A Beacon in the Dark: Grey Literature Data Mining and Machine Learning Enlightening Historical Plankton Seasonality Dynamics in the Ligurian Sea
Abstract
1. Introduction
2. Materials and Methods
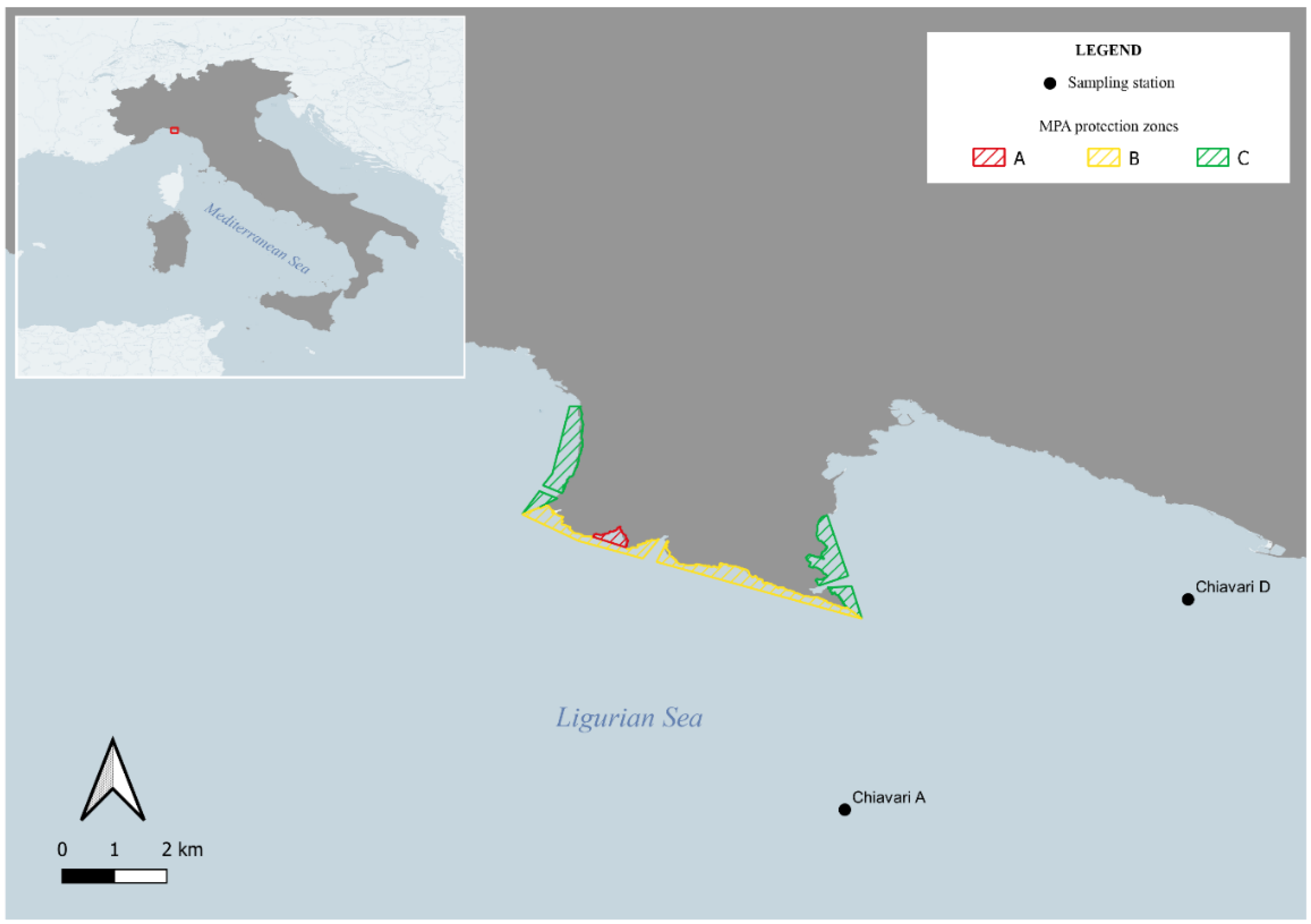
2.1. Study Area
2.2. Data Elaboration and Modeling
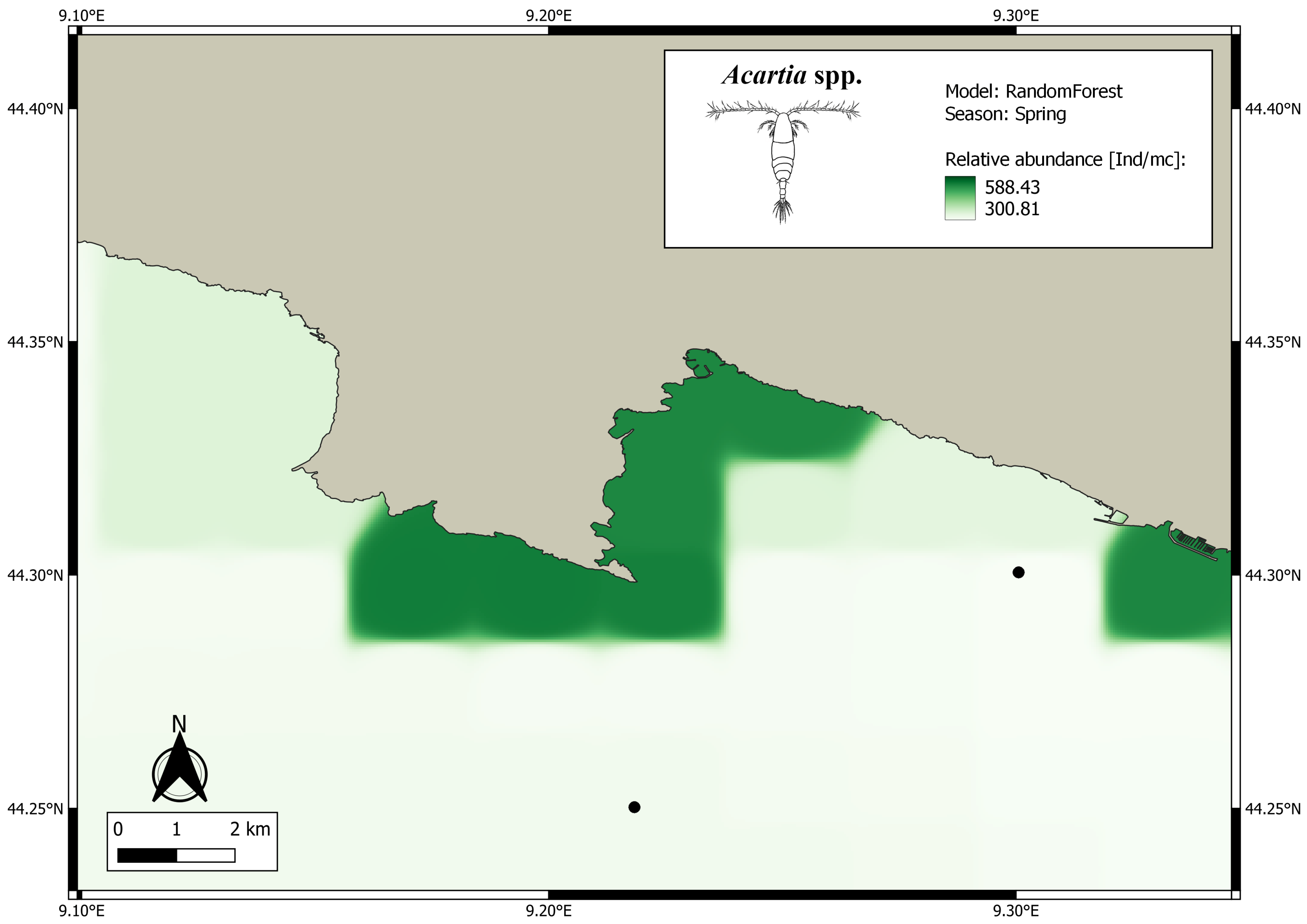
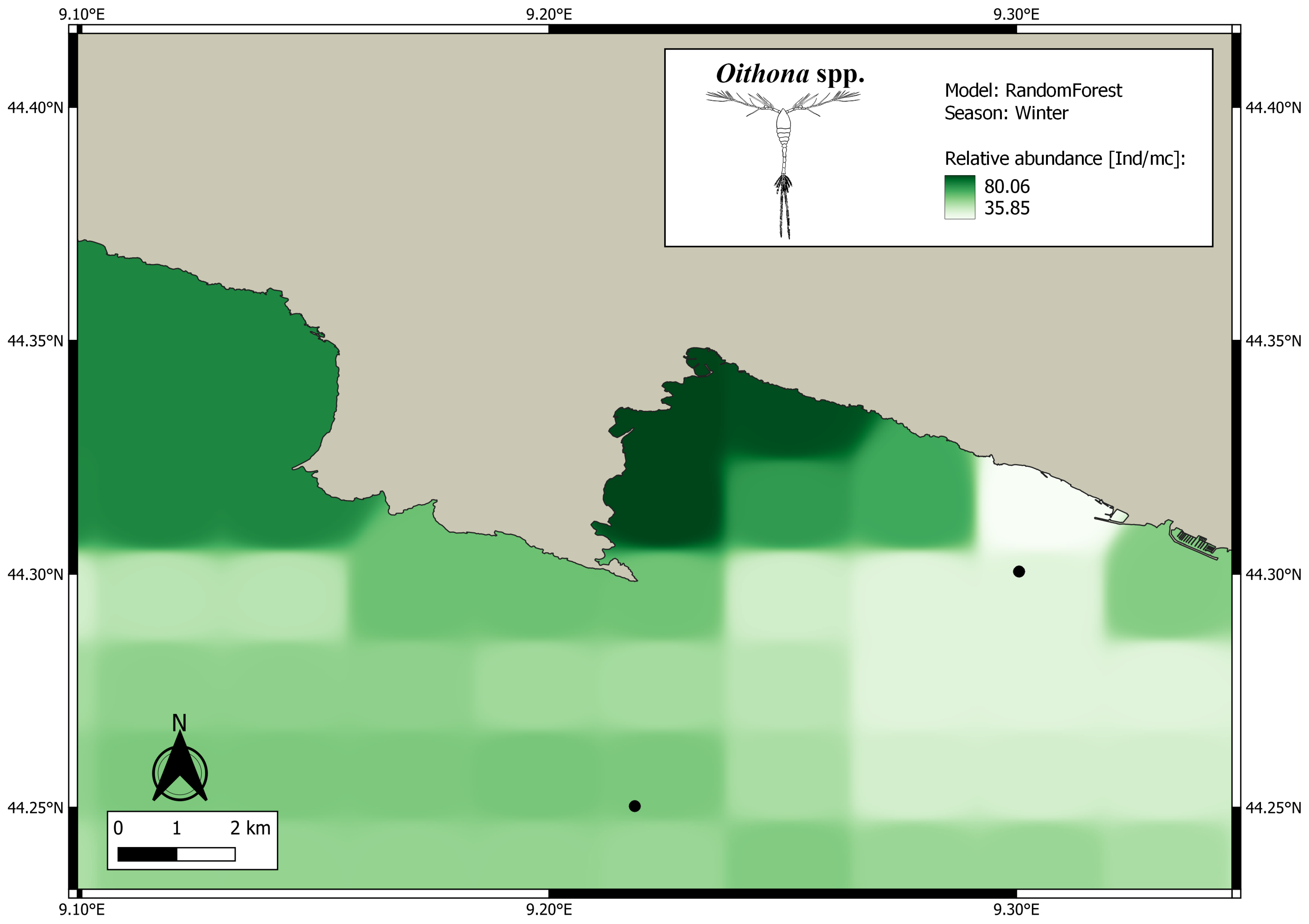
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
References
- Díaz, S.M.; Settele, J.; Brondízio, E.; Ngo, H.; Guèze, M.; Agard, J.; Arneth, A.; Balvanera, P.; Brauman, K.; Butchart, S. The Global Assessment Report on Biodiversity and Ecosystem Services: Summary for Policy Makers; IPBES: Bonn, Germany, 2019. [Google Scholar]
- Feliciano, D.; Recha, J.; Ambaw, G.; MacSween, K.; Solomon, D.; Wollenberg, E. Assessment of Agricultural Emissions, Climate Change Mitigation and Adaptation Practices in Ethiopia. Clim. Policy 2022, 22, 427–444. [Google Scholar] [CrossRef]
- Parmesan, C. Ecological and Evolutionary Responses to Recent Climate Change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Bruno, J.F. The Impact of Climate Change on the World’s Marine Ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Doney, S.C.; Ruckelshaus, M.; Emmett Duffy, J.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate Change Impacts on Marine Ecosystems. Annu. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef]
- Russell, B.D.; Harley, C.D.G.; Wernberg, T.; Mieszkowska, N.; Widdicombe, S.; Hall-Spencer, J.M.; Connell, S.D. Predicting Ecosystem Shifts Requires New Approaches That Integrate the Effects of Climate Change across Entire Systems. Biol. Lett. 2012, 8, 164–166. [Google Scholar] [CrossRef]
- Nadeau, C.P.; Urban, M.C.; Bridle, J.R. Climates Past, Present, and Yet-to-Come Shape Climate Change Vulnerabilities. Trends Ecol. Evol. 2017, 32, 786–800. [Google Scholar] [CrossRef]
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.-C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B.; et al. Biodiversity Redistribution under Climate Change: Impacts on Ecosystems and Human Well-Being. Science 2017, 355, eaai9214. [Google Scholar] [CrossRef]
- Manes, S.; Costello, M.J.; Beckett, H.; Debnath, A.; Devenish-Nelson, E.; Grey, K.-A.; Jenkins, R.; Khan, T.M.; Kiessling, W.; Krause, C. Endemism Increases Species’ Climate Change Risk in Areas of Global Biodiversity Importance. Biol. Conserv. 2021, 257, 109070. [Google Scholar] [CrossRef]
- Pörtner, H.O.; Knust, R. Climate Change Affects Marine Fishes through the Oxygen Limitation of Thermal Tolerance. Science 2007, 315, 95–97. [Google Scholar] [CrossRef]
- Hofmann, G.E.; Todgham, A.E. Living in the Now: Physiological Mechanisms to Tolerate a Rapidly Changing Environment. Annu. Rev. Physiol. 2010, 72, 127–145. [Google Scholar] [CrossRef]
- Sorte, C.J.; Jones, S.J.; Miller, L.P. Geographic Variation in Temperature Tolerance as an Indicator of Potential Population Responses to Climate Change. J. Exp. Mar. Biol. Ecol. 2011, 400, 209–217. [Google Scholar] [CrossRef]
- Minuti, J.J.; Byrne, M.; Hemraj, D.A.; Russell, B.D. Capacity of an Ecologically Key Urchin to Recover from Extreme Events: Physiological Impacts of Heatwaves and the Road to Recovery. Sci. Total Environ. 2021, 785, 147281. [Google Scholar] [CrossRef]
- Dupont, S.; Dorey, N.; Thorndyke, M. What Meta-Analysis Can Tell Us about Vulnerability of Marine Biodiversity to Ocean Acidification? Estuar. Coast. Shelf Sci. 2010, 89, 182–185. [Google Scholar] [CrossRef]
- Byrne, M.; Przeslawski, R. Multistressor Impacts of Warming and Acidification of the Ocean on Marine Invertebrates’ Life Histories. Integr. Comp. Biol. 2013, 53, 582–596. [Google Scholar] [CrossRef]
- Reusch, T.B.H. Climate Change in the Oceans: Evolutionary versus Phenotypically Plastic Responses of Marine Animals and Plants. Evol. Appl. 2014, 7, 104–122. [Google Scholar] [CrossRef]
- Leung, J.Y.; Russell, B.D.; Connell, S.D. Adaptive Responses of Marine Gastropods to Heatwaves. One Earth 2019, 1, 374–381. [Google Scholar] [CrossRef]
- Leung, J.Y.; Russell, B.D.; Connell, S.D. Linking Energy Budget to Physiological Adaptation: How a Calcifying Gastropod Adjusts or Succumbs to Ocean Acidification and Warming. Sci. Total Environ. 2020, 715, 136939. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Sgrò, C.M. Climate Change and Evolutionary Adaptation. Nature 2011, 470, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Silbiger, N.J.; Sorte, C.J. Biophysical Feedbacks Mediate Carbonate Chemistry in Coastal Ecosystems across Spatiotemporal Gradients. Sci. Rep. 2018, 8, 796. [Google Scholar] [CrossRef]
- Eriander, L.; Wrange, A.-L.; Havenhand, J.N. Simulated Diurnal pH Fluctuations Radically Increase Variance in—But Not the Mean of—Growth in the Barnacle Balanus Improvisus. ICES J. Mar. Sci. 2016, 73, 596–603. [Google Scholar] [CrossRef]
- Boyd, P.W.; Lennartz, S.T.; Glover, D.M.; Doney, S.C. Biological Ramifications of Climate-Change-Mediated Oceanic Multi-Stressors. Nat. Clim. Chang. 2015, 5, 71–79. [Google Scholar] [CrossRef]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Ben Rais Lasram, F.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T. The Biodiversity of the Mediterranean Sea: Estimates, Patterns, and Threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [PubMed]
- Chatzimentor, A.; Doxa, A.; Katsanevakis, S.; Mazaris, A.D. Are Mediterranean Marine Threatened Species at High Risk by Climate Change? Glob. Chang. Biol. 2023, 29, 1809–1821. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.N.; Morri, C. Marine Biodiversity of the Mediterranean Sea: Situation, Problems and Prospects for Future Research. Mar. Pollut. Bull. 2000, 40, 367–376. [Google Scholar] [CrossRef]
- Garrabou, J.; Gómez-Gras, D.; Medrano, A.; Cerrano, C.; Ponti, M.; Schlegel, R.; Bensoussan, N.; Turicchia, E.; Sini, M.; Gerovasileiou, V.; et al. Marine Heatwaves Drive Recurrent Mass Mortalities in the Mediterranean Sea. Glob. Chang. Biol. 2022, 28, 5708–5725. [Google Scholar] [CrossRef] [PubMed]
- Pörtner, H.-O.; Roberts, D.C.; Masson-Delmotte, V.; Zhai, P.; Tignor, M.; Poloczanska, E.; Weyer, N.M. The Ocean and Cryosphere in a Changing Climate; Special Report on the Ocean and Cryosphere in a Changing Climate; IPCC: Geneva, Switzerland, 2019; Volume 1155. [Google Scholar]
- Marbà, N.; Jordà, G.; Agustí, S.; Girard, C.; Duarte, C.M. Footprints of Climate Change on Mediterranean Sea Biota. Front. Mar. Sci. 2015, 2, 56. [Google Scholar] [CrossRef]
- Albouy, C.; Guilhaumon, F.; Araújo, M.B.; Mouillot, D.; Leprieur, F. Combining Projected Changes in Species Richness and Composition Reveals Climate Change Impacts on Coastal Mediterranean Fish Assemblages. Glob. Chang. Biol. 2012, 18, 2995–3003. [Google Scholar] [CrossRef]
- Albouy, C.; Guilhaumon, F.; Leprieur, F.; Lasram, F.B.R.; Somot, S.; Aznar, R.; Velez, L.; Le Loc’h, F.; Mouillot, D. Projected Climate Change and the Changing Biogeography of Coastal Mediterranean Fishes. J. Biogeogr. 2013, 40, 534–547. [Google Scholar] [CrossRef]
- Thomas, M.K.; Kremer, C.T.; Klausmeier, C.A.; Litchman, E. A Global Pattern of Thermal Adaptation in Marine Phytoplankton. Science 2012, 338, 1085–1088. [Google Scholar] [CrossRef]
- Hazen, E.L.; Jorgensen, S.; Rykaczewski, R.R.; Bograd, S.J.; Foley, D.G.; Jonsen, I.D.; Shaffer, S.A.; Dunne, J.P.; Costa, D.P.; Crowder, L.B. Predicted Habitat Shifts of Pacific Top Predators in a Changing Climate. Nat. Clim. Chang. 2013, 3, 234–238. [Google Scholar] [CrossRef]
- Pinsky, M.L.; Worm, B.; Fogarty, M.J.; Sarmiento, J.L.; Levin, S.A. Marine Taxa Track Local Climate Velocities. Science 2013, 341, 1239–1242. [Google Scholar] [CrossRef]
- Barton, A.D.; Irwin, A.J.; Finkel, Z.V.; Stock, C.A. Anthropogenic Climate Change Drives Shift and Shuffle in North Atlantic Phytoplankton Communities. Proc. Natl. Acad. Sci. USA 2016, 113, 2964–2969. [Google Scholar] [CrossRef]
- Record, N.R.; Runge, J.A.; Pendleton, D.E.; Balch, W.M.; Davies, K.T.; Pershing, A.J.; Johnson, C.L.; Stamieszkin, K.; Ji, R.; Feng, Z. Rapid Climate-Driven Circulation Changes Threaten Conservation of Endangered North Atlantic Right Whales. Oceanography 2019, 32, 162–169. [Google Scholar] [CrossRef]
- Striebel, M.; Kallajoki, L.; Kunze, C.; Wollschläger, J.; Deininger, A.; Hillebrand, H. Marine Primary Producers in a Darker Future: A Meta-analysis of Light Effects on Pelagic and Benthic Autotrophs. Oikos 2023, 2023, e09501. [Google Scholar] [CrossRef]
- McGinty, N.; Barton, A.D.; Record, N.R.; Finkel, Z.V.; Johns, D.G.; Stock, C.A.; Irwin, A.J. Anthropogenic Climate Change Impacts on Copepod Trait Biogeography. Glob. Chang. Biol. 2021, 27, 1431–1442. [Google Scholar] [CrossRef]
- Hays, G.C.; Richardson, A.J.; Robinson, C. Climate Change and Marine Plankton. Trends Ecol. Evol. 2005, 20, 337–344. [Google Scholar] [CrossRef]
- Steinberg, D.K.; Landry, M.R. Zooplankton and the Ocean Carbon Cycle. Annu. Rev. Mar. Sci. 2017, 9, 413–444. [Google Scholar] [CrossRef]
- Drago, L.; Panaïotis, T.; Irisson, J.-O.; Babin, M.; Biard, T.; Carlotti, F.; Coppola, L.; Guidi, L.; Hauss, H.; Karp-Boss, L. Global Distribution of Zooplankton Biomass Estimated by in Situ Imaging and Machine Learning. Front. Mar. Sci. 2022, 9, 894372. [Google Scholar] [CrossRef]
- Richardson, A.J. In Hot Water: Zooplankton and Climate Change. ICES J. Mar. Sci. 2008, 65, 279–295. [Google Scholar] [CrossRef]
- Bedford, J.; Ostle, C.; Johns, D.G.; Atkinson, A.; Best, M.; Bresnan, E.; Machairopoulou, M.; Graves, C.A.; Devlin, M.; Milligan, A.; et al. Lifeform Indicators Reveal Large-scale Shifts in Plankton across the North-West European Shelf. Glob. Chang. Biol. 2020, 26, 3482–3497. [Google Scholar] [CrossRef]
- McQuatters-Gollop, A.; Edwards, M.; Helaouët, P.; Johns, D.G.; Owens, N.J.; Raitsos, D.E.; Schroeder, D.; Skinner, J.; Stern, R.F. The Continuous Plankton Recorder Survey: How Can Long-Term Phytoplankton Datasets Contribute to the Assessment of Good Environmental Status? Estuar. Coast. Shelf Sci. 2015, 162, 88–97. [Google Scholar] [CrossRef]
- Morabito, G.; Mazzocchi, M.G.; Salmaso, N.; Zingone, A.; Bergami, C.; Flaim, G.; Accoroni, S.; Basset, A.; Bastianini, M.; Belmonte, G. Plankton Dynamics across the Freshwater, Transitional and Marine Research Sites of the LTER-Italy Network. Patterns, Fluctuations, Drivers. Sci. Total Environ. 2018, 627, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Vassallo, P.; Bellardini, D.; Castellano, M.; Dapueto, G.; Povero, P. Structure and Functionality of the Mesozooplankton Community in a Coastal Marine Environment: Portofino Marine Protected Area (Liguria). Diversity 2021, 14, 19. [Google Scholar] [CrossRef]
- Humphries, G.; Magness, D.R.; Huettmann, F. (Eds.) Machine Learning for Ecology and Sustainable Natural Resource Management; Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-96976-3. [Google Scholar]
- Steiner, M.; Huettmann, F.; Bryans, N.; Barker, B. With Super SDMs (Machine Learning, Open Access Big Data, and The Cloud) towards a More Holistic and Inclusive Inference: Insights from Progressing the Marginalized Case of the World’s Squirrel Hotspots and Coldspots. Sci. Rep. 2024, 14, 5204. [Google Scholar] [CrossRef]
- Fabiano, M.; Medica, D.; Ossola, C.; Zunini Sertorio, T. Materiale Particellato e Zooplancton in Acque Superficiali. Chiavari: Marzo 1985–Marzo 1986. Ibid 1988, 25, 14. [Google Scholar]
- Culina, A.; Baglioni, M.; Crowther, T.W.; Visser, M.E.; Woutersen-Windhouwer, S.; Manghi, P. Navigating the Unfolding Open Data Landscape in Ecology and Evolution. Nat. Ecol. Evol. 2018, 2, 420–426. [Google Scholar] [CrossRef]
- Aliani, S.; Griffa, A.; Molcard, A. Floating Debris in the Ligurian Sea, North-Western Mediterranean. Mar. Pollut. Bull. 2003, 46, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Astraldi, M.; Gasparini, G.P. The Seasonal Characteristics of the Circulation in the North Mediterranean Basin and Their Relationship with the Atmospheric-climatic Conditions. J. Geophys. Res. 1992, 97, 9531–9540. [Google Scholar] [CrossRef]
- Doglioli, A.M.; Magaldi, M.G.; Vezzulli, L.; Tucci, S. Development of a Numerical Model to Study the Dispersion of Wastes Coming from a Marine Fish Farm in the Ligurian Sea (Western Mediterranean). Aquaculture 2004, 231, 215–235. [Google Scholar] [CrossRef]
- Doglioli, A.M.; Griffa, A.; Magaldi, M.G. Numerical Study of a Coastal Current on a Steep Slope in Presence of a Cape: The Case of the Promontorio Di Portofino. J. Geophys. Res. 2004, 109, 2004JC002422. [Google Scholar] [CrossRef]
- Vietti, R.C.; Albertelli, G.; Aliani, S.; Bava, S.; Bavestrello, G.; Cecchi, L.B.; Bianchi, C.N.; Bozzo, E.; Capello, M.; Castellano, M.; et al. The Ligurian Sea: Present Status, Problems and Perspectives. Chem. Ecol. 2010, 26, 319–340. [Google Scholar] [CrossRef]
- Cattaneo-Vietti, R.; Cappanera, V.; Castellano, M.; Povero, P. Yield and Catch Changes in a M Editerranean Small Tuna Trap: A Warming Change Effect? Mar. Ecol. 2015, 36, 155–166. [Google Scholar] [CrossRef]
- Oprandi, A.; Mucerino, L.; De Leo, F.; Bianchi, C.N.; Morri, C.; Azzola, A.; Benelli, F.; Besio, G.; Ferrari, M.; Montefalcone, M. Effects of a Severe Storm on Seagrass Meadows. Sci. Total Environ. 2020, 748, 141373. [Google Scholar] [CrossRef]
- Grillo, M.; Huettmann, F.; Guglielmo, L.; Schiaparelli, S. Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data. Diversity 2022, 14, 355. [Google Scholar] [CrossRef]
- Team: QGIS Geographic Information System; Open Source Geospat. Found Proj. 2020. Available online: https://qgis.org/en/site/ (accessed on 1 February 2024).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2010. [Google Scholar]
- Schlitzer, R. Ocean Data View 4.0. Oceanography 1998, 11, 19–21. [Google Scholar]
- Hardy, S.M.; Lindgren, M.; Konakanchi, H.; Huettmann, F. Predicting the distribution and ecological niche of unexploited snow crab (Chionoecetes opilio) populations in Alaskan waters: A first open-access ensemble model. Integr. Comp. Biol. 2011, 51, 608–622. [Google Scholar] [CrossRef][Green Version]
- Meißner, K.; Fiorentino, D.; Schnurr, S.; Arbizu, P.M.; Huettmann, F.; Holst, S.; Brix, S.; Svavarsson, J. Distribution of Benthic Marine Invertebrates at Northern latitudes―An Evaluation Applying Multi-Algorithm Species Distribution Models. J. Sea Res. 2014, 85, 241–254. [Google Scholar] [CrossRef]
- Huettmann, F.; Schmid, M. 9.1. Climate Change and Predictions of Pelagic Biodiversity Components. Biogeogr. Atlas South. Ocean. Sci. Comm. Antarct. Res. Camb 2014, 16, 470–475. [Google Scholar]
- Guisan, A.; Thuiller, W.; Zimmermann, N.E. Habitat Suitability and Distribution Models: With Applications in R; Ecology, Biodiversity and Conservation; Cambridge University Press: Cambridge, UK, 2017; ISBN 978-0-521-76513-8. [Google Scholar]
- Kim, S.; Kim, H. A New Metric of Absolute Percentage Error for Intermittent Demand Forecasts. Int. J. Forecast. 2016, 32, 669–679. [Google Scholar] [CrossRef]
- Armstrong, J.S. Long-Range Forecasting: From Crystal Ball to Computer; Wiley-Interscience: Hoboken, NJ, USA, 1985. [Google Scholar]
- Chicco, D.; Warrens, M.J.; Jurman, G. The Coefficient of Determination R-Squared Is More Informative than SMAPE, MAE, MAPE, MSE and RMSE in Regression Analysis Evaluation. PeerJ Comput. Sci. 2021, 7, e623. [Google Scholar] [CrossRef]
- Houghton, J.T.; Ding, Y.; Griggs, D.J.; Noguer, M.; van der Linden, P.J.; Dai, X.; Maskell, K.; Johnson, C.A. Climate Change 2001: The Scientific Basis; Cambridge University Press: Cambridge, UK, 2001; Volume 881. [Google Scholar]
- Edwards, M. Sea Life (Pelagic and Planktonic Ecosystems) as an Indicator of Climate and Global Change. In Climate Change; Elsevier: Amsterdam, The Netherlands, 2009; pp. 233–251. ISBN 978-0-444-53301-2. [Google Scholar]
- Carli, A.; Feletti, M.; Pane, L. Zooplankton Biomass and Copepod Abundance of Terra Nova Bay, Ross Sea Antarctic Campaign 1994/1995. Terra Antarct. Rep. B 2002, 1, 51–55. [Google Scholar]
- Barton, A.D.; Pershing, A.J.; Litchman, E.; Record, N.R.; Edwards, K.F.; Finkel, Z.V.; Kiørboe, T.; Ward, B.A. The Biogeography of Marine Plankton Traits. Ecol. Lett. 2013, 16, 522–534. [Google Scholar] [CrossRef]
- Turner, J.T. The Importance of Small Planktonic Copepods and Their Roles in Pelagic Marine Food Webs. Zool. Stud 2004, 43, 255–266. [Google Scholar]
- Azzurro, E.; Soto, S.; Garofalo, G.; Maynou, F. Fistularia Commersonii in the Mediterranean Sea: Invasion History and Distribution Modeling Based on Presence-Only Records. Biol. Invasions 2013, 15, 977–990. [Google Scholar] [CrossRef]
- Giannoulaki, M.; Markoglou, E.; Valavanis, V.D.; Alexiadou, P.; Cucknell, A.; Frantzis, A. Linking Small Pelagic Fish and Cetacean Distribution to Model Suitable Habitat for Coastal Dolphin Species, Delphinus Delphis and Tursiops Truncatus, in the Greek Seas (Eastern Mediterranean). Aquat. Conserv. 2017, 27, 436–451. [Google Scholar] [CrossRef]
- Azzolin, M.; Arcangeli, A.; Cipriano, G.; Crosti, R.; Maglietta, R.; Pietroluongo, G.; Saintingan, S.; Zampollo, A.; Fanizza, C.; Carlucci, R. Spatial Distribution Modelling of Striped Dolphin (Stenella coeruleoalba) at Different Geographical Scales within the EU Adriatic and Ionian Sea Region, Central-eastern Mediterranean Sea. Aquat. Conserv. 2020, 30, 1194–1207. [Google Scholar] [CrossRef]
- Ranù, M.; Vanacore, A.; Mandich, A.; Alessi, J. Bottlenose Dolphins and Seabirds Distribution Analysis for the Identification of a Marine Biodiversity Hotspot in Agrigento Waters. J. Mar. Sci. Eng. 2022, 10, 345. [Google Scholar] [CrossRef]
- Licandro, P.; Ibanez, F. Changes of Zooplankton Communities in the Gulf of Tigullio (Ligurian Sea, Western Mediterranean) from 1985 to 1995. Influence of Hydroclimatic Factors. J. Plankton Res. 2000, 22, 2225–2253. [Google Scholar] [CrossRef]
- Pane, L.; Boccardo, S.; Bonfiglioli, F.; Mariottini, G.L.; Priano, F.; Conio, O. Polycyclic Aromatic Hydrocarbons in Water, Seston and Copepods in a Harbour Area in the Western Mediterranean (Ligurian Sea). Mar. Ecol. 2005, 26, 89–99. [Google Scholar] [CrossRef]
- Zagami, G.; Granata, A.; Brugnano, C.; Minutoli, R.; Bonanzinga, V.; Guglielmo, L. Spring Copepod Vertical Zonation Pattern and Diel Migration in the Open Ligurian Sea (North-Western Mediterranean). Prog. Oceanogr. 2020, 183, 102297. [Google Scholar] [CrossRef]
- Mazzocchi, M.G.; Dubroca, L.; García-Comas, C.; Capua, I.D.; Ribera d’Alcalà, M. Stability and Resilience in Coastal Copepod Assemblages: The Case of the Mediterranean Long-Term Ecological Research at Station MC (LTER-MC). Prog. Oceanogr. 2012, 97–100, 135–151. [Google Scholar] [CrossRef]
- Borme, D.; Tirelli, V.; Brandt, S.; Fonda Umani, S.; Arneri, E. Diet of Engraulis Encrasicolus in the Northern Adriatic Sea (Mediterranean): Ontogenetic Changes and Feeding Selectivity. Mar. Ecol. Prog. Ser. 2009, 392, 193–209. [Google Scholar] [CrossRef]
- Lillia, M.; Hafferssas, A.; Hamri, F.; Alioua, Z.; Yacine, K.G.E. Importance of Copepods in the Diet of Sardina Pilchardus and Sardinella Aurita: Preliminary Investigation in Bou Ismail Bay (Algerian Basin—SW Mediterranean Sea). Thalassas 2024, 1–17. [Google Scholar] [CrossRef]
- Granata, A.; Bergamasco, A.; Zagami, G.; Guglielmo, R.; Bonanzinga, V.; Minutoli, R.; Geraci, A.; Pagano, L.; Swadling, K.; Battaglia, P.; et al. Daily Vertical Distribution and Diet of Cyclothone braueri (Gonostomatidae) in the Polcevera Submarine Canyon (Ligurian Sea, North-Western Mediterranean). Deep Sea Res. Part I Oceanogr. Res. Pap. 2023, 199, 104113. [Google Scholar] [CrossRef]
- Coll, M.; Santojanni, A.; Palomera, I.; Tudela, S.; Arneri, E. An Ecological Model of the Northern and Central Adriatic Sea: Analysis of Ecosystem Structure and Fishing Impacts. J. Mar. Syst. 2007, 67, 119–154. [Google Scholar] [CrossRef]
- La Tonnarella e La Pesca Tradizionale a Camogli—Riccardo Vietti Cattaneo, Simone Bava—Libro Le Mani-Microart’S 2009, Le Mani. Natura, Ambiente, Flora e Fauna | Libraccio. Available online: https://www.libraccio.it/libro/9788880124948/riccardo-vietti-cattaneo-simone-bava/tonnarella-e-pesca-tradizionale-a-camogli.html (accessed on 29 February 2024).
- Relini, M.; Palandri, G.; ORSI, L. Monitoraggio Di Grandi Pesci Pelagici Dalla Tonnarella Di Camogli: Il Caso Della Palamita, Sarda Sarda (Bloch). Biol. Mar. Mediterr 2007, 14, 116–117. [Google Scholar]
- Plandri, G.; Lanteri, L.; Garibaldi, F.; Relini, L.O. Biological Parameters of Bullet Tuna in the Ligurian Sea. Collect. Vol. Sci. Pap. ICCAT 2009, 64, 2272–2279. [Google Scholar]
- Escudier, R.; Clementi, E.; Omar, M.; Cipollone, A.; Pistoia, J.; Aydogdu, A.; Drudi, M.; Grandi, A.; Lyubartsev, V.; Lecci, R.; et al. Mediterranean Sea Physical Reanalysis (CMEMS MED-Currents) (Version 1) [Data Set]; Copernicus Monitoring Environment Marine Service (CMEMS): Ramonville-Saint-Agne, France, 2020. [Google Scholar]
- Escudier, R.; Clementi, E.; Cipollone, A.; Pistoia, J.; Drudi, M.; Grandi, A.; Lyubartsev, V.; Lecci, R.; Aydogdu, A.; Delrosso, D.; et al. A High Resolution Reanalysis for the Mediterranean Sea. Front. Earth Sci. 2021, 9, 1060. [Google Scholar] [CrossRef]
- Nigam, T.; Escudier, R.; Pistoia, J.; Aydogdu, A.; Omar, M.; Clementi, E.; Cipollone, A.; Drudi, M.; Grandi, A.; Mariani, A.; et al. Mediterranean Sea Physical Reanalysis INTERIM (CMEMS MED-Currents, E3R1i System) (Version 1) [Data Set]; Copernicus Monitoring Environment Marine Service (CMEMS): Ramonville-Saint-Agne, France, 2021. [Google Scholar]
- Chau, T.T.T.; Gehlen, M.; Chevallier, F. A seamless ensemble-based reconstruction of surface ocean pCO2 and air–sea CO2 fluxes over the global coastal and open oceans. Biogeosciences 2022, 19, 1087–1109. [Google Scholar] [CrossRef]
| Family | Genus | Worms Aphia ID | Trophic Guilds |
|---|---|---|---|
| Acartiidae | Acartia spp. Dana, 1846 | 104108 | Filter feeder |
| Oithonidae | Oithona spp. Baird, 1843 | 106485 | Ambush feeder |
| Centropagidae | Centropages spp. Krøyer, 1849 | 104081 | Suspension feeder |
| Temoridae | Temora spp. Baird, 1850 | 104241 | Filter feeder/Ambush feeder |
| Corycaeidae | Corycaceus spp. Dana, 1845 | 128634 | Predator |
| sMAPE Index | |||||
|---|---|---|---|---|---|
| Season | Acartia spp. | Centropages spp. | Oithona spp. | Temora spp. | Coryceus spp. |
| Autumn | 1.3698 | 1.0204 | 0.7357 | 1.0047 | 0.5111 |
| Winter | 1.0429 | 0.7985 | 0.4567 | 1.3779 | 0.8969 |
| Spring | 0.4105 | 0.9100 | 0.5558 | 1.8411 | 1.8448 |
| Summer | 0.9639 | 1.4982 | 1.0322 | 1.9648 | 0.8390 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzzi, A.; Schiaparelli, S.; Balan, M.; Grillo, M. A Beacon in the Dark: Grey Literature Data Mining and Machine Learning Enlightening Historical Plankton Seasonality Dynamics in the Ligurian Sea. Diversity 2024, 16, 189. https://doi.org/10.3390/d16030189
Guzzi A, Schiaparelli S, Balan M, Grillo M. A Beacon in the Dark: Grey Literature Data Mining and Machine Learning Enlightening Historical Plankton Seasonality Dynamics in the Ligurian Sea. Diversity. 2024; 16(3):189. https://doi.org/10.3390/d16030189
Chicago/Turabian StyleGuzzi, Alice, Stefano Schiaparelli, Maria Balan, and Marco Grillo. 2024. "A Beacon in the Dark: Grey Literature Data Mining and Machine Learning Enlightening Historical Plankton Seasonality Dynamics in the Ligurian Sea" Diversity 16, no. 3: 189. https://doi.org/10.3390/d16030189
APA StyleGuzzi, A., Schiaparelli, S., Balan, M., & Grillo, M. (2024). A Beacon in the Dark: Grey Literature Data Mining and Machine Learning Enlightening Historical Plankton Seasonality Dynamics in the Ligurian Sea. Diversity, 16(3), 189. https://doi.org/10.3390/d16030189









