DNA Barcodes for Marine Biodiversity: Moving Fast Forward?
Abstract
:1. Introduction
2. Marine Biodiversity
| Marine group | Bouchet (2006) | WoRMS (February 2010) Valid species |
|---|---|---|
| Bacteria | 4,800 | 625 |
| Fungi | 500 | 1,061 |
| Rhodophyta | 6,200 | 6,302 |
| Acanthocephala | 600 | 410 |
| Annelida | 12,148a | 12,631 |
| Arthropoda | 47,217b | 44,591 |
| Brachiopoda | 550 | 386 |
| Bryozoa | 5,700c | 1,525 |
| Chaetognatha | 121 | 208 |
| Cnidaria | 9,795 | 11,071 |
| Ctenophora | 166 | 170 |
| Cycliophora | 1 | 2 |
| Echinodermata | 7,000 | 5,764 |
| Echiura | 170 | 203 |
| Entoprocta | 165–170 | 161 |
| Gastrotricha | 390–400 | 524 |
| Gnathostomulida | 97 | 97 |
| Hemichordata | 106 | 106 |
| Mesozoa | 106d | 115 |
| Mollusca | 52,525 | 23,689 |
| Nematoda | 12,000 | 5,889 |
| Nemertea | 1,180–1,230 | 1,371 |
| Phoronida | 10 | 11 |
| Platyhelminthes | 15,000 | 3,348 |
| Porifera | 5,500 | 8,174 |
| Rotifera | 50 | 185 |
| Sipuncula | 144 | 158 |
| Tardigrada | 212 | 170 |
| Chordata | 21,517e | 21,944 |
| Total | 203,887 | 150,891 |
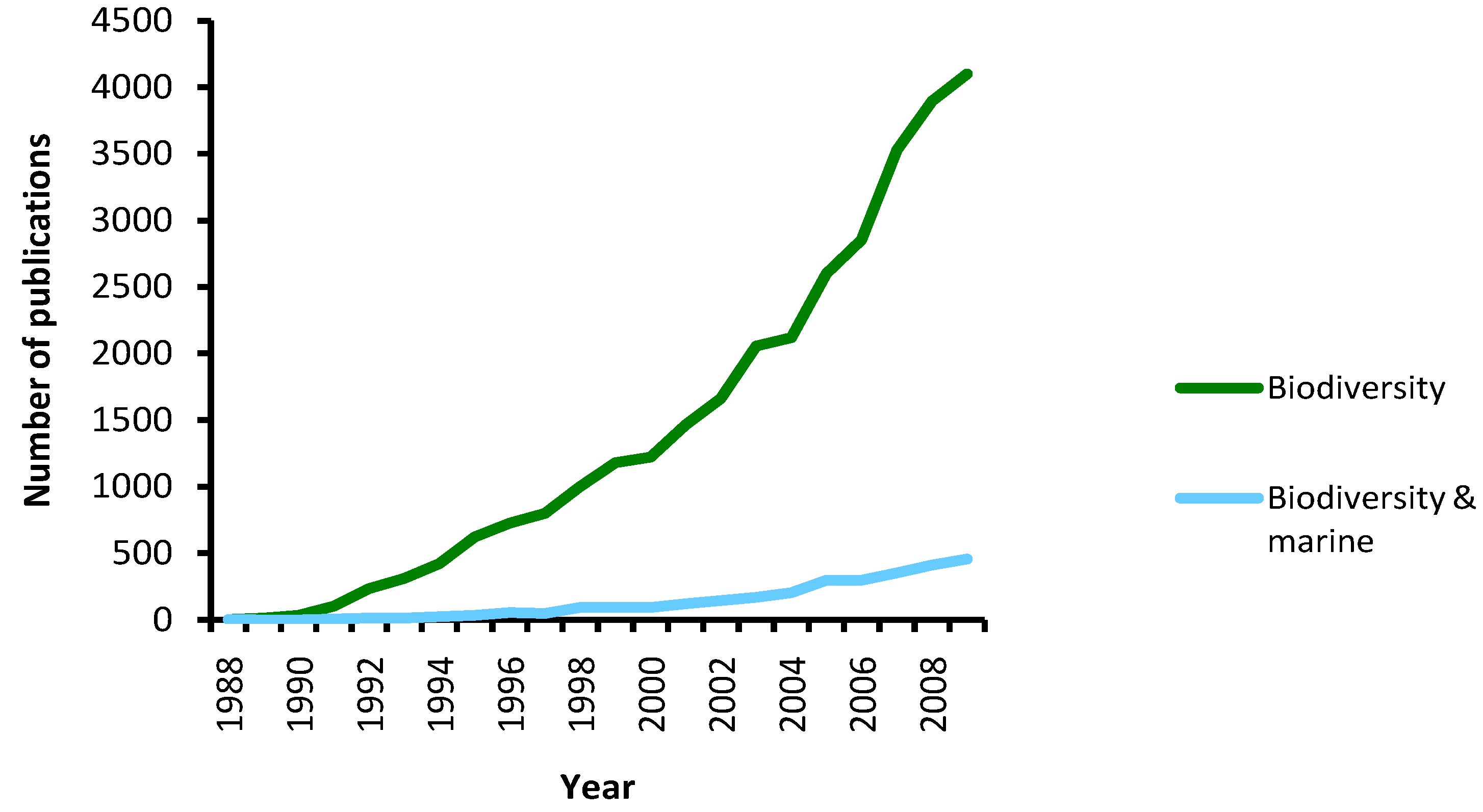
3. Molecular Methods for Species Diversity
4. DNA Barcoding for Species Identification and Discovery
4.1. The Concept: Advantages and Limitations
4.2. Practical Applications for the Marine Environment
4.3. Progress in DNA-based Inventories of Marine Groups
| Marine group | NoS | Intra (%) | Inter (%) | Reference |
|---|---|---|---|---|
| Crustaceans | ||||
| Malacostracans | 80 | 0.91a | 13.6 | [58] |
| Decapods | 54 | 0.46 | 17.16 | [68] |
| Copepods | 24 | 0.75b | 27.05 | [67] |
| Molluscs | ||||
| Heteropods | 9 | 3.28 | 21.7 | [69] |
| Pteropods | 31 | 3.02 | 17.6 | [69] |
| Corals | 30 | 0.05 | 1.90 | [65] |
| Chaetognaths | 14 | 1.45 | 34.5 | [70] |
| Echinoderms | 191 | 0.62 | 15.33 | [71] |
| Fishes | 207 | 0.39 | 9.93 | [72] |
5. Current Status
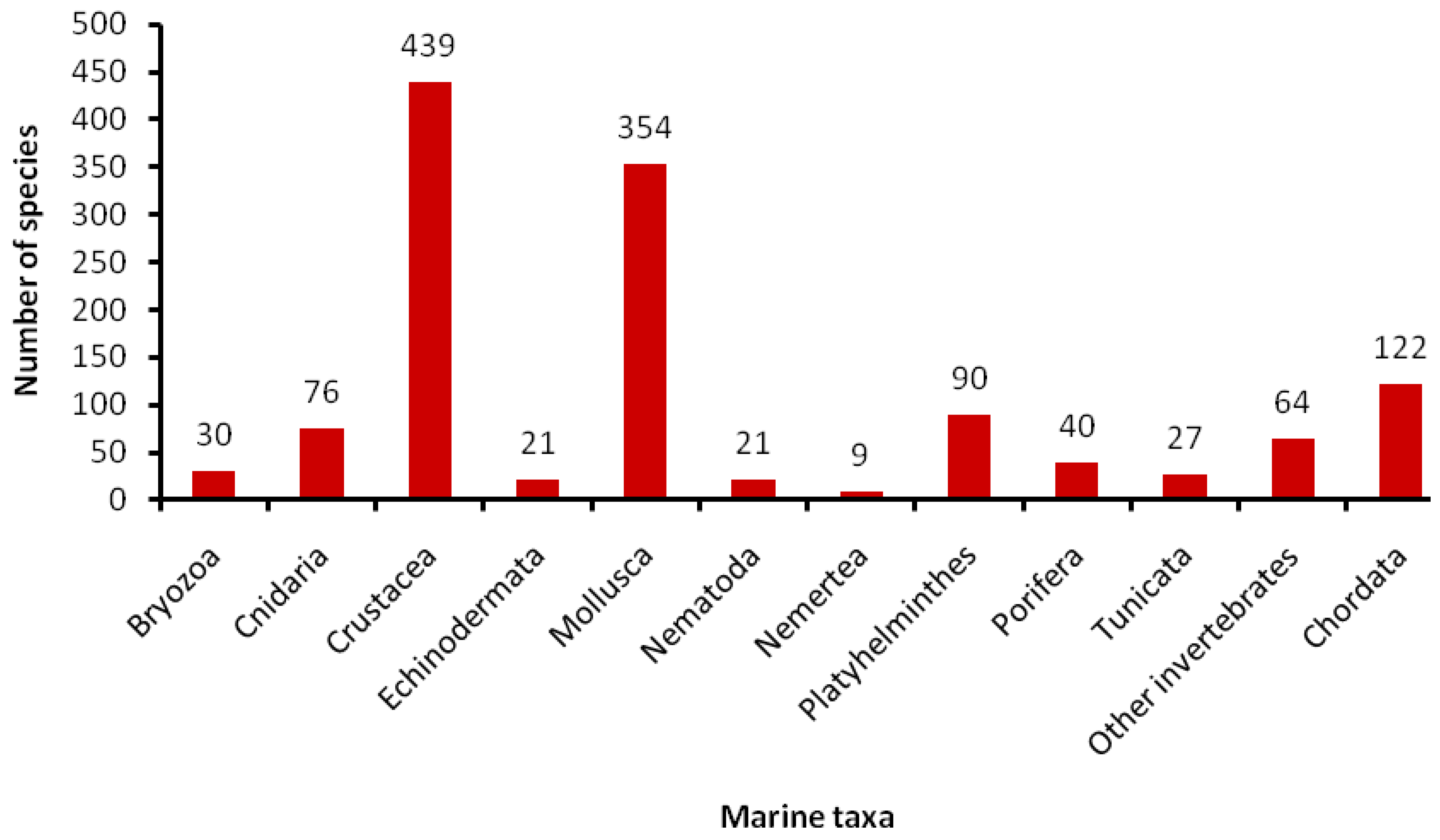
5.1. How Many Marine Barcodes?
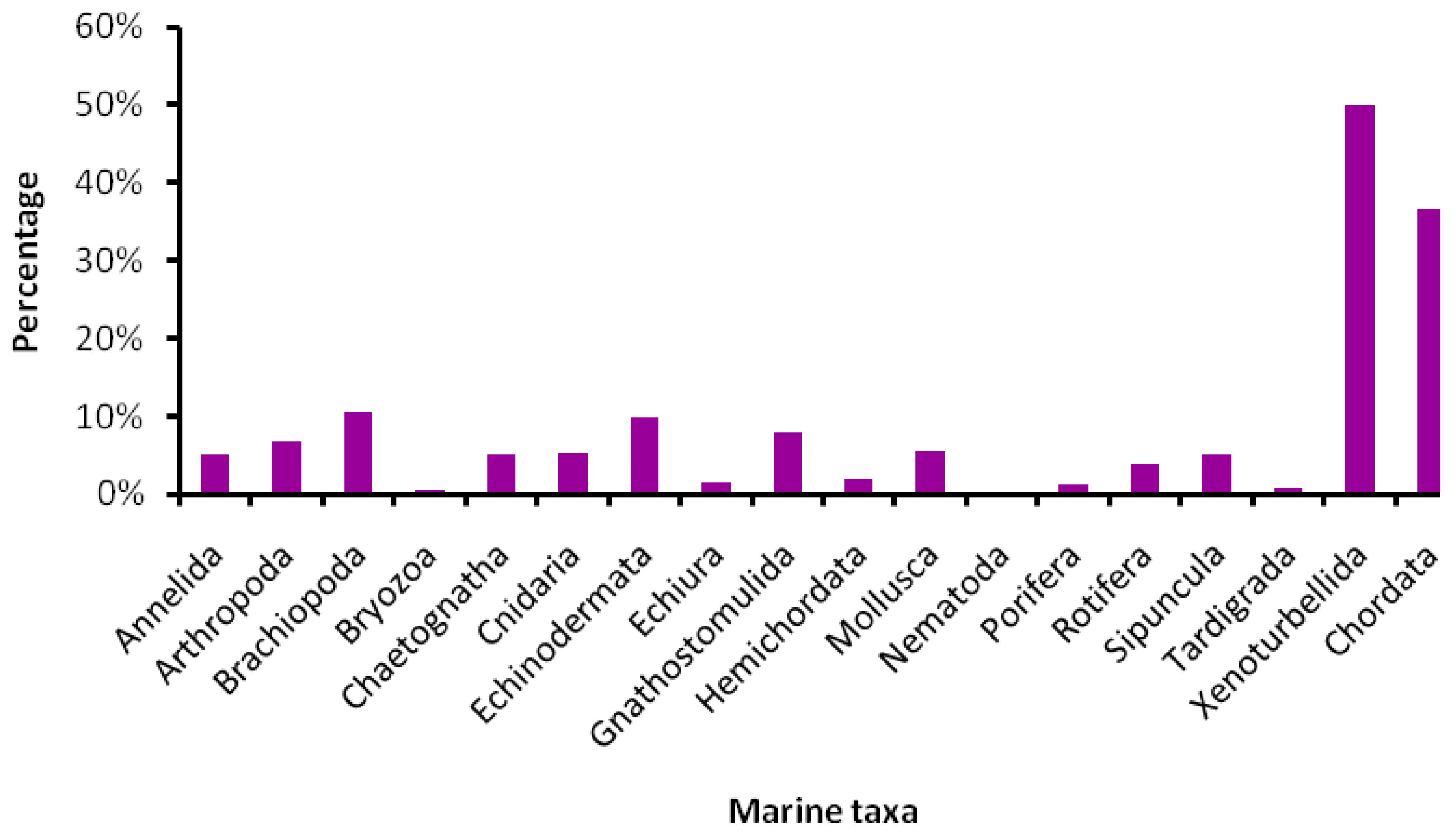
5.2. Special Issues with Marine Taxa
5.3. Taxonomy and Barcoding
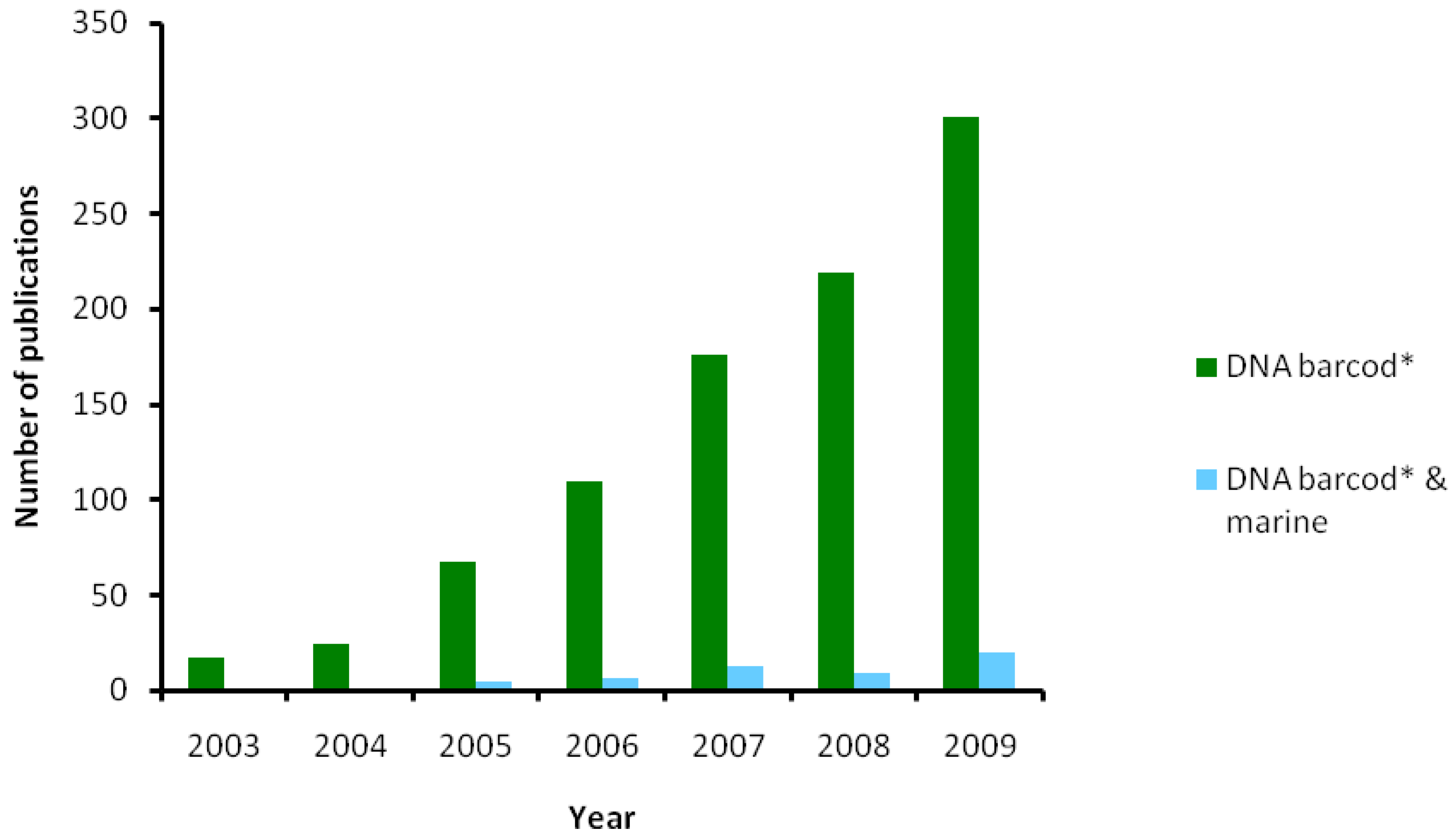
5.4. Future Directions
5.5. Species as Currency for Biodiversity
6. Conclusions
Acknowledgements
References
- Biodiversity; Wilson, E.O.; Peter, F.M. (Eds.) National Academy Press: Washington, DC, USA, 1988.
- Pimm, S.L.; Russell, G.J.; Gittleman, J.L.; Brooks, T.M. The future of biodiversity. Science 1995, 269, 347–350. [Google Scholar]
- Chapman, A.D. Numbers of Living Species in Australia and the World, 2nd ed.; Australian Biological Resources Study: Canberra, Australia, 2009. [Google Scholar]
- Coyne, J.A.; Orr, H.A. Speciation; Sinauer Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Packer, L.; Gibbs, J.; Sheffield, C.; Hanner, R. DNA barcoding and the mediocrity of morphology. Mol. Ecol. Resour. 2009, 9, 42–50. [Google Scholar] [CrossRef]
- Bouchet, P. The magnitude of marine biodiversity. In The Exploration of Marine Biodiversity: Scientific and Technological Challenges; Duarte, C.M., Ed.; Fundacion BBVA: Bilbao, Spain, 2006; pp. 31–64. [Google Scholar]
- Grassle, J.F.; Maciolek, N.J. Deep-sea species richness—regional and local diversity estimates from quantitative bottom samples. Am. Nat. 1992, 139, 313–341. [Google Scholar]
- Vrijenhoek, R.C. Cryptic species, phenotypic plasticity, and complex life histories: Assessing deep-sea faunal diversity with molecular markers. Deep-Sea Res. II 2009, 56, 1713–1723. [Google Scholar] [CrossRef]
- Knowlton, N. Sibling species in the sea. Annu. Rev. Ecol. Syst. 1993, 24, 189–216. [Google Scholar]
- Briggs, J.C. Species-diversity—land and sea compared. Syst. Biol. 1994, 43, 130–135. [Google Scholar] [CrossRef]
- Gray, J.S. Marine biodiversity: patterns, threats and conservation needs. Biodivers. Conserv. 1997, 6, 153–175. [Google Scholar]
- Brunel, P. Visages de la biodiversite marine. VertigO 2005, 6, 1–13. [Google Scholar]
- Worm, B.; Barbier, E.B.; Beaumont, N.; Duffy, J.E.; Folke, C.; Halpern, B.S.; Jackson, J.B.C.; Lotze, H.K.; Micheli, F.; Palumbi, S.R.; Sala, E.; Selkoe, K.A.; Stachowicz, J.J.; Watson, R. Impacts of biodiversity loss on ocean ecosystem services. Science 2006, 314, 787–790. [Google Scholar] [CrossRef]
- Molnar, J.L.; Gamboa, R.L.; Revenga, C.; Spalding, M.D. Assessing the global threat of invasive species to marine biodiversity. Front. Ecol. Environ. 2008, 6, 485–492. [Google Scholar]
- Mosquin, T.; Whiting, P.G.; McAllister, D.E. Canada’s Biodiversity: The Variety of Life, Its Status, Economic Benefits, Conservation Costs and Unmeet Needs; Canadian Museum of Nature: Ottawa, Canada, 1995. [Google Scholar]
- Schander, C.; Willassen, E. What can biological barcoding do for marine biology? Mar. Biol. Res. 2005, 1, 79–83. [Google Scholar] [CrossRef]
- Avise, J.C. Systematic value of electrophoretic data. Syst. Zool. 1975, 23, 465–481. [Google Scholar] [CrossRef]
- Sévigny, J.M.; McLaren, I.A.; Frost, B.W. Discrimination among and variation within species of Pseudocalanus based on the GPI locus. Mar. Biol. 1989, 102, 321–327. [Google Scholar] [CrossRef]
- Hu, Y.P.; Lutz, R.A.; Vrijenhoek, R.C. Electrophoretic identification and genetic-analysis of bivalve larvae. Mar. Biol. 1992, 113, 227–230. [Google Scholar]
- Wong, E.H.K.; Hanner, R.H. DNA barcoding detects market substitution in North American seafood. Food Res. Int. 2008, 41, 828–837. [Google Scholar] [CrossRef]
- Bartlett, S.E.; Davidson, W.S. Identification of Thunnus tuna species by the polymerase chain-reaction and direct sequence-analysis of their mitochondrial cytochrome-b genes. Can. J. Fish. Aquat. Sci. 1991, 48, 309–317. [Google Scholar] [CrossRef]
- Medeiros-Bergen, D.E.; Olson, R.R.; Conroy, J.A.; Kocher, T.D. Distribution of holothurian larvae determined with species-specific genetic probes. Limnol. Oceanogr. 1995, 40, 1225–1235. [Google Scholar] [CrossRef]
- Bucklin, A.; Guarnieri, M.; Hill, R.S.; Bentley, A.M.; Kaartvedt, S. Taxonomic and systematic assessment of planktonic copepods using mitochondrial COI sequence variation and competitive, species-specific PCR. Hydrobiologia 1999, 401, 239–254. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; DeWaard, J.R. Biological identifications through DNA barcodes. Proc. R. Soc. Lond. B Biol. Sci. 2003, 270, 313–321. [Google Scholar] [CrossRef]
- Galtier, N.; Nabholz, B.; Glémin, S.; Hurst, G.D.D. Mitochondrial DNA as a marker of molecular diversity: a reappraisal. Mol. Ecol. 2009, 18, 4541–4550. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Stoeckle, M.Y.; Zemlak, T.S.; Francis, C.M. Identification of birds through DNA barcodes. PLoS Biol. 2004, 2, 1657–1663. [Google Scholar]
- Hollingsworth, P.M.; Forrest, L.L.; Spouge, J.L.; Hajibabaei, M.; Ratnasingham, S.; van der Bank, M.; Chase, M.W.; Cowan, R.S.; Erickson, D.L.; Fazekas, A.J.; Graham, S.W.; James, K.E.; Kim, K.J.; Kress, W.J.; Schneider, H.; van AlphenStahl, J.; Barrett, S.C.H.; van den Berg, C.; Bogarin, D.; Burgess, K.S.; Cameron, K.M.; Carine, M.; Chacón, J.; Clark, A.; Clarkson, J.J.; Conrad, F.; Devey, D.S.; Ford, C.S.; Hedderson, T.A.J.; Hollingsworth, M.L.; Husband, B.C.; Kelly, L.J.; Kesanakurti, P.R.; Kim, J.S.; Kim, Y.D.; Lahaye, R.; Lee, H.L.; Long, D.G.; Madriñan, S.; Maurin, O.; Meusnier, I.; Newmaster, S.G.; Park, C.W.; Percy, D.M.; Petersen, G.; Richardson, J.E.; Salazar, G.A.; Savolainen, V.; Seberg, O.; Wilkinson, M.J.; Yi, D.K.; Little, D.P. A DNA barcode for land plants. Proc. Natl. Acad. Sci. USA 2009, 106, 12794–12797. [Google Scholar] [CrossRef]
- Saunders, G.W. Applying DNA barcoding to red macroalgae: a preliminary appraisal holds promise for future applications. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 2005, 360, 1879–1888. [Google Scholar] [CrossRef]
- Seifert, K.A.; Samson, R.A.; deWaard, J.R.; Houbraken, J.; Levesque, C.A.; Moncalvo, J.M.; Louis-Seize, G.; Hebert, P.D.N. Prospects for fungus identification using CO1 DNA barcodes, with Penicillium as a test case. Proc. Natl. Acad. Sci. USA 2007, 104, 3901–3906. [Google Scholar]
- Pleijel, F.; Jondelius, U.; Norlinder, E.; Nygren, A.; Oxelman, B.; Schander, C.; Sundberg, P.; Thollesson, M. Phylogenies without roots? A plea for the use of vouchers in molecular phylogenetic studies. Mol. Phylogenet. Evol. 2008, 48, 369–371. [Google Scholar] [CrossRef]
- Ratnasingham, S.; Hebert, P.D.N. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar]
- Harris, D.J. Can you bank on GenBank? Trends Ecol. Evol. 2003, 18, 317–319. [Google Scholar] [CrossRef]
- Shokralla, S.; Singer, G.A.C.; Hajibabaei, M. Direct PCR amplification and sequencing of specimens’ DNA from preservative ethanol. BioTechniques 2010, 48, 305–306. [Google Scholar]
- Will, K.W.; Mishler, B.D.; Wheeler, Q.D. The perils of DNA barcoding and the need for integrative taxonomy. Syst. Biol. 2005, 54, 844–851. [Google Scholar] [CrossRef]
- Will, K.W.; Rubinoff, D. Myth of the molecule: DNA barcodes for species cannot replace morphology for identification and classification. Cladistics 2004, 20, 47–55. [Google Scholar] [CrossRef]
- Rubinoff, D.; Cameron, S.; Will, K. A genomic perspective on the shortcomings of mitochondrial DNA for ‘barcoding’ identification. J. Hered. 2006, 97, 581–594. [Google Scholar] [CrossRef]
- Rubinoff, D. Utility of mitochondrial DNA barcodes in species conservation. Conserv. Biol. 2006, 20, 1026–1033. [Google Scholar] [CrossRef]
- Song, H.; Buhay, J.E.; Whiting, M.F.; Crandall, K.A. Many species in one: DNA barcoding overestimates the number of species when nuclear mitochondrial pseudogenes are coamplified. Proc. Natl. Acad. Sci. USA 2008, 105, 13486–13491. [Google Scholar]
- Siddall, M.E.; Fontanella, F.M.; Watson, S.C.; Kvist, S.; Erséus, C. Barcoding bamboozled by Bacteria: convergence to metazoan mitochondrial primer targets by marine microbes. Syst. Biol. 2009, 58, 445–451. [Google Scholar] [CrossRef]
- Kemppainen, P.; Panova, M.; Hollander, J.; Johannesson, K. Complete lack of mitochondrial divergence between two species of NE Atlantic marine intertidal gastropods. J. Evol. Biol. 2009, 22, 2000–2011. [Google Scholar] [CrossRef]
- Frézal, L.; Leblois, R. Four years of DNA barcoding: Current advances and prospects. Infect. Genet. Evol. 2008, 8, 727–736. [Google Scholar] [CrossRef]
- Mitchell, A. DNA barcoding demystified. Aust. J. Entomol. 2008, 47, 169–173. [Google Scholar] [CrossRef]
- Meyer, C.P.; Paulay, G. DNA barcoding: Error rates based on comprehensive sampling. PLoS Biol. 2005, 3, 2229–2238. [Google Scholar]
- Ekrem, T.; Willassen, E.; Stur, E. A comprehensive DNA sequence library is essential for identification with DNA barcodes. Mol. Phylogenet. Evol. 2007, 43, 530–542. [Google Scholar] [CrossRef]
- Nielsen, R.; Matz, M. Statistical approaches for DNA barcoding. Syst. Biol. 2006, 55, 162–169. [Google Scholar] [CrossRef]
- Gómez, A.; Wright, P.J.; Lunt, D.H.; Cancino, J.M.; Carvalho, G.R.; Hughes, R.N. Mating trials validate the use of DNA barcoding to reveal cryptic speciation of a marine bryozoan taxon. Proc. R. Soc. Lond., B, Biol. Sci. 2007, 274, 199–207. [Google Scholar] [CrossRef]
- Ivanova, N.V.; Borisenko, A.V.; Hebert, P.D.N. Express barcodes: racing from specimen to identification. Mol. Ecol. Resour. 2009, 9, 35–41. [Google Scholar] [CrossRef]
- Marko, P.B.; Lee, S.C.; Rice, A.M.; Gramling, J.M.; Fitzhenry, T.M.; McAlister, J.S.; Harper, G.R.; Moran, A.L. Mislabelling of a depleted reef fish. Nature 2004, 430, 309–310. [Google Scholar]
- Smith, P.J.; McVeagh, S.M.; Steinke, D. DNA barcoding for the identification of smoked fish products. J. Fish Biol. 2008, 72, 464–471. [Google Scholar] [CrossRef]
- Barbuto, M.; Galimberti, A.; Ferri, E.; Labra, M.; Malandra, R.; Galli, P.; Casiraghi, M. DNA barcoding reveals fraudulent substitutions in shark seafood products: the Italian case of ‘palombo’ (Mustelus spp.). Food Res. Int. 2010, 43, 376–381. [Google Scholar] [CrossRef]
- Holmes, B.H.; Steinke, D.; Ward, R.D. Identification of shark and ray fins using DNA barcoding. Fish. Res. 2009, 95, 280–288. [Google Scholar] [CrossRef]
- Cohen, N.J.; Deeds, J.R.; Wong, E.S.; Hanner, R.H.; Yancy, H.F.; White, K.D.; Thompson, T.M.; Wahl, M.; Pham, T.D.; Guichard, F.M.; Huh, I.; Austin, C.; Dizikes, G.; Gerber, S.I. Public health response to puffer fish (tetrodotoxin) poisoning from mislabeled product. J. Food Prot. 2009, 72, 810–817. [Google Scholar]
- Barber, P.; Boyce, S.L. Estimating diversity of Indo-Pacific coral reef stomatopods through DNA barcoding of stomatopod larvae. Proc. R. Soc. Lond., B, Biol. Sci. 2006, 273, 2053–2061. [Google Scholar] [CrossRef]
- Webb, K.E.; Barnes, D.K.A.; Clark, M.S.; Bowden, D.A. DNA barcoding: a molecular tool to identify Antarctic marine larvae. Deep Sea Res. II 2006, 53, 1053–1060. [Google Scholar] [CrossRef]
- Pegg, G.G.; Sinclair, B.; Briskey, L.; Aspden, W.J. MtDNA barcode identification of fish larvae in the southern Great Barrier Reef, Australia. Sci. Mar. 2006, 70, 7–12. [Google Scholar]
- Steinke, D.; Zemlak, T.S.; Hebert, P.D.N. Barcoding Nemo: DNA-based identifications for the ornamental fish trade. PLoS One 2009, 4, e6300. [Google Scholar] [CrossRef]
- Saunders, G.W. Routine DNA barcoding of Canadian Gracilariales (Rhodophyta) reveals the invasive species Gracilaria vermiculophylla in British Columbia. Mol. Ecol. Resour. 2009, 9, 140–150. [Google Scholar] [CrossRef]
- Radulovici, A.E.; Sainte-Marie, B.; Dufresne, F. DNA barcoding of marine crustaceans from the Estuary and Gulf of St Lawrence: a regional-scale approach. Mol. Ecol. Resour. 2009, 9, 181–187. [Google Scholar]
- Saunders, G.W. A DNA barcode examination of the red algal family Dumontiaceae in Canadian waters reveals substantial cryptic species diversity. 1. The foliose Dilsea-Neodilsea complex and Weeksia. Botany-Botanique 2008, 86, 773–789. [Google Scholar] [CrossRef]
- Conklin, K.Y.; Kurihara, A.; Sherwood, A.R. A molecular method for identification of the morphologically plastic invasive algal genera Eucheuma and Kappaphycus (Rhodophyta, Gigartinales) in Hawaii. J. Appl. Phycol. 2009, 21, 691–699. [Google Scholar] [CrossRef]
- McDevit, D.C.; Saunders, G.W. On the utility of DNA barcoding for species differentiation among brown macroalgae (Phaeophyceae) including a novel extraction protocol. Phycol. Res. 2009, 57, 131–141. [Google Scholar] [CrossRef]
- Moniz, M.B.J.; Kaczmarska, I. Barcoding of diatoms: nuclear encoded ITS revisited. Protist 2010, 161, 7–34. [Google Scholar] [CrossRef]
- Wörheide, G.; Erpenbeck, D. DNA taxonomy of sponges—progress and perspectives. J. Mar. Biolog. Assoc. U.K. 2007, 87, 1629–1633. [Google Scholar]
- Erpenbeck, D.; Hooper, J.N.A.; Wörheide, G. CO1 phylogenies in diploblasts and the ‘Barcoding of Life’—are we sequencing a suboptimal partition? Mol. Ecol. Notes 2006, 6, 550–553. [Google Scholar] [CrossRef]
- Shearer, T.L.; Coffroth, M.A. Barcoding corals: limited by interspecific divergence, not intraspecific variation. Mol. Ecol. Resour. 2008, 8, 247–255. [Google Scholar] [CrossRef]
- Moura, C.J.; Harris, D.J.; Cunha, M.R.; Rogers, A.D. DNA barcoding reveals cryptic diversity in marine hydroids (Cnidaria, Hydrozoa) from coastal and deep-sea environments. Zool. Scr. 2008, 37, 93–108. [Google Scholar]
- Bucklin, A.; Hopcroft, R.R.; Kosobokova, K.N.; Nigro, L.M.; Ortman, B.D.; Jennings, R.M.; Sweetman, C.J. DNA barcoding of Arctic Ocean holozooplankton for species identification and recognition. Deep-Sea Res. II 2010, 57, 40–48. [Google Scholar] [CrossRef]
- Costa, F.O.; DeWaard, J.R.; Boutillier, J.; Ratnasingham, S.; Dooh, R.T.; Hajibabaei, M.; Hebert, P.D.N. Biological identifications through DNA barcodes: the case of the Crustacea. Can. J. Fish. Aquat. Sci. 2007, 64, 272–295. [Google Scholar] [CrossRef]
- Jennings, R.M.; Bucklin, A.; Ossenbrügger, H.; Hopcroft, R.R. Species diversity of planktonic gastropods (Pteropoda and Heteropoda) from six ocean basins based on DNA barcode analysis. Deep-Sea Res. II. 2010. accepted for publication. [Google Scholar]
- Jennings, R.M.; Bucklin, A.; Pierrot-Bults, A. Barcoding of arrow worms (Phylum Chaetognatha) from three oceans: genetic diversity and evolution within an enigmatic phylum. PLoS One 2010. accepted for publication. [Google Scholar]
- Ward, R.D.; Holmes, B.H.; O’Hara, T.D. DNA barcoding discriminates echinoderm species. Mol. Ecol. Resour. 2008, 8, 1202–1211. [Google Scholar]
- Ward, R.D.; Zemlak, T.S.; Innes, B.H.; Last, P.R.; Hebert, P.D.N. DNA barcoding Australia’s fish species. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 2005, 360, 1847–1857. [Google Scholar] [CrossRef]
- Puillandre, N.; Strong, E.E.; Bouchet, P.; Boisselier, M.C.; Couloux, A.; Samadi, S. Identifying gastropod spawn from DNA barcodes: possible but not yet practicable. Mol. Ecol. Resour. 2009, 9, 1311–1321. [Google Scholar] [CrossRef]
- Mikkelsen, N.T.; Schander, C.; Willassen, E. Local scale DNA barcoding of bivalves (Mollusca): a case study. Zool. Scr. 2007, 36, 455–463. [Google Scholar] [CrossRef]
- Bucklin, A.; Wiebe, P.H.; Smolenack, S.B.; Copley, N.J.; Beaudet, J.G.; Bonner, K.G.; Farber-Lorda, J.; Pierson, J.J. DNA barcodes for species identification of euphausiids (Euphausiacea, Crustacea). J. Plankton Res. 2007, 29, 483–493. [Google Scholar] [CrossRef]
- Plaisance, L.; Knowlton, N.; Paulay, G.; Meyer, C. Reef-associated crustacean fauna: biodiversity estimates using semi-quantitative sampling and DNA barcoding. Coral Reefs 2009, 28, 977–986. [Google Scholar] [CrossRef]
- Nielsen, J.F.; Lavery, S.; Lorz, A.N. Synopsis of a new collection of sea spiders (Arthropoda: Pycnogonida) from the Ross Sea, Antarctica. Polar Biol. 2009, 32, 1147–1155. [Google Scholar] [CrossRef]
- Krabbe, K.; Leese, F.; Mayer, C.; Tollrian, R.; Held, C. Cryptic mitochondrial lineages in the widespread pycnogonid Colossendeis megalonyx Hoek, 1881 from Antarctic and Subantarctic waters. Polar Biol. 2010, 33, 281–292. [Google Scholar] [CrossRef]
- Bhadury, P.; Austen, M.C.; Bilton, D.T.; Lambshead, P.J.D.; Rogers, A.D.; Smerdon, G.R. Development and evaluation of a DNA-barcoding approach for the rapid identification of nematodes. Mar. Ecol. Prog. Ser. 2006, 320, 1–9. [Google Scholar] [CrossRef]
- Leung, T.L.F.; Donald, K.M.; Keeney, D.B.; Koehler, A.V.; Peoples, R.C.; Poulin, R. Trematode parasites of Otago Harbour (New Zealand) soft-sediment intertidal ecosystems: life cycles, ecological roles and DNA barcodes. N.Z. J. Mar. Freshwater Res. 2009, 43, 857–865. [Google Scholar] [CrossRef]
- Locke, S.A.; McLaughlin, J.D.; Dayanandan, S.; Marcogliese, D.J. Diversity and specificity in Diplostomum spp. metacercariae in freshwater fishes revealed by cytochrome c oxidase I and internal transcribed spacer sequences. Int. J. Parasitol. 2010, 40, 333–343. [Google Scholar] [CrossRef]
- Ward, R.D.; Hanner, R.; Hebert, P.D.N. The campaign to DNA barcode all fishes, FISH-BOL. J. Fish Biol. 2009, 74, 329–356. [Google Scholar] [CrossRef]
- Steinke, D.; Zemlak, T.S.; Boutillier, J.A.; Hebert, P.D.N. DNA barcoding of Pacific Canada’s fishes. Mar. Biol. 2009, 156, 2641–2647. [Google Scholar] [CrossRef]
- Ward, R.D.; Costa, F.O.; Holmes, B.H.; Steinke, D. DNA barcoding of shared fish species from the North Atlantic and Australasia: minimal divergence for most taxa, but Zeus faber and Lepidopus caudatus each probably constitute two species. Aquatic Biol. 2008, 3, 71–78. [Google Scholar]
- Zemlak, T.S.; Ward, R.D.; Connell, A.D.; Holmes, B.H.; Hebert, P.D.N. DNA barcoding reveals overlooked marine fishes. Mol. Ecol. Resour. 2009, 9, 237–242. [Google Scholar] [CrossRef]
- Vargas, S.M.; Araujo, F.C.F.; Santos, F.R. DNA barcoding of Brazilian sea turtles (Testudines). Genet. Mol. Biol. 2009, 32, 608–612. [Google Scholar] [CrossRef]
- Naro-Maciel, E.; Reid, B.; Fitzsimmons, N.N.; Le, M.; DeSalle, R.; Amato, G. DNA barcodes for globally threatened marine turtles: a registry approach to documenting biodiversity. Mol. Ecol. Resour. 2009, 10, 252–263. [Google Scholar]
- Järnegren, J.; Schander, C.; Sneli, J.A.; Rønningen, V.; Young, C.M. Four genes, morphology and ecology: distinguishing a new species of Acesta (Mollusca; Bivalvia) from the Gulf of Mexico. Mar. Biol. 2007, 152, 43–55. [Google Scholar] [CrossRef]
- Krug, P.J.; Ellingson, R.A.; Burton, R.; Valdés, Á. A new Poecilogonous species of sea slug (Opisthobranchia: Sacoglossa) from California: comparison with the planktotrophic congener Alderia modesta (Loven, 1844). J. Molluscan Stud. 2007, 73, 29–38. [Google Scholar] [CrossRef]
- Derycke, S.; Fonseca, G.; Vierstraete, A.; Vanfleteren, J.; Vincx, M.; Moens, T. Disentangling taxonomy within the Rhabditis (Pellioditis) marina (Nematoda, Rhabditidae) species complex using molecular and morhological tools. Zool. J. Linn. Soc. 2008, 152, 1–15. [Google Scholar] [CrossRef]
- Cardenas, P.; Menegola, C.; Rapp, H.T.; Diaz, M.C. Morphological description and DNA barcodes of shallow-water Tetractinellida (Porifera: Demospongiae) from Bocas del Toro, Panama, with description of a new species. Zootaxa 2009, 2276, 1–39. [Google Scholar]
- De Wit, P.; Rota, E.; Erséus, C. Grania (Annelida: Clitellata: Enchytraeidae) of the Great Barrier Reef, Australia, including four new species and a re-description of Grania trichaeta Jamieson, 1977. Zootaxa 2009, 2165, 16–38. [Google Scholar]
- Byrkjedal, I.; Rees, D.J.; Willassen, E. Lumping lumpsuckers: molecular and morphological insights into the taxonomic status of Eumicrotremus spinosus (Fabricius, 1776) and Eumicrotremus eggvinii Koefoed, 1956 (Teleostei: Cyclopteridae). J. Fish Biol. 2007, 71, 111–131. [Google Scholar]
- Kappner, I.; Bieler, R. Phylogeny of venus clams (Bivalvia: Venerinae) as inferred from nuclear and mitochondrial gene sequences. Mol. Phylogenet. Evol. 2006, 40, 317–331. [Google Scholar] [CrossRef]
- Larsson, K.; Ahmadzadeh, A.; Jondelius, U. DNA taxonomy of Swedish Catenulida (Platyhelminthes) and a phylogenetic framework for catenulid classification. Org. Divers. Evol. 2008, 8, 399–412. [Google Scholar] [CrossRef]
- Costa, F.O.; Henzler, C.M.; Lunt, D H.; Whiteley, N.M.; Rock, J. Probing marine Gammarus (Amphipoda) taxonomy with DNA barcodes. Syst. Biodivers. 2009, 7, 365–379. [Google Scholar]
- Porco, D.; Rougerie, R.; Deharveng, L.; Hebert, P.D.N. Coupling non-destructive DNA extraction and voucher retrieval for small soft-bodied Arthropods in a high-throughput context: the example of Collembola. Mol. Ecol. Resour. 2010. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Miaud, C.; Pompanon, F.; Taberlet, P. Species detection using environmental DNA from water samples. Biol. Lett. 2008, 4, 423–425. [Google Scholar]
- Packer, L.; Grixti, J.C.; Roughley, R.E.; Hanner, R. The status of taxonomy in Canada and the impact of DNA barcoding. Can. J. Zool. 2009, 87, 1097–1110. [Google Scholar] [CrossRef]
- Boero, F. The study of species in the era of biodiversity: a tale of stupidity. Diversity 2010, 2, 115–126. [Google Scholar] [CrossRef]
- Machida, R.J.; Hashiguchi, Y.; Nishida, M.; Nishida, S. Zooplankton diversity analysis through single-gene sequencing of a community sample. BMC Genomics 2009, 10, 438. [Google Scholar]
- Kochzius, M.; Nölte, M.; Weber, H.; Silkenbeumer, N.; Hjörleifsdottir, S.; Hreggvidsson, G.O.; Marteinsson, V.; Kappel, K.; Planes, S.; Tinti, F.; Magoulas, A.; Vazquez, E.G.; Turan, C.; Hervet, C.; Falgueras, D.C.; Antoniou, A.; Landi, M.; Blohm, D. DNA microarrays for identifying fishes. Mar. Biotechnol. 2008, 10, 207–217. [Google Scholar] [CrossRef]
- Warwick, R.M.; Somerfield, P.J. All animals are equal, but some animals are more equal than others. J. Exp. Mar. Biol. Ecol. 2008, 366, 184–186. [Google Scholar] [CrossRef]
- Warwick, R.M.; Clarke, K.R. New ‘biodiversity’ measures reveal a decrease in taxonomic distinctness with increasing stress. Mar. Ecol. Prog. Ser. 1995, 129, 301–305. [Google Scholar] [CrossRef]
- Faith, D.P. Phylogenetic pattern and the quantification of organismal biodiversity. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 1994, 345, 45–58. [Google Scholar] [CrossRef]
- Faith, D.P.; Baker, A.M. Phylogenetic diversity (PD) and biodiversity conservation: some bioinformatics challenges. Evol. Bioinform. 2006, 2, 121–128. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Radulovici, A.E.; Archambault, P.; Dufresne, F. DNA Barcodes for Marine Biodiversity: Moving Fast Forward? Diversity 2010, 2, 450-472. https://doi.org/10.3390/d2040450
Radulovici AE, Archambault P, Dufresne F. DNA Barcodes for Marine Biodiversity: Moving Fast Forward? Diversity. 2010; 2(4):450-472. https://doi.org/10.3390/d2040450
Chicago/Turabian StyleRadulovici, Adriana E., Philippe Archambault, and France Dufresne. 2010. "DNA Barcodes for Marine Biodiversity: Moving Fast Forward?" Diversity 2, no. 4: 450-472. https://doi.org/10.3390/d2040450
APA StyleRadulovici, A. E., Archambault, P., & Dufresne, F. (2010). DNA Barcodes for Marine Biodiversity: Moving Fast Forward? Diversity, 2(4), 450-472. https://doi.org/10.3390/d2040450





