Crassoascoma gen. nov. (Lentitheciaceae, Pleosporales): Unrevealing Microfungi from the Qinghai-Tibet Plateau in China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection, Isolation and Morphological Examination
2.2. DNA Extration, PCR Amplification and Sequencing
2.3. Sequence Alignment and Phylogenetic Analysis
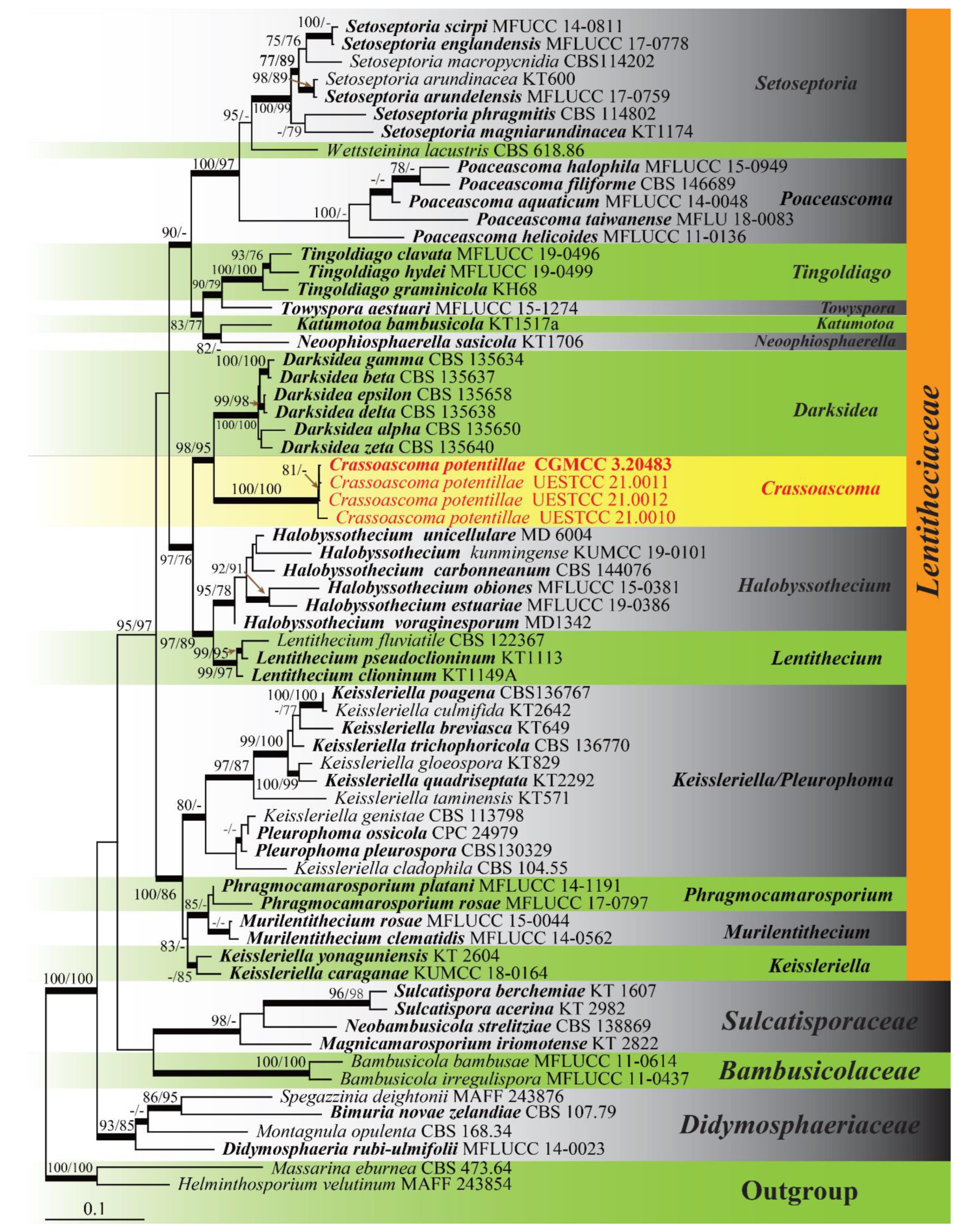
3. Results
3.1. Phylogenetic Analyses
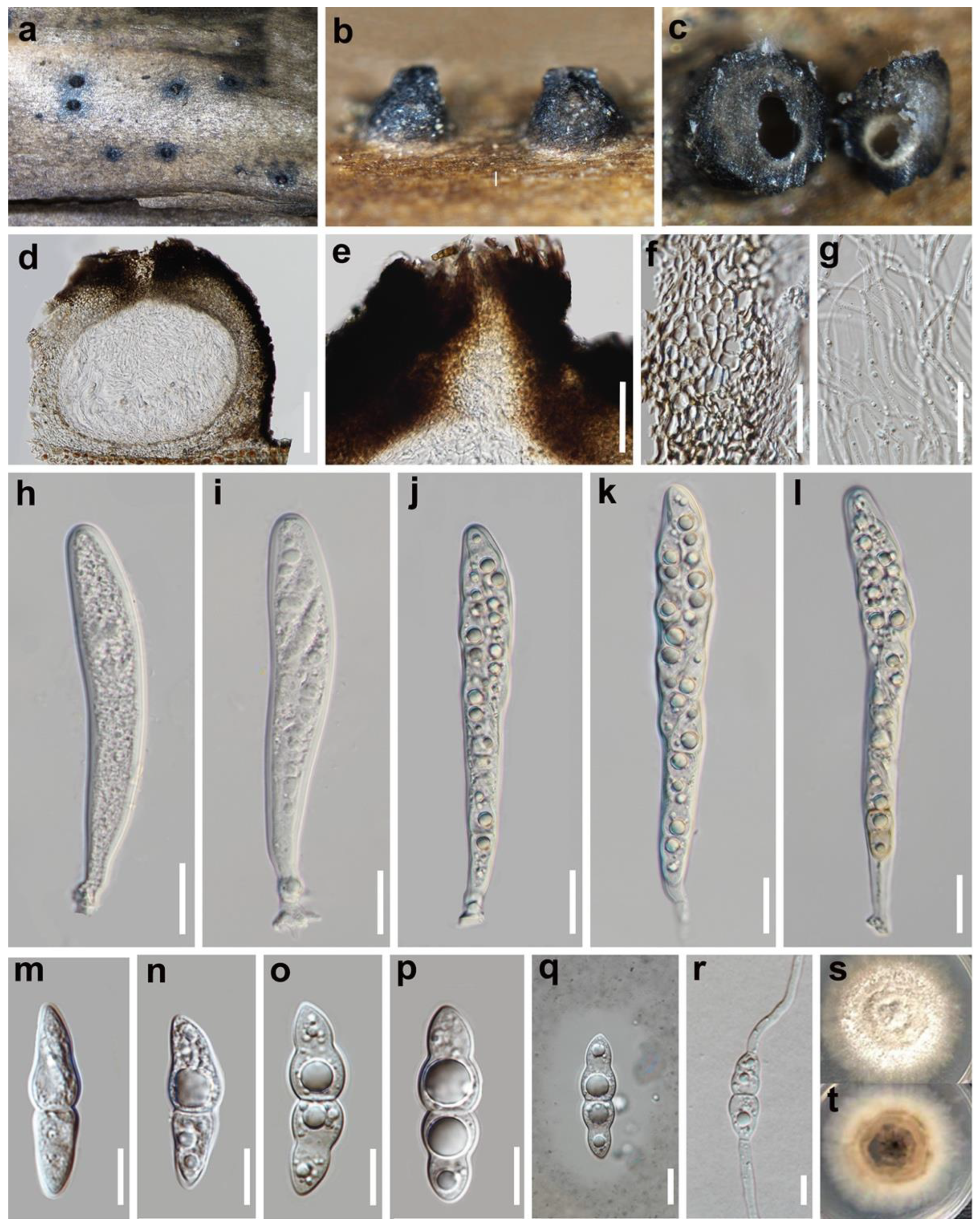
3.2. Taxonomy
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hongsanan, S.; Hyde, K.D.; Phookamsak, R.; Wanasinghe, D.N.; McKenzie, E.H.C.; Sarma, V.V.; Boonmee, S.; Lücking, R.; Bhat, D.J.; Liu, N.G.; et al. Refined families of Dothideomycetes: Dothideomycetidae and Pleosporomycetidae. Mycosphere 2020, 11, 1553–2107. [Google Scholar] [CrossRef]
- Hongsanan, S.; Hyde, K.D.; Phookamsak, R.; Wanasinghe, D.N.; McKenzie, E.H.C.; Sarma, V.V.; Lücking, R.; Boonmee, S.; Bhat, J.D.; Liu, N.G.; et al. Refined families of Dothideomycetes: Orders and families incertae sedis in Dothideomycetes. Fungal Divers. 2020, 105, 17–318. [Google Scholar] [CrossRef]
- Wijayawardene, N.N.; Hyde, K.D.; Al-Ani, L.K.T.; Tedersoo, L.; Haelewaters, D.; Rajeshkumar, K.C.; Zhao, R.L.; Aptroot, A.; Leontyev, D.; Saxena, R.K.; et al. Outline of Fungi and fungus-like taxa. Mycosphere 2020, 11, 1060–1456. [Google Scholar] [CrossRef]
- Atienza, V.; Hawksworth, D.L.; Pérez-Ortega, S. Verrucoccum (Dothideomycetes, Dictyosporiaceae), a new genus of lichenicolous fungi on Lobaria s. lat. for the Dothidea hymeniicola species complex. Mycologia 2021, 113, 1233–1252. [Google Scholar] [CrossRef]
- Hongsanan, S.; Phookamsak, R.; Goonasekara, I.D.; Thambugala, K.M.; Hyde, K.D.; Bhat, J.D.; Suwannarach, N.; Cheewangkoon, R. Introducing a new pleosporalean family Sublophiostomataceae fam. nov. to accommodate Sublophiostoma gen. nov. Sci. Rep. 2021, 11, 9496. [Google Scholar] [CrossRef]
- Li, W.L.; Bao, D.F.; Liu, N.G.; Hyde, K.D.; Liu, J.K. Aquatisphaeria thailandica gen. et sp. nov. (Tetraplosphaeriaceae, Pleosporales) from freshwater habitat in Thailand. Phytotaxa 2021, 513, 118–128. [Google Scholar] [CrossRef]
- Pintye, A.; Knapp, D.G. Two pleosporalean root-colonizing fungi, Fuscosphaeria hungarica gen. et sp. nov. and Delitschia chaetomioides, from a semiarid grassland in Hungary. Mycol. Prog. 2021, 20, 39–50. [Google Scholar] [CrossRef]
- Zhang, Y.; Schoch, C.L.; Fournier, J.; Crous, P.W.; de Gruyter, J.; Woudenberg, J.H.C.; Hirayama, K.; Tanaka, K.; Pointing, S.B.; Spatafora, J.W.; et al. Multi-locus phylogeny of Pleosporales: A taxonomic, ecological and evolutionary re-evaluation. Stud. Mycol. 2009, 64, 85–102. [Google Scholar] [CrossRef]
- Knapp, D.G.; Kovacs, G.M.; Zajta, E.; Groenewald, J.Z.; Crous, P.W. Dark septate endophytic pleosporalean genera from semiarid areas. Persoonia 2015, 35, 87–100. [Google Scholar] [CrossRef] [Green Version]
- Dayarathne, M.C.; Wanasinghe, D.N.; Jones, E.B.G.; Chomnunti, P.; Hyde, K.D. A novel marine genus, Halobyssothecium (Lentitheciaceae) and epitypification of Halobyssothecium obiones comb. nov. Mycol. Prog. 2018, 17, 1161–1171. [Google Scholar] [CrossRef]
- Tanaka, K.; Harada, Y. Bambusicolous fungi in Japan (6): Katumotoa, a new genus of phaeosphaeriaceous ascomycetes. Mycoscience 2005, 46, 313–318. [Google Scholar] [CrossRef]
- Höhnel, F. Fragmente zur Mykologie. XXIII Mitteilung, Nr. 1154 bis 1188. Sitz. Kais. Akad. Wiss. Math.-Nat. Kl. Abt. I 1919, 128, 535–625. Available online: https://www.biodiversitylibrary.org/page/36043822 (accessed on 10 June 2021).
- Wanasinghe, D.N.; Jones, E.B.G.; Camporesi, E.; Boonmee, S.; Ariyawansa, H.A.; Wijayawardene, N.N.; Mortimer, P.E.; Xu, J.; Yang, J.B.; Hyde, K.D. An Exciting Novel Member of Lentitheciaceae in Italy from Clematis vitalba. Cryptogam. Mycol. 2014, 35, 323–337. [Google Scholar] [CrossRef]
- Tanaka, K.; Hirayama, K.; Yonezawa, H.; Sato, G.; Toriyabe, A.; Kudo, H.; Hashimoto, A.; Matsumura, M.; Harada, Y.; Kurihara, Y.; et al. Revision of the Massarineae (Pleosporales, Dothideomycetes). Stud. Mycol. 2015, 82, 75–136. [Google Scholar] [CrossRef] [Green Version]
- Wijayawardene, N.N.; Hyde, K.D.; Bhat, D.J.; Goonasekara, I.D.; Nadeeshan, D.; Camporesi, E.; Schumacher, R.K.; Wang, Y. Additions to Brown Spored Coelomycetous Taxa in Massarinae, Pleosporales: Introducing Phragmocamarosporium gen. nov. and Suttonomyces gen. nov. Cryptogam. Mycol. 2015, 36, 213–224. [Google Scholar] [CrossRef]
- Crous, P.W.; Wingfield, M.J.; Guarro, J.; Hernández-Restrepo, M.; Sutton, D.A.; Acharya, K.; Barber, P.A.; Boekhout, T.; Dimitrov, R.A.; Dueñas, M.; et al. Fungal Planet description sheets: 320–370. Persoonia 2015, 34, 167–266. [Google Scholar] [CrossRef]
- Phookamsak, R.; Manamgoda, D.S.; Li, W.J.; Dai, D.Q.; Singtripop, C.; Hyde, K.D. Poaceascoma helicoides gen et sp. nov., a new genus with scolecospores in Lentitheciaceae. Cryptogam. Mycol. 2015, 36, 225–236. [Google Scholar] [CrossRef]
- Hyde, K.D.; Dong, Y.; Phookamsak, R.; Jeewon, R.; Bhat, D.J.; Jones, E.B.G.; Liu, N.G.; Abeywickrama, P.D.; Mapook, A.; Wei, D.; et al. Fungal diversity notes 1151–1276: Taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers. 2020, 100, 5–277. [Google Scholar] [CrossRef] [Green Version]
- Quaedvlieg, W.; Verkley, G.J.; Shin, H.D.; Barreto, R.W.; Alfenas, A.C.; Swart, W.J.; Groenewald, J.Z.; Crous, P.W. Sizing up Septoria. Stud. Mycol. 2013, 75, 307–390. [Google Scholar] [CrossRef] [Green Version]
- Hirayama, K.; Tanaka, K.; Raja, H.A.; Miller, A.N.; Shearer, C.A. A molecular phylogenetic assessment of Massarina ingoldiana sensu lato. Mycologia 2010, 102, 729–746. [Google Scholar] [CrossRef] [Green Version]
- Li, G.J.; Hyde, K.D.; Zhao, R.L.; Hongsanan, S.; Abdel-Aziz, F.A.; Abdel-Wahab, M.A.; Alvarado, P.; Alves-Silva, G.; Ammirati, J.F.; Ariyawansa, H.A.; et al. Fungal diversity notes 253–366: Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2016, 78, 1–237. [Google Scholar] [CrossRef]
- Tibpromma, S.; Hyde, K.D.; Jeewon, R.; Maharachchikumbura, S.S.N.; Liu, J.K.; Bhat, D.J.; Jones, E.B.G.; McKenzie, E.H.C.; Camporesi, E.; Bulgakov, T.S.; et al. Fungal diversity notes 491–602: Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2017, 83, 1–261. [Google Scholar] [CrossRef]
- Hyde, K.D.; Chaiwan, N.; Norphanphoun, C.; Boonmee, S.; Camporesi, E.; Chethana, K.W.T.; Dayarathne, M.C.; de Silva, N.I.; Dissanayake, A.J.; Ekanayaka, A.H.; et al. Mycosphere notes 169–224. Mycosphere 2018, 9, 271–430. [Google Scholar] [CrossRef]
- Jiang, H.B.; Phookamsak, R.; Doilom, M.; Mortimer, P.E.; Xu, J.C.; Lumyong, S.; Hyde, K.D.; Karunarathna, S.C. Taxonomic and phylogenetic characterizations of Keissleriella bambusicola sp. nov. (Lentitheciaceae, Pleosporales) from Yunnan, China. Phytotaxa 2019, 423, 129–144. [Google Scholar] [CrossRef]
- Xu, L.; Bao, D.F.; Luo, Z.L.; Su, X.J.; Shen, H.W.; Su, H.Y. Lignicolous freshwater ascomycota from Thailand: Phylogenetic and morphological characterisation of two new freshwater fungi: Tingoldiago hydei sp. nov. and T. clavata sp. nov. from Eastern Thailand. MycoKeys 2020, 65, 119–138. [Google Scholar] [CrossRef] [Green Version]
- Pubu, C.R.; Wang, M.; Liu, X.Y. A review of investigation of fungi in Tibet. Mycosystema 2016, 35, 1025–1047. [Google Scholar] [CrossRef]
- Senanayake, I.C.; Rathnayaka, A.R.; Marasinghe, D.S.; Calabon, M.S.; Gentekaki, E.; Lee, H.B.; Hurdeal, V.G.; Pem, D.; Dissanayake, L.S.; Wijesinghe, S.N.; et al. Morphological approaches in studying fungi: Collection, examination, isolation, sporulation and preservation. Mycosphere 2020, 11, 2678–2754. [Google Scholar] [CrossRef]
- Kornerup, A.; Wanscher, J.H. Methuen Handbook of Colour; Eyre Methuen: London, UK, 1978; pp. 1–252. [Google Scholar]
- Crous, P.W.; Gams, W.; Stalpers, J.A.; Robert, V.; Stegehuis, G. MycoBank: An online initiative to launch mycology into the 21st century. Stud. Mycol. 2004, 50, 19–22. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [Green Version]
- Rehner, S.A.; Buckley, E. A Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 2005, 97, 84–98. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef] [Green Version]
- Mesquite Project. Available online: https://www.mesquiteproject.org/ (accessed on 25 August 2021).
- Glez-Peña, D.; Gómez-Blanco, D.; Reboiro-Jato, M.; Fdez-Riverola, F.; Posada, D. ALTER: Program-oriented format conversion of DNA and protein alignments. Nucleic Acids Res. 2010, 38, W14–W18. [Google Scholar] [CrossRef]
- Dissanayake, A.J.; Bhunjun, C.S.; Maharachchikumbura, S.S.N.; Liu, J.K. Applied aspects of methods to infer phylogenetic relationships amongst fungi. Mycosphere 2020, 11, 2652–2676. [Google Scholar] [CrossRef]
- Swofford, D.L. PAUP*: Phylogenetic Analysis Using Paesimony (*and Other Methods); Sinauer Associates: Sunderland, UK, 2002. [Google Scholar]
- Silvestro, D.; Michalak, I. raxmlGUI: A graphical front-end for RAxML. Org. Divers. Evol. 2011, 12, 335–337. [Google Scholar] [CrossRef]
- Rannala, B.; Yang, Z. Probability distribution of molecular evolutionary trees: A new method of phylogenetic inference. J. Mol. Evol. 1996, 43, 304–311. [Google Scholar] [CrossRef]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef] [Green Version]
- Nylander, J.A.A. MrModeltest, Version 2. Program Distributed by the Author. Evolutionary Biology Centre, Uppsala University: Uppsala, Sweden, 2004.
- FigTree 1.4.4. Available online: http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 25 August 2021).
- Dai, D.Q.; Bhat, D.J.; Liu, J.K.; Chukeatirote, E.; Zhao, R.L.; Hyde, K.D. Bambusicola, a new genus from bamboo with asexual and sexual morphs. Cryptogam. Mycol. 2012, 33, 363–379. [Google Scholar] [CrossRef]
- Crous, P.W.; Wingfield, M.J.; Burgess, T.I.; Hardy, G.E.S.J.; Gené, J.; Guarro, J.; Baseia, I.G.; García, D.; Gusmão, L.F.P.; Souza-Motta, C.M.; et al. Fungal planet description sheets: 716–784. Persoonia 2018, 40, 240–393. [Google Scholar] [CrossRef]
- Devadatha, B.; Calabon, M.S.; Abeywickrama, P.D.; Hyde, K.D.; Jones, E.B.G. Molecular data reveals a new holomorphic marine fungus, Halobyssothecium estuariae, and the asexual morph of Keissleriella phragmiticola. Mycology 2020, 11, 167–183. [Google Scholar] [CrossRef] [Green Version]
- Dong, W.; Wang, B.; Hyde, K.D.; McKenzie, E.H.C.; Raja, H.A.; Tanaka, K.; Abdel-Wahab, M.A.; Abdel-Aziz, F.A.; Doilom, M.; Phookamsak, R.; et al. Freshwater Dothideomycetes. Fungal Divers. 2020, 105, 319–575. [Google Scholar] [CrossRef]
- Hyde, K.D.; Hongsanan, S.; Jeewon, R.; Bhat, D.J.; McKenzie, E.H.C.; Jones, E.B.G.; Phookamsak, R.; Ariyawansa, H.A.; Boonmee, S.; Zhao, Q.; et al. Fungal diversity notes 367–490: Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2016, 80, 1–270. [Google Scholar] [CrossRef]
- Wanasinghe, D.N.; Phukhamsakda, C.; Hyde, K.D.; Jeewon, R.; Lee, H.B.; Jones, E.B.G.; Tibpromma, S.; Tennakoon, D.S.; Dissanayake, A.J.; Jayasiri, S.C.; et al. Fungal diversity notes 709–839: Taxonomic and phylogenetic contributions to fungal taxa with an emphasis on fungi on Rosaceae. Fungal Divers. 2018, 89, 1–236. [Google Scholar] [CrossRef]
- Luo, Z.L.; Bahkali, A.H.; Liu, X.Y.; Phookamsak, R.; Zhao, Y.C.; Zhou, D.Q.; Su, H.Y.; Hyde, K.D. Poaceascoma aquaticum sp. nov. (Lentitheciaceae), a new species from submerged bamboo in freshwater. Phytotaxa 2016, 253, 71–80. [Google Scholar] [CrossRef]
- Crous, P.W.; Wingfield, M.J.; Chooi, Y.H.; Gilchrist, C.L.M.; Lacey, E.; Pitt, J.I.; Roets, F.; Swart, W.J.; Cano-Lira, J.F.; Valenzuela-Lopez, N.; et al. Fungal planet description sheets: 1042–1111. Persoonia 2020, 44, 301–459. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Norphanphoun, C.; Abreu, V.P.; Bazzicalupo, A.; Chethana, K.W.T.; Clericuzio, M.; Dayarathne, M.C.; Dissanayake, A.J.; Ekanayaka, A.H.; He, M.Q.; et al. Fungal diversity notes 603–708: Taxonomic and phylogenetic notes on genera and species. Fungal Divers. 2017, 87, 1–235. [Google Scholar] [CrossRef]
- Zhang, Y.; Crous, P.W.; Schoch, C.L.; Hyde, K.D. Pleosporales. Fungal Divers. 2012, 53, 1–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyde, K.D.; Jones, E.B.G.; Liu, J.K.; Ariyawansa, H.; Boehm, E.; Boonmee, S.; Braun, U.; Chomnunti, P.; Crous, P.W.; Dai, D.Q.; et al. Families of Dothideomycetes. Fungal Divers. 2013, 63, 1–313. [Google Scholar] [CrossRef]
- Calabon, M.S.; Jones, E.B.G.; Hyde, K.D.; Boonmee, S.; Tibell, S.; Tibell, L.; Pang, K.L.; Phookamsak, R. Phylogenetic assessment and taxonomic revision of Halobyssothecium and Lentithecium (Lentitheciaceae, Pleosporales). Mycol. Prog. 2021, 20, 701–720. [Google Scholar] [CrossRef]
| Taxa | Strain/Voucher | LSU | ITS | SSU | TEF1-α | References |
|---|---|---|---|---|---|---|
| Bambusicola bambusae | MFLUCC 11-0614 | JX442035 | NR121546 | JX442039 | KP761722 | [44] |
| Bambusicola irregulispora | MFLUCC 11-0437 | JX442036 | NR121547 | JX442040 | KP761723 | [25] |
| Bimuria novae-zelandiae | CBS 107.79 | AY016356 | – | AY016338 | DQ471087 | [14] |
| Darksidea alpha | CBS 135650 | KP184019 | KP183998 | KP184049 | KP184166 | [9] |
| Darksidea beta | CBS 135637 | KP184023 | KP183978 | KP184074 | KP184189 | [9] |
| Darksidea delta | CBS 135638 | KP184024 | KP183981 | KP184069 | KP184184 | [9] |
| Darksidea epsilon | CBS 135658 | KP184029 | KP183983 | KP184070 | KP184186 | [9] |
| Darksidea gamma | CBS 135634 | KP184028 | KP183985 | KP184073 | KP184188 | [9] |
| Darksidea zeta | CBS 135640 | KP184013 | KP183979 | KP184071 | KP184191 | [9] |
| Didymosphaeria rubi-ulmifolii | MFLUCC 14-0023 | KJ436586 | – | KJ436588 | – | [14] |
| Crassoascoma potentillae | UESTCC 21.0010 * | OK161254 | OK161237 | OK161233 | OK181165 | This study |
| Crassoascoma potentillae | UESTCC 21.0011 * | OK161255 | OK161238 | OK161234 | OK181166 | This study |
| Crassoascoma potentillae | UESTCC 21.0012 * | OK161256 | OK161239 | OK161235 | OK181167 | This study |
| Crassoascoma potentillae | CGMCC 3.20483 * | OK161257 | OK161240 | OK161236 | OK181168 | This study |
| Halobyssothecium carbonneanum | CBS 144076 | MH069699 | MH062991 | – | – | [45] |
| Halobyssothecium estuariae | MFLUCC 19-0386 | MN598871 | MN598890 | MN598868 | MN597050 | [46] |
| Halobyssothecium kunmingense | KUMCC 19-0101 | MN913732 | MT627715 | MT864313 | – | [47] |
| Halobyssothecium obiones | MFLUCC 15-0381 | MH376744 | MH377060 | MH376745 | MH376746 | [10] |
| Halobyssothecium unicellulare | MD 6004 | KX505376 | – | KX505374 | – | [48] |
| Halobyssothecium voraginesporum | MD1342 | KX499520 | – | KX499519 | – | [48] |
| Helminthosporium velutinum | MAFF 243854 | AB807530 | LC014556 | AB797240 | AB808505 | [14] |
| Katumotoa bambusicola | KT1517a | AB524595 | LC014560 | AB524454 | AB539108 | [8] |
| Keissleriella breviasca | KT649 | AB807588 | AB811455 | AB797298 | AB808567 | [14] |
| Keissleriella caraganae | KUMCC 18-0164 | MK359439 | MK359434 | MK359444 | MK359073 | [47] |
| Keissleriella cladophila | CBS 104.55 | GU301822 | – | GU296155 | GU349043 | [14] |
| Keissleriella culmifida | KT2642 | AB807592 | LC014562 | AB797302 | AB808571 | [14] |
| Keissleriella genistae | CBS 113798 | GU205222 | – | GU205242 | – | [14] |
| Keissleriella gloeospora | KT829 | AB807589 | LC014563 | AB797299 | AB808568 | [14] |
| Keissleriella poagena | CBS136767 | KJ869170 | KJ869112 | – | – | [14] |
| Keissleriella quadriseptata | KT2292 | AB807593 | AB811456 | AB797303 | AB808572 | [14] |
| Keissleriella taminensis | KT571 | AB807595 | LC014564 | AB797305 | AB808574 | [14] |
| Keissleriella trichophoricola | CBS 136770 | KJ869171 | KJ869113 | – | – | [14] |
| Keissleriella yonaguniensis | KT 2604 | AB807594 | AB811457 | AB797304 | AB808573 | [14] |
| Lentithecium clioninum | KT1149A | AB807540 | LC014566 | AB797250 | AB808515 | [14] |
| Lentithecium fluviatile | CBS 122367 | GU301825 | – | GU296158 | GU349074 | [14] |
| Lentithecium pseudoclioninum | KT1113 | AB807545 | AB809633 | AB797255 | AB808521 | [14] |
| Magnicamarosporium iriomotense | KT2822 | AB807509 | AB809640 | – | AB808485 | [14] |
| Massarina eburnea | CBS 473.64 | GU301840 | – | GU296170 | GU349040 | [14] |
| Montagnula opulenta | CBS 168.34 | DQ678086 | – | AF164370 | – | [14] |
| Murilentithecium clematidis | MFLUCC 14-0562 | KM408759 | KM408757 | KM408761 | KM454445 | [13] |
| Murilentithecium rosae | MFLUCC 15-0044 | MG829030 | MG828920 | MG829137 | – | [49] |
| Neobambusicola strelitziae | CBS 138869 | KP004495 | – | – | – | [14] |
| Neoophiosphaerella sasicola | KT1706 | AB524599 | LC014577 | AB524458 | AB539111 | [14] |
| Phragmocamarosporium platani | MFLUCC 14-1191 | KP842916 | – | KP842919 | – | [15] |
| Phragmocamarosporium rosae | MFLUCC 17-0797 | MG829051 | – | MG829156 | MG829225 | [49] |
| Pleurophoma ossicola | CPC 24979 | KR476769 | KR476736 | – | – | [16] |
| Pleurophoma pleurospora | CBS130329 | JF740327 | – | – | – | [14] |
| Poaceascoma aquaticum | MFLUCC 14-0048 | KT324690 | – | KT324691 | – | [50] |
| Poaceascoma filiforme | CBS 146689 | MT373345 | MT373362 | – | – | [51] |
| Poaceascoma halophila | MFLUCC 15-0949 | MF615399 | – | MF615400 | – | [52] |
| Poaceascoma helicoides | MFLUCC 11-0136 | KP998462 | KP998459 | KP998463 | KP998461 | [17] |
| Poaceascoma taiwanense | MFLU 18-0083 | MG831567 | MG831569 | MG831568 | – | [23] |
| Setoseptoria arundelensis | MFLUCC 17-0759 | MG829073 | MG828962 | MG829173 | – | [49] |
| Setoseptoria arundinacea | KT600 | AB807575 | LC014595 | AB797285 | AB808551 | [14] |
| Setoseptoria englandensis | MFLUCC 17-0778 | MG829074 | MG828963 | MG829174 | – | [49] |
| Setoseptoria macropycnidia | CBS114202 | GU301873 | – | GU296198 | GU349026 | [14] |
| Setoseptoria magniarundinacea | KT1174 | AB807576 | LC014596 | AB797286 | AB808552 | [14] |
| Setoseptoria phragmitis | CBS 114802 | KF251752 | KF251249 | – | – | [19] |
| Setoseptoria scirpi | MFUCC 14-0811 | KY770982 | MF939637 | KY770980 | KY770981 | [52] |
| Spegazzinia deightonii | MAFF 243876 | AB807581 | – | AB797291 | AB808557 | [14] |
| Sulcatispora acerina | KT 2982 | LC014610 | LC014597 | LC014605 | LC014615 | [14] |
| Sulcatispora berchemiae | KT 1607 | AB807534 | AB809635 | AB797244 | AB808509 | [14] |
| Tingoldiago clavata | MFLUCC 19-0496 | MN857178 | MN857182 | MN857186 | – | [25] |
| Tingoldiago graminicola | KH68 | AB521743 | LC014598 | AB521726 | AB808561 | [14] |
| Tingoldiago hydei | MFLUCC 19-0499 | MN857177 | MN857181 | – | – | [25] |
| Towyspora aestuari | MFLUCC 15-1274 | KU248852 | KU248851 | KU248853 | – | [21] |
| Wettsteinina lacustris | CBS 618.86 | – | AF250831 | DQ678023 | DQ677919 | [8] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.-P.; Zhang, S.-N.; Cheewangkoon, R.; Zhao, Q.; Liu, J.-K. Crassoascoma gen. nov. (Lentitheciaceae, Pleosporales): Unrevealing Microfungi from the Qinghai-Tibet Plateau in China. Diversity 2022, 14, 15. https://doi.org/10.3390/d14010015
Liu Z-P, Zhang S-N, Cheewangkoon R, Zhao Q, Liu J-K. Crassoascoma gen. nov. (Lentitheciaceae, Pleosporales): Unrevealing Microfungi from the Qinghai-Tibet Plateau in China. Diversity. 2022; 14(1):15. https://doi.org/10.3390/d14010015
Chicago/Turabian StyleLiu, Zuo-Peng, Sheng-Nan Zhang, Ratchadawan Cheewangkoon, Qi Zhao, and Jian-Kui Liu. 2022. "Crassoascoma gen. nov. (Lentitheciaceae, Pleosporales): Unrevealing Microfungi from the Qinghai-Tibet Plateau in China" Diversity 14, no. 1: 15. https://doi.org/10.3390/d14010015
APA StyleLiu, Z.-P., Zhang, S.-N., Cheewangkoon, R., Zhao, Q., & Liu, J.-K. (2022). Crassoascoma gen. nov. (Lentitheciaceae, Pleosporales): Unrevealing Microfungi from the Qinghai-Tibet Plateau in China. Diversity, 14(1), 15. https://doi.org/10.3390/d14010015







