Diversity and Abundance of Roadkilled Bats in the Brazilian Atlantic Forest
Abstract
:1. Introduction
2. Materials and Methods
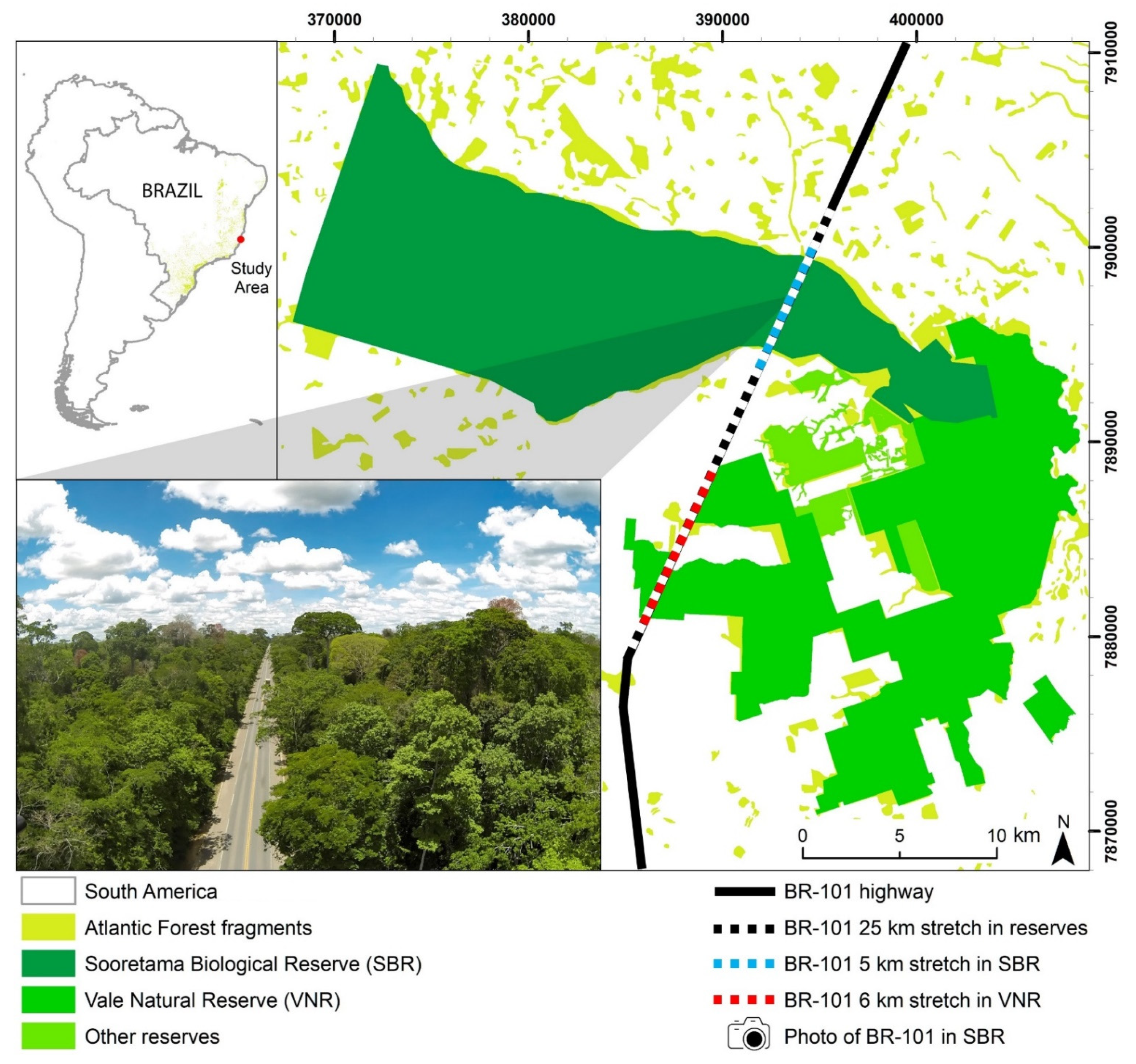
2.1. Study Area
2.2. Data Collection
2.3. Species Identification
2.4. Data Analysis
3. Results
3.1. Bat Diversity
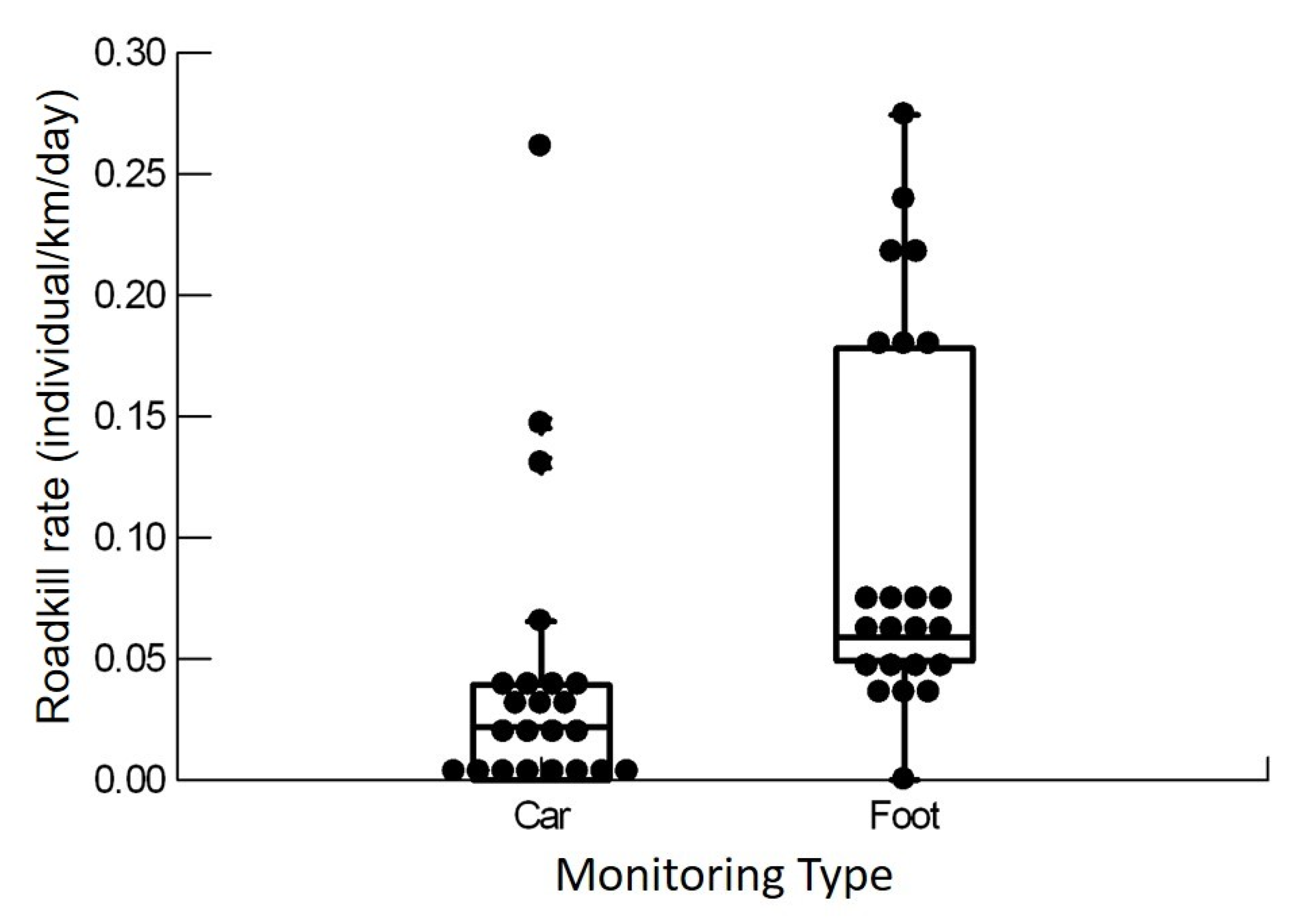
3.2. Diversity and Abundance by Method and Monitoring Stretch
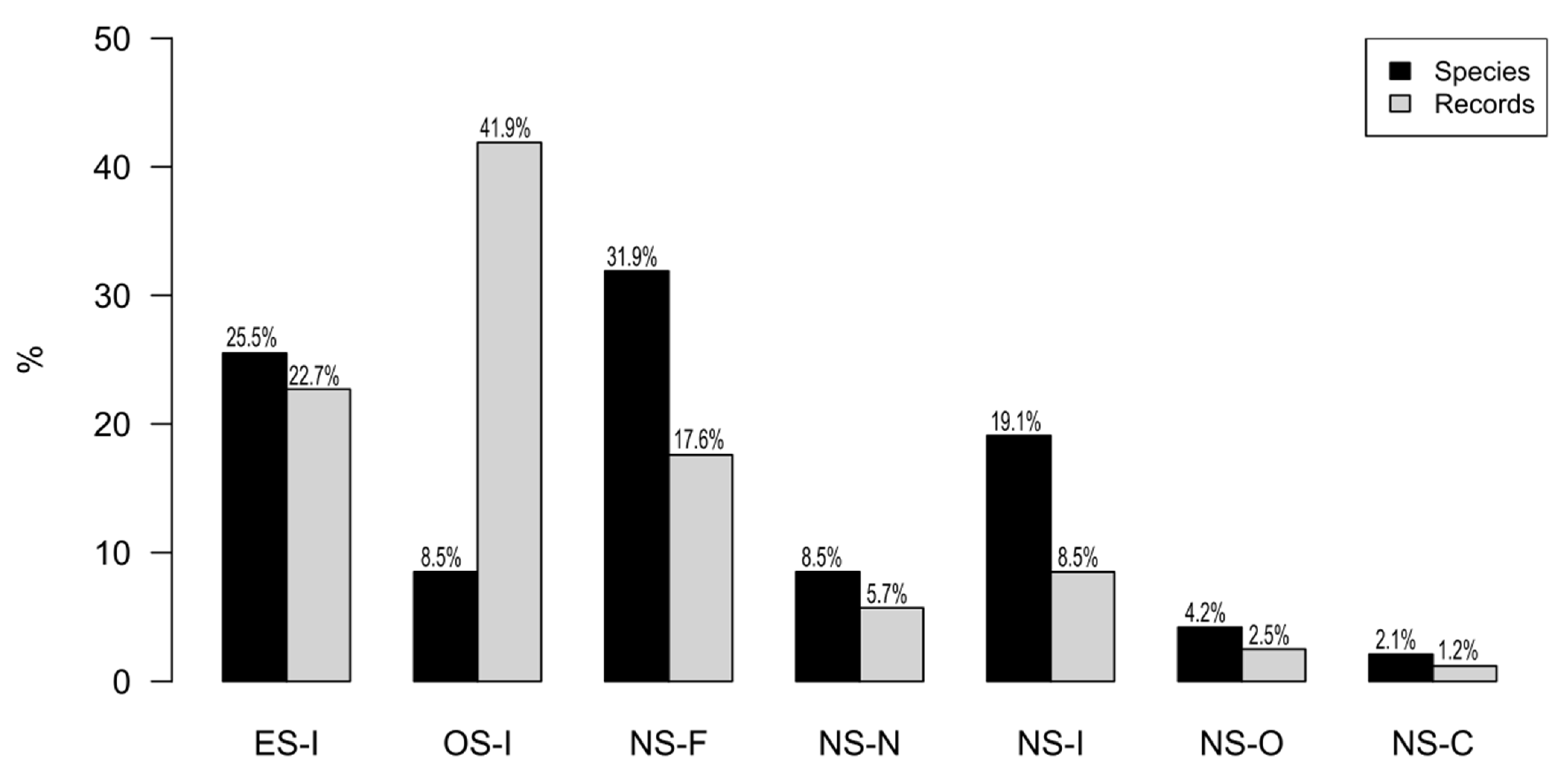
3.3. Feeding Guild and Flight Type
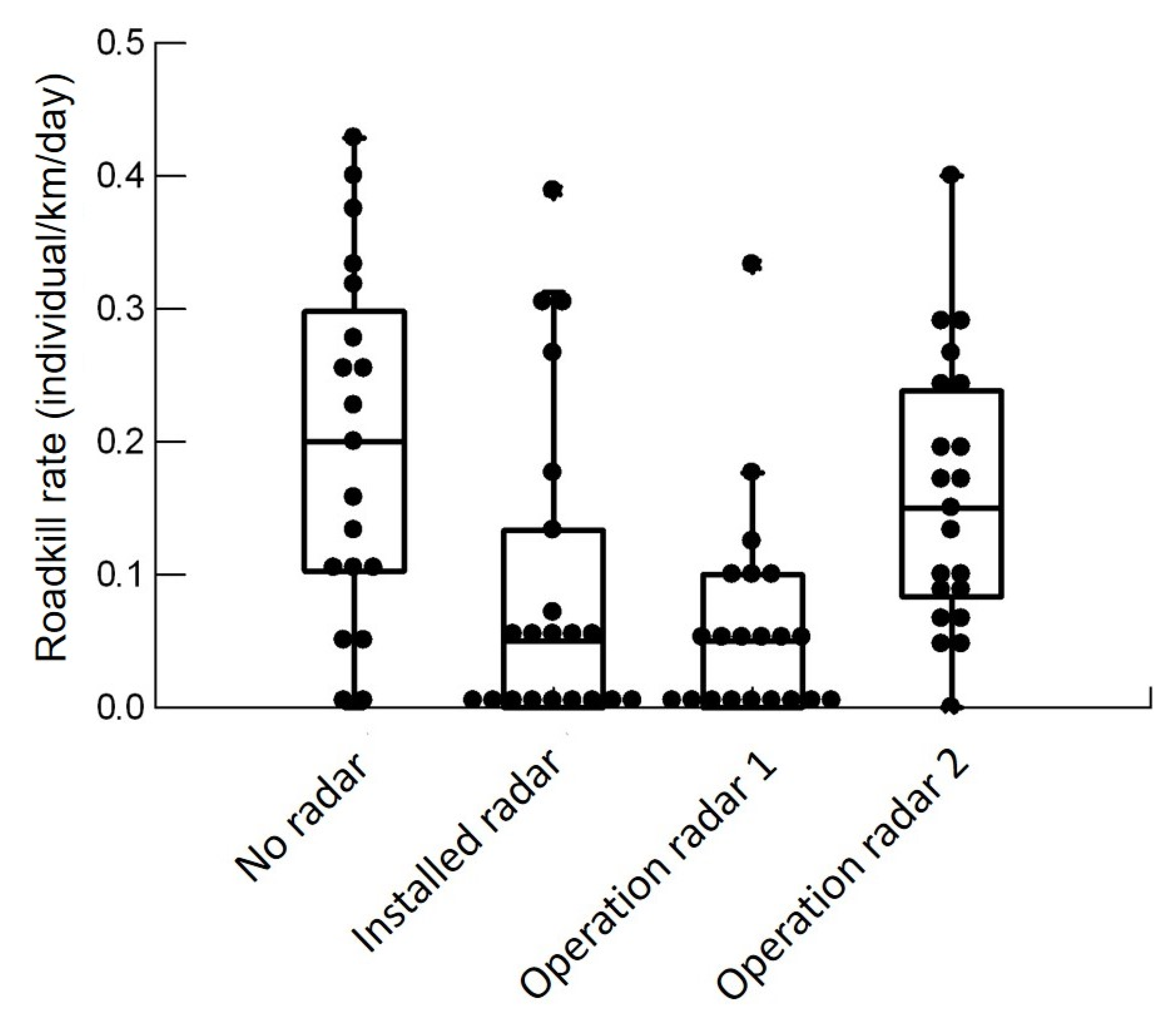
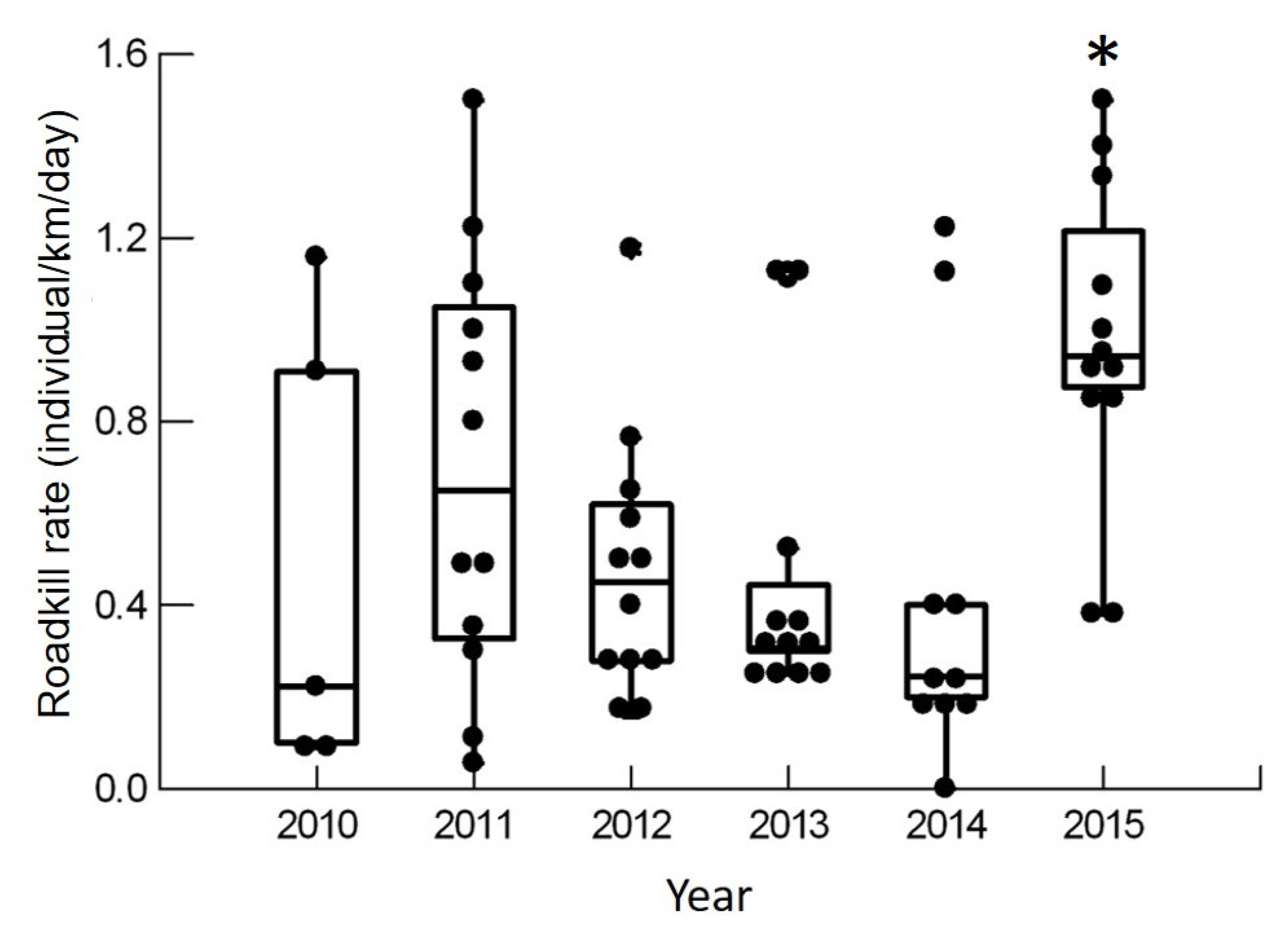
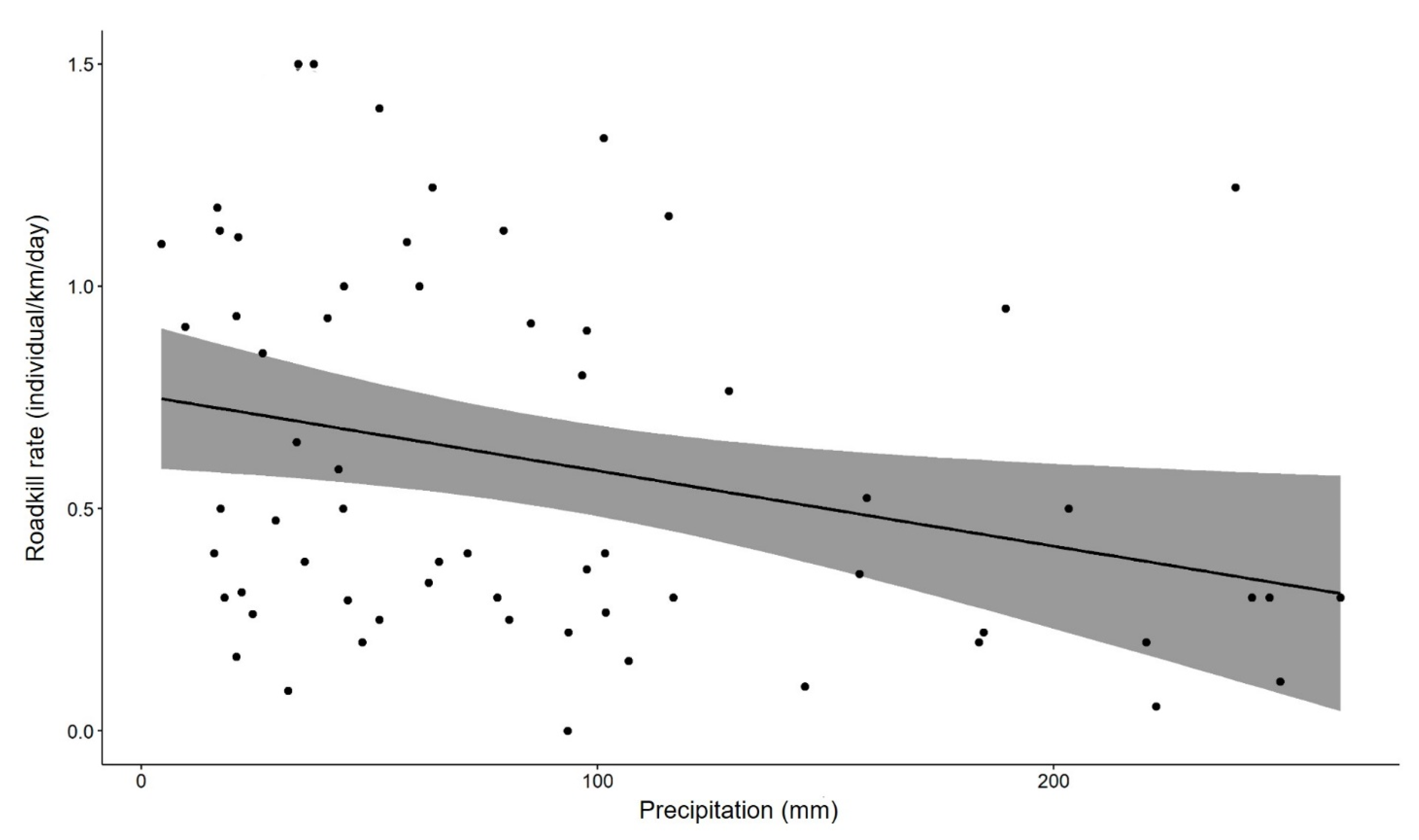
3.4. Rates and Pattern of Roadkill
4. Discussion
4.1. Roadkilled Bat Diversity
4.2. Feeding Guild and Type of Flight of Roadkilled Bats
4.3. Roadkill Rates and Pattern
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Forman, R.T.T.; Alexander, L.E. Roads and Their Major Ecological Effects. Annu. Rev. Ecol. Syst. 1998, 29, 207–231. [Google Scholar] [CrossRef] [Green Version]
- Trombulak, C.S.; Frissel, A.C. Review of Ecological Effects of Roads on Terrestrial and Aquatic Communities. Conserv. Biol. 2000, 14, 18–30. [Google Scholar] [CrossRef]
- Coffin, A.W. From roadkill to road ecology: A review of the ecological effects of roads. J. Transp. Geogr. 2007, 15, 396–406. [Google Scholar] [CrossRef]
- Laurance, W.F.; Goosem, M.; Laurance, S.G.W. Impacts of roads and linear clearings on tropical forests. Trends Ecol. Evol. 2009, 24, 659–669. [Google Scholar] [CrossRef]
- Balkenhol, N.; Waits, L.P. Molecular road ecology: Exploring the potential of genetics for investigating transportation impacts on wildlife. Mol. Ecol. 2009, 18, 4151–4164. [Google Scholar] [CrossRef]
- Holderegger, R.; Di Giulio, M. The genetic effects of roads: A review of empirical evidence. Basic Appl. Ecol. 2010, 11, 522–531. [Google Scholar] [CrossRef]
- Altringham, J.; Kerth, G. Bats and Roads. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Tigga, K., Eds.; Springer Nature: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Ramalho, D.F.; Aguiar, L.M.S. Bats on the Road—A Review of the Impacts of Roads and Highways on Bats. Acta Chiropterol. 2020, 22, 417–433. [Google Scholar] [CrossRef]
- Burgin, C.J.; Colella, J.P.; Kahn, P.L.; Upham, N.S. How many species of mammals are there? J. Mammal. 2018, 99, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Rackow, W.; Harz, O.; Schlegel, D. Fledermäuse (Chiroptera) als Verkehrsopfer in Niedersachsen. Nyctalus 1994, 5, 11–18. [Google Scholar]
- Kiefer, A.; Merz, H.; Rackow, W.; Roer, H.; Schlegel, D. Bats as traffic casualties in Germany. Myotis 1995, 32–33, 215–220. [Google Scholar]
- Haensel, J.; Rackow, W. Fledermause als Verkehrsopferein neuer report. Nyctalus 1996, 6, 29–47. [Google Scholar]
- Bafaluy, J.J. Mortandad De Murciélagos Por Atropello En Carreteras Del Sur De La Provincia De Huesca. Galemys 2000, 12, 15–23. [Google Scholar]
- Capo, G.; Chaut, J.J.; Arthur, L. Quatre ans d’étude de mortalité des Chiroptères sur deux kilomèters routiers proches d’un site d’hibernation. Symbioses 2006, 15, 45–46. [Google Scholar]
- Lesiński, G. Bat road casualties and factors determining their number. Mammalia 2007, 71, 138–142. [Google Scholar] [CrossRef]
- Lesiński, G. Linear landscape elements and bat casualties on roads—An example. Ann. Zool. Fenn. 2008, 45, 277–280. [Google Scholar] [CrossRef]
- Gaisler, J.; Řehák, Z.; Bartonička, T. Bat casualties by road traffic (Brno-Vienna). Acta Theriol. 2009, 54, 147–155. [Google Scholar] [CrossRef]
- Russell, A.; Butchkoski, C.; Saidak, L.; McCracken, G. Road-killed bats, highway design, and the commuting ecology of bats. Endanger. Species Res. 2009, 8, 49–60. [Google Scholar] [CrossRef]
- Lesiński, G.; Sikora, A.; Olszewski, A. Bat casualties on a road crossing a mosaic landscape. Eur. J. Wildl. Res. 2011, 57, 217–223. [Google Scholar] [CrossRef] [Green Version]
- Medinas, D.; Marques, J.T.; Mira, A. Assessing road effects on bats: The role of landscape, road features, and bat activity on road-kills. Ecol. Res. 2013, 28, 227–237. [Google Scholar] [CrossRef] [Green Version]
- Iković, V.; Đurović, M.; Prestenik, P. First data on bat traffic casualties in Montenegro. Vespertilio 2014, 17, 89–94. [Google Scholar]
- Parise, C. Etude de la mortalité des chauves-souris (etautres animaux sauvages) par collision routière das deus secteurs de Champagne-Ardenne. Naturale 2014, 5, 58–64. [Google Scholar]
- Alves, D.M.D.; Barros, R.F.; Nepomuceno, C. Levantamento de vertebrados silvestres atropelados com enfoque em indivíduos da ordem Chiroptera: Estudo de caso da rodovia MGC-354, Minas Gerais, Brasil. Perquirere 2015, 12, 176–193. [Google Scholar]
- Ceron, K.; Bôlla, D.A.S.; Mattia, D.L.; de Carvalho, F.; Zocche, J.J. Roadkilled bats (Mammalia: Chiroptera) in two highways of Santa Catarina state, Southern Brazil. Oecol. Aust. 2017, 21, 207–212. [Google Scholar] [CrossRef]
- Secco, H.; Gomes, L.A.; Lemos, H.; Mayer, F.; Machado, T.; Guerreiro, M.; Gregorin, R. Road and landscape features that affect bat roadkills in Southeastern Brazil. Oecol. Aust. 2017, 21, 323–336. [Google Scholar] [CrossRef]
- Grilo, C.; Coimbra, M.R.; Cerqueira, R.C.; Barbosa, P.; Dornas, R.A.P.; Gonçalves, L.O.; Teixeira, F.Z.; Coelho, I.P.; Schmidt, B.R.; Pacheco, D.L.K.; et al. Brazil road-kill: A data set of wildlife terrestrial vertebrate road-kills. Ecology 2018, 99, 2625. [Google Scholar] [CrossRef]
- Novaes, R.L.M.; Laurindo, R.S.; Dornas, R.A.P.; Esbérard, C.E.L.; Bueno, C. On a collision course: The vulnerability of bats to roadkills in Brazil. Mastozool. Neotrop. 2018, 25, 115–128. [Google Scholar] [CrossRef]
- Stoianova, D.; Karaivanov, N.; Simov, N. Bat (Mammalia: Chiroptera) road casualties in the Kresna Gorge, Bulgaria. In ARPHA Conference Abstracts; Pensoft Publishers: Sofia, Bulgaria, 2019; Volume 2, p. e46469. [Google Scholar] [CrossRef]
- Ramalho, D.F.; Resende, D.; de Oliveira, T.F.; Santos, R.A.L.; Aguiar, L.M.S. Factors influencing bat road casualties in a Neotropical savanna. Perspect. Ecol. Conserv. 2021, 19, 189–194. [Google Scholar] [CrossRef]
- Clare, E.L.; Lim, B.K.; Fenton, M.B.; Hebert, P.D.N. Neotropical Bats: Estimating Species Diversity with DNA Barcodes. PLoS ONE 2011, 6, e22648. [Google Scholar] [CrossRef]
- Nogueira, M.R.; de Lima, I.P.; Moratelli, R.; Tavares, V.D.C.; Gregorin, R.; Peracchi, A.L. Checklist of Brazilian bats, with comments on original records. Check List 2014, 10, 808–821. [Google Scholar] [CrossRef] [Green Version]
- Varzinczak, L.H.; Bernardi, I.P.; Passos, F.C. Is the knowledge of bat distribution in the Atlantic Rainforest sufficient? Comments about new findings and a case study in the Paraná State coastal area, Brazil. Mammalia 2015, 80, 263–269. [Google Scholar] [CrossRef]
- Graipel, M.E.; Cherem, J.J.; Monteiro-Filho, E.L.A.; Carmignotto, A.P. Mamíferos da Mata Atlântica. In Revisões Em Zoologia: Mata Atlântica; Monteiro-Filho, E., Conte, C., Eds.; Universidade Federal do Paraná: Curitiba, Brazil, 2017; pp. 391–482. [Google Scholar]
- Mittermeier, R.A.; Giland, P.R.; Mittermeier, G.G. Megadiversity—Earth’s Biologically Wealthiest Nations; CEMEX and Agrupacion Sierra Madre: Monterrey, Mexico, 1997. [Google Scholar]
- Garbino, G.S.T.; Gregorin, R.; Lima, I.P.; Loureiro, L.; Moras, L.M.; Moratelli, R.; Nogueira, M.R.; Pavan, A.C.; Tavares, V.C.; Nascimento, M.C.; et al. Updated Checklist of Brazilian Bats: Versão 2020. Comitê da Lista de Morcegos do Brasil—CLMB. Sociedade Brasileira Para o Estudo de Quirópteros (Sbeq). Available online: https://www.sbeq.net/lista-de-especies (accessed on 22 September 2020).
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.W.; Banhos, A.; Barbosa, N.P.U.; Barbosa, M.; Bergallo, H.G.; Loureiro, C.G.; Overbeck, G.E.; Solar, R.; Strassburg, B.B.N.; Vale, M.M. Restoring Brazil’s road margins could help the country offset its CO2 emissions and comply with the Bonn and Paris Agreements. Perspect. Ecol. Conserv. 2018, 16, 105–112. [Google Scholar] [CrossRef]
- Dean, W. A Ferro e Fogo: A História e a Devastação da Floresta Atlântica Brasileira; Companhia das Letras: São Paulo, Brazil, 2004. [Google Scholar]
- Ribeiro, M.C.; Metzger, J.P.; Martensen, A.C.; Ponzoni, F.J.; Hirota, M.M. The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biol. Conserv. 2009, 142, 1141–1153. [Google Scholar] [CrossRef]
- Instituto Brasileiro de Desenvolvimento Florestal. Plano de Manejo Reserva Biológica de Sooretama; Instituto Brasileiro de Desenvolvimento Florestal: Brasília, Brazil, 1981. [Google Scholar]
- United Nations Educational, Scientific and Cultural Organization. Twenty-Third Session of the World Heritage Committee—Marrakesh, Morocco. 1999. Available online: http://whc.unesco.org/archive/1999/whc-99-conf209-22e.pdf (accessed on 22 September 2020).
- Klippel, A.H.; Oliveira, P.V.; Britto, K.B.; Freire, B.F.; Moreno, M.R.; dos Santos, A.R.; Banhos, A.; Paneto, G.G. Using DNA Barcodes to Identify Road-Killed Animals in Two Atlantic Forest Nature Reserves, Brazil. PLoS ONE 2015, 10, e0134877. [Google Scholar] [CrossRef] [PubMed]
- Srbek-Araujo, A.; Mendes, S.; Chiarello, A. Jaguar (Panthera onca Linnaeus, 1758) roadkill in Brazilian Atlantic Forest and implications for species conservation. Braz. J. Biol. 2015, 75, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Rolim, S.G.; Menezes, L.F.T.; Srbek-Araujo, A.C. Floresta Atlântica de Tabuleiro: Diversidade e Endemismo na Reserva Natural Vale; Editora Rona: Belo Horizonte, Brazil, 2016. [Google Scholar]
- Jesus, R.M.; Souza, A.L.; Garcia, A. Produção Sustentável de Floresta Atlântica, Documento Sociedade de Investigações Florestais; Sociedade de Investigações Florestais: Viçosa, Brazil, 1992. [Google Scholar]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.D.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z. 2013, 22, 711–728. [Google Scholar] [CrossRef]
- Engel, V.L.; Martins, F.R. Reproductive phenology of Atlantic forest tree species in Brazil: An eleven year study. Trop. Ecol. 2005, 46, 1–16. [Google Scholar]
- Jesus, R.M.; Rolim, S.G. Fitossociologia da Mata Atlântica de Tabuleiro; Boletim Técnico da Sociedade de Investigações Florestais: Viçosa, Brazil, 2005. [Google Scholar]
- Instituto Nacional de Meteorologia. Dados Meteorológicos. Available online: https://tempo.inmet.gov.br/ (accessed on 22 September 2020).
- Díaz, M.M.; Solari, S.; Aguirre, L.F.; Aguiar, L.M.S.; Barquez, R.M. Clave de Identificación de los Murciélagos de Sudamérica/Chave de Identificação dos Morcegos da América Do Sul, 2nd ed.; Programa de Conservación de los Murciélagos de Argentina: Tucumán, Argentina, 2016. [Google Scholar]
- Gregorin, R.; Taddei, V. Chave artificial para a identificação de molossídeos brasileiros (Mammalia, Chiroptera). Mastozool. Neotrop. 2002, 9, 13–32. [Google Scholar]
- Ratnasingham, S.; Hebert, P.D. BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef] [Green Version]
- Benson, D.A.; Cavanaugh, M.; Clark, K.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Sayers, E.W. GenBank. Nucleic Acids Res. 2012, 41, 36–42. [Google Scholar] [CrossRef] [Green Version]
- Reis, N.R.; Fregonezi, M.N.; Peracchi, A.L.; Shibatta, O.A. Morcegos do Brasil: Guia de Campo; Technical Books Editora: Rio de Janeiro, Brazil, 2013. [Google Scholar]
- Kalko, E.K.V.; Villegas, S.E.; Schmidt, M.; Wegmann, M.; Meyer, C.F.J. Flying high—Assessing the use of the aerosphere by bats. Integr. Comp. Biol. 2008, 48, 60–73. [Google Scholar] [CrossRef] [Green Version]
- Pimenta, V.T. Segregação de Recursos por Diferentes Espécies de Morcegos (Mammalia: Chiroptera) na Reserva Biológica de Sooretama. Master’s Thesis, Universidade Federal do Espírito Santo, Vitória, Brazil, 2013. [Google Scholar]
- Gnocchi, A.P.; Huber, S.; Srbek-Araujo, A.C. Diet in a bat assemblage in Atlantic Forest in southeastern Brazil. Trop. Ecol. 2019, 60, 389–404. [Google Scholar] [CrossRef]
- Peracchi, A.L.; Nogueira, M.R.; de Lima, I.P. Novos achegos à lista dos quirópteros do município de Linhares, estado do Espírito Santo, sudeste do Brasil (Mammalia, Chiroptera). Chiropt. Neotrop. 2011, 17, 842–852. [Google Scholar]
- Hoppe, J.P.M.; Pimenta, V.T.; Ditchfield, A.D. First occurrence of the recently described Patricia’s Disk-winged bat Thyroptera wynneae (Chiroptera: Thyropteridae) in Espírito Santo, southeastern Brazil. Check List 2014, 10, 645–647. [Google Scholar] [CrossRef]
- Vela-Ulian, C.M.; Hoppe, M.J.P.; Ditchfield, A.D. New records of bats (Chiroptera) in the Atlantic Forest of Espírito Santo, southeastern Brazil. Mammalia 2021, 85, 52–63. [Google Scholar] [CrossRef]
- Emmons, L.; Feer, F. Neotropical Rainforest Mammals: A Field Guide, 2nd ed.; University of Chicago Press: Chicago, IL, USA, 1997. [Google Scholar]
- Keeley, B.; Tuttle, M. Bats in American Bridges; Bat Conservation International: Austin, TX, USA, 1999. [Google Scholar]
- Russo, D.; Cistrone, L.; Jones, G. Emergence time in forest bats: The influence of canopy closure. Acta Oecol. 2007, 31, 119–126. [Google Scholar] [CrossRef]
- Amorim, F.; Alves, P.; Rebelo, H. Bridges over the troubled Conservation of Iberian Bats. Barbastella 2013, 6, 3–12. [Google Scholar] [CrossRef]
- Instituto Chico Mendes de Conservação da Biodiversidade. Livro Vermelho da Fauna Brasileira Ameaçada de Extinção, 1st ed.; Instituto Chico Mendes de Conservação da Biodiversidade: Brasília, Brazil, 2018.
- Santos, S.M.; Carvalho, F.; Mira, A. How Long Do the Dead Survive on the Road? Carcass Persistence Probability and Implications for Road-Kill Monitoring Surveys. PLoS ONE 2011, 6, e25383. [Google Scholar] [CrossRef] [Green Version]
- Popovici, P.V.; Ile, G.A. Variations of road mortality in 24 hours on a local road from Eastern Romania: Implications for monitoring. S. West. J. Hortic. Biol. Environ. 2018, 9, 35–46. [Google Scholar]
- Péron, G.; Hines, J.E.; Nichols, J.D.; Kendall, W.L.; Peters, K.A.; Mizrahi, D.S. Estimation of bird and bat mortality at wind-power farms with superpopulation models. J. Appl. Ecol. 2013, 50, 902–911. [Google Scholar] [CrossRef]
- Avila-Flores, R.; Fenton, M.B. Use of Spatial Features by Foraging Insectivorous Bats in a Large Urban Landscape. J. Mammal. 2005, 86, 1193–1204. [Google Scholar] [CrossRef]
- Jung, K.; Kalko, E.K.V. Where forest meets urbanization: Foraging plasticity of aerial insectivorous bats in an anthropogenically altered environment. J. Mammal. 2010, 91, 144–153. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Soanes, K.; Straka, T.M.; Lahoz-Monfort, J.J.; Lumsden, L.F.; van der Ree, R. Differential use of highway underpasses by bats. Biol. Conserv. 2017, 212, 22–28. [Google Scholar] [CrossRef]
- Berthinussen, A.; Altringham, J. The effect of a major road on bat activity and diversity. J. Appl. Ecol. 2012, 49, 82–89. [Google Scholar] [CrossRef]
- Fensome, A.G.; Mathews, F. Roads and bats: A meta-analysis and review of the evidence on vehicle collisions and barrier effects. Mamm. Rev. 2016, 46, 311–323. [Google Scholar] [CrossRef] [Green Version]
- Bennett, V.J.; Zurcher, A.A. When Corridors Collide: Road-Related Disturbance in Commuting Bats. J. Wildl. Manag. 2012, 77, 93–101. [Google Scholar] [CrossRef]
- Schaub, A.; Ostwald, J.; Siemers, B.M. Foraging bats avoid noise. J. Exp. Biol. 2008, 211, 3174–3180. [Google Scholar] [CrossRef] [Green Version]
- Siemers, B.M.; Schaub, A. Hunting at the highway: Traffic noise reduces foraging efficiency in acoustic predators. Proc. R. Soc. B 2011, 278, 1646–1652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stone, E.L.; Harris, S.; Jones, G. Impacts of artificial lighting on bats: A review of challenges and solutions. Mamm. Biol. 2015, 80, 213–219. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Soanes, K.; Lahoz-Monfort, J.J.; Lumsden, L.F.; van der Ree, R. Insectivorous bats are less active near freeways. PLoS ONE 2021, 16, e0247400. [Google Scholar] [CrossRef]
- Kunz, T.H.; de Torrez, E.B.; Bauer, D.; Lobova, T.; Fleming, T.H. Ecosystem services provided by bats. Ann. N. Y. Acad. Sci. 2011, 1223, 1–38. [Google Scholar] [CrossRef]
- Cleveland, C.J.; Betke, M.; Federico, P.; Frank, J.D.; Hallam, T.G.; Horn, J.; López, J.D.; McCracken, G.F.; Medellín, R.A.; Moreno-Valdez, A.; et al. Economic value of the pest control service provided by Brazilian free-tailed bats in south-central Texas. Front. Ecol. Environ. 2006, 4, 238–243. [Google Scholar] [CrossRef]
- Boyles, J.G.; Cryan, P.M.; McCracken, G.F.; Kunz, T.H. Economic Importance of Bats in Agriculture. Science 2011, 332, 41–42. [Google Scholar] [CrossRef]
- Teixeira, F.Z.; Coelho, A.V.P.; Esperandio, I.B.; Kindel, A. Vertebrate road mortality estimates: Effects of sampling methods and carcass removal. Biol. Conserv. 2013, 157, 317–323. [Google Scholar] [CrossRef]
- Branco, F.E.R.; dos Santos, A.R.; Pezzopane, J.E.M.; dos Santos, A.B.; Alexandre, R.S.; Bernardes, V.P.; da Silva, R.G.; de Souza, K.B.; Moura, M.M. Space-time analysis of vegetation trends and drought occurrence in domain area of tropical forest. J. Environ. Manag. 2019, 246, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, L.M.S.; Bernard, E.; Ribeiro, V.; Machado, R.B.; Jones, G. Should I stay or should I go? Climate change effects on the future of Neotropical savannah bats. Glob. Ecol. Conserv. 2016, 5, 22–33. [Google Scholar] [CrossRef] [Green Version]
- Chaverri, G.; Vonhof, M.J. Reproduction and growth in a Neotropical insectivorous bat. Acta Chiropterol. 2011, 13, 147–155. [Google Scholar] [CrossRef]
- Jones, D.N.; Griffiths, M.R.; Griffiths, J.R.; Hacker, J.L.F.; Hacker, J.B. Implications of upgrading a minor forest road on traffic and road-kill in southeast Queensland. Australas. J. Environ. Manag. 2014, 21, 429–440. [Google Scholar] [CrossRef]
- Filius, J.; Hoek, Y.; Jarrín-V, P.; Hooft, P. Wildlife roadkill patterns in a fragmented landscape of the Western Amazon. Ecol. Evol. 2020, 10, 6623–6635. [Google Scholar] [CrossRef]
- Claireau, F.; Bas, Y.; Julien, J.F.; Machon, N.; Allegrini, B.; Puechmaille, S.J.; Kerbiriou, C. Bat overpasses as an alternative solution to restore habitat connectivity in the context of road requalification. Ecol. Eng. 2019, 131, 34–38. [Google Scholar] [CrossRef]
| Roadkilled Species Grouped by Family and Subfamily | Spatial Habitat Use | Feeding Guild | SBR on Foot | SBR by Car | SBR Sporadic | VNR on Foot | VNR by Car | VNR Sporadic | 13.9 km by Car | 13.9 km Sporadic | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Emballonuridae | |||||||||||
| Unidentified species | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 | ||
| Emballonurinae | |||||||||||
| Centronycteris maximiliani (J. Fisher, 1829) | ES | I | 2 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 3 |
| Diclidurus albus Wied-Neuwied, 1820 | OS | I | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Rhynchonycteris naso (Wied-Neuwied, 1820) | ES | I | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Saccopteryx spp. | ES | I | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Saccopteryx bilineata (Temminck 1838) | ES | I | 8 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 10 |
| Saccopteryx leptura (Schreber, 1774) | ES | I | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Furipteridae | |||||||||||
| Furipterus horrens (F. Cuvier, 1828) | ES | I | 3 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 5 |
| Natalidae | |||||||||||
| Unidentified species | ES | I | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Molossidae | |||||||||||
| Unidentified species | OS | I | 62 | 4 | 1 | 1 | 0 | 0 | 0 | 1 | 69 |
| Molossus sp. | OS | I | 11 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 13 |
| Molossus molossus (Pallas, 1766) | OS | I | 36 | 4 | 5 | 4 | 1 | 1 | 4 | 10 | 65 |
| Molossus rufus E. Geoffroy, 1805 | OS | I | 2 | 2 | 0 | 0 | 0 | 0 | 1 | 0 | 5 |
| Nyctinomops sp. | OS | I | 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 |
| Nyctinomops laticaudatus (E. Geoffroy, 1805) | OS | I | 3 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 6 |
| Promops nasutus (Spix, 1823) | OS | I | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| Phyllostomidae | |||||||||||
| Unidentified species | 13 | 2 | 1 | 2 | 0 | 0 | 0 | 0 | 18 | ||
| Carollinae | |||||||||||
| Carollia sp. | NS | F | 6 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 9 |
| Carollia perspicillata (Linnaeus, 1758) | NS | F | 2 | 1 | 1 | 1 | 1 | 0 | 0 | 2 | 8 |
| Rhinophylla pumilio Peters, 1865 | NS | F | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Glossophaginae | |||||||||||
| Unidentified species | NS | N | 5 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 7 |
| Anoura sp. | NS | N | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Anoura geoffroyi Gray, 1838 | NS | N | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 |
| Dryadonycteris capixaba (Nogueira, Lima, Peracchi & Simmons, 2012) | NS | N | 4 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 5 |
| Glossophaga soricina (Pallas, 1766) | NS | N | 0 | 0 | 0 | 1 | 0 | 0 | 2 | 1 | 4 |
| Lonchophylla sp. | NS | N | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Lonchophylla peracchii (Días, Esbérard & Moratelli, 2013) | NS | N | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 |
| Phyllostominae | |||||||||||
| Unidentified species | 8 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 9 | ||
| Lampronycteris brachyotis (Dobson, 1879) | NS | I | 2 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 4 |
| Lophostoma sp. | NS | I | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Lophostoma brasiliense Peters, 1867 | NS | I | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Macrophyllum macrophyllum (Schinz, 1821) | ES | I | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Micronycteris sp. | NS | I | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 |
| Micronycteris hirsuta (Peters, 1869) | NS | I | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Micronycteris megalotis (Gray, 1842) | NS | I | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Micronycteris minuta (Gervais, 1856) | NS | I | 2 | 0 | 1 | 2 | 0 | 0 | 0 | 0 | 5 |
| Micronycteris microtis Miller, 1898 | NS | I | 3 | 0 | 2 | 1 | 0 | 0 | 0 | 0 | 6 |
| Micronycteris schmidtorum Sanborn, 1935 | NS | I | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 3 |
| Gardnerycteris crenulatum E. Geoffroy, 1803 | NS | I | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 3 |
| Phyllostomus sp. | NS | O | 3 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 5 |
| Phyllostomus hastatus Pallas, 1767 | NS | O | 2 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 4 |
| Phyllostomus discolor Wagner, 1843 | NS | O | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Trachops cirrhosus (Spix,1823) | NS | C | 3 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 5 |
| Stenodermatinae | |||||||||||
| Unidentified species | F | 12 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 15 | |
| Dermanura sp. | NS | F | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Dermanura cinerea (Gervais, 1856) | NS | F | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| Dermanura gnoma (Handley, 1987) | NS | F | 1 | 1 | 2 | 0 | 0 | 0 | 0 | 0 | 4 |
| Artibeus sp. | NS | F | 3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 4 |
| Artibeus fimbriatus Gray, 1838 | NS | F | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Artibeus lituratus Olfers, 1818 | NS | F | 2 | 0 | 2 | 1 | 0 | 0 | 0 | 0 | 5 |
| Artibeus obscurus (Schinz, 1821) | NS | F | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Chiroderma villosum Peters, 1860 | NS | F | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 5 |
| Platyrrhinus sp. | NS | F | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Platyrrhinus recifinus (Thomas, 1901) | NS | F | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 |
| Platyrrhinus incarum (Thomas, 1912) | NS | F | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Platyrrhinus lineatus (E. Geoffroy, 1810) | NS | F | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 |
| Pygoderma bilabiatum (Wagner, 1843) | NS | F | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Sturnira lilium (E. Geoffroy, 1810) | NS | F | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Uroderma magnirostrum Davis, 1968 | NS | F | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Vampyressa pusilla (Wagner, 1843) | NS | F | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Thyropteridae | |||||||||||
| Thyroptera sp. | ES | I | 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 |
| Thyroptera wynneae Velazco et. al., 2014 | ES | I | 6 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 7 |
| Vespertilionidae | |||||||||||
| Unidentified species | ES | I | 9 | 1 | 2 | 3 | 0 | 0 | 0 | 0 | 15 |
| Eptesicus furinales (d’Orbigny & Gervais, 1847) | ES | I | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Lasiurus sp. | ES | I | 6 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 7 |
| Lasiurus blossevillii (Lesson & Garnot, 1826) | ES | I | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| Lasiurus ega (Gervais, 1856) | ES | I | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 4 |
| Myotis sp. | ES | I | 13 | 0 | 2 | 1 | 0 | 0 | 0 | 2 | 18 |
| Myotis nigricans (Schinz, 1821) | ES | I | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Myotis riparius Handley, 1960 | ES | I | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 |
| Unidentified species at taxonomic levels | 289 | 10 | 16 | 10 | 1 | 0 | 6 | 9 | 341 | ||
| Total | 592 | 35 | 58 | 33 | 3 | 1 | 16 | 35 | 773 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damásio, L.; Ferreira, L.A.; Pimenta, V.T.; Paneto, G.G.; dos Santos, A.R.; Ditchfield, A.D.; Bergallo, H.G.; Banhos, A. Diversity and Abundance of Roadkilled Bats in the Brazilian Atlantic Forest. Diversity 2021, 13, 335. https://doi.org/10.3390/d13070335
Damásio L, Ferreira LA, Pimenta VT, Paneto GG, dos Santos AR, Ditchfield AD, Bergallo HG, Banhos A. Diversity and Abundance of Roadkilled Bats in the Brazilian Atlantic Forest. Diversity. 2021; 13(7):335. https://doi.org/10.3390/d13070335
Chicago/Turabian StyleDamásio, Lucas, Laís Amorim Ferreira, Vinícius Teixeira Pimenta, Greiciane Gaburro Paneto, Alexandre Rosa dos Santos, Albert David Ditchfield, Helena Godoy Bergallo, and Aureo Banhos. 2021. "Diversity and Abundance of Roadkilled Bats in the Brazilian Atlantic Forest" Diversity 13, no. 7: 335. https://doi.org/10.3390/d13070335
APA StyleDamásio, L., Ferreira, L. A., Pimenta, V. T., Paneto, G. G., dos Santos, A. R., Ditchfield, A. D., Bergallo, H. G., & Banhos, A. (2021). Diversity and Abundance of Roadkilled Bats in the Brazilian Atlantic Forest. Diversity, 13(7), 335. https://doi.org/10.3390/d13070335







