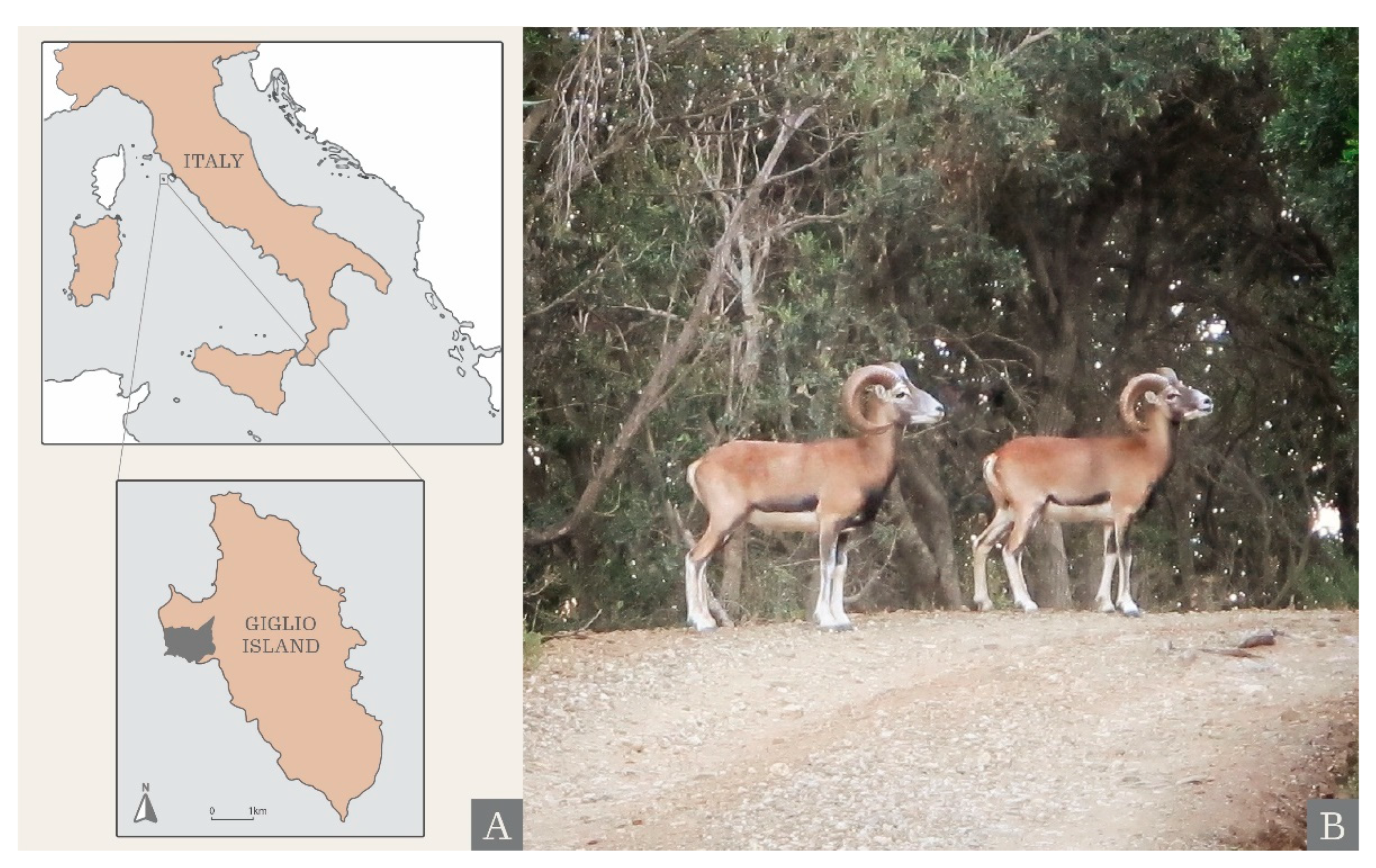
Islands as Time Capsules for Genetic Diversity Conservation: The Case of the Giglio Island Mouflon
Abstract
:1. Introduction
2. Materials and Methods
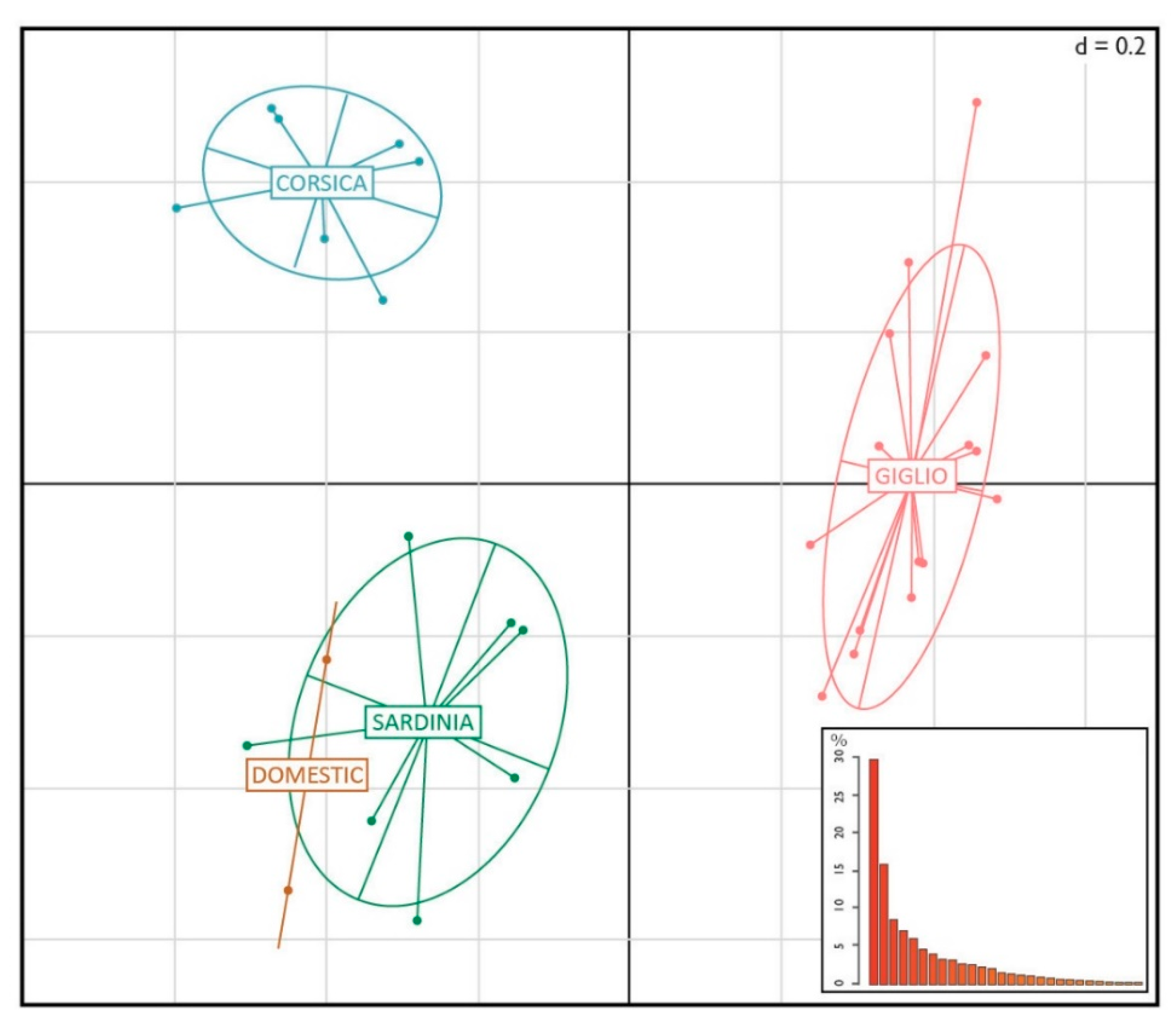
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poplin, F. Origine Du Mouflone de Corse Dans Une Nouvelle Perspective Paléontologique: Par Marronnage. Ann. Genet. Sel. Anim. 1979, 11, 133–143. [Google Scholar] [PubMed]
- Masseti, M. The Prehistoric Diffusion of the Asiatic Mouflon, Ovis Gmelini Blyth, 1841, and the Bezoar Goat, Capra Aegagrus Erxleben, 1777, in the Mediterranean Area beyond Their Natural Distributions. In Proceedings of the Second International Symposium on Mediterranean Mouflon “The Mediterranean Mouflon: Management and Conservation”; Hadjisterkotis, E., Ed.; Game Fund of Cyprus/IUCN Species Survival Commission, Caprinae Specialist Group: Nicosia, Cyprus, 1997. [Google Scholar]
- Barbato, M.; Hailer, F.; Orozco-terWengel, P.; Kijas, J.W.; Mereu, P.; Cabras, P.; Mazza, R.; Pirastru, M.; Bruford, M.W. Genomic Signatures of Adaptive Introgression from European Mouflon into Domestic Sheep. Sci. Rep. 2017, 7, 7623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ciani, E.; Mastrangelo, S.; da Silva, A.; Marroni, F.; Ferenčaković, M.; Ajmone-Marsan, P.; Baird, H.; Barbato, M.; Colli, L.; Delvento, C.; et al. On the Origin of European Sheep as Revealed by the Diversity of the Balkan Breeds and by Optimizing Population-Genetic Analysis Tools. Genet. Sel. Evol. 2020, 52, 25. [Google Scholar] [CrossRef]
- Cugnasse, J.-M. Révision Taxonomique Des Mouflons Des Ȋles Méditerranée. Mammalia 1994, 58, 507–512. [Google Scholar]
- Michel, S.; Ghoddousi, A. Ovis Vignei. The IUCN Red List of Threatened Species 2020; E. T54940655A54940728; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2020. [Google Scholar]
- Perco, F. Ungulati; Carlo Lorenzini editore: Udine, Italy, 1987. [Google Scholar]
- Cassola, F. Il Muflone Di Sardegna: Importanza, Stato Attuale e Problemi Di Conservazione; World Wildlife Fund: Roma, Italy, 1976. [Google Scholar]
- Perco, F. Il Muflone; Edagricole: Bologna, Italy, 1977. [Google Scholar]
- Masserini, V. Diana; Editoriale Olimpia: Firenze, Italy, 1958; pp. 11–14. [Google Scholar]
- Baldacci, U. La riserva di Caccia; Ente Produttori Selvaggina: Roma, Italy, 1975; pp. 1–4. [Google Scholar]
- Masseti, M. Fauna Toscana. Galliformi Non Migratori, Lagormorfi e Artiodattili; Agenzia Regionale per lo Sviluppo e l’Innovazione nel settore Agricolo-forestale (ARSIA): Firenze, Italy, 2003; ISBN 8882950441. [Google Scholar]
- LifeGoGiglio. Available online: https://www.lifegogiglio.eu/en/highlights/actions/ (accessed on 8 July 2022).
- Satta, V.; Mereu, P.; Barbato, M.; Pirastru, M.; Bassu, G.; Manca, L.; Naitana, S.; Leoni, G.G. Genetic Characterization and Implications for Conservation of the Last Autochthonous Mouflon Population in Europe. Sci. Rep. 2021, 11, 14729. [Google Scholar] [CrossRef]
- Hall, T. BioEdit: A User-Friendly Biological Sequence Alignment Editor and Analysis Program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA Sequence Polymorphism Analysis of Large Data Sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef]
- Bandelt, H.J.; Forster, P.; Rohl, A. Median-Joining Networks for Inferring Intraspecific Phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Scali, M.; Vignani, R.; Quintetti, M.; Bigliazzi, J.; Paolucci, E.; Cresti, M. Genetic Traceability of Cinta Senese Pig (Sus scrofa domesticus L.): A Study of the Meat and Processed Products by Microsatellite Markers. Food Nutr. Sci. 2015, 06, 712–726. [Google Scholar] [CrossRef] [Green Version]
- Dray, S.; Dufour, A.-B. The Ade4 Package: Implementing the Duality Diagram for Ecologists. J. Stat. Softw. 2007, 22, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Sanna, D.; Barbato, M.; Hadjisterkotis, E.; Cossu, P.; Decandia, L.; Trova, S.; Pirastru, M.; Leoni, G.G.; Naitana, S.; Francalacci, P.; et al. The First Mitogenome of the Cyprus Mouflon (Ovis Gmelini Ophion): New Insights into the Phylogeny of the Genus Ovis. PLoS ONE 2015, 10, e0144257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drake, D.R.; Mulder, C.P.H.; Towns, D.R.; Daugherty, C.H. The Biology of Insularity: An Introduction. J. Biogeogr. 2002, 29, 563–569. [Google Scholar] [CrossRef] [Green Version]
- Hébert, K.; Millien, V.; Lessard, J.-P.; Masters, J. Source Pool Diversity and Proximity Shape the Compositional Uniqueness of Insular Mammal Assemblages Worldwide. J. Biogeogr. 2021, 48, 2337–2349. [Google Scholar] [CrossRef]
- Fernández-Palacios, J.M.; Kreft, H.; Irl, S.D.H.; Norder, S.; Ah-Peng, C.; Borges, P.A.V.; Burns, K.C.; de Nascimento, L.; Meyer, J.Y.; Montes, E.; et al. Scientists’ Warning—The Outstanding Biodiversity of Islands Is in Peril. Glob. Ecol. Conserv. 2021, 31, e01847. [Google Scholar] [CrossRef]
- Reumer, J.W.F. Shrews (Soricidae) on Islands, with Special Reference to Crocidura Zimmermanni from Crete. In Pleistocene and Holocene Fauna of Crete and its First Settlers; Prehistory Press: Madison, WI, USA, 1996; pp. 173–179. [Google Scholar]
- Masseti, M. Mammals of the Mediterranean Islands: Homogenisation and the Loss of Biodiversity. Mammalia 2009, 73, 169–202. [Google Scholar] [CrossRef]
- Sarà, M.; Vitturi, R. Crocidura Populations (Mammalia, Soricidae) from the Sicilian-Maltese Insular Area. Hystrix Ital. J. Mammal. 1996, 8, 121–132. [Google Scholar] [CrossRef]
- Masseti, M.; Vernesi, C. Historic Zoology of the European Fallow Deer, Dama Dama Dama: Evidence from Biogeography, Archaeology and Genetics. In Deer and People; Oxbow Books: Oxford, UK, 2014; pp. 13–22. [Google Scholar]
- Masseti, M.; Pecchioli, E.; Vernesi, C. Phylogeography of the Last Surviving Populations of Rhodian and Anatolian Fallow Deer (Dama dama dama L., 1758). Biol. J. Linn. Soc. 2008, 93, 835–844. [Google Scholar] [CrossRef] [Green Version]
- Cucchi, T.; Orth, A.; Auffray, J.-C.; Renaud, S.; Fabre, L.; Catalan, J.; Hadjisterkotis, E.; Bonhomme, F.; Vigne, J.-D. A New Endemic Species of the Subgenus Mus (Rodentia, Mammalia) on the Island of Cyprus. Zootaxa 2006, 1241, 1–36. [Google Scholar]
- Mereu, P.; Satta, V.; Frongia, G.N.; Berlinguer, F.; Muzzeddu, M.; Campus, A.; Decandia, L.; Pirastru, M.; Manca, L.; Naitana, S.; et al. The Complete MtDNA Sequence of the Griffon Vulture (Gyps Fulvus): Phylogenetic Analysis and Haplotype Frequency Variations after Restocking in the Sardinian Population. Biol. Conserv. 2017, 214, 195–205. [Google Scholar] [CrossRef]
- Morelli, L.; Useli, A.; Sanna, D.; Barbato, M.; Contu, D.; Pala, M.; Cancedda, M.; Francalacci, P. Mitochondrial DNA Lineages of Italian Giara and Sarcidano Horses. Genet. Mol. Res. 2014, 13, 8241–8257. [Google Scholar] [CrossRef]
- Doan, K.; Zachos, F.E.; Wilkens, B.; Vigne, J.-D.; Piotrowska, N.; Stanković, A.; Jędrzejewska, B.; Stefaniak, K.; Niedziałkowska, M. Phylogeography of the Tyrrhenian Red Deer (Cervus elaphus corsicanus) Resolved Using Ancient DNA of Radiocarbon-Dated Subfossils. Sci. Rep. 2017, 7, 2331. [Google Scholar] [CrossRef] [PubMed]
- Hobohm, C. Nature-Culture Dichotomy and Environmental Consciousness: Do We Fear the Right Things? In Perspectives for Biodiversity and Ecosystems; Springer: Cham, Switzerland, 2021; pp. 17–41. [Google Scholar]
- Masseti, M.; Zuffi, M.A.L. On the Origin of the Population of Asp Viper, Vipera Aspis Hugyi Schinz, 1833, of the Island of Montecristo, in the Northern Tyrrhenian Sea (Tuscan Archipelago, Italy). Herpetol. Bull. 2011, 117. [Google Scholar]
- Somenzi, E.; Senczuk, G.; Ciampolini, R.; Cortellari, M.; Vajana, E.; Tosser-Klopp, G.; Pilla, F.; Ajmone-Marsan, P.; Crepaldi, P.; Colli, L. The SNP-Based Profiling of Montecristo Feral Goat Populations Reveals a History of Isolation, Bottlenecks, and the Effects of Management. Genes 2022, 13, 213. [Google Scholar] [CrossRef] [PubMed]
- Mereu, P.; Pirastru, M.; Barbato, M.; Satta, V.; Hadjisterkotis, E.; Manca, L.; Naitana, S.; Leoni, G.G. Identification of an Ancestral Haplotype in the Mitochondrial Phylogeny of the Ovine Haplogroup B. PeerJ 2019, 7, e7895. [Google Scholar] [CrossRef] [PubMed]
- CASSOLA, F. Management and Conservation of the Sardinian Moufflon (Ovis Musimon). In The Biology and Management of Mountain Ungulates; Croom-Helm: London, UK, 1985; p. 197. [Google Scholar]
- Myers, J.H.; Simberloff, D.; Kuris, A.M.; Carey, J.R. Eradication Revisited: Dealing with Exotic Species. Trends Ecol. Evol. 2000, 15, 316–320. [Google Scholar] [CrossRef]
- Hess, S.C.; Kawakami, B.; Okita, D.; Medeiros, K. A Preliminary Assessment of Mouflon Abundance at the Kahuku Unit of Hawai‘i Volcanoes National Park; U.S. Geological Survey: Reston, VA, USA, 2006. [Google Scholar]
- Bertolino, S.; Sciandra, C.; Bosso, L.; Russo, D.; Lurz, P.W.W.; di Febbraro, M. Spatially Explicit Models as Tools for Implementing Effective Management Strategies for Invasive Alien Mammals. Mammal Rev. 2020, 50, 187–199. [Google Scholar] [CrossRef]
- Sundseth, K. Invasive Alien Species: A European Union Response; Publications Office of the European Union: Luxembourg, 2014; ISBN 9279354868. [Google Scholar]
- Hadjisterkotis, E.; Konstantinou, G.; Sanna, D.; Pirastru, M.; Mereu, P. First Mtdna Sequences and Body Measurements for Rattus Norvegicus from the Mediterranean Island of Cyprus. Life 2020, 10, 136. [Google Scholar] [CrossRef] [PubMed]
- Bani, L.; Orioli, V.; Trasforini, S.; Puzzi, C.M.; Sibilia, A.; Dondina, O.; Tirozzi, P. The Spread of Exotic Fish Species in Italian Rivers and Their Effect on Native Fish Fauna since 1990. Biodiversity 2021, 22, 4–12. [Google Scholar] [CrossRef]
- Shackleton, D. Objectives, Format and Limitations of the Caprinae Survey and Action Plan; IUCN International Union for Conservation of Nature: Gland, Switzerland, 1997; ISBN 2-8317-0353-0. [Google Scholar]
- Kijas, J.W.; Lenstra, J.A.; Hayes, B.J.; Boitard, S.; Porto Neto, L.R.; San Cristobal, M.; Servin, B.; McCulloch, R.; Whan, V.; Gietzen, K.; et al. Genome-Wide Analysis of the World’s Sheep Breeds Reveals High Levels of Historic Mixture and Strong Recent Selection. PLoS Biol. 2012, 10, e1001258. [Google Scholar] [CrossRef] [Green Version]
- Dotsev, A.V.; Kunz, E.; Shakhin, A.V.; Petrov, S.N.; Kostyunina, O.V.; Okhlopkov, I.M.; Deniskova, T.E.; Barbato, M.; Bagirov, V.A.; Medvedev, D.G.; et al. The First Complete Mitochondrial Genomes of Snow Sheep ( Ovis nivicola ) and Thinhorn Sheep (Ovis dalli) and Their Phylogenetic Implications for the Genus Ovis. Mitochondrial DNA Part B 2019, 4, 1332–1333. [Google Scholar] [CrossRef] [Green Version]
- Cao, Y.-H.; Xu, S.-S.; Shen, M.; Chen, Z.-H.; Gao, L.; Lv, F.-H.; Xie, X.-L.; Wang, X.-H.; Yang, H.; Liu, C.-B.; et al. Historical Introgression from Wild Relatives Enhanced Climatic Adaptation and Resistance to Pneumonia in Sheep. Mol. Biol. Evol. 2020, 38, 838–855. [Google Scholar] [CrossRef]
- Lv, F.-H.; Cao, Y.-H.; Liu, G.-J.; Luo, L.-Y.; Lu, R.; Liu, M.-J.; Li, W.-R.; Zhou, P.; Wang, X.-H.; Shen, M.; et al. Whole-Genome Resequencing of Worldwide Wild and Domestic Sheep Elucidates Genetic Diversity, Introgression and Agronomically Important Loci. Mol. Biol. Evol. 2021, 39, msab353. [Google Scholar] [CrossRef] [PubMed]
- Alberto, F.J.; Boyer, F.; Orozco-terWengel, P.; Streeter, I.; Servin, B.; de Villemereuil, P.; Benjelloun, B.; Librado, P.; Biscarini, F.; Colli, L.; et al. Convergent Genomic Signatures of Domestication in Sheep and Goats. Nat. Commun. 2018, 9, 813. [Google Scholar] [CrossRef] [PubMed]
- Cortellari, M.; Barbato, M.; Talenti, A.; Bionda, A.; Carta, A.; Ciampolini, R.; Ciani, E.; Crisà, A.; Frattini, S.; Lasagna, E.; et al. The Climatic and Genetic Heritage of Italian Goat Breeds with Genomic SNP Data. Sci. Rep. 2021, 11, 10986. [Google Scholar] [CrossRef]
- Passamonti, M.M.; Somenzi, E.; Barbato, M.; Chillemi, G.; Colli, L.; Joost, S.; Milanesi, M.; Negrini, R.; Santini, M.; Vajana, E.; et al. The Quest for Genes Involved in Adaptation to Climate Change in Ruminant Livestock. Animals 2021, 11, 2833. [Google Scholar] [CrossRef]
- Somenzi, E.; Ajmone-Marsan, P.; Barbato, M. Identification of Ancestry Informative Marker (AIM) Panels to Assess Hybridisation between Feral and Domestic Sheep. Animals 2020, 10, 582. [Google Scholar] [CrossRef] [Green Version]
- Lorenzini, R.; Cabras, P.; Fanelli, R.; Carboni, G.L. Wildlife Molecular Forensics: Identification of the Sardinian Mouflon Using STR Profiling and the Bayesian Assignment Test. Forensic Sci. Int. Genet. 2011, 5, 345–349. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbato, M.; Masseti, M.; Pirastru, M.; Columbano, N.; Scali, M.; Vignani, R.; Mereu, P. Islands as Time Capsules for Genetic Diversity Conservation: The Case of the Giglio Island Mouflon. Diversity 2022, 14, 609. https://doi.org/10.3390/d14080609
Barbato M, Masseti M, Pirastru M, Columbano N, Scali M, Vignani R, Mereu P. Islands as Time Capsules for Genetic Diversity Conservation: The Case of the Giglio Island Mouflon. Diversity. 2022; 14(8):609. https://doi.org/10.3390/d14080609
Chicago/Turabian StyleBarbato, Mario, Marco Masseti, Monica Pirastru, Nicolò Columbano, Monica Scali, Rita Vignani, and Paolo Mereu. 2022. "Islands as Time Capsules for Genetic Diversity Conservation: The Case of the Giglio Island Mouflon" Diversity 14, no. 8: 609. https://doi.org/10.3390/d14080609
APA StyleBarbato, M., Masseti, M., Pirastru, M., Columbano, N., Scali, M., Vignani, R., & Mereu, P. (2022). Islands as Time Capsules for Genetic Diversity Conservation: The Case of the Giglio Island Mouflon. Diversity, 14(8), 609. https://doi.org/10.3390/d14080609








