A Comprehensive Review of Disease-Causing Agents in Freshwater Turtles: Implications for Conservation and Public Health
Abstract
1. Introduction
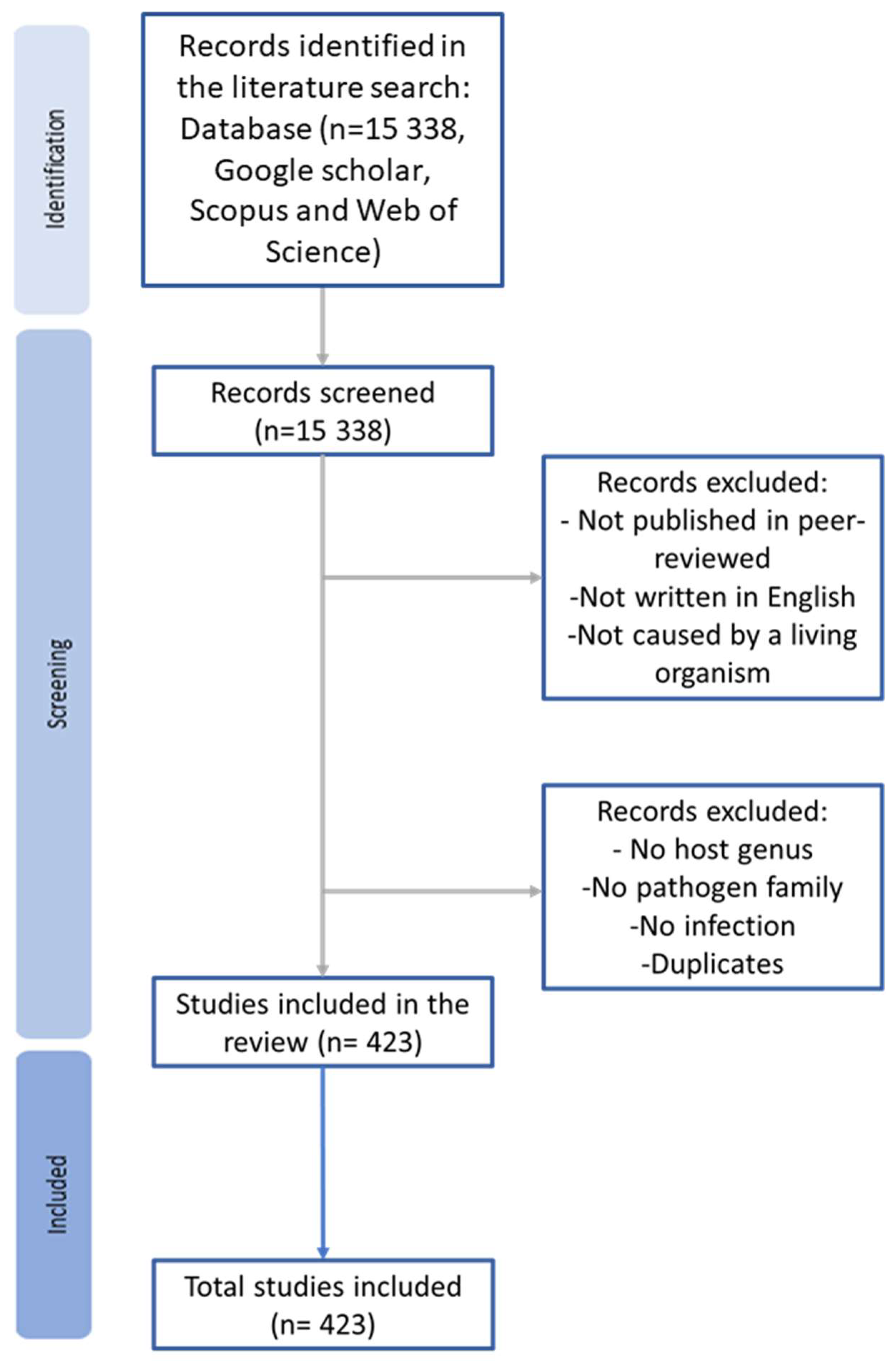
2. Methods
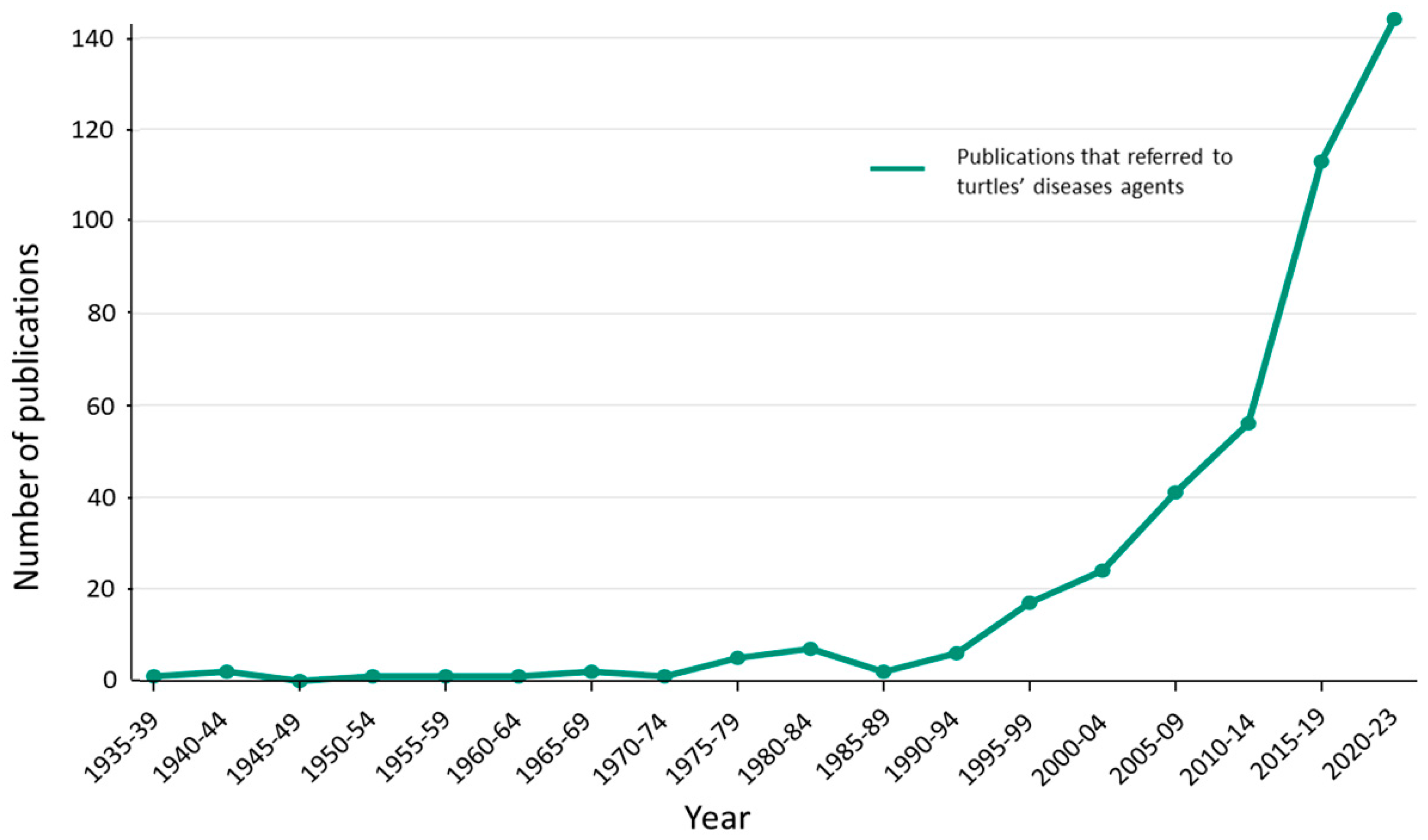
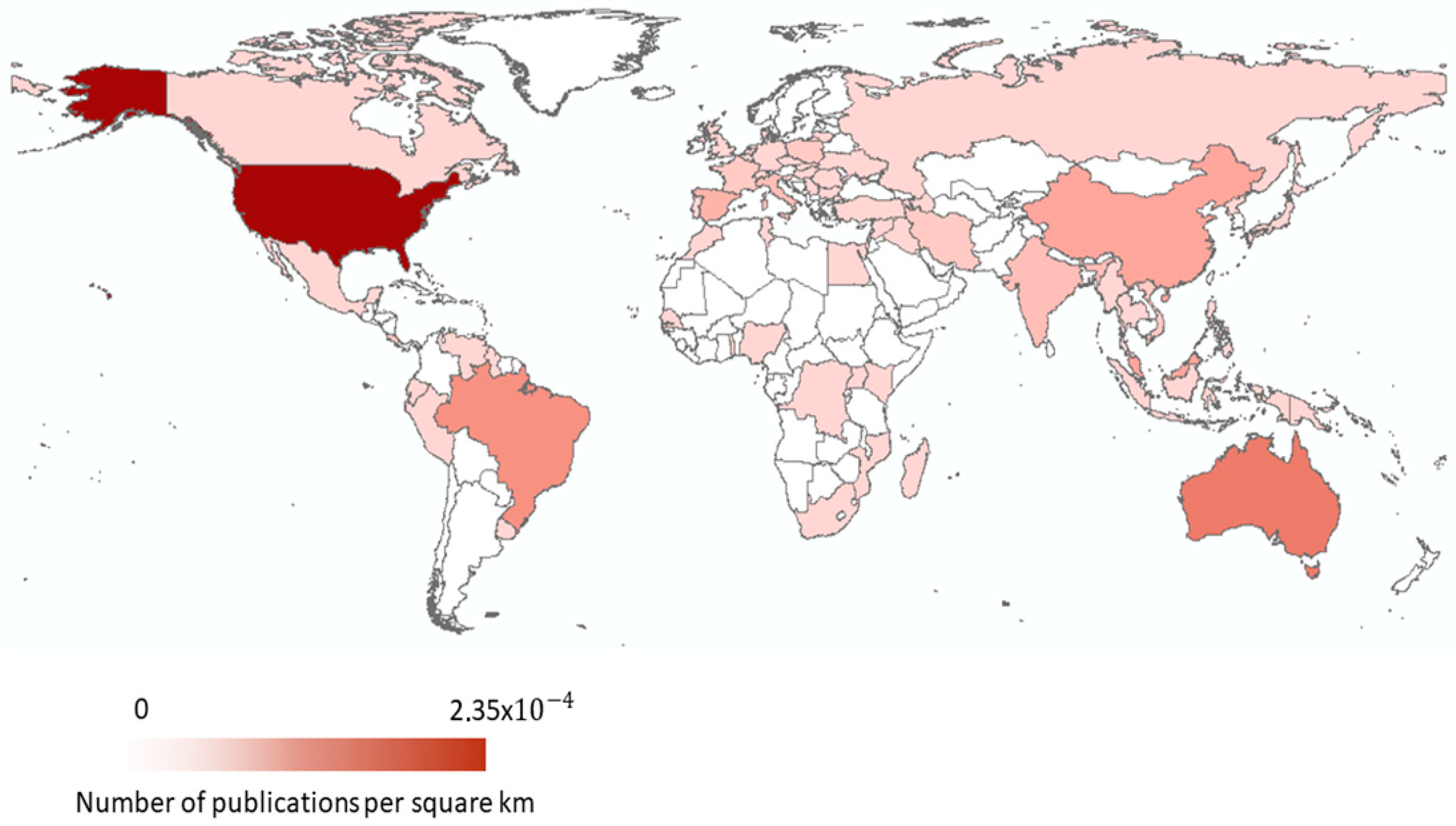
3. Timeline and Global Distribution
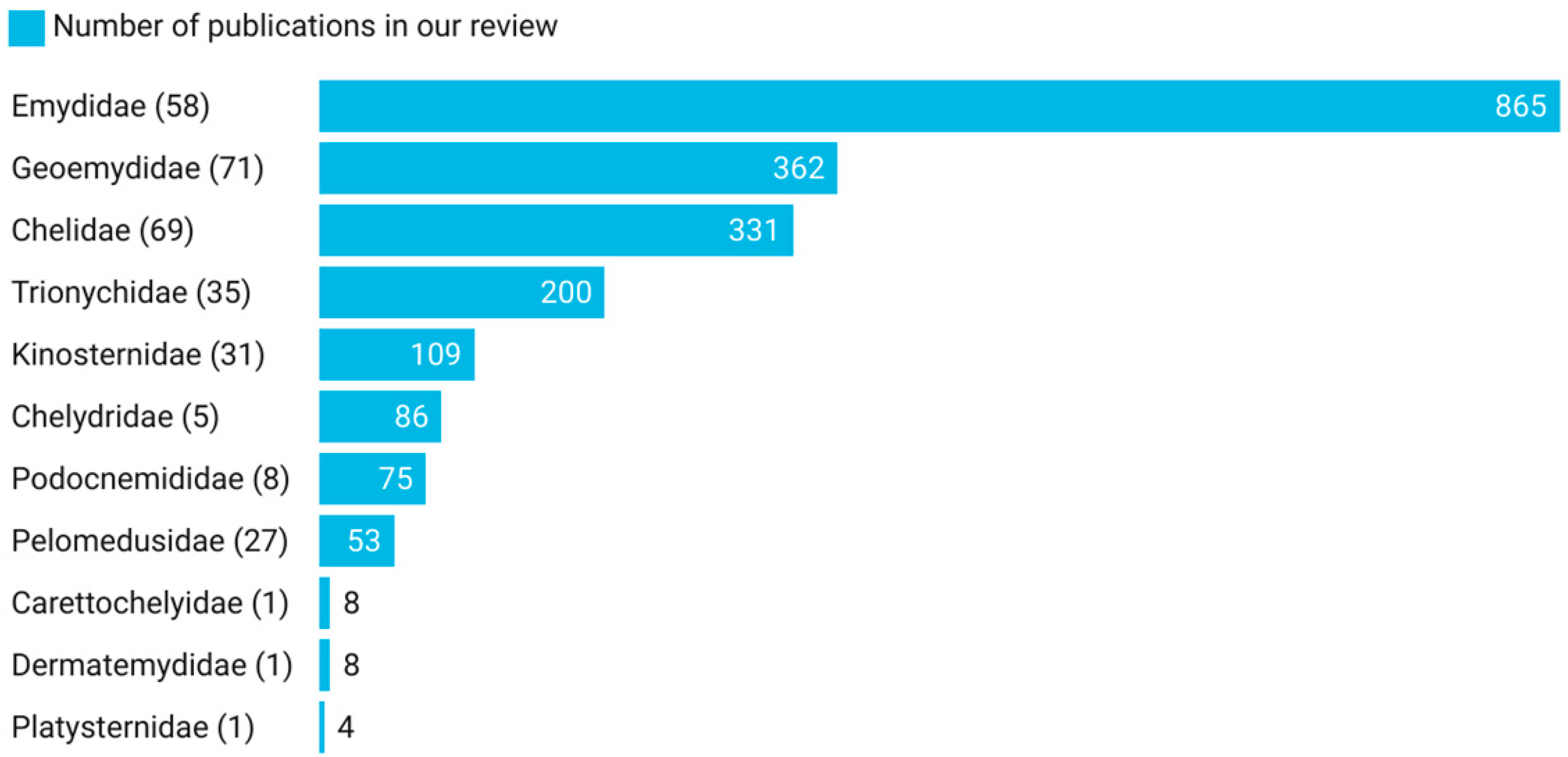
4. Hosts
5. Disease Agents
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bour, R. Global Diversity of Turtles (Chelonii; Reptilia) in Freshwater. Hydrobiologia 2008, 595, 593–598. [Google Scholar] [CrossRef]
- Uetz, P.; Freed, P.; Reyes, F.; Aguilar, R.; Kudera, J.; Hošek, J. (Eds.) The Reptile Database. 2023. Available online: http://www.reptile-database.org/ (accessed on 11 January 2024).
- Colon, V.A.; Lugsomya, K.; Lam, H.K.; Wahl, L.C.; Parkes, R.S.V.; Cormack, C.A.; Horlbog, J.A.; Stevens, M.; Stephan, R.; Magouras, I. Serotype Diversity and Antimicrobial Resistance Profile of Salmonella enterica Isolates from Freshwater Turtles Sold for Human Consumption in Wet Markets in Hong Kong. Front. Vet. Sci. 2022, 9, 912693. [Google Scholar] [CrossRef]
- Hoang, H.; McCormack, T.E.M.; Lo, H.; Nguyen, M.; Tapley, B. Hunting and Trade of Big-Headed Turtles (Platysternon megacephalum Gray 1831) in Two Protected Areas in Northern Vietnam. Herpetol. Notes 2021, 14, 1077–1085. [Google Scholar]
- Pezzuti, J.C.B.; Lima, J.P.; Da Silva, D.F.; Begossi, A. Uses and Taboos of Turtles and Tortoises Along Rio Negro, Amazon Basin. J. Ethnobiol. 2010, 30, 153–168. [Google Scholar] [CrossRef]
- EFSA. Public Health Risks Involved in the Human Consumption of Reptile Meat. EFSA J. 2007, 578, 1–55. [Google Scholar] [CrossRef]
- Magnino, S.; Colin, P.; Dei-Cas, E.; Madsen, M.; McLauchlin, J.; Nöckler, K.; Prieto Maradona, M.; Tsigarida, E.; Vanopdenbosch, E.; Van Peteghem, C. Biological Risks Associated with Consumption of Reptile Products. Int. J. Food Microbiol. 2009, 134, 163–175. [Google Scholar] [CrossRef]
- Bush, E.R.; Baker, S.E.; Macdonald, D.W. Global Trade in Exotic Pets 2006-2012: Exotic Pet Trade. Conserv. Biol. 2014, 28, 663–676. [Google Scholar] [CrossRef]
- Banha, F.; Diniz, A.; Anastácio, P.M. Patterns and Drivers of Aquarium Pet Discharge in the Wild. Ecol. Indic. 2019, 106, 105513. [Google Scholar] [CrossRef]
- Young, H.S.; Parker, I.M.; Gilbert, G.S.; Sofia Guerra, A.; Nunn, C.L. Introduced Species, Disease Ecology, and Biodiversity–Disease Relationships. Trends Ecol. Evol. 2017, 32, 41–54. [Google Scholar] [CrossRef]
- Bosch, S.; Tauxe, R.V.; Behravesh, C.B. Turtle-Associated Salmonellosis, United States, 2006–2014. Emerg. Infect. Dis. 2015, 22, 1149. [Google Scholar] [CrossRef]
- Alves, R.R.; Vieira, W.L.; Santana, G.G. Reptiles Used in Traditional Folk Medicine: Conservation Implications. Biodivers. Conserv. 2008, 17, 2037–2049. [Google Scholar] [CrossRef]
- Chen, T.-H.; Chang, H.-C.; Lue, K.-Y. Unregulated Trade in Turtle Shells for Chinese Traditional Medicine in East and Southeast Asia: The Case of Taiwan. Chelonian Conserv. Biol. 2009, 8, 11–18. [Google Scholar] [CrossRef]
- Alves, R.R.; Rosa, I.M. Biodiversity, Traditional Medicine and Public Health: Where Do They Meet? J. Ethnobiol. Ethnomedicine 2007, 3, 14. [Google Scholar] [CrossRef]
- Friant, S.; Bonwitt, J.; Ayambem, W.A.; Ifebueme, N.M.; Alobi, A.O.; Otukpa, O.M.; Bennett, A.J.; Shea, C.; Rothman, J.M.; Goldberg, T.L.; et al. Zootherapy as a Potential Pathway for Zoonotic Spillover: A Mixed-Methods Study of the Use of Animal Products in Medicinal and Cultural Practices in Nigeria. One Health Outlook 2022, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Finlaison, D.S.; Frost, M.J.; Gestier, S.; Gu, X.; Hall, J.; Jenkins, C.; Parrish, K.; Read, A.J.; Srivastava, M.; et al. Identification of a Novel Nidovirus as a Potential Cause of Large Scale Mortalities in the Endangered Bellinger River Snapping Turtle (Myuchelys georgesi). PLoS ONE 2018, 13, e0205209. [Google Scholar] [CrossRef]
- Filho, W.; Ternova, L.; Parasnis, S.A.; Kovaleva, M.; Nagy, G.J. Climate Change and Zoonoses: A Review of Concepts, Definitions, and Bibliometrics. Int. J. Environ. Res. Public. Health 2022, 19, 893. [Google Scholar] [CrossRef] [PubMed]
- Barroso, P.; Acevedo, P.; Vicente, J. The Importance of Long-term Studies on Wildlife Diseases and Their Interfaces with Humans and Domestic Animals: A Review. Transbound. Emerg. Dis. 2021, 68, 1895–1909. [Google Scholar] [CrossRef]
- Hossain, S.; Wimalasena, S.; Zoysa, M.; Heo, G.-J. Prevalence of Citrobacter Spp. From Pet Turtles and Their Environment. J. Exot. Pet Med. 2017, 26, 7–12. [Google Scholar] [CrossRef]
- Hossain, S.; Heo, G.-J. Pet-Turtles: A Potential Source of Human Pathogenic Bacteria. Arch. Microbiol. 2021, 203, 3785–3792. [Google Scholar] [CrossRef]
- Marin, C.; Martín-Maldonado, B.; Cerdà-Cuéllar, M.; Sevilla-Navarro, S.; Lorenzo-Rebenaque, L.; Montoro-Dasi, L.; Manzanares, A.; Ayats, T.; Mencía-Gutiérrez, A.; Jordá, J.; et al. Antimicrobial Resistant Salmonella in Chelonians: Assessing Its Potential Risk in Zoological Institutions in Spain. Vet. Sci. 2022, 9, 264. [Google Scholar] [CrossRef]
- Mendoza-Roldan, J.A.; Modry, D.; Otranto, D. Zoonotic Parasites of Reptiles: A Crawling Threat. Trends Parasitol. 2020, 36, 677–687. [Google Scholar] [CrossRef]
- Tompkins, D.M.; Carver, S.; Jones, M.E.; Krkošek, M.; Skerratt, L.F. Emerging Infectious Diseases of Wildlife: A Critical Perspective. Trends Parasitol. 2015, 31, 149–159. [Google Scholar] [CrossRef]
- Wiethoelter, A.K.; Beltrán-Alcrudo, D.; Kock, R.; Mor, S.M. Global Trends in Infectious Diseases at the Wildlife–Livestock Interface. Proc. Natl. Acad. Sci. USA 2015, 112, 9662–9667. [Google Scholar] [CrossRef] [PubMed]
- Guedes, J.J.M.; Moura, M.R.; Alexandre, F.; Diniz-Filho, J. Species out of Sight: Elucidating the Determinants of Research Effort in Global Reptiles. Ecography 2023, 2023, e06491. [Google Scholar] [CrossRef]
- Iverson, J.B. A Review of Chelonian Type Specimens (Order Testudines). Megataxa 2022, 7, 1–85. [Google Scholar] [CrossRef]
- Francis, R.A. (Ed.) A Handbook of Global Freshwater Invasive Species; Earthscan: London, UK; New York, NY, USA, 2012; ISBN 978-1-84971-228-6. [Google Scholar]
- Ramsay, N.F.; Ng, P.K.A.; O’Riordan, M.; Chou, L.M. The Red-Eared Slider (Trachemys scripta elegans) in Asia: A Review. In Biological Invaders in Inland Waters: Profiles, Distribution, and Threats; Gherardi, F., Ed.; Springer: Dordrecht, The Netherlands, 2007; pp. 161–174. [Google Scholar]
- Kraus, F. Alien Reptiles and Amphibians: A Scientific Compendium and Analysis; Invading Nature: Springer Series in Invasion Ecology; Springer: Dordrecht, The Netherlands, 2009; ISBN 978-1-4020-8945-9. [Google Scholar]
- Meyer, L.; Du Preez, L.; Bonneau, E.; Héritier, L.; Quintana, M.; Valdeón, A.; Sadaoui, A.; Kechemir-Issad, N.; Palacios, C.; Verneau, O. Parasite Host-Switching from the Invasive American Red-Eared Slider, Trachemys scripta elegans, to the Native Mediterranean Pond Turtle, Mauremys leprosa, in Natural Environments. Aquat. Invasions 2015, 10, 79–91. [Google Scholar] [CrossRef]
- Demkowska-Kutrzepa, M.; Studzińska, M.; Roczeń-Karczmarz, M.; Tomczuk, K.; Abbas, Z.; Różański, P. A Review of the Helminths Co-Introduced with Trachemys scripta elegans—A Threat to European Native Turtle Health. Amphib.-Reptil. 2018, 39, 177–189. [Google Scholar] [CrossRef]
- Héritier, L.; Valdeón, A.; Sadaoui, A.; Gendre, T.; Ficheux, S.; Bouamer, S.; Kechemir-Issad, N.; Du Preez, L.; Palacios, C.; Verneau, O. Introduction and Invasion of the Red-Eared Slider and Its Parasites in Freshwater Ecosystems of Southern Europe: Risk Assessment for the European Pond Turtle in Wild Environments. Biodivers. Conserv. 2017, 26, 1817–1843. [Google Scholar] [CrossRef]
- Verneau, O.; Palacios, C.; Platt, T.; Alday, M.; Billard, E.; Allienne, J.-F.; Basso, C.; Du Preez, L.H. Invasive Species Threat: Parasite Phylogenetics Reveals Patterns and Processes of Host-Switching between Non-Native and Native Captive Freshwater Turtles. Parasitology 2011, 138, 1778–1792. [Google Scholar] [CrossRef]
- Beunen, R. European Nature Conservation Legislation and Spatial Planning: For Better or for Worse? J. Environ. Plan. Manag. 2006, 49, 605–619. [Google Scholar] [CrossRef]
- Engelhardt, E.K.; Bowler, D.E.; Hof, C. European Habitats Directive Has Fostered Monitoring but Not Prevented Species Declines. Conserv. Lett. 2023, 16, e12948. [Google Scholar] [CrossRef]
- Velo-Antón, G.; Wink, M.; Schneeweiß, N.; Fritz, U. Native or Not? Tracing the Origin of Wild-Caught and Captive Freshwater Turtles in a Threatened and Widely Distributed Species (Emys orbicularis). Conserv. Genet. 2011, 12, 583–588. [Google Scholar] [CrossRef]
- Destoumieux-Garzón, D.; Mavingui, P.; Boetsch, G.; Boissier, J.; Darriet, F.; Duboz, P.; Fritsch, C.; Giraudoux, P.; Le Roux, F.; Morand, S.; et al. The One Health Concept: 10 Years Old and a Long Road Ahead. Front. Vet. Sci. 2018, 5, 14. [Google Scholar] [CrossRef]
- Köster, P.C.; Martínez-Nevado, E.; González, A.; Abelló-Poveda, M.T.; Fernández-Bellon, H.; De La Riva-Fraga, M.; Marquet, B.; Guéry, J.-P.; Knauf-Witzens, T.; Weigold, A.; et al. Intestinal Protists in Captive Non-Human Primates and Their Handlers in Six European Zoological Gardens. Molecular Evidence of Zoonotic Transmission. Front. Vet. Sci. 2022, 8, 819887. [Google Scholar] [CrossRef]
- Thakur, R.; Sharma, R.; Aulakh, R.S.; Gill, J.P.S.; Singh, B.B. Seroprevalence and Risk Factor Investigation for the Exposure of Toxoplasma Gondii among Veterinary Personnel in Punjab, India. Comp. Immunol. Microbiol. Infect. Dis. 2022, 80, 101739. [Google Scholar] [CrossRef] [PubMed]
- Carstairs, S.J.; Kyle, C.J.; Vilaça, S.T. High Prevalence of Subclinical Frog Virus 3 Infection in Freshwater Turtles of Ontario, Canada. Virology 2020, 543, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Ossiboff, R.J.; Raphael, B.L.; Ammazzalorso, A.D.; Seimon, T.A.; Newton, A.L.; Chang, T.Y.; Zarate, B.; Whitlock, A.L.; McAloose, D. Three Novel Herpesviruses of Endangered Clemmys and Glyptemys Turtles. PLoS ONE 2015, 10, e0122901. [Google Scholar] [CrossRef] [PubMed]
- Deem, S.L.; Karesh, W.B.; Weisman, W. Putting Theory into Practice: Wildlife Health in Conservation. Conserv. Biol. 2008, 15, 1224–1233. [Google Scholar] [CrossRef]
- Bradley, M.J.; Kutz, S.J.; Jenkins, E.; O’Hara, T.M. The Potential Impact of Climate Change on Infectious Diseases of Arctic Fauna. Int. J. Circumpolar Health 2005, 64, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Torchin, M.E.; Lafferty, K.D.; Dobson, A.P.; McKenzie, V.J.; Kuris, A.M. Introduced Species and Their Missing Parasites. Nature 2003, 421, 628–630. [Google Scholar] [CrossRef] [PubMed]
- Rhodin, A.G.J.; Stanford, C.B.; Dijk, P.P.V.; Eisemberg, C.; Luiselli, L.; Mittermeier, R.A.; Hudson, R.; Horne, B.D.; Goode, E.V.; Kuchling, G.; et al. Global Conservation Status of Turtles and Tortoises (Order Testudines). Chelonian Conserv. Biol. 2018, 17, 135. [Google Scholar] [CrossRef]
- Martínez-Ríos, M.; Martín-Torrijos, L.; Diéguez-Uribeondo, J. The Invasive Alien Red-Eared Slider Turtle, Trachemys scripta, as a Carrier of STEF-Disease Pathogens. Fungal Biol. 2022, 126, 113–121. [Google Scholar] [CrossRef]
- Smyth, C.W.; Sarmiento-Ramírez, J.M.; Short, D.P.G.; Diéguez-Uribeondo, J.; O’Donnell, K.; Geiser, D.M. Unraveling the Ecology and Epidemiology of an Emerging Fungal Disease, Sea Turtle Egg Fusariosis (STEF). PLOS Pathog. 2019, 15, e1007682. [Google Scholar] [CrossRef]
- Carranco, A.S.; Gillingham, M.A.F.; Wilhelm, K.; Torres, M.D.L.; Sommer, S.; Romo, D. Transcending Sea Turtles: First Report of Hatching Failure in Eggs of an Amazonian Freshwater Turtle with Symptoms of the Fungal Emerging Disease Fusariosis. Transbound. Emerg. Dis. 2022, 69, e3282–e3288. [Google Scholar] [CrossRef]
- Sterrett, S.C.; Smith, L.L.; Schweitzer, S.H.; Maerz, J.C. An Assssment Of Two Methods For Sampling River Turtle Assemblages. Herpetol. Conserv. Biol. 2010, 5, 490–497. [Google Scholar]
- Tesche, M.R.; Hodges, K.E. Unreliable Population Inferences from Common Trapping Practices for Freshwater Turtles. Glob. Ecol. Conserv. 2015, 3, 802–813. [Google Scholar] [CrossRef]
- Sánchez, C.A.; Becker, D.J.; Teitelbaum, C.S.; Barriga, P.; Brown, L.M.; Majewska, A.A.; Hall, R.J.; Altizer, S. On the Relationship between Body Condition and Parasite Infection in Wildlife: A Review and Meta-analysis. Ecol. Lett. 2018, 21, 1869–1884. [Google Scholar] [CrossRef]
- Foufopoulos, J.; Wobeser, G.A.; McCallum, H. Infectious Disease Ecology and Conservation, 1st ed.; Oxford University Press: Oxford, UK, 2022; ISBN 978-0-19-958350-8. [Google Scholar]
- Carlson, C.J.; Dallas, T.A.; Alexander, L.W.; Phelan, A.L.; Phillips, A.J. What Would It Take to Describe the Global Diversity of Parasites? Proc. R Soc. B Biol. Sci. 2020, 287, 20201841. [Google Scholar] [CrossRef]
- Poulin, R.; Presswell, B.; Bennett, J.; De Angeli Dutra, D.; Salloum, P.M. Biases in Parasite Biodiversity Research: Why Some Helminth Species Attract More Research than Others. Int. J. Parasitol. Parasites Wildl. 2023, 21, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Poulin, R.; Morand, S. The Diversity of Parasites. Q. Rev. Biol. 2000, 75, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Abebe, E.; Decraemer, W.; Ley, P. Global Diversity of Nematodes (Nematoda) in Freshwater. Hydrobiologia 2008, 595, 67–78. [Google Scholar] [CrossRef]
- Jones, J.T.; Haegeman, A.; Danchin, E.G.J.; Gaur, H.S.; Helder, J.; Jones, M.G.K.; Kikuchi, T.; Manzanilla-López, R.; Palomares-Rius, J.E.; Wesemael, W.M.L.; et al. Top 10 Plant-Parasitic Nematodes in Molecular Plant Pathology: Top 10 Plant-Parasitic Nematodes. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.; Viney, M. The Population Genetics of Parasitic Nematodes of Wild Animals. Parasit. Vectors 2018, 11, 590. [Google Scholar] [CrossRef]
- Fitze, P.S.; Tschirren, B.; Richner, H. Life History and Fitness Consequences of Ectoparasites. J. Anim. Ecol. 2004, 73, 216–226. [Google Scholar] [CrossRef]
- Werner, R.E. Parasites in the Diamondback Terrapin, Malaclemys terrapin: A Review. J. Herpetol. Med. Surg. 2003, 13, 5–9. [Google Scholar] [CrossRef]
- Mendoza-Roldan, J.A.; Ribeiro, S.R.; Castilho-Onofrio, V.; Marcili, A.; Simonato, B.B.; Latrofa, M.S.; Benelli, G.; Otranto, D.; Barros-Battesti, D.M. Molecular Detection of Vector-Borne Agents in Ectoparasites and Reptiles from Brazil. Ticks Tick-Borne Dis. 2021, 12, 101585. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Roldan, J.A.; Colella, V.; Lia, R.P.; Nguyen, V.L.; Barros-Battesti, D.M.; Iatta, R.; Dantas-Torres, F.; Otranto, D. Borrelia burgdorferi (Sensu Lato) in Ectoparasites and Reptiles in Southern Italy. Parasit. Vectors 2019, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Siddall, M.E.; Desser, S.S. Transmission of Haemogregarina balli From Painted Turtles to Snapping Turtles Through the Leech Placobdella Ornata. J. Parasitol. 2001, 87, 1217–1218. [Google Scholar] [CrossRef] [PubMed]
- Verneau, O.; Melliti, S.; Kimdil, L.; El Mouden, E.H.; Achouri, M.S.; Rouag, R. Molecular Phylogenies of Leeches and Haemoparasites Infecting Freshwater Turtles in Aquatic Ecosystems of Northern Africa Suggest Phylogenetic Congruence between Placobdella costata Sensu Lato and Haemogregarina stepanowi Sensu Lato. Microorganisms 2023, 11, 1584. [Google Scholar] [CrossRef]
- Chlebicz, A.; Śliżewska, K. Campylobacteriosis, Salmonellosis, Yersiniosis, and Listeriosis as Zoonotic Foodborne Diseases: A Review. Int. J. Environ. Res. Public. Health 2018, 15, 863. [Google Scholar] [CrossRef]
- Back, D.-S.; Shin, G.-W.; Wendt, M.; Heo, G.-J. Prevalence of Salmonella Spp. in Pet Turtles and Their Environment. Lab. Anim. Res. 2016, 32, 166. [Google Scholar] [CrossRef]
- Beale, D.J.; Bissett, A.; Nilsson, S.; Bose, U.; Nelis, J.L.D.; Nahar, A.; Smith, M.; Gonzalez-Astudillo, V.; Braun, C.; Baddiley, B.; et al. Perturbation of the Gut Microbiome in Wild-Caught Freshwater Turtles (Emydura macquarii macquarii) Exposed to Elevated PFAS Levels. Sci. Total Environ. 2022, 838, 156324. [Google Scholar] [CrossRef] [PubMed]
- Parks, M.; Kedy, C.; Skalla, C. Consistent Patterns in 16S and 18S Microbial Diversity from the Shells of the Common and Widespread Red-Eared Slider Turtle (Trachemys scripta). PLoS ONE 2020, 15, e0244489. [Google Scholar] [CrossRef] [PubMed]
- Zancolli, G.; Mahsberg, D.; Sickel, W.; Keller, A. Reptiles as Reservoirs of Bacterial Infections: Real Threat or Methodological Bias? Microb. Ecol. 2015, 70, 579–584. [Google Scholar] [CrossRef]
- Harding, E.F.; Russo, A.G.; Yan, G.J.H.; Mercer, L.K.; White, P.A. Revealing the Uncharacterised Diversity of Amphibian and Reptile Viruses. ISME Commun. 2022, 2, 95. [Google Scholar] [CrossRef]
- Correa, J.K.C.; Picelli, A.M.; Da Silva, M.R.L.; Valadão, R.M.; Hernández-Ruz, E.J.; Viana, L.A. Phylogenetic Analysis of Chelonian Hemogregarines Reveals Shared Species among the Amazonian Freshwater Turtle Podocnemis Spp. and Provides a Description of Two New Species of Haemogregarina. Parasitol. Res. 2022, 121, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Laghzaoui, E.-M.; Perera, A.; Harris, D.J.; El Mouden, E.H. Characterization and Identification of Haemogregarine Hemoparasites (Apicomplexa: Adeleina: Hepatozoidae) in Natural Populations of Mauremys leprosa leprosa and M. leprosa saharica from Morocco. Syst. Parasitol. 2021, 98, 547–557. [Google Scholar] [CrossRef]
- Johnson, J.G.; Watson, M.K. Diseases of the Reptile Renal System. Veterinary Clin. N. Am. Exot. Anim. Pract. 2020, 23, 115–129. [Google Scholar] [CrossRef]
- Reavill, D.R.; Schmidt, R.E. Urinary Tract Diseases of Reptiles. J. Exot. Pet Med. 2010, 19, 280–289. [Google Scholar] [CrossRef]
- Dezzutto, D.; Barbero, R.; Canale, G.; Acutis, P.; Biolatti, C.; Dogliero, A.; Mitzy, M.; Francone, P.; Colzani, A.; Bergagna, S.; et al. Detection of Leptospira Spp. in Water Turtle (Trachemys scripta) Living in Ponds of Urban Parks. Vet. Sci. 2017, 4, 51. [Google Scholar] [CrossRef]
- Oliveira, J.P.; Kawanami, A.E.; Silva, A.S.L.; Chung, D.G.; Werther, K. Detection of Leptospira Spp. in Wild Phrynops geoffroanus (Geoffroy’s Side-Necked Turtle) in Urban Environment. Acta Trop. 2016, 164, 165–168. [Google Scholar] [CrossRef] [PubMed]


| Number of Publications | Publication Percentage | ||
|---|---|---|---|
| Age | Adult | 56 | 13.2% |
| Juvenile | 12 | 2.8% | |
| Adult and Juvenile | 41 | 9.7% | |
| Egg | 7 | 1.7% | |
| Unspecified | 303 | 71.6% | |
| Status | Alive | 275 | 65.0% |
| Dead | 35 | 8.3% | |
| Both | 31 | 7.3% | |
| Unspecified | 82 | 19.4% | |
| Origin | Wild | 237 | 56.0% |
| Captive | 83 | 19.6% | |
| Both | 33 | 7.8% | |
| Unspecified | 70 | 16.6% | |
| Native/Non-native | Native | 343 | 81.1% |
| Non-native | 38 | 8.9% | |
| Both | 13 | 3.1% | |
| Unspecified | 29 | 6.9% | |
| Symptoms | Yes | 75 | 17.8% |
| No | 70 | 16.5% | |
| Unspecified | 278 | 65.7% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rato, J.; Xavier, R.; Harris, D.J.; Banha, F.; Anastácio, P. A Comprehensive Review of Disease-Causing Agents in Freshwater Turtles: Implications for Conservation and Public Health. Diversity 2024, 16, 171. https://doi.org/10.3390/d16030171
Rato J, Xavier R, Harris DJ, Banha F, Anastácio P. A Comprehensive Review of Disease-Causing Agents in Freshwater Turtles: Implications for Conservation and Public Health. Diversity. 2024; 16(3):171. https://doi.org/10.3390/d16030171
Chicago/Turabian StyleRato, João, Raquel Xavier, D. James Harris, Filipe Banha, and Pedro Anastácio. 2024. "A Comprehensive Review of Disease-Causing Agents in Freshwater Turtles: Implications for Conservation and Public Health" Diversity 16, no. 3: 171. https://doi.org/10.3390/d16030171
APA StyleRato, J., Xavier, R., Harris, D. J., Banha, F., & Anastácio, P. (2024). A Comprehensive Review of Disease-Causing Agents in Freshwater Turtles: Implications for Conservation and Public Health. Diversity, 16(3), 171. https://doi.org/10.3390/d16030171







