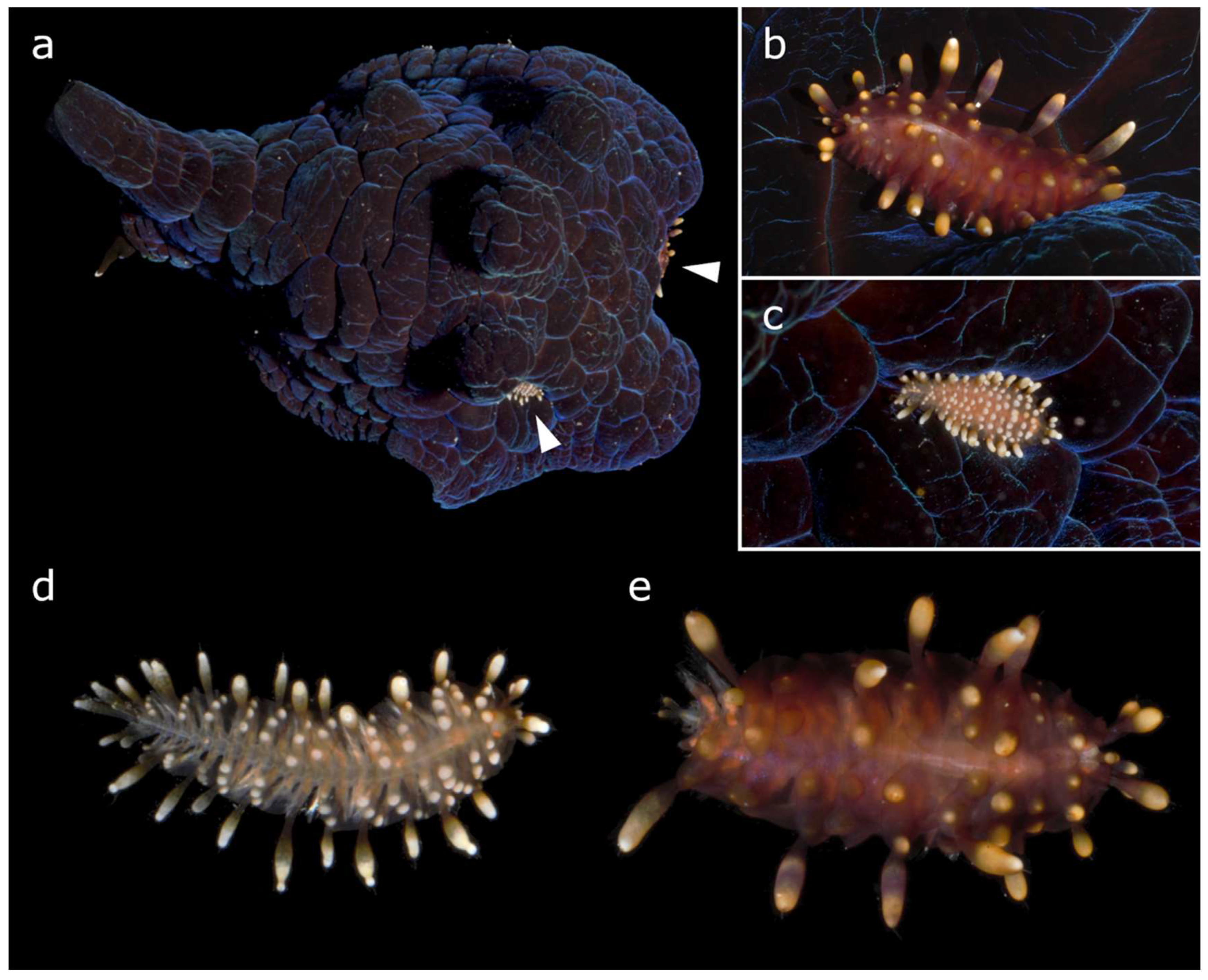
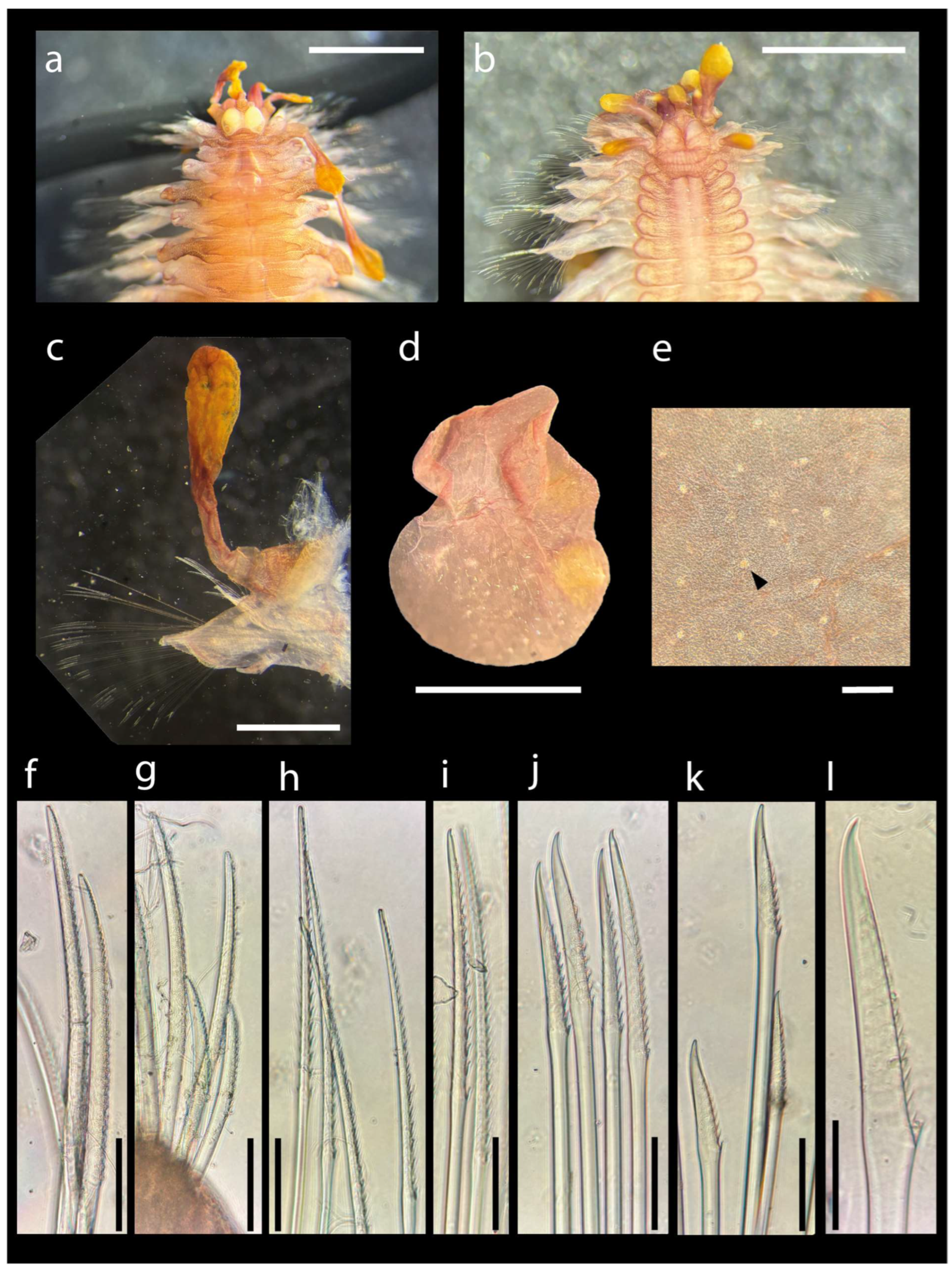
Coriocella and the Worms: First Record of Scale-Worm Asterophilia cf. culcitae Ectosymbiotic on a Mollusc
Abstract
Supplementary Materials
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fassio, G.; Modica, M.V.; Alvaro, M.C.; Buge, B.; Salvi, D.; Oliverio, M.; Schiaparelli, S. An Antarctic flock under the Thorson’s rule: Diversity and larval development of Antarctic Velutinidae (Mollusca: Gastropoda). Mol. Phylogenetics Evol. 2019, 132, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Fassio, G.; Stefani, M.; Russini, V.; Buge, B.; Bouchet, P.; Treneman, N.; Malaquias, M.A.E.; Schiaparelli, S.; Modica, M.V.; Oliverio, M. Neither slugs nor snails: A molecular reappraisal of the gastropod family Velutinidae. Zool. J. Linn. Soc. 2023, 197, 924–964. [Google Scholar] [CrossRef]
- Gulbin, V.V. Prosobranch family Velutinidae (Gastropoda) in cold and temperate waters of the northern hemisphere: History, biogeography, evolution and chorology. Ocean. Sci. J. 2005, 40, 45–54. [Google Scholar] [CrossRef]
- Behrens, D.W.; Ornelas, E.; Valdés, Á. Two new species of Velutinidae Gray, 1840 (Gastropoda) from the North Pacific with a preliminary molecular phylogeny of the family. Nautilus 2014, 128, 114–121. [Google Scholar]
- Andersen, R.J.; Faulkner, D.J.; He, C.H.; Van Duyne, G.D.; Clardy, J. Metabolites of the marine prosobranch mollusk Lamellaria sp. J. Am. Chem. Soc. 1985, 107, 5492–5495. [Google Scholar] [CrossRef]
- Cantrell, C.L.; Groweiss, A.; Gustafson, K.R.; Boyd, M.R. A new staurosporine analog from the prosobranch mollusk Coriocella nigra. Nat. Prod. Lett. 1999, 14, 39–46. [Google Scholar] [CrossRef]
- Bailly, C. Anticancer Properties of Lamellarins. Mar. Drugs 2015, 13, 1105–1123. [Google Scholar] [CrossRef] [PubMed]
- Maggioni, D.; Saponari, L.; Montano, S. Shrimps with a coat: An amphipod hiding in the mantle of Coriocella hibyae (Gastropoda, Velutinidae). Coral Reefs 2018, 37, 647. [Google Scholar] [CrossRef]
- Kumar, J.Y.; Raghunathan, C.; Venkataraman, K. A report on some symbiotic shrimps (Crustacea: Decapoda) from the Andaman and Nicobar Islands, India. Sch. Acad. J. Biosci. 2015, 3, 113–119. [Google Scholar]
- Martin, D.; Britayev, T.A. Symbiotic polychaetes: Review of known species. Oceanogr. Mar. Biol. Ann. Rev. 1998, 36, 217–340. [Google Scholar]
- Martin, D.; Britayev, T.A. Symbiotic polychaetes revisited: An update of the known species and relationships (1998–2017). Oceanogr. Mar. Biol. Ann. Rev. 2018, 56, 371–448. [Google Scholar]
- Sugiyama, T.; Jimi, N.; Goto, R. Widening the host range of the ectosymbiotic scale-worm Asterophilia culcitae (Annelida: Polynoidae) to three echinoderm classes, with data on its body color variation. Plankton Benthos Res. 2020, 15, 289–295. [Google Scholar] [CrossRef]
- Britayev, T.A.; Fauchald, K. New species of symbiotic scaleworms Asterophilia (Polychaeta, Polynoidae) from Vietnam. Invertebr. Zool. 2005, 2, 15–22. [Google Scholar] [CrossRef]
- Hanley, J.R. Revision of the scaleworm genera Arctonoe Chamberlin and Gastrolepidia Schmarda (Polychaeta: Polynoidae) with the erection of a new subfamily Arctonoinae. Beagle 1989, 6, 1–34. [Google Scholar] [CrossRef]
- Fassio, G.; Bouchet, P.; Lozouet, P.; Modica, M.V.; Russini, V.; Schiaparelli, S.; Oliverio, M. Becoming a limpet: An ‘intermittent limpetization’ process driven by host features in the kleptoparasitic gastropod family Capulidae. Mol. Phylogenetics Evol. 2021, 155, 107014. [Google Scholar] [CrossRef] [PubMed]
- Fassio, G.; Modica, M.V.; Alvaro, M.C.; Schiaparelli, S.; Oliverio, M. Developmental trade-offs in Southern Ocean mollusc kleptoparasitic species. Hydrobiologia 2015, 761, 121–141. [Google Scholar] [CrossRef]
- Hoeksema, B.W.; Fransen, C.H. Host switch by the Caribbean anemone shrimp Periclimenes rathbunae in Curaçao. Coral Reefs 2017, 36, 607. [Google Scholar] [CrossRef][Green Version]
- Fransen, C.H.; Hoeksema, B.W. Going for the stars: Extending the host record for the reef-dwelling Emperor shrimp, Periclimenes imperator (Pontoniinae). Mar. Biodivers. 2014, 44, 465–466. [Google Scholar] [CrossRef]
- Horká, I.; De Grave, S.; Fransen, C.H.; Petrusek, A.; Ďuriš, Z. Multiple host switching events shape the evolution of symbiotic palaemonid shrimps (Crustacea: Decapoda). Sci. Rep. 2016, 6, 26486. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.; Atkinson, R.; Gordon, J.; Smith, I.; Hughes, D. Coral-associated invertebrates: Diversity, ecological importance and vulnerability to disturbance. In Oceanography and Marine Biology; Taylor & Francis: Oxford, UK, 2011; Volume 49, pp. 43–104. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fassio, G. Coriocella and the Worms: First Record of Scale-Worm Asterophilia cf. culcitae Ectosymbiotic on a Mollusc. Diversity 2024, 16, 65. https://doi.org/10.3390/d16010065
Fassio G. Coriocella and the Worms: First Record of Scale-Worm Asterophilia cf. culcitae Ectosymbiotic on a Mollusc. Diversity. 2024; 16(1):65. https://doi.org/10.3390/d16010065
Chicago/Turabian StyleFassio, Giulia. 2024. "Coriocella and the Worms: First Record of Scale-Worm Asterophilia cf. culcitae Ectosymbiotic on a Mollusc" Diversity 16, no. 1: 65. https://doi.org/10.3390/d16010065
APA StyleFassio, G. (2024). Coriocella and the Worms: First Record of Scale-Worm Asterophilia cf. culcitae Ectosymbiotic on a Mollusc. Diversity, 16(1), 65. https://doi.org/10.3390/d16010065





