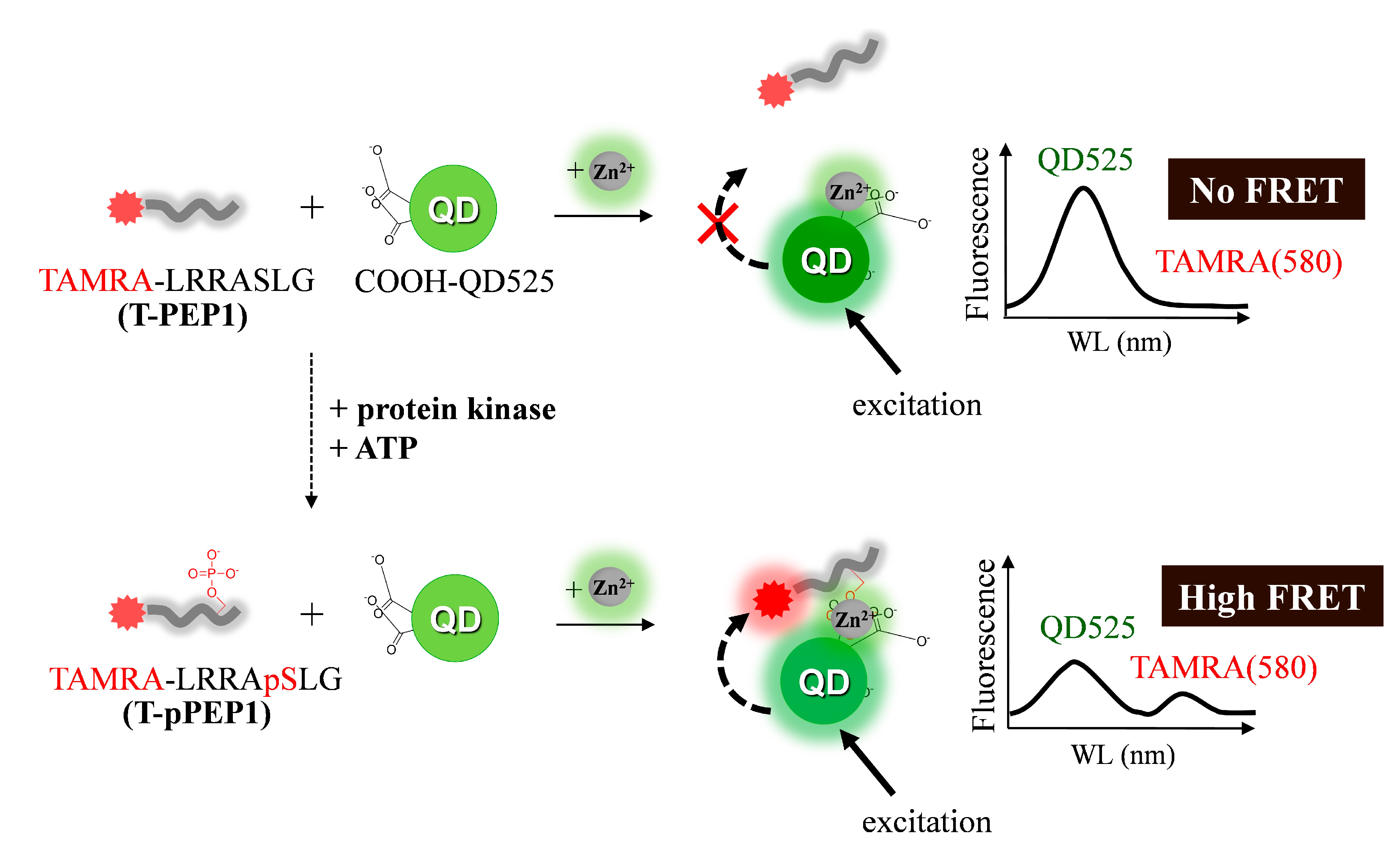
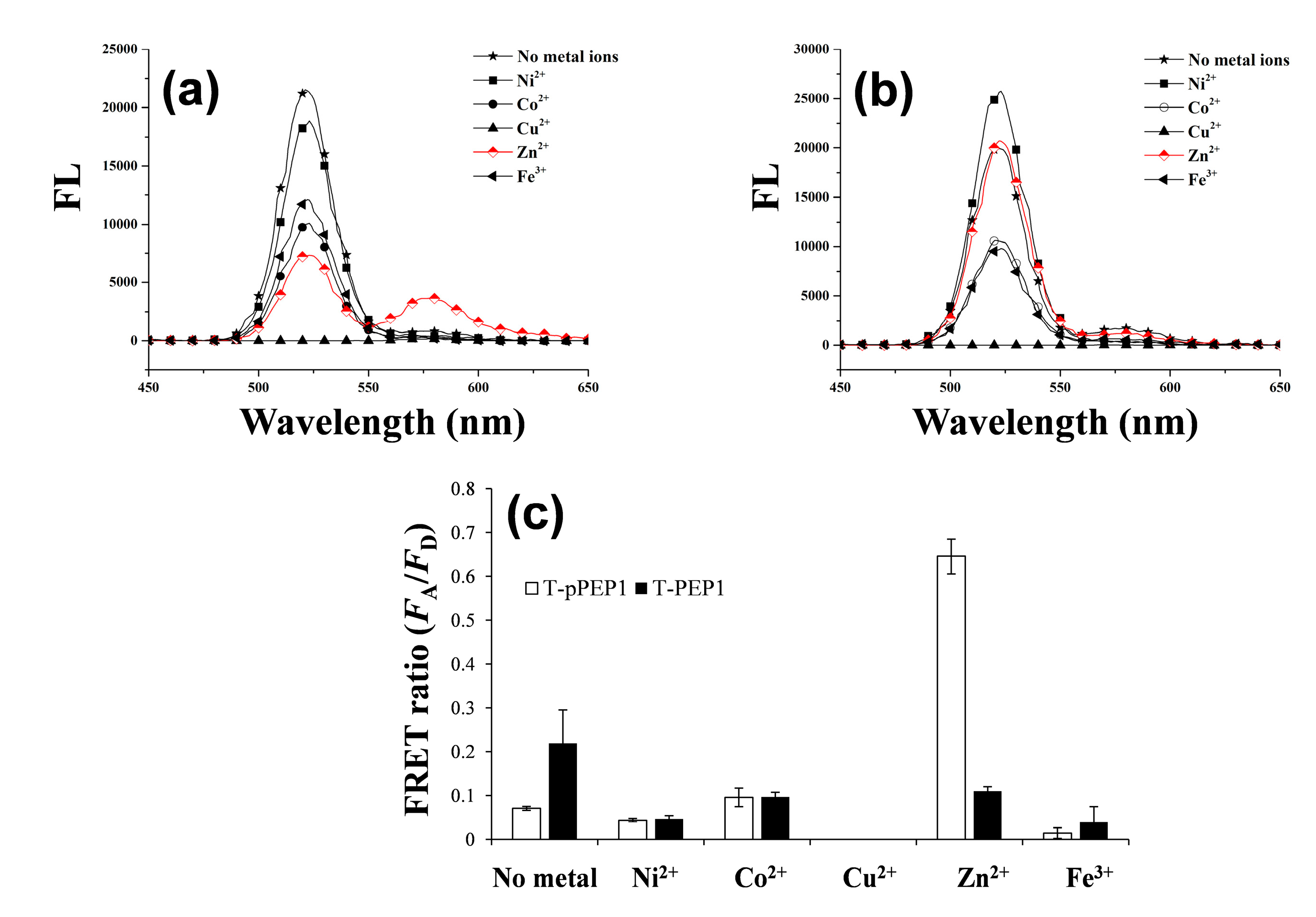
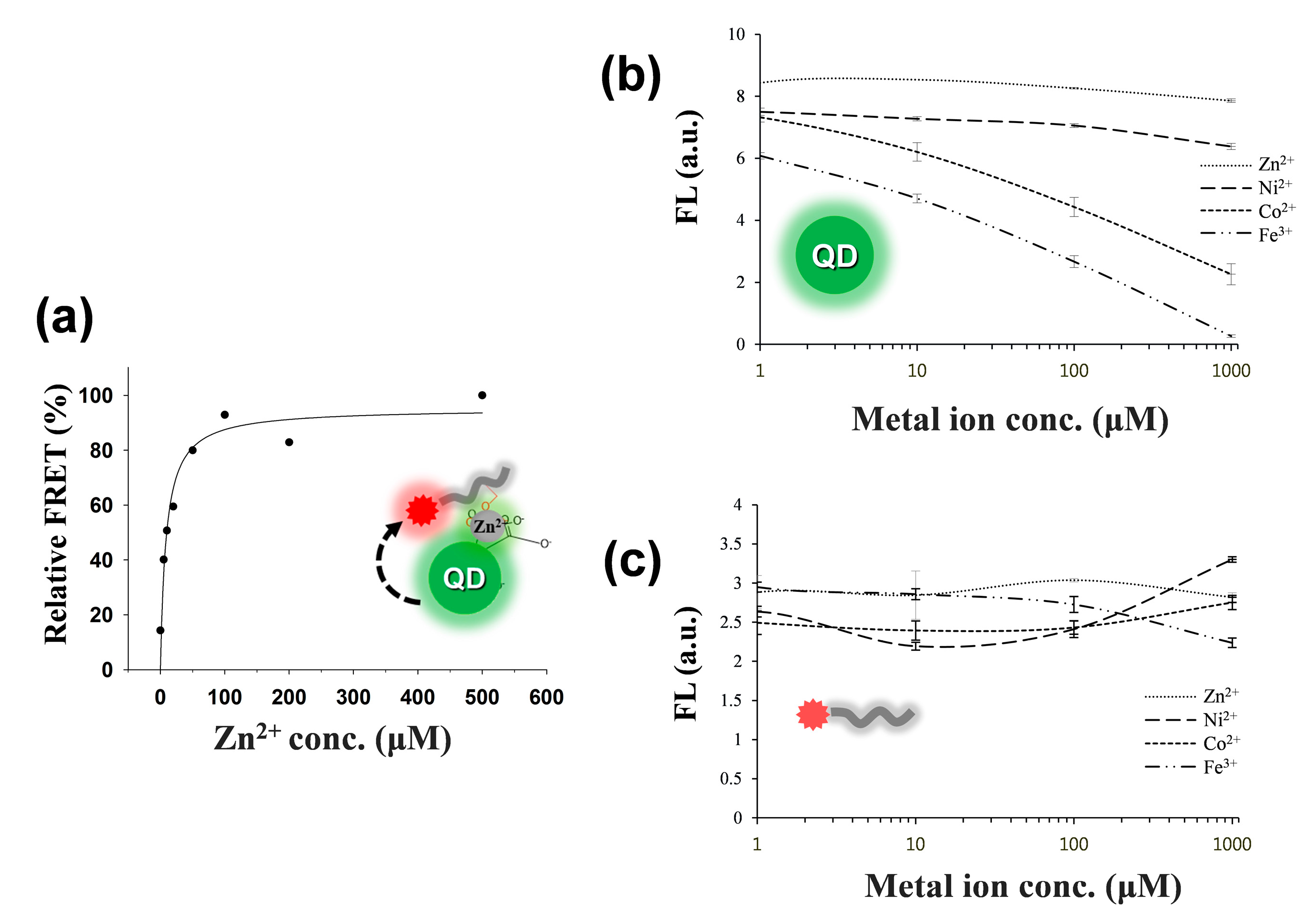
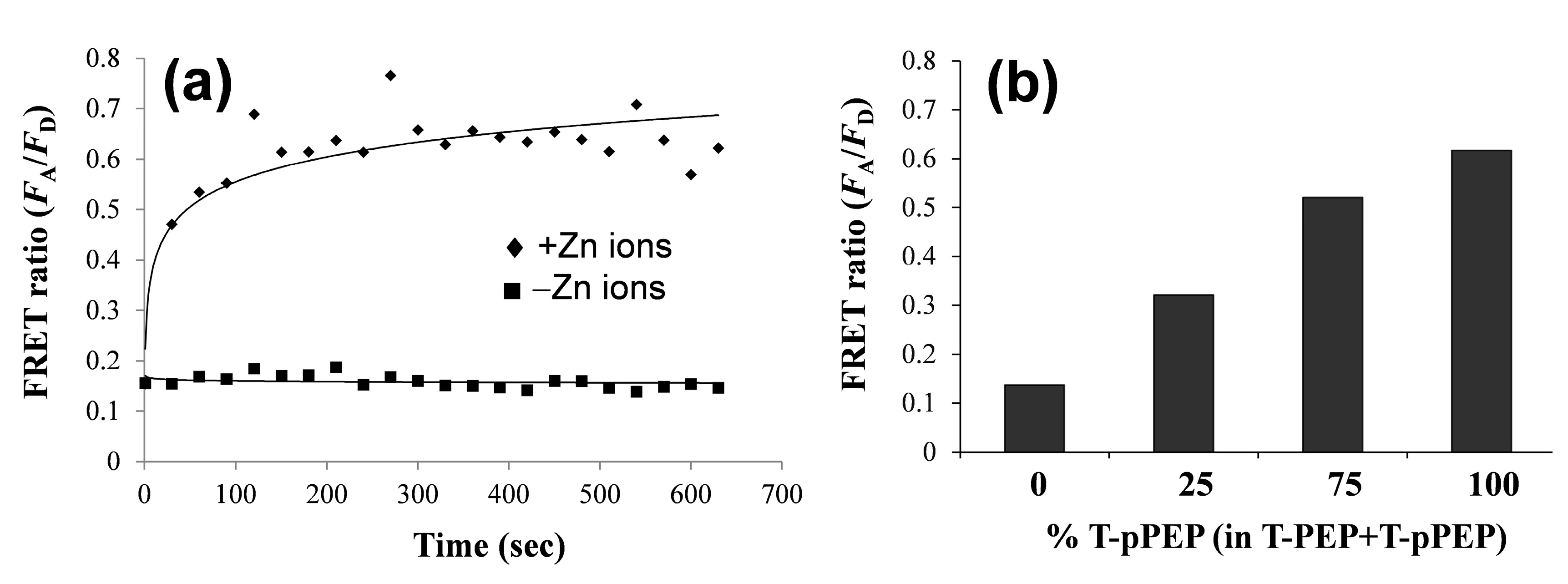
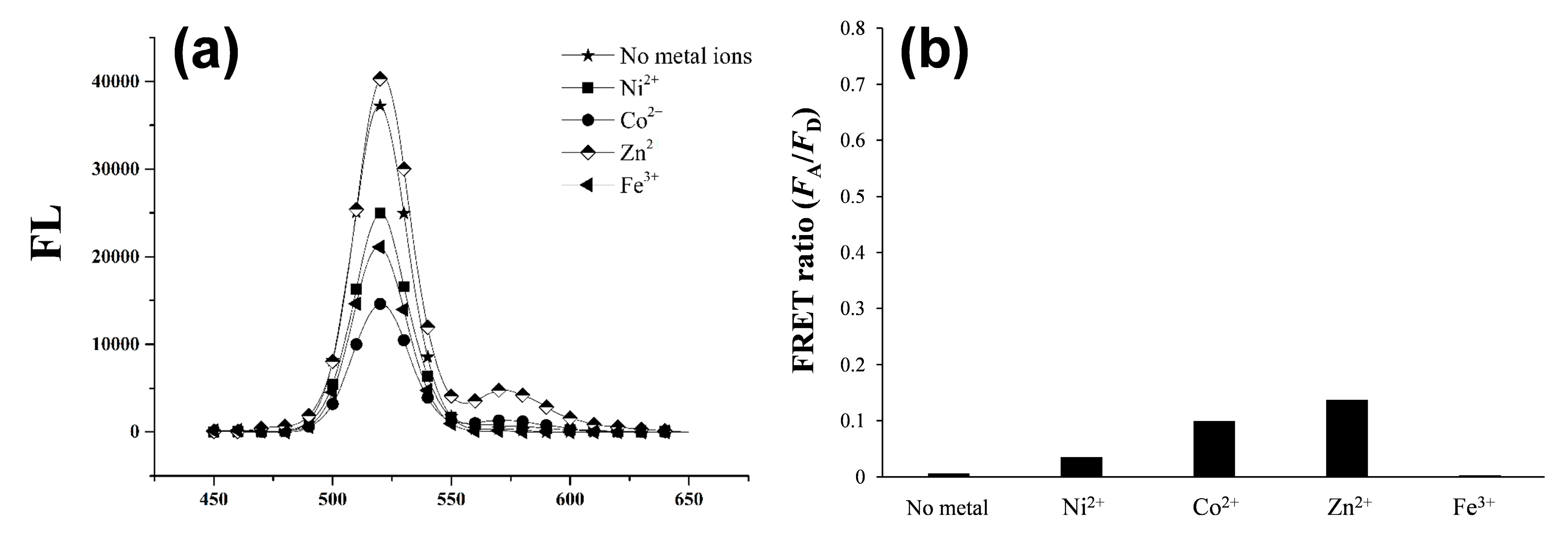
3.2. Changes in QD-FRET Ratio by Phosphorylation Degree
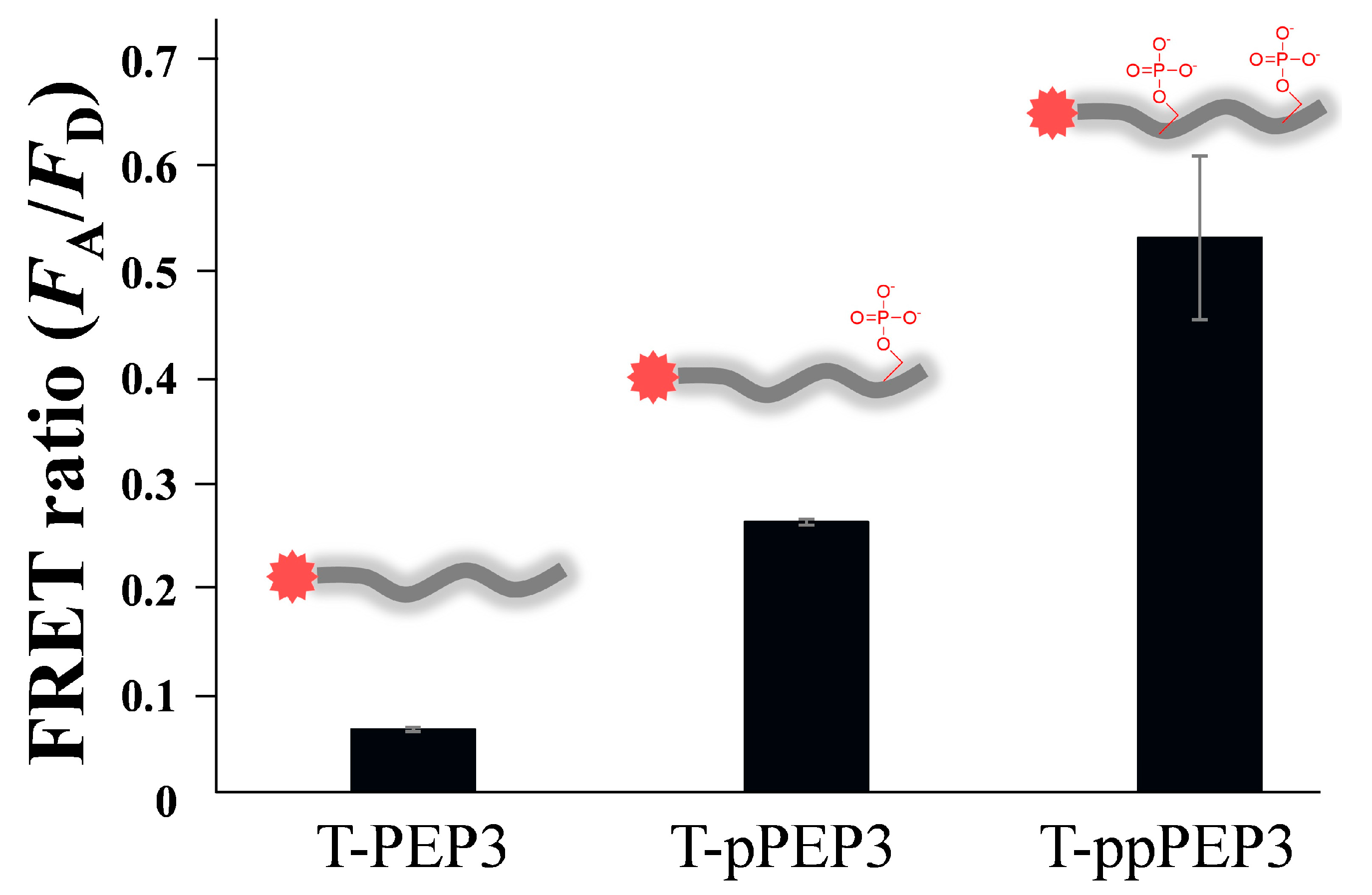
3.3. Protein Kinase Assay by QD-FRET
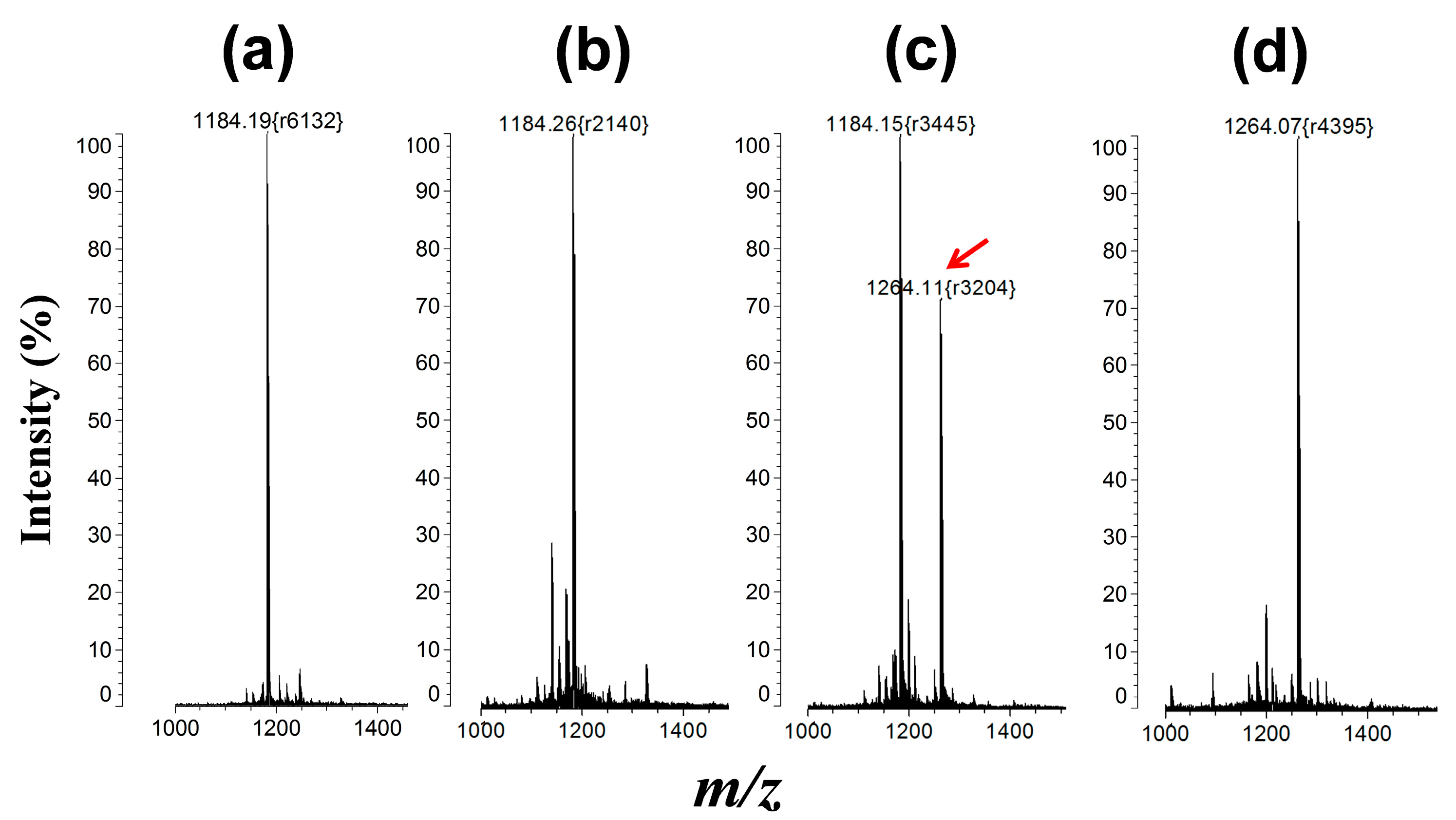
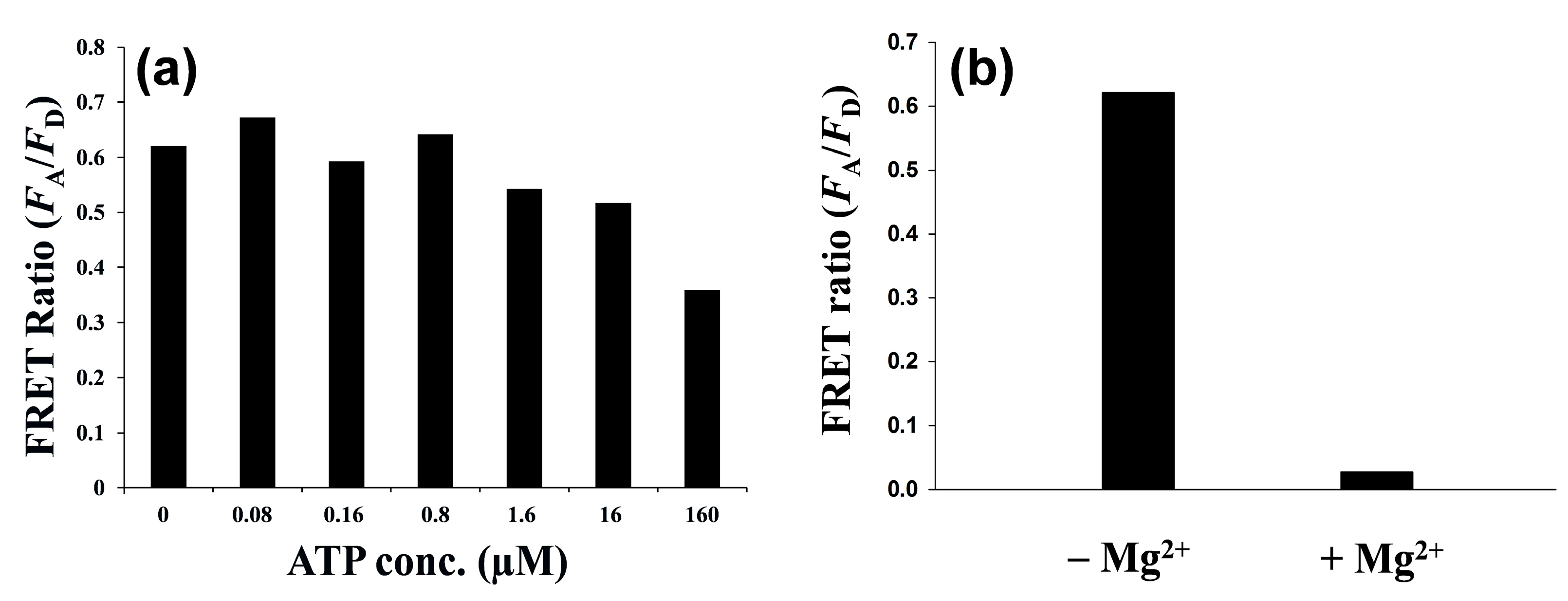
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Manning, G.; Whyte, D.B.; Martinez, R.; Hunter, T.; Sudarsanam, S. The protein kinase complement of the human genome. Science 2002, 298, 1912–1934. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.S.; Kornev, A.P. Protein kinases: Evolution of dynamic regulatory proteins. Trends Biochem. Sci. 2011, 36, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, P.T.; Michell, R.H.; Kirk, C.J. A simple assay-method for determination of the specific radioactivity of the gamma-phosphate group of 32P-labeled ATP. Biochem. J. 1983, 210, 717–720. [Google Scholar] [PubMed]
- Wooten, M.W. In-gel kinase assay as a method to identify kinase substrates. Sci. STKE 2002, 2002. [Google Scholar] [CrossRef] [PubMed]
- Ross, H.; Armstrong, C.G.; Cohen, P. A non-radioactive method for the assay of many serine/threonine-specific protein kinases. Biochem. J. 2002, 366, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Steinberg, T.H.; Cooley, L.A.; Gee, K.R.; Beechem, J.M.; Patton, W.F. Quantitative analysis of protein phosphorylation status and protein kinase activity on microarrays using a novel fluorescent phosphorylation sensor dye. Proteomics 2003, 3, 1244–1255. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.P.; Oh, Y.H.; Kim, H.S. Protein kinase assay on peptide-conjugated gold nanoparticles. Biosens. Bioelectron. 2008, 23, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Ni, Q.; Titov, D.V.; Zhang, J. Analyzing protein kinase dynamics in living cells with FRET reporters. Methods 2006, 40, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Allen, M.D. FRET-based biosensors for protein kinases: Illuminating the kinome. Mol. Biosyst. 2007, 3, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Rodems, S.M.; Hamman, B.D.; Lin, C.; Zhao, J.; Shah, S.; Heidary, D.; Makings, L.; Stack, J.H.; Pollok, B.A. A fret-based assay platform for ultra-high density drug screening of protein kinases and phosphatases. ASSAY Drug Dev. Technol. 2002, 1, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Liu, C.H.; Yang, T.; Wang, F.F.; Li, Z.P. A versatile platform for highly sensitive detection of kinase activity based on metal ion-mediated FRET using an anionic conjugated polymer. Chem. Commun. 2013, 49, 3887–3889. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.C.; Deacon, S.; Horiuchi, K. The challenge of selecting protein kinase assays for lead discovery optimization. Expert Opin. Drug Discov. 2008, 3, 607–621. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C. Fluorescent biosensors–probing protein kinase function in cancer and drug discovery. Biochim. Biophys. Acta-Proteins Proteom. 2013, 1834, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.W.; Hildebrandt, N. Semiconductor quantum dots for in vitro diagnostics and cellular imaging. Trends Biotechnol. 2012, 30, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Clapp, A.R.; Medintz, I.L.; Mauro, J.M.; Fisher, B.R.; Bawendi, M.G.; Mattoussi, H. Fluorescence resonance energy transfer between quantum dot donors and dye-labeled protein acceptors. J. Am. Chem. Soc. 2004, 126, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Yeh, H.C.; Kuroki, M.T.; Wang, T.H. Single-quantum-dot-based DNA nanosensor. Nat. Mater. 2005, 4, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.P.; Oh, Y.H.; Oh, E.; Ko, S.; Han, M.K.; Kim, H.S. Energy transfer-based multiplexed assay of proteases by using gold nanoparticle and quantum dot conjugates on a surface. Anal. Chem. 2008, 80, 4634–4641. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Han, B.; Park, C.; Jeong, J.S.; Ahn, J.J.; Ha, S.M.; Hwang, S.Y.; Ahn, Y. Development of microbiochip for detection of metalloproteinase 7 using fluorescence resonance energy transfer. Biochip J. 2013, 7, 164–172. [Google Scholar] [CrossRef]
- Shiosaki, S.; Nobori, T.; Mori, T.; Toita, R.; Nakamura, Y.; Kim, C.W.; Yamamoto, T.; Niidome, T.; Katayama, Y. A protein kinase assay based on fret between quantum dots and fluorescently-labeled peptides. Chem. Commun. 2013, 49, 5592–5594. [Google Scholar] [CrossRef] [PubMed]
- Ghadiali, J.E.; Cohen, B.E.; Stevens, M.M. Protein kinase-actuated resonance energy transfer in quantum dot-peptide conjugates. ACS Nano 2010, 4, 4915–4919. [Google Scholar] [CrossRef] [PubMed]
- Freeman, R.; Finder, T.; Gill, R.; Willner, I. Probing protein kinase (CK2) and alkaline phosphatase with cdse/zns quantum dots. Nano Lett. 2010, 10, 2192–2196. [Google Scholar] [CrossRef] [PubMed]
- Kimura, E. Roles of Zinc(II) ion in zinc enzymes. Pure Appl. Chem. 1993, 65, 355–359. [Google Scholar] [CrossRef]
- Bobyr, E.; Lassila, J.K.; Wiersma-Koch, H.I.; Fenn, T.D.; Lee, J.J.; Nikolic-Hughes, I.; Hodgson, K.O.; Rees, D.C.; Hedman, B.; Herschlag, D. High-resolution analysis of Zn2+ coordination in the alkaline phosphatase superfamily by exafs and X-ray crystallography. J. Mol. Biol. 2012, 415, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Evers, T.H.; Appelhof, M.A.M.; Meijer, E.W.; Merkx, M. His-tags as Zn(II) binding motifs in a protein-based fluorescent sensor. Protein Eng. Des. Sel. 2008, 21, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.O.; Kim, E.J.; Lim, B.; Kim, T.W.; Kim, Y.P. Rapid detection of protein phosphatase activity using Zn(II)-coordinated gold nanosensors based on his-tagged phosphopeptides. Anal. Chem. 2015, 87, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Pomerantz, A.H.; Allfrey, V.G.; Merrifield, R.B.; Johnson, E.M. Studies on mechanism of phosphorylation of synthetic polypeptides by a calf thymus cyclic amp-dependent protein-kinase. Proc. Natl Acad. Sci. 1977, 74, 4261–4265. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, E.; Takahashi, M.; Takeda, H.; Shiro, M.; Koike, T. Recognition of phosphate monoester dianion by an alkoxide-bridged dinuclear Zinc(II) complex. Dalton Trans. 2004, 8, 1189–1193. [Google Scholar] [CrossRef] [PubMed]
- Chu, B.Y.; Soncin, F.; Price, B.D.; Stevenson, M.A.; Calderwood, S.K. Sequential phosphorylation by mitogen-activated protein kinase and glycogen synthase kinase 3 represses transcriptional activation by heat shock factor-1. J. Biol. Chem. 1996, 271, 30847–30857. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Meng, Y.H.; Mivechi, N.F. Glycogen synthase kinase 3 beta and extracellular signal-regulated kinase inactivate heat shock transcription factor 1 by facilitating the disappearance of transcriptionally active granules after heat shock. Mol. Cell. Biol. 1998, 18, 6624–6633. [Google Scholar] [PubMed]
- Ye, J.Y.; Zhang, X.M.; Young, C.; Zhao, X.L.; Hao, Q.; Cheng, L.; Jensen, O.N. Optimized imac-imac protocol for phosphopeptide recovery from complex biological samples. J. Prot. Res. 2010, 9, 3561–3573. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.T.; Hsu, C.C.; Tsai, C.F.; Lin, P.C.; Lin, C.C.; Chen, Y.J. Nanoprobe-based immobilized metal affinity chromatography for sensitive and complementary enrichment of multiply phosphorylated peptides. Proteomics 2011, 11, 2639–2653. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, B.; Park, J.-I.; Lee, K.J.; Lee, J.-W.; Kim, T.-W.; Kim, Y.-P. Zn(II)-Coordinated Quantum Dot-FRET Nanosensors for the Detection of Protein Kinase Activity. Sensors 2015, 15, 17977-17989. https://doi.org/10.3390/s150817977
Lim B, Park J-I, Lee KJ, Lee J-W, Kim T-W, Kim Y-P. Zn(II)-Coordinated Quantum Dot-FRET Nanosensors for the Detection of Protein Kinase Activity. Sensors. 2015; 15(8):17977-17989. https://doi.org/10.3390/s150817977
Chicago/Turabian StyleLim, Butaek, Ji-In Park, Kyung Jin Lee, Jin-Won Lee, Tae-Wuk Kim, and Young-Pil Kim. 2015. "Zn(II)-Coordinated Quantum Dot-FRET Nanosensors for the Detection of Protein Kinase Activity" Sensors 15, no. 8: 17977-17989. https://doi.org/10.3390/s150817977
APA StyleLim, B., Park, J.-I., Lee, K. J., Lee, J.-W., Kim, T.-W., & Kim, Y.-P. (2015). Zn(II)-Coordinated Quantum Dot-FRET Nanosensors for the Detection of Protein Kinase Activity. Sensors, 15(8), 17977-17989. https://doi.org/10.3390/s150817977






