Mollusk Thanatocoenoses Unravel the Diversity of Heterogeneous Rhodolith Beds (Italy, Tyrrhenian Sea)
Abstract
1. Introduction
2. Materials and Methods
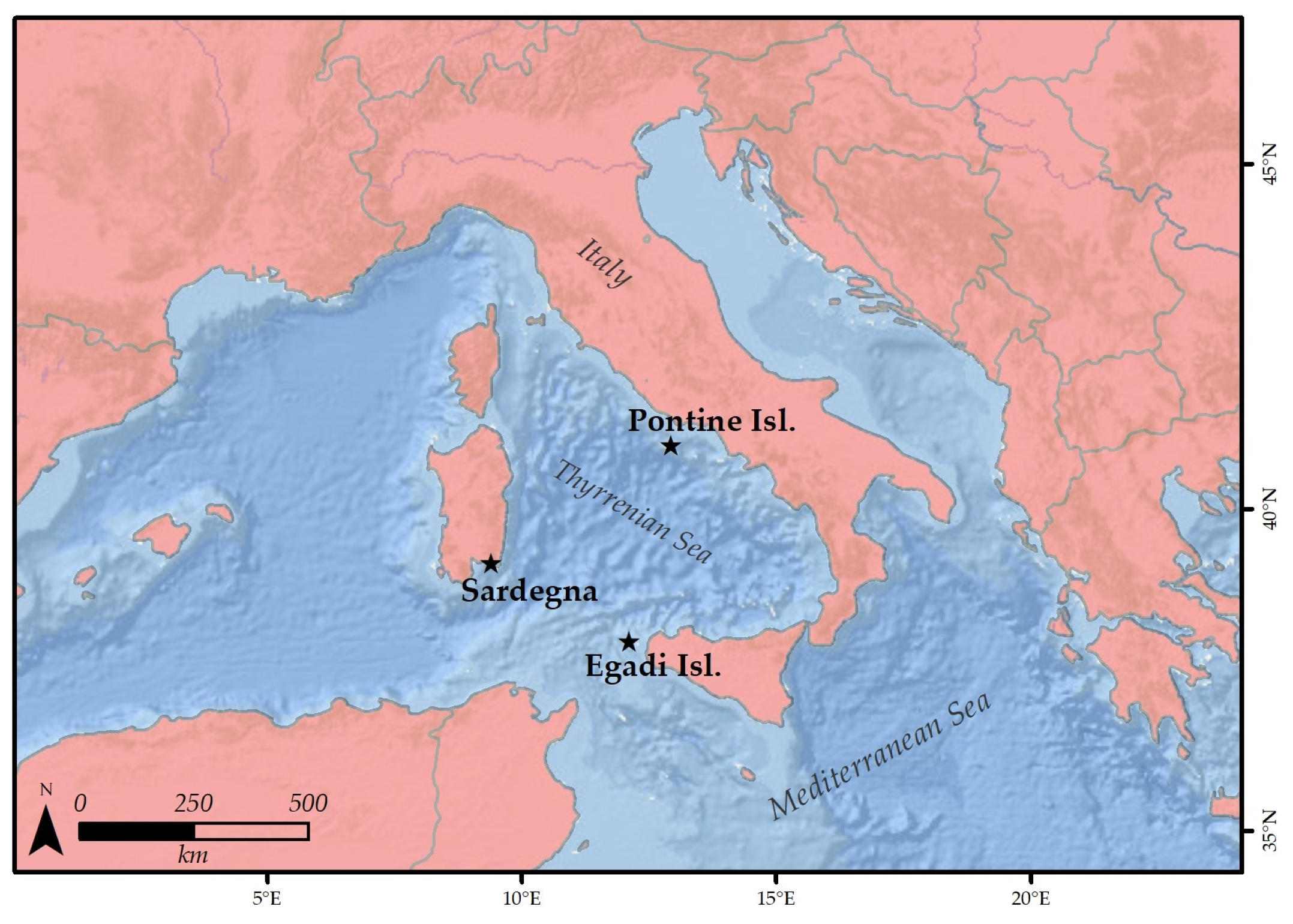
2.1. Sampling
2.2. Rhodoliths
2.3. Grain Size and Mollusks
2.4. Environmental Data
2.5. Statistics
3. Results
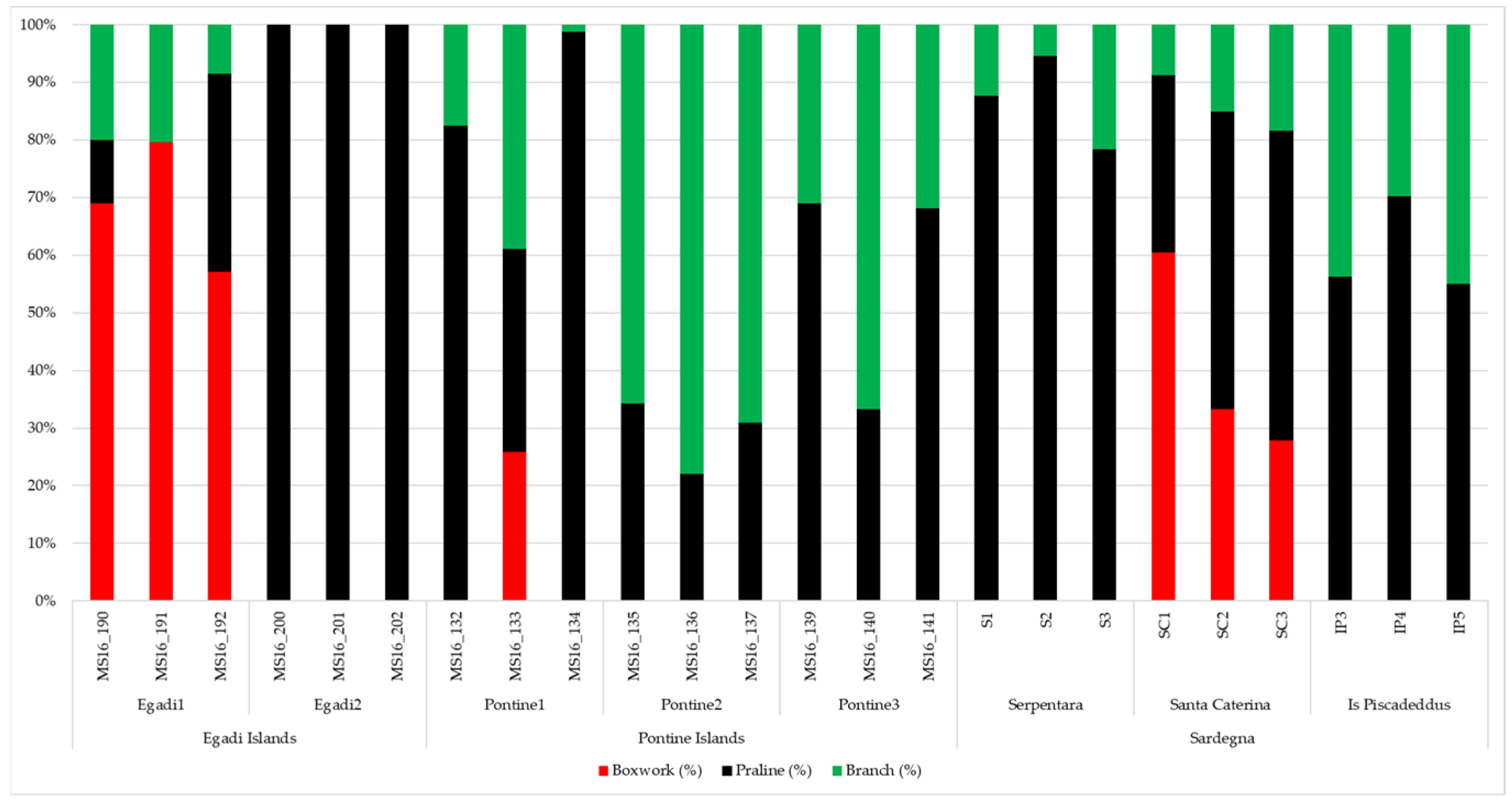
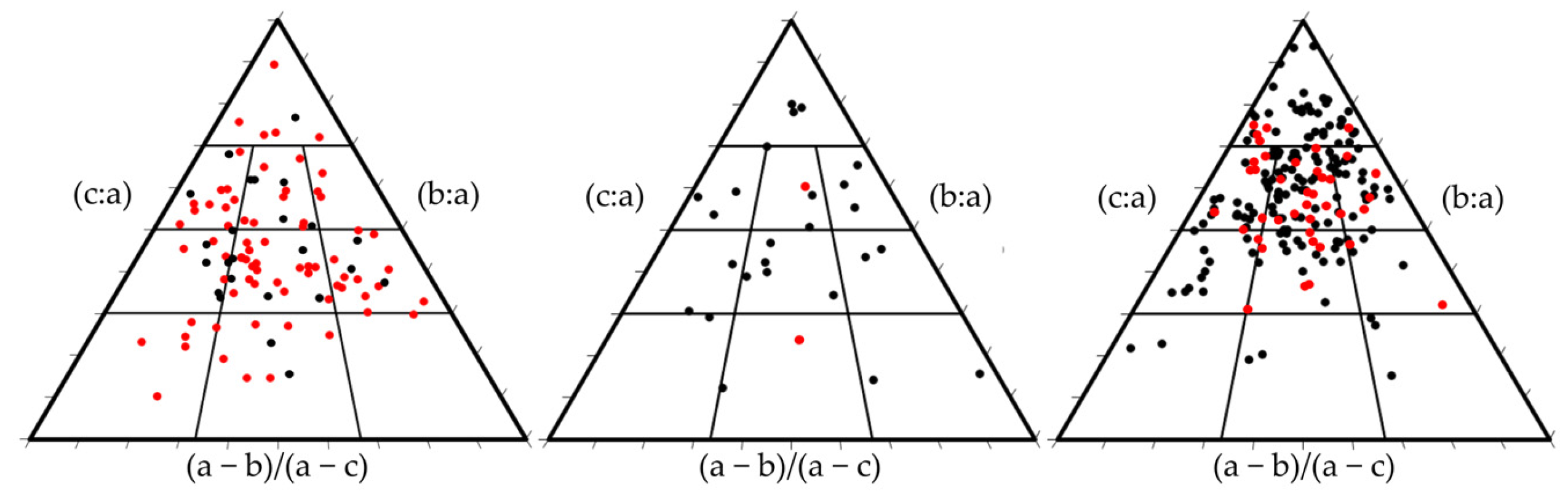
3.1. Rhodoliths
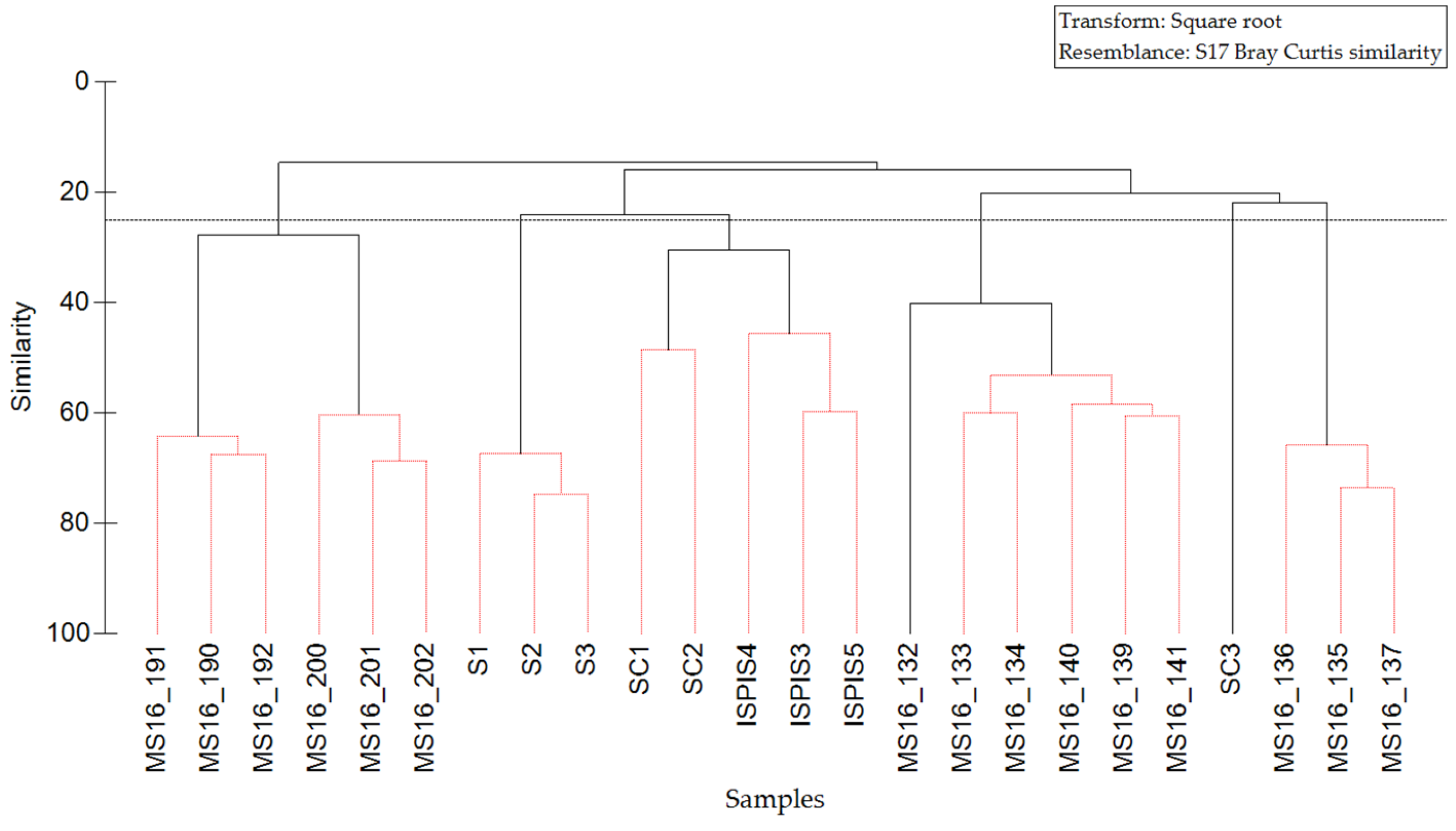
3.2. Mollusks
3.3. Grain Size
3.4. Environmental Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bosellini, A.; Ginsburg, R.N. Form and Internal Structure of Recent Algal Nodules (Rhodolites) from Bermuda. J. Geol. 1971, 79, 669–682. [Google Scholar] [CrossRef]
- Foster, M.S.; Amado Filho, G.M.; Kamenos, K.A.; Riosmena-Rodríguez, R.; Steller, D.L. Rhodoliths and Rhodolith Beds. In Research and Discoveries: The Revolution of Science Through SCUBA; American Academy of Underwater Sciences: Mobile, AL, USA, 2013; pp. 143–155. [Google Scholar]
- Basso, D.; Babbini, L.; Ramos-Esplá, A.A.; Salomidi, M. Mediterranean Rhodolith Beds. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Coastal Research Library; Springer: Cham, Switzerland, 2017; Volume 15, pp. 281–298. [Google Scholar]
- Riosmena-Rodríguez, R.; Nelson, W.; Aguirre, J. Rhodolith/Maërl Beds: A Global Perspective; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Basso, D.; Babbini, L.; Kaleb, S.; Bracchi, V.A.; Falace, A. Monitoring deep Mediterranean rhodolith beds. Aquat. Conserv. Mar. Freshw. Ecosyst. 2015, 26, 549–561. [Google Scholar] [CrossRef]
- UNEP-MAP-RAC/SPA. Action Plan for the Conservation of the Coralligenous and Other Calcareous Bio-Concretions in the Mediterranean Sea; Pergent-Martini, C., Brichet, M., Eds.; RAC/SPA: Tunis, Tunisia, 2008; p. 21. [Google Scholar]
- Basso, D.; Nalin, R.; Massari, F. Genesis and composition of the Pleistocene Coralligène de plateau of the Cutro Terrace (Calabria, southern Italy). Neues Jahrb. Für Geol. Und Paläontologie–Abh. 2009, 244, 73–182. [Google Scholar] [CrossRef]
- Foster, M.S. Rhodoliths: Between Rocks and Soft Places. J. Phycol. 2001, 37, 659–667. [Google Scholar] [CrossRef]
- Steller, D.L.; Riosmena-Rodríguez, R.; Foster, M.S.; Roberts, C.A. Rhodolith bed diversity in the Gulf of California: The importance of rhodolith structure and consequences of disturbance. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, S5–S20. [Google Scholar] [CrossRef]
- Bosence, D.W.J. Ecological studies on two unattached coralline algae from Western Ireland. Palaeontology 1976, 19, 365–395. [Google Scholar]
- BIOMAERL Team. Conservation and management of northeast Atlantic and Mediterranean maërl beds. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, S65–S76. [Google Scholar] [CrossRef]
- Bordehore, C.; Borg, J.A.; Lanfranco, E.; Ramos-Esplá, A.A.; Rizzo, F.; Schembri, P.J. Trawling as a major threat to Mediterranean maërl beds. In Proceedings of the First Mediterranean symposium on Marine Vegetation, Mednature, Ajaccio, France, 3–4 October 2000; Volume 1, pp. 105–109. [Google Scholar]
- Bordehore, C.; Ramos-Esplá, A.A.; Riosmena-Rodríguez, R. Comparative study of two maërl beds with different otter trawling history, southeast Iberian Peninsula. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, S43–S54. [Google Scholar] [CrossRef]
- Bahia, R.G.; Abrantes, D.P.; Brasileiro, P.S.; Filho, G.H.P.; Filho, G.M.A. Rhodolith bed structure along a depth gradient on the northern coast of Bahia state, Brazil. Braz. J. Oceanogr. 2010, 58, 323–337. [Google Scholar] [CrossRef]
- Riul, P.; Targino, C.H.; Farias, J.D.N.; Visscher, P.T.; Horta, P.A. Decrease of Lithothamnion sp. (Rhodophyta) primary production due to the deposition of a thin sediment layer. J. Mar. Biol. Ass. UK 2008, 88, 17–19. [Google Scholar] [CrossRef]
- Barberá, C.; Moranta, J.; Ordines, F.; Ramón, M.; de Mesa, A.; Díaz-Valdés, M.; Grau, A.M.; Massutí, E. Biodiversity and habitat mapping of Menorca Channel (western Mediterranean): Implications for conservation. Biodivers. Conserv. 2012, 21, 701–728. [Google Scholar] [CrossRef]
- Micallef, A.; Le Bas, T.P.; Huvenne, V.A.; Blondel, P.; Hühnerbach, V.; Deidun, A. A multi-method approach for benthic habitat mapping of shallow coastal areas with high-resolution multibeam data. Cont. Shelf Res. 2012, 39–40, 14–26. [Google Scholar] [CrossRef]
- Sañé, E.; Chiocci, F.; Basso, D.; Martorelli, E. Environmental factors controlling the distribution of rhodoliths: An integrated study based on seafloor sampling, ROV and side scan sonar data, offshore the W-Pontine Archipelago. Cont. Shelf Res. 2016, 129, 10–22. [Google Scholar] [CrossRef]
- Millar, K.; Gagnon, P. Mechanisms of stability of rhodolith beds: Sedimentological aspects. Mar. Ecol. Prog. Ser. 2018, 594, 65–83. [Google Scholar] [CrossRef]
- O’Connell, L.G.; James, N.P.; Harvey, A.S.; Luick, J.; Bone, Y.; Shepherd, S.A. Reevaluation of the Inferred Relationship Between Living Rhodolith Morphologies, Their Movement, and Water Energy: Implications for Interpreting Paleoceanographic Conditions. Palaios 2020, 35, 543–556. [Google Scholar] [CrossRef]
- Marrack, E.C. The Relationship between Water Motion and Living Rhodolith Beds in the Southwestern Gulf of California, Mexico. Palaios 1999, 14, 159–171. [Google Scholar] [CrossRef]
- Basso, D.; Tomaselli, V. Paleoecological potentiality of rhodoliths: A Mediterranean case history. Boll. Soc. Paleontol. Ital. 1994, 2, 17–27. [Google Scholar]
- Basso, D.; Nalin, R.; Nelson, C.S. Shallow-water Sporolithon rhodoliths from north island (New Zealand). Palaios 2009, 24, 92–103. [Google Scholar] [CrossRef]
- Bracchi, V.A.; Caronni, S.; Meroni, A.N.; Burguett, E.G.; Atzori, F.; Cadoni, N.; Marchese, F.; Basso, D. Morphostructural Characterization of the Heterogeneous Rhodolith Bed at the Marine Protected Area “Capo Carbonara” (Italy) and Hydrodynamics. Diversity 2022, 14, 51. [Google Scholar] [CrossRef]
- Jardim, V.L.; Gauthier, O.; Toumi, C.; Grall, J. Quantifying maerl (rhodolith) habitat complexity along an environmental gradient at regional scale in the Northeast Atlantic. Mar. Environ. Res. 2022, 181, 105768. [Google Scholar] [CrossRef]
- Basso, D.; Vrsaljko, D.; Grgasović, T. The coralline flora of a Miocene maërl: The Croatian “Litavac”. Geol. Croat. 2008, 61, 333–340. [Google Scholar] [CrossRef]
- Bassi, D.; Nebelsick, J.H.; Checconi, A.; Hohenegger, J.; Iryu, Y. Present-day and fossil rhodolith pavements compared: Their potential for analysing shallow-water carbonate deposits. Sediment. Geol. 2009, 214, 74–84. [Google Scholar] [CrossRef]
- Gagnon, P.; Matheson, K.; Stapleton, M. Variation in rhodolith morphology and biogenic potential of newly discovered rhodolith beds in Newfoundland and Labrador (Canada). Bot. Mar. 2012, 55, 85–99. [Google Scholar] [CrossRef]
- De Queiroz, E.V.; Araújo, P.V.D.N.; Hammill, E.; Amaral, R.F.D. Morphological characteristics of rhodolith and correlations with associated sediment in a sandstone reef: Northeast Brazil. Reg. Stud. Mar. Sci. 2016, 8, 133–140. [Google Scholar] [CrossRef]
- Bracchi, V.A.; Angeletti, L.; Marchese, F.; Taviani, M.; Cardone, F.; Hajdas, I.; Grande, V.; Prampolini, M.; Caragnano, A.; Corselli, C.; et al. A resilient deep-water rhodolith bed off the Egadi Archipelago (Mediterranean Sea) and its actual paleontological significance. Alp. Mediterr. Quat. 2019, 32, 1–20. [Google Scholar]
- Basso, D. Deep rhodolith distribution in the Pontian Islands, Italy: A model for the paleoecology of a temperate sea. Palaeogeogr. Palaeoclim. Palaeoecol. 1998, 137, 173–187. [Google Scholar] [CrossRef]
- Bosence, D.W.J. Description and Classification of Rhodoliths (Rhodoids, Rhodolites). In Coated Grains; Peryt, T.M., Ed.; Springer: Berlin/Heidelberg, Germany, 1983; pp. 217–224. [Google Scholar]
- Carvalho, V.F.; Assis, J.; Serrão, E.A.; Nunes, J.M.; Anderson, A.B.; Batista, M.B.; Barufi, J.B.; Silva, J.; Pereira, S.M.; Horta, P.A. Environmental drivers of rhodolith beds and epiphytes community along the South Western Atlantic coast. Mar. Environ. Res. 2019, 154, 104827. [Google Scholar] [CrossRef]
- Adey, W.H.; Macintyre, I.G. Crustose Coralline Algae: A Re-evaluation in the Geological Sciences. GSA Bull. 1973, 84, 883–904. [Google Scholar] [CrossRef]
- Steller, D.L.; Foster, M.S. Environmental factors influencing distribution and morphology of rhodoliths on Bahía Concepción, B.CS, México. J. Exp. Mar. Biol. Ecol. 1995, 194, 201–212. [Google Scholar] [CrossRef]
- Lund, M.; Davies, P.J.; Braga, J.C. Coralline algal nodules off Fraser Island, eastern Australia. Facies 2000, 42, 25–34. [Google Scholar] [CrossRef]
- Foster, M.S.; Riosmena-Rodriguez, R.; Steller, D.L.; Woelkerling, W.J. Living rhodolith beds in the Gulf of California and their implications. In Pliocene Carbonates and Related Facies Flanking the Gulf of California, Baja California, Mexico; Special Paper; Johnson, M.E., Ledesma-Vazquez, J., Eds.; Geological Society of America: Boulder, CO, USA, 1997; pp. 27–139. [Google Scholar]
- Minnery, G.A.; Rezak, R.; Bright, T.J. Depth Zonation and Growth Form of Crustose Coralline Algae: Flower Garden Banks, Northwestern Gulf of Mexico. In Paleoalgology: Contemporary Research and Applications; Toomey, D.F., Nitecki, M.H., Eds.; Springer: Berlin/Heidelberg, Germany, 1985; pp. 237–247. [Google Scholar]
- Amado-Filho, G.M.; Maneveldt, G.; Manso, R.C.C.; Marins Rosa, B.V.; Pacheco, M.R. Guimarães SMPB 2007 Structure of rhodolith beds from 4 to 55 meters deep along the southern coast of Espírito Santo State, Brazil. Cienc Mar 2007, 33, 399–410. [Google Scholar] [CrossRef]
- Littler, M.M.; Littler, D.S.; Hanisak, M.D. Deep-water rhodolith distribution, productivity and growth history at sites of formation and subsequent degradation. J. Mar. Biol. Ecol. 1991, 150, 163–182. [Google Scholar] [CrossRef]
- Basso, D.; Brusoni, F. The molluscan assemblage of a transitional environment: The Mediterranean maërl from off the Elba Island (Tuscan Archipelago, Tyrrhenian Sea). Boll. Malacol. 2004, 40, 37–45. [Google Scholar]
- Staff, G.; Powell, E. The paleoecological significance of diversity: The effect of time averaging and differential preservation on macroinvertebrate species richness in death assemblages. Palaeogeogr. Palaeoclim. Palaeoecol. 1988, 63, 73–89. [Google Scholar] [CrossRef]
- Kidwell, S.M. Time-averaging in the marine fossil record: Overview of strategies and uncertainties. GEOBIOS 1998, 30, 977–995. [Google Scholar] [CrossRef]
- Basso, D.; Corselli, C. Molluscan Paleoecology in the Reconstruction of Coastal Changes. In Black Sea Flood Question: Changes in Coastline, Climate, and Human Settlement; NATO Science Series IV, Earth and Environmental Science; Yanko-Hombach, V., Gilbert, A.S., Panin, N., Dolukhanov, P.M., Eds.; Kluwer Academic Press: Dordrecht, The Netherlands, 2007; pp. 23–46. [Google Scholar]
- Bracchi, V.A.; Nalin, R.; Basso, D. Paleoecology and dynamics of coralline-dominated facies during a Pleistocene transgressive-regressive cycle (Capo Colonna marine terrace, Southern Italy). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2014, 414, 296–309. [Google Scholar] [CrossRef]
- Bracchi, V.A.; Nalin, R.; Basso, D. Morpho-structural heterogeneity of shallow-water coralligenous in a Pleistocene marine Terrace (Le Castella, Italy). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2016, 454, 101–112. [Google Scholar] [CrossRef]
- Petersen, C.G.J. On the animal communities of the sea bottom in the Skagerrak, the Christiania Fjord and the Danish waters. Rep. Dan. Biol. Stn. 1915, 23, 31–38. [Google Scholar]
- Möbius, K. Die Auster und die Austernwirtschaft. Wiegundt; Hampel and Parey: Berlin, Germany, 1877; pp. 683–751. [Google Scholar]
- Basso, D.; Corselli, C. Community versus biocoenosis in multivariate analysis of benthic molluscan thanatocoenosis. Riv. It. Paleont. Strat. 2002, 108, 153–172. [Google Scholar]
- Pérès, J.M.; Picard, J. Nouveau manuel de bionomie benthique de la Mer Méditerranée. Recl. De Trav. De La Stn. Mar. D’endoume 1964, 31, 1–137. [Google Scholar]
- Pérès, J.M. Major Benthic Assemblages. In Marine Ecology; Kinne, O., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 1982; Volume 1, Part 1; pp. 373–522. [Google Scholar]
- Official Journal of the European Union. Directive 2008/56/EC. Directive of the European Parliament and of the Council of 17 June 2008 Establishing a Framework for Community Action in the Field of Marine Environmental Policy (Marine Strategy Framework Directive); Official Journal of the European Union: Brussels, Belgium, 2008. [Google Scholar]
- Sañé, E.; Ingrassia, M.; Chiocci, F.L.; Argenti, L.; Martorelli, E. Characterization of rhodolith beds-related backscatter facies from the western Pontine Archipelago (Mediterranean Sea). Mar. Environ. Res. 2021, 169, 105339. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.J.; Midgley, N.G. Graphical representation of particle shape using triangular diagrams: An Excel spreadsheet method. Earth Surf. Proc. Landf. 2000, 25, 1473–1477. [Google Scholar] [CrossRef]
- Mediterranean Sea Monitoring and Forecasting Centre. Available online: https://resources.marine.copernicus.eu/product-detail/MEDSEA_MULTIYEAR_PHY_006_004/ (accessed on 15 November 2022).
- Field, J.G.; Clarke, K.R.; Varwick, R.M. A practical strategy for analysing multispecies distribution patterns. Mar. Ecol. Prog. Ser. 1982, 8, 37–52. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; Primer-E Ltd.: Plymouth, UK, 2001. [Google Scholar]
- Basso, D. Considerazioni sulla distribuzione di Acmaea virginea (O.F. Mu¨ller, 1776) (Gastropoda, Acmaeidae) in tanatocenosi tirreniche. Bolettino Malacol. 1992, 28, 177–186. [Google Scholar]
- Dominici, S. Taphonomy and paleoecology of shallow marine macrofossil assemblages in a collisional setting (late Pliocene-early Pleistocene, western Emilia, Italy). Palaios 2001, 16, 336–353. [Google Scholar] [CrossRef]
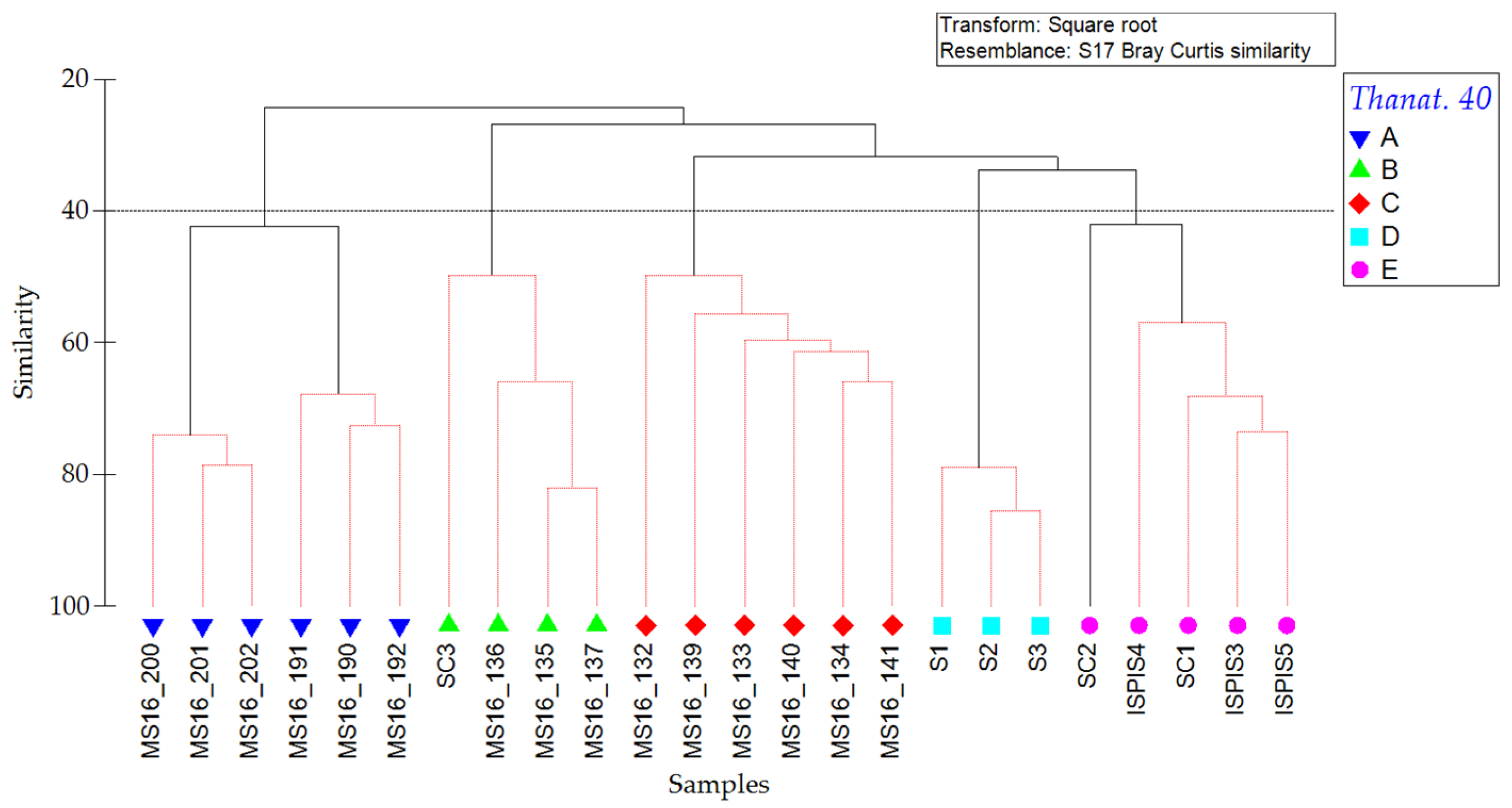


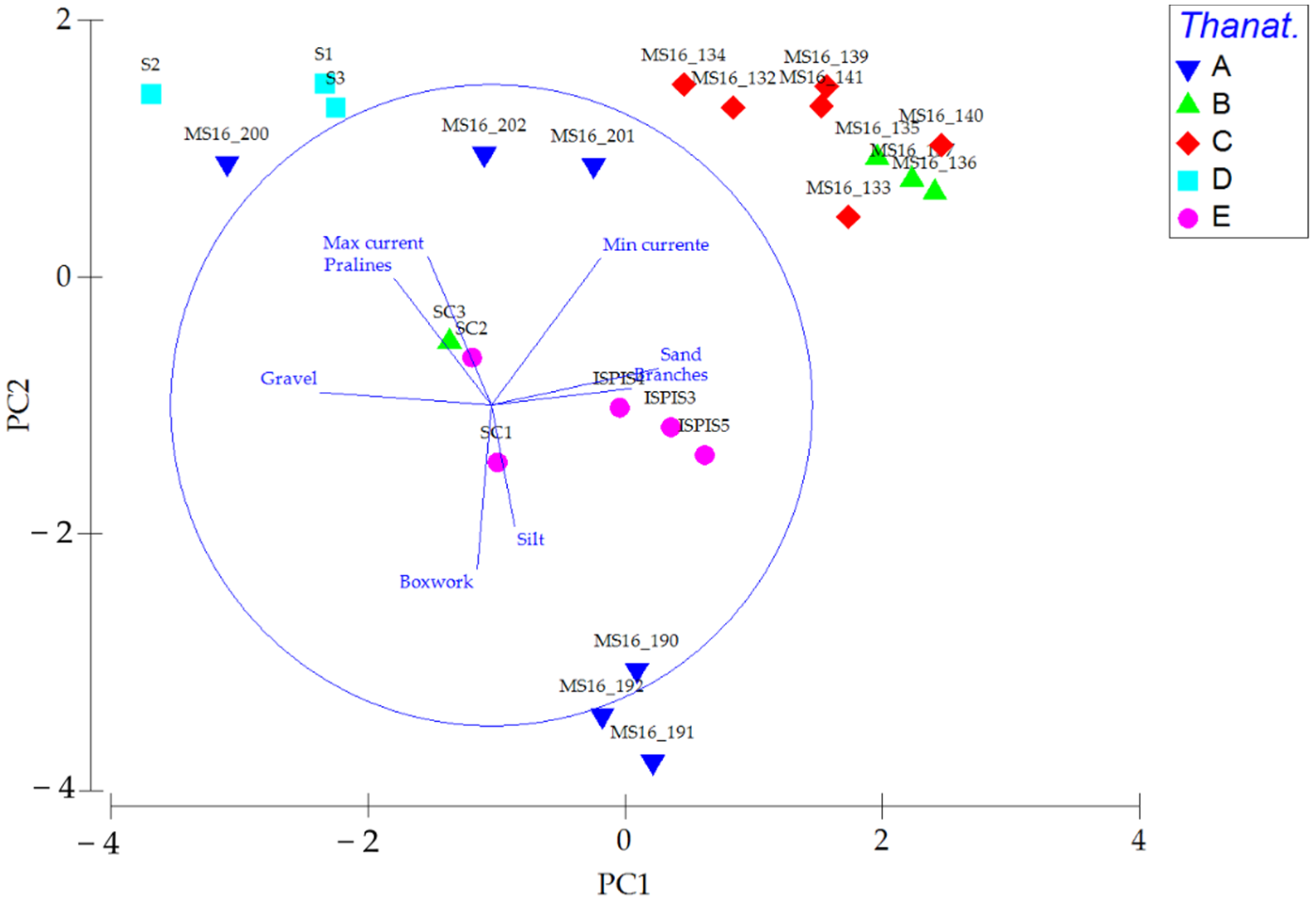
| Station | Site | Replicate | Depth (m) | Latitude | Longitude | Species | Abundance | Shannon Index |
|---|---|---|---|---|---|---|---|---|
| Egadi Islands | Egadi1 | MS16_190 | 103 | 37.9236 | 12.1412 | 38 | 243 | 0.79 |
| MS16_191 | 102.8 | 37.9236 | 12.1412 | 35 | 161 | 0.81 | ||
| MS16_192 | 103.1 | 37.9236 | 12.1412 | 35 | 142 | 0.77 | ||
| Egadi2 | MS16_200 | 86.5 | 37.9500 | 12.1205 | 35 | 162 | 0.85 | |
| MS16_201 | 86.5 | 37.9500 | 12.1205 | 39 | 296 | 0.80 | ||
| MS16_202 | 86.5 | 37.9501 | 12.1205 | 31 | 208 | 0.84 | ||
| Pontine Islands | Pontine1 | MS16_132 | 66.6 | 40.9102 | 12.8693 | 12 | 46 | 0.81 |
| MS16_133 | 66.9 | 40.9102 | 12.8693 | 26 | 153 | 0.74 | ||
| MS16_134 | 66.9 | 40.9102 | 12.8693 | 28 | 124 | 0.74 | ||
| Pontine2 | MS16_135 | 65.9 | 40.9155 | 12.8853 | 15 | 65 | 0.84 | |
| MS16_136 | 66.4 | 40.9155 | 12.8854 | 17 | 214 | 0.71 | ||
| MS16_137 | 66.1 | 40.9155 | 12.8853 | 14 | 96 | 0.86 | ||
| Pontine3 | MS16_139 | 65 | 40.9113 | 12.8828 | 27 | 177 | 0.69 | |
| MS16_140 | 64.9 | 40.9113 | 12.8829 | 24 | 175 | 0.66 | ||
| MS16_141 | 64.8 | 40.9113 | 12.8828 | 31 | 384 | 0.66 | ||
| Sardinia | Serpentara | S1 | 59 | 39.1499 | 9.6127 | 44 | 618 | 0.58 |
| S2 | 59 | 39.1499 | 9.6127 | 45 | 774 | 0.60 | ||
| S3 | 59 | 39.1499 | 9.6127 | 43 | 712 | 0.58 | ||
| Santa Caterina | SC1 | 40 | 39.0865 | 9.4966 | 32 | 57 | 0.95 | |
| SC2 | 40 | 39.0863 | 9.4964 | 20 | 35 | 0.86 | ||
| SC3 | 40 | 39.0861 | 9.4963 | 52 | 682 | 0.64 | ||
| Is Piscadeddus | ISPIS3 | 45 | 36.1120 | 9.4518 | 16 | 70 | 0.77 | |
| ISPIS4 | 45 | 36.1120 | 9.4518 | 22 | 80 | 0.76 | ||
| ISPIS5 | 45 | 36.1074 | 9.4558 | 18 | 57 | 0.79 |
| Thanat. | Species | Sediment | Morphotype | Current |
|---|---|---|---|---|
| A | DC/VP | Mixed | Boxwork-praline | Medium-low |
| B | AP/HP/DC | Sand | Branch-praline | Medium-high |
| C | DE/DL/SGCF | Sand | Praline-branch | Medium-high |
| D | DE/DL/DC/VP | Gravelly sand | Praline | High |
| E | DE/DL/DC/VP/C | Mixed | Praline-branch | Medium |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bracchi, V.A.; Meroni, A.N.; Epis, V.; Basso, D. Mollusk Thanatocoenoses Unravel the Diversity of Heterogeneous Rhodolith Beds (Italy, Tyrrhenian Sea). Diversity 2023, 15, 526. https://doi.org/10.3390/d15040526
Bracchi VA, Meroni AN, Epis V, Basso D. Mollusk Thanatocoenoses Unravel the Diversity of Heterogeneous Rhodolith Beds (Italy, Tyrrhenian Sea). Diversity. 2023; 15(4):526. https://doi.org/10.3390/d15040526
Chicago/Turabian StyleBracchi, Valentina Alice, Agostino Niyonkuru Meroni, Vivien Epis, and Daniela Basso. 2023. "Mollusk Thanatocoenoses Unravel the Diversity of Heterogeneous Rhodolith Beds (Italy, Tyrrhenian Sea)" Diversity 15, no. 4: 526. https://doi.org/10.3390/d15040526
APA StyleBracchi, V. A., Meroni, A. N., Epis, V., & Basso, D. (2023). Mollusk Thanatocoenoses Unravel the Diversity of Heterogeneous Rhodolith Beds (Italy, Tyrrhenian Sea). Diversity, 15(4), 526. https://doi.org/10.3390/d15040526







