Diversity of Land Snail Tribe Helicini (Gastropoda: Stylommatophora: Helicidae): Where Do We Stand after 20 Years of Sequencing Mitochondrial Markers?
Abstract
1. Introduction
2. Materials and Methods
2.1. The Model Group
2.2. Data Acquisition
2.3. Phylogenetic Analyses
2.3.1. Outgroup Selection
2.3.2. Alignment
2.3.3. Maximum Likelihood Analysis of Backbone Phylogeny
2.3.4. Complete Phylogeny of Helicini
2.4. Distribution Maps of Intraspecific Lineages
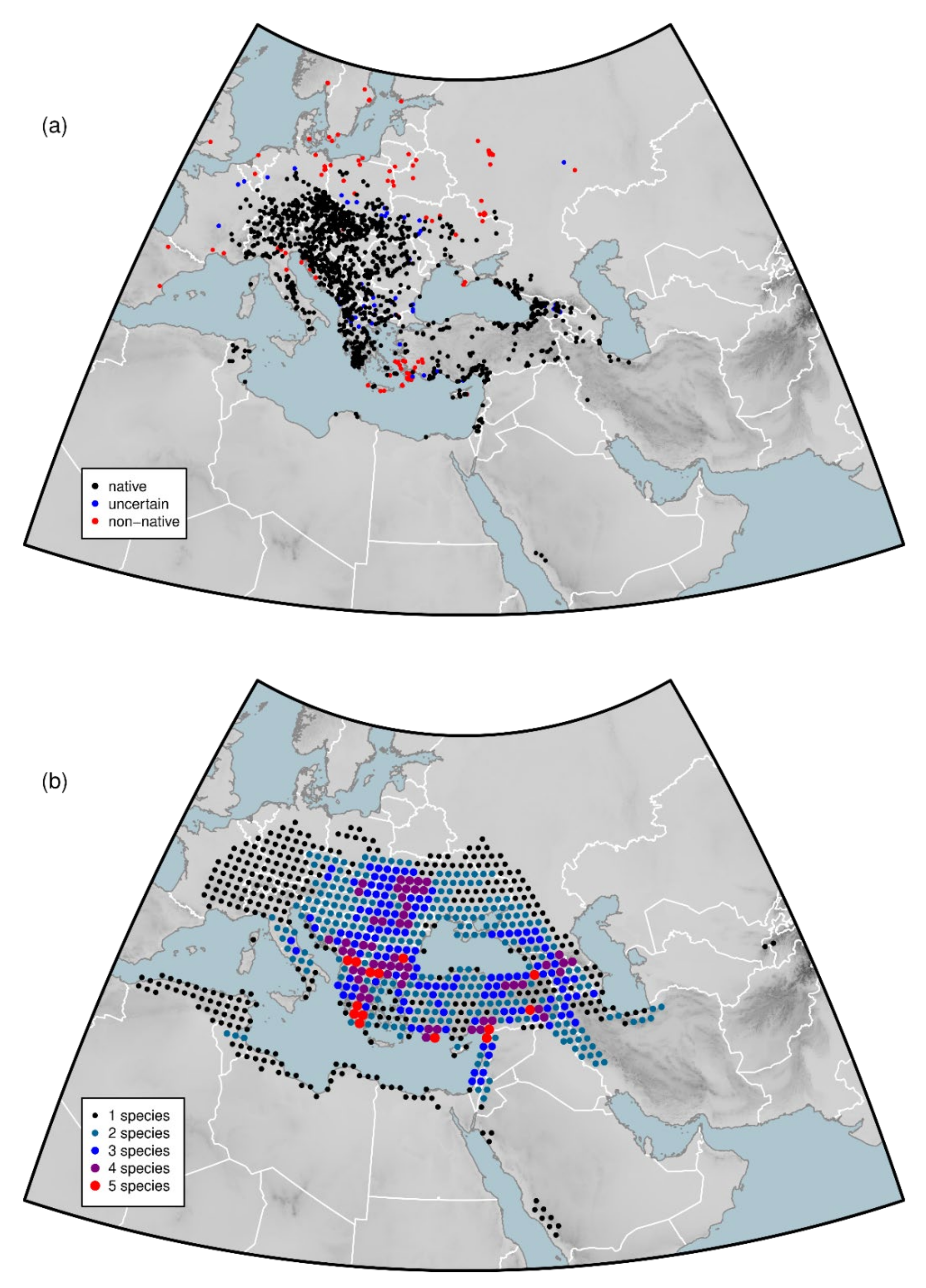
2.5. Non-Native Populations
3. Results and Discussion
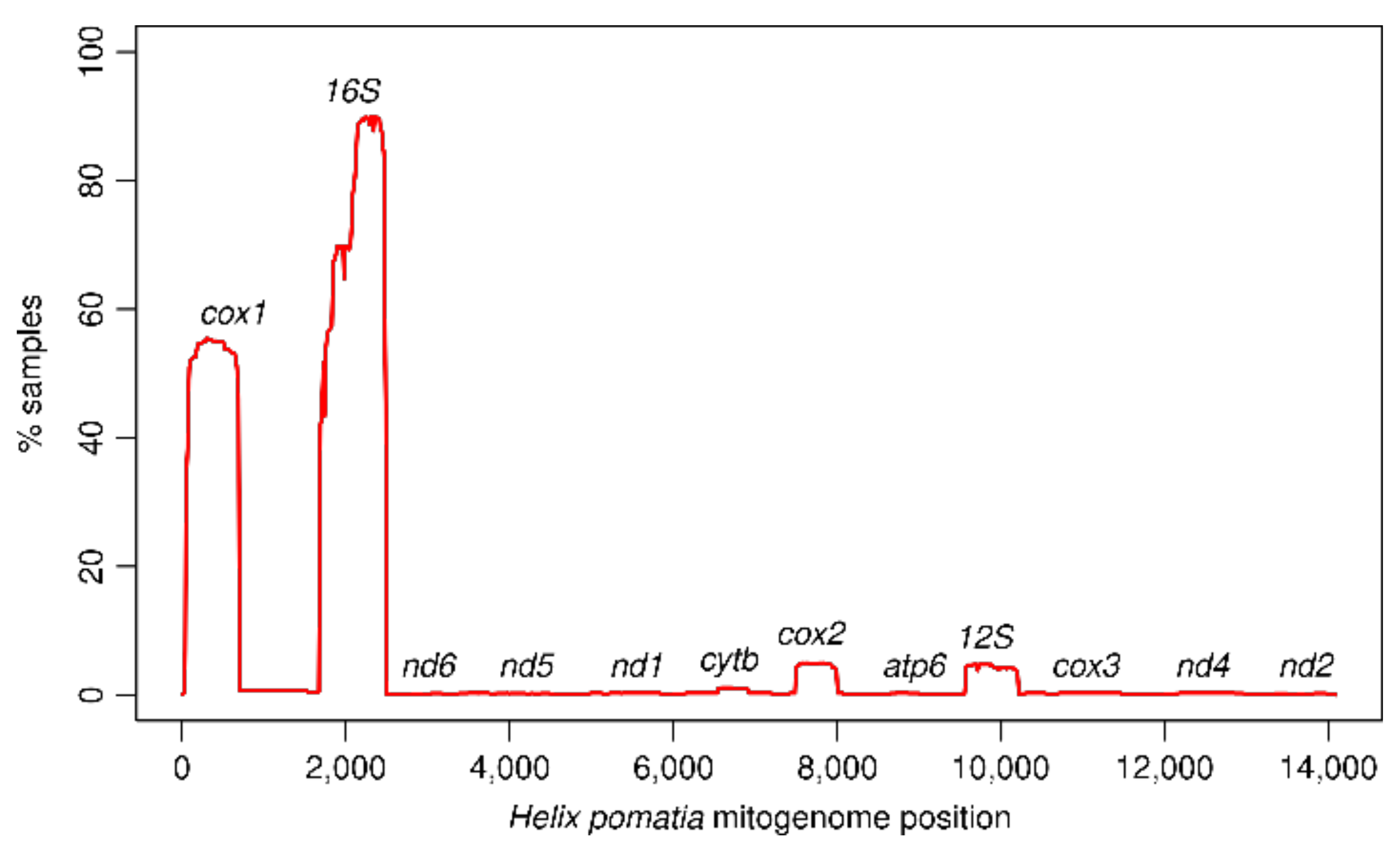
3.1. Mitogenome Representation
3.2. Dataset Coverage
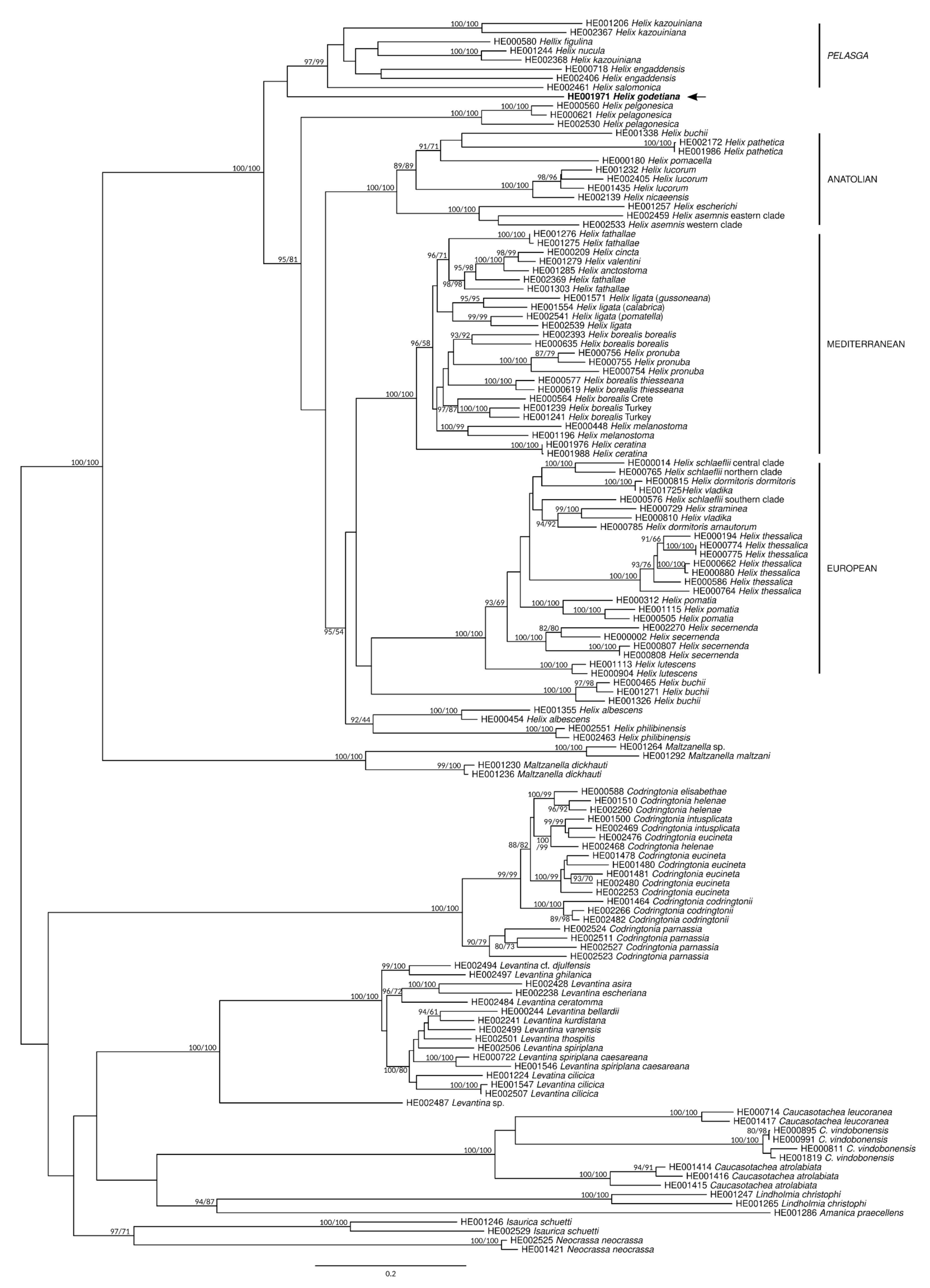
3.3. Phylogeny
3.4. Diversity of Helicini
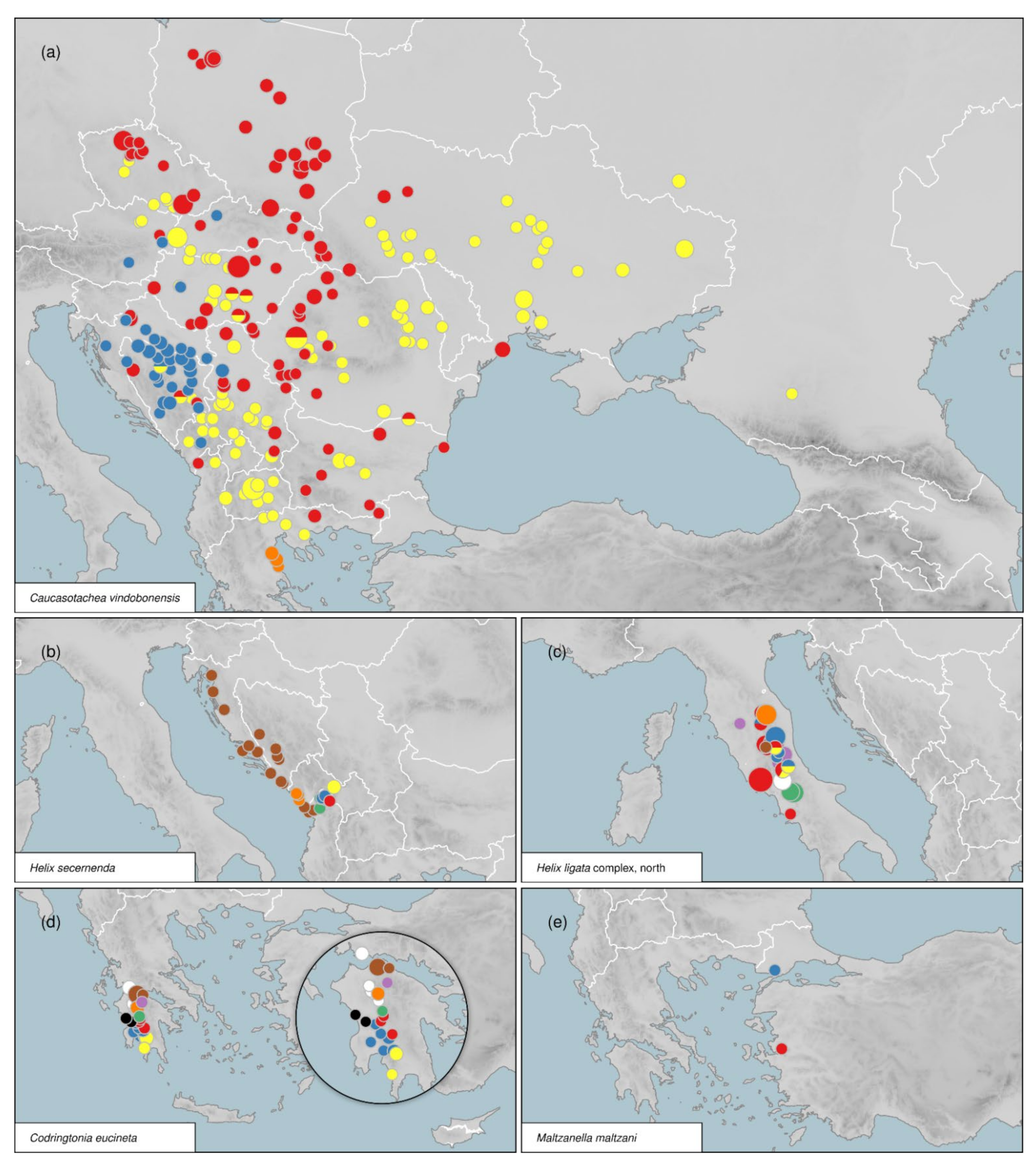
3.4.1. Caucasotachea C. Boettger, 1909
3.4.2. Neocrassa Subai, 2005
3.4.3. Isaurica Kobelt, 1901
3.4.4. Amanica Nordsieck, 2017
3.4.5. Levantina Kobelt, 1871
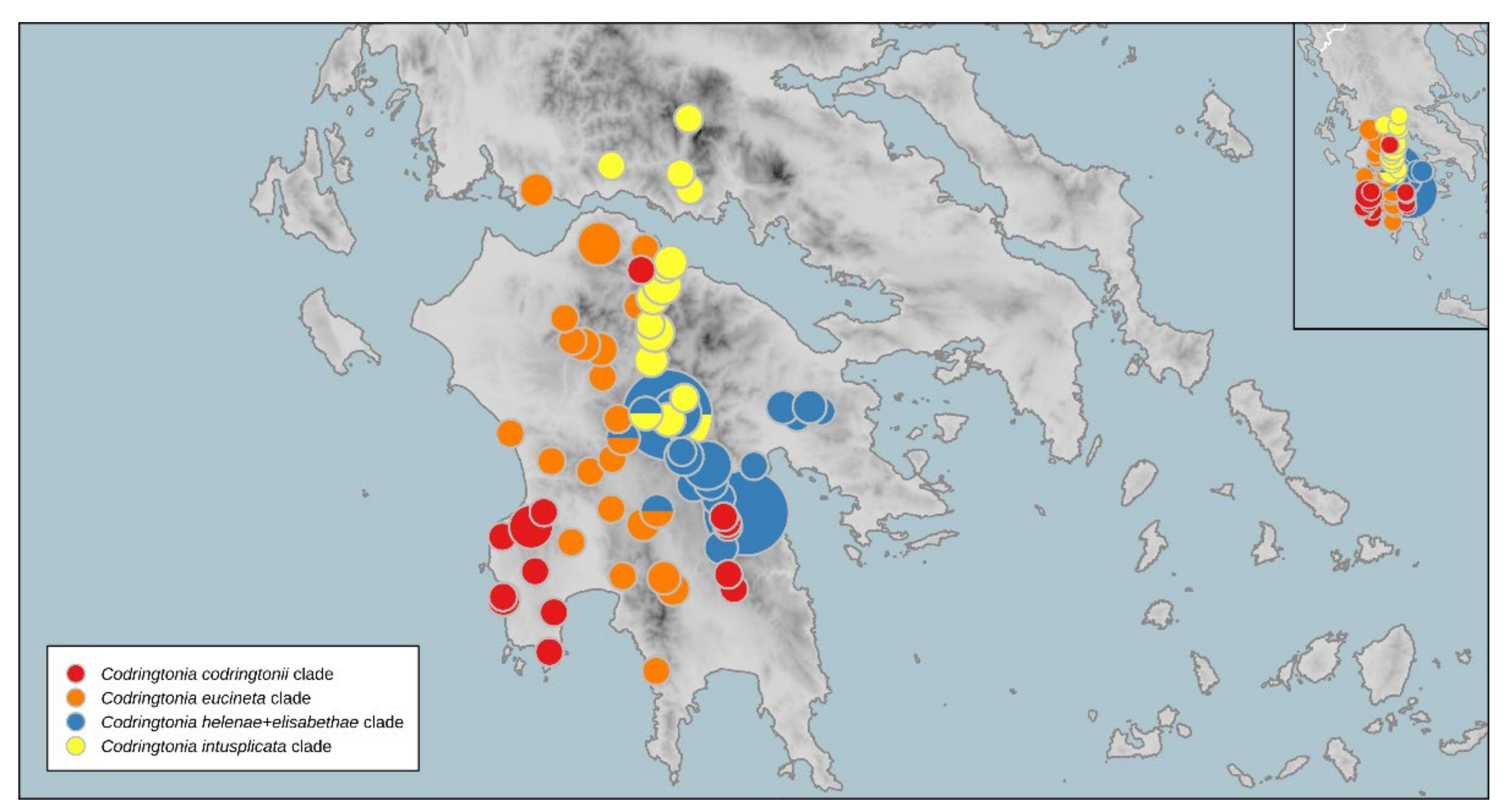
3.4.6. Codringtonia Kobelt, 1898
3.4.7. Lindholmia P. Hesse, 1919
3.4.8. Maltzanella P. Hesse, 1917
3.4.9. Helix Linnaeus, 1758
Subgenus Pelasga P. Hesse, 1908
European Clade
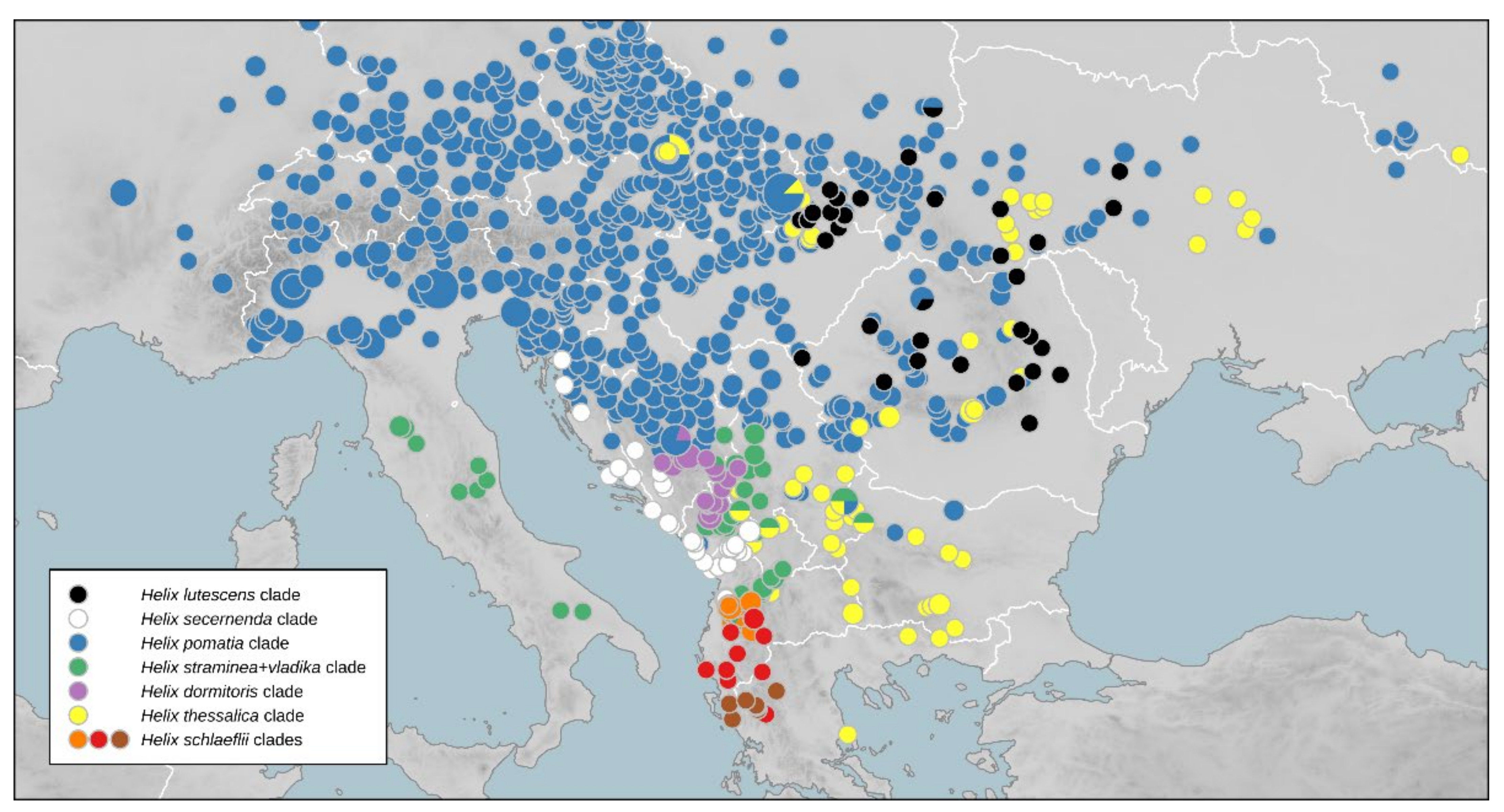
Mediterranean Clade
Anatolian Clade
Species Unassigned to Clades
3.5. Distribution of Intraspecific Diversity
3.6. Outlook
3.6.1. Mitochondrial Data in Helicini Taxonomy
3.6.2. Mitochondrial Data in Phylogeography
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schluter, D.; Pennell, M.W. Speciation gradients and the distribution of biodiversity. Nature 2017, 546, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Miraldo, A.; Li, S.; Borregaard, M.K.; Flórez-Rodríguez, A.; Gopalakrishnan, S.; Rizvanovic, M.; Wang, Z.; Rahbek, C.; Marske, K.A.; Nogués-Bravo, D. An Anthropocene map of genetic diversity. Science 2016, 353, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Carstens, B.C.; Morales, A.E.; Field, K.; Pelletier, T.A. A global analysis of bats using automated comparative phylogeography uncovers a surprising impact of Pleistocene glaciation. J. Biogeogr. 2018, 45, 1795–1805. [Google Scholar] [CrossRef]
- Reid, B.N.; Naro-Maciel, E.; Torres Hahn, A.; FitzSimmons, N.N.; Gehara, M. Geography best explains global patterns of genetic diversity and postglacial co-expansion in marine turtles. Mol. Ecol. 2019, 28, 3358–3370. [Google Scholar] [CrossRef]
- Theodoridis, S.; Fordham, D.A.; Brown, S.C.; Li, S.; Rahbek, C.; Nogues-Bravo, D. Evolutionary history and past climate change shape the distribution of genetic diversity in terrestrial mammals. Nat. Commun. 2020, 11, 2557. [Google Scholar] [CrossRef]
- Lawrence, E.R.; Benavente, J.N.; Matte, J.-M.; Marin, K.; Wells, Z.R.R.; Bernos, T.A.; Krasteva, N.; Habrich, A.; Nessel, G.A.; Koumrouyan, R.A.; et al. Geo-referenced population-specific microsatellite data across American continents. The MacroPopGen Database. Sci. Data 2019, 6, 14. [Google Scholar] [CrossRef]
- Manel, S.; Guerin, P.-E.; Mouillot, D.; Blanchet, S.; Velez, L.; Albouy, C.; Pellissier, L. Global determinants of freshwater and marine fish genetic diversity. Nat. Commun. 2020, 11, 692. [Google Scholar] [CrossRef]
- Paz-Vinas, I.; Jensen, E.L.; Bertola, L.D.; Breed, M.F.; Hand, B.K.; Hunter, M.E.; Kershaw, F.; Leigh, D.M.; Luikart, G.; Mergeay, J.; et al. Macrogenetic studies must not ignore limitations of genetic markers and scale. Ecol. Lett. 2021, 24, 1282–1284. [Google Scholar] [CrossRef]
- Gratton, P.; Marta, S.; Bocksberger, G.; Winter, M.; Trucchi, E.; Kühl, H. A world of sequences: Can we use georeferenced nucleotide databases for a robust automated phylogeography? J. Biogeogr. 2017, 44, 475–486. [Google Scholar] [CrossRef]
- Neubert, E. Revision of Helix Linnaeus, 1758 in its eastern Mediterranean distribution area, and reassignment of Helix godetiana Kobelt, 1878 to Maltzanella Hesse, 1917 (Gastropoda, Pulmonata, Helicidae). Contrib. Nat. Hist. 2014, 26, 1–200. [Google Scholar]
- Neiber, M.T.; Hausdorf, B. Molecular phylogeny reveals the polyphyly of the snail genus Cepaea (Gastropoda: Helicidae). Mol. Phylogenet. Evol. 2015, 93, 143–149. [Google Scholar] [CrossRef]
- Hausdorf, B.; Bamberger, S.; Walther, F. A Sicilian-Cretan biogeographical disjunction in the land snail group Cornu (Gastropoda: Helicidae). Zool. J. Linn. Soc. 2021, 192, 59–74. [Google Scholar] [CrossRef]
- Chueca, L.J.; Madeira, M.J.; Gómez-Moliner, B.J. Biogeography of the land snail genus Allognathus (Helicidae): Middle Miocene colonization of the Balearic Islands. J. Biogeogr. 2015, 42, 1845–1857. [Google Scholar] [CrossRef]
- Neiber, M.T.; Chueca, L.J.; Caro, A.; Teixeira, D.; Schlegel, K.A.; Gómez-Moliner, B.J.; Walther, F.; Glaubrecht, M.; Hausdorf, B. Incorporating palaeogeography into ancestral area estimation can explain the disjunct distribution of land snails in Macaronesia and the Balearic Islands (Helicidae: Allognathini). Mol. Phylogenet. Evol. 2021, 162, 107196. [Google Scholar] [CrossRef]
- Harzhauser, M.; Neubauer, T.A. A review of the land snail faunas of the European Cenozoic—Composition, diversity and turnovers. Earth-Sci. Rev. 2021, 217, 103610. [Google Scholar] [CrossRef]
- Guiller, A.; Coutellec-Vreto, M.A.; Madec, L.; Deunff, J. Evolutionary history of the land snail Helix aspersa in the Western Mediterranean: Preliminary results inferred from mitochondrial DNA sequences. Mol. Ecol. 2001, 10, 81–87. [Google Scholar] [CrossRef]
- Abdulmawjood, A.; Bülte, M. Snail species identification by RFLP-PCR and designing of species-specific oligonucleotide primers. J. Food Sci. 2001, 66, 1287–1293. [Google Scholar] [CrossRef]
- Manganelli, G.; Salomone, N.; Giusti, F. A molecular approach to the phylogenetic relationships of the western palaearctic Helicoidea (Gastropoda: Stylommatophora). Biol. J. Linn. Soc. 2005, 85, 501–512. [Google Scholar] [CrossRef]
- Ketmaier, K.; Fiorentino, V.; Tiedemann, R.; Manganelli, G.; Giusti, F. Morphological and molecular characterization of the Roman snail Helix pomatia with data on the phylogeny of the genus Helix (Pulmonata, Helicidae). In Proceedings of the World Congress of Malacology, Antwerp, Belgium, 15–20 July 2007. [Google Scholar]
- Fiorentino, V.; Manganelli, G.; Giusti, F.; Ketmaier, V. Recent expansion and relic survival: Phylogeography of the land snail genus Helix (Mollusca, Gastropoda) from south to north Europe. Mol. Phylogenet. Evol. 2016, 98, 358–372. [Google Scholar] [CrossRef]
- Kotsakiozi, P.; Parmakelis, A.; Giokas, S.; Papanikolaou, I.; Valakos, E.D. Mitochondrial phylogeny and biogeographic history of the Greek endemic land-snail genus Codringtonia Kobelt 1898 (Gastropoda, Pulmonata, Helicidae). Mol. Phylogenet. Evol. 2012, 62, 681–692. [Google Scholar] [CrossRef]
- Razkin, O.; Gómez-Moliner, B.J.; Prieto, C.E.; Martínez-Ortí, A.; Arrébola, J.R.; Muñoz, B.; Chueca, L.J.; Madeira, M.J. Molecular phylogeny of the western Palaearctic Helicoidea (Gastropoda, Stylommatophora). Mol. Phylogenet. Evol. 2015, 83, 99–117. [Google Scholar] [CrossRef]
- Korábek, O.; Petrusek, A.; Neubert, E.; Juřičková, L. Molecular phylogeny of the genus Helix (Pulmonata: Helicidae). Zool. Scr. 2015, 44, 263–280. [Google Scholar] [CrossRef]
- Neiber, M.T.; Sagorny, C.; Hausdorf, B. Increasing the number of molecular markers resolves the phylogenetic relationship of ‘Cepaea’ vindobonensis (Pfeiffer 1828) with Caucasotachea Boettger 1909 (Gastropoda: Pulmonata: Helicidae). J. Zool. Syst. Evol. Res. 2016, 54, 40–45. [Google Scholar] [CrossRef]
- Korábek, O.; Petrusek, A.; Rovatsos, M. Mitogenome of Helix pomatia and the basal phylogeny of Helicinae (Gastropoda, Stylommatophora, Helicidae). ZooKeys 2019, 827, 19–30. [Google Scholar] [CrossRef]
- Groenenberg, D.S.J.; Duijm, E. The complete mitogenome of the Roman snail Helix pomatia Linnaeus 1758 (Stylommatophora: Helicidae). Mitochondrial DNA Part B 2019, 4, 1494–1495. [Google Scholar] [CrossRef]
- Neiber, M.T.; Korábek, O.; Glaubrecht, M.; Hausdorf, B. A misinterpreted disjunction: The phylogenetic relationships of the Libyan land snail Gyrostomella (Gastropoda: Stylommatophora: Helicidae). Zool. J. Linn. Soc. 2021, in press. [Google Scholar] [CrossRef]
- Groenenberg, D.S.J.; Subai, P.; Gittenberger, E. Systematics of Ariantinae (Gastropoda, Pulmonata, Helicidae), a new approach to an old problem. Contrib. Zool. 2016, 85, 37–65. [Google Scholar] [CrossRef]
- Bouaziz-Yahiatene, H.; Inäbnit, T.; Medjdoub-Bensaad, F.; Colomba, M.S.; Sparacio, I.; Gregorini, A.; Liberto, F.; Neubert, E. Revisited—the species of Tweeting vineyard snails, genus Cantareus Risso, 1826 (Stylommatophora, Helicidae, Helicinae, Otalini). ZooKeys 2019, 876, 1–26. [Google Scholar] [CrossRef]
- Bober, S.; Glaubrecht, M.; Hausdorf, B.; Neiber, M.T. One, two or three? Integrative species delimitation of short-range endemic Hemicycla species (Gastropoda: Helicidae) from the Canary Islands based on morphology, barcoding, AFLP and ddRADseq data. Mol. Phylogenet. Evol. 2021, 161, 107153. [Google Scholar] [CrossRef]
- Saenko, S.V.; Groenenberg, D.S.J.; Davison, A.; Schilthuizen, M. The draft genome sequence of the grove snail Cepaea nemoralis. G3 Genes Genomes Genet. 2021, 11, jkaa071. [Google Scholar] [CrossRef]
- Gittenberger, E.; Piel, W.H.; Groenenberg, D.S.J. The Pleistocene glaciations and the evolutionary history of the polytypic snail species Arianta arbustorum (Gastropoda, Pulmonata, Helicidae). Mol. Phylogenet. Evol. 2004, 30, 64–73. [Google Scholar] [CrossRef]
- Mumladze, L.; Tarkhnishvili, D.; Murtskhvaladze, M. Systematics and evolutionary history of large endemic snails from the Caucasus (Helix buchii and H. goderdziana) (Helicidae). Am. Malacol. Bull. 2013, 31, 225–234. [Google Scholar] [CrossRef]
- Parmakelis, A.; Kotsakiozi, P.; Rand, D. Animal mitochondria, positive selection and cyto-nuclear coevolution: Insights from Pulmonates. PLoS ONE 2013, 8, e61970. [Google Scholar] [CrossRef]
- Cadahía, L.; Harl, J.; Duda, M.; Sattmann, H.; Kruckenhauser, L.; Fehér, Z.; Zopp, L.; Haring, E. New data on the phylogeny of Ariantinae (Pulmonata, Helicidae) and the systematic position of Cylindrus obtusus based on nuclear and mitochondrial DNA marker sequences. J. Zool. Syst. Evol. Res. 2014, 52, 163–169. [Google Scholar] [CrossRef]
- Korábek, O.; Juřičková, L.; Petrusek, A. Resurrecting Helix straminea, a forgotten escargot with trans-Adriatic distribution: First insights into the genetic variation within the genus Helix (Gastropoda: Pulmonata). Zool. J. Linn. Soc. 2014, 171, 72–91. [Google Scholar] [CrossRef]
- Korábek, O.; Juřičková, L.; Petrusek, A. Splitting the Roman snail Helix pomatia Linnaeus, 1758 (Stylommatophora: Helicidae) into two: Redescription of the forgotten Helix thessalica Boettger, 1886. J. Molluscan Stud. 2016, 82, 11–22. [Google Scholar] [CrossRef]
- Korábek, O.; Petrusek, A.; Juřičková, L. Glacial refugia and postglacial spread of an iconic large European land snail, Helix pomatia (Pulmonata: Helicidae). Biol. J. Linn. Soc. 2018, 123, 218–234. [Google Scholar] [CrossRef]
- Korábek, O.; Juřičková, L.; Balashov, I.; Petrusek, A. The contribution of ancient and modern anthropogenic introductions to the colonization of Europe by the land snail Helix lucorum Linnaeus, 1758 (Helicidae). Contrib. Zool. 2018, 87, 61–74. [Google Scholar] [CrossRef]
- Korábek, O.; Juřičková, L.; Petrusek, A. Inferring the sources of postglacial range expansion in two large European land snails. J. Zool. Syst. Evol. Res. 2020, 58, 944–956. [Google Scholar] [CrossRef]
- Korábek, O.; Kosová, T.; Dolejš, P.; Petrusek, A.; Neubert, E.; Juřičková, L. Geographic isolation and human-assisted dispersal in land snails: A Mediterranean story of Helix borealis and its relatives (Gastropoda: Stylommatophora: Helicidae). Zool. J. Linn. Soc. 2021, 193, 1310–1335. [Google Scholar] [CrossRef]
- Korábek, O.; Glaubrecht, M.; Hausdorf, B.; Neiber, M.T. Phylogeny of the land snail Levantina reveals long-distance dispersal in the Middle East. Zool. Scr. in review.
- Dahirel, M.; Olivier, E.; Guiller, A.; Martin, M.-C.; Madec, L.; Ansart, A. Movement propensity and ability correlate with ecological specialization in European land snails: Comparative analysis of a dispersal syndrome. J. Anim. Ecol. 2015, 84, 228–238. [Google Scholar] [CrossRef]
- Giusti, F.; Fiorentino, V.; Manganelli, G. A neotype for Helix cincta Müller, 1774 (Gastropoda, Pulmonata, Helicidae). J. Conchol. 2015, 42, 209–212. [Google Scholar]
- Psonis, N.; Vardinoyannis, K.; Mylonas, M.; Poulakakis, N. Evaluation of the taxonomy of Helix cincta (Muller, 1774) and Helix nucula (Mousson, 1854); insights using mitochondrial DNA sequence data. J. Nat. Hist. 2015, 49, 383–392. [Google Scholar] [CrossRef]
- Jaksch, K.; Eschner, A.; von Rintelen, T.; Haring, E. DNA analysis of molluscs from a museum wet collection: A comparison of different extraction methods. BMC Res. Notes 2016, 9, 348. [Google Scholar] [CrossRef]
- Neiber, M.T.; Sagorny, C.; Sauer, J.; Walther, F.; Hausdorf, B. Phylogeographic analyses reveal Transpontic long distance dispersal in land snails belonging to the Caucasotachea atrolabiata complex (Gastropoda: Helicidae). Mol. Phylogenet. Evol. 2016, 103, 172–183. [Google Scholar] [CrossRef]
- Schmera, D.; Pizá, J.; Reinartz, E.; Ursenbacher, S.; Baur, B. Breeding system, shell size and age at sexual maturity affect sperm length in stylommatophoran gastropods. BMC Evol. Biol. 2016, 16, 89. [Google Scholar] [CrossRef]
- Bouaziz-Yahiatene, H.; Pfarrer, B.; Medjdoub-Bensaad, F.; Neubert, E. Revision of Massylaea Möllendorff, 1898 (Stylommatophora, Helicidae). ZooKeys 2017, 694, 109–133. [Google Scholar] [CrossRef]
- Cesaroni, D.; De Felici, S.; Riccarducci, G.; Ciambotta, M.; Ventura, A.; Bianchi, E.; Sbordoni, V. DNA Barcodes of the animal species occurring in Italy under the European “Habitats Directive” (92/43/EEC): A reference library for the Italian National Biodiversity Network. Biogeogr. J. Integr. Biogeogr. 2017, 32, 5–23. [Google Scholar] [CrossRef]
- Kajtoch, Ł.; Davison, A.; Grindon, A.; Deli, T.; Sramkó, G.; Gwardjan, M.; Kramarenko, S.; Mierzwa-Szymkowiak, D.; Ruta, R.; Tóth, J.P.; et al. Reconstructed historical distribution and phylogeography unravels non-steppic origin of Caucasotachea vindobonensis (Gastropoda: Helicidae). Org. Divers. Evol. 2017, 17, 679–692. [Google Scholar] [CrossRef]
- Sei, M.; Robinson, D.G.; Geneva, A.J.; Rosenberg, G. Doubled helix: Sagdoidea is the overlooked sister group of Helicoidea (Mollusca: Gastropoda: Pulmonata). Biol. J. Linn. Soc. 2017, 122, 697–728. [Google Scholar] [CrossRef]
- Dimzas, D.; Morelli, S.; Traversa, D.; Di Cesare, A.; Van Bourgonie, Y.R.; Breugelmans, K.; Backeljau, T.; Frangipane di Regalbono, A.; Diakou, A. Intermediate gastropod hosts of major feline cardiopulmonary nematodes in an area of wildcat and domestic cat sympatry in Greece. Parasites Vectors 2020, 13, 345. [Google Scholar] [CrossRef]
- Petraccioli, A.; Niero, A.; Carandente, F.; Crovato, P.; de Vico, G.; Odierna, G.; Picariello, O.L.A.; Tardy, E.; Viglietti, S.; Guarino, F.M.; et al. Helix straminea Briganti, 1825 in Italy (Gastropoda: Pulmonata): Taxonomic history, morphology, biology, distribution and phylogeny. Eur. Zool. J. 2021, 88, 390–416. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Chiba, S. Accelerated evolution of land snails Mandarina in the oceanic Bonin Islands: Evidence from mitochondrial DNA sequences. Evolution 1999, 53, 460–471. [Google Scholar] [CrossRef]
- Neiber, M.T.; Razkin, O.; Hausdorf, B. Molecular phylogeny and biogeography of the land snail family Hygromiidae (Gastropoda: Helicoidea). Mol. Phylogenet. Evol. 2017, 111, 169–184. [Google Scholar] [CrossRef]
- Misof, B.; Erpenbeck, D.; Sauer, K.P. Mitochondrial gene fragments suggest paraphyly of the genus Panorpa (Mecoptera, Panorpidae). Mol. Phylogenet. Evol. 2000, 17, 76–84. [Google Scholar] [CrossRef]
- Hugall, A.; Moritz, C.; Moussalli, A.; Stanisic, J. Reconciling paleodistribution models and comparative phylogeography in the Wet Tropics rainforest land snail Gnarosophia bellendenkerensis (Brazier 1875). Proc. Natl. Acad. Sci. USA 2002, 99, 6112–6117. [Google Scholar] [CrossRef]
- Hausdorf, B.; Röpstorf, P.; Riedel, F. Relationships and origin of endemic Lake Baikal gastropods (Caenogastropoda: Rissooidea) based on mitochondrial DNA sequences. Mol. Phylogenet. Evol. 2003, 26, 435–443. [Google Scholar] [CrossRef]
- Merritt, T.J.S.; Shi, L.; Chase, M.C.; Rex, M.A.; Etter, R.J.; Quattro, J.M. Universal cytochrome b primers facilitate intraspecific studies in molluscan taxa. Mol. Mar. Biol. Biotechnol. 1998, 7, 7–11. [Google Scholar]
- Palumbi, S.; Martin, A.; Romano, S.; McMillan, W.O.; Stice, L.; Grabowski, G. The Simple Fool’s Guide to PCR, Version 2; Department of Zoology, University of Hawaii: Honolulu, HI, USA, 1991. [Google Scholar]
- Song, H.; Buhay, J.E.; Whiting, M.F.; Crandall, K.A. Many species in one: DNA barcoding overestimates the number of species when nuclear mitochondrial pseudogenes are coamplified. Proc. Natl. Acad. Sci. USA 2008, 105, 13486–13491. [Google Scholar] [CrossRef] [PubMed]
- Grindon, A.J.; Davison, A. Irish Cepaea nemoralis land snails have a cryptic Franco-Iberian origin that is most easily explained by the movements of mesolithic humans. PLoS ONE 2013, 8, e65792. [Google Scholar] [CrossRef] [PubMed]
- Holyoak, D.T.; Holyoak, G.A.; Gómez-Moliner, B.J.; Chueca, L.J. Phylogeny, species-limits and taxonomic revision of Otalini (Helicidae) from north-west Africa. J. Conchol. 2020, 43, 551–611. [Google Scholar]
- Elejalde, M.A.; Muñoz, B.; Arrébola, J.R.; Gómez-Moliner, B.J. Phylogenetic relationships of Iberus gualtieranus and I. alonensis (Gastropoda: Helicidae) based on partial mitochondrial 16S rRNA and COI gene sequences. J. Molluscan Stud. 2005, 71, 349–355. [Google Scholar] [CrossRef]
- Elejalde, M.A.; Madeira, M.J.; Arrébola, J.R.; Muñoz, B.; Gómez-Moliner, B.J. Molecular phylogeny, taxonomy and evolution of the land snail genus Iberus (Pulmonata: Helicidae). J. Zool. Syst. Evol. Res. 2008, 46, 193–202. [Google Scholar] [CrossRef]
- Greve, C.; Hutterer, R.; Groh, K.; Haase, M.; Misof, B. Evolutionary diversification of the genus Theba (Gastropoda: Helicidae) in space and time: A land snail conquering islands and continents. Mol. Phylogenet. Evol. 2010, 57, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Greve, C.; Gimnich, F.; Hutterer, R.; Misof, B.; Haase, M. Radiating on oceanic islands: Patterns and processes of speciation in the land snail genus Theba (Risso 1826). PLoS ONE 2012, 7, e34339. [Google Scholar] [CrossRef]
- Greve, C.; Haase, M.; Hutterer, R.; Rödder, D.; Ihlow, F.; Misof, B. Snails in the desert: Species diversification of Theba (Gastropoda: Helicidae) along the Atlantic coast of NW Africa. Ecol. Evol. 2017, 7, 5524–5538. [Google Scholar] [CrossRef]
- Neiber, M.T.; Vega-Luz, R.; Vega-Luz, R.; Koenemann, S. Hemicycla (Adiverticula) diegoi (Gastropoda: Pulmonata: Helicidae), a new species from Tenerife, Canary Islands, with a phylogenetic analysis of conchologically similar species in the genus Hemicycla Swainson, 1840. Zootaxa 2011, 2757, 29–46. [Google Scholar] [CrossRef]
- Guiller, A.; Martin, M.-C.; Hiraux, C.; Madec, L. Tracing the invasion of the mediterranean land snail Cornu aspersum aspersum becoming an agricultural and garden pest in areas recently introduced. PLoS ONE 2012, 7, e49674. [Google Scholar] [CrossRef]
- Gaitán-Espitia, J.D.; Nespolo, R.F.; Opazo, J. C The complete mitochondrial genome of the land snail Cornu aspersum (Helicidae: Mollusca): Intra-specific divergence of protein-coding genes and phylogenetic considerations within Euthyneura. PLoS ONE 2013, 8, e67299. [Google Scholar] [CrossRef]
- Martínez-Ortí, A.; Robles, F. El viaje del Prof. Emil, A. Rossmässler en 1853 por España y la localidad tipo de Iberus angustatus (Rossmässler, 1854) (Gastropoda, Helicidae). Anim. Biodivers. Conserv. 2013, 36, 187–194. [Google Scholar] [CrossRef]
- Haase, M.; Greve, C.; Hutterer, R.; Misof, M. Amplified fragment length polymorphisms, the evolution of the land snail genus Theba (Stylommatophora: Helicidae), and an objective approach for relating fossils to internal nodes of a phylogenetic tree using geometric morphometrics. Zool. J. Linn. Soc. 2014, 171, 92–107. [Google Scholar] [CrossRef]
- Colomba, M.S.; Gregorini, A.; Liberto, F.; Reitano, A.; Giglio, S.; Sparacio, I. The genus Erctella Monterosato, 1894: New molecular evidence (Pulmonata Stylommatophora Helicidae). Biodivers. J. 2015, 6, 401–411. [Google Scholar]
- Ali, R.F.; Neiber, M.T.; Walther, F.; Hausdorf, B. Morphological and genetic differentiation of Eremina desertorum (Gastropoda, Pulmonata, Helicidae) in Egypt. Zool. Scr. 2016, 45, 48–61. [Google Scholar] [CrossRef]
- Böckers, A.; Greve, C.; Hutterer, R.; Misof, B.; Haase, M. Testing heterogeneous base composition as potential cause for conflicting phylogenetic signal between mitochondrial and nuclear DNA in the land snail genus Theba Risso 1826 (Gastropoda: Stylommatophora: Helicoidea). Org. Divers. Evol. 2016, 16, 825–846. [Google Scholar] [CrossRef]
- Walther, F.; Neiber, M.T.; Hausdorf, B. Species complex or complex species? Integrative taxonomy of the land snail genus Rossmaessleria (Gastropoda, Helicidae) from Morocco and Gibraltar. Syst. Biodivers. 2016, 14, 394–416. [Google Scholar] [CrossRef]
- Neiber, M.T. On the status of Rossmaessleria scherzeri scherzeri (Zelebor in Pfeiffer & Zelebor, 1867) (Gastropoda: Pulmonata: Helicidae). Zootaxa 2017, 4286, 116–120. [Google Scholar] [CrossRef]
- Holyoak, D.T.; Holyoak, G.A.; Chueca, L.J.; Gómez-Moliner, B.J. Evolution and taxonomy of the populations of Eremina (Gastropoda, Pulmonata, Helicidae) in Morocco. J. Conchol. 2018, 43, 17–57. [Google Scholar]
- Kneubühler, J.; Hutterer, R.; Pfarrer, B.; Neubert, E. Anatomical and phylogenetic investigation of the genera Alabastrina Kobelt, 1904, Siretia Pallary, 1926, and Otala Schumacher, 1817 (Stylommatophora, Helicidae). ZooKeys 2019, 843, 1–37. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. Molecular Evolutionary Genetics Analysis Version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordjik, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef]
- Smirnov, V.; Warnow, T. Phylogeny estimation given sequence length heterogeneity. Syst. Biol. 2021, 70, 268–282. [Google Scholar] [CrossRef]
- Upham, N.S.; Esselstyn, J.A.; Jetz, W. Inferring the mammal tree: Species-level sets of phylogenies for questions in ecology, evolution, and conservation. PLoS Biol. 2019, 17, e3000494. [Google Scholar] [CrossRef]
- Bouckaert, R.; Vaughan, T.G.; Barido-Sottani, J.; Duchêne, S.; Fourment, M.; Gavryushkina, A.; Heled, J.; Jones, G.; Kühnert, D.; De Maio, N.; et al. BEAST 2.5: An advanced software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 2019, 15, e1006650. [Google Scholar] [CrossRef]
- Ritchie, A.M.; Lo, N.; Ho, S.Y.W. The impact of the tree prior on molecular dating of data sets containing a mixture of inter- and intraspecies sampling. Syst. Biol. 2017, 66, 413–425. [Google Scholar] [CrossRef]
- Groenenberg, D.S.J.; Pirovano, W.; Gittenberger, E.; Schilthuizen, M. The complete mitogenome of Cylindrus obtusus (Helicidae, Ariantinae) using Illumina next generation sequencing. BMC Genom. 2012, 13, 114. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Yang, S.-P.; Lin, J.-H.; Zhang, M.-Z.; Zhou, W.-C. The mitochondrial genome of the land snail Theba pisana (Müller, 1774) (Stylommatophora: Helicidae): The first complete sequence in the genus Theba. Mitochondrial DNA Part B Resour. 2018, 3, 798–800. [Google Scholar] [CrossRef] [PubMed]
- Neubert, E.; Bank, R. Notes on the species of Caucasotachea, C. Boettger 1909 and Lindholmia, P. Hesse 1919, with annotations to the Helicidae. Arch. Molluskenkd. 2006, 135, 101–132. [Google Scholar] [CrossRef]
- Subai, P. Revision der Gattung Codringtonia Kobelt 1898 [Revision of the genus Codringtonia Kobelt 1898] (Gastropoda: Pulmonata: Helicidae: Helicinae). Arch. Molluskenkd. 2005, 134, 56–119. [Google Scholar]
- Subai, P. Vergleich der mit Levantina verwandten großen Heliciden, sowie Revision der Gattung Isaurica (Kobelt). Arch. Molluskenkd. 1994, 123, 49–87. [Google Scholar] [CrossRef]
- Nordsieck, H. Amanica n. gen., a new genus of Helicidae (Helicini) from Turkey. In Pulmonata, Stylommatophora, Helicoidea: Systematics with Comments; Nordsieck, H., Ed.; ConchBooks: Harxheim, Germany, 2017; pp. 82–89. [Google Scholar]
- Pfeiffer, K.L.; Wächtler, W. Über Codringtonia and Isaurica. Arch. Molluskenkd. 1939, 71, 57–74. [Google Scholar]
- Glaubrecht, M. Systematics and biogeography of Assyriella rechingeri (Gastropoda, Pulmonata, Helicidae) on Kárpathos, Greece. Verh. Nat. Ver. Hambg. 1994, 34, 373–384. [Google Scholar]
- Grano, M.; Cattaneo, C. Rediscovery of Assyriella rechingeri (Fuchs et Käufel, 1936) (Gastropoda Helicidae) in Karpathos Island (Dodecanese, Greece). Biodivers. J. 2021, 12, 467–474. [Google Scholar] [CrossRef]
- Schütt, H.; Subai, P. Revision der Gattung Assyriella, P. Hesse 1908 (Gastropoda: Pulmonata: Helicidae: Helicinae). Arch. Molluskenkd. 1996, 125, 117–161. [Google Scholar] [CrossRef]
- Dohrn, H. Ueber einige centralasiatische Landschnecken. Jahrbücher Dtsch. Malakozool. Ges. 1882, 9, 115–120. [Google Scholar]
- Neubert, E. Annotated checklist of the terrestrial and freshwater molluscs of the Arabian Peninsula with descriptions of new species. Fauna Arab. 1998, 17, 333–461. [Google Scholar]
- Schütt, H. Über die Helix-Untergattung Maltzanella Hesse 1917. Arch. Molluskenkd. 1976, 107, 63–71. [Google Scholar]
- Wade, C.M.; Hudelot, C.; Davison, A.; Naggs, F.; Mordan, P.B. Molecular phylogeny of the helicoid land snails (Pulmonata: Stylommatophora: Helicoidea), with special emphasis on the Camaenidae. J. Molluscan Stud. 2007, 73, 411–415. [Google Scholar] [CrossRef]
- Schileyko, A. Nazemnye mollyuski nadsemeystva Helicoidea. In Fauna SSSR, Mollyuski; Skarlato, O.A., Ed.; Nauka: Leningrad, Russia, 1978; Volume III, pp. 1–384. [Google Scholar]
- Giusti, F.; Manganelli, G.; Schembri, P.J. The non-marine molluscs of the Maltese Islands. Monogr. Mus. Reg. Sci. Nat. 1995, 15, 1–607. [Google Scholar]
- Heller, J.; Ittiel, H. Natural history and population dynamics of the land snail Helix texta in Israel (Pulmonata: Helicidae). J. Molluscan Stud. 1990, 56, 189–204. [Google Scholar] [CrossRef]
- Heller, J. Land Snails of the Land of Israel. Natural History and Field Guide; Pensoft: Sofia, Bulgaria; Moscow, Russia, 2009. [Google Scholar]
- Ložek, V. Quartärmollusken der Tschechoslowakei. Rozpr. Ústředního Ústavu Geol. 1964, 31, 1–374. [Google Scholar]
- Popiuk, Y.; Ridush, B.; Solovey, T. Middle and Late Pleistocene terrestrial snails from the Middle Dniester area, Ukraine (based on Mykola Kunytsia’s collections). Geol. Q. 2021, 65, 6. [Google Scholar] [CrossRef]
- Blume, W. Einige mazedonische Schnecken. Arch. Molluskenkd. 1920, 52, 89–92. [Google Scholar]
- Knipper, H. Systematische, anatomische, ökologische und tiergeographische Studien an südosteuropäischen Heliciden (Moll. Pulm.). Z. Wiss. Zool. Abt. B Archiv. Nat. Neue Folge. 1939, 8, 327–517. [Google Scholar]
- Urbański, J. Beiträge zur Kenntnis balkanischer Vertreter des Genus Helix, L. I (Systematische, zoogeographische und ökologische Studien über die Mollusken der Balkan-Halbinsel X). Bull. Société Amis Sci. Lett. Poznań Série D 1970, 11, 63–79. [Google Scholar]
- Urbański, J. Beiträge zur Kenntnis balkanischer Vertreter des Genus Helix, L. II. (Systematische, zoogeographische und ökologische Studien über die Mollusken der Balkan-Halbinsel XIV). Bull. Société Amis Sci. Lett. Poznań Série D 1975, 15, 99–107. [Google Scholar]
- Phuong, M.A.; Bi, K.; Moritz, C. Range instability leads to cytonuclear discordance in a morphologically cryptic ground squirrel species complex. Mol. Ecol. 2017, 26, 4743–4755. [Google Scholar] [CrossRef]
- Dufresnes, C.; Nicieza, A.G.; Litvinchuk, S.N.; Rodrigues, N.; Jeffries, D.L.; Vences, M.; Perrin, N.; Martínez-Solano, Í. Are glacial refugia hotspots of speciation and cytonuclear discordances? Answers from the genomic phylogeography of Spanish common frogs. Mol. Ecol. 2020, 29, 986–1000. [Google Scholar] [CrossRef]
- Bouchet, P.; Ripken, T.; Recorbet, B. Redécouverte de l’escargot de Corse Helix ceratina au bord de l’extinction. Rev. Écologie 1997, 52, 97–111. [Google Scholar]
- Giusti, F. Notulae malacologicae XVI. I molluschi terrestri e di acqua dolce viventi sul massiccio dei Monti Reatini (Appennino Centrale). Lav. Soc. Ital. Biogeogr. Nuova Ser. 1971, 11, 423–574, pl. 1–7. [Google Scholar] [CrossRef]
- Welter-Schultes, F.W. European Non-Marine Molluscs, a Guide for Species Identification; Planet Poster Editions: Göttingen, Germany, 2012. [Google Scholar]
- Douka, K.; Jacobs, Z.; Lane, C.; Grün, R.; Farr, L.; Hunt, C.; Inglis, R.H.; Reynolds, T.; Albert, P.; Aubert, M.; et al. The chronostratigraphy of the Haua Fteah cave (Cyrenaica, northeast Libya). J. Hum. Evol. 2014, 66, 39–63. [Google Scholar] [CrossRef]
- Schütt, H.; Subai, P. Beitrag zur Kenntnis von Tacheopsis nicaeensis (FÉRUSSAC) (Gastropoda Pulmonata: Helicidae: Helicinae). Mitt. Dtsch. Malakozool. Ges. 1989, 44/45, 31–34. [Google Scholar]
- Mumladze, L. Sympatry without co-occurrence: Exploring the pattern of distribution of two Helix species in Georgia using an ecological niche modelling approach. J. Molluscan Stud. 2014, 80, 249–255. [Google Scholar] [CrossRef]
- Grossu, A.V. Gastropoda Romaniae 4, Ordo Stylommatophora, Suprafam.: Arionacea, Zonitacea, Ariophantacea și Helicacea; Editura Litera: Bucuresti, Romania, 1983. [Google Scholar]
- Molak, M.; Ho, S.Y.W. Prolonged decay of molecular rate estimates for metazoan mitochondrial DNA. PeerJ 2015, 3, e821. [Google Scholar] [CrossRef]
- Ho, S.Y.W.; Duchêne, S.; Molak, M.; Shapiro, B. Time-dependent estimates of molecular evolutionary rates: Evidence and causes. Mol. Ecol. 2015, 24, 6007–6012. [Google Scholar] [CrossRef]
- Kojumdgieva, E. Fosilite na Balgaria, VII, Sarmat; Bulgarian Academy of Science: Sofia, Bulgaria, 1969. [Google Scholar]
- Dufresnes, C.; Mazepa, G.; Jablonski, D.; Caliari Oliveira, R.; Wenseleers, T.; Shabanov, D.A.; Auer, M.; Ernst, R.; Koch, C.; Ramírez-Chaves, H.E.; et al. Fifteen shades of green: The evolution of Bufotes toads revisited. Mol. Phylogenet. Evol. 2019, 141, 106615. [Google Scholar] [CrossRef] [PubMed]
- Heller, J. Distribution, hybridization and variation in the Israeli landsnail Levantina (Pulmonata: Helicidae). Zool. J. Linn. Soc. 1979, 67, 115–148. [Google Scholar] [CrossRef]
| Forward Primer | Reverse Primer | Notes on Use | References |
|---|---|---|---|
| cox1 | |||
| LCO1490: 5′-GGTCAACAAATCATAAAGATATTGG-3′ | HC02198: 5′-TAAACTTCAGGGTGACCAAAAAATCA-3′ | most samples | [55] * |
| p-cox1-f: 5′-TCGGGACGGGTCTCTCTTTG-3′ | HC02198: 5′-TAAACTTCAGGGTGACCAAAAAATCA-3′ | a few Helix pomatia samples | forward: this study |
| COI-vind-f: 5′-TACTGTTTGGTGTTTGATGTGG-3′ | COI-vind-r: 5′-ACAACATAG TAATTGCCCCAGC-3′ | Caucasotachea vindobonensis | [40] |
| LCO1490: 5′-GGTCAACAAATCATAAAGATATTGG-3′ | H2198-Alb: 5′-TATACTTCAGGATGACCAAAAAATCA-3′ | a few samples | reverse [32] |
| g-COX1-f: 5′-TGGGACAGGTTTATCGTTACTG-3′ | H2198-Alb: 5′-TATACTTCAGGATGACCAAAAAATCA-3′ | 3′ part of Helix godetiana sequence | forward: this study |
| COI_OK1F: 5′-TTGTWACTGCYCAYGCRTTTG-3′ | HC02198: 5′-TAAACTTCAGGGTGACCAAAAAATCA-3′ | used for some samples that failed to amplify with the standard primer pair, mostly from museum ethanol material | forward [42] |
| LCO1490: 5′-GGTCAACAAATCATAAAGATATTGG-3′ | COI_OK3R: 5′-AAAGGTGGRTAAACAGTYCANCC-3′ | reverse [42] | |
| 16S rRNA | |||
| 16Scs1: 5′-AAACATACCTTTTGCATAATGG-3′ | 16Scs2: 5′-AGAAACTGACCTGGCTTACG-3′ | most samples | both [56] ** |
| Scs1-p: 5′-GAATTACCTTTTGCATAATGGA-3′ | Scs2-p: 5′-GAAACTGACCTGGCTTACG-3′ | a couple of Helix pomatia samples | this study |
| 16Scs1: 5′-AAACATACCTTTTGCATAATGG-3′ | 16S_MN3R: 5′-GCTACCTTTGCACAGTCAGWG-3′ | the 16S sequence in two parts, mainly used for older or improperly preserved samples | reverse [57] |
| 16S-F: 5′-CGGCCGCCTGTTTATCAAAAACAT-3′ | 16S-R: 5′-GGAGCTCCGGTTTGAACTCAGATC-3′ | [58] *** | |
| 16S-F: 5′-CGGCCGCCTGTTTATCAAAAACAT-3′ | 16S-Helcentr-R: 5′-AAGYTTCTAGGGTCTTCTCGTCT-3′ | the 3′ half of the 16S sequence in three parts, used for particularly fragmented templates | reverse [23] |
| 16S-Helcentr-F: 5′-AGACGAGAAGACCCTAGAARCTT-3′ | 16S-R: 5′-GGAGCTCCGGTTTGAACTCAGATC-3′ | forward [23] | |
| 16S-Helinter-F: 5′-GTACYYTGACTGTGCAAAGGT-3′ | 16S-Helinter-R: 5′-CTAGTCCAACATCGAGGTCAC-3′ | [23] | |
| 16S-F: 5′-CGGCCGCCTGTTTATCAAAAACAT-3′ | g-centr-R: 5′-AGACAGTTACCGCCCATGCT-3′ | the 3′ half of the 16S sequence in three parts, for an old sample of Helix godetiana | reverse: this study |
| g-centr-F: 5′-AGCATGGGCGGTAACTGTCT-3′ | 16S-R: 5′-GGAGCTCCGGTTTGAACTCAGATC-3′ | forward: this study | |
| g-inter-F: 5′-TGGCCCATGATTGGGGTCTA-3′ | 16S-Helinter-R: 5′-CTAGTCCAACATCGAGGTCAC-3′ | this study | |
| cox2 | |||
| COII-F: 5′-AAATAATGCTATTTCATGAYCAYGC-3′ | COII-R: 5′-GCTCCGCAAATCTCTGARCAYTG-3′ | [59] | |
| 12S rRNA | |||
| 12SGast_fwd2: 5′-AGTGACGGGCGATTTGT-3′ | 12SGast_rev3: 5′-TAAGCTGTTGGGCTCATAAC-3′ | most samples | [35] |
| 12Sam: 5′-AACTAGGATTAGATACCCCAYTAT-3′ | 12bm: 5′-CGAGAGTGACGGGCGATTTGT-3′ | a few samples, shorter amplicon than with the other pair | [60] |
| cytb | |||
| UCYTB151F: 5′-TGTGGRGCNACYGTWATYACTAA-3′ | UCYTB270R: 5′-AANAGGAARTAYCAYTCNGGYTG-3′ | [61] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korábek, O.; Juřičková, L.; Petrusek, A. Diversity of Land Snail Tribe Helicini (Gastropoda: Stylommatophora: Helicidae): Where Do We Stand after 20 Years of Sequencing Mitochondrial Markers? Diversity 2022, 14, 24. https://doi.org/10.3390/d14010024
Korábek O, Juřičková L, Petrusek A. Diversity of Land Snail Tribe Helicini (Gastropoda: Stylommatophora: Helicidae): Where Do We Stand after 20 Years of Sequencing Mitochondrial Markers? Diversity. 2022; 14(1):24. https://doi.org/10.3390/d14010024
Chicago/Turabian StyleKorábek, Ondřej, Lucie Juřičková, and Adam Petrusek. 2022. "Diversity of Land Snail Tribe Helicini (Gastropoda: Stylommatophora: Helicidae): Where Do We Stand after 20 Years of Sequencing Mitochondrial Markers?" Diversity 14, no. 1: 24. https://doi.org/10.3390/d14010024
APA StyleKorábek, O., Juřičková, L., & Petrusek, A. (2022). Diversity of Land Snail Tribe Helicini (Gastropoda: Stylommatophora: Helicidae): Where Do We Stand after 20 Years of Sequencing Mitochondrial Markers? Diversity, 14(1), 24. https://doi.org/10.3390/d14010024







