The Indian Sundarban Mangrove Forests: History, Utilization, Conservation Strategies and Local Perception
Abstract
:1. Introduction
2. Study Area
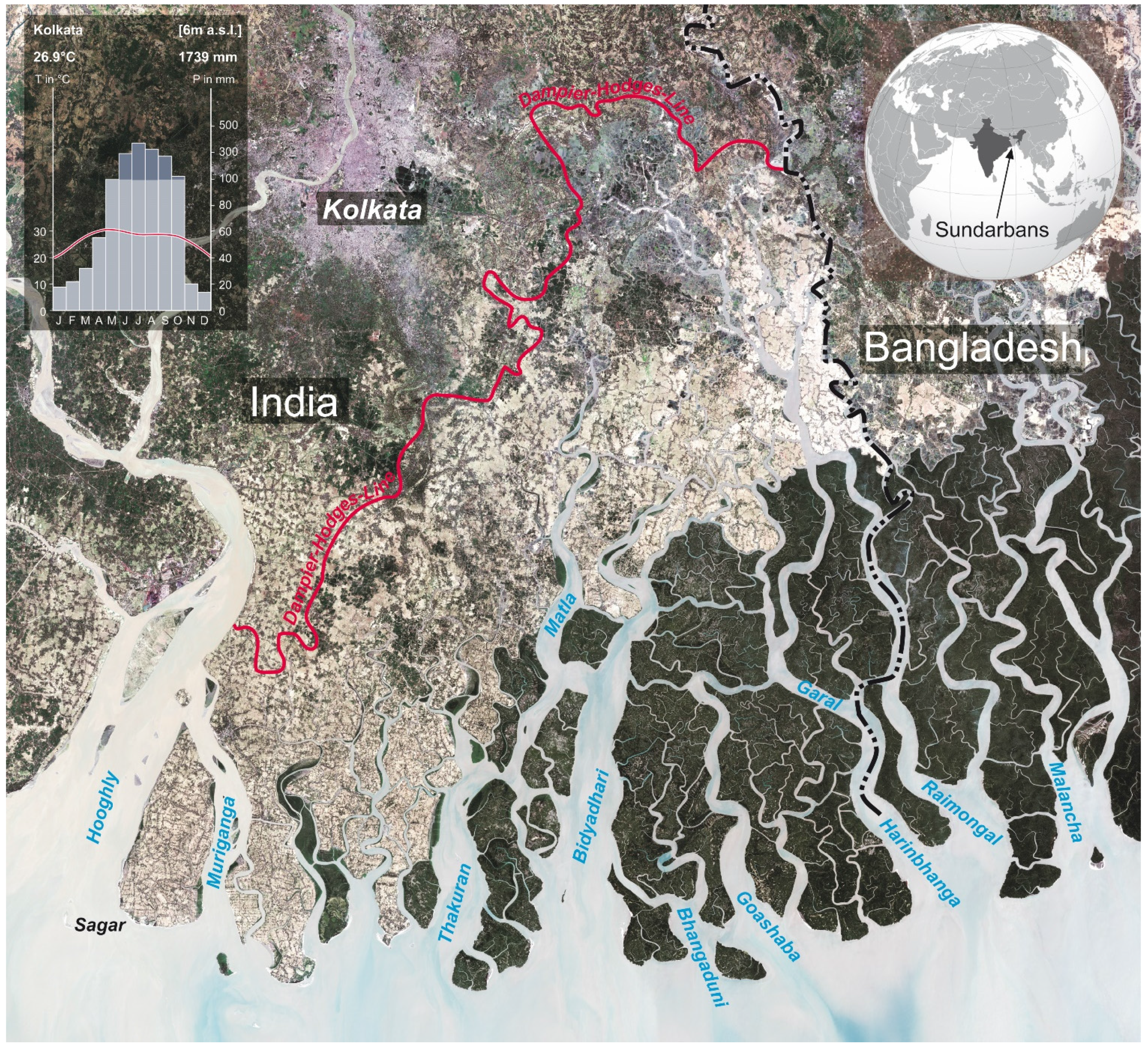
3. Data and Methods
| Maps | Year | Reference | Scale | |
|---|---|---|---|---|
| Rennel’s Map | 1776 | [49] | 60 Geogr. Miles to 1 Degree | |
| Hunter’s Map | 1873 | [50] | 16 Miles to 1 Inch | |
| Sensor Type * | Acquired Data | ID | Spatial Resolution [m × m²] | Spectral Resolution |
| Corona KH | 1968/02/06 | DS1045-2196DA128 | 2 | pan |
| DS1045-2196DA129 | ||||
| DS1045-2196DA130 | ||||
| DS1045-2196DA131 | ||||
| Landsat 5 TM | 1989/01/19 | LT41380451989019AAA02 | 30 | VIS, IR |
| Landsat 7 ETM | 2001/01/04 | LE71380452001004SGS00 | 30 | VIS, IR |
| Landsat 7 ETM | 2002/04/13 | LE71380452002103SGS00 | 30 | pan |
| LE71380442002103SGS00 | ||||
| Landsat 8 OLI | 2014/03/05 | LC81380452014064LGN00 | 30 | VIS, IR |
4. Historical Overview of the Sunderbans’ Development
“…the area was depopulated for all practical purposes. The forest reclaimed the previously inhabited area and when the British East India Company set up their headquarters at Calcutta in 1757, it was at the edge of the forest”[52]
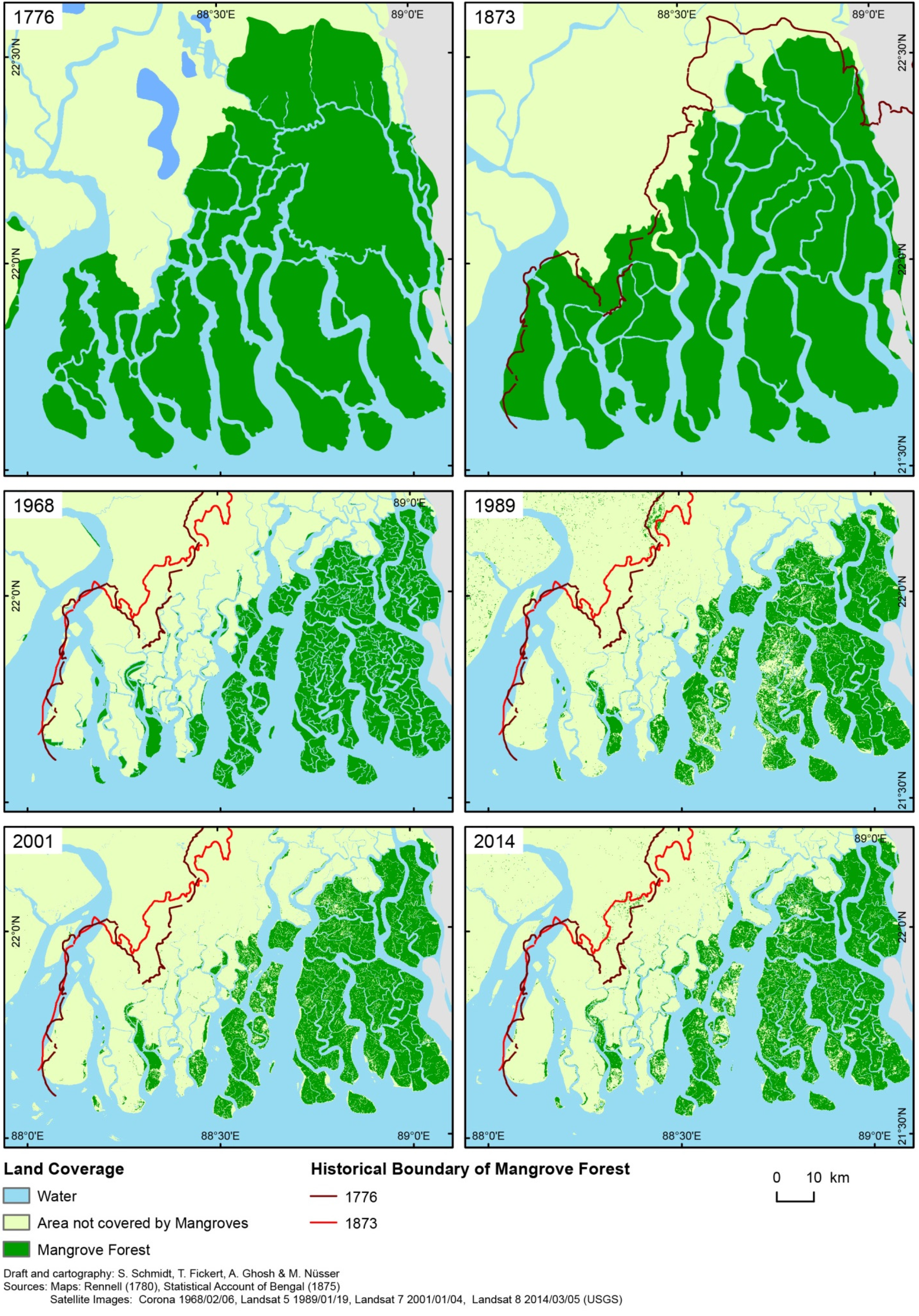
| Year of Observation | Mangrove Forest [km2] | Area Change in % | |
|---|---|---|---|
| relating to previous observation | per decade | ||
| 1776 | 6588 | ||
| 1873 | 6068 | −7.9 | −0.8 |
| 1968 | 2307 | −62.0 | −6.5 |
| 1989 | 1983 | −14.0 | −6.7 |
| 2001 | 1926 | −2.9 | −2.4 |
| 2014 | 1852 | −3.8 | −3.0 |
5. Post-Colonial Policies: Legislative Protection and Community Participation
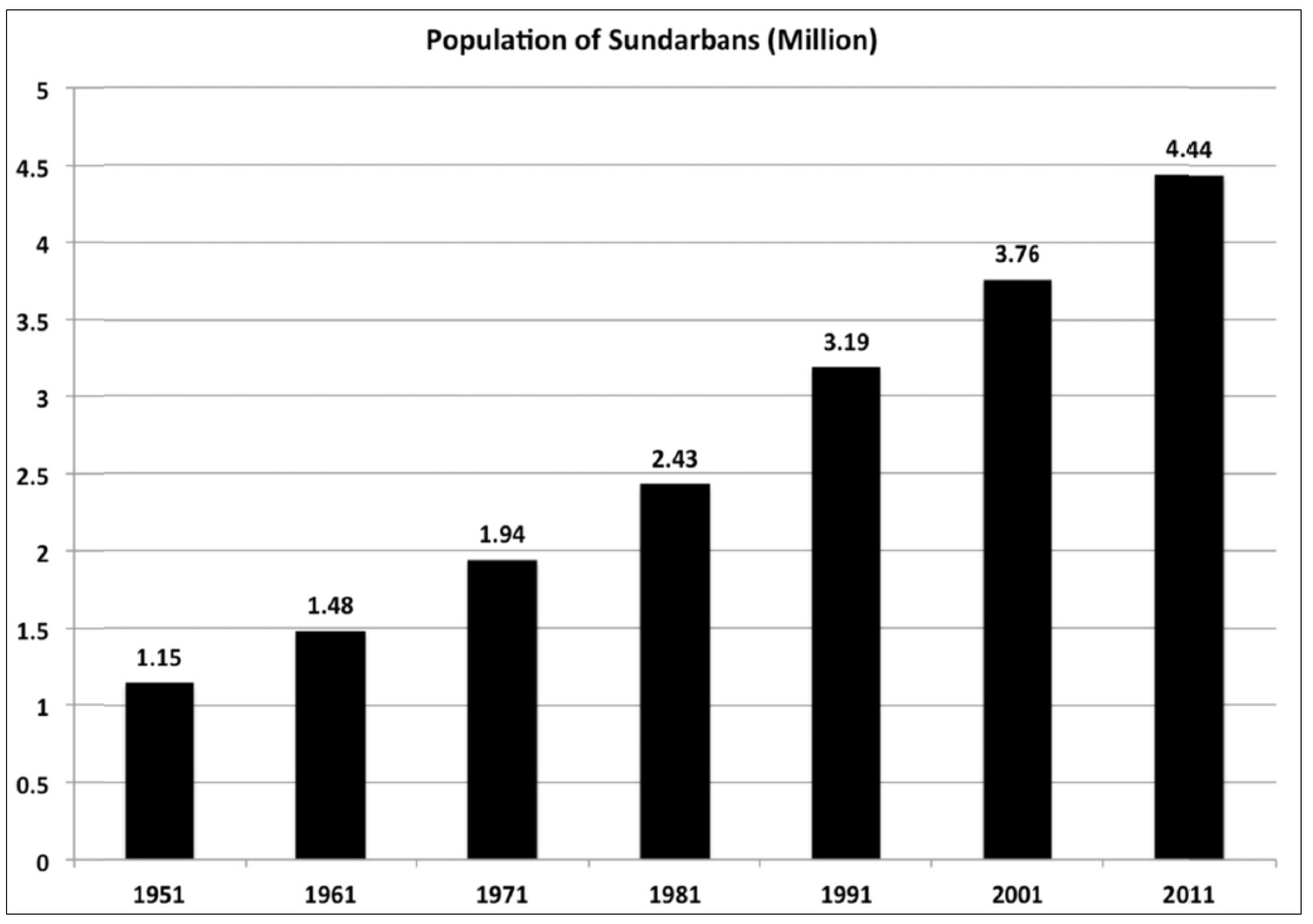
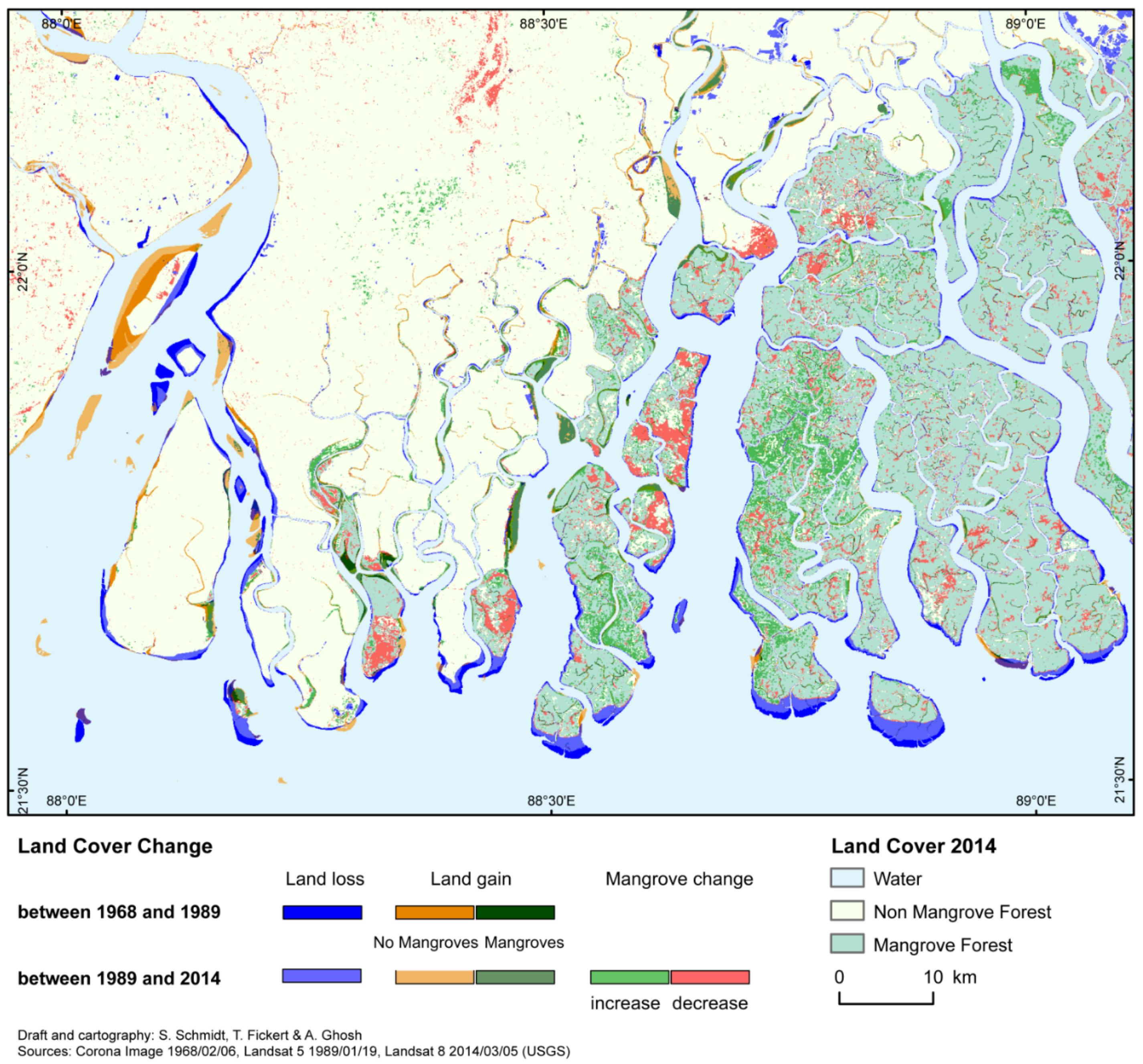
6. Internal Mangrove Dynamics—Climate Change and Human Impacts
| Differences Between Observations | |||||
|---|---|---|---|---|---|
| 1968–1989 [km²] | 1968–2014 [km²] | 1989–2001 [km²] | 2001–2014 [km²] | 1989–2014 [km²] | |
| Corona and Landsat | Landsat | ||||
| Total Land Loss | 136 | 299 | 74 | 263 | 213 |
| Total Land Gain | 163 | 270 | 219 | 60 | 159 |
| No-Mangroves | 123 | 163 | 204 | 55 | 107 |
| Mangroves | 40 | 107 | 15 | 5 | 52 |
| Mangrove Change | |||||
| Increase | 54 | 235 | 137 | 240 | |
| decrease | 475 | 154 | 220 | 262 | |
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saenger, P. Mangrove Ecology, Silviculture and Conservation; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Hogarth, P.J. The Biology of Mangroves and Seagrasses; Oxford University Press: Oxford, UK, 2007; p. 273. [Google Scholar]
- Ewel, K.C.; Twilley, R.R.; Ong, J.E. Different kinds of mangrove forests provide different goods and services. Glob. Ecol. Biogeogr. Lett. 1998, 7, 83–94. [Google Scholar] [CrossRef]
- Nagelkerken, I.; Blaber, S.J.M.; Bouillon, S.; Green, P.; Haywood, M.; Kirton, L.G.; Meynecke, J.-O.; Pawlik, J.; Penrose, H.M.; Sasekumar, A.; et al. The habitat function of mangroves for terrestrial and marine fauna: A review. Aquat. Bot. 2008, 89, 155–185. [Google Scholar] [CrossRef]
- Dahdouh-Guebas, F.; Jayatissa, L.P.; di Nitto, D.; Bosire, J.O.; Lo Seen, D.; Koedam, N. How effective were mangroves as a defence against the recent tsunami? Curr. Biol. 2005, 15, 443–447. [Google Scholar] [CrossRef]
- Danielsen, F.; Sørensen, M.K.; Olwig, M.F.; Selvam, V.; Parish, F.; Burgess, N.D.; Hiraishi, T.; Karunagaran, V.M.; Rasmussen, M.S.; Hansen, L.B.; et al. The Asian Tsunami: A protective role for coastal vegetation. Science 2005, 310, 643. [Google Scholar] [CrossRef] [PubMed]
- Kathiresan, K.; Rajendran, N. Coastal mangrove forests mitigated tsunami. Estuar. Coast. Shelf Sci. 2005, 65, 601–606. [Google Scholar] [CrossRef]
- Williams, N. Tsunami insight to mangrove value. Curr. Biol. 2005, 15, 73. [Google Scholar] [CrossRef] [PubMed]
- Mumby, P.J.; Hastings, A. The impact of ecosystem connectivity on coral reef resilience. J. Appl. Ecol. 2008, 45, 854–862. [Google Scholar] [CrossRef]
- Kristensen, E.; Bouillon, S.; Dittmar, T.; Marchand, C. Organic carbon dynamics in mangrove ecosystems: A review. Aquat. Bot. 2008, 89, 201–219. [Google Scholar] [CrossRef]
- Costanza, R.; Farber, S.C.; Maxwell, J. The valuation and management of wetland ecosystems. Ecol. Econ. 1989, 1, 335–361. [Google Scholar] [CrossRef]
- Costanza, R.; de Groot, R.; Sutton, P.; van der Ploeg, S.; Anderson, S.J.; Kubiszewski, I.; Farber, S.; Turner, R.K. Changes in the global value of ecosystem services. Glob. Environ. Change 2014, 26, 152–158. [Google Scholar] [CrossRef]
- Bann, C. The Economic Valuation of Mangroves: A Manual for Researchers; International Development Research Centre: Ottawa, ON, Canada, 1997; p. 54. [Google Scholar]
- Barbier, E.B. Valuing ecosystem services as productive inputs. Econ. Policy 2007, 49, 177–229. [Google Scholar]
- Walters, B.B.; Rönnbäck, P.; Kovacs, J.M.; Crona, B.; Hussain, S.A.; Badola, R.; Primavera, J.H.; Barbier, E.; Dahdouh-Guebas, F. Ethnobiology, socio-economics and management of mangrove forests: A review. Aquat. Bot. 2008, 89, 220–236. [Google Scholar] [CrossRef]
- Salem, M.E.; Mercer, D.E. The economic value of mangroves: A meta-analysis. Sustainability 2012, 4, 359–383. [Google Scholar] [CrossRef]
- Russi, D.; ten Brink, P.; Farmer, A.; Badura, T.; Coates, D.; Förster, J.; Kumar, R.; Davidson, N. The Economics of Ecosystems and Biodiversity for Water and Wetlands; IEEP: London, UK, 2013; p. 84. [Google Scholar]
- Valiela, I.; Bowen, J.L.; York, J.K. Mangrove forests: One of the world’s threatened major tropical environment. Bioscience 2001, 51, 807–815. [Google Scholar] [CrossRef]
- Ellison, A.M. Macroecology of mangroves: Large scale patterns and processes in tropical coastal forests. Trees Struct. Funct. 2002, 16, 181–194. [Google Scholar] [CrossRef]
- Duke, N.C.; Meynecke, J.-O.; Dittmann, S.; Ellison, A.M.; Anger, A.M.; Berger, U.; Cannicci, S.; Diele, K.; Ewel, K.C.; Field, C.D.; et al. A world without mangroves. Science 2007, 317, 41–42. [Google Scholar] [CrossRef] [PubMed]
- World Bank. Building Resilience for Sustainable Development of the Sundarbans: Strategy Report; World Bank Group: Washington, DC, USA, 2014; p. 290. [Google Scholar]
- Islam, M.T. Vegetation changes of Sundarbans based on Landsat Imagery analysis between 1975 and 2006. Landsc. Environ. 2014, 8, 1–9. [Google Scholar]
- Spalding, M.; Kainuma, M.; Collins, L. World Atlas of Mangroves; Earthscan: London, UK, 2010; p. 319. [Google Scholar]
- Gopal, B.; Chauhan, M. Biodiversity and its conservation in the Sundarban mangrove ecosystem. Aquat. Sci. 2006, 68, 338–354. [Google Scholar] [CrossRef]
- Banerjee, L.K. Sundarbans. In Floristic Diversity and Conservation Strategies in India, vol. V, Botanical Survey of India; Shing, N.P., Shing, K.P., Eds.; Ministry of Environment and Forests: New Delhi, India, 2002; pp. 2801–2829. [Google Scholar]
- Duke, N.C. Mangrove floristics and biogeography. In Tropical Mangrove Ecosystems; Robertson, A.I., Alongi, D.M., Eds.; American Geophysical Union: Washington, DC, USA, 1992; pp. 63–100. [Google Scholar]
- Barik, J.; Chowdhury, S. True mangrove species of Sundarbans delta, West Bengal, Eastern India. Check List 2014, 10, 329–334. [Google Scholar] [CrossRef]
- Goodbred, S.L.; Kuehl, S.A. The significance of large sediment supply, active tectonism, and eustasy on margin sequence development: Late Quaternary stratigraphy and evolution of the Ganges—Brahmaputra delta. Sediment. Geol. 2000, 133, 227–248. [Google Scholar] [CrossRef]
- Syvitski, J.P.M.; Kettner, A.J.; Overeem, I.; Hutton, E.W.H.; Hannon, M.T.; Brakenridge, G.R.; Day, J.; Vörösmarty, C.; Saito, Y.; Giosan, L.; et al. Sinking deltas due to human activities. Nat. Geosci. 2009, 2, 681–686. [Google Scholar] [CrossRef]
- Hanebuth, T.J.J.; Kudrass, H.R.; Linstädter, J.; Islam, B.; Zander, A.M. Rapid coastal subsidence in the central Ganges-Brahmaputra Delta (Bangladesh) since the 17th century deduced from submerged saltproducing kilns. Geology 2013, 41, 987–990. [Google Scholar] [CrossRef]
- Blasco, F.; Saenger, P.; Janodet, E. Mangroves as indicators of coastal change. Catena 1996, 27, 167–178. [Google Scholar] [CrossRef]
- IUCN. International Union for the Conservation of Nature—Bangaldesh: The Bangladesh Sundarbans: A Photo Real Sojourn; IUCN Bangladesh Country Office: Dhaka, Bangladesh, 2001. [Google Scholar]
- Banerjee, A.K. Forests of Sundarbans, Centenary Commemoration Volume, West Bengal Forests. Planning and Statistical Cell, Writer’s Building; Calcutta: Bengal, India, 1964; p. 188. [Google Scholar]
- Iftekhar, M.S. An overview of mangrove management strategies in three South Asian countries: Bangladesh, India and Sri Lanka. Int. For. Rev. 2008, 10, 38–51. [Google Scholar]
- Shams-Uddin, M.; Shah, M.A.R.; Khanom, S.; Nesha, M.K. Climate change impacts on the Sundarbans mangrove ecosystem services and dependent livelihoods in Bangladesh. Asian J. Conserv. Biol. 2013, 2, 152–156. [Google Scholar]
- De Groot, R.S.; Wilson, M.A.; Boumans, R.M.J. A typology for the classification, description and valuation of ecosystem functions, goods and services. Ecol. Econ. 2002, 41, 393–408. [Google Scholar] [CrossRef]
- Saenger, P. Mangroves: Sustainable management in Bangladesh. In Tropical Forestry 8, Silviculture in the Tropics; Günter, S., Weber, M., Stimm, B., Mosandl, R., Eds.; Springer: Berlin, Germany, 2011; pp. 339–347. [Google Scholar]
- Lillesand, T.M.; Kiefer, R.W.; Chipman, J.W. Remote Sensing and Image Interpretation; John Wiley & Sons: Hoboken, NJ, USA, 2004; p. 704. [Google Scholar]
- Dashora, A.; Lohani, B.; Malik, J.N. A repository of earth resource information—CORONA satellite programme. Curr. Sci. India 2007, 92, 926–932. [Google Scholar]
- Green, E.P.; Clark, C.D.; Mumby, P.J.; Edwards, A.J.; Ellis, A.C. Remote sensing techniques for mangrove mapping. Int. J. Remote Sens. 1998, 19, 935–956. [Google Scholar] [CrossRef]
- Giri, C.; Pengra, B.; Zhu, Z.; Singh, A.; Tieszen, L.L. Monitoring mangrove forest dynamics of the Sundarbans in Bangladesh and India using multi-temporal satellite data from 1973 to 2000. Estuar. Coast. Shelf Sci. 2007, 73, 91–100. [Google Scholar] [CrossRef]
- Lee, T.-M.; Yeh, H.-C. Applying remote sensing techniques to monitor shifting wetland vegetation: A case study of Danshui River estuary mangrove communities, Taiwan. Ecol. Eng. 2009, 35, 487–496. [Google Scholar] [CrossRef]
- Kuenzer, C.; Bluemel, A.; Gebhardt, S.; Vo Quov, T.; Dech, S. Remote sensing of mangrove ecosystems: A Review. Remote Sens. 2011, 3, 879–928. [Google Scholar] [CrossRef]
- Heumann, B.W. Satellite remote sensing of mangrove forests: Recent advances and future opportunities. Prog. Phys. Geogr. 2011, 35, 87–108. [Google Scholar] [CrossRef]
- Gray, D.; Zisman, S.; Corves, C. Mapping of the Mangroves of Belize; Technical Report; University of Edinburgh: Edinburgh, UK, 1990; p. 33. [Google Scholar]
- Eaton, R.M. Human settlement and colonization in the Sundarbans, 1200–1750. Agric. Hum. Values 1990, 7, 6–16. [Google Scholar] [CrossRef]
- O’Malley, L.S.S. Eastern Bengal District Gazetteers, Khulna; Calcutta: Bengal, India, 1908; pp. 193–194. [Google Scholar]
- Sarkar, S.C. Bengal’s southern frontier, 1757 to 1948. Stud. Hist. 2012, 28, 69–97. [Google Scholar] [CrossRef]
- Rennel, J. A Bengal Atlas Containing Maps of the Theatre of War and Commerce on that Side of Hindoostan; East India Company: London, UK, 1780. [Google Scholar]
- Hunter, W.W. Statistical Account of Bengal, 24 Parganas; Trübner: London, UK, 1876. [Google Scholar]
- Chakrabarty, R. Local people and the global tiger: An environmental history of the sundarbans. Glob. Environ. 2009, 3, 72–95. [Google Scholar]
- Anon. District Human Development Report, South 24 Parganas; Development and Planning Department, Government of West Bengal: Kolkata, India, 2010.
- Chakraborty, S.C. The Sundarbans—Terrain, legends, Gods & myths. Geogr. Rev. India 2005, 67, 1–11. [Google Scholar]
- Hunter, W.W. Statistical Account of Bengal; Parganas and Sundarbans: London, UK, 1885. [Google Scholar]
- Greenough, P.R. Hunter’s drowned land: An environmental fantasy of the Victorian Sundarbans. In Nature and the Orient: The Environmental History of South and Southeast Asia; Grove, R.H., Damodaran, V., Sangwan, S., Eds.; Oxford University Press: Delhi, India, 1998; pp. 237–272. [Google Scholar]
- Iqbal, I. The Bengal Delta: Ecology, State and Social Change, 1840–1943; Palgrave Macmillan: London, UK, 2011; p. 268. [Google Scholar]
- Danda, A.A. Surviving in the Sundarbans: Threats and Responses. Ph.D. Thesis, University of Twente, Enschede, The Netherlands, 2007; p. 199. [Google Scholar]
- DasGupta, R.; Shaw, R. Changing perspectives of mangrove management in India: An analytical overview. Ocean. Coast. Manag. 2013, 80, 107–118. [Google Scholar] [CrossRef]
- Iftekhar, M.S.; Islam, M.R. Degeneration of Bangladesh’s Sundarbans mangroves: A management issue. Int. For. Rev. 2004, 6, 123–135. [Google Scholar]
- Jalais, A. The Sundarbans: Whose world heritage site? Conserv. Soc. 2007, 5, 1–8. [Google Scholar]
- Ghosh, A. Living with Changing Climate—Impact, Vulnerability and Adaptation Challenges in Indian Sundarbans; Centre for Science and Environment: New Dehli, India, 2012; p. 108. [Google Scholar]
- Banerjee, A. Joint forest management in west Bengal. In Forests People and Power: The Political Ecology of Reform in South Asia; Springate-Baginski, O., Blaikie, P., Eds.; Routledge: London, UK, 2013; pp. 177–220. [Google Scholar]
- Raha, A.K.; Zaman, S.; Sengupta, K.; Bhattacharyya, S.B.; Raha, S.; Banerjee, K.; Mitra, A. Climate change and sustainable livelihood programme: A case study from Indian Sundarbans. J. Ecol. 2013, 107, 335–348. [Google Scholar]
- Sivaramakrishnan, K. Crafting the public sphere in the forests of West Bengal: Democracy, development, and political action. Am. Ethnol. 2000, 27, 431–461. [Google Scholar] [CrossRef]
- Raha, A.; Das, S.; Banerjee, K.; Mitra, A. Climate change impacts on Indian Sunderbans: A time series analysis (1924–2008). Biodivers. Conserv. 2012, 21, 1289–1307. [Google Scholar] [CrossRef]
- Jalais, A. Forest of Tigers: People, Politics and Environment in the Sundarbans; Routledge: London, UK, 2014; p. 268. [Google Scholar]
- Ostrom, E. A polycentric approach for coping with climate change. Ann. Econ. Financ. 2014, 15, 71–108. [Google Scholar]
- Mitra, A.; Banerjee, K.; Sengupta, K.; Gangopadhyay, A. Pulse of climate change in Indian Sundarbans: A myth or reality. Natl. Acad. Sci. Lett. 2009, 32, 19–25. [Google Scholar]
- Banerjee, K. Decadal change in the surface water salinity profile of Indian Sundarbans: A potential indicator of climate change. J. Mar. Sci. Res. Dev. 2013, 7. [Google Scholar]
- Iftekhar, M.S.; Saenger, P. Vegetation dynamics in the Bangladesh Sundarbans mangroves: A review of forest inventories. Wetl. Ecol. Manag. 2008, 16, 291–312. [Google Scholar] [CrossRef]
- Mitra, A.; Gangopadhyay, A.; Dube, A.; Schmidt, A.C.; Banerjee, K. Observed changes in water mass properties in the Indian Sundarbans (northwestern Bay of Bengal) during 1980–2007. Curr. Sci. India 2009, 97, 1445–1452. [Google Scholar]
- Gupta, A.; Basu, J.; Bhatta, A. “@110/hr”. In Down to Earth; Society for Environmental Communications: New Delhi, India, 2009; Volume 18. [Google Scholar]
- Singh, O.P. International variability and predictability of sea level along the Indian coast. Theor. Appl. Climatol. 2002, 72, 11–28. [Google Scholar] [CrossRef]
- Unnikrishnan, A.S.; Kumar, M.R.R.; Sindhu, B. Tropical cyclones in the Bay of Bengal and extreme sea-level projections along the east coast of India in a future climate scenario. Curr. Sci. India 2011, 101, 327–331. [Google Scholar]
- Parth Sarthi, P.; Agrawal, A.; Rana, A. Possible future changes in cyclonic storms in the Bay of Bengal, India under warmer climate. Int. J. Climatol. 2014, 35, 11. [Google Scholar]
- Harun-or-Rashid, S.; Biswas, S.R.; Böcker, R.; Kruse, M. Mangrove community recovery potential after catastrophic disturbances in Bangladesh. For. Ecol. Manag. 2009, 257, 923–930. [Google Scholar] [CrossRef]
- Gopinath, G.; Seralathan, P. Rapid erosion of the coast of Sagar island, West Bengal, India. Environ. Geol. 2005, 48, 1058–1067. [Google Scholar] [CrossRef]
- Dinesh Kumar, P.K.; Gopinath, G.; Laluraj, C.M.; Seralathan, P.; Mitra, D. Change detection studies of Sagar island, India, using Indian remote sensing satellite 1C linear imaging self-scan sensor III data. J. Coast. Res. 2007, 23, 1498–1502. [Google Scholar] [CrossRef]
- Das, M. Deformation of the Jambudwip island of Sundarban region, Eastern India. Int. J. Geomat. Geosci. 2014, 5, 9–18. [Google Scholar]
- Hazra, S.; Ghosh, T.; DasGupta, R.; Sen, G. Sea level and associated changes in the Sundarbans. Sci. C. 2002, 68, 309–321. [Google Scholar]
- Nandy, S.; Bandopadhyay, S. Trend of sea level change in the Hugli estuary, India. Indian J. Geo-Mar. Sci. 2011, 40, 802–812. [Google Scholar]
- DasGupta, S.; Huq, M.; Khan, Z.H.; Ahmed, M.M.Z.; Mukherjee, N.; Khan, M.F.; Pandey, K. Cyclones in a changing climate: The case of Bangladesh. Clim. Dev. 2014, 6, 96–110. [Google Scholar] [CrossRef]
- Cornforth, W.A.; Fatoyinbo, T.E.; Freemantle, T.P.; Pettorelli, N. Advanced land observing satellite phased array type L-Band SAR (ALOS PALSAR) to inform the conservation of mangroves: Sundarbans as a case study. Remote Sens. 2013, 5, 224–237. [Google Scholar] [CrossRef]
- Ganguly, D.; Mukhopadhyay, A.; Pandey, R.K.; Mitra, D. Geomorphological study of Sundarban deltaic estuary. J. Indian Soc. Remote Sens. 2006, 34, 431–435. [Google Scholar] [CrossRef]
- Hazra, S.; Samanta, K.; Mukhopadhyay, A.; Akhand, A. Temporal Change Detection (2001–2008) of the Sundarban; Unpublished Report; WWF-India: New Delhi, India, 2010. [Google Scholar]
- Ghosh, T.; Hajra, R.; Mukhopadhyay, A. Island erosion and afflicted population: Crisis and policies to handle climate change. In International Perspectives on Climate Change; Filho, W.L., Alves, F., Caeiro, S., Azeiteiro, U.M., Eds.; Springer: Berlin, Germany, 2014; pp. 217–225. [Google Scholar]
- Mitra, A.; Banerjee, K. Trace elements in edible shellfish species from the lower Gangetic delta. Ecotoxicol. Environ. Saf. 2011, 74, 1512–1517. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A. A comprehensive report on sundry (Heritiera fomes) trees with particular reference to top dying in the Sundarbans. In Seminar on Top Dying of Sundri (Heritiera fomes) Trees; Rahman, M.A., Khandakar, M.A., Ahmed, F.U., Ali, M.O., Eds.; Bangladesh Agricultural Research Council: Dhaka, Bangladesh, 1990; p. 256. [Google Scholar]
- Zaman, S.; Bhattacharyya, S.B.; Pramanick, P.; Raha, A.K.; Chakraborty, S.; Mitra, A. Rising water salinity: A threat to mangroves of Indian Sundarbans. In Water Insecurity: A Social Dilemma; Abedin, M.A., Habiba, U., Shaw, R., Eds.; Emerald Group Publishing Limited: Bingley, UK, 2013; pp. 167–183. [Google Scholar]
- Selvam, V. Environmental classification of mangrove wetlands of India. Curr. Sci. India 2003, 84, 757–765. [Google Scholar]
- Loucks, C.; Barber-Meyer, S.; Hossain, M.A.A.; Barlow, A.; Chowdhury, R.M. Sea level rise and tigers: Predicted impacts to Bangladesh’s Sundarbans mangroves. Clim. Change 2010, 9, 291–298. [Google Scholar] [CrossRef]
- Siddiqi, N.A. Mangrove Forestry in Bangladesh; University of Chittagong: Chittagong, Bangladesh, 2001. [Google Scholar]
- Bhattacharya, S. Sundarban—Dying a slow death. The Hindu Survey of Environment, 1998; 89–94. [Google Scholar]
- Rudra, K. Changing river courses in the western part of the Ganga–Brahmaputra delta. Geomorphology 2014, 227, 87–100. [Google Scholar] [CrossRef]
- Bandyopadhyay, S.; Bandyopadhyay, M.K. Retrogradation of the western Ganga-Brahmaputra delta: Possible reasons. Natl. Geogr. 1996, 31, 105–128. [Google Scholar]
- Inman, M. Working with water. Nat. Rep. Clim. Change 2010. [Google Scholar] [CrossRef]
- Kazmierczak, A.; Carter, J. Adaptation to Climate Change Using Green and Blue Infrastructure. A Database of Case Studies; GraBS Project; University of Manchester: Manchester, UK, 2010. [Google Scholar]
- Dinda, A. Evaluation of eco-tourism activity: A case study of Sundarban tiger reserve. In Man in Biosphere: A Case Study of Sundarban Biosphere Reserve; Gyan: New Delhi, India, 2007. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghosh, A.; Schmidt, S.; Fickert, T.; Nüsser, M. The Indian Sundarban Mangrove Forests: History, Utilization, Conservation Strategies and Local Perception. Diversity 2015, 7, 149-169. https://doi.org/10.3390/d7020149
Ghosh A, Schmidt S, Fickert T, Nüsser M. The Indian Sundarban Mangrove Forests: History, Utilization, Conservation Strategies and Local Perception. Diversity. 2015; 7(2):149-169. https://doi.org/10.3390/d7020149
Chicago/Turabian StyleGhosh, Aditya, Susanne Schmidt, Thomas Fickert, and Marcus Nüsser. 2015. "The Indian Sundarban Mangrove Forests: History, Utilization, Conservation Strategies and Local Perception" Diversity 7, no. 2: 149-169. https://doi.org/10.3390/d7020149
APA StyleGhosh, A., Schmidt, S., Fickert, T., & Nüsser, M. (2015). The Indian Sundarban Mangrove Forests: History, Utilization, Conservation Strategies and Local Perception. Diversity, 7(2), 149-169. https://doi.org/10.3390/d7020149







