The Role of Old Relicts in Structuring the Boreal/Tropical Transitional Zone: The Case of East Asian Planktonic Cladocera (Crustacea)
Abstract
1. Introduction
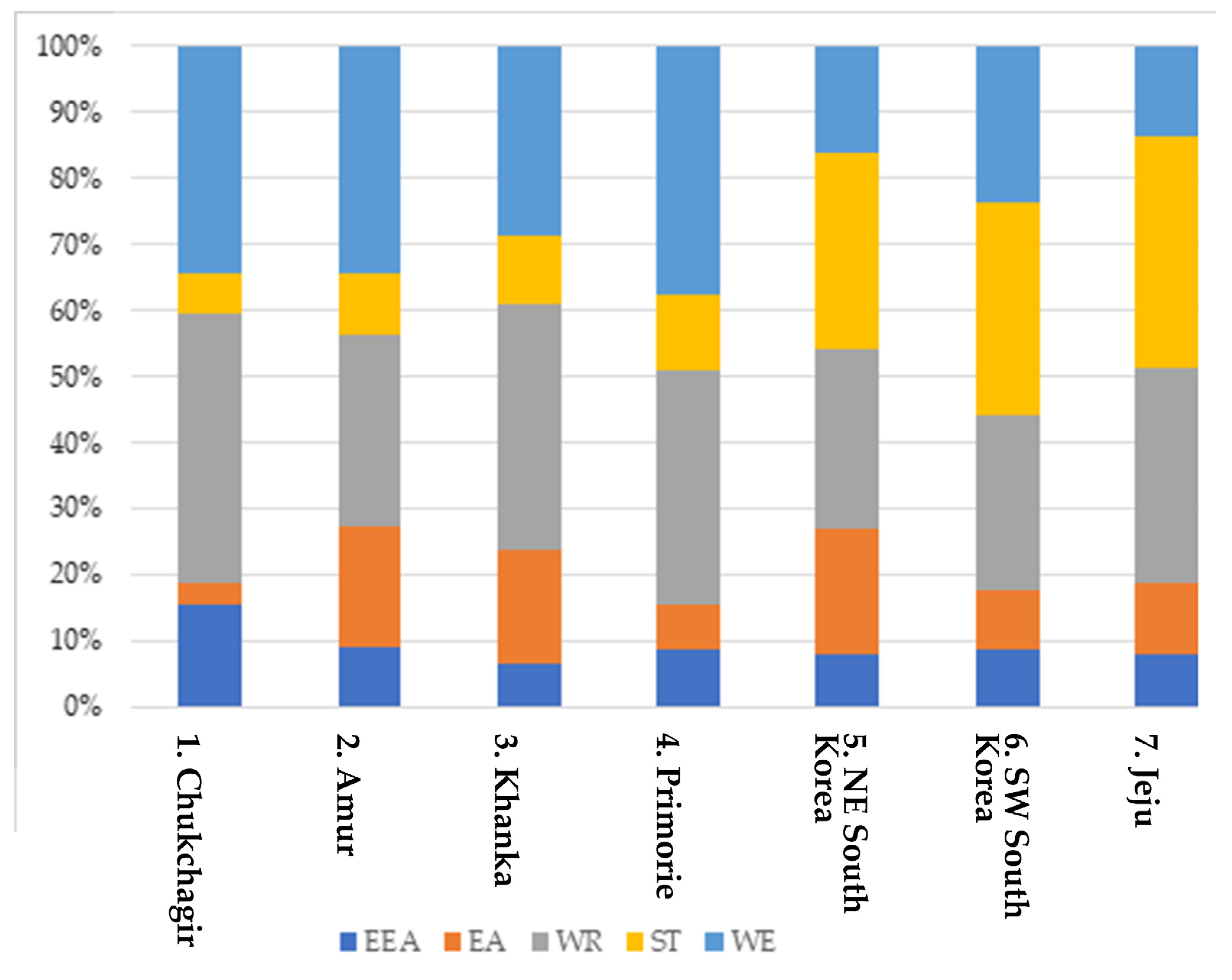
- Widely distributed Eurasian (WE).
- Widely distributed in East Asia and North America (EAA).
- Endemic species belonging to the Far Eastern zone of endemism (EA).
- Southern tropical (ST).
2. Materials and Methods
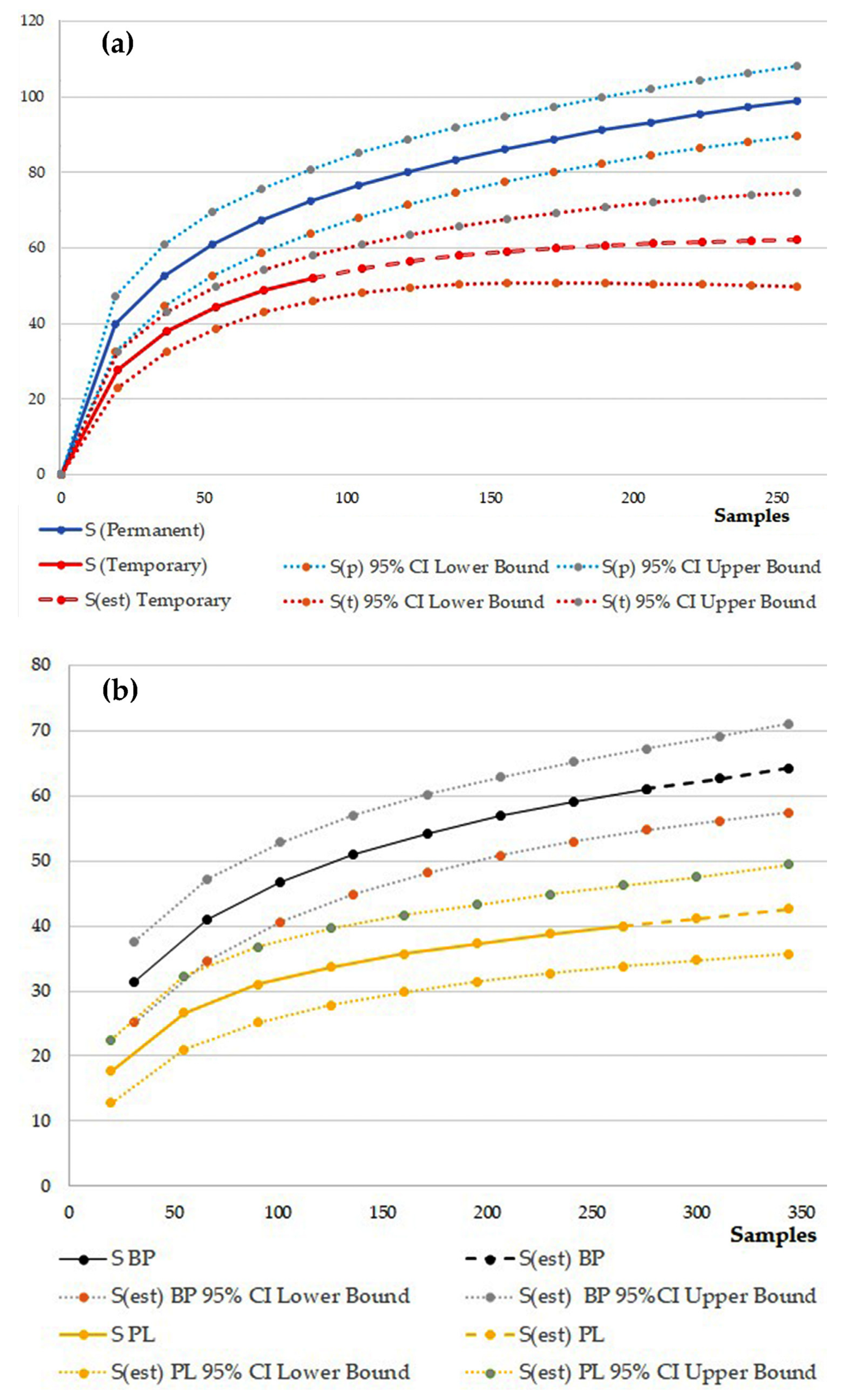
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simpson, G.G. Too many lines; the limits of the Oriental and Australian zoogeographic regions. Proc. Am. Philos. Soc. 1977, 121, 107–120. [Google Scholar]
- Burton, R.S. Intraspecific phylogeography across the point conception biogeographic boundary. Evolution 1998, 52, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Kreft, H.; Jetz, W. A framework for delineating biogeographical regions based on species distributions. J. Biogeogr. 2010, 37, 2029–2053. [Google Scholar] [CrossRef]
- Morrone, J.J. Fundamental biogeographic patterns across the Mexican Transition Zone: An evolutionary approach. Ecography 2010, 33, 355–361. [Google Scholar] [CrossRef]
- Ferro, I.; Morrone, J.J. Biogeographical transition zones: A search for conceptual synthesis. Biol. J. Linn. Soc. 2014, 113, 1–12. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Mazel, F.; Thuiller, W. Global determinants of zoogeographical boundaries. Nat. Ecol. Evol. 2017, 1, 89. [Google Scholar] [CrossRef]
- El Ayari, T.; Trigui El Menif, N.; Hamer, B.; Cahill, A.E.; Bierne, N. The hidden side of a major marine biogeographic boundary: A wide mosaic hybrid zone at the Atlantic-Mediterranean divide reveals the complex interaction between natural and genetic barriers in mussels. Heredity 2019, 122, 770–784. [Google Scholar] [CrossRef]
- Bolotov, I.N.; Kondakov, A.V.; Konopleva, E.S.; Vikhrev, I.V.; Aksenova, O.V.; Aksenov, A.S.; Bespalaya, Y.V.; Borovskoy, A.V.; Danilov, P.P.; Dvoryankin, G.A.; et al. Integrative taxonomy, biogeography and conservation of freshwater mussels (Unionidae) in Russia. Sci. Rep. 2020, 10, 3072. [Google Scholar] [CrossRef]
- Wallace, A.R. The Geographical Distribution of Animals; Harper and Brothers: New York, NY, USA, 1876. [Google Scholar]
- Chen, L.; Song, Y.; Xu, S. The boundary of palaearctic and oriental realms in western China. Prog. Nat. Sci. 2008, 18, 833–841. [Google Scholar] [CrossRef]
- Rueda, M.; Rodríguez, M.Á.; Hawkins, B.A. Identifying global zoogeographical regions: Lessons from Wallace. J. Biogeogr. 2013, 40, 2215–2225. [Google Scholar] [CrossRef]
- Holt, B.G.; Lessard, J.-P.; Borregaard, M.K.; Fritz, S.A.; Araújo, M.B.; Dimitrov, D.; Fabre, P.-H.; Graham, C.H.; Graves, G.R.; Jønsson, K.A.; et al. An update of Wallace’s zoogeographic regions of the world. Science 2013, 339, 74–78. [Google Scholar] [CrossRef]
- Berg, L.S. The Division of the Palaearctic and the Amur Region into Zoogeographic Regions on the Basis of the Distribution of Freshwater Fishes; Izdatelstvo AN SSSR: Moscow, Russia, 1962. [Google Scholar]
- Starobogatov, Y. The Molluscan Fauna and the Zoogeographical Zonation of the Continental Water Reservoirs of the Earth; Nauka Press: Leningrad, Russia, 1970. [Google Scholar]
- Chertoprud, M.V. Biogeographic zonation of the Eurasian fresh waters based on the macrobenthic faunas. Zh. Obshch. Biol. 2010, 71, 144–162. [Google Scholar]
- Bledzki, L.A.; Rybak, J.I. Freshwater Crustacean Zooplankton of Europe; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-29870-2. [Google Scholar]
- Adamowicz, S.J.; Purvis, A. How many branchiopod crustacean species are there? Quantifying the components of underestimation. Glob. Ecol. Biogeogr. 2005, 14, 455–468. [Google Scholar] [CrossRef]
- Korovchinsky, N.M.; Kotov, A.A.; Sinev, A.Y.; Neretina, A.N.; Garibian, P.G. Water Fleas (Crustacea: Cladocera) of North Eurasia; KMK Press: Moscow, Russia, 2021; Volume 2. [Google Scholar]
- Van Damme, K.; Kotov, A.A. The fossil record of the Cladocera (Crustacea: Branchiopoda): Evidence and hypotheses. Earth-Sci. Rev. 2016, 163, 162–189. [Google Scholar] [CrossRef]
- Incagnone, G.; Marrone, F.; Barone, R.; Robba, L.; Naselli-Flores, L. How do freshwater organisms cross the “dry ocean”? A review on passive dispersal and colonization processes with a special focus on temporary ponds. Hydrobiologia 2015, 750, 103–123. [Google Scholar] [CrossRef]
- Figuerola, J.; Green, A.J. Dispersal of aquatic organisms by waterbirds: A review of past research and priorities for future studies. Freshw. Biol. 2002, 47, 483–494. [Google Scholar] [CrossRef]
- Lampert, W. Daphnia: Development of a Model Organism in Ecology and Evolution; International Ecology Institute: Oldendorf/Luhe, Germany, 2011; ISBN 978-3-946729-21-1. [Google Scholar]
- Smirnov, N.N. Physiology of the Cladocera: Second Edition; Academic Press: Amsterdam, The Netherlands, 2017; ISBN 9780128051948. [Google Scholar]
- Jeppesen, E.; Nõges, P.; Davidson, T.A.; Haberman, J.; Nõges, T.; Blank, K.; Lauridsen, T.L.; Søndergaard, M.; Sayer, C.; Laugaste, R.; et al. Zooplankton as indicators in lakes: A scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- Chen, G.; Dalton, C.; Taylor, D. Cladocera as indicators of trophic state in Irish lakes. J. Paleolimnol. 2010, 44, 465–481. [Google Scholar] [CrossRef]
- Mehner, T.; Keeling, C.; Emmrich, M.; Holmgren, K.; Argillier, C.; Volta, P.; Winfield, I.J.; Brucet, S. Effects of fish predation on density and size spectra of prey fish communities in lakes. Can. J. Fish. Aquat. Sci. 2016, 73, 506–518. [Google Scholar] [CrossRef]
- Kirillova, I.V.; Argant, J.; Lapteva, E.G.; Korona, O.M.; van der Plicht, J.; Zinovyev, E.V.; Kotov, A.; Chernova, O.F.; Fadeeva, E.; Baturina, O.A.; et al. The diet and environment of mammoths in North-East Russia reconstructed from the contents of their feces. Quat. Int. 2016, 406, 147–161. [Google Scholar] [CrossRef]
- Korponai, J.L.; Kövér, C.; López-Blanco, C.; Gyulai, I.; Forró, L.; Katalinic, A.; Ketola, M.; Nevalainen, L.; Luoto, T.P.; Sarmaja-Korjonen, K.; et al. Effect of Temperature on the Size of Sedimentary Remains of Littoral Chydorids. Water 2020, 12, 1309. [Google Scholar] [CrossRef]
- Korponai, J.; Braun, M.; Forró, L.; Gyulai, I.; Kövér, C.; Nédli, J.; Urák, I.; Buczkó, K. Taxonomic, functional and phylogenetic diversity: How subfossil cladocerans mirror contemporary community for ecosystem functioning: A comparative study in two oxbows. Limnetica 2019, 38, 431–456. [Google Scholar] [CrossRef]
- Frey, D.G. Cladocera analysis. In Handbook of the Holocene Palaeoecology and Palaeohydrology; Berglund, B.E., Ralska-Jasiewiczowa, M., Eds.; J. Wiley & Sons Ltd.: Caldwell, NJ, USA, 1986; pp. 667–692. [Google Scholar]
- Jeong, H.; Kotov, A.A.; Lee, W. Checklist of the freshwater Cladocera (Crustacea: Branchiopoda) of South Korea. Proc. Biol. Soc. Wash. 2014, 127, 216–228. [Google Scholar] [CrossRef]
- Ji, G.-H.; Xiang, X.-F.; Chen, S.-Z.; Yu, G.-L.; Kotov, A.A.; Dumont, H.J. Annotated checklist of Chinese Cladocera (Crustacea: Branchiopoda). Part II. Order Anomopoda (families Macrotrichidae, Eurycercidae and Chydoridae). Zootaxa 2015, 4044, 241–269. [Google Scholar] [CrossRef]
- Xiang, X.-F.; Ji, G.-H.; Chen, S.-Z.; Yu, G.-L.; Xu, L.; Han, B.-P.; Kotov, A.A.; Dumont, H.J. Annotated Checklist of Chinese Cladocera (Crustacea: Branchiopoda). Part I. Haplopoda, Ctenopoda, Onychopoda and Anomopoda (families Daphniidae, Moinidae, Bosminidae, Ilyocryptidae). Zootaxa 2015, 3904, 1–27. [Google Scholar] [CrossRef]
- Kotov, A.A.; Korovchinsky, N.M.; Sinev, A.Y.; Smirnov, N.N. Cladocera (Crustacea, Branchiopoda) of the Zeya basin (Amurskaya Area, Russian Federation). 3. Systematic-faunistic and zoogeographic analysis. Zool. Zh. 2011, 90, 402–411. [Google Scholar]
- Kotov, A.A. Faunistic complexes of the Cladocera (Crustacea, Branchiopoda) of Eastern Siberia and the Far East of Russia. Zool. Zh. 2016, 95, 748–768. [Google Scholar] [CrossRef]
- Garibian, P.G.; Neretina, A.N.; Korovchinsky, N.M.; Sinev, A.Y.; Tchabovsky, A.V.; Kotov, A.A.; Smirnov, N.N. The Southern part of Russian Far East and Korean Peninsula as a transition zone between the boreal and tropical faunas of the waterfleas (Cladocera, Crustacea). Zool. Zh. 2020, 99, 1094–1109. [Google Scholar] [CrossRef]
- Yoon, S.M.; Kim, H.S. A systematic study on the freshwater Cladocera from Korea. Korean J. Syst. Zool. 1987, 3, 175–207. [Google Scholar]
- Yoon, S.M. Arthropoda: Branchiopoda: Anostraca, Notostraca, Spinicaudata, Laevicaudata, Ctenopoda, Anomopoda, Haplopoda Branchiopods. Invertebr. Fauna Korea 2010, 21, 1–156. [Google Scholar]
- Kotov, A.A.; Jeong, H.G.I.; Lee, W. Cladocera (Crustacea: Branchiopoda) of the south-east of the Korean Peninsula, with twenty new records for Korea. Zootaxa 2012, 3368, 50–90. [Google Scholar] [CrossRef]
- Korovchinsky, N.M. Cladocerans of the Order Ctenopoda of the World Fauna (Morphology, Systematics, Ecology, Biogeography); KMK Press: Moscow, Russia, 2004. [Google Scholar]
- Kotov, A.A.; Sinev, A.Y. Cladocera (Crustacea, Branchiopoda) of the Zeya basin (Amurskaya Area, Russian Federation). 2. Descriptions of new taxa. Zool. Zh. 2011, 90, 272–284. [Google Scholar]
- Garibian, P.G. Nicsmirnovius eximius (Kiser 1948) (Cladocera, Chydoridae) from the Primorsky Territory: The first record of the genus from Russia. Zool. Zh. 2017, 96, 1359–1363. [Google Scholar] [CrossRef]
- Korovchinsky, N.M. Identification Guides to the Plankton and Benthos of Inland Waters; Backhuys Publishers: Kerkwerve, The Netherlands; Margraf Publishers: Weikersheim, Germany, 2018; ISBN 978-3-8236-1756-3. [Google Scholar]
- Garibian, P.G.; Neretina, A.N.; Taylor, D.J.; Kotov, A.A. Partial revision of the neustonic genus Scapholeberis Schoedler, 1858 (Crustacea: Cladocera): Decoding of the barcoding results. PeerJ 2020, 8, e10410. [Google Scholar] [CrossRef]
- Ishida, S.; Taylor, D.J. Mature habitats associated with genetic divergence despite strong dispersal ability in an arthropod. BMC Evol. Biol. 2007, 7, 52. [Google Scholar] [CrossRef]
- Ma, X.; Petrusek, A.; Wolinska, J.; Hu, W.; Yin, M. Lineage diversity and reproductive modes of the Daphnia pulex group in Chinese lakes and reservoirs. Mol. Phylogenet. Evol. 2019, 130, 424–433. [Google Scholar] [CrossRef]
- Ma, X.; Ni, Y.; Wang, X.; Hu, W.; Yin, M. Lineage diversity, morphological and genetic divergence in Daphnia magna (Crustacea) among Chinese lakes at different altitudes. Contrib. Zool. 2020, 89, 450–470. [Google Scholar] [CrossRef]
- Maruoka, N.; Ohtsuki, H.; Makino, W.; Urabe, J. Rediscovery after Almost 120 Years: Morphological and genetic evidence supporting the validity of Daphnia mitsukuri (Crustacea: Cladocera). Zoolog. Sci. 2018, 35, 468–475. [Google Scholar] [CrossRef]
- Wang, L.; Zhuang, H.; Zhang, Y.; Wei, W. Diversity of the Bosmina (Cladocera: Bosminidae) in China, revealed by analysis of two genetic markers (mtDNA 16S and a nuclear ITS). BMC Evol. Biol. 2019, 19, 145. [Google Scholar] [CrossRef]
- Makino, W.; Machida, R.J.; Okitsu, J.; Usio, N. Underestimated species diversity and hidden habitat preference in Moina (Crustacea, Cladocera) revealed by integrative taxonomy. Hydrobiologia 2020, 847, 857–878. [Google Scholar] [CrossRef]
- Yamamoto, A.; Makino, W.; Urabe, J. The taxonomic position of Asian Holopedium (Crustacea: Cladocera) confirmed by morphological and genetic analyses. Limnology 2020, 21, 97–106. [Google Scholar] [CrossRef]
- Deng, Z.; Yao, Y.; Blair, D.; Hu, W.; Yin, M. Ceriodaphnia (Cladocera: Daphniidae) in China: Lineage diversity, phylogeography and possible interspecific hybridization. Mol. Phylogenet. Evol. 2022, 175, 107586. [Google Scholar] [CrossRef] [PubMed]
- Kotov, A.A.; Karabanov, D.P.; Bekker, E.I.; Neretina, T.V.; Taylor, D.J. Phylogeography of the Chydorus sphaericus group (Cladocera: Chydoridae) in the Northern Palearctic. PLoS ONE 2016, 11, e0168711. [Google Scholar] [CrossRef] [PubMed]
- Kotov, A.A.; Taylor, D.J. Contrasting endemism in pond-dwelling cyclic parthenogens: The Daphnia curvirostris species group (Crustacea: Cladocera). Sci. Rep. 2019, 9, 6812. [Google Scholar] [CrossRef] [PubMed]
- Kotov, A.A.; Garibian, P.G.; Bekker, E.I.; Taylor, D.J.; Karabanov, D.P. A new species group from the Daphnia curvirostris species complex (Cladocera: Anomopoda) from the eastern Palaearctic: Taxonomy, phylogeny and phylogeography. Zool. J. Linn. Soc. 2021, 191, 772–822. [Google Scholar] [CrossRef]
- Xu, S.; Hebert, P.D.N.; Kotov, A.A.; Cristescu, M.E. The noncosmopolitanism paradigm of freshwater zooplankton: Insights from the global phylogeography of the predatory cladoceran Polyphemus pediculus (Linnaeus, 1761) (Crustacea, Onychopoda). Mol. Ecol. 2009, 18, 5161–5179. [Google Scholar] [CrossRef]
- Xu, L.; Han, B.-P.; van Damme, K.; Vierstraete, A.; Vanfleteren, J.R.; Dumont, H.J. Biogeography and evolution of the Holarctic zooplankton genus Leptodora (Crustacea: Branchiopoda: Haplopoda). J. Biogeogr. 2011, 38, 359–370. [Google Scholar] [CrossRef]
- Korovchinsky, N.M. The Cladocera (Crustacea: Branchiopoda) as a relict group. Zool. J. Linn. Soc. 2006, 147, 109–124. [Google Scholar] [CrossRef][Green Version]
- Alonso, M. Review of Iberian Cladocera with remarks on ecology and biogeography. Hydrobiologia 1991, 225, 37–43. [Google Scholar] [CrossRef]
- Marrone, F. The microcrustacean fauna of Sicily and the central Mediterranean sea area—Current knowledge and gaps to be filled. Pol. J. Ecol. 2006, 54, 681–685. [Google Scholar]
- Alonso, M.; Neretina, A.N.; Ventura, M. Ceriodaphnia smirnovi (Crustacea: Cladocera), a new species from the Mediterranean Region, and a phylogenetic analysis of the commonest species. Zootaxa 2021, 4974, 146. [Google Scholar] [CrossRef]
- Kotov, A.A.; Seleznev, D.G.; Garibian, P.G.; Korovchnsky, N.M.; Neretina, A.N.; Sinev, A.Y.; Jeong, H.-G.; Yang, H.-M.; Lee, W. History of Colonization of Jeju Island (Republic of Korea) by the Water Fleas (Crustacea: Cladocera) Is Reflected by the Seasonal Changes in Their Fauna and Species Associations. Water 2022, 14, 3394. [Google Scholar] [CrossRef]
- Chertoptud, E.S.; Seleznev, D.G.; Garibian, P.G.; Kotov, A.A. Microcrustaceans (Cladocera and Copepoda) of the Boreal/Tropical Transition Zone in the Russian Far East: A Case Study of Species Associations in Three Large Lakes. Diversity 2023, 15, 338. [Google Scholar] [CrossRef]
- Bow, S.-T. Pattern Recognition: Applications to Large Data-Set Problems/Sing-Tze Bow; Dekker: New York, NY, USA, 1984; ISBN 0824771761. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology, 2nd ed.; Pierre Legendre and Louis Legendre; Elsevier: Amsterdam, The Netherlands; Oxford, UK, 1998; ISBN 0444892508. [Google Scholar]
- Gotelli, N.J.; Ellison, A.M. A Primer of Ecological Statistics, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2013; ISBN 1605350648. [Google Scholar]
- Abell, R.; Thieme, M.L.; Revenga, C.; Bryer, M.; Kottelat, M.; Bogutskaya, N.; Coad, B.; Mandrak, N.; Balderas, S.C.; Bussing, W.; et al. Freshwater Ecoregions of the World: A New Map of Biogeographic Units for Freshwater Biodiversity Conservation. Bioscience 2008, 58, 403–414. [Google Scholar] [CrossRef]
- WWF/TNC. Freshwater Ecoregions of the World. Available online: www.feow.org (accessed on 2 April 2023).
- Colwell, R.K.; Elsensohn, J.E. EstimateS turns 20: Statistical estimation of species richness and shared species from samples, with non-parametric extrapolation. Ecography 2014, 37, 609–613. [Google Scholar] [CrossRef]
- Stephens, M.A. Use of the Kolmogorov-Smirnov, Cramer-von Mises and related statistics without extensive tables. J. R. Stat. Soc. Ser. B 1970, 32, 115–122. [Google Scholar] [CrossRef]
- Pettitt, A.N. A Two-Sample Anderson—Darling Rank Statistic. Biometrika 1976, 63, 161. [Google Scholar] [CrossRef]
- Mann, H.B.; Whitney, D.R. On a Test of Whether one of Two Random Variables is Stochastically Larger than the Other. Ann. Math. Statist. 1947, 18, 50–60. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Sandel, B.; Arge, L.; Dalsgaard, B.; Davies, R.G.; Gaston, K.J.; Sutherland, W.J.; Svenning, J.-C. The influence of Late Quaternary climate-change velocity on species endemism. Science 2011, 334, 660–664. [Google Scholar] [CrossRef]
- Kerfoot, W.C.; Lynch, M. Branchiopod communities: Associations with planktivorous fish in space and time. In Predation. Direct and Indirect Impacts on Aquatic Communities; Academic Press: Hanover, NH, USA; London, UK, 1987; pp. 367–378. [Google Scholar]
- Pinder, A.M.; Halse, S.A.; Shiel, R.J.; McRae, J.M. Granite outcrop pools in south-western Australia: Foci of diversification and refugia for aquatic invertebrates. J. R. Soc. West. Aust. 2000, 83, 149–161. [Google Scholar]
- Céréghino, R.; Biggs, J.; Oertli, B.; Declerck, S. The ecology of European ponds: Defining the characteristics of a neglected freshwater habitat. Hydrobiologia 2008, 597, 1–6. [Google Scholar] [CrossRef]
- Fryer, G. Structure and habits of living branchiopod crustaceans and their bearing on the interpretation of fossil forms. Earth Environ. Sci. Trans. R. Soc. Edinb. 1985, 76, 103–113. [Google Scholar] [CrossRef]
- Zharov, A.A.; Neretina, A.N.; Rogers, D.C.; Reshetova, S.A.; Sinitsa, S.M.; Kotov, A.A. Pleistocene Branchiopods (Cladocera, Anostraca) from Transbaikalian Siberia demonstrate morphological and ecological stasis. Water 2020, 12, 3063. [Google Scholar] [CrossRef]
- Walseng, B.; Hessen, D.O.; Halvorsen, G.; Schartau, A.K. Major contribution from littoral crustaceans to zooplankton species richness in lakes. Limnol. Oceanogr. 2006, 51, 2600–2606. [Google Scholar] [CrossRef]
- Mordukhai-Boltovskoi, P.; Rivier, I.K. Predatory Cladocera: Podonidae, Polyphenidae, Cercopagidae and Leptodoridae of the world fauna. Opredeliteli Po Faune SSSR 1968, 148, 1–182. [Google Scholar]
- Rivier, I.K. The Predatory Cladocera, (Onychopoda: Podonidae, Polyphemidae, Cercopagidae), and Leptodorida of the World; by Irina K. Rivier; with an extended introduction by Henri J. Dumont; Backhuys: Leiden, The Netherlands, 1998; ISBN 9073348854. [Google Scholar]
- Panov, V.E.; Rodionova, N.V.; Bolshagin, P.V.; Bychek, E.A. Invasion biology of Ponto-Caspian onychopod cladocerans (Crustacea: Cladocera: Onychopoda). Hydrobiologia 2007, 590, 3–14. [Google Scholar] [CrossRef]
- Cristescu, M.E.A.; Hebert, P.D.N.; Witt, J.D.S.; MacIsaac, H.J.; Grigorovich, I.A. An invasion history for Cercopagis pengoi based on mitochondrial gene sequences. Limnol. Oceanogr. 2001, 46, 224–229. [Google Scholar] [CrossRef]
- Kotov, A.A.; Karabanov, D.P.; Van Damme, K. Non-Indigenous Cladocera (Crustacea: Branchiopoda): From a Few Notorious Cases to a Potential Global Faunal Mixing in Aquatic Ecosystems. Water 2022, 14, 2806. [Google Scholar] [CrossRef]
- Cristescu, M.E.A.; Hebert, P.D.N. Phylogeny and adaptive radiation in the Onychopoda (Crustacea, Cladocera): Evidence from multiple gene sequences. J. Evol. Biol. 2002, 15, 838–849. [Google Scholar] [CrossRef]
- Xu, S.-L.; Han, B.-P.; Martínez, A.; Schwentner, M.; Fontaneto, D.; Dumont, H.J.; Kotov, A.A. Mitogenomics of Cladocera (Branchiopoda): Marked gene order rearrangements and independent predation roots. Mol. Phylogenet. Evol. 2021, 164, 107275. [Google Scholar] [CrossRef]
- Cristescu, M.E.A.; Hebert, P.D.N.; Onciu, T.M. Phylogeography of Ponto-Caspian crustaceans: A benthic-planktonic comparison. Mol. Ecol. 2003, 12, 985–996. [Google Scholar] [CrossRef]
- Smirnov, N.N. Cladocerans. In Systematics and Evolution of Invertebrates from Baikal; Linevich, A.A., Ed.; Nauka Press: Novosibirsk, Russia, 1984; pp. 75–114. [Google Scholar]
- Sheveleva, N.G.; Pomazkova, G.I.; Melnik, N.G. Eco-taxonomical review of Rotatoria, Cladocera, Calanoida and Cyclopoida of Lake Baikal. Jpn. J. Limnol. 1995, 56, 49–62. [Google Scholar] [CrossRef]
- Dorogostaiskiy, V.C. Vertical and horizontal distribution of the fauna in Lake Baikal. Sb. Tr. Profr. I Prepod. Irkutsk. Univ. 1923, 4, 103–131. [Google Scholar]
- Kozhov, M.M. Biology of Lake Baikal; AN SSSR Press: Moscow, Russia, 1962. [Google Scholar]
- Sherbakov, D.Y.; Kovalenkova, M.V.; Maikova, O.O. Some results of molecular phylogenetic studies of Baikal endemic invertebrates. Russ. J. Genet. Appl. Res. 2017, 7, 345–349. [Google Scholar] [CrossRef]
- Chertoprud, E.S.; Garlitska, L.A.; Azovsky, A.I. Large-scale patterns in marine harpacticoid (Crustacea, Copepoda) diversity and distribution. Mar. Biodivers. 2010, 40, 301–315. [Google Scholar] [CrossRef]
- Kallimanis, A.S.; Bergmeier, E.; Panitsa, M.; Georghiou, K.; Delipetrou, P.; Dimopoulos, P. Biogeographical determinants for total and endemic species richness in a continental archipelago. Biodivers. Conserv. 2010, 19, 1225–1235. [Google Scholar] [CrossRef]
- Pinel-Alloul, B.; André, A.; Legendre, P.; Cardille, J.A.; Patalas, K.; Salki, A. Large-scale geographic patterns of diversity and community structure of pelagic crustacean zooplankton in Canadian lakes. Glob. Ecol. Biogeogr. 2013, 22, 784–795. [Google Scholar] [CrossRef]
- Karpowicz, M.; Ejsmont-Karabin, J. Diversity and Structure of Pelagic Zooplankton (Crustacea, Rotifera) in NE Poland. Water 2021, 13, 456. [Google Scholar] [CrossRef]
- Kotov, A.A.; Ishida, S.; Taylor, D.J. Revision of the genus Bosmina Baird, 1845 (Cladocera: Bosminidae), based on evidence from male morphological characters and molecular phylogenies. Zool. J. Linn. Soc. 2009, 156, 1–51. [Google Scholar] [CrossRef][Green Version]
- Crease, T.J.; Omilian, A.R.; Costanzo, K.S.; Taylor, D.J. Transcontinental phylogeography of the Daphnia pulex species complex. PLoS ONE 2012, 7, e46620. [Google Scholar] [CrossRef] [PubMed]
- Zafirah, N.; Nurin, N.A.; Samsurijan, M.S.; Zuknik, M.H.; Rafatullah, M.; Syakir, M.I. Sustainable Ecosystem Services Framework for Tropical Catchment Management: A Review. Sustainability 2017, 9, 546. [Google Scholar] [CrossRef]
- The Global Learning and Observations to Benefit the Environment (GLOBE) Program. Available online: https://www.globe.gov/web/globescientist/blog/-/blogs/ (accessed on 1 April 2023).
- Lee, S.H.; Lee, Y.I.; Yoon, H.I.; Yoo, K.-C. East Asian monsoon variation and climate changes in Jeju Island, Korea, during the latest Pleistocene to early Holocene. Quat. Res. 2008, 70, 265–274. [Google Scholar] [CrossRef]
- Wiens, J.J. The niche, biogeography and species interactions. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 2336–2350. [Google Scholar] [CrossRef]
- Connell, J.H. The Influence of Interspecific Competition and Other Factors on the Distribution of the Barnacle Chthamalus Stellatus. Ecology 1961, 42, 710–723. [Google Scholar] [CrossRef]
- Davis, A.J.; Jenkinson, L.S.; Lawton, J.H.; Shorrocks, B.; Wood, S. Making mistakes when predicting shifts in species range in response to global warming. Nature 1998, 391, 783–786. [Google Scholar] [CrossRef]
- Parmesan, C.; Gaines, S.; Gonzalez, L.; Kaufman, D.M.; Kingsolver, J.; Townsend Peterson, A.; Sagarin, R. Empirical perspectives on species borders: From traditional biogeography to global change. Oikos 2005, 108, 58–75. [Google Scholar] [CrossRef]
- Cunningham, H.R.; Rissler, L.J.; Apodaca, J.J. Competition at the range boundary in the slimy salamander: Using reciprocal transplants for studies on the role of biotic interactions in spatial distributions. J. Anim. Ecol. 2009, 78, 52–62. [Google Scholar] [CrossRef]
- Svenning, J.-C.; Gravel, D.; Holt, R.D.; Schurr, F.M.; Thuiller, W.; Münkemüller, T.; Schiffers, K.H.; Dullinger, S.; Edwards, T.C.; Hickler, T.; et al. The influence of interspecific interactions on species range expansion rates. Ecography 2014, 37, 1198–1209. [Google Scholar] [CrossRef]
- Hu, J.; Jiang, J. Inferring ecological explanations for biogeographic boundaries of parapatric Asian mountain frogs. BMC Ecol. 2018, 18, 3. [Google Scholar] [CrossRef]
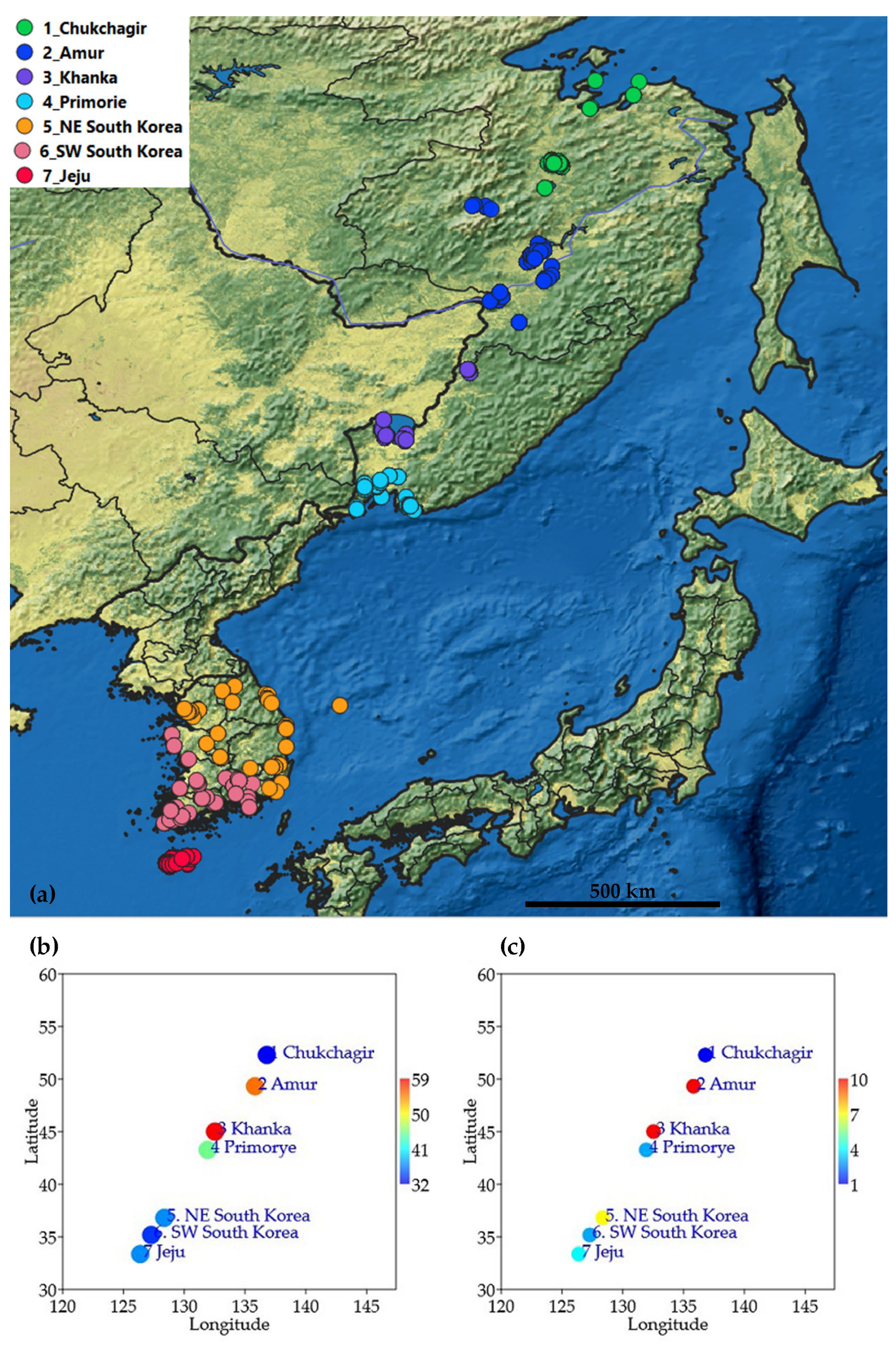


| Sub-Region | Water Bodies Total | Permanent | Temporary | Number of Identifications | Freshwater Ecoregion | Mean Latitude, °N | Mean Longitude, °E |
|---|---|---|---|---|---|---|---|
| 1. Chukchagir | 34 | 33 | 1 | 197 | 615, 616 | 52.29 | 136.79 |
| 2. Amur | 44 | 31 | 13 | 276 | 616 | 49.32 | 135.82 |
| 3. Khanka | 49 | 41 | 8 | 273 | 616 | 45.01 | 132.53 |
| 4. Primorye | 43 | 17 | 26 | 161 | 641 | 43.27 | 131.93 |
| 5. NE South Korea | 40 | 35 | 5 | 151 | 638, 639 | 36.81 | 128.36 |
| 6. SW South Korea | 51 | 35 | 16 | 185 | 638, 639 | 35.18 | 127.28 |
| 7. Jeju | 82 | 63 | 19 | 419 | 639 | 33.36 | 126.38 |
| Taxon | 1. Chukchagir | 2. Amur | 3. Khanka | 4. Primorye | 5. NE South Korea | 6. SW South Korea | 7. Jeju | Biotope | Faunistic Complex |
|---|---|---|---|---|---|---|---|---|---|
| Acroperus angustatus | + | + | + | + | BP | WE | |||
| A. harpae | + | + | + | + | BP | WE | |||
| Alona guttata | + | + | + | + | + | + | BP | WS | |
| A. quadrangularis | + | + | BP | WE | |||||
| Alonella excisa | + | + | + | + | + | + | + | BP | WS |
| Anchistropus emarginatus | + | BP | WE | ||||||
| Biapertura affinis | + | + | + | + | + | + | BP | WS | |
| B. sibirica | + | + | + | BP | WE | ||||
| Bosmina fatalis | + | + | + | PL | EA | ||||
| B. longirostris | + | + | + | + | + | + | + | PL | WS |
| Bosminopsis zernowi | + | + | + | + | + | PL | WE | ||
| Bunops serricaudata | + | + | BP | WE | |||||
| Camptocercus fennicus | + | BP | WE | ||||||
| C. smirnovi | + | + | BP | EEA | |||||
| C. uncinatus | + | + | + | + | + | + | + | BP | EEA |
| C. vietnamensis | + | BP | ST | ||||||
| Ceriodaphnia cornuta | + | + | + | PL | ST | ||||
| C. laticaudata | + | + | PL | WS | |||||
| C. megops | + | PL | WS | ||||||
| C. pulchella | + | + | + | + | PL | WS | |||
| C. quadrangula | + | + | + | PL | WS | ||||
| C. reticulata | + | + | + | PL | WS | ||||
| C. rotunda | + | PL | WS | ||||||
| Chydorus cf. sphaericus | + | + | + | + | + | + | + | BP | EEA |
| Coronatella jejuana | + | BP | EA | ||||||
| C. rectangula | + | + | + | + | + | + | + | BP | WS |
| C. trachystriata | + | + | BP | EA | |||||
| Daphnia cristata | + | + | PL | WE | |||||
| D. curvirostris | + | + | + | + | PL | WE | |||
| D. galeata | + | + | + | + | + | + | PL | WE | |
| D. jejuana | + | PL | EA | ||||||
| D. koreana | + | PL | EA | ||||||
| D. korovchinskyi | + | PL | EA | ||||||
| D. longispina | + | PL | WS | ||||||
| D. obtusa | + | PL | WS | ||||||
| D. pulex | + | PL | WS | ||||||
| D. sinensis | + | + | + | + | + | + | PL | WE | |
| D. sinevi | + | + | + | PL | EA | ||||
| Diaphanosoma amurensis | + | + | + | PL | EA | ||||
| D. brachyurum | + | + | PL | WE | |||||
| D. chankensis | + | PL | EA | ||||||
| D. dubium | + | + | + | + | + | + | + | PL | EA |
| D. macrophtalma | + | + | + | + | + | PL | EA | ||
| D. pseudodubium | + | PL | EA | ||||||
| D. sarsi | + | PL | ST | ||||||
| D. transamurensis | + | + | PL | EA | |||||
| Disparalona chappuisi | + | + | + | + | + | BP | ST | ||
| D. ikarus | + | + | + | + | + | + | + | BP | ST |
| D. rostrata | + | BP | WE | ||||||
| Drepanothrix dentata | + | BP | WE | ||||||
| Dunhevedia crassa | + | + | BP | ST | |||||
| Eurycercus macracanthus | + | + | + | + | BP | EEA | |||
| Flavalona costata | + | + | + | + | + | + | + | BP | WS |
| F. rustica | + | BP | WS | ||||||
| Graptoleberis testudinaria | + | + | + | + | BP | WS | |||
| Illyocryptus acutifrons | + | BP | WE | ||||||
| I. cuneatus | + | + | BP | WE | |||||
| I. raridentatus | + | + | + | BP | ST | ||||
| I. spinifer | + | + | + | + | + | BP | ST | ||
| I. yooni | + | + | + | + | BP | ST | |||
| Kurzia latissima | + | + | + | BP | WE | ||||
| K. longirostris | + | BP | ST | ||||||
| Leberis diaphanus | + | BP | ST | ||||||
| Leptodora kindtii | + | + | PL | WE | |||||
| L. richardi | + | PL | EA | ||||||
| Leydigia acanthocercoides | + | BP | WE | ||||||
| L. ciliata | + | BP | ST | ||||||
| L. louisi | + | BP | ST | ||||||
| Limnosida frontosa | + | BP | WE | ||||||
| Macrothrix laticornis | + | BP | WE | ||||||
| M. rosea | + | + | + | + | + | BP | WE | ||
| M. triserialis | + | + | BP | ST | |||||
| M. vietnamensis | + | + | BP | ST | |||||
| Megafenestra aurita | + | PL | WE | ||||||
| Moina affinis | + | PL | WS | ||||||
| M. lipini | + | + | PL | WE | |||||
| M. macrocopa | + | + | + | + | PL | WE | |||
| M. micrura | + | + | + | + | + | PL | WS | ||
| M. weismanni | + | + | + | PL | EA | ||||
| Monospilus daedalus | + | BP | EA | ||||||
| Nedorchynchotalona chiangi | + | + | BP | EA | |||||
| Picripleuroxus denticulatus | + | BP | ST | ||||||
| P. laevis | + | + | + | BP | WE | ||||
| P. quasidenticulatus | + | + | + | + | + | BP | ST | ||
| P. striatus | + | + | BP | WE | |||||
| Pleuroxus aduncus | + | + | + | BP | WS | ||||
| P. truncatus | + | + | BP | WE | |||||
| Polyphemus pediculus | + | + | + | PL | WS | ||||
| Pseudochydorus globosus | + | + | + | + | BP | WE | |||
| Pseudosida szalayi | + | + | BP | ST | |||||
| Scapholeberis mucronata | + | + | + | + | PL | WS | |||
| S. smirnovi | + | + | + | + | + | + | PL | ST | |
| Sida crystallina | + | + | + | + | BP | WE | |||
| S. ortiva | + | + | + | + | + | + | BP | EEA | |
| Simocephalus congener | + | BP | WS | ||||||
| S. exspinosus | + | + | + | BP | WS | ||||
| S. mixtus | + | + | + | + | + | + | + | BP | WS |
| S. serrulatus | + | + | + | + | + | BP | WS | ||
| S. vetuloides | + | BP | EEA | ||||||
| S. vetulus | + | + | + | + | BP | WS | |||
| Streblocerus serricaudatus | + | BP | WE | ||||||
| Total | 32 | 55 | 59 | 45 | 37 | 34 | 37 | 101 | 101 |
| Sub-Region | BP | PL | Total |
|---|---|---|---|
| 1. Chukchagir | 147/25 | 50/7 | 197/32 |
| 2. Amur | 193/32 | 83/23 | 276/55 |
| 3. Khanka | 158/34 | 115/25 | 273/59 |
| 4. Primorye | 89/28 | 72/27 | 161/45 |
| 5. NE South Korea | 100/21 | 51/16 | 151/37 |
| 6. SW South Korea | 109/24 | 76/10 | 185/34 |
| 7. Jeju | 283/22 | 136/15 | 419/37 |
| Total | 1079/61 | 583/40 | 1662/101 |
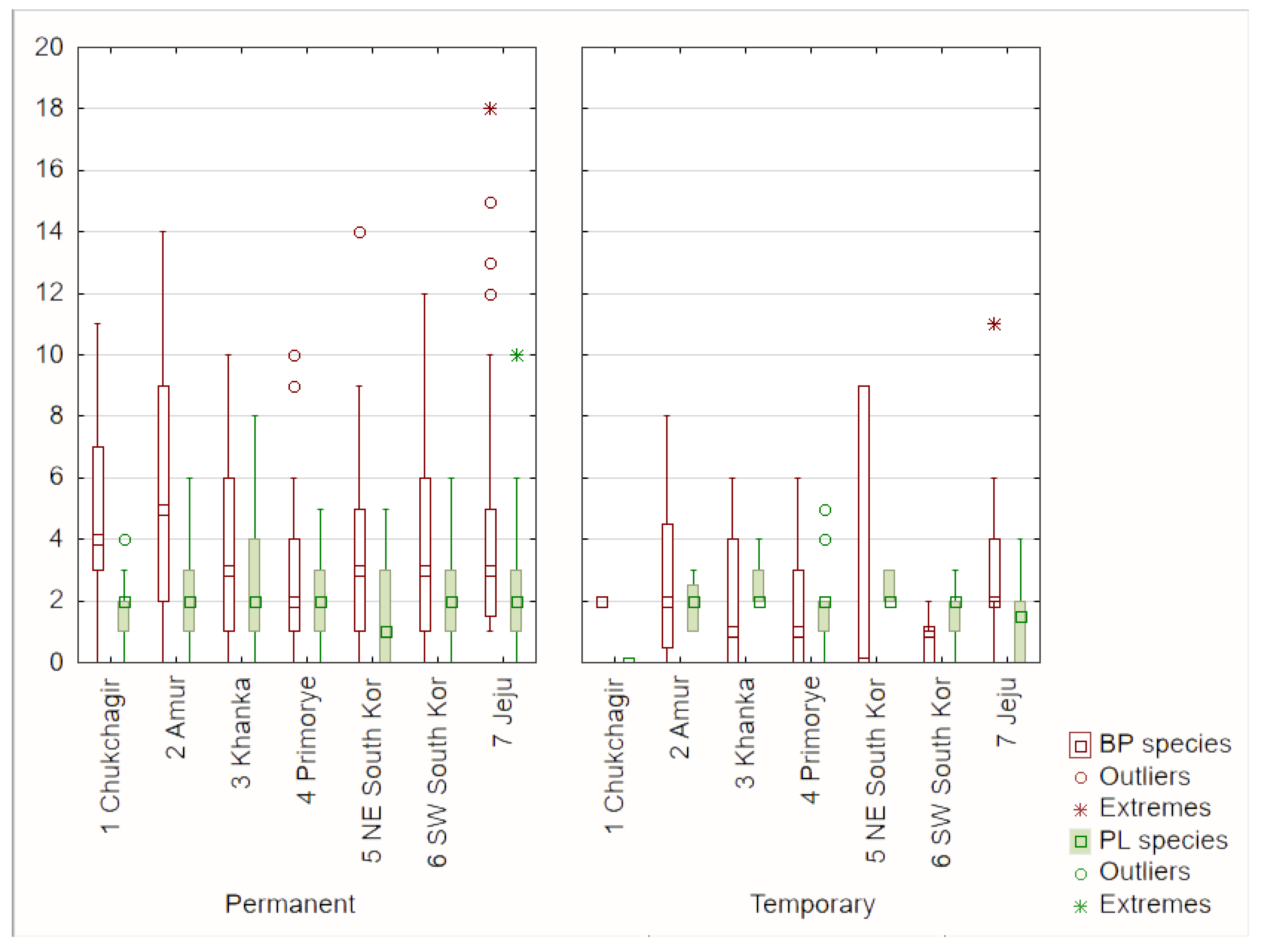
| Water Bodies | BP Species (n ± SD) | PL Species (n ± SD) | EA (n ± SD) | |
|---|---|---|---|---|
| Permanent | 218 | 4.17 ± 3.48 | 2.06 ± 1.67 | 0.41 ± 0.77 |
| Temporary | 60 | 2.18 ± 2.42 | 1.77 ± 1.09 | 0.30 ± 0.57 |
| Total | 278 | 3.74 ± 3.37 | 2.00 ± 1.56 | 0.38 ± 0.72 |
| Rank Sum Permanent | Rank Sum Temporary | U | Z | p-Value | Z Adjusted | p-Value | Valid N Permanent | Valid N Temporary | |
|---|---|---|---|---|---|---|---|---|---|
| PL species | 30,701.50 | 8079.50 | 6249.50 | 0.52678 | 0.59834 | 0.54154 | 0.58813 | 218 | 60 |
| BP species | 32,886.00 | 5895.00 | 4065.00 | 4.48807 | 0.00007 | 4.51964 | 0.00006 | 218 | 60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krolenko, I.I.; Garibian, P.G.; Kotov, A.A. The Role of Old Relicts in Structuring the Boreal/Tropical Transitional Zone: The Case of East Asian Planktonic Cladocera (Crustacea). Diversity 2023, 15, 713. https://doi.org/10.3390/d15060713
Krolenko II, Garibian PG, Kotov AA. The Role of Old Relicts in Structuring the Boreal/Tropical Transitional Zone: The Case of East Asian Planktonic Cladocera (Crustacea). Diversity. 2023; 15(6):713. https://doi.org/10.3390/d15060713
Chicago/Turabian StyleKrolenko, Ivan I., Petr G. Garibian, and Alexey A. Kotov. 2023. "The Role of Old Relicts in Structuring the Boreal/Tropical Transitional Zone: The Case of East Asian Planktonic Cladocera (Crustacea)" Diversity 15, no. 6: 713. https://doi.org/10.3390/d15060713
APA StyleKrolenko, I. I., Garibian, P. G., & Kotov, A. A. (2023). The Role of Old Relicts in Structuring the Boreal/Tropical Transitional Zone: The Case of East Asian Planktonic Cladocera (Crustacea). Diversity, 15(6), 713. https://doi.org/10.3390/d15060713







