Comparative Prevalence and Intensity of Endoparasites in a Dynamic Boreal Ungulate Community
Abstract
1. Introduction
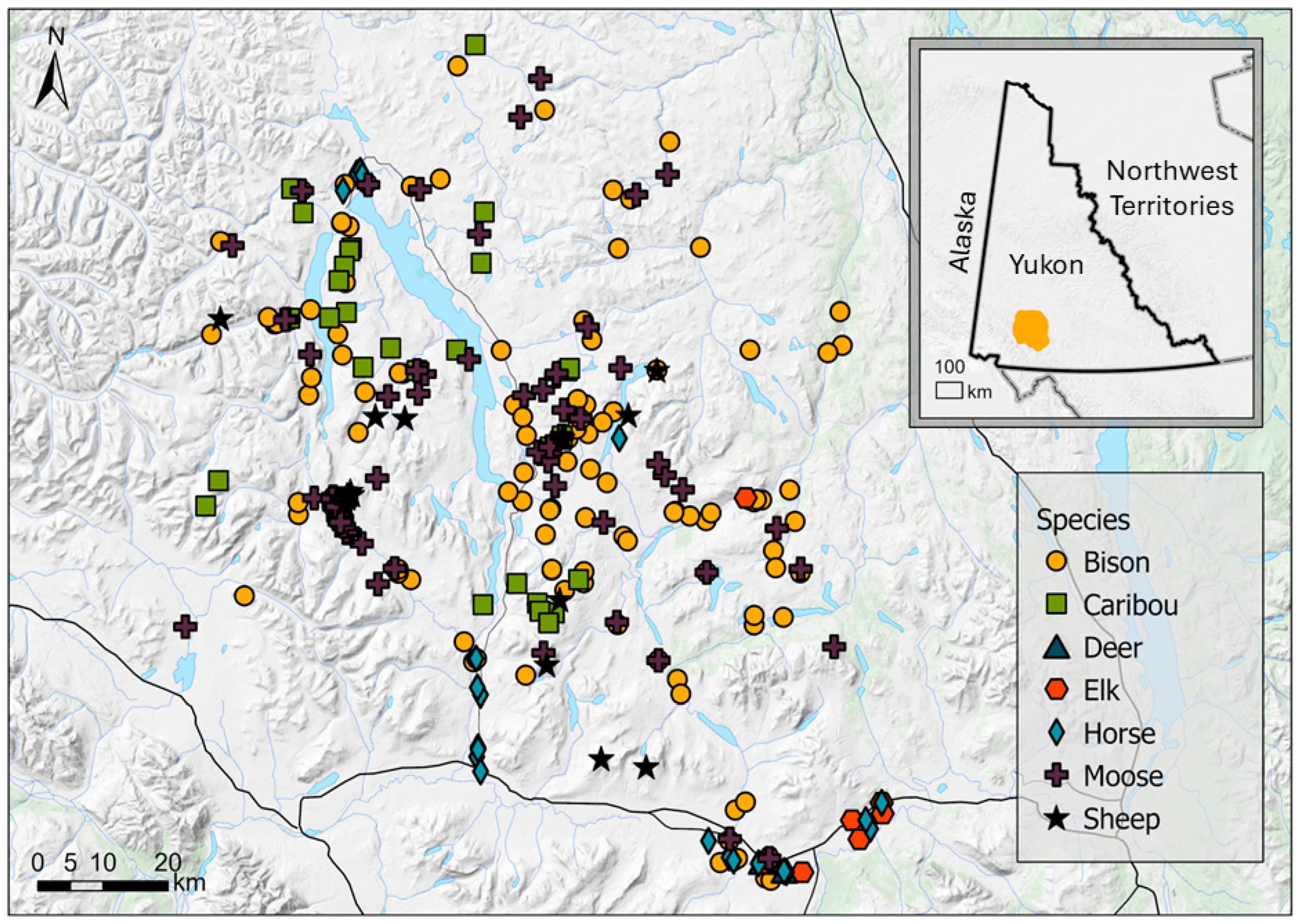
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoberg, E.P.; Kocan, A.A.; Rickard, L.G. Gastrointestinal strongyles in wild ruminants. In Parasitic Diseases of Wild Mammals; Samuel, W.M., Pybus, M.J., Kocan, A.A., Eds.; Wiley: New York, NY, USA, 2001; pp. 193–227. [Google Scholar]
- Gulland, F.M.D. The role of nematode parasites in soay sheep (Ovis aries L.) mortality during a population crash. Parasitology 1992, 105, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Lankester, M.W. Extrapulmonary lungworms of cervids. In Parasitic Diseases of Wild Mammals; Samuel, W.M., Pybus, M.J., Kocan, A.A., Eds.; Wiley: New York, NY, USA, 2001; pp. 228–278. [Google Scholar]
- Eljaki, A.A.; Al Kappany, Y.M.; Grosz, D.D.; Smart, A.J.; Hildreth, M.B. Molecular survey of trichostrongyle nematodes in a Bison bison herd experiencing clinical parasitism, and effects of avermectin treatment. Vet. Parasitol. 2016, 227, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Kutz, S.J.; Asmundsson, I.; Hoberg, E.P.; Appleyard, G.D.; Jenkins, E.J.; Beckmen, K.; Branigan, M.; Butler, L.; Chilton, N.B.; Cooley, D.; et al. Serendipitous discovery of a novel protostrongylid (Nematoda: Metastrongyloidea) in caribou, muskoxen, and moose from high latitudes of North America based on DNA sequence comparisons. Can. J. Zool. 2007, 85, 1143–1156. [Google Scholar] [CrossRef]
- Jung, T.S.; Stotyn, S.A.; Czetwertynski, S.M. Dietary overlap and potential competition in a dynamic ungulate community in Northwestern Canada. J. Wildl. Manag. 2015, 79, 1277–1285. [Google Scholar] [CrossRef]
- Jung, T.S.; Hegel, T.M.; Stotyn, S.A.; Czetwertynski, S.M. Co-occurrence of reintroduced and resident ungulates on a shared winter range in northwestern Canada. Écoscience 2015, 22, 7–16. [Google Scholar]
- Jung, T.S. Investigating local concerns regarding large mammal restoration: Group size in a growing population of reintroduced bison (Bison bison). Glob. Ecol. Conserv. 2020, 24, e01303. [Google Scholar] [CrossRef]
- Jung, T.S.; Czetwertynski, S.M.; Schmiegelow, F.K.A. Boreal forest titans do not clash: Low overlap in winter habitat selection by moose (Alces americanus) and reintroduced bison (Bison bison). Eur. J. Wildl. Res. 2018, 64, 25. [Google Scholar] [CrossRef]
- Strong, W.L.; Chambers, J.H.S.; Jung, T.S. Range constraints for introduced elk in southwest Yukon, Canada. Arctic 2013, 66, 470–482. [Google Scholar] [CrossRef]
- Hoefs, M. Mule, Odocoileus hemionus, and white-tailed, O. virginianus, deer in the Yukon. Can. Field-Nat. 2001, 115, 296–300. [Google Scholar] [CrossRef]
- Jenkins, E.J.; Veitch, A.M.; Kutz, S.J.; Hoberg, E.P.; Polley, L. Climate change and the epidemiology of protostrongylid nematodes in northern ecosystems: Parelaphostrongylus odocoilei and Protostrongylus stilesi in Dall’s sheep (Ovis d. dalli). Parasitology 2006, 132, 387–401. [Google Scholar] [CrossRef][Green Version]
- Hoberg, E.P.; Brooks, D.R. A macroevolutionary mosaic: Episodic host-switching, geographical colonization and diversification in complex host-parasite systems. J. Biogeogr. 2008, 35, 1533–1550. [Google Scholar] [CrossRef]
- Kutz, S.J.; Jenkins, E.J.; Veitch, A.M.; Ducrocq, J.; Polley, L.; Elkin, B.; Lair, S. The Arctic as a model for anticipating, preventing, and mitigating climate change impacts on host–parasite interactions. Vet. Parasitol. 2009, 163, 217–228. [Google Scholar] [CrossRef] [PubMed]
- de Bruyn, N.P. Gastrointestinal Nematodes of Western Canadian Cervids: Molecular Diagnostics, Faunal Baselines and Management Considerations. Master’s Thesis, University of Calgary, Calgary, AB, Canada, 2010. [Google Scholar]
- Kutz, S.J.; Ducrocq, J.; Verocai, G.G.; Hoar, B.M.; Colwell, D.D.; Beckmen, K.B.; Polley, L.; Elkin, B.T.; Hoberg, E.P. Parasites in ungulates of arctic North America and Greenland. Adv. Parasitol. 2012, 79, 99–252. [Google Scholar] [PubMed]
- Kutz, S.J.; Hoberg, E.P.; Polley, L.; Jenkins, E.J. Global warming is changing the dynamics of Arctic host–parasite systems. Proc. R. Soc. B. 2005, 272, 2571–2576. [Google Scholar] [CrossRef] [PubMed]
- Kafle, P.; Peller, P.; Massolo, A.; Hoberg, E.; Leclerc, L.-M.; Tomaselli, M.; Kutz, S.J. Range expansion of muskox lungworms track rapid arctic warming: Implications for geographic colonization under climate forcing. Sci. Rep. 2020, 10, 17323. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.J.; Peters-Kennedy, J.; Stokol, T.; Gerhold, R.W.; Beckstead, R.B.; Divers, T.J. Diagnosis of Parelaphostrongylus spp. infection as a cause of meningomyelitis in calves. J. Vet. Diagn. Investig. 2011, 23, 1097–1103. [Google Scholar] [CrossRef] [PubMed]
- Harms, N.J.; Jung, T.S.; Andrew, C.L.; Surujballi, O.P.; VanderKop, M.; Savic, M.; Powell, T. Health status of reintroduced wood bison (Bison bison athabascae): Assessing the conservation value of an isolated population in northwestern Canada. J. Wildl. Dis. 2019, 55, 44–53. [Google Scholar] [PubMed]
- Ecological Stratification Working Group. A National Ecological Framework for Canada; Agriculture and Agri-Food Canada and Environment Canada: Ottawa, ON, Canada, 1995.
- Jung, T.S.; Kukka, P.M. Influence of habitat type on decay and disappearance of elk (Cervus canadensis) pellets in boreal forest of northwestern Canada. Wildl. Biol. 2016, 22, 160–166. [Google Scholar] [CrossRef]
- Verocai, G.G.; Kutz, S.J.; Simard, M.; Hoberg, E.P. Varestrongylus eleguneniensis sp. n. (Nematoda: Protostrongylidae): A widespread, multi-host lungworm of wild North American ungulates, with an emended diagnosis for the genus and explorations of biogeography. Parasit. Vectors 2014, 7, 556. [Google Scholar] [CrossRef] [PubMed]
- Filip-Hutsch, K.; Czopowicz, M.; Świsłocka, M.; Ratkiewicz, M.; Borkowska, A.; Kowalczyk, R.; Demiaszkiewicz, A.W. Patterns of parasite eggs, oocysts and larvae shedding by moose in the Biebrza marshland (NE Poland). Int. J. Parasitol. Parasites Wildl. 2020, 11, 191–197. [Google Scholar] [CrossRef]
- Świsłocka, M.; Borkowska, A.; Matosiuk, M.; Czajkowska, M.; Duda, N.; Kowalczyk, R.; Ratkiewicz, M. Sex-biased polyparasitism in moose (Alces alces) based on molecular analysis of faecal samples. Int. J. Parasitol. Parasites Wildl. 2020, 13, 171–177. [Google Scholar] [CrossRef]
- Idland, L.; Juul, A.M.; Solevåg, E.K.; Tysnes, K.R.; Robertson, L.J.; Utaaker, K.S. Occurrence of faecal endoparasites in reindeer (Rangifer tarandus) in two grazing areas in northern Norway. Acta Vet. Scand. 2021, 63, 13. [Google Scholar] [CrossRef] [PubMed]
- Kafle, P.; Peacock, S.J.; Grond, S.; Orsel, K.; Kutz, S. Temperature-dependent development and freezing survival of protostrongylid nematodes of Arctic ungulates: Implications for transmission. Parasit. Vectors 2018, 11, 400. [Google Scholar] [CrossRef] [PubMed]
- Schurer, J.; Davenport, L.; Wagner, B.; Jenkins, E. Effects of sub-zero storage temperatures on endoparasites in canine and equine feces. Vet. Parasitol. 2014, 204, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Cox, D.D.; Todd, A.C. Survey of gastrointestinal parasitism in Wisconsin dairy cattle. J. Am. Vet. Med. Assoc. 1962, 141, 706–709. [Google Scholar]
- Narsapur, V.S. Pathogenesis and biology of anoplocephaline cestodes of domestic animals. Ann. Rech. Vet. 1988, 19, 1–17. [Google Scholar] [PubMed]
- Samuel, W.M.; Gray, D.R. Parasitic infection in muskoxen. J. Wildl. Manag. 1974, 38, 775–782. [Google Scholar] [CrossRef]
- Verocai, G.G.; Kafle, P.; Sulliotti, V.; Lejeune, M.; Hoberg, E.P.; Kutz, S.J. Morphometry of first-stage larvae of Orthostrongylus macrotis (Nematoda: Protostrongylidae), lungworm of wild ungulates from western North America. J. Parasitol. 2022, 108, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Boddicker, M.L.; Hugghins, E.J. Helminths of big game mammals in South Dakota. J. Parasitol. 1969, 55, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Greiner, E.C.; Worley, D.E.; O’Gara, B.W. Protostrongylus macrotis (Nematoda: Metastrongyloidea) in pronghorn antelope from Montana and Wyoming. J. Wildl. Dis. 1974, 10, 70–73. [Google Scholar] [CrossRef]
- Pybus, M.J. Survey of hepatic and pulmonary helminths of wild cervids in Alberta, Canada. J. Wildl. Dis. 1990, 26, 453–459. [Google Scholar] [CrossRef]
- Belem, A.M.G.; Couvillion, C.E.; Siefker, C.; Griffin, R.N. Evidence for arrested development of abomasal nematodes in white-tailed deer. J. Wildl. Dis. 1993, 29, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Samuel, W.M.; Barrett, M.W.; Lynch, G.M. Helminths in moose of Alberta. Can. J. Zool. 1976, 54, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Thorne, T. Diseases of Wildlife in Wyoming, 2nd ed.; Wyoming Game and Fish Department: Cheyenne, WY, USA, 1982.
- deVos, J.C., Jr.; McKinney, T. Potential impacts of global climate change on abundance and distribution of elk and mule deer in western North America. Final. Rep. West. Assoc. Fish Wildl. Agencies 2007, 32, 1369–1378. [Google Scholar]
- Kutz, S.J.; Hoberg, E.P.; Molnár, P.K.; Dobson, A.; Verocai, G.G. A walk on the tundra: Host–parasite interactions in an extreme environment. Int. J. Parasitol. Parasites Wildl. 2014, 3, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Hoberg, E.P.; Brooks, D.R. Evolution in action: Climate change, biodiversity dynamics and emerging infectious disease. Phil. Trans. R. Soc. B 2015, 370, 20130553. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.R.; Hoberg, E.P.; Boeger, W.A. The Stockholm Paradigm: Climate Change and Emerging Disease; University of Chicago Press: Chicago, IL, USA, 2019. [Google Scholar]
- Cunningham, A.; Daszak, P.; Rodríguez, J. Pathogen pollution: Defining a parasitological threat to biodiversity conservation. J. Parasitol. 2003, 89, S78–S83. [Google Scholar]
- Hoberg, E.P.; Kutz, S.J.; Nagy, J.; Jenkins, E.J.; Elkin, B.; Branigan, M.; Cooley, D. Protostrongylus stilesi (Nematoda: Protostrongylidae): Ecological isolation and putative host-switching between Dall’s sheep and muskoxen in a contact zone. Comp. Parasitol. 2002, 69, 1–9. [Google Scholar] [CrossRef][Green Version]
- Jenkins, E.J.; Appleyard, G.D.; Hoberg, E.P.; Rosenthal, B.M.; Kutz, S.J.; Veitch, A.M.; Schwantje, H.M.; Elkin, B.T.; Polley, L. Geographic distribution of the muscle-dwelling nematode Parelaphostrongylus odocoilei in North America, using molecular identification of first-stage larvae. J. Parasitol. 2005, 91, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Kutz, S.J.; Veitch, A.M.; Hoberg, E.P.; Elkin, B.T.; Jenkins, E.J.; Polley, L. New host and geographic records for two protostrongylids in Dall’s sheep. J. Wildl. Dis. 2001, 37, 761–774. [Google Scholar] [CrossRef]
- Jenkins, E.J.; Veitch, A.M.; Kutz, S.J.; Bollinger, T.K.; Chirino-Trejo, J.M.; Elkin, B.T.; West, K.H.; Hoberg, E.P.; Polley, L. Protostrongylid parasites and pneumonia in captive and wild thinhorn sheep (Ovis dalli). J. Wildl. Dis. 2007, 43, 189–205. [Google Scholar] [CrossRef]
- Johnson, D.; Harms, N.J.; Larter, N.C.; Elkin, B.T.; Tabel, H.; Wei, G. Serum biochemistry, serology, and parasitology of boreal caribou (Rangifer tarandus caribou) in the Northwest Territories, Canada. J. Wildl. Dis. 2010, 46, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Barone, C.D.; Wit, J.; Hoberg, E.P.; Gilleard, J.S.; Zarlenga, D.S. Wild ruminants as reservoirs of domestic livestock gastrointestinal nematodes. Vet. Parasitol. 2020, 279, 109041. [Google Scholar] [CrossRef] [PubMed]
- Tessaro, S.V. Review of the diseases, parasites and miscellaneous pathological conditions of North American bison. Can. Vet. J. 1989, 30, 416–422. [Google Scholar] [PubMed]
- Avramenko, R.W.; Bras, A.; Redman, E.M.; Woodbury, M.R.; Wagner, B.; Shury, T.; Liccioli, S.; Windeyer, M.C.; Gilleard, J.S. High species diversity of trichostrongyle parasite communities within and between western Canadian commercial and conservation bison herds revealed by nemabiome metabarcoding. Parasit. Vectors 2018, 11, 299. [Google Scholar] [CrossRef] [PubMed]
- Beaumelle, C.; Redman, E.M.; de Rijke, J.; Wit, J.; Benabed, S.; Debias, F.; Duhayer, J.; Pardonnet, S.; Poirel, M.T.; Capron, G.; et al. Metabarcoding in two isolated populations of wild roe deer (Capreolus capreolus) reveals variation in gastrointestinal nematode community composition between regions and among age classes. Parasit. Vectors 2021, 14, 594. [Google Scholar] [CrossRef] [PubMed]
- Hoberg, E.P.; Polley, L.; Jenkins, E.J.; Kutz, S.J. Pathogens of domestic and free-ranging ungulates: Global climate change in temperate to boreal latitudes across North America. Rev. Sci. Tech. 2008, 27, 511–528. [Google Scholar] [CrossRef] [PubMed]
- Hoberg, E.P.; Polley, L.; Jenkins, E.J.; Kutz, S.J.; Veitch, A.M.; Elkin, B.T. Integrated approaches and empirical models for investigation of parasitic diseases in northern Wildlife. Emerg. Infect. Dis. 2008, 14, 10–17. [Google Scholar] [CrossRef]
- Festa-Bianchet, M.; Ray, J.C.; Boutin, S.; Côté, S.D.; Gunn, A. Conservation of caribou (Rangifer tarandus) in Canada: An uncertain future. Can. J. Zool. 2011, 89, 419–434. [Google Scholar] [CrossRef]
- van Beest, F.M.; Petersen, H.H.; Krogh, A.K.H.; Frederiksen, M.L.; Schmidt, N.M.; Hansson, S.V. Estimating parasite-condition relationships and potential health effects for fallow deer (Dama dama) and red deer (Cervus elaphus) in Denmark. Int. J. Parasitol. Parasites Wildl. 2023, 21, 143–152. [Google Scholar] [CrossRef]

| Endoparasite Group | Bison (n = 83) | Caribou (n = 38) | Moose (n = 139) | Thinhorn Sheep (n = 35) | Elk (n = 9) | Mule Deer (n = 4) |
|---|---|---|---|---|---|---|
| Trichostrongyle | 1% (1) | 21% (8) | 7% (10) | 6% (2) | 11% (1) | 0 (0) |
| Nematodirid | 0 (0) | 11% (4) | 25% (34) | 49% (17) | 11% (1) | 0 (0) |
| Trichuris sp. | 1% (1) | 3% (1) | 0 (0) | 43% (15) | 0 (0) | 0 (0) |
| Eimeria sp. | 5% (4) | 5% (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Spike-tailed larvae | no data | 5% (2) | 0 (0) | 74% (26) | 22% (2) | 0 (0) |
| Dorsal-spined larvae | no data | 3% (1) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Cestodes | 33% (27) | 11% (4) | 7% (9) | 3% (1) | 0 (0) | 0 (0) |
| Endoparasite Group | Metric | Bison | Caribou | Moose | Thinhorn Sheep | Elk |
|---|---|---|---|---|---|---|
| Trichostrongyle | Median Mean ± SE Range | 1.6 1.6 (1.6) | 0.4 0.6 ± 0.5 (0.2–1.4) | 0.6 1.0 ± 0.9 (0.2–3.0) | 0.3 0.3 ± 0.1 (0.2–0.4) | 0.4 0.4 (0.4) |
| Nematodirid | Median Mean ± SE Range | 0 | 1.4 1.3 ± 0.7 (0.2–2.2) | 0.5 1.3 ± 1.8 (0.2–10.2) | 0.4 1.2 ± 1.2 (0.2–3.4) | 0.2 0.2 (0.2) |
| Trichuris sp. | Median Mean ± SE Range | 0.2 0.2 (0.2) | 1.2 1.2 (1.2) | 0 | 1.2 3.5 ± 5.9 (0.2–20.6) | 0 |
| Spike-tailed larvae | Median Mean ± SE Range | no data | 9.3 9.3 ± 3.5 (5.8–12.8) | 0 | 25.5 181.4 ± 412.5 (0.6–1675.6) | 3.1 3.1 ± 2.3 (0.8–5.4) |
| Dorsal-spined larvae | Median Mean ± SE Range | no data | 51 51 (51) | 0 | 0 | 0 |
| Cestodes | Median Mean ± SE Range | 1.8 4.4 ± 5.2 (0.2–17.2) | 7.5 9.9 ± 8.9 (0.4–10.6) | 6.6 31.0 ± 74.0 (0.2–240.0) | 9.6 9.6 (9.6) | 0 |
| Endoparasite Group | Prevalence | Intensity (Eggs per Gram) | |||
|---|---|---|---|---|---|
| 2010 | 2018 | 2010 | 2018 | ||
| Trichostrongyle | 1% (1) | 93% (81) | 1.6 (1.6 [1.6]) | 7.8 (11.1 ± 10.1 [0.2–42.4]) | |
| Nematodirid | 0 (0) | 0 (0) | 0 (0) | 0 (0) | |
| Trichuris sp. | 1% (1) | 3% (3) | 0.2 (0.2 [0.2]) | 0.4 (0.3 ± 0.1 [0.2–0.4]) | |
| Eimeria sp. | 5% (4) | 2% (2) | no data | no data | |
| Cestodes | 33% (27) | 33% (29) | 1.8 (4.4 ± 5.2 [0.2–17.2]) | 8.2 (12.8 ± 15.6 [0.2–77.8]) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrew, C.L.; Wagner, B.; Harms, N.J.; Jenkins, E.J.; Jung, T.S. Comparative Prevalence and Intensity of Endoparasites in a Dynamic Boreal Ungulate Community. Diversity 2024, 16, 230. https://doi.org/10.3390/d16040230
Andrew CL, Wagner B, Harms NJ, Jenkins EJ, Jung TS. Comparative Prevalence and Intensity of Endoparasites in a Dynamic Boreal Ungulate Community. Diversity. 2024; 16(4):230. https://doi.org/10.3390/d16040230
Chicago/Turabian StyleAndrew, Cassandra L., Brent Wagner, N. Jane Harms, Emily J. Jenkins, and Thomas S. Jung. 2024. "Comparative Prevalence and Intensity of Endoparasites in a Dynamic Boreal Ungulate Community" Diversity 16, no. 4: 230. https://doi.org/10.3390/d16040230
APA StyleAndrew, C. L., Wagner, B., Harms, N. J., Jenkins, E. J., & Jung, T. S. (2024). Comparative Prevalence and Intensity of Endoparasites in a Dynamic Boreal Ungulate Community. Diversity, 16(4), 230. https://doi.org/10.3390/d16040230







