Undara Lava Cave Fauna in Tropical Queensland with an Annotated List of Australian Subterranean Biodiversity Hotspots
Abstract
1. Introduction
2. Materials and Methods
3. Geology, Geomorphology, Hydrology
4. Environment
4.1. Surface
4.2. Underground
5. Results
5.1. Overview of Invertebrate Sampling in the Undara Basalt Lava Tube Systems
- Minimally 78 species of arthropods have been recorded from Undara Basalt lava tubes, along with a cavernicolous fern (Psilotum sp.) and four bat genera (Hipposideros, Miniopterus, Rhinolophus, Vespadelus).
- Of the arthropods: 16 (21%) have been taxonomically described; 30 (39%) identified to genus and/or morpho-species; and 32 (40%) remain unidentified to species or genus level.
- Seven caves harboured obligate subterranean species; all these caves contained deep zone environments with high humidity, of which three also contained bad air (CO2).
- Overall, 30 troglobionts and one stygobiont species were recorded in the Undara Basalt Flow (Table 3). Bayliss Cave harboured the most obligate subterranean species: 23 troglobionts and one stygobiont.
- Twenty-two species of arthropods and one plant are classified as troglophiles, being native cavernicoles capable of living their entire life cycle underground but populations of the same species may also be found in surface habitats (Table S1) [2,21,30,32,50,51,52,53,54]. Some of these may prove to be troglobionts once more is known of their biology.
- Twenty-five species of arthropods are classified as ‘visitors’ (Table S2) [2,30,35,55,56]. Some habitually use caves for shelter or to find food (=trogloxenes). Others occasionally enter caves for shelter, and some wander or fall into caves accidentally (=‘incidental’ or ‘accidental’ cavernicoles). In addition, unidentified mites (Arachnida: Acari) have been reported from most surveyed caves. Many are associated with guano, and a few are parasites of other cavernicoles including bats; however, because both the identity and ecological status of the mites recorded from Undara caves are unknown, they are not enumerated further in Table S2.
- Besides Bayliss, the other caves with a high diversity of troglobionts are Nasty Cave (eight species) and Barkers Cave (seven species), which are located, respectively, 3 km and 3.7 km downflow of Bayliss (Figure 4).
5.2. Bayliss Cave Environment
5.3. Bayliss Cave Invertebrate Fauna Distribution
5.4. Notable Cave Species
5.4.1. Aquatic Fauna
5.4.2. Terrestrial Fauna
6. Discussion
6.1. The Challenge of Classifying Cavernicoles
6.2. Why Is Bayliss Cave So Rich in Troglobionts?
6.3. Comparison with Other Subterranean Hotspots in Australia
- Undara remains the richest subterranean hotspot in humid tropical Australia, which far exceeds the richness of troglobionts recorded in other humid tropical Australian karsts such as Judburra-Gregory [89].
- In the arid Yilgarn and semi-arid Pilbara regions of Western Australia significantly richer subterranean assemblages have been documented in recent decades.
- Most Western Australian hotspots in calcretes and iron-ore terrains do not contain enterable caves, and fauna can only be collected by sampling mesocaverns and microcaverns via constructed wells and drill holes.
- In Eastern Australia, the Jenolan karst in New South Wales recorded the highest species richness (136 taxa) however the majority of these are accidentals and troglophiles, and only 8 taxa are obligate cavernicoles [100].
- Overall species richness in warm temperate (New South Wales) [101,102,103] and cool temperate (Tasmania) [104] karst areas is comparable (median 54 taxa), and these karsts harbour a much lower proportion of obligate subterranean species compared with arid and semi-arid regions, although Tasmania stands out in terms of troglobiont richness in temperate latitudes (maximum 25 species at Precipitous Bluff, Tasmania) [104,105,106,107,108].
- The Peel Valley alluvial aquifer in New South Wales harbours the richest known stygofauna assemblage in eastern Australia; 54 species including 33 stygobionts [109].
- In terms of obligate species, the richest Australian localities are in arid and semi-arid climate regions, where most of the troglobionts and stygobionts are relictual with no close surface relatives. Molecular phylogenetic studies have shown that Quaternary aridification is the likely driving mechanism for troglo/stygogenesis in these regions [110].
| Geographic/Climate Region | Locality Name; Geology, Hydrology Type | Total spp. Richness (No. Sb/Tb) * | Stygofauna spp. (Sb) * | Troglofauna spp. (Tb) * | Sources |
|---|---|---|---|---|---|
| Queensland, humid tropical | Undara Basalt lava tubes | 77 (31) | 1 (1) | 76 (30) | [2], and text |
| Northern Territory, humid tropical | Judbarra-Gregory karst | 56 (7) | 3 (2) | 53 (5) | [89] |
| Yilgarn, WA, arid | Yeelirrie calcrete aquifer | 115 (*) | 70 (*) | 45 (*) | |
| Yilgarn, WA, arid | Uramurdah calcrete | 45 (*) | 36 (*) | 9 (*) | [90,91,92] |
| Yilgarn, WA, arid | Hinkler Well calcrete | 41 (*) | 32 (*) | 9 (*) | [95] |
| Yilgarn, WA, arid | Lake Violet calcrete | 39 (*) | 35 (*) | 4 (*) | [95] |
| Yilgarn, WA, arid | Barwidgee calcrete | 37 (*) | 28 (*) | 9 (*) | [95] |
| Pilbara, WA, arid/semi-arid | Ethel Gorge calcrete aquifer | 84 (45) | 84 (45) | 0 | [95] |
| Pilbara, WA, arid/semi-arid | Cape Range karst | 83 (*) | 42 (*) | 41 (*) | |
| Pilbara, WA, arid/semi-arid | Barrow Island karst | 74 (*) | 56 (*) | 18 (*) | [94] |
| Pilbara, WA, arid/semi-arid | Well PSS016, Robe River calcrete aquifer | 54 (*) | 54 (*) | 0 | [82,98] |
| Pilbara, WA, arid/semi-arid | Mesa A iron pisolite, Robe Valley | 24 (*) | 0 | 24 (*) | [83,99] |
| New South Wales, warm temperate | Jenolan karst | 136 (8) | 10 (2) | 126 (6) | [85,97] |
| New South Wales, warm temperate | Wombeyan karst | 55 (7) | 5 (2) | 50 (5) | [96] |
| New South Wales, warm temperate | Wee Jasper karst | 53 (7) | 5 (3) | 48 (4) | |
| New South Wales, warm temperate | Peel Valley alluvial aquifer | 54 (33) | 54 (33) | 0 | [100] |
| Tasmania, cool temperate | Ida Bay karst | 65 (18) | 17 (6) | 48 (12) | [101,102,103] |
| Tasmania, cool temperate | Junee Florentine karst | 60 (20) | 17 (8) | 43 (12) | [101,102,103] |
| Tasmania, cool temperate | Precipitous Bluff karst | 37 (25) | 11 (11) | 26 (14) | [109] |
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Howarth, F.G. Environmental Ecology of North Queensland Caves: Why are there so many troglobites in Australia? In Proceedings of the Tropicon 1988, 17th Biennial Australian Speleological Conference, Tinaroo, QLD, Australia, 27–31 December 1988; pp. 76–84. [Google Scholar]
- Howarth, F.G.; Stone, F.D. Elevated carbon dioxide levels in Bayliss Cave, Australia: Implications for the evolution of obligate cave species. Pac. Sci. 1990, 44, 207–218. [Google Scholar]
- Culver, D.C.; Sket, B. Hotspots of subterranean biodiversity in caves and wells. J. Cave Karst Stud. 2000, 62, 11–17. [Google Scholar]
- Hoch, H.; Howarth, F.G. The evolution of cave-adapted cixiid planthoppers in volcanic and limestone caves in north Queensland, Australia (Homoptera: Fulgoroidea). Mem. Biospeol. 1989, 16, 17–24. [Google Scholar]
- Hoch, H.; Howarth, F.G. Reductive evolutionary trends in two new cavernicolous species of a new Australian cixiid genus (Homoptera: Fulgroidea). Syst. Entomol. 1989, 14, 179–196. [Google Scholar] [CrossRef]
- Howarth, F.G. High-stress subterranean habitats and evolutionary change in cave-inhabiting arthropods. Am. Nat. 1993, 142, S65–S77. [Google Scholar] [CrossRef] [PubMed]
- Hamilton-Smith, E. The Arthropoda of Australian caves. J. Aust. Entomol. Soc. 1967, 6, 103–118. [Google Scholar] [CrossRef]
- Humphreys, W.F. The significance of the subterranean fauna in biogeographical reconstruction: Examples from Cape Range Peninsula, Western Australia. Rec. West. Aust. Mus. Suppl. 1993, 45, 165–192. [Google Scholar]
- Guzik, M.T.; Austin, A.D.; Cooper, S.; Harvey, M.; Humpherys, W.F.; Bradford, T.; Eberhard, S.; King, R.A.; Leys, R.; Muirhead, K.A.; et al. Is the Australian subterranean fauna uniquely diverse? Invertebr. Syst. 2010, 24, 407–418. [Google Scholar] [CrossRef]
- Vandel, A. Biospeleology—The Biology of Cavernicolous Animals; Elsevier: London, UK, 1965. [Google Scholar]
- Barr, T.C.J. Cave ecology and the evolution of troglobites. In Evolutionary Biology; Hecht, M.K., Steere, W.C., Eds.; Appleton-Century-Crofts: New York, NY, USA, 1968; Volume 2, pp. 35–102. [Google Scholar]
- Howarth, F.G. The tropical cave environment and the evolution of troglobites. In Proceedings of the 9th Congreso Internacional de Espeleologia, Barcelona, Spain, 9–15 August 1986; pp. 153–155. [Google Scholar]
- Leleup, N. Premier Partie. In Mission Zoologique Belge aux iles Galapagos et en Ecuador (N. et J. Leleup, 1964–1965) Resutats Scientifiques; Koninklijk Museum voor Midden-Africa—Musee Royal de l’Afrique Centrale: Tervueren, Belgium, 1968; pp. 9–34. [Google Scholar]
- Howarth, F.G. Cavernicoles in lava tubes on the island of Hawaii. Science 1972, 75, 325–326. [Google Scholar] [CrossRef] [PubMed]
- Elias-Gutierrez, M.; Martinez Jeronimo, F.; Ivanova, N.V.; Valdez-Moreno, M.; Hebert, P.D.N. DNA barcodes for Cladocera and Copepoda from Mexico and Guatemala highlights and new discoveries. Zootaxa 2008, 1839, 1–42. [Google Scholar] [CrossRef]
- Martínez, A.; González, B.C. Volcanic Anchialine Habitats of Lanzarote. In Cave Ecology; Moldovan, O.T., Kovác, L., Halse, S.A., Eds.; Ecological Studies, 235; Springer: Cham, Switzerland, 2018; pp. 195–228. [Google Scholar]
- Peck, S.B. The Invertebrate Fauna of Tropical American Caves, Part III: Jamaica, An Introduction. Int. J. Speleol. 1975, 7, 303–326. [Google Scholar] [CrossRef]
- Culver, D.C.; Deharveng, L.; Bedos, A.; Lewis, J.J.; Madden, M.; Reddell, J.R.; Sket, B.; Trontelj, P.; White, D. The mid-latitude biodiversity ridge in terrestrial cave fauna. Ecography 2006, 29, 120–128. [Google Scholar] [CrossRef]
- Deharveng, L.; Bedos, A. The cave fauna of southeast Asia. Origin, evolution, and ecology. In Subterranean Ecosystems; Wilken, H., Culver, D.C., Humphreys, W.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; pp. 603–632. [Google Scholar]
- Soulier-Perkins, A. Phylogenetic evidence for multiple invasions and speciation in caves: The Australian planthopper genus Solonaima (Hemiptera: Fulgoromorpha: Cixiidae). Syst. Entomol. 2005, 30, 281–288. [Google Scholar] [CrossRef]
- Slaney, D.P.; Blair, D. Molecules and Morphology are Concordant in Discriminating among Populations of Cave Cockroaches in the Genus Paratemnopteryx Saussure (Blattodea: Blattellidae). Ann. Entomol. Soc. Am. 2000, 93, 398–404. [Google Scholar] [CrossRef]
- Medeiros, M.J.; Davis, D.; Howarth, F.G.; Gillespie, R. Evolution of cave living in Hawaiian Schrankia (Lepidoptera: Noctuidae) with description of a remarkable new cave species. Zool. J. Linn. Soc. 2009, 156, 114–139. [Google Scholar] [CrossRef]
- Howarth, F.G.; Hoch, H.; Wessel, A. Adaptive shifts. In Encyclopedia of Caves, 3rd ed.; Culver, D.C., White, W., Eds.; Academic Press: Burlington, MA, USA, 2019; pp. 47–55. [Google Scholar]
- Howarth, F.G. A comparison of volcanic and karstic cave communities. In Proceedings of the 7th International Symposium on Vulcanospeleology, Santa Cruz de la Palma, Canary Islands, Spain, 4–11 November 1994; pp. 63–68. [Google Scholar]
- Howarth, F.G. Ecology of cave arthropods. Annu. Rev. Entomol. 1983, 28, 365–389. [Google Scholar] [CrossRef]
- Howarth, F.G.; Moldovan, O.T. Where cave animals live. In Cave Ecology; Moldovan, O.T., Kováč, Ľ., Halse, S., Eds.; Springer: Cham, Switzerland, 2018; pp. 23–37. [Google Scholar]
- Culver, D.; Pipan, T. The Biology of Caves and Other Subterranean Habitats, 2nd ed.; Oxford University Press: Oxford, UK, 2019; p. 301. [Google Scholar]
- Pipan, T.; Deharveng, L.; Culver, D.C. Hotspots of Subterranean Biodiversity. Diversity 2020, 12, 209. [Google Scholar] [CrossRef]
- Stone, F.D. Bayliss Lava Tube and the Discovery of a Rich Cave Fauna in Tropical Australia. In Proceedings of the 14th International Symposium on Vulcanospeleology, Undara National Park, Mount Surprise, QLD, Australia, 12–17 August 2010; pp. 47–58. [Google Scholar]
- Clarke, A. An Overview of Invertebrate Fauna Collections from the Undara Lava Tube System. In Proceedings of the 14th International Symposium on Vulcanospeleology, Undara National Park, Mount Surprise, QLD, Australia, 12–17 August 2010; pp. 59–76. [Google Scholar]
- Fontaine, B.; Perrad, A.; Bouchet, P.V.N.P. 21 years of shelf life between discovery and description of new species. Curr. Biol. 2012, 22, 943–944. [Google Scholar] [CrossRef]
- Bannink, P. List of invertebrates collected from the McBride Volcanic Province lava tubes and associated rainforest depressions. Unpublished Report. 1–12.
- Godwin, M.D. Undara and Associated Lava Fields of McBride Plateau, a Speleological Field Guide; Chillagoe Caving Club, Inc.: Cairns, Australia, 1993. [Google Scholar]
- Godwin, M.D.; Pearson, L.M. The Murronga lava flow. In Proceedings of the Cave Leeuwin Conference, Margaret River, WA, Australia, 30 December 1990–5 January 1991; pp. 34–54. [Google Scholar]
- Pearson, L.M. Field Guide to the Lava Tubes on the McBride Volcanic Province in North Queensland; Chillagoe Caving Club, Inc.: Cairns, Australia, 2010. [Google Scholar]
- Howarth, F.G.; Moldovan, O.T. The ecological classification of cave animals and their adaptations. In Cave Ecology; Moldovan, O.T., Kovác, L., Halse, S., Eds.; Ecological Studies; Springer: Cham, Switzerland, 2018; pp. 41–67. [Google Scholar]
- Cloudsley-Thompson, J.L. On the responses to environmental stimuli, and the sensory physiology of Millipedes (Diplopoda). Proc. Zool. Soc. Lond. 1951, 121, 253–277. [Google Scholar] [CrossRef]
- Griffin, T.J. The Geology, Mineralogy and Geochemistry of the McBride Basaltic Province, Northern Queensland. Ph.D. Thesis, James Cook University, Douglas, QLD, Australia, 1977. [Google Scholar]
- Cohen, B.E.; Mark, D.F.; Fallon, S.J.; Stephenson, P.J. Holocene-Neogene volcanism in northeastern Australia: Chronology and eruption history. Quat. Geochronol. 2017, 39, 79–91. [Google Scholar] [CrossRef]
- Griffin, T.G.; McDougall, I. Geochronology of the Cainozoic McBride Volcanic Province Northern Queensland. J. Geol. Soc. Aust. 1975, 22, 387–396. [Google Scholar] [CrossRef]
- Atkinson, A. The Undara lava tube system and its caves. Helictite 1990, 28, 3–14. [Google Scholar]
- Malipatil, M.B.; Howarth, F.G. Two new species of Micropolytaxus Elkins from Northern Australia (Hemiptera: Reduviidae: Saicinae). J. Aust. Entomol. Soc. 1990, 29, 37–40. [Google Scholar] [CrossRef]
- Atkinson, A.; Atkinson, V. Undara Volcano and Its Lava Tubes: A Geological Wonder of Australia in Undara Volcanic National Park, North Queensland; Atkinson, Anne & Vernon: Brisbane, Australia, 1995; pp. 1–96. [Google Scholar]
- Rein, T.; Kempe, S.; Dufresne, A. The “Cueva del Viento” on the Canaries, Spain. In Proceedings of the 17th International Vulcanspeleology Symposium Ocean View, Hawaii, HI, USA, 6–12 February 2016; pp. 1–8. [Google Scholar]
- Peterson, D.W.; Holcomb, R.T.; Tilling, R.I.; Christiansen, R.L. Development of lava tubes in the light of observations at Mauna Ulu, Kilauea Volcano, Hawaii. Bull. Volcanol. 1994, 56, 343–360. [Google Scholar] [CrossRef]
- Whitehead, P.W. The Regional Context of the McBride Basalt Province and the Formation of the Undara Lava Flows, Tubes, Rises and Depressions. In Proceedings of the 14th International Symposium on Vulcanospeleology, Undara National Park, Mount Surprise, QLD, Australia, 12–17 August 2010; pp. 9–18. [Google Scholar]
- Atkinson, A.; Griffin, T.J.; Stevenson, P.J. A major lava tube system, North Queensland. Bull. Volcanol. 1975, 39, 1–28. [Google Scholar] [CrossRef]
- Stephenson, P.J.; Griffin, T.J.; Sutherland, F.L.I.E.P. Cainozoic volcanism in north-eastern Australia. In Geology and Geophysics of North-Eastern Australia; Henderson, R.A., Stephenson, P.J., Eds.; Geological Association of Australia, Queensland Division: Douglas, QLD, Australia, 1980; pp. 349–374. [Google Scholar]
- Bureau of Meteorology. Climate Statistics for Australian Locations. Available online: http://www.bom.gov.au/climate/averages/tables/cw_030036.shtml (accessed on 25 April 2021).
- Bland, R.G.; Weinstein, P.; Slaney, D.P. Mouthpart sensilla of cave species of Australian Paratemnopteryx cockroaches (Blattaria: Blattellidae). Int. J. Insect Morphol. Embryol. 1998, 16, 291–300. [Google Scholar] [CrossRef]
- Roth, L.M. A revision of the Australian Parcoblattini (Blattaria: Blattellidae: Blattellinae). Mem. Qld. Mus. Nat. 1990, 28, 531–596. [Google Scholar]
- Smith, G.B. New species of Metrinura Mendes (Zygentoma: Nicoletiidae) from Queensland, Australia. Aust. J. Entomol. 2006, 45, 163–167. [Google Scholar] [CrossRef]
- Smith, G.B. New Atelurinae (Zygentoma: Nicoletiidae) from Northern Australia. Gen. Appl. Entomol. 2016, 44, 21–58. [Google Scholar]
- Stone, F.D.; (University of Hawaii). Personal Communication, 1985.
- Davies, V.T. The huntsman spiders Heteropoda Latreille and Yiinthi gen. nov. (Araneae: Heteropodidae) in Australia. Mem. Qld. Mus. 1994, 35, 75–122. [Google Scholar]
- Dyce, A.L.; Wellings, G. Phlebotomine sandflies (Diptera: Psychodidae) from caves in Australia. Parasitologia 1991, 33, 193–198. [Google Scholar]
- Bradbury, J.H.; (University of Adelaide). Personal Communication, 2000.
- Harvey, M.S. New cave-dwelling schizomids (Schizomida: Hubbardiidae) from Australia. Rec. West. Aust. Mus. Suppl. 2001, 64, 171–185. [Google Scholar] [CrossRef][Green Version]
- Moulds, T.A.; Murphy, N.; Adams, M.; Reardon, T.B.; Harvey, M.S.; Jennings, J.; Austin, A.D. Phylogeography of cave pseudoscorpions in southern Australia. J. Biogeogr. 2007, 34, 951–962. [Google Scholar] [CrossRef]
- Raven, R.J.; Stumkat, K.; Gray, M.R. Revisions of Australian ground-hunting spiders: I. Amauropelma gen. nov. (Araneomorphae: Ctenidae). Rec. West. Aust. Mus. Suppl. 2001, 64, 187–227. [Google Scholar] [CrossRef]
- Gray, M.R. Cavernicolous spiders (Araneae) from Undara, Queensland and Cape Range, Western Australia. Helictite 1989, 27, 87–89. [Google Scholar]
- Main, B.Y. Spiders; Collins: Sydney, Australia, 1984. [Google Scholar]
- Gray, M.R. Survey of the spider fauna of Australian caves. Helictite 1973, 11, 46–75. [Google Scholar]
- Baehr, B.C.; Jocqué, R. The new endemic Australian genus Nosterella and a review of Nostera (Araneae: Zodariidae), including eight new species. Mem. Qld. Mus. Nat. 2017, 60, 53–76. [Google Scholar]
- Slaney, D.P. New species of cave dwelling cockroaches in the genus Neotemnopteryx Princis (Blattaria: Blattellidae: Blattellinae). Mem. Qld. Mus. Nat. 2000, 46, 331–336. [Google Scholar]
- Stone, F.D. The cockroaches of North Queensland caves and the evolution of tropical troglobites. In Proceedings of the Tropicon 1988, 17th Biennial Australian Speleological Conference, Tinaroo, QLD, Australia, 27–31 December 1988; pp. 88–93. [Google Scholar]
- Escalona, H.; Oberprieler, R. Australian National Insect Collection; CSIRO: Canberra, Australia, 2021. [Google Scholar]
- Hoch, H.; Howarth, F.G. Six new cavernicolous cixiid planthoppers in the genus Solonaima from Australia (Homoptera: Fulgoroidea). Syst. Entomol. 1989, 14, 377–402. [Google Scholar] [CrossRef]
- Howarth, F.G. Bioclimatic and geologic factors governing the evolution and distribution of Hawaiian cave insects. Entomol. Gen. 1983, 8, 17–26. [Google Scholar] [CrossRef]
- Eberhard, S.M. Nowranie Caves and the Camooweal karst area, Queensland: Hydrology, geomorphology and speleogenesis, with notes on aquatic biota. Helictite 2003, 38, 27–38. [Google Scholar]
- Eberhard, S.M. Ecology and Hydrology of a Threatened Groundwater-Dependent Ecosystem: The Jewel Cave Karst System in Western Australia. Ph.D. Thesis, School of Environmental Science, Murdoch University, Perth, Australia, 2004. [Google Scholar]
- Wagner, W.H.; (University of Michigan, Ann Arbor, MI, USA). Personal Communication, 1988.
- Howarth, F.G.; Medeiros, M.J.; Stone, F.D. Hawaiian lava tube cave associated Lepidoptera from the collections of Francis G Howarth and Fred D Stone. Bish. Mus. Occas. Pap. 2020, 129, 37–54. [Google Scholar]
- Smith, G.B.; McRae, J.M. New species of subterranean silverfish (Zygentoma: Nicoletiidae: Atelurinae) from Western Australia’s semi-arid Pilbara region. Rec. West. Aust. Mus. 2014, 29, 105–127. [Google Scholar] [CrossRef][Green Version]
- Smith, G.B.; McRae, J.M. Further short range endemic troglobitic silverfish (Zygentoma: Nicoletiidae; Subnicoletiinae and Coletiniinae) from north-western Australia. Rec. West. Aust. Mus. 2016, 31, 41–55. [Google Scholar] [CrossRef][Green Version]
- Slaney, D.P. New species of Australian cockroaches in the genus Paratemnopteryx Saussure (Blattaria, Blattellidae, Blattellinae), and a discussion of some behavioural observations with respect to the evolution and ecology of cave life. J. Nat. Hist. 2001, 35, 1001–1012. [Google Scholar] [CrossRef]
- Brad, T.; Lepure, S.; Sarbu, S.M. The Chemoautotrophically Based Movile Cave Groundwater Ecosystem, a Hotspot of Subterranean Biodiversity. Diversity 2021, 13, 128. [Google Scholar] [CrossRef]
- Deharveng, L.; Bedos, A. Gaz carbonique. In Thai-Maros 85, Rapport Speleologique et Scientifique to Thailand and Sulawesi; Association Pyreneenne de Speleologie: Toulouse, France, 1986; pp. 144–152. [Google Scholar]
- Stone, F.D.; Howarth, F.G.; Hoch, H.; Asche, M. Root communities in lava tubes. In Encyclopedia of Caves, 2nd ed.; White, W.B., Culver, D.C., Eds.; Academic Press: Burlington, MA, USA, 2012; pp. 658–664. [Google Scholar]
- Humphreys, W.F. Where angels fear to tread: Developments in cave ecology. In Cave Ecology; Moldovan, O.T., Kovác, L., Halse, S., Eds.; Ecological Studies; Springer: Cham, Switzerland, 2018; pp. 497–532. [Google Scholar]
- Humphreys, W.F. (Ed.) The Biogeography of Cape Range, Western Australia; Records of the Western Australian Museum Supplement; Western Australian Museum: Perth, Australia, 1993; Volume 45, pp. 1–248. [Google Scholar]
- Poore, G.C.B.; Humphreys, W.F. Bunderanthura bundera gen. et sp. nov. from Western Australia, first anchialine Leptanthuridae (Isopoda) from the Southern Hemisphere. Rec. West. Aust. Mus. 2013, 28, 21–29. [Google Scholar] [CrossRef][Green Version]
- Humphreys, G.; Alexander, J.; Harvey, M.; Humphreys, W.F. The subterranean fauna of Barrow Island, northwestern Australia. Rec. West. Aust. Mus. Suppl. 2013, 83, 145–158. [Google Scholar] [CrossRef][Green Version]
- Eberhard, S.M.; Halse, S.A.; Humphreys, W.F. Stygofauna in the Pilbara region, north-west Western Australia: A review. J. R. Soc. West. Aust. 2005, 88, 167–176. [Google Scholar]
- Eberhard, S.M.; Halse, S.A.; Williams, M.; Scanlon, M.D.; Cocking, J.S.; Barron, H.J. Exploring the relationship between sampling efficiency and short range endemism for groundwater fauna in the Pilbara region, Western Australia. Freshw. Biol. 2009, 54, 885–901. [Google Scholar] [CrossRef]
- Humphreys, W.F. Groundwater calcrete aquifers in the Australian arid zone: The context to an unfolding plethora of stygal diversity. Rec. West. Aust. Mus. Suppl. 2001, 64, 233–234. [Google Scholar] [CrossRef]
- Halse, S.A. Research in calcretes and other deep subterranean habitats outside caves. In Cave Ecology; Moldovan, O.T., Kovác, L., Halse, S.A., Eds.; Ecological Studies; Springer: Cham, Switzerland, 2018; pp. 415–434. [Google Scholar]
- Halse, S.A.; Pearson, G.B. Troglofauna in the vadose zone: Comparison of scraping an dtrapping results and sampling adequacy. Subterr. Biol. 2014, 13, 17–34. [Google Scholar] [CrossRef]
- Moulds, T.; Bannink, P. Preliminary notes on the cavernicolous arthropod fauna of Judburra/Gregory karst area, northern Australia. Helictite 2012, 41, 75–85. [Google Scholar]
- Bennelongia Pty Ltd. Yeelirrie Subterranean Fauna Assessment; Report Prepared for Cameco Australia Pty Ltd.; 2015/236b; Bennelongia Pty Ltd.: Perth, Australia, 2015. [Google Scholar]
- Eberhard, S.M.; Watts, C.H.S.; Callan, S.K.; Leijs, R. Three new subterranean diving beetles (Coleoptera: Dytiscidae) from the Yeelirrie groundwater calcretes, Western Australia, and their distribution between several calcrete deposits including a potential mine site. Rec. West. Aust. Mus. 2016, 31, 27–40. [Google Scholar] [CrossRef]
- Subterranean Ecology Pty Ltd. Yeelirrie Subterranean Fauna Survey; Report Prepared for BHP Billiton Yeelirrie Development Company Pty Ltd.; 2010/14; Subterranean Ecology Pty Ltd.: Perth, Australia, 2011; p. 269. [Google Scholar]
- Subterranean Ecology Pty Ltd. Ethel Gorge Aquifer Threatened Ecological Community—Consolidated Taxonomy; Unpublished Report Prepared for BHP Billiton Iron Ore; Subterranean Ecology Pty Ltd.: Perth, Australia, 2013; p. 96. [Google Scholar]
- Tang, D.; Eberhard, S.M. Two new species of Nitocrella (Crustacea, Copepoda, Harpacticoida) from groundwaters of northwestern Australia expand the geographic range of the genus in a global hotspot of subterranean biodiversity. Subterr. Biol. 2016, 20, 51–76. [Google Scholar] [CrossRef]
- Toro Energy Limited. Extension to the Wiluna Uranium Project: Environmental Management Plan: Subterranean Fauna Management Plan; Toro Energy Limited: Perth, Australia, 2012. [Google Scholar]
- Biota Environmental Services Pty Ltd. Rio Tinto Regional Troglobitic Fauna Study; Unpublished Report Prepared for Rio Tinto Iron Ore; Biota Environmental Services Pty Ltd.: Perth, Australia, 2013. [Google Scholar]
- Halse, S.A.; Scanlon, M.D.; Cocking, J.S.; Barron, H.J.; Richardson, J.B.; Eberhard, S.M. Pilbara stygofauna: Deep groundwater of an arid landscape contains globally significant radiation of biodiversity. Rec. West. Aust. Mus. Suppl. 2014, 78, 443–483. [Google Scholar] [CrossRef][Green Version]
- Bennelongia Pty Ltd. Stygofauna Survey—Exmouth Cape Aquifer: Scoping Document Describing Work Required to Determine Ecological Water Requirements for the Exmouth Cape Aquifer; Prepared for Department of Water; Bennelongia Pty Ltd.: Perth, Australia, 2008; p. 39. [Google Scholar]
- King, R.A.; Fagan-Jeffries, E.; Bradford, T.M.; Stringer, D.N.; Finston, T.; Halse, S.A.; Eberhard, S.M.; Humphreys, G.; Humphreys, W.F.; Austin, A.D.; et al. Cryptic Diversity down under: Defining species in the subterranean amphipod genus Nedsia Barnard and Williams (Hadzioidea: Eriopisidae) from the Pilbara, Western Australia. Invertebr. Syst. 2021. in review. [Google Scholar]
- Eberhard, S.; Smith, G.; Gibian, M.; Smith, H.; Gray, M. Invertebrate Cave Fauna of Jenolan. Proc. Linn. Soc. N. S. W. 2014, 136, 35–67. [Google Scholar]
- Eberhard, S.M.; Spate, A. Cave Invertebrate Survey: Toward an Atlas of NSW Cave Fauna; Report Prepared under the NSW Heritage Assistance Program NEP 94 765; NSW Heritage Assistance Program: Canberra, Australia, 1995; pp. 1–112. [Google Scholar]
- Thurgate, M.E.; Gough, J.S.; Clarke, A.; Serov, P.; Spate, A. Stygofauna diversity and distribution in Eastern Australian cave and karst areas. Rec. West. Aust. Mus. 2001, 64, 49–62. [Google Scholar] [CrossRef][Green Version]
- Thurgate, M.E.; Gough, J.S.; Spate, A.; Eberhard, S.M. Subterranean biodiversity in New South Wales: From rags to riches. Rec. West. Aust. Mus. Suppl. 2001, 64, 37–47. [Google Scholar] [CrossRef]
- Eberhard, S.M.; Richardson, A.M.M.; Swain, R. The Invertebrate Cave Fauna of Tasmania; Report to the National Estate Office, Canberra; Zoology Department, University of Tasmania: Hobart, Australia, 1991; p. 174. [Google Scholar]
- Ahyong, S.T. The Tasmanian Mountain Shrimps, Anaspides Thomson, 1894 (Crustacea, Syncarida, Anaspididae). Rec. Aust. Mus. 2016, 68, 313–364. [Google Scholar] [CrossRef]
- Eberhard, S.M.; Giachino, P.M. Tasmanian Trechinae and psydrinae (Coleoptera, Carabidae): A taxonomic and biogeographic synthesis, with description of new species and evaluation of the impact of Quaternary climate changes on evolution of the subterranean fauna. Subterr. Biol. 2011, 9, 1–72. [Google Scholar]
- Karanovic, I.; Eberhard, M.S.; Perina, G. Austromesocypris bluffensis sp. n. (Crustacea, Ostracoda, Cypridoidea, Scottiinae) from subterranean aquatic habitats in Tasmania, with a key to world species of the subfamily. ZooKeys 2012. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ponder, W.F.; Clark, S.A.; Eberhard, S.M.; Studdert, J.B. A radiation of hydrobiid snails in the caves and streams at Precipitous Bluff, southwest Tasmania, Australia (Mollusca: Caenogastropoda: Rissooidea: Hydrobiidae s.l.). Zootaxa 2005, 1074, 1–66. [Google Scholar] [CrossRef]
- Tomlinson, M. A Framework for Determining Environmental Water Requirements for Alluvial Aquifer Ecosystems. Ph.D. Thesis, University of New England, Armidale, Australia, 2008. [Google Scholar]
- Humphreys, W.F. Australasian subterranean biogeography. In Handbook of Australasian Biogeography; Ebach, M.C., Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 269–293. [Google Scholar]
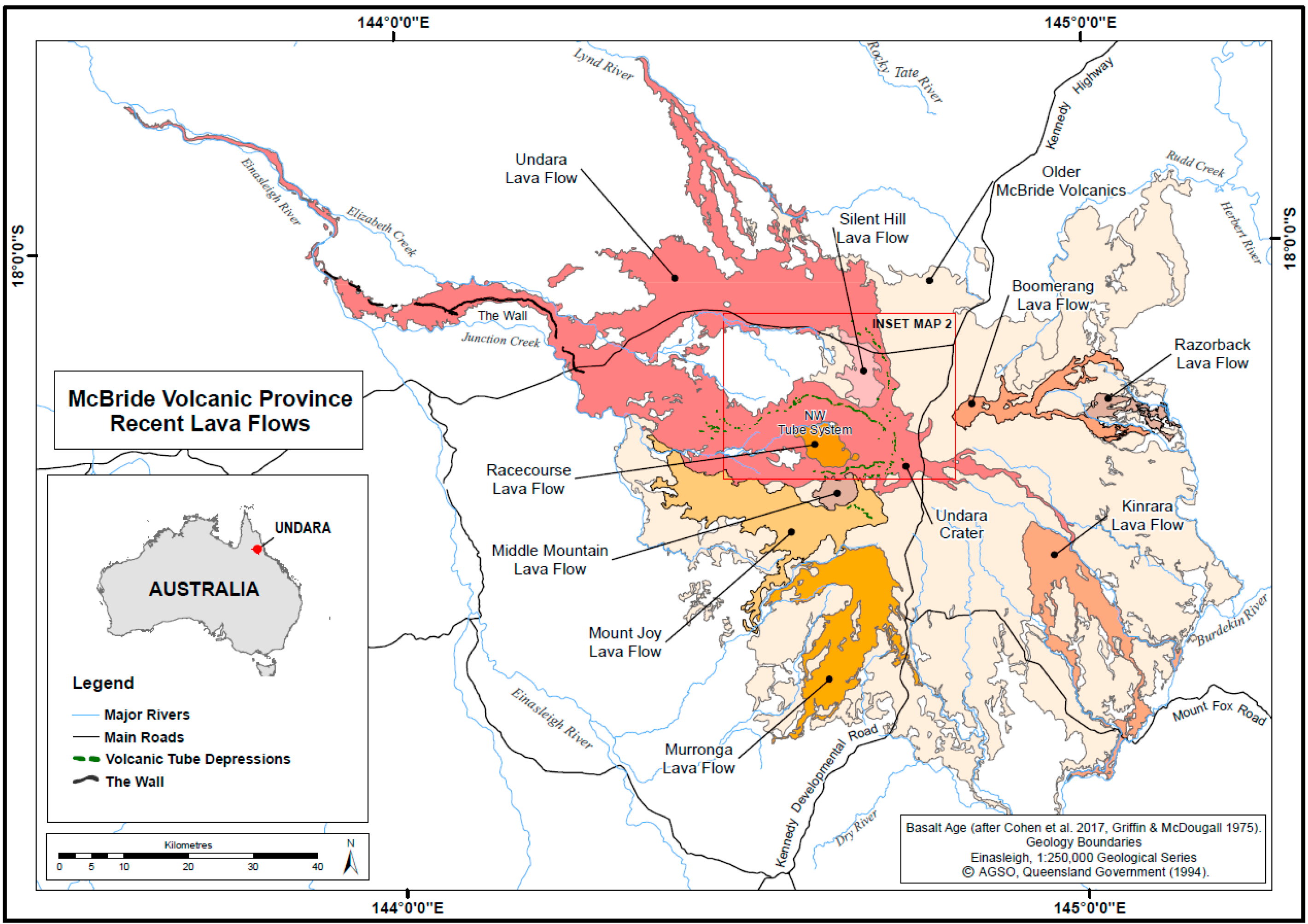
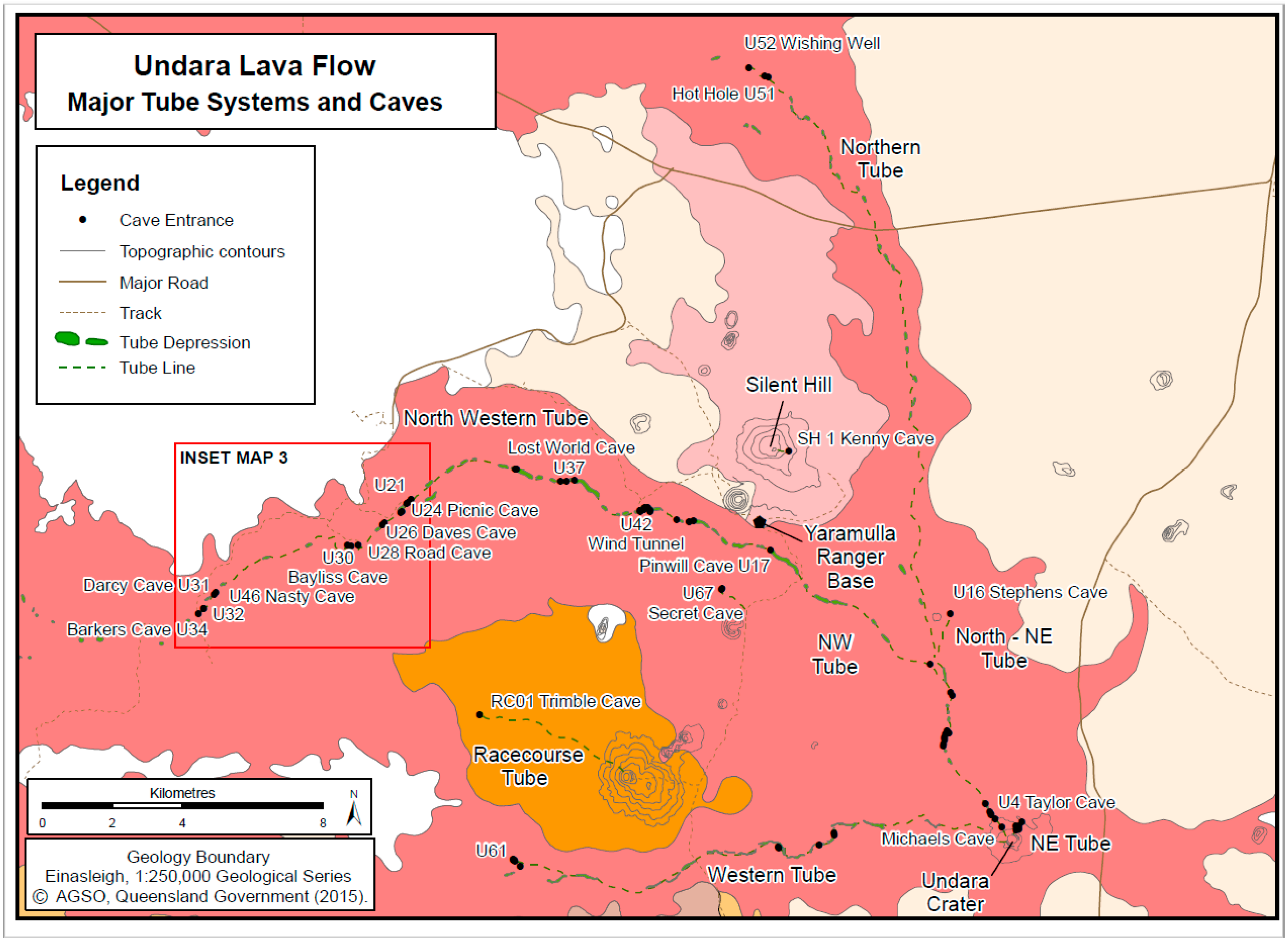

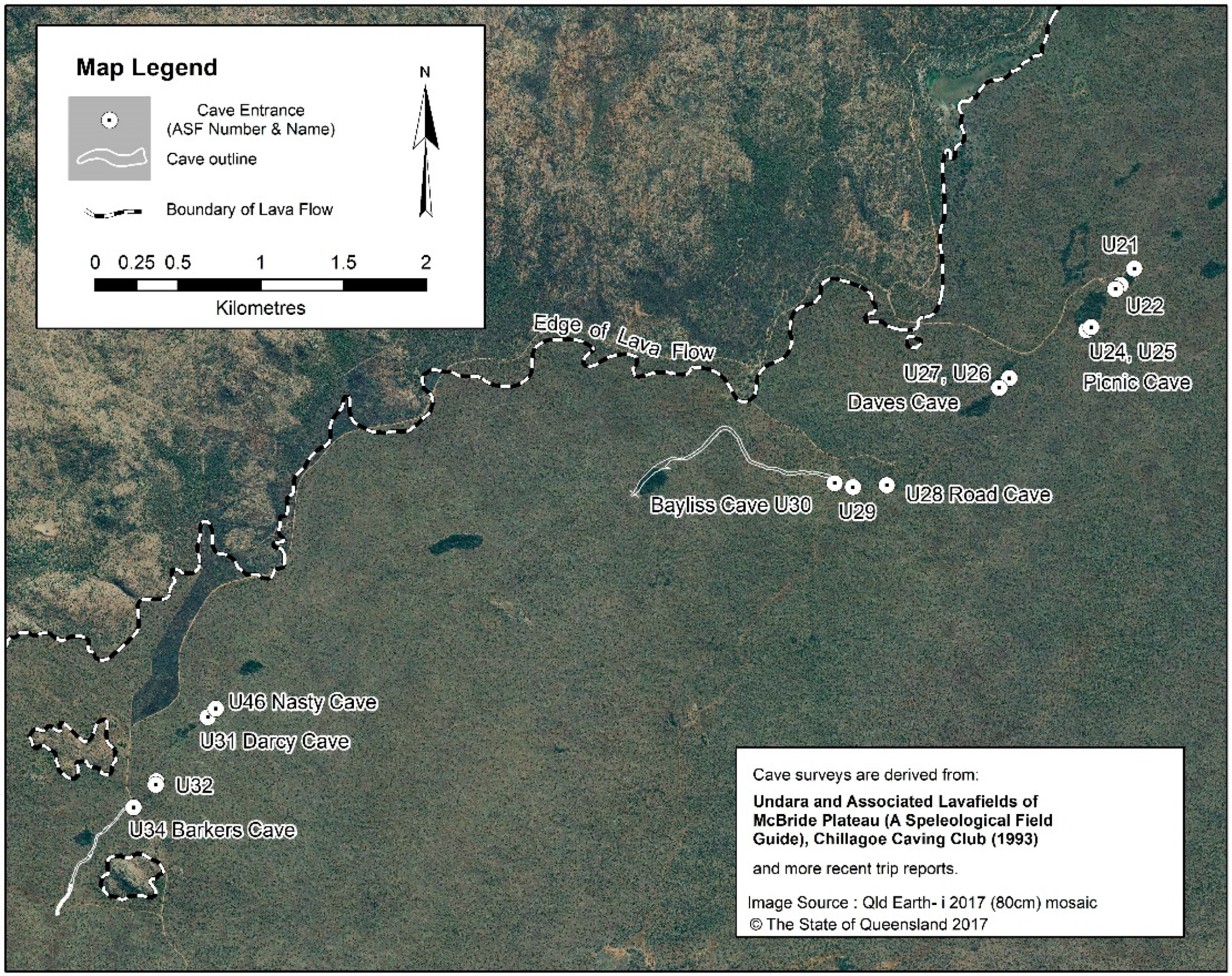




| Basalt Flows (40Ar/39Ar Age, ka/ma) | Lava Tube Systems (Length)/Number of Known Caves | Sampled Caves by Tube System from Crater | Remarks |
|---|---|---|---|
| Kinrara (40Ar/39Ar Age 7 ± 2 ka, 2σ) | 2 tube systems, 11 caves/arches | none | Approximately 25 km SE Undara crater, potentially important for biology. |
| Murronga (40Ar/39Ar Age 153 ± 5 ka, 2σ) | 2 tubes, 12 caves with combined survey length of 1.2 km | Collins No. 1, Collins No. 2, Two Ten, Long Shot | Approximately 15 km S of Undara Crater, potentially important for biology. Major caves with significant fauna. |
| Undara (40Ar/39Ar Age 189 ± 4 ka, 2σ) | 5 tube systems, ~67 caves/arches with combined survey length of 6.38 km | N-NE system: Hot Hole, Wishing Well. N system: Stephens NW system: 10 caves. W system: none E system: none Other: Secret No. 1 and 2 | Fauna collection records obtained from 17 caves, all except 3 sampled caves occur in NW system. 70% of known caves occur in the NW tube system. |
| Racecourse ~ 0.20 ma | 1 tube, 4 caves | None sampled | Caves reported in Pearson [35], not sampled. |
| Boomerang ~ 0.23 ma | None recorded | - | - |
| Razorback < 0.27 ma | None recorded | - | - |
| Silent Hill ~ 0.37 ma | 1 tube system, 1 cave | Kenny Cave SH-1 | Located within 3 km of Undara NW tube system, potentially important for biology. |
| Mount Joy < 0.40 ma | None recorded | - | - |
| Middle Mountain ~ 0.89 ma | None recorded | - | - |
| Older basalt < 3.00 ma | None recorded | - | - |
| Tube System | Cave Name (No. *) | Length | Elevation | Environment | Fauna |
|---|---|---|---|---|---|
| Northwest | Michael’s (no #) | 15 m | 975 m | All twilight | Twilight zone fauna |
| Northwest | Taylor (U-4) | 108 m | 950 m | Deep Zone | A few troglobionts, bats |
| Northwest | Pinwell (U-17) | 150 m | 850 m | Deep Zone | A few troglobionts, bats |
| Northwest | Wind tunnel (U-42) | 293 m | 800 m | Two entrances, limited deep zone | A few possible troglobionts, bats |
| Northwest | Lost World (U-37) | 74 m | 775 m | Two entrances, well-ventilated, transition zone | Blattodea: Macropanesthia rhinoceros, bats |
| Northwest | Picnic (U-24; U-25) | ~450 m | 740 m | Permanent pool, sump, humid, deep zone | Unidentified moths, isopods, beetles, crickets, spiders, bats |
| Northwest | Daves (upflow) (U-26)) | 50 m | 730 m | Limited dark zone | Lepidoptera: Euploia sp. aff. core; unid. spider, phalangid, moths, beetles, bugs, cockroaches, ants, wasps, bats |
| Northwest | Daves II (U-27) | 27 m | 730 m | Limited dark zone | Lepidoptera: Euploia sp. aff. core; unid. spider, phalangid, moths, beetles, bugs, cockroaches, ants, wasps, bats |
| Northwest | Road (U-28) | 220 m | 700 m | Mostly twilight, intermittent stream | Stygobiont Amphipoda: Chillagoe sp.; many surface arthropods in leaflitter |
| Northwest | Bayliss (U-30) | 1300 m | 700 m | Hot humid bad air | 23 troglobionts, one stygobiont, bats |
| Northwest | Nasty (U-46) | 127 m | 670 m | Hot, humid, bad air | 8 troglobionts, bats |
| Northwest | Darcy (U-31) | 99 m | 670 m | Limited deep, elevated CO2 | trogloxenes and accidentals, bats |
| Northwest | Barkers (U-34) | 560+ m | 625 m | Deep zone, Lake at end | 7 troglobionts, bats |
| Tributary of Northwest | Secret Cave No. 1 (U-67) | 150 m | ~825 m | Deep zone | Blind cockroaches |
| North-NE | Stephens (=Stevens) Cave (U-16) | 70 m | 880 m | Mostly transition zone | Troglophilic cockroach, bats |
| Northern | Hot Hole (U-51) | 172 m | ~760 m | Warm, bad air, deep guano | Unid. spider, troglophilic cockroach, bats |
| Northern | Wishing Well (U-52) | 104 m | ~750 m | Humid, bad air | Unid. spider, troglophilic cockroach, slater, bats |
| # | SB/TB | Species | Taxonomic Classification | Caves * | References |
|---|---|---|---|---|---|
| 1 | SB | Chillagoe n. sp. | Crustacea: Amphipoda: Chillagoeidae | U-28; U-30 | [57] |
| 2 | TB | Unidentified genus and species 1 | Crustacea: Isopoda: Porcellionidae | U-30; U-17 | [2] |
| 3 | TB | Unidentified genus and species 2 | Crustacea: Isopoda: Porcellionidae | U-30; U-17 | [2] |
| 4 | TB | Unidentified genus and species 3 (eyeless) | Crustacea: Isopoda: Oniscoidea | U-34 | [30] |
| 5 | TB | Unidentified genus and species 1, juvenile | Arachnida: Schizomida: Hubbardiidae | U-34 | [58] |
| 6 | TB | Protochelifer sp. nr. cavernarum Beier | Arachnida: Pseudoscorpionida: Cheliferidae | U-42; U-17 | [30,59] |
| 7 | TB | Amauropelma undara Raven et al. | Arachnida: Araneae: Ctenidae | U-30; U-46 | [60] |
| 8 | TB | Unidentified genus and species 1 | Arachnida: Araneae: Linyphiidae | U-30 | [2] |
| 9 | TB | Nesticella sp. 1 | Arachnida: Araneae: Nesticidae | U-30 | [61] |
| 10 | TB | Unidentified genus and species | Arachnida: Araneae: Oonopidae | U-30 | [1] |
| 11 | TB | Spermophora sp. 1 | Arachnida: Araneae: Pholcidae | U-30 | [62] (p. 239); [2] |
| 12 | TB | Spermophora sp. 2 | Arachnida: Araneae: Pholcidae | U-34 | [30,63] |
| 13 | TB | Dolomedes sp. 1 | Arachnida: Araneae: Pisauridae | U-34 | [30] |
| 14 | TB | Nosterella cavicola Baehr and Jocqué. | Arachnida: Araneae: Zodariidae | U-30; U-46 | [64] |
| 15 | TB | New genus and species | Chilopoda: Scutigeromorpha: Scutigeridae | U-30; U-34; U-46 | [2] |
| 16 | TB | Unidentified genus and species | Diplopoda: Spirostreptida | U-30; U-46; U-34 | [2] |
| 17 | TB | Unidentified genus and species 1 | Diplopoda: Polydesmida | U-30 | [2] |
| 18 | TB | Unidentified genus and species 2 | Diplopoda: Polydesmida | U-30 | [2] |
| 19 | TB | Unidentified genus and species 1 | Diplopoda: Polyxenida | U-30; U-46 | [2] |
| 20 | TB | Pseudosinella sp. 1 | Collembola: Entomobryidae | U-30; U-34 | [2] |
| 21 | TB | Unidentified genus and species | Collembola: | U-30; U-46 | [2] |
| 22 | TB | Neotemnopteryx baylissensis Slaney | Insecta: Blattodea: Ectobiidae | U-30; SH-1 | [65] |
| 23 | TB | Nocticola sp. 1 | Insecta: Blattodea: Nocticolidae | U-30; U-46; U-17 | [66] |
| 24 | TB | New genus and species 1 | Insecta: Coleoptera: Curculionidae: Entiminae | U-30 | [67] |
| 25 | TB | New genus and species 2 | Insecta: Coleoptera: Curculionidae: Entiminae | U-4 | [67] |
| 26 | TB | Unidentified genus and species | Insecta: Coleoptera: Pselaphinae | U-30 | [2] |
| 27 | TB | Unidentified genus and species | Insecta: Coleoptera: Staphylinidae | U-30; U-34 | [2] |
| 28 | TB | Unidentified genus and species | Insecta: Diplura | U-30 | [2] |
| 29 | TB | Solonaima baylissa Hoch and Howarth | Insecta: Auchenorrhyncha: Cixiidae | U-30; U-46 | [68] |
| 30 | TB | Micropolytoxus cavicolus Malipatil and Howarth | Insecta: Heteroptera: Reduviidae | U-30; U-34 | [42] |
| 31 | TB | Peirates sp. 1 | Insecta: Heteroptera: Reduviidae | U-30 | [2] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eberhard, S.M.; Howarth, F.G. Undara Lava Cave Fauna in Tropical Queensland with an Annotated List of Australian Subterranean Biodiversity Hotspots. Diversity 2021, 13, 326. https://doi.org/10.3390/d13070326
Eberhard SM, Howarth FG. Undara Lava Cave Fauna in Tropical Queensland with an Annotated List of Australian Subterranean Biodiversity Hotspots. Diversity. 2021; 13(7):326. https://doi.org/10.3390/d13070326
Chicago/Turabian StyleEberhard, Stefan M., and Francis G. Howarth. 2021. "Undara Lava Cave Fauna in Tropical Queensland with an Annotated List of Australian Subterranean Biodiversity Hotspots" Diversity 13, no. 7: 326. https://doi.org/10.3390/d13070326
APA StyleEberhard, S. M., & Howarth, F. G. (2021). Undara Lava Cave Fauna in Tropical Queensland with an Annotated List of Australian Subterranean Biodiversity Hotspots. Diversity, 13(7), 326. https://doi.org/10.3390/d13070326







