Thiol Reactive Probes and Chemosensors
Abstract
: Thiols are important molecules in the environment and in biological processes. Cysteine (Cys), homocysteine (Hcy), glutathione (GSH) and hydrogen sulfide (H2S) play critical roles in a variety of physiological and pathological processes. The selective detection of thiols using reaction-based probes and sensors is very important in basic research and in disease diagnosis. This review focuses on the design of fluorescent and colorimetric probes and sensors for thiol detection. Thiol detection methods include probes and labeling agents based on nucleophilic addition and substitution, Michael addition, disulfide bond or Se-N bond cleavage, metal-sulfur interactions and more. Probes for H2S are based on nucleophilic cyclization, reduction and metal sulfide formation. Thiol probe and chemosensor design strategies and mechanism of action are discussed in this review.1. Introduction
Sulfhydryl-containing compounds are often referred to as mercaptans due to their ability to react with mercury. They have unique chemical reactivity and thus special utility in chemical reactions [1] and in biological processes [2–4]. Thiols and thiophenols are widely used intermediates in synthetic chemistry; dithiothreitol (DTT) and 2-mercaptoethanol are common antioxidants used in biology labs [5]; aminothiols, such as cysteine (Cys, 1), homocysteine (Hcy, 2), and γ-L-glutamyl-L-cysteinylglycine (glutathione or GSH, 3) play essential metabolic roles in biological systems. For example, Cys plays versatile roles in protein structure and function [6]. The sulfhydryl group of Cys serves as an ideal nucleophile in nucleophilic enzyme catalysis. Its ability to undergo reversible redox reactions under physiological conditions is essential for maintaining tertiary and quaternary protein structures through disulfide formation [7]. Hcy is a key intermediate generated during the biosynthesis of Cys from the essential amino acid methionine (Met, 4) [3] (Figure 1). It is implicated in the health of the cardiovascular (CV) system [3]. The tripeptide GSH is present at very high levels (0.1–10 mM) in the cell (comprising about 90% of non-protein sulfur) and protects the cells against oxidative stress [2,8] among many other functions. GSH levels in cancer cells can impact the effectiveness of chemotherapy [9]. In addition to the above-mentioned thiols, thiol drugs such as D-penicillamine (D-PEN or PenA) [10] and tiopronin (TP or Thiola) [11], are also widely used in clinical practice. The quantitative detection of these drugs and their metabolites are very important in related clinical research. Hydrogen sulfide (H2S), the simplest mercaptan, has been known as an environmental hazard and toxic gas for many years. Recently, hydrogen sulfide has been recognized as one of the three gasotransmitters [12–15], together with nitric oxide (NO) and carbon monoxide (CO), that are endogenously produced and essential for maintaining the health of cardiovascular system among other roles. Even the unpleasant smell of mercaptans has found important applications. For example, some small molecule thiols such as ethanethiol and butanethiol are added to natural gas to help warn of gas leaks.
The metabolism and transportation of these sulfur-containing compounds in biological systems are closely related to a series of important enzymes and proteins, the deficiency of which could lead to various physiological/pathological conditions [16–18]. Furthermore, fluctuations in the endogenous concentration of these thiols indicate the functional state of the corresponding enzymes/proteins and are correlated with disease [18–20]. Thus, the detection of concentrations of mercaptans has implications and significance in clinical diagnosis. Among all the biologically important mercaptans, Hcy has been extensively studied as a biomarker for various reasons [21]. Deficiency in the expression of enzymes such as cystathionine β-synthase (CBS) and cystathionine γ-lyase (CSE or CGL) or their cofactors may lead to high levels or abnormal accumulation of Hcy, which characterizes inherited diseases such as homocystinuria [22], Down syndrome [23,24] and other clinical conditions such as vitamin (folate [25], cobalamin (vitamin B12) [26] or vitamin B6 [27]) deficiency, cardiovascular disease [28] and renal failure [29]. The tripeptide GSH is found to be present at low micromolar range in plasma [30]. However, the cytosol contains 0.1-10 mM GSH, depending on the cell type, while in most cells the concentration is 1-2 mM [4]. Because GSH provides antioxidant protection for the cell, the GSH concentration is also significant. The ratio of free GSH and its oxidized state glutathione disulfide (GSSG, 5), which is normally >100:1 [31], is an indicator for both the corresponding enzyme (GSSG reductase or enzymes related to de novo GSH synthesis) activity and the redox state of the cell [32]. Low GSH concentration or [GSH]/[GSSG] ratio is related to inflammation and lung diseases, such as cystic fibrosis [19,33].
H2S is synthesized in the cell both enzymatically and non-enzymatically. The enzymatic synthesis of H2S is catalyzed by CBS, CSE [34,35] and cysteine aminotransferase (CAT)/3-mercaptopyruvate sulfurtransferase (3MST) [36,37]. H2S undergoes fast metabolism and is involved in the regulation of various systems, such as the cardiovascular [38–41] and the central nervous system (CNS) [42,43]. Concrete evidence has revealed the physiological and therapeutic significance of H2S, leading to a rapid growth in research activity involving H2S [13,14,44]. Endogenous and exogenous hydrogen sulfide has been demonstrated to exert either beneficial or detrimental effects in many pathological conditions. H2S was found to have therapeutic benefit in ischemia-induced heart failure [39,45] and hyperhomocysteinemia-induced hypertension [46]. The endogenous hydrogen sulfide level is related to Down syndrome [47] and lung diseases [48]. Exogenous hydrogen sulfide may confer myocardial protection against ischemia/reperfusion injury and exerts a protective effect against anti-inflammatory drug-induced gastric mucosal injury [38].
The utility of thiol detection is not limited to biomarker studies. Numerous kinetic assays have been developed based upon the quantification of thiols. These methods are used in studying enzymes that are naturally involved in the metabolism of thiols, such as CBS [49] and S-ribosylhomocysteinase (LuxS) [50], as well as enzymes that use artificial sulfur-containing substrates and can produce free thiols, such as acetylcholine [51], carboxypeptidase A [52], and so on.
From the above, one can clearly recognize the need for the development of chemosensors and probes for mercaptans, which has been reviewed in refs [53,54]. In the following sections, we present a variety of reaction-based chemoprobes reported for the selective detection of individual mercaptans, with a focus on strategies used for designing such chemoprobes. Due to page limitation, for applications of commonly used probes in biological samples such as plasma, urine and cell cultures, and detection procedures, readers are referred to recent reviews [21,55,56]. It should be noted that there are common challenges in the detection of mercaptans. They are easily oxidizable, and do not have readily detectable and distinguishable spectroscopic properties. Thus, the detection of thiols usually consists of reduction, chemical derivatization or labeling, and chromatographic separation followed by quantitative detection. Disulfides, such as GSSG, cystine, and homocystine, are commonly brought to their reduced state by the treatment of reducing agents such as DTT, tris(2-carboxyethyl)phosphine (TCEP) or NaBH4. In addition, selectivity among various thiols is a key issue, and methods for the accurate detection with minimal or no work-up, or in vivo, are the most desirable.
2. Probes for Biological Thiols
2.1. Labeling and Detection of Thiols Based on Nucleophilic Substitution
Due to the strong nucleophilicity of the sulfhydryl group, it can react readily with electrophiles such as Michael acceptors and alkylating agents [1]. Therefore, some probes for thiols are either Michael acceptors or alkylating agents conjugated to a chromophore or fluorophore. The most widely used alkylating/labeling agents include monobromobimane (mBrB or mBB, 6, Figure 2), 4-fluoro-7-sulfamoylbenzofurazan (ABD-F, 9), 7-fluorobenzo-2-oxa-1,3-diazole-4-sulfonic acid ammonium salt (SBD-F, 10), 5-iodoacetamidofluorescein (5-IAF, 11), 2-chloro-1-methylpyridinium iodide (CMPI, 12) and 2-chloro-1-methylquinolinium tetrafluoroborate (CMQT, 13) (Figure 2). One common feature of these reagents is that they all bear a halogen that can undergo nucleophilic substitution with thiols. mBrB, ABD-F, SBD-F and 5-IAF form fluorescent conjugates with thiols, while CMPI and CMQT yield UV-absorbing conjugates. The samples could then be analyzed using high performance liquid chromatography (HPLC) or capillary electrophoresis (CE) coupled with a UV-vis detector (such as a diode array detector or DAD) or a fluorescence detector. Several commonly used fluorogenic and chromogenic labeling agents are compared in Table 1 in terms of their reactivity and limit of detection (LOD). Compared with absorption detection (such as CMPI and CMQT), fluorescent probes (such as mBrB) show higher sensitivity with detection limits in the picomolar range. This is due to the low background and less interference from the matrix. Compared to fluorescence detection, absorption methods, especially the absorptions in the UV range (<400 nm), are more prone to interference from other biological substrates. It should be noted that the detection limits listed in Table 1 were obtained from chromatographic methods, which includes an additional separation step. Such data are not directly comparable to the detection limits obtained from direct detections. In addition, detection limits reported for most probes by various labs were measured in different solvent systems (buffers or a mixed solvent of a buffer and an organic solvent). They are not directly comparable either.
Among these agents, mBrB (6) can easily undergo SN2 substitution with a sulfhydryl group at ambient temperature. The thiol-bimane conjugate 7 emits at 480 nm when excited at 380 nm. It has been extensively used in the quantification of thiols [19,57–60]. Automated separation-quantification of biological thiols in plasma and urine samples has been developed using mBrB [74]. Other bimane derivatives, such as monochlorobimane (mCB, 8) [59], have also been used for thiol detection. ABD-F and SBD-F [75] consist another group of useful reagents for the derivatization of thiols, yielding fluorescent conjugates (λex 380 nm, λem 510 nm) and have been used for detection of thiols in both plasma and tissues [62,65,76,77], although these reagents require fairly high temperature and pH for the substitution reaction. 5-IAF reacts rapidly with thiols at room temperature at pH 12.5 and is used in CE analysis of thiols in plasma and bacteria [66–68]. CMPI (12) forms thiol conjugates with an absorption maxima at 310 nm, providing nanomolar detection limits. It has been used for the analysis of thiols in urine and plasma [69,70]. CMQT (13) is user-friendly because it undergoes a fast substitution reaction with thiols. The conjugates show a maximum absorption at 355 nm. It has also been used for the quantitative analysis of thiols in urine and plasma [71–73,78,79]. Other UV labeling agents, such as p-bromophenacyl bromide, have also been reported [80]. However, the short absorption wavelength (263 nm) could limit their use because of spectral interference from the matrix.
These methods are widely used in both research and clinical analysis. However, one drawback of these agents is the lack of any selectivity among various thiols because they rely on simple nucleophilic addition or substitution for chromogenic or fluorescent labeling. As a result, detection and quantification of individual thiols using these techniques relies on separations such as HPLC or CE to differentiate various thiol-derivatives. Another disadvantage is interference from excess amount of labeling agent, especially in the case of fluorescence. This issue can be overcome if the labeling agent show significantly increased signal after conjugation.
Due to the high level of electron-deficiency on the phenyl ring, the 2,4-dinitrophenyl sulfonyl (DNBS) moiety can act as an electron sink when attached to a fluorophore and may incur photoinduced electron transfer (PET) resulting in the quenching of the fluorescence. Furthermore, DNBS can easily undergo de-sulfonylation in the presence of thiols, releasing SO2 gas and the attached fluorophore, thus resulting in a fluorescence increase (Figure 3). This strategy has been used in the development of many thiol probes. Examples include fluorescein derivatives 14 and 15 (Figure 3), which are almost non-fluorescent (ΦFL 0.0007 and 0.0003, respectively in HEPES buffer at pH 7.4). In these two compounds, the hydroxyl group was “capped” as a sulfonate. After reaction with thiols in HEPES buffer, 16 and 17 are produced, leading to strong fluorescence (ΦFL 0.75 and 0.58, λex 460 nm, λem 560 nm). Compound 14 shows a low detection limit of 2 pM for thiols such as GSH and Cys. It was investigated for its application in cholinesterase (ChE) assays [81]. Although 0.7% of compound 14 was observed to be decomposed after 1 h incubation in buffer at 37 °C, high reaction rates with thiols still allowed for high throughput screening of ChE inhibitors using this probe.
In another example (compound 19, Figure 4) the DNBS moiety was conjugated to a red-emissive fluorophore, which can be released through reaction with thiols [82]. A donor-π-acceptor architecture was built into the molecule, which was masked by the DNBS group. After addition of thiol, the sulfonyl ester moiety collapsed and resulted in the release of aniline as an electron donor. This process turned the fluorescence on with on/off ratios of 60 for GSH and 110-120 for Cys and Hcy (λex 560 nm, λem 623 nm), providing a detection limit of about 3 μM. Almost exclusive selectivity was obtained for Cys over other amino acids. Furthermore, no hydrolysis was detected over 12 h at 37 °C. Its compatibility over a wide pH range (5.6–9.5) also makes this an excellent probe. The probe was utilized in the bioimaging of thiols in albino Swiss mouse embryonic fibroblast cells (3T3 lines). The probe showed good cell permeability and reacted with intracellular thiols. The control experiment using N-methylmaleimide to consume all free thiols did not show any fluorescence after incubation with the probe. In another example, a merocyanine fluorophore was conjugated to a DNBS moiety (Compound 20, Figure 4) [83]. Cleavage of the DNBS moiety tunes the intramolecular charge transfer (ICT) state of the molecule and provides an absorption shift from 380 nm to 530 nm (ε∼78,000 M−1·cm−1) and a strong fluorescence emission at 553 nm. Both absorption and emission intensity are linearly related to thiol concentrations in MeOH/H2O 3:7. A nanomolar detection limit was achieved.
High photostability, high quantum yield and low pH sensitivity makes 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY) one of the best fluorescent scaffolds available. Recently a BODIPY-based fluorescent probe (compound 21, Figure 4) for thiols was reported [84]. The DNBS moiety was conjugated to this probe, providing an efficient fluorescence quenching effect. After exposure to Cys or GSH in PBS buffer for 10 min, a 20–25-fold increase in fluorescence (λex 527 nm, λem 570 nm) was observed, presumably as a result of the displacement reaction described. Micromolar detection limit could be achieved with a moderate selectivity (>3-fold) over other amino acids. The probe was stable at pH 7.3 for at least 12 hr at room temperature. Imaging using this probe was demonstrated in monkey renal fibroblast COS-7 cell lines. In 2011, another BODIPY-based red-emitting off-on fluorescent probe (compound 22, Figure 4) was reported [85]. This probe showed a 46-fold fluorescent enhancement after exposure to thiols in MeOH/H2O 4:1 (λex 520 nm, λem 590 nm) with a detection limit of 7 μM. Probe 22 showed moderate specificity toward Cys over Hcy with about 3-fold difference, while no obvious response was observed for GSH. The fluorescence of the reaction product remained pH-independent over a wide pH range (pH 2–8). Cellular thiol imaging was performed in SGC-H446 cells. The specificity for thiols was also confirmed by N-methylmaleimide control experiments.
Compared to fluorescence, phosphorescence has the advantages of large Stokes shift, long luminescence lifetimes and ease of measurement. Phosphorescent probes utilizing a ruthenium complex have been developed for the detection of thiols based on the nucleophilic addition-fragmentation of DNBS (Figure 5, compound 23) [86]. The probe 23 consists of a DNBS moiety attached to a Ru(II) poly(1,10-phenanthroline) complex as the luminophore, taking advantage of the metal-to-ligand charge transfer (MLCT) red emission. The addition of 20 μM of Cys led to the formation of 24 and a 90-fold increase in phosphorescence emission at 600 nm in a mixed solvent of acetonitrile/water 4:1 v/v, providing a detection limit in the high nanomolar range. The selectivity for Cys over other amino acids was over 40 fold. A fluorescent imaging study of this probe was performed using NCI-H446 cells. Control experiments were carried out with cells pre-treated with N-methylmaleimide, which covalently conjugate to thiols (this is also discussed in the next section).
2.2. Labeling of Thiols Based on Michael Addition
Because of their excellent nucleophilicity, thiols react readily with Michael acceptors [1]. Recently, the Michael addition reaction has been widely used in the development of chemoprobes for thiols. Maleimide as one excellent Michael acceptor has found numerous applications in various studies. DBPM 27 and BIPM 28 (Figure 6) are such examples with off-on behaviors. Thus, maleimide has been conjugated to fluorophores of long wavelength leading to some commercially available fluorescent thiol-probes including ThioGlo™1 29 (λex 379 nm, λem 513 nm), ThioGlo™3 30 (λex 378 nm, λem 446 nm), and ThioGlo™5 31 (λex 365 nm, λem 536 nm). These probes react with thiols rapidly (usually in 2-5 min) and are used for fluorescent labeling of proteins. A HPLC-based study for the detection of thiols using ThioGlo™3 shows a detection limit as low as 50 fM for the derivative of GSH [87].
A number of other probes bearing a maleimide group have been reported as fluorescent labels for thiols. In a recent example, a maleimide group was introduced at the ortho position of the phenyl ring of BODIPY derivative (32, Figure 7) [88], resulting in quenched fluorescence due to PET from BODIPY to maleimide. Experimental results showed that ortho substitution is crucial for the PET quenching. In terms of fluorescence quantum yields, they were 0.002 (ortho), 0.37 (meta) and 0.54 (para) respectively, which were determined using fluorescein (0.85) as the reference. After reaction with thiol, the PET effect was inhibited and the fluorescence of 32 was restored, affording a 350-fold intensity increase [89], acieving detection in the nanomolar range. Another example of a maleimide functionalized fluorescent probe is tetraphenylethene derivative 33 (TPE-MI, Figure 7). It showed an aggregation-induced emission (AIE) through thiol-maleimide addition in aqueous solution [90]. Though the selectivity among thiol containing molecules Cys, Hcy and GSH was low, the detection limit was as low as 1 ppb and the AIE occurred both in aqueous solution and the solid state.
Recently, another fluorescent probe for thiols using a Michael addition to the maleimide moiety was reported (Compound 34, Figure 7) [91]. The detection limit of this probe was ∼20 nM for Cys based on S/N = 3. This probe was also evaluated in human metastatic breast cancer cells (MDA-MB 231) for imaging biothiols, using N-phenylmaleimide treated cells as a negative control.
Besides maleimide, the quinone moiety is also frequently chosen as a Michael acceptor for construction of thiol probes. For example, a donor-acceptor compound 35 was designed as a colorimetric probe (Figure 7) [92]. The probe showed an absorption band at 582 nm due to ICT. This band decreased upon addition of thiols such as Cys and GSH, mostly likely due to the nucleophilic addition of thiols on the quinone ring. A 17-fold fluorescence increase was observed for Cys in a 1:1 mixed solvent of H2O and THF The linear relationship between the absorption intensity change and the thiol concentration could be used for quantitation.
Open chain Michael acceptors are another option for the design of fluorescent sensor of thiols. A real-time thiol quantitation method reported was based on the modulation of intramolecular PET quenching upon addition of mercapto species [93]. Water soluble sensor 36 (Figure 8) reacts rapidly with thiols to form conjugate 37 with a rate constant of 7.0 × 104 M−1·s−1 in Tris buffer at 25 °C. The reaction of 50 μM probe 36 and 50 mM β-mercaptoethiol has a t1/2 of 3 ms. The conjugate 37 (λex 400 nm, λem 470 nm) has a fluorescence quantum yield more than 470-fold higher than that of 36. The detection limit was as low as 0.5 nM. This has enabled the development of a high-throughput fluorescence assay for glutathione reductase, since the assay requires probes with very short response time. A similar idea was used in the design of α, β-unsaturated ketone derivative 38 (Figure 8) [94].
Upon thiol addition, the conjugation is disrupted and the fluorescence of the coumarin fluorophore is restored. The probe 38 is a highly sensitive thiol reagent showing over 200-fold increases in fluorescence (λex 444 nm, λem 496 nm) by forming 39 through Michael addition. The detection limit was found to be 1 μM for Cys in 25 mM phosphate buffer (pH 7.4). The malononitrile group is an effective fluorescent quencher and has been utilized in another coumarin-based biothiol probe 40 (Figure 8) [95], which forms fluorescent product 41 through reaction with thiols. However compound 40 only showed a relatively low fluorescent enhancement upon reaction with thiols (5.6–12 fold, λex 394 nm, λem 475 nm) in DMSO/HEPES buffer 1:2 (v/v).
The nucleophilic addition of a sulfhydryl group to electron-deficient squaraines has also been used in the detection of thiols [96]. Compounds 42 and 43 are such derivatives (Figure 9). They showed strong absorption at 640 nm in acetonitrile/H2O. The addition of thiols to the solution of the probe in acetonitrile/MES buffer (pH 6.5) leads to the formation of adducts 44 and 45, where the absorption band at 640 nm is significantly decreased, which is associated with a color change from blue to colorless. Near infrared (NIR) spectroscopy is emerging as a very powerful tool in tissue imaging because light in the 650–900 nm range is known to penetrate much deeper than visible light [97]. In 2009, a π-extended NIR squaraine dye 46 (Figure 9) formed by linking two bispyrrole molecules was reported [98]. Upon addition of thiols such as Cys and GSH, the π-conjugation of probe 46 is interrupted leading to the formation of 47, which shows significantly increased fluorescence (λex 410 nm, λem 595 nm and λex 730 nm, λem 802 nm) in both visible and NIR region. The results also confirmed that the level of the aminothiols in blood doubles after smoking. Probes of similar structures have also been reported for cyanide detection [99]. However, since one would not normally expect cyanide in blood, the lack of selectivity over cyanide does not pose a significant interference problem.
Michael addition triggered ring-opening reaction is another strategy in developing thiol reactive probes (Figure 10). In 2009, a chromene-based colorimetric probe 48 (λmax 292 nm) was reported [100]. The 4-nitrophenolate moiety was generated after Michael addition, leading to the formation of 49 (λmax 405 nm) and a fast (within 10 s) visual color change from colorless to yellow. An analogous fluorescein-based probe 50 was reported for the detection of biothiol molecules [101]. Probe 50 responds to Cys, Hcy and GSH, forming fluorescent conjugate 52 through intermediate 51 with a detection limit in the high nM range. Further applications of the probe for intracellular thiol detection and monitoring of thiols in zebrafish were examined. The results showed that thiol species in the eye, gallbladder, egg and fin were detected by the probe after 5 μM 1 h exposure.
2.3. Detection of Thiols Based on Disulfide and Se-N Bond Cleavage
Reducing ability is one of the most important properties of thiols. It imparts thiols with biological function and also enables the selective detection of mercaptans. Probes based on disulfide cleavage are commonly used for estimation of total sulfhydryl groups in protein samples. These probes, which usually exhibit weak absorption, share the structure of disulfide-linked chromophores. The reduction of the disulfide bond in the probe by sulfhydryl groups in the sample results in the production of a free chromophore, which absorbs strongly within the UV-vis range. The absorption could be recorded on a UV-vis spectrophotometer [102] or a multi-label counter [103]. One prominent example is the Ellman's reagent (5,5′-dithiobis-2-nitrobenzoic acid or DTNB, 53, Figure 11), which was introduced in 1959 [104]. DTNB has two electron-deficient phenyl groups linked by a disulfide bond. This “activated” disulfide readily undergoes a transsulfuration reaction or reduction by a sulfhydryl group, releasing the conjugate 54 and the highly chromogenic product 5-thio-2-nitrobenzoate (TNB, 55) with a strong absorption at 412 nm (ε = 14.15 × 103 M−1cm−1 at 25 °C) [105].
One problem with Ellman's reagent is its low stability. DTNB is relatively stable at pH below 8. Under more basic conditions DTNB undergoes obvious degradation, leading to undesired background absorption increase. However, slightly basic conditions (pH values greater than 8) are necessary for optimal reaction rates. Due to this reason, other reagents with relatively higher stability have been reported as alternatives. Such examples include n-octyldithionitrobenzoate (ODNB, 56, Figure 12) [106] and 5-(2-aminoethyl)dithio-2-nitrobenzoate (ADNB, 57) [107,108]. Both are mixed disulfides bearing one electron donating aliphatic chain. Because of the less activated disulfide bond and one less TNB group, they both show lower background than Ellman's reagent for thiol detection. ADNB was reported to show similar reactivity with thiols as Ellman's reagent with much slower hydrolysis [107,108]. On the other hand, the octyl group renders ODNB much more reactive to the sulfhydryl groups in proteins that reside in a hydrophobic environment. ADNB is more reactive to sulfhydryl groups in an anionic environment. These sulfhydryl groups usually react much slower with doubly negatively charged DTNB [106].
2,2′-Dipyridyl disulfide (2-PDS, 58) and 4,4′-dipyridyl disulfide (4-PDS, 59) [109,110] were reported for the determination of thiols such as GSH and protein thiols. When reduced by thiols, 2-PDS and 4-PDS forms 2-thiopyridone (2-TP) and 4-thiopyridone (4-TP), which absorb strongly at 343 and 324 nm, respectively. These two probes are reported to be more reactive to thiols at lower pH (3-6) compared with DTNB [111]. The protonation of the nitrogen on pyridinyl ring is believed to further activate the disulfide bond. Besides, 4-PDS reacts with GSH about 3 (pH 7)-30 (pH 4) times faster than 2-PDS [109].
Another interesting probe, Ratio-HPSSC (60) was developed by the Lin group [112]. In this probe (60, Figure 12), tetrakis(4-hydroxyphenyl)porphyrin is linked to coumarin by a disulfide bond. Due to the overlap of the coumarin emission (λex 350 nm, λem 459 nm) with the porphyrin absorption (421 nm, Soret band) and subsequent Förster resonance energy transfer (FRET), the fluorescence of coumarin in Ratio-HPSSC is almost completely quenched. When exposed to thiols, the disulfide bond is cleaved, switching off FRET and thus restoring the fluorescence of coumarin. This probe shows good selectivity and sensitivity to thiols such as Cys with a detection limit lower than 1 μM in PBS buffer/ethanol. Thiol imaging in live Hela cells was also studied.
Cyclization-release is one of the most popular strategies used in directed drug release. This was also found to be useful in the development of thiol probes. In a typical example, a disulfide bond is linked to a fluorophore by a carbamate linkage (61, Figure 13). After the disulfide is reduced by thiols to form 62, the newly formed sulfhydryl group could undergo an intramolecular cyclization with the carbonyl, releasing the linked fluorophore as a free amine. In the first example published in this class [113], 63 (RhoSS, Figure 13) released rhodamine 110 after incubation with thiols for 1–2 h at 37 °C resulting in a fluorescence increase at 535 nm. This probe was tested in cellular imaging studies in live Hela cells. Strong fluorescence response was observed after incubating with cells. When incubated with N-ethylmaleimide pre-treated cells, where sulfhydryl groups on thiols are capped, no such fluorescence response was observed. The cellular distribution of the probe was studied by co-staining and the probe seems to be localized within the cytosol.
Two-photon microscopy (TPM) [114,115] is a relatively new technology that has received great interest in the past several years owing to its applications in deep tissue imaging (>500 μm). Specifically, a two-photon microscope generates pulsed laser beams, focusing within less than femtoliter volumes in the objective, and employs two photons of lower energy to excite the fluorophore. TPM allows for increased penetration depth, localized excitation, reduced photo-damage and prolonged observation time [116,117], and thus is superior to conventional one-photon excitation and confocal microscopy. Recently, two-photon fluorescent probes that could selectively image biothiols have been developed. For example, ASS (64, Figure 13) is a two-photon fluorescent probe derived from 2-methylamino-6-acetylnaphthalene. In the presence of thiols the disulfide bond is cleaved, leading to intramolecular cyclization and release of the fluorophore, which could be detected by TPM. Fluorescence imaging using this probe in live cells and rat tissue in a depth of 120 μm has been demonstrated [118]. Based on an analogous concept, SSH-Mito (65, Figure 13) bearing a triphenylphosphonium (TPP) moiety for mitochondrial targeting was developed recently [119]. TPP has been demonstrated to specifically transport cargo molecules to mitochondria due to electrostatic interactions of the positively charged phosphine and the negative potential across the inner mitochondrial membrane [120].
Recently, a thiol probe aimed at targeting liver cells was described [121]. This probe (66, Figure 13) contains a galactose subunit and a disulfide-linked naphthalimide. The terminal galactose residue is recognized by the asialoglycoprotein receptor (ASGP-R), which is expressed on the plasma membrane of mammalian hepatocytes. This directs the probe selectively to hepatocytes. In the presence of biothiols, such as GSH, thioredoxin (Trx), Hcy and Cys in PBS buffer, the disulfide bond is cleaved, followed by an intramolecular cyclization. This uncaps the amino group on the naphthalimide moiety and leads to a substantial increase in fluorescence intensity at 540 nm. A 10-fold fluorescence increase was observed for thiols at 5.0 mM when 1.0 μM of probe 66 was used in PBS buffer. The target specificity was confirmed by cellular imaging experiments, in which fluorescence was observed only in HepG2 cells, but not in other non-hepatocytes such as C2C12, HaCaT, and N2a cells. The results of tissue imaging experiments using male Sprague-Dawley rats have also confirmed the specificity.
Probes for the detection of thiols based on gold nanoparticles (AuNPs) have also been reported [122]. Specifically, disulfide linked AuNP clusters (68, Figure 14) are formed by treating AuNPs coated with dithiobis(succinimidylpropinate) (DSP, 67).
The formation of AuNP clusters was confirmed by localized surface plasmon resonance (LSPR) spectroscopy and transmission electron microscopy (TEM). When the AuNP clusters are exposed to small molecule thiols, the GSSG disulfide linkage can be readily reduced, resulting in the reversal of the cluster formation to form 69 and 70. This is characterized by a significant blue shift (610 nm to 520 nm) of the LSPR spectra. This probe is especially useful for the detection of low molecular weight thiols because by controlling the size of the linker and the NPs, a steric environment is created for the easy access of low molecular weight thiols but not larger molecules. Different responses were obtained for various thiols with the best detection limit (to NaSH) being in the low micromolar range.
Glutathione peroxidase (GPx) is an antioxidant enzyme that catalyzes the oxidation of GSH to GSSG by H2O2. It has a selenocysteine in its active site, forming a catalytic triad with tryptophan (Trp) and glutamine (Gln). The catalytic mechanism involves the formation of selenyl sulfide as an intermediate [123]. An anti-inflammatory drug Ebselen (71, 2-phenyl-1,2-benzisoselenazol-3(2H)-one, PZ 51 or DR3305, Figure 15) [124–126], bearing a Se-N bond, could also catalyze the reduction of H2O2 by GSH with a similar mechanism. Based on this well-known reaction between thiols and the Se-N bond, Tang and co-workers developed a fluorescent probe for thiols [127]. Probe 72 is only weakly fluorescent in aqueous solution. After incubation at 25 °C for 30 min with GSH, the Se-N bond is cleaved, leading to the formation of rhodamine 6G and the concomitant significant increase of fluorescence (λex 525 nm, λem 550 nm). The reaction is fast and quantitative with a linear correlation curve between fluorescence intensity and thiol concentrations. The detection limit is lower than 100 nM in PBS buffer. This probe was used to sense the difference in thiol concentrations in normal human liver cell line HL-7702 and human hepatoma cell line HepG2.
Another example based on Se-N bond cleavage has also been reported [128]. This probe (73, Rh-Se-2, Figure 15) reacts with thiols at 25 °C in PBS buffer with the maximum fluorescence at 522 nm observed after only 5-10 min. This method also provides a linear calibration curve with a detection limit lower than 100 nM. Cellular imaging using this probe has also been tested in HL-7702 and HepG2 cells.
2.4. Sensors Showing Selectivity among Thiols
Almost all the labeling and detection methods described thus far are based on the reactivity of the sulfhydryl group itself, which is present in all thiols, such as Cys, Hcy, GSH, and Cys residues in proteins. Therefore, there is no reason to expect more than limited selectivity among various thiols for idiosynchratic reasons. However, considering the difference in the biological roles of each thiol, the quantification of individual thiols instead of total thiols, is very important. For example, total Hcy concentration (tHcy) in healthy plasma is lower than 15 μM [21], while the Cys concentration is 20–30 times that of tHcy. Because tHcy is related to many diseases, its selective determination is necessary. Differentiation among various thiols in current methods is mainly based on chromatographic separation [56,78,129]. Specifically, thiols are derivatized with labeling agents such as mBrB or CMQT and analyzed by HPLC or CE equipped with a DAD or fluorescence detector. Data could then be compared with a calibration curve. This process is instrument-dependent and time-consuming. Therefore, the need for the development of chemoprobes, which allow for selectivity among thiols, is urgent.
1,1′-Thiocarbonyldiimidazole (TCDI, 74, Figure 16) [130,131] has been reported to differentiate Cys, Hcy and PenA from other non-aminothiols. Through dual nucleophilic substitution steps accompanied by releasing two molecules of imidazole, TCDI selectively reacts with aminothiols such as Cys, Hcy, PenA and cycteinylglycine. The five-membered ring adduct (75, TTCA) formed from the reaction with Cys has a maximum UV absorption at 272 nm. The six-membered ring adduct T3CA (76) formed from Hcy absorbs at 283 nm. The reaction completes within 20 min at 37 °C under basic aqueous conditions. HPLC can then be used to quantitatively analyze those adducts.
In 2003, the Strongin group reported the synthesis and evaluation of a bisaldehyde based fluorescent sensor 77 (Figure 17) for thiol detection [132]. The formation of a thiazolidine (78) between aldehyde and Cys led to a 25 nm red shift in the absorption spectrum (from 480 to 505 nm) and a decrease in fluorescence intensity.
This sensor responds to Hcy as well, giving rise to a six-membered ring product 79 with very similar spectral changes (about 25 nm of red shift in the absorption spectrum and a decrease in fluorescence intensity). The same group has also synthesized the monoaldehyde 80 [133]. Interestingly, the results have shown that both sensors exhibited moderate selectivity to Cys over Hcy. This might be due to the favored formation of a five-membered ring structure. In 2004, the Barbas group reported another probe 81 bearing an aldehyde group [134]. This probe showed a fluorescence increase after condensation with Cys. Although the authors did not mention the study of homocysteine, this sensor showed a significantly higher reaction rate with Cys compared to GSH. Based on the same design strategy, the Hong group described in 2008 a fluorescent probe 82 bearing an aldehyde moiety on the coumarin dye. This probe was reported to show about 3-fold selectivity toward Cys over Hcy with no response to GSH [135]. In addition, two photon fluorescent probes have been reported recently bearing an aldehyde moiety, showing selectivity for cysteine [136].
In 2005, the Strongin group reported the selective detection of Cys using a commercially available aldehyde (Figure 18, 83) [133]. This probe shows exclusive selectivity to Cys over Hcy, forming an adduct 84, which is demonstrated by a decrease in absorbance at 400 nm. The addition of Hcy did not lead to any obvious change in absorption. This Cys-selective sensor is very useful, because it could be used to determine background concentration of Cys in the biological sample. Then the concentration of Hcy may be calculated by subtracting the concentration of Cys from the total concentration of biological amino thiols, which normally includes Cys and Hcy. One way of achieving such selective detection is to use excessive amount of 83 to selectively pre-saturate the Cys in the sample. This is then followed by a direct detection of Hcy using probe 77. This turned out to be a very effective method. After pre-treatment of 84, the absorption decrease of 77 was only due to the concentration of Hcy.
In 2007, a phosphorescent probe was reported for the detection of biothiols. A homocysteine-selective iridium(III)-based sensor 85 was synthesized [137]. This probe reacts with Cys to form 86, showing a color change from orange to green and an increase in phosphorescence (λex 360 nm, λem 525 nm) in DMSO/HEPES buffer. It also exhibited a 19-fold selectivity for Hcy over Cys. This was the first luminescent chemosensor with high selectivity for Hcy over other biothiols (including Cys and GSH).
In addition to the sensors based on aldehydes, another approach for selective detection of Hcy was described by the Strongin group [133,138]. Thiyl radicals (87, 89 and 90, Figure 19) are the intermediates in the oxidation of thiols such as Cys, Hcy and GSH. They can either lead to disulfide formation, or a “repairing” process, which results in the formation of a carbon centered radical (88). The intramolecular formation of the carbon centered radical is favored for Hcy, because it goes through a 5-membered ring transition state, and is less favored for Cys and GSH. In the presence of a compound that develops a colored radical species, the carbon-centered radical can be detected. Methyl Viologen (91) is a good candidate. After gently refluxing for 5 min, the mixture of 91 and Hcy in Tris buffer showed a blue color through formation of 92, demonstrated by absorption at 398 and 605 nm. The same response was not observed for either Cys or GSH. This method was also applied to the selective detection of Hcy in human plasma [139]. Figure 19(b) shows an optimization of the method for total homocysteine in human serum standard calibrators using 91 (unpublished results). Fluorone black (93) can also be used for Hcy detection using this mechanism. This is demonstrated by an absorption increase at 510 nm and emission increase at 540 nm in 70% MeOH in H2O.
The most recent work on the selective detection of Hcy and Cys reported by the Strongin group takes advantage of a Michael addition-lactam formation cascade [140]. The probe (95, Figure 20) undergoes a Michael addition with the sulfhydryl group to generate conjugates 96. Then an intramolecular nucleophilic attack from the amino group leads to the formation of lactams 97, release of a HMBT fluorophore 98, and a subsequent fluorescence change. The difference in reaction rates with Cys and Hcy provides an option for discrimination of Cys and Hcy. Very recently, Yoon lab has also reported a cyanine-based NIR emitting fluorescent probe showing selectivity for cysteine using the same strategy [141].
3. Probes for Aromatic Thiols
Aromatic thiols, such as thiophenols, are widely used in the production of pharmaceutical intermediates, polymers and pesticides [142]. Exposure to thiophenol may lead to damage to the central nervous system, kidney and liver [143]. Due to conjugation effect with the phenyl ring, the sulfhydryl group of thiophenol has a pKa much lower than that of alkyl thiols. Thus it is conceivable that fluorescent labeling agents for thiols would also label thiophenols, and probably more efficiently. The significant difference in the nucleophilicity under physiological pH conditions has been successfully used in the selective detection of thiophenols. The Wang group has reported two fluorescent chemoprobes for thiophenols [144,145]. Compound 99 is based on a nitrobenzofuranzan fluorophore bearing a DNBS moiety (Figure 21), which shows no fluorescence before the addition of thiophenol. When thiophenol is added, the sulfhydryl group attacks the dinitrophenyl and releases the free fluorophore NBD (100), which leads to a dramatic fluorescence increase at an emission wavelength of 550 nm. A similar design also imparted the same selectivity to compound 101, which releases a benzoxazole fluorophone 102 after the addition of thiophenol. Both probes have a detection limit in the low micromolar range.
Besides the strategy of using DNBS moiety, another probe (103, Figure 21, ΦFL 0.006) based on the nucleophilic attack on the dinitrophenyl group was developed by Lin and co-workers [146]. The probe releases a fluorescent coumarin derivative 104 (λex 461 nm, λem 494 nm, ΦFL 0.50) upon reaction with thiophenol, showing good selectivity for thiolphenol over 2-mercaptoethanol. The detection limit was reported to be 1.8 nM for benzenethiols. This method has been tested in water, soil and cell cultures.
4. Probes for H2S
H2S has been recognized as an endogenously produced gasotransmitter. It plays regulatory roles in multiple systems, such as the cardiovascular [38–41] and the central nervous system (CNS) [42,43]. It also shows therapeutic effects in heart diseases [39,45]. Due to the newly recognized biological significance of H2S, more and more research interest has been focused on the molecular mechanisms of its biological functions and related therapeutic applications. However, H2S is very unstable because of its high volatility and high propensity to be oxidized under physiological conditions. Such properties have made the accurate measurement of hydrogen sulfide a difficult task. A number of methods have been reported for the detection of H2S [147,148]. These methods include gas chromatography (GC) [149–152], HPLC [153], and electrochemical methods, which mostly rely on sulfide ion selective electrodes [154,155] or polarographic methods [156]. Despite the availability of numerous detection methods, literature reported hydrogen sulfide concentrations vary significantly among publications, ranging from high micromolar [48,157,158] to low nanomolar [159]. Part of the reason could be the difference in sample preparation and intrinsic fluctuation of hydrogen sulfide concentrations. Other reasons might be due to the lack of methods for fast and selective detection. In any case, it is generally agreed that the development of new approaches is necessary not only for the selective and instantaneous detection, but also for intracellular imaging.
4.1. Probes for H2S based on Nucleophilic Cyclization Reactions
Hydrogen sulfide (H2S) dissociates in aqueous solutions in two sequential steps to HS− and S2− with a pKa1 of 6.9 and pKa2 of 12, which means over 75% of H2S exists in the anionic state at physiological pH. Sulfide is a stronger nucleophile than commonly encountered anions such as chloride and hydroxide under physiological conditions. This provides the possibility of selective detection of sulfide amongst various anions. One of the classical methods for the quantification of sulfide is the methylene blue method [160]. Sulfide reacts with N,N-dimethyl-p-phenylenediamine (105, Figure 22) in the presence of Zn(OAc)2 and FeCl3 under acidic conditions, yielding methylene blue (106), which absorbs strongly at 670 nm.
Samples containing H2S are often preserved by addition of Zn2+ to trap H2S in the form of ZnS. During analysis, solutions of compound 105 and FeCl3 in HCl are added into the sample. A blue color, which could be measured by a UV-vis spectrophotometer, usually develops in minutes [161]. This method shows both good selectivity and sensitivity with a nanomolar detection limit. It has been approved by the US Environmental Protection Agency (EPA) as a standard method for sulfide quantitation and has been utilized for hydrogen sulfide determination in many studies [153,157,162]. However, because methylene blue tends to form dimers and trimers [163] at concentrations over 10 μM, a linear calibration curve could not be obtained for sulfide at 10 μM or higher concentrations. In addition, the use of corrosive reagents and the non-instantaneous nature of this method have also limited the application of this method.
Being the simplest molecule in the thiol family, hydrogen sulfide can undergo two deprotonation steps. In other words, it can undergo two nucleophilic reactions. This distinguishes it from other thiols and provides a strategy for sulfide detection with high selectivity. The Xian group reported a fluorescent probe for H2S, which was designed based on this principle [164]. As shown in Figure 23, compound 107 reacts with sulfide, releasing mercaptopyridine 108 to form an intermediate 109, which undergoes a cyclization in situ and releases fluorophore 110 and benzodithiolone 111. This probe is very selective for H2S in aqueous solution (PBS/acetonitrile 9:1) among thiols such as Cys and GSH, and gives a linear correlation to sulfide concentrations with a detection limit of low micromolar concentrations. Fluorescent imaging using this probe and exogenous H2S has been studied in COS7 cells.
Another strategy reported by the He group uses a Michael addition reaction followed by cyclization [165]. In this study, two fluorescent probes, SFP-1 (112) and SFP-2 (114, Figure 24) were synthesized. These probes bear an α, β-unsaturated ester group at the ortho position of a benzaldehyde, which is linked to a fluorophore. The nucleophilic attack by sulfide on the formyl group yields hemithioacetals, which positions the sulfhydryl group for the following Michael addition to form the trapped thioacetal 113 and 115, in which the PET effect is interrupted and the fluorescence is recovered.
Both SFP-1 and SFP-2 show 50–100 fold selectivity for sulfide over other thiols including β-mercaptoethanol, Cys and GSH. The detection limit is about 5–10 μM with a S/N ratio of 3:1. SFP-2 was used in the in vivo imaging of endogenously generated H2S triggered by the addition of GSH and Cys in Hela cells. Along a similar line, probes 116 and 117 have been reported recently by the Xian group. These probes are based on a Michael addition-cyclization reaction [166]. In probes 116 and 117, the Michael acceptor is activated by two electron withdrawing groups. After incubating the probes (5 μM) with 100 μM sulfide for 30 min in phosphate buffer, Michael addition-cyclization takes place to release the fluorophore 110 to form thiolactones 118 and 119, leading to 11 (116) or 160 (117)-fold fluorescence increase (λex = 465 nm, λem = 510 nm), respectively. Imaging of exogenous H2S was performed in COS7 cells.
Furthermore, chemoprobes developed for other thiols could also be used for H2S detection. For example, compound 14 (Figure 3), reported by Maeda for the fluorescent detection of thiols, has also been used for the fluorescent detection of H2S [167]. Of course, in such a case, selectivity is an issue.
4.2. Probes for H2S Based on Reduction Reactions
Sulfide is a fairly strong reducing agent. This is another chemical property of H2S that can be used in probe design. Azides are known to be reduced by sulfide anion [168]. However, it is not until very recently that this reaction was utilized in the selective detection of sulfide independently by the labs of Chang and Wang. Specifically, the Chang group reported the synthesis and evaluation of two fluorescent probes (SF1, 120 and SF2, 121, Figure 25) based on this strategy [169]. Both probes bear a fluorescein moiety attached directly to an azido group, which is easily reduced to an amino group by hydrogen sulfide, resulting in a significant increase in fluorescence. The selectivity of these two probes was demonstrated among various RSS (reactive sulfur species), RNS (reactive nitrogen species) and ROS (reactive oxygen species) including GSH, Cys, Na2SO3, NO, H2O2 and O2−. The probes were also evaluated for in vivo cell imaging in HEK293T cells using exogenous H2S [169].
At the same time, another fluorescent probe for H2S was reported by the Wang group [170]. It is based on the reduction of the azido group attached to a sulfonylnaphthalene fluorophore, the dansyl dye (DNS-Az, 122, Figure 25). This probe showed a dramatic fluorescence increase (λex 340 nm, λem 517 nm, over 40 fold) upon addition of 25 μM of sulfide into phosphate buffer with Tween-20. The probe was evaluated in different aqueous media, including phosphate buffer and commercial bovine serum. Good selectivity of DNS-Az for H2S was observed among various anions (>50 fold) and other reducing agents, including Cys (60-fold) and thiophenol (4-7.5 fold). Another important advantage of this probe over other reported sulfide probes is the fast response [171]. The reduction reaction is complete in seconds in bovine serum at room temperature. This is among the fastest H2S fluorescent probes reported so far. Considering the volatility and low stability of H2S, an “instant” probe is very important because it allows for accurate detection of sulfide concentration without sample pre-treatment such as precipitation. The fluorescence intensity could be recorded on a fluorometer or a multi-label counter. Excellent linear calibration curves were obtained for H2S in different solvents, including buffer and bovine serum. The probe was used for the measurement of H2S concentration in blood using the C57BL6/J mouse model. Specifically, a stock solution of DNS-Az was added to the blood, followed by mixing and reaction at room temperature for 5 min. Blood plasma was then obtained by centrifugation and fluorescence intensity was analyzed in a 96-well plate and multi-label counter. The zero point was obtained by trapping sulfide with ZnCl2 and the calibration curve was obtained by using an internal standard method. The blood H2S concentration obtained (31.9 ± 9.4 μM) is very similar to most previously reported numbers.
Besides DNS-Az, compound 123 has also been synthesized by the Wang group (unpublished results). This probe is composed of an azido group and the naphthalimide fluorophore moiety. The synthesis of this compound consists of two steps from 4-bromo-1,8-naphthalic anhydride through amide formation with n-butylamine and substitution of the bromo group by azido using NaN3. This compound is sensitive toward sulfide with a 100-fold fluorescence increase (λex 420 nm, λem 530 nm) in response to the addition of 100 μM of H2S in a mixed solvent of 1:1 phosphate buffer/acetonitrile. The reaction is complete in 30 s in acetonitrile and 20 min in phosphate buffer/acetonitrile at room temperature. However, the lower solubility and stability of compound 123 compared with 122 may limit its application. Further structure optimization is ongoing to obtain improved fluorescent probes for H2S. Two other naphthalimide probes (compounds 124 and 125, Figure 25) have also been reported recently [172]. Probe 124 is based on the reduction of an azido group and probe 125 is based on the reduction of a nitro group by sulfide. Both probes can be converted to the corresponding 4-aminonaphthalimide (λex 432 nm, λem 542 nm) by reduction. They were found to show more than 10-fold selectivity toward H2S over other reactive oxygen, nitrogen, and sulfur species. In PIPES buffer, the reduction reaction is complete in 90 min (124) and 45 min (125), respectively, upon treatment with 0.5 mM of sulfide. Fluorescent imaging of exogenous H2S using these two probes were demonstrated in Hela cells.
Recently, a NIR fluorescent probe (126, Figure 25) for H2S was reported by Han and co-workers [173]. When the azido group is reduced by H2S, a 50-nm red shift from 610 nm to 660 nm in absorption and a 40 nm red shift from 710 nm to 750 nm (λex 625 nm) in emission were observed in HEPES buffer. The reaction took 20 min to complete with a micromolar detection limit. This probe was tested in RAW 264.7 macrophage cells for imaging exogenously added H2S.
Due to its high selectivity and reactivity, redox-based strategy is becoming very useful in the detection of H2S. The level of activities is very high in this area. Recently, several other probes have been reported based on this strategy [174–177]. These probes include two-photon fluorescent probes and protein-based probes.
4.3. Probes for H2S Based on Metal Sulfide Formation
The selective interaction between sulfide and copper has been utilized in the detection of sulfide. A fluorescein derivative (127, Figure 26) with a dipicolylamine (DPA) binding site for Cu2+ was developed by the Chang group [178]. The fluorescence intensity of the DPA conjugate (λex 470 nm, λem 517 nm) was almost completely quenched by Cu2+. However, when exposed to sulfide in 10 mM HEPES buffer at pH 7.0, Cu2+ was extracted, leading to the formation of CuS and the free fluorophore ligand, and subsequently a significant fluorescence increase. Selectivity for sulfide was demonstrated among various anions. A similar strategy was recently reported by Nagano and co-workers in the development of a fluorescent probe for hydrogen sulfide (Hsip-1, 128, Figure 26) [179]. Instead of the DPA ligand, cyclen (1,4,7,10-tetraazacyclododecane) was used as the ligand for copper complexation. Hsip-1 exhibited significant fluorescence enhancement (λex 491 nm, λem 516 nm) when exposed to sulfide in HEPES buffer at pH 7.4. Compared with the DPA ligand probe 127, Hsip-1 showed about 10-fold selectivity for sulfide over GSH. Visualization of H2S using Hsip1 was demonstrated in live Hela cells. Recently, another fluorescein based probe (129, Figure 26) based on a similar strategy was described [180]. The fluorescence is almost completely quenched by copper. When exposed to sulfide in HEPES/CH3CN (pH 7.0, 6: 4, v/v), fluorescence is restored (λex 494 nm, λem 523 nm). Although the selectivity was only 1.6 fold over Cys, the probe was reported to be reversible (up to 10 cycles) upon addition of Cu(II)/H2S. This could be used as an on-off-on probe, which could have potential applications for continuous monitoring. Recently, additional probes have been developed based on metal sulfide formation [181–183].
The selective interaction of sulfur with gold was also utilized in the detection of H2S and other thiols. Lewis and co-workers have developed a mercaptan gas detector using amine capped Au nanocrystal films (Figure 27) [184]. The film consists of dodecylamine-capped, 6-7 nm diameter Au nanocrystals. When exposed to gaseous thiols such as CH3SH, the amine cap is displaced by thiols, resulting in an electrical resistance drop due to the reduced distance amongst Au cores. The slope of the resistance curve shows a linear response to CH3SH between 0.153 and 1.53 ppm in air with the detection limit of 1.7 ppb. Although the film detector does not show a comparable response to larger molecules such as octanethiol, it still provides a very sensitive detection of small molecular gaseous mercaptans such as CH3SH and H2S.
Because of the biological significance of H2S, the value for the quantitative detection of this small molecule has been recognized both for diagnostic applications and in basic research. Fluorescent probes for H2S based on nucleophilic cyclization, reduction and metal sulfide formation have been reported. Quantitation of H2S was achieved in mouse blood samples with DNS-Az (122). Fluorescence imaging of endogenous and exogenous H2S was demonstrated by various groups using different cell lines. AuNPs was also used in the detection of mercaptan gas including H2S and CH3SH. These probes provide new chemical tools for biomedical researchers in their investigation of the functions and properties of H2S in biological systems.
5. Conclusions
With the widely recognized importance of biological thiols, there has been increasing interest in developing new detection methods. Numerous fluorescent probes have been reported so far for the selective detection of thiols. The probes for biological amino-thiols include those based on nucleophilic substitution, Michael addition, and disulfide/Se-N bond cleavage. Among these probes, nucleophilic substitution-based probes (such as mBrB) are commonly used in the fluorometric/colorimetric labeling of thiols for HPLC quantitation. Disulfide cleavage-based probes (such as Ellman's reagent) are frequently used for estimation of total sulfhydryl groups in a protein sample. Methods based on other probes are still in development. Fluorescent probes for H2S include those based on nucleophilic cyclization, reduction, and metal sulfide formation. These probes are emerging as important detection tools of H2S. Generally speaking, excellent methods are available for the quantitative and selective concentration determinations of various thiols in solution or in sera. Still needed are methods that are fast enough to address the issues of rapid fluctuations of intracellular concentrations or in vivo, especially for hydrogen sulfide. Recently, there is growing interest in the intracellular imaging of various thiols. Fluorescent probes are needed that are: (1) selective, (2) fast response, (3) cell permeable, and (4) of long wavelength. Among these, especially important is the availability of probes that are fast in response because reaction times on the scale of minutes run the risk of perturbing the thiol homeostasis mechanisms in such a way that the amount of probe consumed (and thus fluorescent intensity) may not be correlated with the true concentration at a given time. However, based on the level of research interest in this area, there are good reasons to be optimistic that excellent solutions will be found in the not too distant future.
Acknowledgments
We gratefully acknowledge the financial support of this work in the authors' lab by the Molecular Basis of Disease Program at GSU through fellowships to HJP, WXC and YFC, the GSU University fellowship program through a fellowship to HJP, and the National Institutes of Health (GM084933 to BW).
References
- Koval, I.V. Reactions of thiols. Russ. J. Org. Chem. 2007, 43, 319–346. [Google Scholar]
- Meister, A.; Anderson, M.E. Glutathione. Ann. Rev. Biochem. 1983, 52, 711–760. [Google Scholar]
- Selhub, J. Homocysteine metabolism. Annu. Rev. Nutr. 1999, 19, 217–246. [Google Scholar]
- Forman, H.J.; Zhang, H.Q.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Aspects Med. 2009, 30, 1–12. [Google Scholar]
- Cleland, W.W. Dithiothreitol, a new protective reagent for SH groups. Biochemistry 1964, 3, 480–482. [Google Scholar]
- Lipton, S.A.; Choi, Y.B.; Takahashi, H.; Zhang, D.X.; Li, W.Z.; Godzik, A.; Bankston, L.A. Cysteine regulation of protein function—As exemplified by NMDA-receptor modulation. Trends Neurosci. 2002, 25, 474–480. [Google Scholar]
- Petsko, G.A.; Ringe, D. Protein Structure and Function: From Sequence to Consequence; New Science Press Ltd.: London, UK, 2004. [Google Scholar]
- Meister, A. Glutathione metabolism and its selective modification. J. Biol. Chem. 1988, 263, 17205–17208. [Google Scholar]
- Conklin, K.A. Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness. Integr. Cancer. Ther. 2004, 3, 294–300. [Google Scholar]
- Peisach, J.; Blumberg, W.E. A mechanism for the action of penicillamine in the treatment of Wilson's disease. Mol. Pharmacol. 1969, 5, 200–209. [Google Scholar]
- Zhang, J.G.; Lindup, W.E. Tiopronin protects against the nephrotoxicity of cisplatin in rat renal cortical slices in vitro. Toxicol. Appl. Pharm. 1996, 141, 425–433. [Google Scholar]
- Boehning, D.; Snyder, S.H. Novel neural modulators. Annu. Rev. Neurosci. 2003, 26, 105–131. [Google Scholar]
- Martelli, A.; Testai, L.; Breschi, M.C.; Blandizzi, C.; Virdis, A.; Teddei, S.; Calderone, V. Hydrogen sulfide: novel oppotunity for drug discovery. Med. Res. Rev. 2011. [Google Scholar] [CrossRef]
- Szabo, C. Hydrogen sulphide and its therapeutic potential. Nat. Rev. Drug Discov. 2007, 6, 917–935. [Google Scholar]
- Wang, R. Two's company, three's a crowd: Can H2S be the third endogenous gaseous transmitter? FASEB J. 2002, 16, 1792–1798. [Google Scholar]
- Kluijtmans, L.A.J.; van den Heuvel, L.P.W.J.; Boers, G.H.J.; Frosst, P.; Stevens, E.M.B.; vanOost, B.A.; den Heijer, M.; Trijbels, F.J.M.; Rozen, R.; Blom, H.J. Molecular genetic analysis in mild hyperhomocysteinemia: A common mutation in the methylenetetrahydrofolate reductase gene is a genetic risk factor for cardiovascular disease. Am. J. Hum. Genet. 1996, 58, 35–41. [Google Scholar]
- Calonge, M.T.; Gasparini, P.; Chillaron, J.; Chillon, M.; Gallucci, M.; Rousaud, F.; Zelante, L.; Testar, X.; Dallapiccola, B.; Disilverio, F.; et al. Cystinuria caused by mutations in rbat, a gene involved in the transport of cystine. Nat. Genet. 1994, 6, 420–425. [Google Scholar]
- Lowicka, E.; Beltowski, J. Hydrogen sulfide (H2S)—The third gas of interest for pharmacologists. Pharmacol. Rep. 2007, 59, 4–24. [Google Scholar]
- Wu, G.Y.; Fang, Y.Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar]
- Droge, W.; Eck, H.P.; Mihm, S. Hiv-Induced Cysteine Deficiency and T-Cell Dysfunction—A rationale for treatment with N-Acetylcysteine. Immunol. Today 1992, 13, 211–214. [Google Scholar]
- Refsum, H.; Smith, A.D.; Ueland, P.M.; Nexo, E.; Clarke, R.; McPartlin, J.; Johnston, C.; Engbaek, F.; Schneede, J.; McPartlin, C.; et al. Facts and recommendations about total homocysteine determinations: An expert opinion. Clin. Chem. 2004, 50, 3–32. [Google Scholar]
- Levy, H.L.; Kraus, J.P.; Mudd, S.H. Disorders of Transsulfuration. In The Metabolic and Molecular Bases of Inherited Disease; Valle, D., Beaudet, A.L., Vogelstein, B., Kinzler, K.W., Antonarakis, S.E., Ballabio, A., Scriver, C.R., William, S.S., Childs, B., Eds.; McGraw-Hill: New York, NY, USA, 1995. [Google Scholar]
- Chadefaux, B.; Ceballos, I.; Hamet, M.; Coude, M.; Poissonnier, M.; Kamoun, P.; Allard, D. Is absence of atheroma in Down syndrome due to decreased homocysteine levels? Lancet 1988, 2, 741. [Google Scholar]
- James, S.J.; Pogribna, M.; Melnyk, S.; Pogribny, I.; Chango, A.; Yi, P. Homocysteine metabolism in children with Down syndrome: In vitro modulation. Am. J. Hum. Genet. 2001, 69, 88–95. [Google Scholar]
- Kang, S.S.; Wong, P.W.K.; Norusis, M. Homocysteinemia due to folate deficiency. Metabolism 1987, 36, 458–462. [Google Scholar]
- Klee, G.G. Cobalamin and folate evaluation: Measurement of methylmalonic acid and homocysteine vs vitamin B-12 and folate. Clin. Chem. 2000, 46, 1277–1283. [Google Scholar]
- Ubbink, J.B.; van der Merwe, A.; Delport, R.; Allen, R.H.; Stabler, S.P.; Riezler, R.; Vermaak, W.J.H. The effect of a subnormal vitamin B-6 status on homocysteine metabolism. J. Clin. Invest. 1996, 98, 177–184. [Google Scholar]
- Medina, M.A.; Amores-Sanchez, M.I. Homocysteine: an emergent cardiovascular risk factor? Eur. J. Clin. Invest. 2000, 30, 754–762. [Google Scholar]
- Bostom, A.G.; Culleton, B.F. Hyperhomocysteinemia in chronic renal disease. J. Am. Soc. Nephrol. 1999, 10, 891–900. [Google Scholar]
- Jones, D.P.; Carlson, J.L.; Samiec, P.S.; Sternberg, P.; Mody, V.C.; Reed, R.L.; Brown, L.A.S. Glutathione measurement in human plasma Evaluation of sample collection, storage and derivatization conditions for analysis of dansyl derivatives by HPLC. Clin. Chim. Acta 1998, 275, 175–184. [Google Scholar]
- Akerboom, T.P.M.; Bilzer, M.; Sies, H. Relationship of biliary glutathione disulfide efflux and intra cellular glutathione disulfide content in perfused rat liver. J. Biol. Chem. 1982, 257, 4248–4252. [Google Scholar]
- Griffith, O.W. Biologic and pharmacologic regulation of mammalian glutathione synthesis. Free Radic. Bio. Med. 1999, 27, 922–935. [Google Scholar]
- Rahman, I.; Biswas, S.K. Environmental toxicity, redox signaling and lung inflammation: The role of glutathione. Mol. Aspects Med. 2009, 30, 60–76. [Google Scholar]
- Kruger, W.D.; Chen, X.L.; Jhee, K.H. Production of the neuromodulator H2S by cystathionine beta-synthase via the condensation of cysteine and homocysteine. J. Biol. Chem. 2004, 279, 52082–52086. [Google Scholar]
- Ishii, I.; Akahoshi, N.; Yu, X.N.; Kobayashi, Y.; Namekata, K.; Komaki, G.; Kimura, H. Murine cystathionine gamma-lyase: Complete cDNA and genomic sequences, promoter activity, tissue distribution and developmental expression. Biochem. J. 2004, 381, 113–123. [Google Scholar]
- Shibuya, N.; Mikami, Y.; Kimura, Y.; Nagahara, N.; Kimura, H. Vascular endothelium expresses 3-mercaptopyruvate sulfurtransferase and produces hydrogen sulfide. J. Biochem. 2009, 146, 623–626. [Google Scholar]
- Tanizawa, K. Production of H2S by 3-mercaptopyruvate sulphurtransferase. J. Biochem. 2011, 149, 357–359. [Google Scholar]
- Calvert, J.W.; Jha, S.; Gundewar, S.; Elrod, J.W.; Ramachandran, A.; Pattillo, C.B.; Kevil, C.G.; Lefer, D.J. Hydrogen sulfide mediates cardioprotection through Nrf2 signaling. Circ. Res. 2009, 105, 365–U105. [Google Scholar]
- Calvert, J.W.; Elston, M.; Nicholson, C.K.; Gundewar, S.; Jha, S.; Elrod, J.W.; Ramachandran, A.; Lefer, D.J. Genetic and pharmacologic hydrogen sulfide therapy attenuates ischemia-induced heart failure in mice. Circulation 2010, 122, 11–19. [Google Scholar]
- Zhao, W.M.; Zhang, J.; Lu, Y.J.; Wang, R. The vasorelaxant effect of H2S as a novel endogenous gaseous K-ATP channel opener. EMBO J. 2001, 20, 6008–6016. [Google Scholar]
- Lefer, D.J. A new gaseous signaling molecule emerges: Cardioprotective role of hydrogen sulfide. P. Natl. Acad. Sci. USA 2007, 104, 17907–17908. [Google Scholar]
- Abe, K.; Kimura, H. The possible role of hydrogen sulfide as an endogenous neuromodulator. J. Neurosci. 1996, 16, 1066–1071. [Google Scholar]
- Kimura, H. Hydrogen sulfide as a neuromodulator. Mol. Neurobiol. 2002, 26, 13–19. [Google Scholar]
- Moore, P.K.; Bhatia, M.; Moochhala, S. Hydrogen sulfide: from the smell of the past to the mediator of the future? Trends Pharmacol. Sci. 2003, 24, 609–611. [Google Scholar]
- Elrod, J.W.; Calvert, J.W.; Morrison, J.; Doeller, J.E.; Kraus, D.W.; Tao, L.; Jiao, X.Y.; Scalia, R.; Kiss, L.; Szabo, C.; et al. Hydrogen sulfide attenuates myocardial ischemia-reperfusion injury by preservation of mitochondrial function. Proc. Natl. Acad. Sci. USA 2007, 104, 15560–15565. [Google Scholar]
- Sen, U.; Mishra, P.K.; Tyagi, N.; Tyagi, S.C. Homocysteine to hydrogen sulfide or hypertension. Cell Biochem. Biophys. 2010, 57, 49–58. [Google Scholar]
- Kamoun, P.; Belardinelli, M.-C.; Chabli, A.; Lallouchi, K. Chadefaux-Vekemans, B. Endogenous hydrogen sulfide overproduction in down syndrom. Am. J. Med. Genet. 2003, 116A, 310–311. [Google Scholar]
- Chen, Y.-H.; Yao, W.-Z.; Geng, B.; Ding, Y.-L.; Lu, M.; Zhao, M.-W.; Tang, C.-S. Endogenous hydrogen sulfide in patients with COPD. Chest 2005, 128, 3205–3211. [Google Scholar]
- Aitken, S.M.; Kirsch, J.F. Kinetics of the yeast cystathionine beta-synthase forward and reverse reactions: Continuous assays and the equilibrium constant for the reaction. Biochemistry 2003, 42, 571–578. [Google Scholar]
- Zhu, J.G.; Dizin, E.; Hu, X.B.; Wavreille, A.S.; Park, J.; Pei, D.H. S-ribosylhomocysteinase (LuxS) is a mononuclear iron protein. Biochemistry 2003, 42, 4717–4726. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Feather-Stone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar]
- Brown, K.S.; Kingsbury, W.D.; Hall, N.M.; Dunn, G.L.; Gilvarg, C. Determination of carboxypeptidase A using N-acetyl-phenylalanyl-3-thiaphenylalanine as substrate application to a direct serum assay. Anal. Biochem. 1987, 161, 219–225. [Google Scholar]
- Chen, X.; Zhou, Y.; Peng, X.J.; Yoon, J. Fluorescent and colorimetric probes for detection of thiols. Chem. Soc. Rev. 2010, 39, 2120–2135. [Google Scholar]
- Yin, L.L.; Chen, Z.Z.; Tong, L.L.; Xu, K.H.; Tang, B. Progress on fluorescent probes for thiols. Chinese J. Anal. Chem. 2009, 37, 1073–1081. [Google Scholar]
- Toyo'oka, T. Review Recent advances in separation and detection methods for thiol compounds in biological samples. J. Chromatogr. B. 2009, 877, 3318–3330. [Google Scholar]
- Nekrassova, O.; Lawrence, N.S.; Compton, R.G. Analytical determination of homocysteine: A review. Talanta 2003, 60, 1085–1095. [Google Scholar]
- Sakhi, A.K.; Blomhoff, R.; Gundersen, T.E. Simultaneous and trace determination of reduced and oxidized glutathione in minute plasma samples using dual mode fluorescence detection and column switching high performance liquid chromatography. J. Chromatogr. A 2007, 1142, 178–184. [Google Scholar]
- Ivanov, A.R.; Nazimov, I.V.; Baratova, L.A. Qualitative and quantitative determination of biologically active low-molecular-mass thiols in human blood by reversed-phase high-performance liquid chromatography with photometry and fluorescence detection. J. Chromatogr. A 2000, 870, 433–442. [Google Scholar]
- Kamencic, H.; Lyon, A.; Paterson, P.G.; Juurlink, B.H.J. Monochlorobimane fluorometric method to measure tissue glutathione. Anal. Biochem. 2000, 286, 35–37. [Google Scholar]
- Chou, S.T.; Ko, L.E.; Yang, C.S. High performance liquid chromatography with fluorimetric detection for the determination of total homocysteine in human plasma: Method and clinical applications. Anal. Chim. Acta 2001, 429, 331–336. [Google Scholar]
- Toyo'oka, T.; Tanabe, J.; Kashihara, Y. Determination of intracellular glutathione in rat hepatocytes after treatment of environmental pollutants by capillary electrophoresis with laser-induced fluorescence detection. Anal. Chim. Acta 2001, 433, 1–12. [Google Scholar]
- Ogasawara, Y.; Mukai, Y.; Togawa, T.; Suzuki, T.; Tanabe, S.; Ishii, K. Determination of plasma thiol bound to albumin using affinity chromatography and high-performance liquid chromatography with fluorescence detection: Ratio of cysteinyl albumin as a possible biomarker of oxidative stress. J. Chromatogr. B. 2007, 845, 157–163. [Google Scholar]
- Satoh, S.; Shindoh, M.; Min, J.Z.; Toyo'oka, T.; Fukushima, T.; Inagaki, S. Selective and sensitive determination of lipoyllysine (protein-bound alpha-lipoic acid) in biological specimens by high-performance liquid chromatography with fluorescence detection. Anal. Chim. Acta 2008, 618, 210–217. [Google Scholar]
- Feussner, A.; Rolinski, B.; Weiss, N.; Deufel, T.; Wolfram, G.; Roscher, A.A. Determination of total homocysteine in human plasma by isocratic high-performance liquid chromatography. Eur. J. Clin. Chem. Clin. 1997, 35, 687–691. [Google Scholar]
- Nolin, T.D.; McMenamin, M.E.; Himmelfarb, J. Simultaneous determination of total homocysteine, cysteine, cysteinylglycine, and glutathione in human plasma by high-performance liquid chromatography: Application to studies of oxidative stress. J. Chromatogr. B. 2007, 852, 554–561. [Google Scholar]
- Carru, C.; Deiana, L.; Sotgia, S.; Pes, G.M.; Zinellu, A. Plasma thiols redox status by laser-induced fluorescence capillary electrophoresis. Electrophoresis 2004, 25, 882–889. [Google Scholar]
- Zinellu, A.; Carru, C.; Sotgia, S.; Deiana, L. Plasma D-penicillamine redox state evaluation by capillary electrophoresis with laser-induced fluorescence. J. Chromatogr. B. 2004, 803, 299–304. [Google Scholar]
- Musenga, A.; Mandrioli, R.; Bonifazi, P.; Kenndler, E.; Pompei, A.; Raggi, M.A. Sensitive and selective determination of glutathione in probiotic bacteria by capillary electrophoresis-laser induced fluorescence. Anal. Bioanal. Chem. 2007, 387, 917–924. [Google Scholar]
- Kaniowska, E.; Chwatko, G.; Glowacki, R.; Kubalczyk, P.; Bald, E. Urinary excretion measurement of cysteine and homocysteine in the form of their S-pyridinium derivatives by high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. A 1998, 798, 27–35. [Google Scholar]
- Bald, E.; Kaniowska, E.; Chwatko, G.; Glowacki, R. Liquid chromatographic assessment of total and protein-bound homocysteine in human plasma. Talanta 2000, 50, 1233–1243. [Google Scholar]
- Kuimierek, K.; Bald, E. Measurement of reduced and total mercaptamine in urine using liquid chromatography with ultraviolet detection. Biomed. Chromatogr. 2008, 22, 441–445. [Google Scholar]
- Kubalczyk, P.; Bald, E. Transient pseudo-isotachophoretic stacking in analysis of plasma for homocysteine by capillary zone electrophoresis. Anal. Bioanal. Chem. 2006, 384, 1181–1185. [Google Scholar]
- Bald, E.; Chwatko, G.; Glowacki, R.; Kusmierek, K. Analysis of plasma thiols by high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. A 2004, 1032, 109–115. [Google Scholar]
- Federici, G.; Pastore, A.; Massoud, R.; Motti, C.; Lo Russo, A.; Fucci, G.; Cortese, C. Fully automated assay for total homocysteine, cysteine, cysteinylglycine, glutathione, cysteamine, and 2-mercaptopropionylglycine in plasma and urine. Clin. Chem. 1998, 44, 825–832. [Google Scholar]
- Imai, K.; Toyo'oka, T. Fluorometric assay of thiols with fluorobenzoxadiazoles. Methods Enzymol. 1987, 143, 67–75. [Google Scholar]
- Toyo'oka, T.; Tanabe, J.; Jinno, H. Determination of rat hepatocellular glutathione by reversed-phase liquid chromatography with fluorescence detection and cytotoxicity evaluation of environmental pollutants based on the concentration change. Biomed. Chromatogr. 2001, 15, 240–247. [Google Scholar]
- Satoh, S.; Toyo'oka, T.; Fukushima, T.; Inagaki, S. Simultaneous determination of alpha-lipoic acid and its reduced form by high-performance liquid chromatography with fluorescence detection. J. Chromatogr. B. 2007, 854, 109–115. [Google Scholar]
- Kusmierek, K.; Glowacki, R.; Bald, E. Analysis of urine for cysteine, cysteinylglycine, and homocysteine by high-performance liquid chromatography. Anal. Bioanal. Chem. 2006, 385, 855–860. [Google Scholar]
- Kusmierek, K.; Bald, E. Simultaneous determination of tiopronin and D-penicillamine in human urine by liquid chromatography with ultraviolet detection. Anal. Chim. Acta 2007, 590, 132–137. [Google Scholar]
- Huang, T.M.; Yang, B.; Yu, Y.J.; Zheng, X.W.; Duan, G.L. Reverse-phase high performance liquid chromatography for the determination of tiopronin in human plasma after derivatization with p-bromophenacyl bromide. Anal. Chim. Acta 2006, 565, 178–182. [Google Scholar]
- Maeda, H.; Matsuno, H.; Ushida, M.; Katayama, K.; Saeki, K.; Itoh, N. 2,4-Dinitrobenzenesulfonyl fluoresceins as fluorescent alternatives to Ellman's reagent in thiol-quantification enzyme assays. Angew. Chem. Int. Ed. 2005, 44, 2922–2925. [Google Scholar]
- Bouffard, J.; Kim, Y.; Swager, T.M.; Weissleder, R.; Hilderbrand, S.A. A highly selective fluorescent probe for thiol bioimaging. Org. Lett. 2008, 10, 37–40. [Google Scholar]
- Wang, S.-P.; Deng, W.-J.; Sun, D.; Yan, M.; Zheng, H.; Xu, J.-G. A colorimetric and fluorescent merocyanine-based probe for biological thiols. Org. Biomol. Chem. 2009, 7, 4017–4020. [Google Scholar]
- Li, X.; Qian, S.; He, Q.; Yang, B.; Li, J.; Hu, Y. Design and synthesis of a highly selective fluorescent turn-on probe for thiol bioimaging in living cells. Org. Biomol. Chem. 2010, 8, 3627–3630. [Google Scholar]
- Shao, J.; Guo, H.; Ji, S.; Zhao, J. Styryl-BODIPY based red-emitting fluorescent OFF–ON molecular probe for specific detection of cysteine. Biosens. Bioelectron. 2011, 26, 3012–3017. [Google Scholar]
- Ji, S.; Guo, H.; Yuan, X.; Li, X.; Ding, H.; Gao, P.; Zhao, C.; Wu, W.; Wu, W.; Zhao, J. A highly selective off-on red-emitting phosphorescent thiol probe with large stokes shift and long luminescent lifetime. Org. Lett. 2010, 12, 2876–2879. [Google Scholar]
- Ercal, N.; Yang, P.; Aykin, N. Determination of biological thiols by high-performance liquid chromatography following derivatization by ThioGlo maleimide reagents. J. Chromatogr. B 2001, 753, 287–292. [Google Scholar]
- Matsumoto, T.; Urano, Y.; Shoda, T.; Kojima, H.; Nagano, T. A thiol-reactive fluorescence probe based on donor-excited photoinduced electron transfer: Key role of ortho substitution. Org. Lett. 2007, 9, 3375–3377. [Google Scholar]
- Guo, X.-F.; Wang, H.; Guo, Y.-H.; Zhang, H.-S. Selective spectrofluorimetric determination of glutathione in clinical and biological samples using 1,3,5,7-tetramethyl-8-phenyl-(2-maleimide)-difluoroboradiaza-s-indacene. Anal. Chim. Acta 2009, 633, 71–75. [Google Scholar]
- Liu, Y.; Yu, Y.; Lam, J.W.Y.; Hong, Y.; Faisal, M.; Yuan, W.Z.; Tang, B.Z. Simple biosensor with high selectivity and sensitivity: Thiol-specific biomolecular probing and intracellular imaging by AIE fluorogen on a TLC plate through a thiol–ene click mechanism. Chem. Eur. J. 2010, 16, 8433–8438. [Google Scholar]
- Kand, D.; Kalle, A.M.; Varma, S.J.; Talukdar, P. A chromenoquinoline-based fluorescent off-on thiol probe for bioimaging. Chem. Commun. 2012, 48, 2722–2724. [Google Scholar]
- Zeng, Y.; Zhang, G.; Zhang, D. A selective colorimetric chemosensor for thiols based on intramolecular charge transfer mechanism. Anal. Chim. Acta 2008, 627, 254–257. [Google Scholar]
- Yi, L.; Li, H.Y.; Sun, L.; Liu, L.L.; Zhang, C.H.; Xi, Z. A Highly Sensitive Fluorescence Probe for Fast Thiol-Quantification Assay of Glutathione Reductase. Angew. Chem. Int. Ed. 2009, 48, 4034–4037. [Google Scholar]
- Lin, W.; Yuan, L.; Cao, Z.; Feng, Y.; Long, L. A sensitive and selective fluorescent thiol probe in water based on the conjugate 1,4-addition of thiols to α,β-unsaturated ketones. Chem. Eur. J. 2009, 15, 5096–5103. [Google Scholar]
- Kwon, H.; Lee, K.; Kim, H.-J. Coumarin-malonitrile conjugate as a fluorescence turn-on probe for biothiols and its cellular expression. Chem. Commun. 2011, 47, 1773–1775. [Google Scholar]
- Ros-Lis, J.V.; García, B.; Jiménez, D.; Martínez-Máñez, R.; Sancenón, F.; Soto, J.; Gonzalvo, F.; Valldecabres, M.C. Squaraines as fluoro−chromogenic probes for thiol-containing compounds and their application to the detection of biorelevant thiols. J. Am. Chem. Soc. 2004, 126, 4064–4065. [Google Scholar]
- Stolik, S.; Delgado, J.A.; Perez, A.; Anasagasti, L. Measurement of the penetration depths of red and near infrared light in human “ex vivo” tissues. J. Photochem. Photobiol. B 2000, 57, 90–93. [Google Scholar]
- Sreejith, S.; Divya, K.P.; Ajayaghosh, A. A near-infrared squaraine dye as a latent ratiometric fluorophore for the detection of aminothiol content in blood plasma. Angew. Chem. Int. Ed. 2008, 47, 7883–7887. [Google Scholar]
- Ros-Lis, J.V.; Martinez-Manez, R.; Soto, J. A selective chromogenic reagent for cyanide determination. Chem. Commun. 2002, 2248–2249. [Google Scholar]
- Huo, F.-J.; Sun, Y.-Q.; Su, J.; Chao, J.-B.; Zhi, H.-J.; Yin, C.-X. Colorimetric detection of thiols using a chromene molecule. Org. Lett. 2009, 11, 4918–4921. [Google Scholar]
- Chen, X.; Ko, S.-K.; Kim, M.J.; Shin, I.; Yoon, J. A thiol-specific fluorescent probe and its application for bioimaging. Chem. Commun. 2010, 46, 2751–2753. [Google Scholar]
- Riddles, P.W.; Blakeley, R.L.; Zerner, B. Reassessment of Ellman's reagent. Methods Enzymol. 1983, 91, 49–60. [Google Scholar]
- Taylan, E.; Resmi, H. The Analytical Performance of a Microplate Method for Total Sulfhydryl Measurement in Biological Samples. Turk. J. Biochem. 2010, 35, 275–278. [Google Scholar]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar]
- Eyer, P.; Worek, F.; Kiderlen, D.; Sinko, G.; Stuglin, A.; Simeon-Rudolf, V.; Reiner, E. Molar absorption coefficients for the reduced Ellman reagent: reassessment. Anal. Biochem. 2003, 312, 224–227. [Google Scholar]
- Faulstich, H.; Tews, P.; Heintz, D. Determination and derivatization of protein thiols by N-octyldithionitrobenzoic acid. Anal. Biochem. 1993, 208, 357–362. [Google Scholar]
- Zhang, H.; Le, M.; Means, G.E. A kinetic approach to characterize the electrostatic environments of thiol groups in proteins. Bioorg. Chem. 1998, 26, 356–364. [Google Scholar]
- Zhu, J.G.; Dhimitruka, I.; Pei, D. 5-(2-aminoethyl)dithio-2-nitrobenzoate as a more base-stable alternative to Ellman's reagent. Org. Lett. 2004, 6, 3809–3812. [Google Scholar]
- Le, M.; Means, G.E. A procedure for the determination of monothiols in the presence of dithiothreitol -an improved assay for the reduction of disulfides. Anal. Biochem. 1995, 229, 264–271. [Google Scholar]
- Grassetti, D.R.; Murray, J.F. Determination of sulfhydryl groups with 2,2′- or 4,4′-dithiodipyridine. Arch. Biochem. Biophys. 1967, 119, 41–49. [Google Scholar]
- Riener, C.K.; Kada, G.; Gruber, H.J. Quick measurement of protein sulfhydryls with Ellman's reagent and with 4,4′-dithiodipyridine. Anal. Bioanal. Chem. 2002, 373, 266–276. [Google Scholar]
- Cao, X.W.; Lin, W.Y.; Yu, Q.X. A ratiometric fluorescent probe for thiols based on a tetrakis (4-hydroxyphenyl)porphyrin-coumarin scaffold. J. Org. Chem. 2011, 76, 7423–7430. [Google Scholar]
- Pires, M.M.; Chmielewski, J. Fluorescence imaging of cellular glutathione using a latent rhodamine. Org. Lett. 2008, 10, 837–840. [Google Scholar]
- Denk, W.; Strickler, J.H.; Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 1990, 248, 73–6. [Google Scholar]
- Rubart, M. Two-photon microscopy of cells and tissue. Circ. Res. 2004, 95, 1154–1166. [Google Scholar]
- Helmchen, F.; Denk, W. Deep tissue two-photon microscopy. Nat. Methods 2005, 2, 932–940. [Google Scholar]
- Zipfel, W.R.; Williams, R.M.; Webb, W.W. Nonlinear magic: multiphoton microscopy in the biosciences. Nat. Biotechnol. 2003, 21, 1368–1376. [Google Scholar]
- Lee, J.H.; Lim, C.S.; Tian, Y.S.; Han, J.H.; Cho, B.R. A two-photon fluorescent probe for thiols in live cells and tissues. J. Am. Chem. Soc. 2010, 132, 1216–1217. [Google Scholar]
- Lim, C.S.; Masanta, G.; Kim, H.J.; Han, J.H.; Kim, H.M.; Cho, B.R. Ratiometric detection of mitochondrial thiols with a two-photon fluorescent probe. J. Am. Chem. Soc. 2011, 133, 11132–11135. [Google Scholar]
- Yousif, L.F.; Stewart, K.M.; Kelley, S.O. Targeting mitochondria with organelle-specific compounds: Strategies and applications. Chembiochem 2009, 10, 1939–1950. [Google Scholar]
- Lee, M.H.; Han, J.H.; Kwon, P.-S.; Bhuniya, S.; Kim, J.Y.; Sessler, J.L.; Kang, C.; Kim, J.S. Hepatocyte-targeting single galactose-appended naphthalimide: A tool for intracellular thiol imaging in vivo. J. Am. Chem. Soc. 2012, 134, 1316–1322. [Google Scholar]
- Mutus, B.; Durocher, S.; Rezaee, A.; Hamm, C.; Rangan, C.; Mittler, S. Disulfide-linked, gold nanoparticle based reagent for detecting small molecular weight thiols. J. Am. Chem. Soc. 2009, 131, 2475–2477. [Google Scholar]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.B.; Hafeman, D.G.; Hoekstra, W.G. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar]
- Bhabak, K.P.; Mugesh, G. Functional mimics of glutathione peroxidase: Bioinspired synthetic antioxidants. Acc. Chem. Res. 2010, 43, 1408–1419. [Google Scholar]
- Mugesh, G.; du Mont, W.W.; Sies, H. Chemistry of biologically important synthetic organoselenium compounds. Chem. Rev. 2001, 101, 2125–2179. [Google Scholar]
- Müller, A.; Cadenas, E.; Graf, P.; Sies, H. A novel biologically active seleno-organic compound-I. Glutathione peroxidase-like activity in vitro and antioxidant capacity of PZ 51 (Ebselen). Biochem. Pharmacol. 1984, 33, 3235–3239. [Google Scholar]
- Tang, B.; Xing, Y.L.; Li, P.; Zhang, N.; Yu, F.B.; Yang, G.W. A rhodamine-based fluorescent probe containing a Se-N bond for detecting thiols and its application in living cells. J. Am. Chem. Soc. 2007, 129, 11666–11667. [Google Scholar]
- Tang, B.; Yin, L.; Wang, X.; Chen, Z.; Tong, L.; Xu, K. A fast-response, highly sensitive and specific organoselenium fluorescent probe for thiols and its application in bioimaging. Chem. Commun. 2009, 5293–5295. [Google Scholar]
- Goodman, S.I.; Elsas, L.J.; Rosenblatt, D.S. ASHG/ACMG STATEMENT—Measurement and use of total plasma homocysteine. Am. J. Hum. Genet. 1998, 63, 1541–1543. [Google Scholar]
- Amarnath, V.; Amarnath, K. Specific determination of cysteine and penicillamine through cyclization to 2-thioxothiazolidine-4-carboxylic acids. Talanta 2002, 56, 745–751. [Google Scholar]
- Amarnath, K.; Amarnath, V.; Valentine, H.L.; Valentine, W.M. A specific HPLC-UV method for the determination of cysteine and related aminothiols in biological samples. Talanta 2003, 60, 1229–1238. [Google Scholar]
- Strongin, R.M.; Rusin, O.; St Luce, N.N.; Agbaria, R.A.; Escobedo, J.O.; Jiang, S.; Warner, I.M.; Dawan, F.B.; Lian, K. Visual detection of cysteine and homocysteine. J. Am. Chem. Soc. 2004, 126, 438–439. [Google Scholar]
- Rusin, O.; Wang, W.H.; Xu, X.Y.; Kim, K.K.; Escobedo, J.O.; Fakayode, S.O.; Fletcher, K.A.; Lowry, M.; Schowalter, C.M.; Lawrence, C.M.; et al. Detection of homocysteine and cysteine. J. Am. Chem. Soc. 2005, 127, 15949–15958. [Google Scholar]
- Tanaka, F.; Mase, N.; Barbas, C.F. Determination of cysteine concentration by fluorescence increase: Reaction of cysteine with a fluorogenic aldehyde. Chem. Commun. 2004, 15, 1762–1763. [Google Scholar]
- Lee, K.S.; Kim, T.K.; Lee, J.H.; Kim, H.J.; Hong, J.I. Fluorescence turn-on probe for homocysteine and cysteine in water. Chem. Commun. 2008, 46, 6173–6175. [Google Scholar]
- Yang, Z.; Zhao, N.; Sun, Y.; Miao, F.; Liu, Y.; Liu, X.; Zhang, Y.; Ai, W.; Song, G.; Shen, X.; Yu, X.; Sun, J.; Wong, W.-Y. Highly selective red- and green-emitting two-photon fluorescent probes for cysteine detection and their bio-imaging in living cells. Chem. Commun. 2012, 48, 3442–3444. [Google Scholar]
- Chen, H.; Zhao, Q.; Wu, Y.; Li, F.; Yang, H.; Yi, T.; Huang, C. Selective phosphorescence chemosensor for homocysteine based on an iridium(III) complex. Inorg. Chem. 2007, 46, 11075–11081. [Google Scholar]
- Strongin, R.M.; Wang, W.H.; Escobedo, J.O.; Lawrence, C.M. Direct detection of homocysteine. J. Am. Chem. Soc. 2004, 126, 3400–3401. [Google Scholar]
- Escobedo, J.O.; Wang, W.H.; Strongin, R.M. Use of a commercially available reagent for the selective detection of homocysteine in plasma. Nat. Protoc. 2006, 1, 2759–2762. [Google Scholar]
- Yang, X.; Guo, Y.; Strongin, R.M. Conjugate addition/cyclization sequence enables selective and simultaneous fluorescence detection of cysteine and homocysteine. Angew. Chem. Int. Ed. 2011, 50, 10690–10693. [Google Scholar]
- Guo, Z.; Nam, S.; Park, S.; Yoon, J. A highly selective ratiometric near-infrared fluorescent cyanine sensor for cysteine with remarkable shift and its application in bioimaging. Chem. Sci. 2012, 3, 2760–2765. [Google Scholar]
- Harris, R.L.; Bingham, E.; Cohrssen, B. Patty's Industrial Hygiene and Toxicology, 5th ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2001; Volume 7. [Google Scholar]
- Hathaway, G.J.; Proctor, N.H. Proctor and Hughes' Chemical Hazards of the Workplase; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Wang, W.; Jiang, W.; Fu, Q.Q.; Fan, H.Y.; Ho, J. A highly selective fluorescent probe for thiophenols. Angew. Chem. Int. Ed. 2007, 46, 8445–8448. [Google Scholar]
- Wang, W.; Jiang, W.; Cao, Y.T.; Liu, Y.A. Rational design of a highly selective and sensitive fluorescent PET probe for discrimination of thiophenols and aliphatic thiols. Chem. Commun. 2010, 46, 1944–1946. [Google Scholar]
- Lin, W.Y.; Long, L.L.; Tan, W. A highly sensitive fluorescent probe for detection of benzenethiols in environmental samples and living cells. Chem. Commun. 2010, 46, 1503–1505. [Google Scholar]
- Lawrence, N.S.; Davis, J.; Compton, R.G. Analytical strategies for the detection of sulfide: A review. Talanta 2000, 52, 771–784. [Google Scholar]
- Hughes, M.N.; Centelles, M.N.; Moore, K.P. Making and working with hydrogen sulfide the chemistry and generation of hydrogen sulfide in vitro and its measurement in vivo: A review. Free Radic. Biol. Med. 2009, 47, 1346–1353. [Google Scholar]
- Radfordknoery, J.; Cutter, G.A. Determination of carbonyl sulfide and hydrogen sulfide species in natural waters using specialized collection procedures and gas chromatography with flame photometric detection. Anal. Chem. 1993, 65, 976–982. [Google Scholar]
- Berube, P.R.; Parkinson, P.D.; Hall, E.R. Measurement of reduced sulphur compounds contained in aqueous matrices by direct injection into a gas chromatograph with a flame photometric detector. J. Chromatogr. A 1999, 830, 485–489. [Google Scholar]
- Hill, P.G.; Smith, R.M. Determination of sulphur compounds in beer using headspace solid-phase microextraction and gas chromatographic analysis with pulsed flame photometric detection. J. Chromatogr. A 2000, 872, 203–213. [Google Scholar]
- Hyspler, R.; Tichá, A.; Indrová, M.; Zadák, Z.; Hysplerová, L.; Gasparic, J.; Churácek, J. A simple, optimized method for the determination of sulphide in whole blood by GC–MS as a marker of bowel fermentation processes. J. Chromatogr. B. 2002, 770, 255–259. [Google Scholar]
- Savage, J.C.; Gould, D.H. Determination of sulfide in brain tissue and rumen fluid by ion-interaction reversed-phase high-performance liquid chromatography. J. Chromatogr. Biomed. 1990, 526, 540–545. [Google Scholar]
- Schiavon, G.; Zotti, G.; Toniolo, R.; Bontempelli, G. Electrochemical detection of trace hydrogen-sulfide in gaseous samples by porous silver electrodes supported on ion-exchange membranes (solid polymer electrolytes). Anal. Chem. 1995, 67, 318–323. [Google Scholar]
- Spilker, B.; Randhahn, J.; Grabow, H.; Beikirch, H.; Jeroschewski, P. New electrochemical sensor for the detection of hydrogen sulfide and other redox active species. J. Electroanal. Chem. 2008, 612, 121–130. [Google Scholar]
- Doeller, J.E.; Isbell, T.S.; Benavides, G.; Koenitzer, J.; Patel, H.; Patel, R.P.; Lancaster, J.R.; Darley-Usmar, V.M.; Kraus, D.W. Polarographic measurement of hydrogen sulfide production and consumption by mammalian tissues. Anal. Biochem. 2005, 341, 40–51. [Google Scholar]
- Li, L.; Bhatia, M.; Zhu, Y.Z.; Ramnath, R.D.; Wang, Z.J.; Anuar, F.B. M.; Whiteman, M.; Salto-Tellez, M.; Moore, P.K. Hydrogen sulfide is a novel mediator of lipopolysaccharide-induced inflammation in the mouse. FASEB J. 2005, 19, 1196–1198. [Google Scholar]
- Kamoun, P. Endogenous production of hydrogen sulfide in mammals. Amino Acids 2004, 26, 243–254. [Google Scholar]
- Whitfield, N.L.; Kreimier, E.L.; Verdial, F.C.; Skovgaard, N.; Olson, K.R. Reappraisal of H2S/sulfide concentration in vertebrate blood and its potential significance in ischemic preconditioning and vascular signaling. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1930–R1937. [Google Scholar]
- Lei, W.; Dasgupta, P.K. Determination of sulfide and mercaptans in caustic scrubbing liquor. Anal. Chim. Acta 1989, 226, 165–170. [Google Scholar]
- Fonselius, S.; Dyrssen, D.; Yhlen, B. Determination of Hydrogen Sulphide. In Methods of Seawater Analysis; Grasshoff, K., Kremling, K., Ehrhardt, M., Eds.; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2007. [Google Scholar]
- Sowmya, S.; Swathi, Y.; Yeo, A.L.; Shoon, M.L.; Moore, P.K.; Bhatia, M. Hydrogen sulfide: regulatory role on blood pressure in hyperhomocysteinemia. Vasc. Pharmacol. 2010, 53, 138–143. [Google Scholar]
- Lewis, G.N.; Godschmid, O.; Magel, T.T.; Bigeleisen, J. Dimetric and other forms of methylene blue: absorption and fluorescence of the pure monomer. J. Am. Chem. Soc. 1943, 65, 1150–1154. [Google Scholar]
- Liu, C.; Pan, J.; Li, S.; Zhao, Y.; Wu, L.Y.; Berkman, C.E.; Whorton, A.R.; Xian, M. Capture and visualization of hydrogen sulfide by a fluorescent probe. Angew. Chem. Int. Ed. 2011, 50, 10327–10329. [Google Scholar]
- Qian, Y.; Karpus, J.; Kabil, O.; Zhu, H.-L.; Banerjee, R.; Zhao, J.; He, C. Selective fluorescent probes for live-cell monitoring of sulfide. Nat. Commun. 2011, 2, 495. [Google Scholar]
- Liu, C.; Peng, B.; Li, S.; Park, C.-M.; Whorton, A.R.; Xian, M. Reaction based fluorescent probes for hydrogen sulfide. Org. Lett. 2012, 14, 2184–2187. [Google Scholar]
- Yang, X.F.; Wang, L.P.; Xu, H.M.; Zhao, M.L. A fluorescein-based fluorogenic and chromogenic chemodosimeter for the sensitive detection of sulfide anion in aqueous solution. Anal. Chim. Acta 2009, 631, 91–95. [Google Scholar]
- Kazemi, F.; Kiasat, A.R.; Sayyahi, S. Chemoselective reduction of azides with sodium sulfide hydrate under solvent free conditions. Phosphorus Sulfur 2004, 179, 1813–1817. [Google Scholar]
- Lippert, A.R.; New, E.J.; Chang, C.J. Reaction-based fluorescent probes for selective imaging of hydrogen sulfide in living cells. J. Am. Chem. Soc. 2011, 133, 10078–10080. [Google Scholar]
- Peng, H.; Cheng, Y.; Dai, C.; Wang, B. A fluorescent probe for fast and quantitative detection of hydrogen sulfide in blood. Angew. Chem. Int. Ed. 2011, 50, 9672–9675. [Google Scholar]
- Xuan, W.M.; Sheng, C.Q.; Cao, Y.T.; He, W.H.; Wang, W. Fluorescent probes for the detection of hydrogen sulfide in biological systems. Angew. Chem. Int. Ed. 2012, 51, 2282–2284. [Google Scholar]
- Montoya, L.A.; Pluth, M.D. Selective turn-on fluorescent probes for imaging hydrogen sulfide in living cells. Chem. Commun. 2012, 48, 4767–4769. [Google Scholar]
- Yu, F.; Li, P.; Song, P.; Wang, B.; Zhao, J.; Han, K. An ICT-based strategy to a colorimetric and ratiometric fluorescence probe for hydrogen sulfide in living cells. Chem. Commun. 2012, 48, 2852–2854. [Google Scholar]
- Chen, S.; Chen, Z.-j.; Ren, W.; Ai, H.-w. Reaction-based genetically encoded fluorescent hydrogen sulfide sensors. J. Am. Chem. Soc. 2012, 134, 9589–9592. [Google Scholar]
- Das, S.K.; Lim, C.S.; Yang, S.Y.; Han, J.H.; Cho, B.R. A small molecule two-photon probe for hydrogen sulfide in live tissues. Chem. Commun. 2012, 48, 8395–8397. [Google Scholar]
- Wu, Z.S.; Li, Z.; Yang, L.; Han, J.H.; Han, S.F. Fluorogenic detection of hydrogen sulfide via reductive unmasking of o-azidomethylbenzoyl-coumarin conjugate. Chem. Commun. 2012, 48, 10120–10122. [Google Scholar]
- Li, W.; Sun, W.; Yu, X.; Du, L.; Li, M. Coumarin-based Fluorescent Probes for H2S Detection. J. Fluoresc. 2012. [Google Scholar] [CrossRef]
- Chang, S.K.; Choi, M.G.; Cha, S.; Lee, H.; Jeon, H.L. Sulfide-selective chemosignaling by a Cu(2+) complex of dipicolylamine appended fluorescein. Chem. Commun. 2009, 47, 7390–7392. [Google Scholar]
- Sasakura, K.; Hanaoka, K.; Shibuya, N.; Mikami, Y.; Kimura, Y.; Komatsu, T.; Ueno, T.; Terai, T.; Kimura, H.; Nagano, T. Development of a highly selective fluorescence probe for hydrogen sulfide. J. Am. Chem. Soc. 2011, 133, 18003–18005. [Google Scholar]
- Hou, F.; Cheng, J.; Xi, P.; Chen, F.; Huang, L.; Xie, G.; Shi, Y.; Liu, H.; Bai, D.; Zeng, Z. Recognition of copper and hydrogen sulfide in vitro using a fluorescein derivative indicator. Dalton Trans. 2012, 41, 5799–5804. [Google Scholar]
- Strianese, M.; Palm, G.J.; Milione, S.; Kuhl, O.; Hinrichs, W.; Pellecchia, C. A FRET enzyme-based probe for monitoring hydrogen sulfide. Inorg. Chem. 2012, 51, 11220–11222. [Google Scholar]
- Hou, F.P.; Huang, L.; Xi, P.X.; Cheng, J.; Zhao, X.F.; Xie, G.Q.; Shi, Y.J.; Cheng, F.J.; Yao, X.J.; Bai, D.C.; Zeng, Z.Z. A Retrievable and highly selective fluorescent probe for monitoring sulfide and imaging in living cells. Inorg. Chem. 2012, 51, 2454–2460. [Google Scholar]
- Wang, M.Q.; Li, K.; Hou, J.T.; Wu, M.Y.; Huang, Z.; Yu, X.Q. BINOL-based fluorescent sensor for recognition of Cu(II) and sulfide anion in water. J. Org. Chem. 2012, 77, 8350–8354. [Google Scholar]
- Briglin, S.M.; Gao, T.; Lewis, N.S. Detection of organic mercaptan vapors using thin films of alkylamine-passivated gold nanocrystals. Langmuir 2004, 20, 299–305. [Google Scholar]
Abbreviations
| ABD-F: | 4-Fluoro-7-sulfamoylbenzofurazan |
| ADNB: | 5-(2-aminoethyl)dithio-2-nitrobenzoate |
| AIE: | aggregation-induced emission |
| ATP: | adenosine triphosphate |
| AuNPs: | gold nanoparticles |
| BHMT: | betaine-homocysteine S-methyltransferase |
| BIPM: | N-[4-(2-benzimidazolyl)phenyl]maleimide |
| BODIPY: | 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene |
| CAT: | cysteine aminotransferase |
| CBS: | cystathionine β-synthase |
| CE: | capillary electrophoresis |
| ChE: | cholinesterase |
| CMPI | 2-chloro-1-methylpyridinium iodide |
| CMQT | 2-chloro-1-methylquinolinium tetrafluoroborate |
| CNS: | central nervous system |
| CSE: | cystathionine γ-lyase or CGL |
| CV: | cardiovascular |
| Cys: | cysteine |
| DAD: | diode array detector |
| DNBS: | 2,4-dinitrophenyl sulfonyl group |
| D-PEN: | D-penicillamine or PenA |
| DSP: | dithiobis(succinimidylpropinate) |
| DTT: | dithiothreitol |
| DTNB: | 5,5′-dithiobis(2-nitrobenzoic acid or Ellman's reagent |
| EPA: | US Environmental Protection Agency |
| FRET: | Förster resonance energy transfer |
| GCL: | γ-glutamate-cysteine ligase or γ-glutamylcysteine synthetase (γ-GCS) |
| Gly: | glycine |
| Glu: | glutamate |
| GPx: | glutathione peroxidase |
| GSH: | glutathione or γ-L-glutamyl-L-cysteinylglycine |
| GSR: | glutathione reductase |
| GSSG: | glutathione disulfide |
| Hcy: | homocysteine |
| HEPES: | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid |
| HPLC: | high performance liquid chromatography |
| 5-IAF: | 5-iodoacetamidofluorescein |
| ICT: | intramolecular charge transfer |
| LSPR: | localized surface plasmon resonance |
| LuxS: | S-ribosylhomocysteinase |
| MAT: | methionine adenosyltransferase |
| MLCT: | metal-to-ligand charge transfer |
| mBrB: | monobromobimane |
| MES: | 2-(N-morpholino)ethanesulfonic acid |
| 3MST: | 3-mercaptopyruvate sulfurtransferase |
| MTs: | AdoMet-dependent methyltransfersess |
| N-DBPM: | N-[4-(dimethylamino-2-benzofuranyl)phenyl]maleimide |
| NIR: | near infrared |
| ODNB: | 5-octyldithio-2-nitrobenzoate |
| PBS: | phosphate buffered saline |
| 2-PDS: | 2,2′-dipyridyl disulfide |
| PET: | photoinduced electron transfer |
| PIPES: | piperazine-N,N′-bis(2-ethanesulfonic acid) |
| RNS: | reactive nitrogen species |
| ROS: | reactive oxygen species |
| RSS: | reactive sulfur species |
| SAHH: | S-adenosylhomocysteine hydrolase |
| SBD-F: | 7-Fluorobenzo-2-oxa-1,3-diazole-4-sulfonic acid ammonium salt |
| Ser: | serine |
| TCDI: | 1,1′-thiocarbonyldiimidazole |
| TCEP | tris(2-carboxyethyl)phosphine |
| tHcy: | total Hcy concentration |
| TNB: | 5-thio-2-nitrobenzoate |
| TP: | tiopronin or Thiola |
| 2-TP: | 2-thiopyridone |
| 4-TP: | 4-thiopyridone |
| TPM: | two-photon microscopy |
| TPP: | triphenylphosphonium |
| Trx: | thioredoxin |
| UV: | ultraviolet |
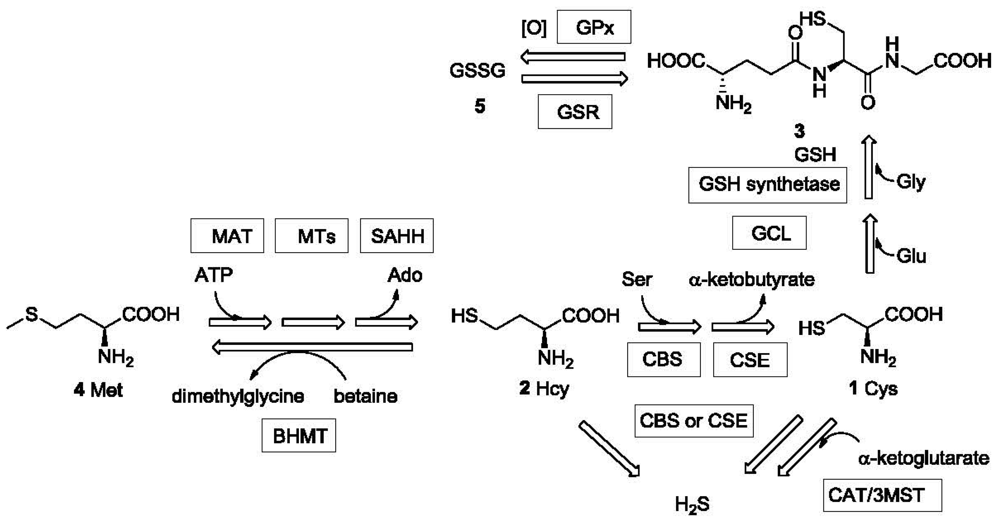
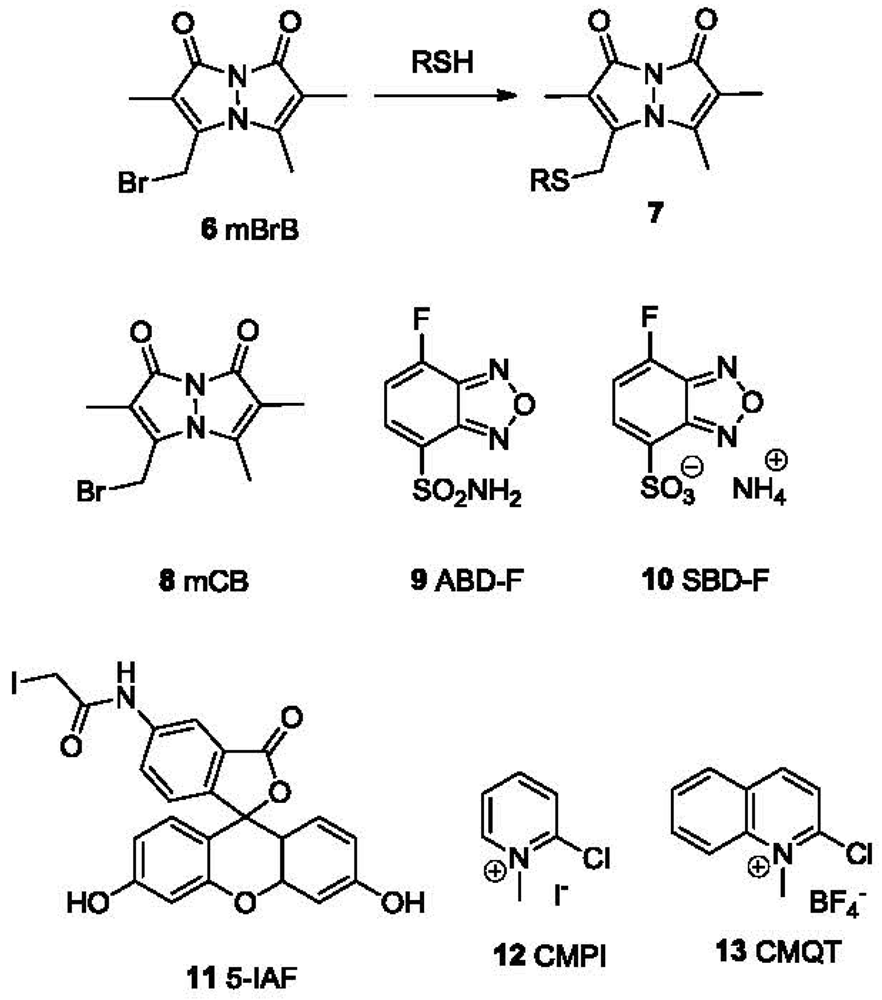


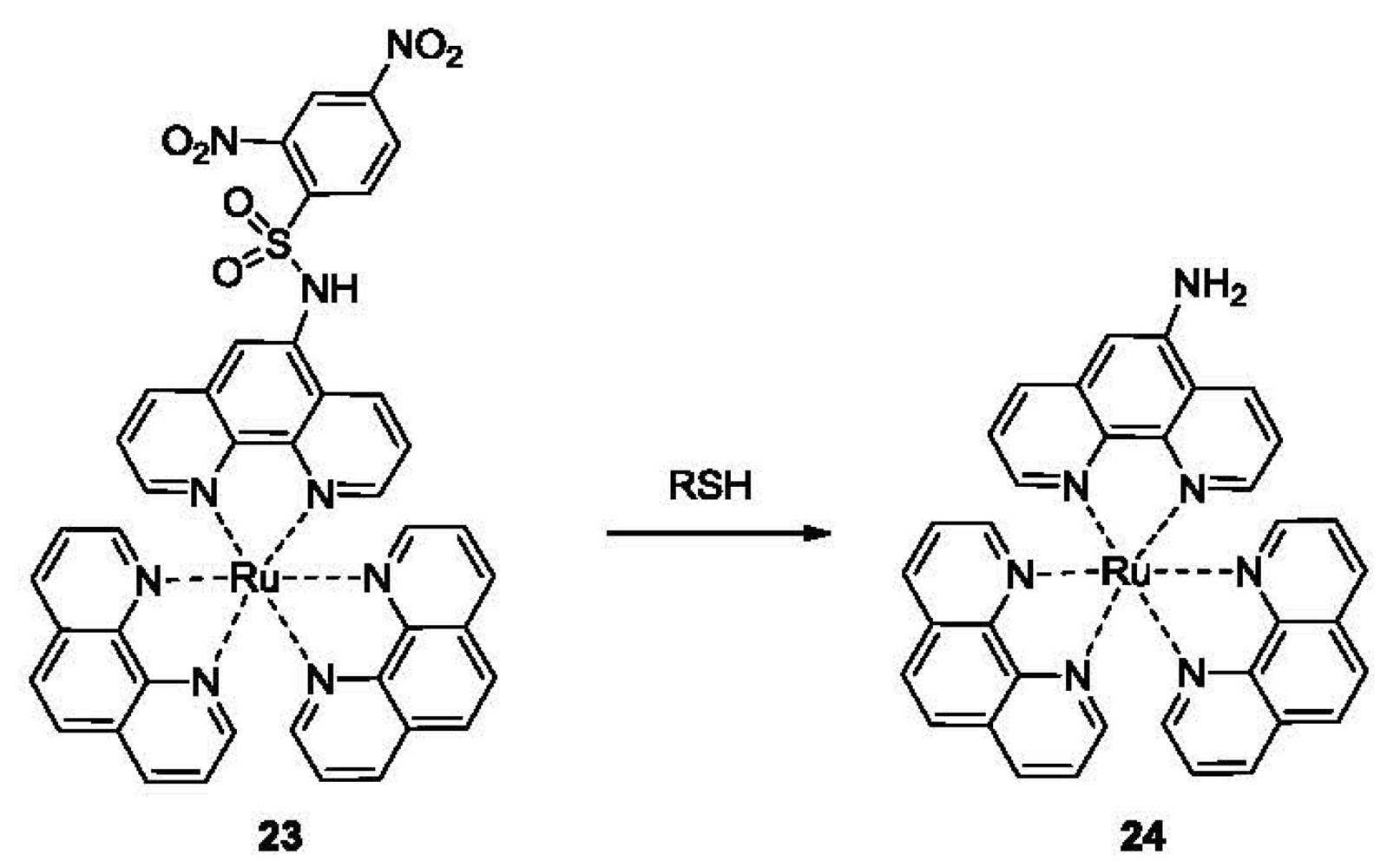
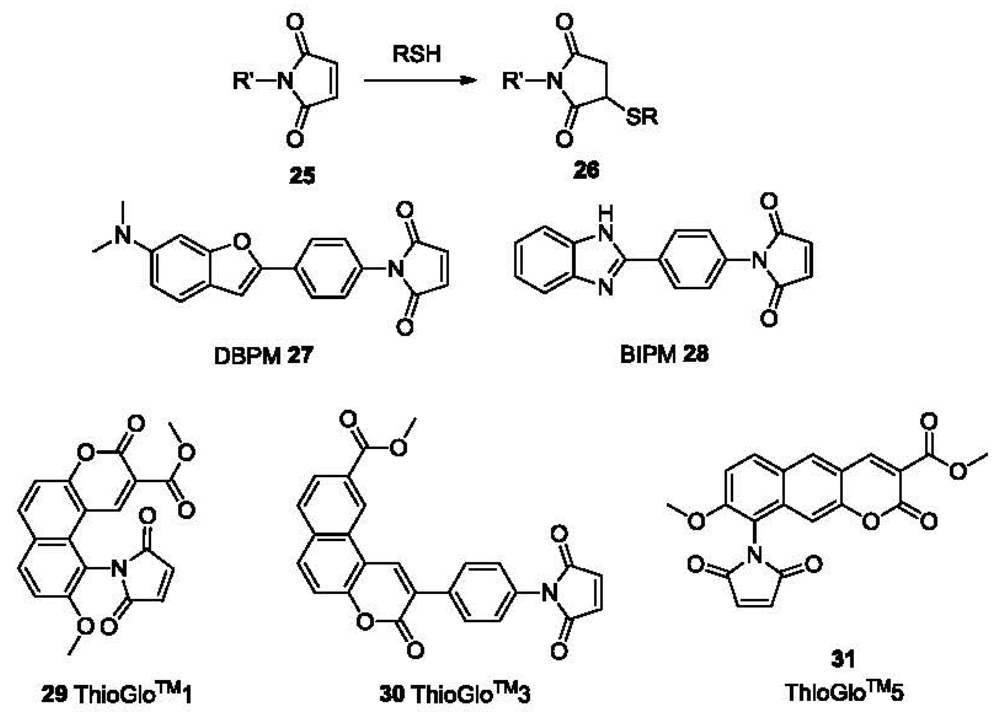
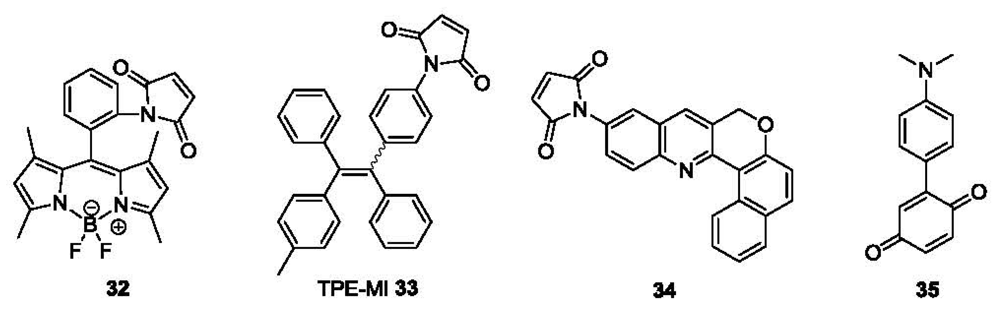


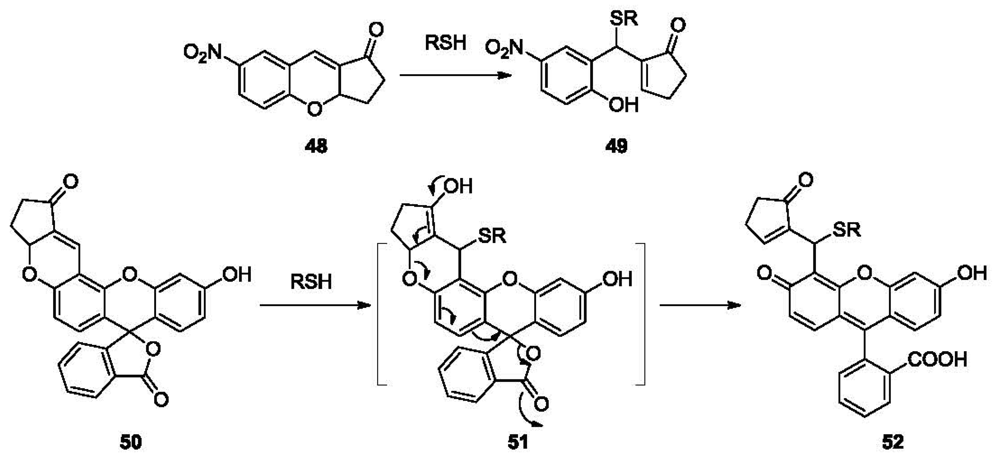
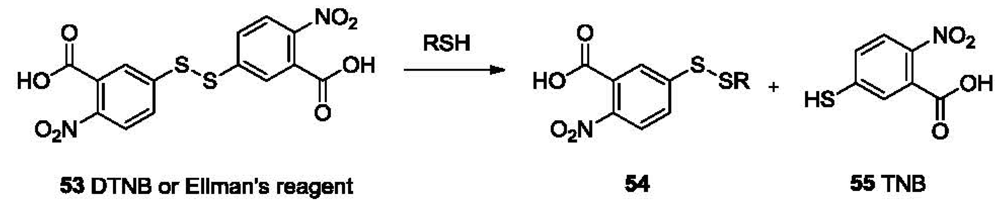

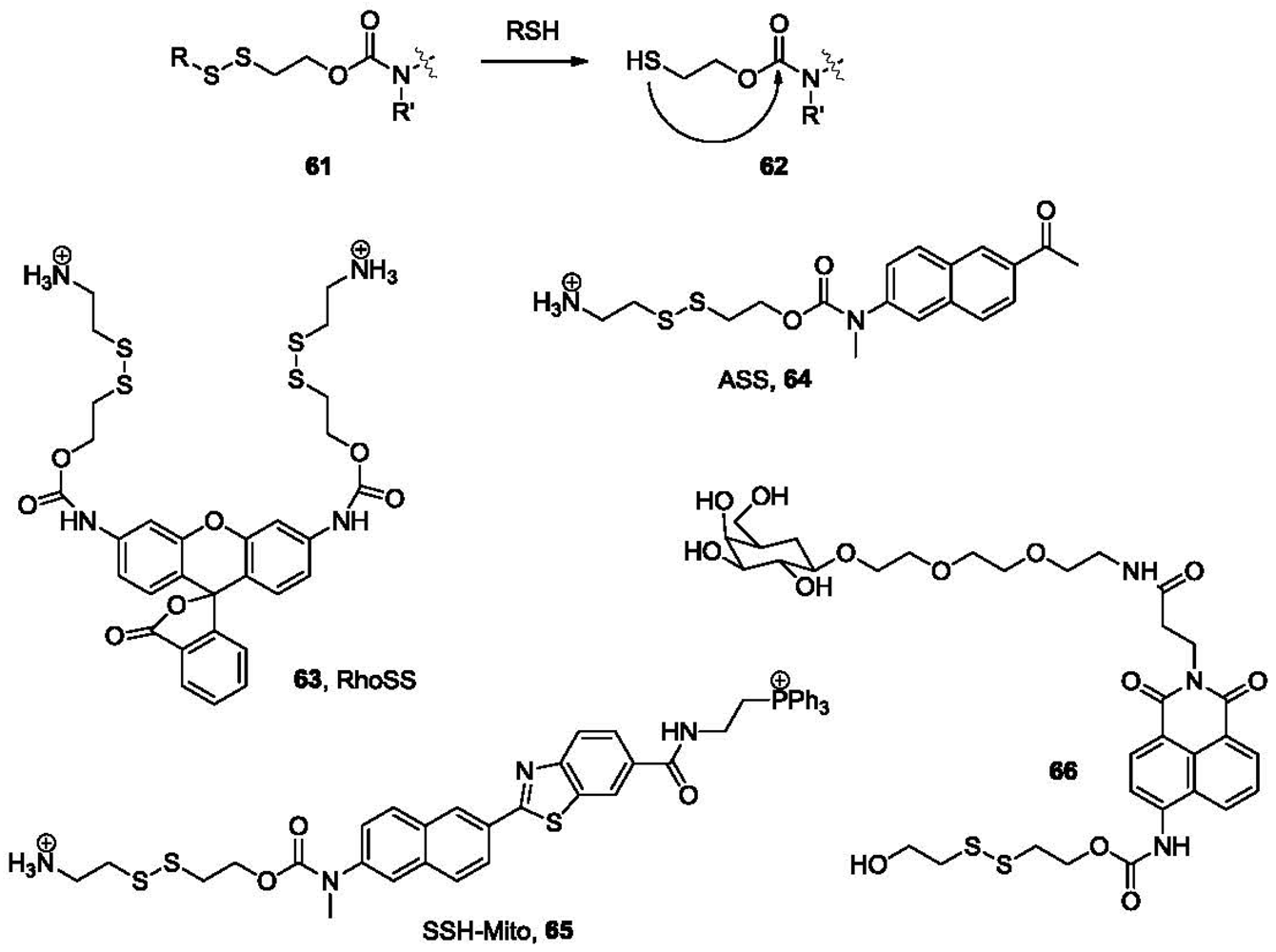
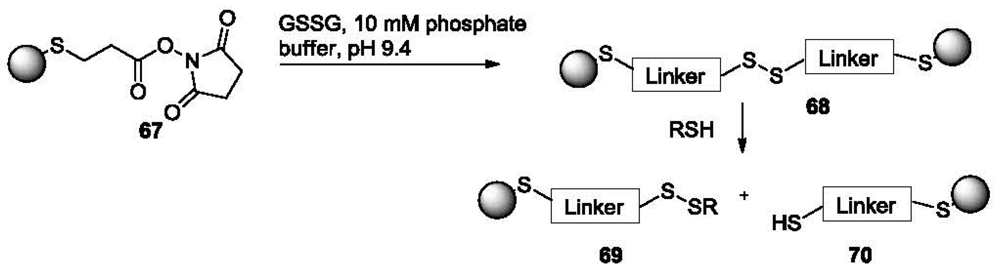
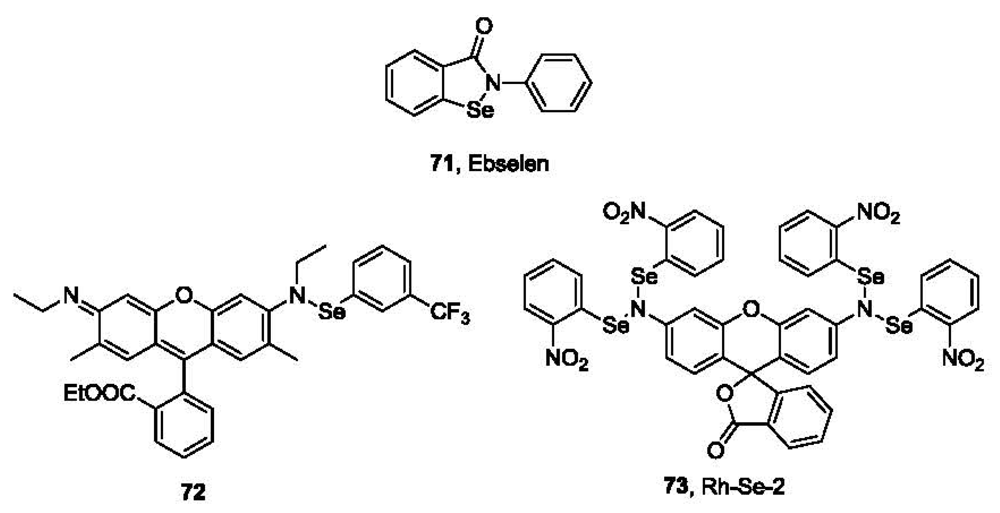
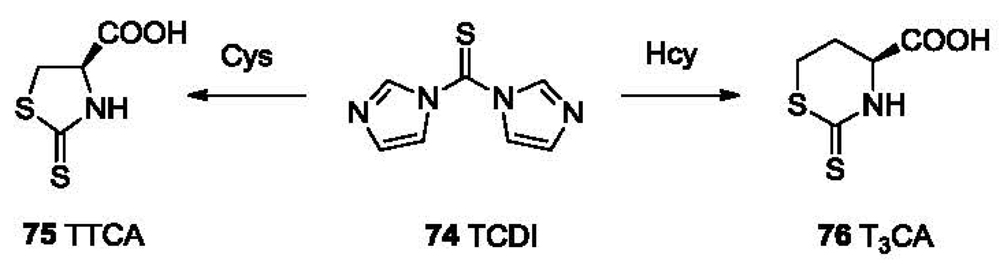





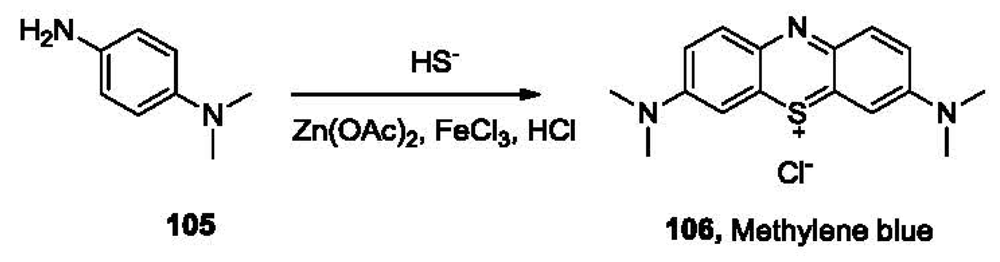
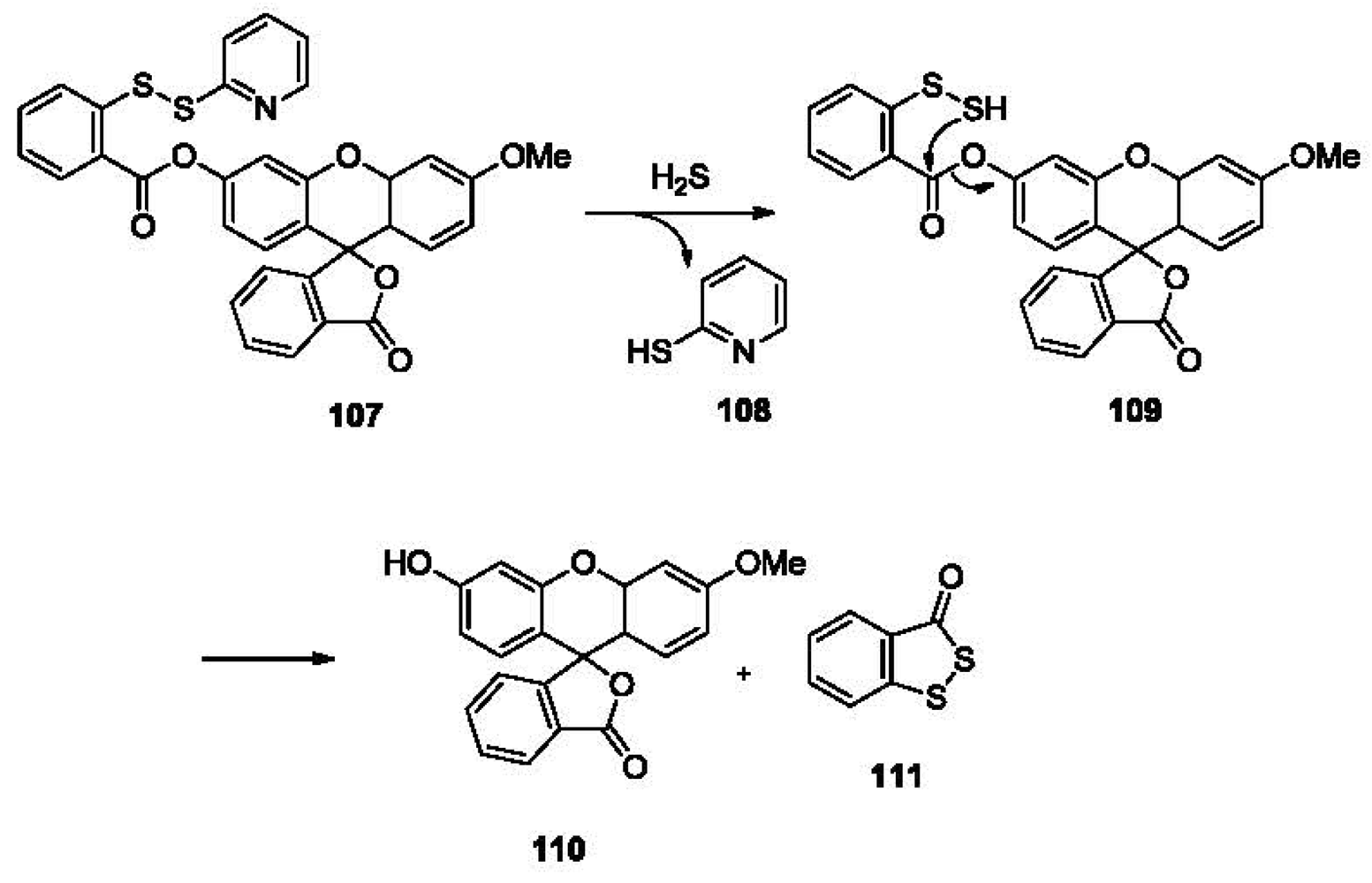

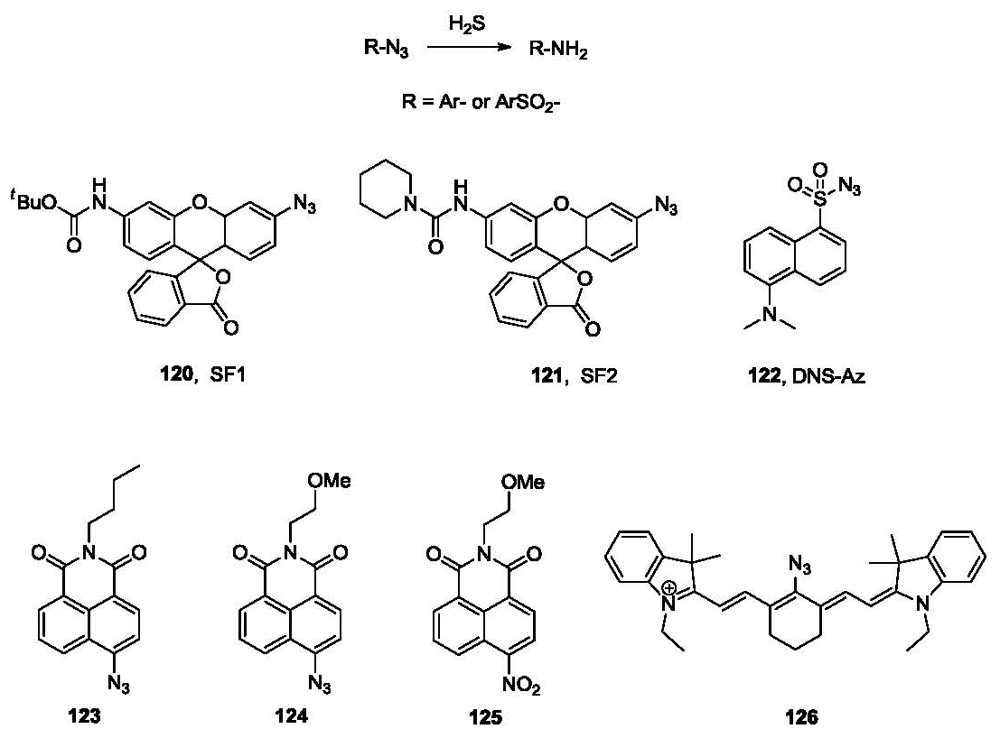
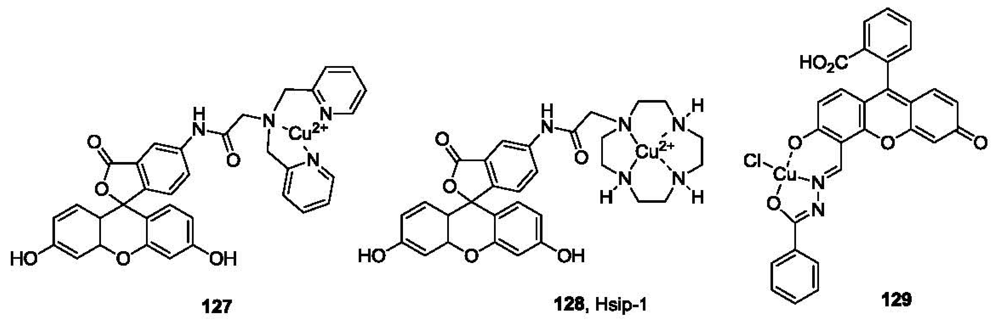

| Compound | λex/λem (nm) or Abs. (nm) | Reaction Time (min) | Reaction Temperature/pH | LOD | Biological Sample | Refs. |
|---|---|---|---|---|---|---|
| mBrB | 380/480 | 2–20 | 25 °C/7.4–8.9 | 0.5 pM | plasma | [57–60] |
| ABD-F | 380/510 | 5–60 | 37–60 °C /7.5–9.3 | 0.3 pM | plasma, cell | [61–63] |
| SBD-F | 380/510 | 60 | 60 °C/9.5–10.5 | 0.1–1.5 μM | plasma | [64,65] |
| 5-IAF | 494/521 | 15–120 | 25–40 °C/12.5 | 2–30 nM | plasma | [66–68] |
| CMPI | 310 | 15–30 | 25 °C/8.2 | 0.3–20 nM | urine | [69,70] |
| CMQT | 355 | 1–4 | 25 °C/7.5 | 50 nM | urine, plasma | [71–73] |
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Peng, H.; Chen, W.; Cheng, Y.; Hakuna, L.; Strongin, R.; Wang, B. Thiol Reactive Probes and Chemosensors. Sensors 2012, 12, 15907-15946. https://doi.org/10.3390/s121115907
Peng H, Chen W, Cheng Y, Hakuna L, Strongin R, Wang B. Thiol Reactive Probes and Chemosensors. Sensors. 2012; 12(11):15907-15946. https://doi.org/10.3390/s121115907
Chicago/Turabian StylePeng, Hanjing, Weixuan Chen, Yunfeng Cheng, Lovemore Hakuna, Robert Strongin, and Binghe Wang. 2012. "Thiol Reactive Probes and Chemosensors" Sensors 12, no. 11: 15907-15946. https://doi.org/10.3390/s121115907
APA StylePeng, H., Chen, W., Cheng, Y., Hakuna, L., Strongin, R., & Wang, B. (2012). Thiol Reactive Probes and Chemosensors. Sensors, 12(11), 15907-15946. https://doi.org/10.3390/s121115907






