Mangroves on the Edge: Anthrome-Dependent Fragmentation Influences Ecological Condition (Turbo, Colombia, Southern Caribbean)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
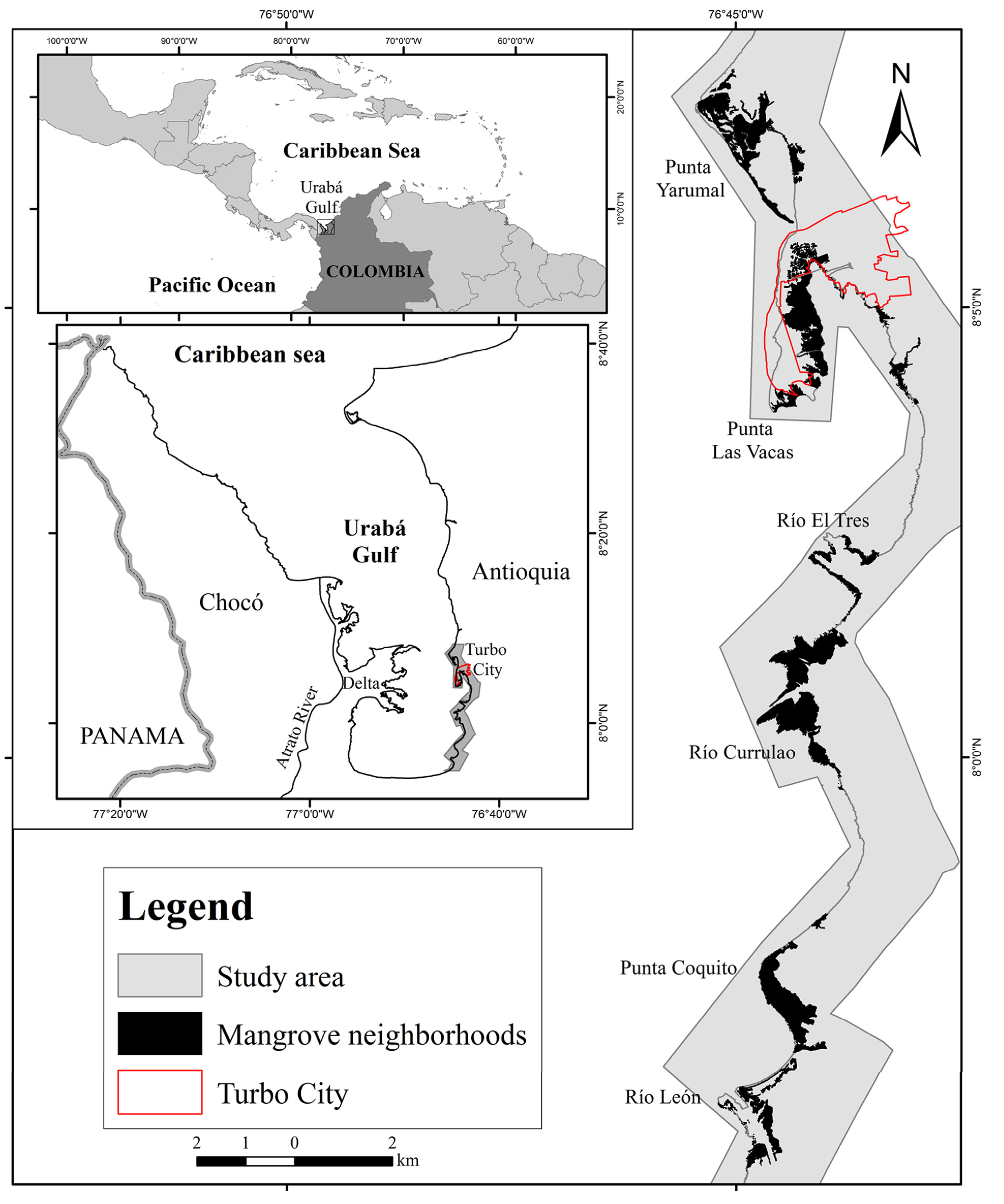
2.2. Spatial Data, GIS Methods and Mangrove Change Calculations
- Peri-urban: Equivalent to “Dense settlements”, where significant urban area is found. The landscape is a mixture of private and public areas, but “green areas”, “lowlands” and “inter-tidal lands” are public domain.
- Rural: Equivalent to “Croplands or Rangelands”, where dense agricultural or pastoral land cover are dominant, respectively. The “green elements” of the landscape are a mixture of private and public lands.
- Military-protected: Equivalent to “Semi-natural”, where forests with minor human presence occur. It is mostly dominated by native tree species, and land-tenure is public.
- Wild: Equivalent to “Wildlands”, where no land with human populations, agriculture or pastures occur. The land-tenure is public, mostly dominated by coastal freshwater wetlands and mangroves.
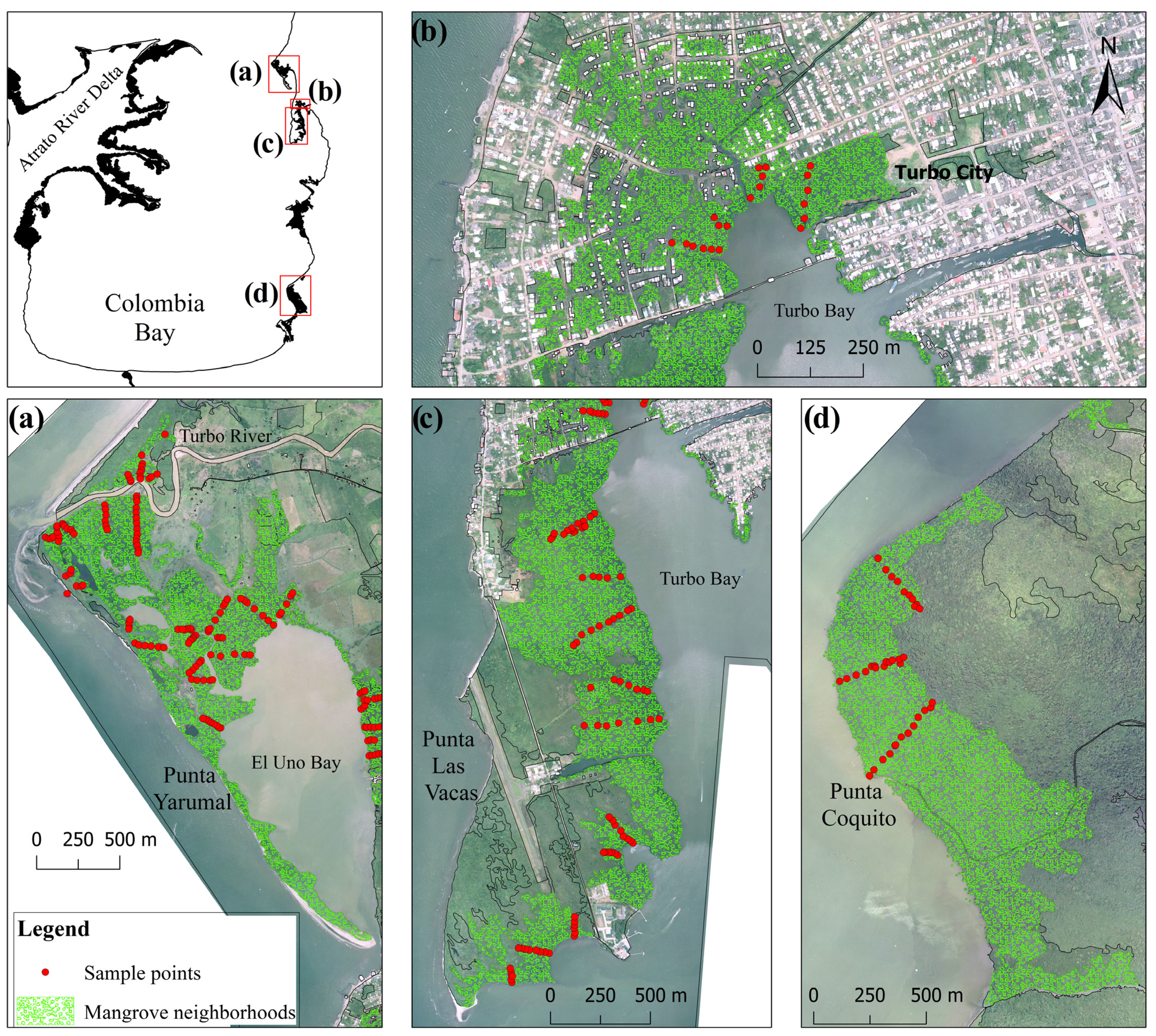
2.3. Mangrove Neighborhood Fragmentation
2.4. Mangrove Ecological Condition: Vegetation, Anthropogenic Disturbance Index (ADI) and Dominant Snail Presence
- Trampling (T): Presence of human and livestock footprints.
- Logging (L): Evidence of selective logging on mangrove species, such as stumps and downed wood and logs.
- Wastes (W): Presence of solid wastes on the forest floor and entangled on the roots.
- Structures (S): Evidence of human modifications of mangrove hydrology and topography (diggings, infillings, cement canals and pipelines), and other structures related to pastoral activities (fences and troughs).
2.5. Statistical Methods
3. Results and Discussion
3.1. Deforestation Rates
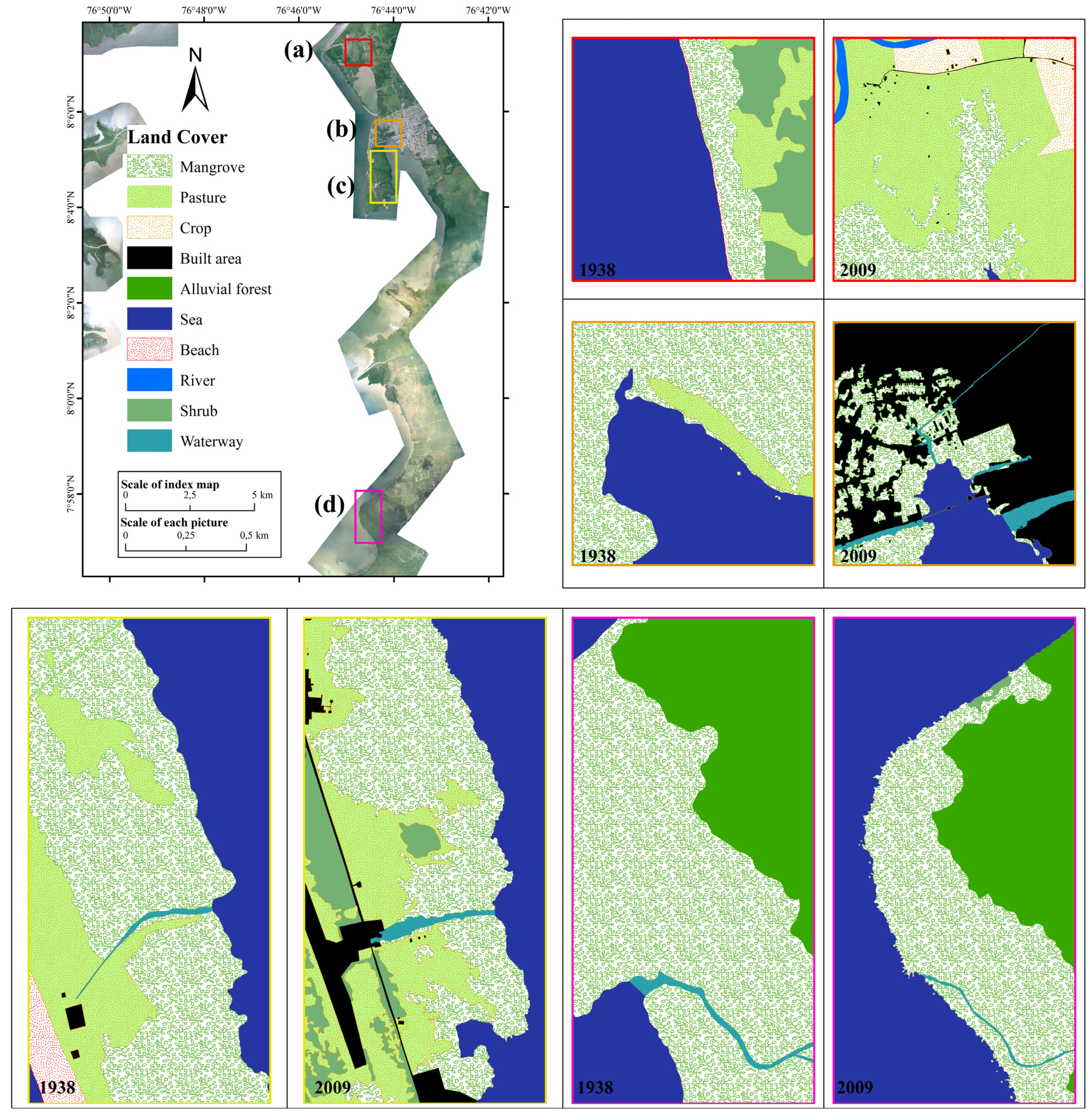
| Context | Area 1938 (km2) | Area 2009 (km2) | r (%·year−1) |
|---|---|---|---|
| Rural | 0.15 | 0.25 | 0.8 |
| Peri-urban | 0.55 | 0.24 | −1.2 |
| Military | 1.01 | 0.77 | −0.4 |
| Wild | 1.18 | 0.66 | −0.8 |
3.2. Mangrove Neighborhood Fragmentation
| Variable | Context | |||
|---|---|---|---|---|
| Peri-urban | Rural | Military | Wild | |
| Agricultural edge (km) | 0 | 1.6 | 0 | 0 |
| Urban edge (km) | 17.0 | 0.4 | 0.4 | 0 |
| Pasture edge (km) | 0 | 9.8 | 5.4 | 1.0 |
| Total edge (km) | 20.2 | 29.3 | 18.6 | 14.5 |
| Anthropogenic edge (%) | 84.3 | 40.2 | 31.3 | 6.6 |
| Total number of patches | 52 | 15 | 5 | 5 |
| Patch density (patches/ km2) | 237.3 | 17.0 | 4.9 | 5.5 |
| Mean patch area (km2) | <0.1 | 0.1 | 0.2 | 0.2 |
| Total neighborhood area (km2) | 0.2 | 0.9 | 1.0 | 0.9 |
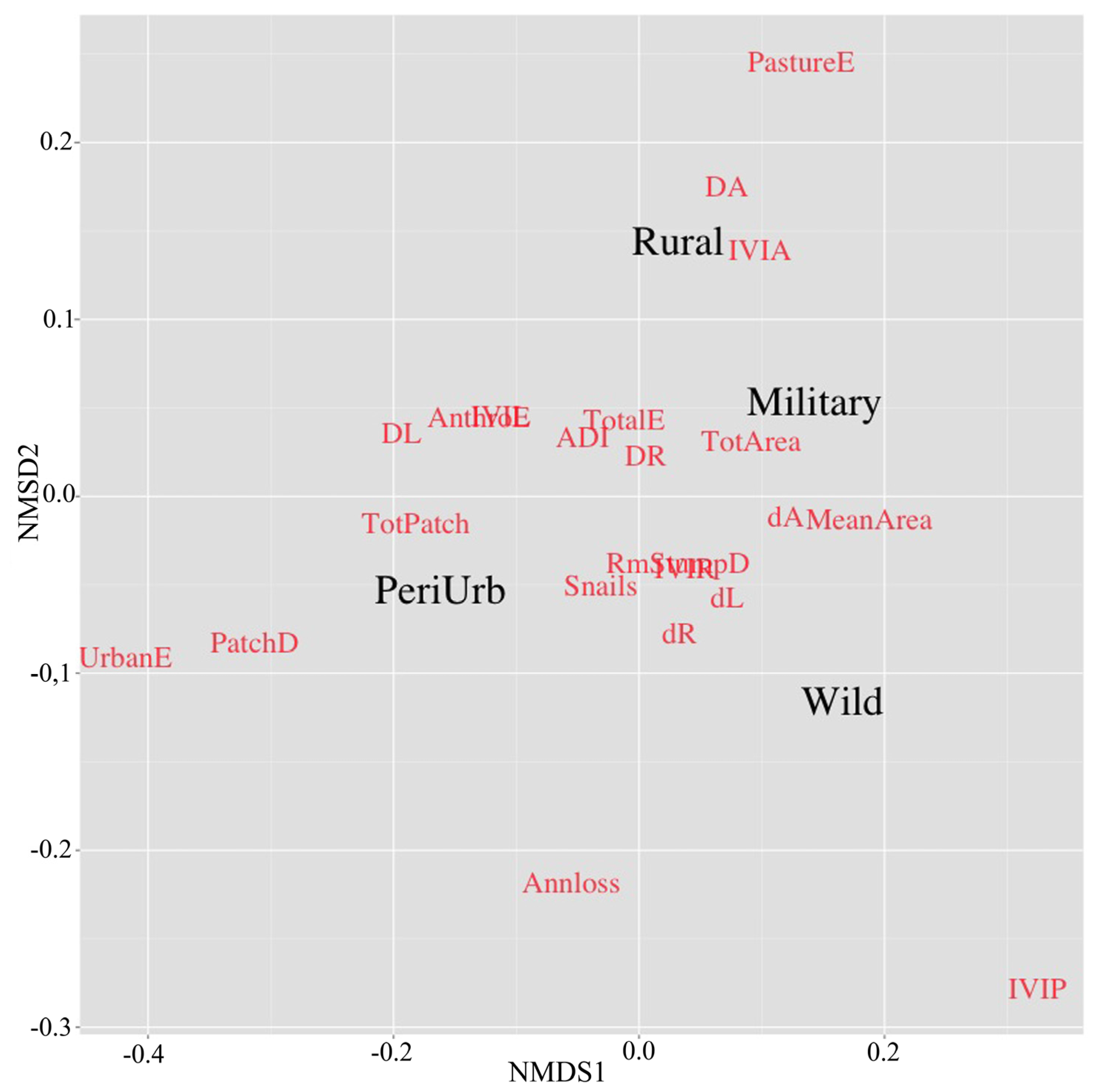
3.3. Fragmentation and Anthropogenic Disturbance Influence on Ecological Condition
3.3.1. ADI Relative to Context
| Species | Peri-urban | Rural | Military | Wild | ||||
|---|---|---|---|---|---|---|---|---|
| Number | Diameter (cm) | Number | Diameter (cm) | Number | Diameter (cm) | Number | Diameter (cm) | |
| Rhizophora mangle | 4 | 7.7 (6–9.5) | 60 | 6.7 (4.1–11.4) | 65 | 8.5 (5–15.6) | 14 | 12.5 (9–23) |
| Laguncularia racemosa | 22 | 8.6 (6–17.8) | 68 | 7.5 (5.1–15.6) | 19 | 11 (5–20) | 0 | N.A. |
| Avicennia germinans | 0 | N.A. | 4 | 23.0 (6.3–37.2) | 3 | 9.4 (5.4–14) | 1 | 47 |
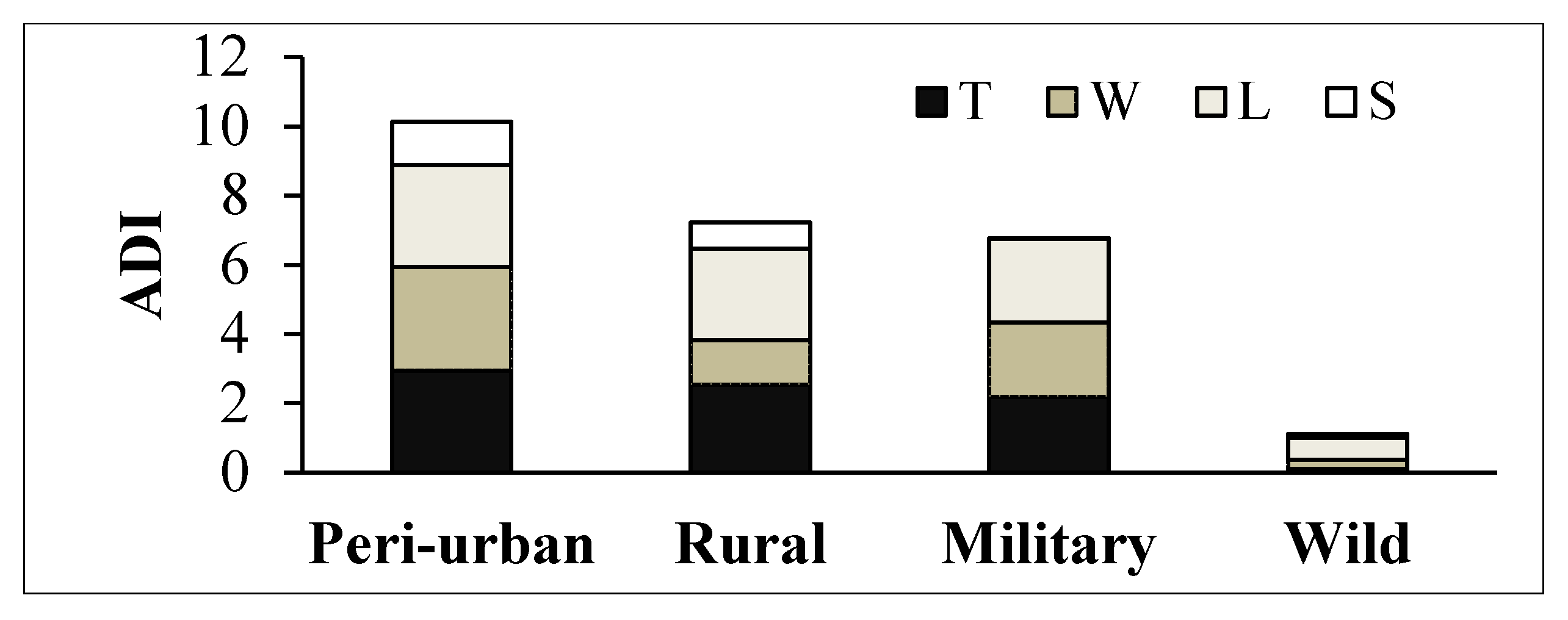
3.3.2. Mangrove Vegetation Relative to ADI
| Context | Species | DBH (cm) (mean ± s.d.) | Density (trees/0.1 ha) | Basal area (m2/0.1 ha) | Frequency (%) | IVI (%) |
|---|---|---|---|---|---|---|
| Peri-urban | Rm | 11.0 ± 6 | 69.7 ± 42.1 | 5.3 ± 6.9 | 19 ± 5 | 35 ± 14 |
| Lr | 6.0 ± 4.4 | 119.6 ± 56 | 3.6 ± 2.1 | 79 ± 5 | 64 ± 15 | |
| Ag | 3.5 * | 8.2 * | <0.01 * | 6 * | 3 * | |
| Rural | Rm | 5.5 ± 8.4 | 63.8 ± 33.5 | 2.9 ± 2.6 | 80 ± 18 | 35 ± 13 |
| Lr | 8.3 ± 8.3 | 48.6 ± 38 | 7.9 ± 12.4 | 67 ± 17 | 33 ± 19 | |
| Ag | 16.3 ± 8.1 | 35.0 ± 23.5 | 8.1 ± 11.8 | 55 ± 24 | 32 ± 17 | |
| Military | Rm | 10.5 ± 6.3 | 96.2 ± 67.1 | 16.7 ± 6.9 | 67 ± 13 | 78 ± 13 |
| Lr | 8.0 ± 6.3 | 9.9 ± 17.1 | 0.7 ± 2 | 13 ± 15 | 8 ± 12 | |
| Ag | 17.5 ± 6.3 | 13.3 ± 17.1 | 1.3 ± 2.1 | 18 ± 14 | 13 ± 13 | |
| Pr | 10.1 ± 6.2 | 1.4 ± 3.3 | 0.03 ± 0.08 | 2 ± 4 | 1 ± 3 | |
| Wild | Rm | 18.6 ± 16 | 47.9 ± 21.8 | 71.0 ± 6.5 | 56 ± 5 | 70 ± 10 |
| Lr | 20.8 ± 19.1 | 4.4 ± 4.1 | 1.4 ± 2.5 | 12 ± 10 | 9 ± 11 | |
| Ag | 29.5 ± 18.8 | 3.3 ± 3.1 | 0.9 ± 2.5 | 9 ± 12 | 7 ± 9 | |
| Pr | 11.9 ± 17.9 | 12.8 ± 11.1 | 1.8 ± 2.4 | 23 ± 20 | 14 ± 12 |
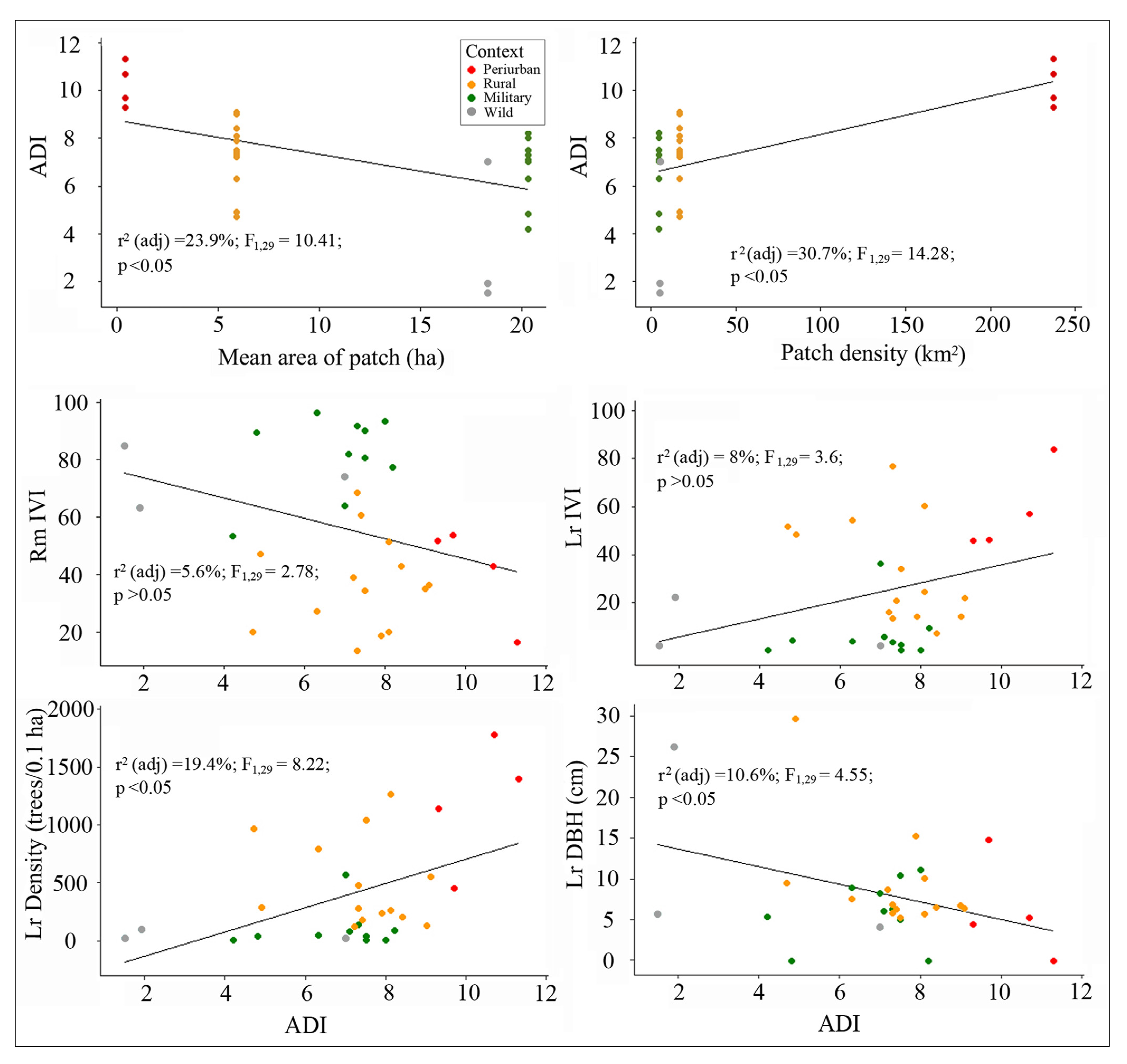
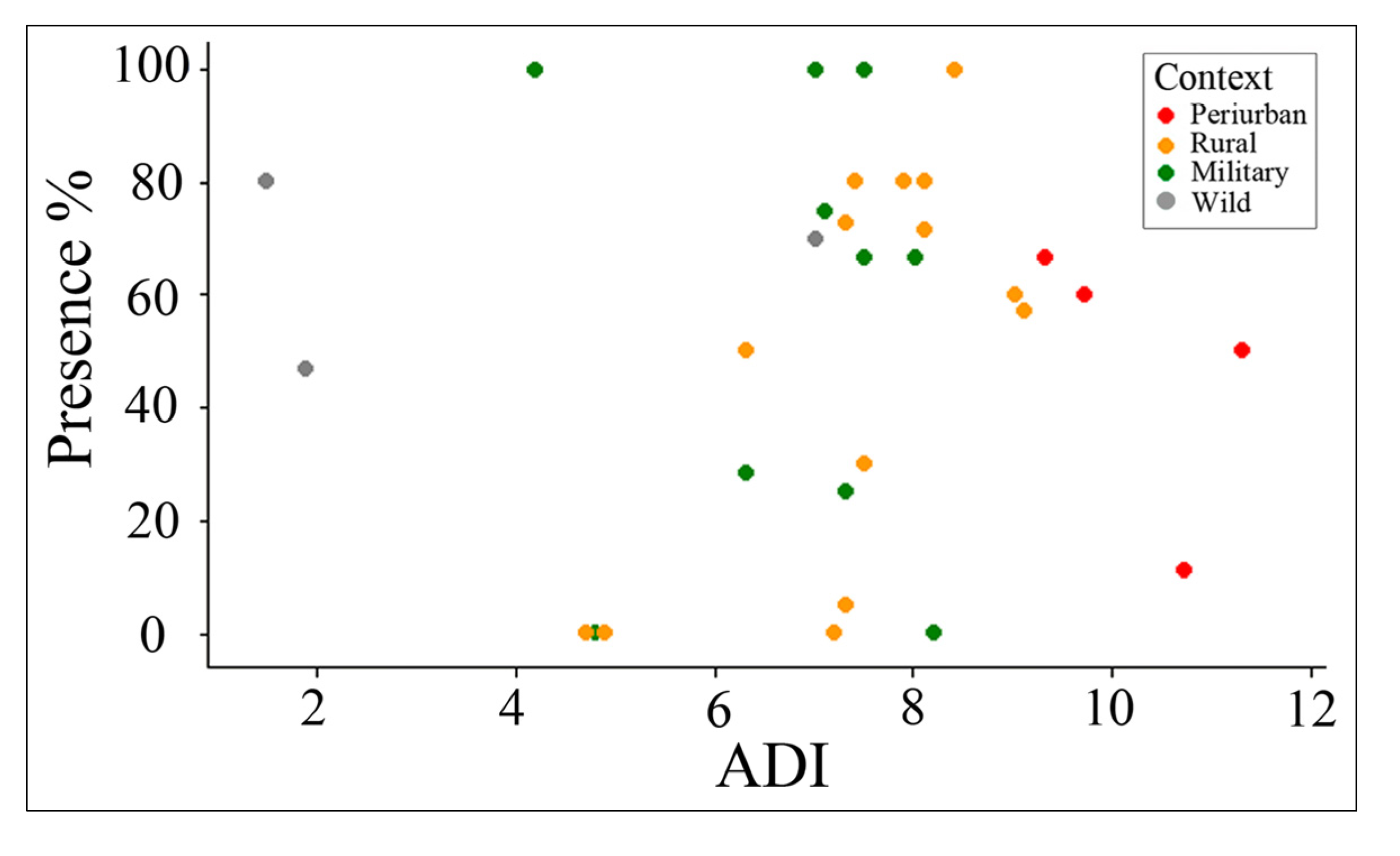
3.3.3. Snail Presence Relative to ADI
4. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Malhi, Y.; Gardner, T.A.; Goldsmith, G.R.; Silman, M.R.; Zelazowski, P. Tropical forests in the Anthropocene. Ann. Rev. Environ. Resour. 2014, 39, 125–159. [Google Scholar] [CrossRef]
- Lugo, A.E.; Medina, E.; McGinley, K. Issues and challenges of mangrove conservation in the Anthropocene. Madera y Bosques 2014, 20, 11–38. [Google Scholar]
- Ellis, E.C.; Ramankutty, N. Putting people in the map: Anthropogenic biomes of the world. Front. Ecol. Environ. 2008, 6, 439–447. [Google Scholar] [CrossRef]
- Ellis, E.C. Anthropogenic transformation of the terrestrial biosphere. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 2011, 369, 1010–1035. [Google Scholar] [CrossRef] [PubMed]
- Ellis, E.C.; Antill, E.C.; Kreft, H. All Is Not Loss: Plant Biodiversity in the Anthropocene. PLoS ONE 2012, 7, e30535. [Google Scholar] [CrossRef] [PubMed]
- DeFries, R.S.; Ellis, E.C.; Chapin, F.S.; Matson, P.A.; Turner, B.L.; Agrawal, A.; Crutzen, P.J.; Peter, C.F.; Kareiva, P.M.; Lambin, E.; et al. Planetary opportunities: A social contract for global change science to contribute to a sustainable future. BioScience 2012, 62, 603–606. [Google Scholar]
- Ellis, E.C. Sustaining biodiversity and people in the world’s anthropogenic biomes. Curr. Opin. Environ. Sustain. 2013, 5, 368–372. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Higgs, E.; Hall, C.M.; Bridgewater, P.; Chapin, F.S., III; Ellis, E.C.; Ewel, J.J.; Hallett, L.M.; Harris, J.; Hulvey, K.B.; et al. Managing the whole landscape: Historical, hybrid, and novel ecosystems. Front. Ecol. Environ. 2014, 12, 557–564. [Google Scholar] [CrossRef]
- Martin, L.J.; Quinn, J.E.; Ellis, E.C.; Shaw, M.R.; Dorning, M.A.; Hallett, L.M.; Heller, N.E.; Hobbs, R.J.; Kraft, C.E.; Law, E.; et al. Conservation opportunities across the world’s anthromes. Divers. Distrib. 2014, 20, 745–755. [Google Scholar] [CrossRef]
- Ellis, E.C.; Haff, P.K. Earth science in the Anthropocene: New epoch, new paradigm, new responsibilities. Eos, Trans. Am. Geophys. Union 2009, 90, 473. [Google Scholar] [CrossRef]
- Macintosh, D.J.; Ashton, E.C.; Havanon, S. Mangrove rehabilitation and intertidal biodiversity: A study in the Ranong mangrove ecosystem, Thailand. Estuar. Coast. Shelf Sci. 2002, 55, 331–345. [Google Scholar] [CrossRef]
- Primavera, J.H.; Esteban, J.M.A. A review of mangrove rehabilitation in the Philippines: Successes, failures and future prospects. Wetl. Ecol. Manag. 2008, 16, 345–358. [Google Scholar] [CrossRef]
- Ellison, A.M.; Farnsworth, E.J. Anthropogenic disturbance of Caribbean mangrove ecosystems: Past impacts, present trends, and future predictions. Biotropica 1996, 28, 549–565. [Google Scholar] [CrossRef]
- Laurance, W.F.; Useche, D.C.; Rendeiro, J.; Kalka, M.; Bradshaw, C.J.A.; Sloan, S.P.; Laurance, S.G.; Campbell, M.; Abernethy, K.; Alvarez, P.; et al. Averting biodiversity collapse in tropical forest protected areas. Nature 2012, 489, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Stein, B.A.; Scott, C.; Benton, N. Federal lands and endangered species: The role of military and other federal lands in sustaining biodiversity. Bioscience 2008, 58, 339–347. [Google Scholar] [CrossRef]
- Cohn, J.P. New defenders of wildlife. BioScience 1996, 46, 1–14. [Google Scholar] [CrossRef]
- Castilla, J.C. Roles of experimental marine ecology in coastal management and conservation. J. Exp. Mar. Biol. Ecol. 2000, 250, 3–21. [Google Scholar] [CrossRef]
- Bosire, J.O.; Kaino, J.J.; Olagoke, A.O.; Mwihaki, L.M.; Ogendi, G.M.; Kairo, J.G.; Berger, U.; Macharia, D. Mangroves in peril: Unprecedented degradation rates of peri-urban mangroves in Kenya. Biogeosciences Discuss 2013, 10, 16371–16404. [Google Scholar] [CrossRef]
- Martinuzzi, S.; Gould, W.A.; Lugo, A.E.; Medina, E. Conversion and recovery of Puerto Rican mangroves: 200 years of change. For. Ecol. Manag. 2009, 257, 75–84. [Google Scholar] [CrossRef]
- Fahrig, L. Effects of habitat fragmentation on biodiversity. Ann. Rev. Ecol. Evol. Syst. 2003, 34, 487–515. [Google Scholar] [CrossRef]
- Ries, L.; Fletcher, R.J., Jr.; Battin, J.; Sisk, T.D. Ecological responses to habitat edges: Mechanisms, models, and variability explained. Ann. Rev. Ecol. Evol. Syst. 2004, 35, 491–522. [Google Scholar] [CrossRef]
- Friess, D.A.; Phelps, J.; Leong, R.C.; Lee, W.K.; Wee, A.K.S.; Sivasothi, N.; Oh, R.R.Y.; Webb, E.L. Mandai mangrove, Singapore: Lessons for the conservation of Southeast Asia’s mangroves. Raffles Bull. Zool. 2012, 25, 55–65. [Google Scholar]
- Friess, D.A.; Webb, E.L. Variability in mangrove change estimates and implications for the assessment of ecosystem service provision. Glob. Ecol. Biogeogr. 2014, 23, 715–725. [Google Scholar] [CrossRef]
- Sanchez-Azofeifa, G.A.; Daily, G.C.; Pfaff, A.S.P.; Busch, C. Integrity and isolation of Costa Rica’s national parks and biological reserves: Examining the dynamics of land-cover change. Biol. Conserv. 2003, 109, 123–135. [Google Scholar] [CrossRef]
- Kuenzer, C.; Bluemel, A.; Gebhardt, S.; Tuan Vo, Q.; Dech, S. Remote sensing of mangrove ecosystems: A review. Remote Sens. 2011, 3, 878–928. [Google Scholar] [CrossRef]
- Giri, C.; Ochieng, E.; Tieszen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Skilleter, G.A. Validation of rapid assessment of damage in urban mangrove forests and relationships with molluscan assemblages. J. Mar. Biol. Assoc. UK. 1996, 76, 701–716. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Pickett, S.T.A. Ecosystem structure and function along urban rural gradients: An unexploited opportunity for ecology. Ecology. 1990, 71, 1232–1237. [Google Scholar] [CrossRef]
- Laurance, W.F.; Lovejoy, T.E.; Vasconcelos, H.L.; Bruna, E.M.; Didham, R.K.; Stouffer, P.C.; Gascon, C.; Bierregaard, R.O.; Laurance, S.G.; Sampaio, E. Ecosystem decay of Amazonian forest fragments: A 22-year investigation. Conserv. Biol. 2002, 16, 605–618. [Google Scholar] [CrossRef]
- Laurance, W.F.; Camargo, J.L.C.; Luizao, R.C.C.; Laurance, S.G.; Pimm, S.L.; Bruna, E.M.; Stouffer, P.C.; Williamson, G.B.; Benitez-Malvido, J.; Vasconcelos, H.L.; et al. The fate of Amazonian forest fragments: A 32-year investigation. Biol. Conserv. 2011, 144, 56–67. [Google Scholar] [CrossRef]
- Blanco, J.F.; Castaño, M.C. Effects mangrove conversion to pasture on density and shell size of two gastropods in the turbo river delta (Urabá Gulf, Caribbean Coast of Colombia). Rev. Biol. Trop. 2012, 60, 1707–1719. [Google Scholar] [PubMed]
- Amortegui-Torres, V.; Taborda-Marín, A.; Blanco, J.F. Edge effect on a Neritina virginea (Neritimorpha, Neritinidae) population in a black mangrove stand (Magnoliopsida, Avicenniaceae: Avicennia germinans) in the Southern Caribbean. Pan-Am. J. Aquat. Sci. 2013, 8, 68–78. [Google Scholar]
- Satyanarayana, B.; Mulder, S.; Jayatissa, L.P.; Dahdouh-Guebas, F. Are the mangroves in the Galle-Unawatuna area (Sri Lanka) at risk? A social-ecological approach involving local stakeholders for a better conservation policy. Ocean Coast. Manag. 2013, 71, 225–237. [Google Scholar] [CrossRef]
- Blanco, J.F.; Estrada, E.A.; Ortiz, L.F.; Urrego, L.E. Ecosystem-wide impacts of deforestation in mangroves: The Urabá Gulf (Colombian Caribbean) case study. ISRN Ecol. 2012, 2012, 1–14. [Google Scholar] [CrossRef]
- Urrego, L.E.; Molina, E.C.; Suárez, J.A. Environmental and anthropogenic influences on the distribution, structure, and floristic composition of mangrove forests of the Gulf of Urabá (Colombian Caribbean). Aquat. Bot. 2014, 114, 42–49. [Google Scholar] [CrossRef]
- Blanco, J.F.; Taborda-Marín, A.; Amortegui-Torres, V.; Arroyave-Rincón, A.; Sandoval, A.; Estrada, E.A.; Leal-Flórez, J.; Vásquez-Arango, J.G.; Vivas-Narváez, A. Deforestation and sedimentation in Uraba Gulf mangroves. A synthesis of the impacts on macrobenthos and fishes in the Turbo River Delta. Rev. Gest. Ambient. 2013, 16, 19–36. [Google Scholar]
- Arroyave-Rincón, A.; Amortegui-Torres, V.; Blanco-Libreros, J.F.; Taborda-Marín, A. Edge effect on a blue crab population Cardisoma guanhumi (Decapoda: Gecarcinidae) in the mangrove of El Uno Bay, Urabá Gulf (Colombia): An approximation to the folk catchery. Actual. Biol. (Medellín) 2014, 36, 47–57. [Google Scholar]
- Colombia, D.A.N.E. General Census 2005, Population Conciliated. Available online: https://www.dane.gov.co/index.php/poblacion-y-demografia/sistema-de-consulta (accessed on 30 January 2015).
- CORPOURABA (Corporación para el Desarrollo Sostenible de Urabá). Caracterización y zonificación de los manglares del Golfo de Urabá Departamento de Antioquia. Proyecto Zonificación y Ordenamiento de los manglares de Urabá; Convenio 201671 FONADE-CORPOURABA; Documento técnico; CORPOURABA: Apartadó, Colombia, 2003. (In Spanish) [Google Scholar]
- CORPOURABA. Plan de manejo integral de los manglares del golfo de Urabá y mar Caribe antioqueño; Corporación para el Desarrollo Sostenible de Urabá CORPOURABA; Documento técnico; CORPOURABA: Apartadó, Colombia, 2005. (In Spanish) [Google Scholar]
- Blanco, J.F.; Londoño-Mesa, M.; Quan-Young, L.; Urrego, L.; Polanía, J.; Osorio, A.; Bernal, G.; Correa, I. The Urabá Gulf mangrove expedition of Colombia. ISME/GLOMIS 2011, 9, 8–10. [Google Scholar]
- Klijn, F.; Dehaes, H.A.U. A hierarchical approach to ecosystems and its implications for ecological land classification. Landsc. Ecol. 1994, 9, 89–104. [Google Scholar] [CrossRef]
- Dahdouh-Guebas, F.; Triest, L.; Verneirt, M. The importance of a hierarchical ecosystem classification for the biological evaluation and selection of least valuable sites. Impact Assess. Proj. Apprais. 1998, 16, 185–194. [Google Scholar] [CrossRef]
- Colombia Instituto de Hidrología, Meteorología y Estudios Ambientales (IDEAM). Leyenda Nacional de Coberturas de la Tierra. Metodología CORINE LandCover adaptada para Colombia Escala 1:100.000; Editorial Scripto Ltda IDEAM: Bogotá, D.C., Colombia, 2010; p. 72. (In Spanish) [Google Scholar]
- Melo-Wilches, L.H.; Camacho-Chávez, M.A. Interpretación visual de imágenes de sensores remotos y su aplicación en levantamientos de cobertura y uso de la Tierra; Instituto Geográfico Agustín Codazzi IGAC: Bogota, D.C., Colombia, 2005. (In Spanish) [Google Scholar]
- Congalton, R.G. A review of assessing the accuracy of classifications of remotely sensed data. Remote Sens. Environ. 1991, 37, 35–46. [Google Scholar] [CrossRef]
- Foody, G.M. Status of land cover classification accuracy assessment. Remote Sens. Environ. 2002, 80, 185–201. [Google Scholar] [CrossRef]
- Puyravaud, J.P. Standardizing the calculation of the annual rate of deforestation. For. Ecol. Manag. 2003, 177, 593–596. [Google Scholar] [CrossRef]
- Hargis, C.D.; Bissonette, J.A.; David, J.L. The behavior of landscape metrics commonly used in the study of habitat fragmentation. Landsc. Ecol. 1998, 13, 167–186. [Google Scholar] [CrossRef]
- Cintrón, G.; Schaeffer-Novelli, Y. Methods for studying mangrove structure. In The Mangrove Ecosystem: Research Methods. Monographs on Oceanographic Methodology, No. 8; Snedaker, S.C., Snedaker, J.G., Eds.; United Nations Educational, Scientific and Cultural Organization Publisher, Richard Clay (The Chaucer Press), UNESCO: Paris, France, 1985; pp. 91–113. [Google Scholar]
- Dahdouh-Guebas, F.; Koedam, N. Empirical estimate of the reliability of the use of the Point-Centred Quarter Method (PCQM): Solutions to ambiguous field situations and description of the PCQM+ protocol. For. Ecol. Manag. 2006, 228, 1–18. [Google Scholar] [CrossRef]
- Ortiz, L.F.; Blanco, J.F. Distribution of the mangrove gastropods Neritina virginea (Neritidae) and Littoraria angulifera (Littorinidae) within the Colombian Caribbean, Darién Ecoregion. Rev. Biol. Trop. 2012, 60, 219–232. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008; Available online: http//www.R-project.org (accessed on 29 May 2015).
- Gotelli, N.J.; Ellison, A.M. A Primer of Ecological Statistics; Sinauer Associates, Inc.: Sunderland, MA, USA, 2004; p. 510. [Google Scholar]
- FAO. The World’s Mangroves 1980–2005; FAO Forestry Paper 153; Food and Agricultural: Rome, Italy, 2007; p. 77. [Google Scholar]
- Kirui, K.B.; Kairo, J.G.; Bosire, J.; Viergever, K.M.; Rudra, S.; Huxham, M.; Briers, R.A. Mapping of mangrove forest land cover change along the Kenya coastline using Landsat imagery. Ocean Coast. Manag. 2013, 83, 19–24. [Google Scholar] [CrossRef]
- Rideout, A.J.R.; Joshi, N.P.; Viergever, K.M.; Huxham, M.; Briers, R.A. Making predictions of mangrove deforestation: A comparison of two methods in Kenya. Glob. Chang. Biol. 2013, 19, 3493–3501. [Google Scholar] [CrossRef]
- Thu, P.M.; Populus, J. Status and changes of mangrove forest in Mekong delta: Case study in Tra Vinh, Vietnam. Estuar. Coast. Shelf Sci. 2007, 71, 98–109. [Google Scholar] [CrossRef]
- Hamilton, S. Assessing the role of commercial aquaculture in displacing mangrove forest. Bull. Mar. Sci. 2013, 89, 585–601. [Google Scholar] [CrossRef]
- Webb, E.L.; Friess, D.A.; Krauss, K.W.; Cahoon, D.R.; Guntenspergen, G.R.; Phelps, J. A global standard for monitoring coastal wetland vulnerability to accelerated sea-level rise. Nat. Clim. Chang. 2013, 3, 458–465. [Google Scholar] [CrossRef]
- Gilman, E.L.; Ellison, J.; Duke, N.C.; Field, C. Threats to mangroves from climate change and adaptation options: A review. Aquat. Bot. 2008, 89, 237–250. [Google Scholar] [CrossRef]
- Kairo, J.G.; Kivyatu, B.; Koedam, N. Application of remote sensing and GIS in the management of mangrove forests within and adjacent to Kiunga Marine Protected Area, Lamu, Kenya. Environ. Dev. Sustain. 2002, 4, 153–166. [Google Scholar] [CrossRef]
- Polidoro, B.A.; Carpenter, K.E.; Collins, L.; Duke, N.C.; Ellison, A.M.; Ellison, J.C.; Farnsworth, E.J.; Fernando, E.S.; Kathiresan, K.; Koedam, N.E.; et al. The loss of species: Mangrove extinction risk and geographic areas of global concern. PLoS ONE 2010. [Google Scholar] [CrossRef]
- Cannicci, S.; Burrows, D.; Fratini, S.; Smith, T.J., III; Offenberg, J.; Dahdouh-Guebas, F. Faunal impact on vegetation structure and ecosystem function in mangrove forests: A review. Aquat. Bot. 2008, 89, 186–200. [Google Scholar] [CrossRef]
- Lee, S.Y. Mangrove macrobenthos: Assemblages, services, and linkages. J. Sea Res. 2008, 59, 16–29. [Google Scholar] [CrossRef]
- Diele, K.; Ngoc, D.M.T.; Geist, S.J.; Meyer, F.W.; Pham, Q.H.; Saint-Paul, U.; Tran, T.; Berger, U. Impact of typhoon disturbance on the diversity of key ecosystem engineers in a monoculture mangrove forest plantation, Can Gio Biosphere Reserve, Vietnam. Glob. Planet. Chang. 2013, 110, 236–248. [Google Scholar] [CrossRef]
- Laurance, W.F. Do edge effects occur over large spatial scales? Trends Ecol. Evol. 2000, 15, 134–135. [Google Scholar] [CrossRef]
- Martin, L.J.; Blossey, B.; Ellis, E. Mapping where ecologists work: Biases in the global distribution of terrestrial ecological observations. Front. Ecol. Environ. 2012, 10, 195–201. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blanco-Libreros, J.F.; Estrada-Urrea, E.A. Mangroves on the Edge: Anthrome-Dependent Fragmentation Influences Ecological Condition (Turbo, Colombia, Southern Caribbean). Diversity 2015, 7, 206-228. https://doi.org/10.3390/d7030206
Blanco-Libreros JF, Estrada-Urrea EA. Mangroves on the Edge: Anthrome-Dependent Fragmentation Influences Ecological Condition (Turbo, Colombia, Southern Caribbean). Diversity. 2015; 7(3):206-228. https://doi.org/10.3390/d7030206
Chicago/Turabian StyleBlanco-Libreros, Juan Felipe, and Edgar Andrés Estrada-Urrea. 2015. "Mangroves on the Edge: Anthrome-Dependent Fragmentation Influences Ecological Condition (Turbo, Colombia, Southern Caribbean)" Diversity 7, no. 3: 206-228. https://doi.org/10.3390/d7030206
APA StyleBlanco-Libreros, J. F., & Estrada-Urrea, E. A. (2015). Mangroves on the Edge: Anthrome-Dependent Fragmentation Influences Ecological Condition (Turbo, Colombia, Southern Caribbean). Diversity, 7(3), 206-228. https://doi.org/10.3390/d7030206






