Abstract
This paper presents a study of freshwater zooplankton biodiversity, deemed as a reliable indicator of water quality. The Guatemalan Lake Amatitlán, currently used as a water source, has shown signs of progressive eutrophication, with perceptible variations of the local zooplankton diversity. Biotic and abiotic parameters were determined at four sites of Lake Amatitlán (Este Centro, Oeste Centro, Bahía Playa de Oro, and Michatoya) in 2016 and 2017. The local composition, the species richness and abundance of zooplankton, and the system environmental parameters were analyzed during both years surveyed. Biological data suggesting eutrophication of this tropical system were obtained, including a high rotifer abundance (11 species: the rotifers Brachionus havanaensis (109 ind L−1) and Keratella americana (304 ind L−1) were the most abundant species in this lake). The presumably endemic diaptomid copepod species, Mastigodiaptomus amatitlanensis, was absent in our samples, but we report the unprecedented occurrence of two Asian cyclopoid copepods (i.e., Thermocyclops crassus and Mesocyclops thermocyclopoides) for Lake Amatitlán and Guatemala. The presence of larger zooplankters like adults and immature copepods (i.e., Arctodiaptomus dorsalis) and cladocerans (Ceriodaphnia sp.) at site “Este Centro” indicates a relatively healthy zooplankton community and represents a focal point for managing the conservation of this lake.
1. Introduction
The knowledge of zooplankton in the Neotropical region is growing with fragmented studies. Therefore, it is likely that the species richness of zooplanktonic taxa is underestimated because of the presumably high diversity and scarcity of zooplankton taxonomists [1,2,3]. In addition, the progressive destruction of aquatic habitat and the progressive spread of exotic species threaten native biodiversity, ecosystem health, and environmental services.
The zooplankton community and abundance are closely linked to the trophic state of the water system; for this reason, its diversity has been deemed as an indicator of water quality [4]. In eutrophicated systems (at tropical and temperate latitudes), the dominance of microzooplankton is common, compared with larger organisms, owing to the increased availability of food and water conditions [5,6].
For four decades, the Guatemalan Lake Amatitlán has shown signs of progressive eutrophication related to anthropic factors (i.e., peripheral population growth and urbanization, intensive use of water for agricultural irrigation), thus promoting the advancement towards eutrophication, related to the input of nearly 50% of the untreated residual urban and industrial waters from Guatemala City [7,8,9,10]. Because of this, some actions have been proposed to address this problem, either from the governmental level (i.e., Autoridad para el Manejo Sustentable de la cuenca del lago Amatitlán, AMSA 1996) or from descriptive studies of the lake involving the lake zooplankton biodiversity, like those by Basterrechea-Díaz (1997) [7] and Brandorff (2012) [11]; however, studies related with tropical epicontinental waterbodies have been more focused on environmental factors rather than biological community attributes or general limnology [12,13]; thus, the zooplankton biodiversity in Guatemala remains largely unknown [14], with only a few studies in Guatemalan lakes [15,16]. Most studies in Lake Amatitlán and Guatemala are more focused on current data instead of historical analysis.
Based on the analysis of both, historical and current data of zooplankton biodiversity and environmental conditions of Lake Amatitlán, we present information on the zooplankton distribution, species richness, abundance, and its relation with successive changes of its trophic state.
2. Materials and Methods
2.1. Study Sites and Sampling Methods
Lake Amatitlán is the fourth largest lake in Guatemala, Central America, and one of the most emblematic waterbodies of this country. This lake is a warm monomictic waterbody in the highland of Guatemala, located at an altitude of 1186 m above sea level (m.a.s.l.), with an area of 15.2 km2 and 11 km length and a maximum depth of 23 m. Its formation originated from volcanic activity of Pacaya, Fuego, and Agua in the late Quaternary [10,14,17].
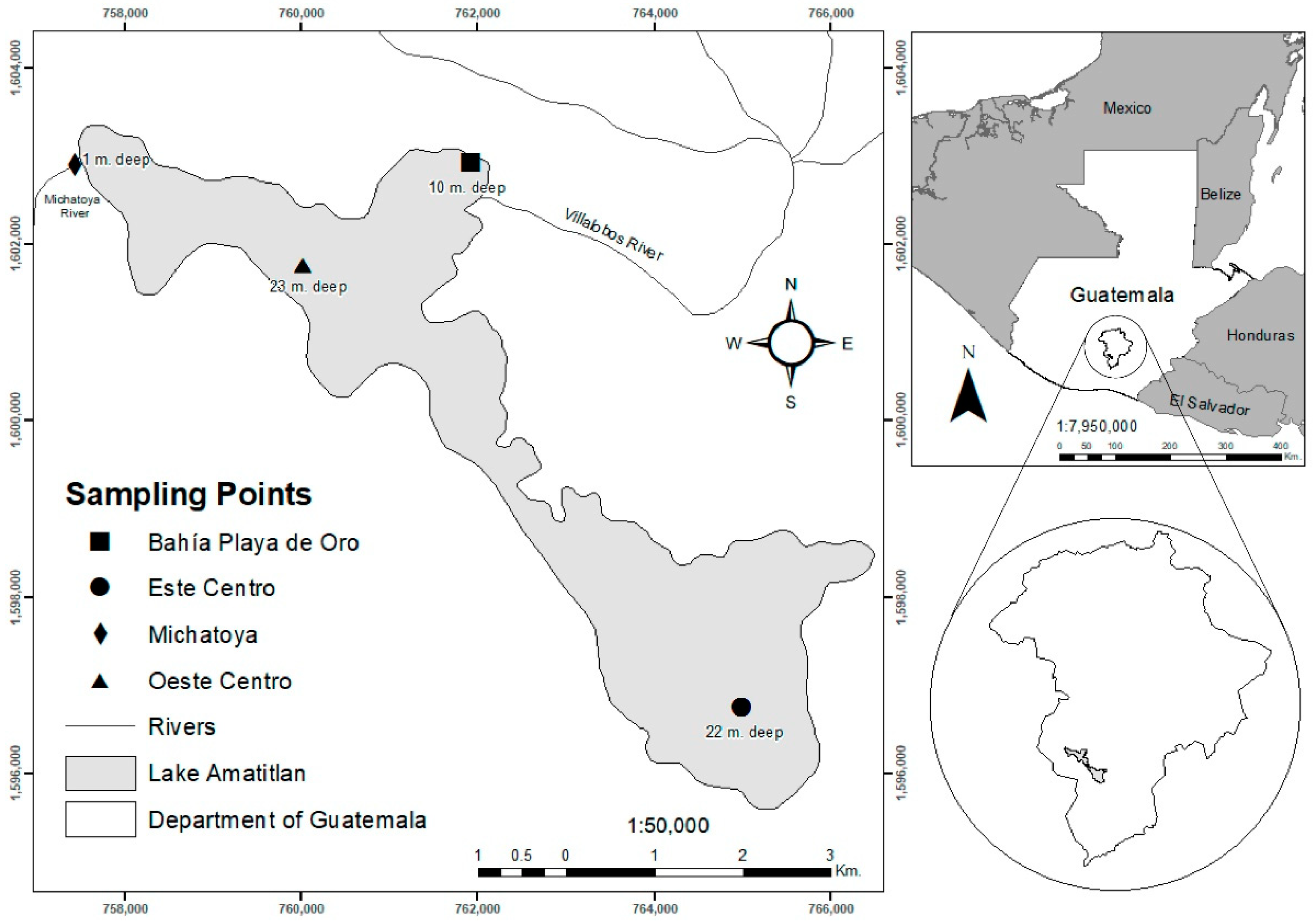
Four sampled sites were considered: Este Centro (EC), Oeste Centro (OC), Bahía Playa de Oro (BPO), and Michatoya (MICH) to analyze the zooplankton species that inhabit the eastern and western regions of Lake Amatitlán (Figure 1). The latter two sites (BPO and MICH) are in the runoff of Villalobos and Michatoya rivers, respectively [14]. Water samples for biotic and abiotic variables were collected for 2016 and 2017 in the rainy (May–October) and dry seasons (November–April).
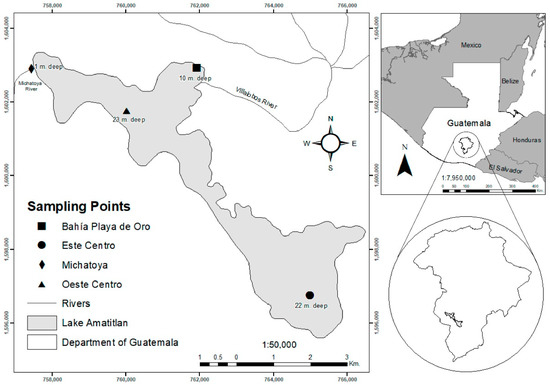
Figure 1.
Location of Lake Amatitlán and sampling points for the biotic collection methodology. Water-filtered, vertical, and horizontal trawls as defined by Cervantes-Martínez & Gutiérrez-Aguirre (2015) [2].
2.1.1. Species Richness
Zooplankton samples (n = 8) were collected by vertical and horizontal trawls with a 45 µm plankton net between 1 and 22 m depth to ensure representative samples to evaluate the species richness in the lake, as it is well known that zooplankton tends to have vertical and horizontal migrations [2].
2.1.2. Species Abundance and Abiotic Variables
To estimate the zooplankton abundance, a known volume of water between 30 and 100 L was filtered through a 45 µm zooplankton net. The water was determined with a 2.1 L−1 capacity Van Dorn bottle [2,18]. Species abundance was determined by the account of two main groups: Rotifera and Copepoda, present in three aliquots of 1 mL each from the filtered samples, then the data were standardized as individuals per liter (ind L−1) in each sampled site [19].
Abiotic variables were measured in situ monthly for both years of study and in all the water columns, with the multiparametric proves WTW Cond 197i, WTW Oxi 1970i, and HACH HQ for water temperature (°C), pH, oxygen concentration O2 (mg L−1), total dissolved solids (mg L−1), and conductivity (µS cm−1). With the actual environmental, richness, and zooplankton abundance data, a description of the trophic state of Lake Amatitlán was proposed.
2.2. Historical and Actual Records of Zooplankton and Environmental Parameters Analysis
Specific classification of Rotifera, Cladocera, and Copepoda of recently collected samples (collected in 2016 and 2017) was done according to Koste (1978) [20], Fontaneto & De Smet (2015) [21], Elías-Gutiérrez et al., (2008) [22], and Suárez-Morales et al., (2020) [23].
The presence/absence of the current zooplankton inventory was compared with previous surveys by Juday (1915) [24], Basterrechea-Díaz (1997) [7], and the record of copepods from the previous surveys of Wilson (1941) [25] and Brandorff (2012) [11], in order to analyze the historical composition of zooplankton of Amatitlán lake.
Historical environmental data recorded by Juday (1915) [24], Brezonik & Fox (1974) [26], Basterrechea-Díaz (1997) [7], and Ellenberg (2014) [27] were compared with the current data surveyed in this study.
3. Results
3.1. Species Richness
A total of 15 species of zooplankters including rotifers and crustaceans were found in the lake for 2016–2017 (Table 1); rotifers showed the highest species richness (80% of zooplankton species recorded), while copepods represented 20% of all zooplankton species in the lake.

Table 1.
Current and historical records of zooplankton species richness in Lake Amatitlán. Currently recorded species are shown in columns (1) EC, (2) OC, (3) BPO, and (4) MICH. Historical records are shown in columns 5–8, following data by Brandorff (2012) [11]; Basterrechea-Díaz (1997) [7]; Wilson (1941) [25]; and Juday (1915) [24], respectively. Presence (x), absence (-), new records (*).
We provide the first record of two cyclopoid exotic species (Mesocyclops thermocyclopoides and Thermocyclops crassus) for Lake Amatitlán and Guatemala. The endemic calanoid copepod, Mastigodiaptomus amatitlanensis, was absent in our current survey and the record of Arctodiaptomus dorsalis in Lake Amatitlán was confirmed here. Cladoceran crustaceans were very scarce in our samples; only a single specimen of Ceriodaphnia sp. was observed. The Brachionidae was the family with the highest species richness among rotifers in 2016 and 2017 (Table 1).
Nowadays, the east region (site EC) of Lake Amatitlán had the highest species richness in the lake (14 species), compared with the western region (9 species including the exotic T. crassus at OC). The largest zooplankters of the lake, including the cladoceran Ceriodaphnia, (~2 mm) [21], the calanoid copepod A. dorsalis, and the cyclopoid copepod M. thermocyclopoides, occurred in eastern region.
Our revision of the zooplankton community (Table 1) indicates that the historical data presented a great microcrustacean richness with the record of eight cladoceran species (Dapnia sp., D. hyalina, Ceriodaphnia sp. C. lacustris, C. pulchella, Bosmina sp., B. longirostris, and Chydorus sphaericus) and the three calanoid copepods: A. dorsalis, Mastigodiaptomus albuquerquensis, and the endemic M. amatitlanensis. The historical record of rotifers had the lowest species richness including three monogonont species. In our survey, the rotifer species richness increased significantly with 12 species not hitherto reported from the lake, including the record of organisms from the Subclass Bdelloidea.
3.2. Species Abundance
In this study, the total rotifer abundance was 522.7 ind L−1. Rotifers represent the most abundant group in the lake; their numerical abundance is considerably higher than that recorded for copepods, including immature stages (7.1 ind L−1). Cladocerans were almost absent from our samples.
Species with the highest abundance at all sites were the rotifers B. havanaensis (109 ind L−1) and K. americana (304 ind L−1), with a considerably lower abundance in the eastern area (9.3 and 121.8 ind L−1, respectively). Species of the family Brachionidae were the most abundant mainly in the western region (sites OC, BPO, and MICH), whereas the lowest abundance of rotifers occurred in the eastern region (site EC) (see Table 2).

Table 2.
Abundance (ind L−1) calculated from zooplankton samples for all the studied points of Lake Amatitlán in 2017.
The local copepod abundance was represented mainly by nauplii and juvenile stages of Calanoida and Cyclopoida (average = 2.6, 2.5 and 1.9 ind L−1, respectively), values resembling those recorded for the Rotifera like B. plicatilis (1.1 ind L−1) and A. sieboldi (2.3 ind L−1) in all the study sites, compared with adult copepods, where the abundance of the adult M. thermocyclopoides present only in EC was 0.23 ind L−1.
3.3. Environmental Variables
Environmental variables values in both analyzed years, in general, presented basic pH values (>8 ± 0.33), dissolved oxygen showed an average of 4.76 ± 5.21 and 4.65 ± 4.92 mg L−1, whereas temperature averaged 24 ± 1.31 °C, conductivity presented average values of 655.95 ± 59.52 and 678.23 ± 68.29 µS cm−1, and finally TDS showed average values of 339.99 ± 47.35 and 341.43 ± 30.30 mg L−1, respectively (Table 3).

Table 3.
Historical and current environmental mean data of the water column recorded by previous surveys and this study. Juday (1915) [24], Brezonik & Fox (1974) [26], Basterrechea-Díaz (1997) [7], Ellenberg (2014) [27]. ND: no data available.
The historical data presented in Table 3 show pH with slightly neutral values in 1969 to 1985–1995, whereas in the first two decades of the XXI century, the pH increased to reach clearly basic values, over 8. The water temperature changed along the time, 19.86 °C in 1910 to 24.23 °C in 2017. Conductivity and total dissolved solids decreased on average by 18.29 and 44.10%, respectively.
4. Discussion
The environmental parameters surveyed in this study can show the progressive eutrophication on Lake Amatitlán, according to the historical data recorded by authors like Juday (1915) [24], Brezonik & Fox (1974) [26] Basterrechea-Díaz (1997) [7], and Ellenberg (2014) [27]. The historical change in environmental and biological variables could reveal strong evidence of the current eutrophication of this lake. For instance, the observed changes of pH values, that is, an average of 8.26 and 8.33 in 2016–2017, differ in contrast from the values recorded in 1969 (7.70) [26], 1985–1995 (7.75) [7], and 2008 (9.3) [17].
The basic pH and the high concentration of dissolved oxygen at the surface promoted an increase of microzooplankters, like rotifers (especially B. havanaensis and K. americana), and a decrease of larger species like cladocerans and adult copepods, indicators of the system trophic state per se. Similar conditions have been recorded in American eutrophicated subtropical and tropical water bodies [4,28,29] as well as in other water bodies (i.e., temperate coastal water bodies) in which the replacement of larger copepod with smaller ones has been reported to the result from the eutrophication process [6].
Recently, phytoplankton blooming has been described as a consequence of this eutrophication progress in Lake Amatitlán, presenting a high concentration mainly in Microcystis sp. and Dolichospermum sp. cyanobacteria preceded by the diatom algae Niszcha sp. at the surface of the lake [9], which in turn allows herbivorous zooplankters like brachionid rotifers to become dominant organisms in eutrophicated epicontinental waterbodies [20].
In earlier studies on Lake Amatitlán, the zooplankton community was largely dominated by cladocerans and copepods. In 1915 [24], zooplankton had a widely different composition compared with our results: rotifers were then the less abundant zooplankton group in the lake (0.3 ind L−1), preceded by copepods (11.6 ind L−1) and cladocerans, the most abundant zooplankton group at that time (14.4 ind L−1). The system trophic state is also related to the zooplankters body size; that is, a stronger level of eutrophication is frequently expressed by a greater abundance and species richness of microzooplankters like small rotifers [4,6,28,29]. A possible explanation of the local absence or scariness of larger zooplankters (i.e., Ceriodaphnia sp., adult cyclopoid and calanoid copepods, including M. amatitlanensis) could result from the competition for available food [5], eventually explaining the strong dominance of small brachionid herbivorous rotifers like B. havanaensis and K. americana.
The presence and high abundance of these latter species, together with another species of Brachionus and Keratella at the east region of Lake Amatitlán, suggest that eutrophic conditions that make food available for these microphagous species [30].
In the case of A. dorsalis, this species is widespread in America [31] and has been recorded as an invasive exotic copepod in Asiatic waterbodies [32,33]. The environmental conditions of Lake Amatitlán seem to be adequate for the development of this species because it shows a selective feeding on phytoplankton; thus, it frequently inhabits moderately to strongly eutrophicate environments [31,32], like Amatitlán lake.
It is well known that many diaptomid copepods tend to have restricted distributional patterns and endemic distributions in neotropical lakes [34]. Then, the local absence of the endemic copepod M. amatitlanensis in this study could be another indicator of the progressive eutrophication of Lake Amatitlán, because, since its description by Wilson (1941) [25], this species has not been recorded in other regional studies (i.e., Elías-Gutiérrez et al., 2008 [35]; Brandorff, 2012 [11]; and Gutiérrez-Aguirre, et al., 2020 [36]). It is probable that M. amatitlanensis occurs in other lakes of Guatemala (or Central America) and it is expected to be collected from adjacent systems. It is also probable that this species dwells at higher depths not easily reached by standard nets.
Our results showed a clear zonification; the eastern region (site EC) diverges from the other sites because of the absence of adjacent rivers (see Figure 1), its distance from the other sampling points (the closest site is OC, 7.04 km away), and its separation from other sites owing to a train riel that divides the lake in two [14]. Therefore, the EC area has the best conservation status of the lake, precisely where we found the greatest species richness and the larger zooplankters, with the copepods T. crassus (average body length of 0.56–0.93 mm) [37], M. thermocyclopoides (0.78–0.89 mm) [38], and A. dorsalis (0.77–1.13 mm) [31] among them. Thus, it is convenient to consider EC as a potential conservation site as it has better environmental conditions for the conservation and preservation of zooplankton biodiversity.
On the other hand, we report the presence of two exotic cyclopoid copepod species for the Central American Lake Amatitlán and Guatemala country, M. thermocyclopoides and T. crassus. M. thermocyclopoides is a native species from Taiwan and is well spread in Asia and Africa, and commonly widespread at tropical latitudes. This species has been recorded in lakes from South Mexico in epicontinental waterbodies from Chiapas state, Mexico, considering that their introduction may be related to anthropic factors (i.e., agriculture and aquaculture) [37,38]. This is the second record of the invasion of this species in Central American countries, as it has been recorded before in Costa Rican water bodies by Collado et al. (1894) [39], and the ecological potential of Mesocyclops use as biocontrol of vector mosquitoes like Aedes aegypti is well known [40,41,42]. Therefore, its finding in Guatemalan lakes represents a source for mass culture of this copepod to be used as biocontrol.
Thermocyclopscrassus is commonly spread at tropical latitudes in Africa, Australia, and Asia; it was also recorded in Laurentian great lakes in the United States of America [43]; recorded for the first time in tropical lakes from Tabasco state, Mexico [37]; as well as in small ponds of San José Province in Costa Rica [39]. Being a thermophilic species, T. crassus has a narrow temperature tolerance [44], so it may be a local indicator of the temperature changes in the lake along time.
Finally, the physical, chemical, and biological conditions of the lake have clearly changed over time, from being a lake with oligotrophic characteristics to one with hypertrophic conditions in a relatively short period of time (100 years, approximately), allowing us to follow and describe the stages and speed of the eutrophication process of a large neotropical lake.
5. Conclusions
The historical analysis of zooplankton composition in the lake presented in this study reinforces the knowledge of its eutrophic state, suggesting a useful role of the zooplankton as a bioindicator and making possible the visualization of the changes in its composition over time, showing the progressive trophic state towards eutrophic or hypereutrophic conditions.
It is likely that the absence of the endemic species M. amatitlanensis is a warning sign regarding the accelerated loss of biodiversity and reinforces the idea that zooplankton is a great tool as a bioindicator of the health status for continental aquatic ecosystems, in both tropical and temperate latitudes.
Further studies analyzing bottom sediments to search resting eggs of zooplankton in Lake Amatitlán and around it can answer the question of the absence of M. amatitlanensis, where this type of knowledge is also scarce in inland aquatic systems of the region.
Finally, is convenient to consider the isolated site EC as a focal point for conservation as it presents better environmental conditions for the conservation and preservation of zooplankton biodiversity, owing to the record of the largest zooplankters found in this site.
Author Contributions
Conceptualization, S.J. and A.C.-M.; methodology, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B.; software, S.J.; validation, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B.; formal analysis, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B.; investigation, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B.; resources, J.R.J.-P. and E.M.R.-S.; data curation, S.J.; writing—original draft preparation, S.J.; writing—review and editing, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B.; visualization, S.J. and A.C.-M.; supervision, S.J. and A.C.-M.; project administration, S.J., A.C.-M., M.A.G.-A., E.S.-M., J.R.J.-P., E.M.R.-S. and V.H.D.-B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Council of Science and Technology (CONACYT) with the Investigator SNI-III Assistant Program, Grant Number 19812.
Institutional Review Board Statement
Zooplankton collected in this study was provided by AMSA in Guatemala. Zooplankton is not under any protection by Guatemalan laws; therefore, no specific permits are required for this kind of field.
Data Availability Statement
The data presented in this study are available on request from the corresponding author due to the need of further use to complete the first author’s postgraduate requirements.
Acknowledgments
We gratefully acknowledge the invitation of Manuel Elías-Gutiérrez to contribute to this special issue of the Selected Papers from 1st International Electronic Conference on Biological Diversity, Ecology, and Evolution, and his valuable suggestions that made possible this research. We acknowledge AMSA for all the biological material and data bases that made possible the present study. Anonymous reviewers made valuable comments and suggestions. University of Quintana Roo, Cozumel with the investigation group vulnerability of continental and coastal water systems and the National Council of Science and Technology (CONACYT) with the Investigator SNI-III Assistant Program (Number 19812) provided financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- José de Paggi, S.B.; Wallace, R.; Fontaneto, D.; Marinone, M.C. Phylum Rotifera. In Thorp and Covich’s Freshwater Invertebrates; Damborenea, C., Rogers, C.D., James, T., Eds.; Elsevier Science Publishing Co Inc.: San Diego, CA, USA, 2020; pp. 145–200. [Google Scholar]
- Cervantes-Martínez, A.; Gutiérrez-Aguirre, M.A. Physicochemistry and Zooplankton of Two Karstic Sinkholes in the Yucatan Peninsula, Mexico. J. Limnol. 2015, 74, 382–393. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Reid, J.W.; Ilige, T.; Fiers, F. Catálogo de Los Copépodos (Crustacea) Continentales de La Península de Yucatán, México; Chetumal, Quintana Roo, México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: Ciudad de México, Mexico, 1996; 296p. [Google Scholar]
- Gómez-Márquez, J.L.; Peña-Mendoza, B.; Guzmán-Santiago, J.L.; Gallardo-Pineda, V. Composición, Abundancia Del Zooplancton y Calidad de Agua En Un Microreservorio En El Estado de Morelos. Hidrobiologica 2013, 23, 227–240. [Google Scholar]
- Gama Flores, J.L.; Sarma, S.S.S.; López Rocha, A.N.; Nandini, S. Effects of Cladoceran-Conditioned Medium on the Demography of Brachionid Rotifers (Rotifera: Brachionidae). Hydrobiologia 2018, 844, 21–30. [Google Scholar] [CrossRef]
- Uye, S.I. Replacement of Large Copepods by Small Ones with Eutrophication of Embayments: Cause and Consequence. Hydrobiologia 1994, 292–293, 513–519. [Google Scholar] [CrossRef]
- Basterrechea-Díaz, M. El Lago de Amatitlán: Década de Estudios Limnológicos 1985–1995; Academia de Ciencias Médicas, Físicas y Naturales de Guatemala: Guatemala City, Guatemala, 1997; 45p. [Google Scholar]
- Sigui, N. ¿Por Qué Continúa La Contaminación de Aguas En Guatemala? Cienc. Tecnol. Salud 2016, 3, 167–176. [Google Scholar] [CrossRef]
- Rodas-Pernillo, E.; Vasquez-Moscoso, C.A.; García, O.F. Dinámica Del Consumo y Aporte de Nutrientes de Fitoplancton, Dominado Por Microcystis sp. (Cyanophyceae) Del Lago de Amatitlán. Cienc. Tecnol. Salud 2020, 7, 2409–3459. [Google Scholar]
- Richardson-Varas, R.; Muñoz-Luza, M.; Landeros-Cáceres, F.; Contreras-Celis, V.; Carranza-González, J.; Nuñez, M.; Hernández, E.; Alfonso-Álvarez, R.; Ramos, E.; Mazariegos, J.; et al. Índice de Fragilidad Ambiental En Las Cuencas Hidro-Geomorfológicas Del Lago Peñuelas, Chile y Del Lago Amatitlán, Guatemala. Rev. Geográfica 2015, 156, 98–109. [Google Scholar]
- Brandorff, G.O. Distribution of Some Calanoida (Crustacea: Copepoda) from the Yucatán Peninsula, Belize and Guatemala. Rev. Biol. Trop. 2012, 60, 187–202. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Martínez, A.; Elías-Gutiérrez, M.; Suárez-Morales, E. Limnological and Morphometrical Data of Eight Karstic Systems “cenotes” of the Yucatan Peninsula, Mexico, during the Dry Season (February–May, 2001). Hydrobiologia 2002, 482, 167–177. [Google Scholar] [CrossRef]
- Schmitter-Soto, J.J.; Comín, F.A.; Escobar-Briones, E.; Herrera-Silveira, J.; Alcocer, J.; Suárez-Morales, E.; Elías-Gutiérrez, M.; Díaz-Arce, V.; Marín, L.E.; Steinich, B. Hydrogeochemical and Biological Characteristics of Cenotes in the Yucatan Peninsula (SE Mexico). Hydrobiologia 2002, 467, 215–228. [Google Scholar] [CrossRef]
- Jiménez, S.; Juárez, J.; Trujillo, L.; Dubón, S.; Valenzuela, O.; Castro, A.M. Calidad de Agua de La Cuenca y Lago de Amatitlán; División de Control, Calidad Ambiental y Manejo de Lagos: Guatemala City, Guatemala, 2015; 32p. [Google Scholar]
- Elías-Gutiérrez, M.; Kotov, A.A.; Garfias-Espejo, T. Cladocera (Crustacea: Ctenopoda, Anomopoda) from Southern Mexico, Belize and Northern Guatemala, with Some Biogeographical Notes. Zootaxa 2006, 1119, 1–27. [Google Scholar] [CrossRef]
- García-Morales, A.E.; Elías-Gutiérrez, M. The Rotifer Fauna of Guatemala and Belize: Survey and Biogeographical Affinities. Rev. Biol. Trop. 2007, 55, 569–584. [Google Scholar]
- Pérez, L.; Bugja, R.; Lorenschat, J.; Brenner, M.; Curtis, J.; Hoelzmann, P.; Islebe, G.; Scharf, B.; Schwalb, A. Aquatic Ecosystems of the Yucatán Peninsula (Mexico), Belize, and Guatemala. Hydrobiologia 2011, 661, 407–433. [Google Scholar] [CrossRef]
- Cervantes-Martínez, A.; Elías-Gutiérrez, M.; Gutiérrez-Aguirre, M.A.; Kotov, A.A. Ecological Remarks on Mastigodiaptomus Nesus Bowman, 1986 (Copepoda: Calanoida) in a Mexican Karstic Sinkhole. Hydrobiologia 2005, 542, 95–102. [Google Scholar] [CrossRef]
- Ramírez García, P.; Nandini, S.; Sarma, S.S.S.; Robles Valderrama, E.; Cuesta, I.; Hurtado, M.D. Seasonal Varations of Zooplankton Abundance in the Freshwater Reservoir Valle de Bravo (Mexico). Hydrobiologia 2002, 467, 99–108. [Google Scholar] [CrossRef]
- Koste, W. Rotatoria: Die Rädertiere Mitteleuropas: Ein Bestimmunswerk Bengründet von Max Voigt Überordnung Monogononta; Gebrüder Borntraeger: Berlin, Germany; Stuttgart, Germany, 1978; 234p. [Google Scholar]
- Fontaneto, D.; De Smet, W.H. Rotifera. In Handbook of Zoology Gastrotricha, Cycloneuralia and Gnathifera; Rhaera, A.S., Ed.; De Gruyter-GmbH: Berlin, Germany, 2015; Volume 3, pp. 196–217. [Google Scholar]
- Elías-Gutiérrez, M.; Suárez-Morales, E.; Gutiérrez-Aguirre, M.A.; Silva-Briano, M.; Granados-Ramírez, J.G.; Garfias-Espejo, T. Cladocera y Copepoda de Las Aguas Continentales de México. Guía Ilustrada; Universidad Autónoma de México: Ciudad de México, Mexico, 2008; 322p. [Google Scholar]
- Suarez-Morales, E.; Gutiérrez-Aguirre, M.A.; Gómez, S.; Perbiche-Neves, G.; Previattelli, D.; Dos Santos-Silva, N.; da Rocha, C.E.F.; Mercado-Salas, N.F.; Manriquez, T.M.; Cruz-Quintana, Y.; et al. Class Copepoda. In Thorp and Covich’s Freshwater Invertebrates: Volume 5: Keys to Neotropical and Antarctic Fauna; Damborenea, C., Rogers, C.D., James, T., Eds.; Elsevier Science Publishing Co Inc.: San Diego, CA, USA, 2020; pp. 663–796. [Google Scholar]
- Juday, C. Limnological Studies on Some Lakes in Central America. Wisconsin Acad. Sci. Arts Lett. 1915, 18, 214–250. [Google Scholar]
- Wilson, M.S. New Species and Distribution Records of Diaptomid Copepods from the Marsh Collection in the United States National Museum. J. Washingt. Acad. Sci. 1941, 31, 509–515. [Google Scholar]
- Brezonik, P.L.; Fox, J.L. The Limnology of Selected Guatemalan Lakes. Hydrobiologia 1974, 45, 467–487. [Google Scholar] [CrossRef]
- Ellenberg, R.L. Limnology of Lake Amatitlán in Guatemala and Its Eutrophication Process. Ph.D. Thesis, Technical University of Berlin, Berlin, Germany, 2014; 128p. [Google Scholar]
- Frutos, S.M.; Poi, A.S.G.; Neiff, J.J. Zooplankton Abundance and Species Diversity in Two Lakes with Different Trophic States (Corrientes, Argentina) Abundância e Diversidade Específica Do Zooplâncton Em Dois Lagos. Acta Limnol. Bras. 2009, 21, 367–375. [Google Scholar]
- Moreno-Gutiérrez, R.M.; Sarma, S.S.S.; Sobrino-Figueroa, A.S.; Nandini, S. Population Growth Potential of Rotifers from a High Altitude Eutrophic Waterbody, Madín Reseivoir (State of Mexico, Mexico): The Importance of Seasonal Sampling. J. Limnol. 2020, 77, 441–451. [Google Scholar] [CrossRef]
- Obertegger, U.; Smith, H.A.; Flaim, G.; Wallace, R.L. Using the Guild Ratio to Characterize Pelagic Rotifer Communities. Hydrobiologia 2011, 662, 157–162. [Google Scholar] [CrossRef]
- Reid, J.W. Arctodiaptomusdorsalis (Marsh): A Case History of Copepod Dispersal. Banisteria 2007, 1860, 3–18. [Google Scholar]
- Papa, R.D.S.; Li, H.; Tordesillas, D.T.; Han, B.; Dumont, H.J. Massive Invasion of Arctodiaptomus dorsalis (Copepoda, Calanoida, Diaptomidae) in Philippine Lakes: A Threat to Asian Zooplankton Biodiversity? Biol. Invasions 2012, 14, 2471–2478. [Google Scholar] [CrossRef]
- Metillo, E.B.; Masorong, A.M.; Macabangkit, S.A.N.; Licayan, J.R.U.; Tordesillas, D.T.; Papa, R.D.S. First Record of the Invasive Arctodiaptomus dorsalis (Marsh, 1907) (Copepoda: Calanoida: Diaptomidae) in Lake Lanao (Mindanao Is., Philippines). Acta Manila. Ser. A 2014, 62, 19–23. [Google Scholar]
- Perbiche-Neves, G.; Previattelli, D.; Pie, M.R.; Duran, A.; Suárez-Morales, E.; Boxshall, G.A.; Nogueira, M.G.; da Rocha, C.E.F. Historical Biogeography of the Neotropical Diaptomidae (Crustacea: Copepoda). Front. Zool. 2014, 11, 36. [Google Scholar] [CrossRef]
- Elías-Gutiérrez, M.; Jerónimo, F.M.; Ivanova, N.V.; Valdez-Moreno, M.; Hebert, P.D.N. DNA Barcodes for Cladocera and Copepoda from Mexico and Guatemala, Highlights and New Discoveries. Zootaxa 2008, 1839, 1–42. [Google Scholar] [CrossRef]
- Gutiérrez-Aguirre, M.A.; Cervantes-Martínez, A.; Elías-Gutiérrez, M.; Lugo-Vázquez, A. Remarks on Mastigodiaptomus (Calanoida: Diaptomidae) from Mexico Using Integrative Taxonomy, with a Key of Identification and Three New Species. PeerJ 2020, 8, e8416. [Google Scholar] [CrossRef]
- Gutiérrez-Aguirre, M.A.; Suárez-Morales, E. The Eurasian Thermocyclops crassus (Fischer, 1853) (Copepoda, Cyclopoida) Found in Southeastern Mexico. Crustaceana 2000, 73, 705–713. [Google Scholar] [CrossRef]
- Gutiérrez-Aguirre, M.A.; Reid, J.W.; Suárez-Morales, E. An Afro-Asian Species of Mesocyclops (Copepoda: Cyclopoida) in Central America and Mexico. J. Crustacean Biol. 2003, 23, 352–363. [Google Scholar] [CrossRef][Green Version]
- Collado, C.; Defaye, D.; Dussart, B.H.; Fernando, C.H. The Freshwater Copepoda (Crustacea) of Costa Rica with Notes on Some Species. Hydrobiologia 1984, 119, 89–99. [Google Scholar] [CrossRef]
- Gutiérrez-Aguirre, M.A.; Suárez-Morales, E.; Cervantes-Martínez, A.; Elías-Gutiérrez, M.; Previattelli, D. The Neotropical Species of Mesocyclops (Copepoda, Cyclopoida): An Upgraded Identification Key and Comments on Selected Taxa. J. Nat. Hist. 2006, 40, 549–570. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Gutiérrez-Aguirre, M.A.; Elías-Gutiérrez, M. Observations on the Structure of Mandibular Gnathobase in Some American Mesocyclops (Copepoda: Cyclopidae). Proc. Biol. Soc. Wash. 2003, 116, 742–753. [Google Scholar]
- Cervantes-Martínez, A.; Gutiérrez-Aguirre, M.A.; Delgado-Blas, V.H.; Ruíz-Ramírez, J.-D. Especies de Zooplancton Dulceacuícola de Cozumel; Quintana Roo, Universidad de Quintana Roo (UQROO): Cozumel, Mexico; Chetumal, Mexico, 2018; 86p. [Google Scholar]
- Connolly, J.K.; Watkins, J.M.; Hinchey, E.K.; Rudstam, L.G.; Reid, J.W. New Cyclopoid Copepod (Thermocyclops crassus) Reported in the Laurentian Great Lakes. J. Great Lakes Res. 2017, 43, 198–203. [Google Scholar] [CrossRef]
- Verbitsky, V.B.; Lazareva, V.I.; Medyantseva, E.N.; Malysheva, O.A.; Zhdanova, S.M.; Verbitskaya, T.I.; Grishanin, A.K. The Preferred and Avoidance Temperatures of Thermocyclops crassus (Fischer, 1853) and Their Relation to the Temperature of Optimal, Pessimal and Normal Performance of the Species. J. Therm. Biol. 2018, 78, 106–113. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).