Biogeography of Iberian Ants (Hymenoptera: Formicidae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Species Included in the Study
2.2. Biogeographical Analysis
2.2.1. Chorology
2.2.2. Distribution Range
2.2.3. Origin and Dispersal Routes
3. Results
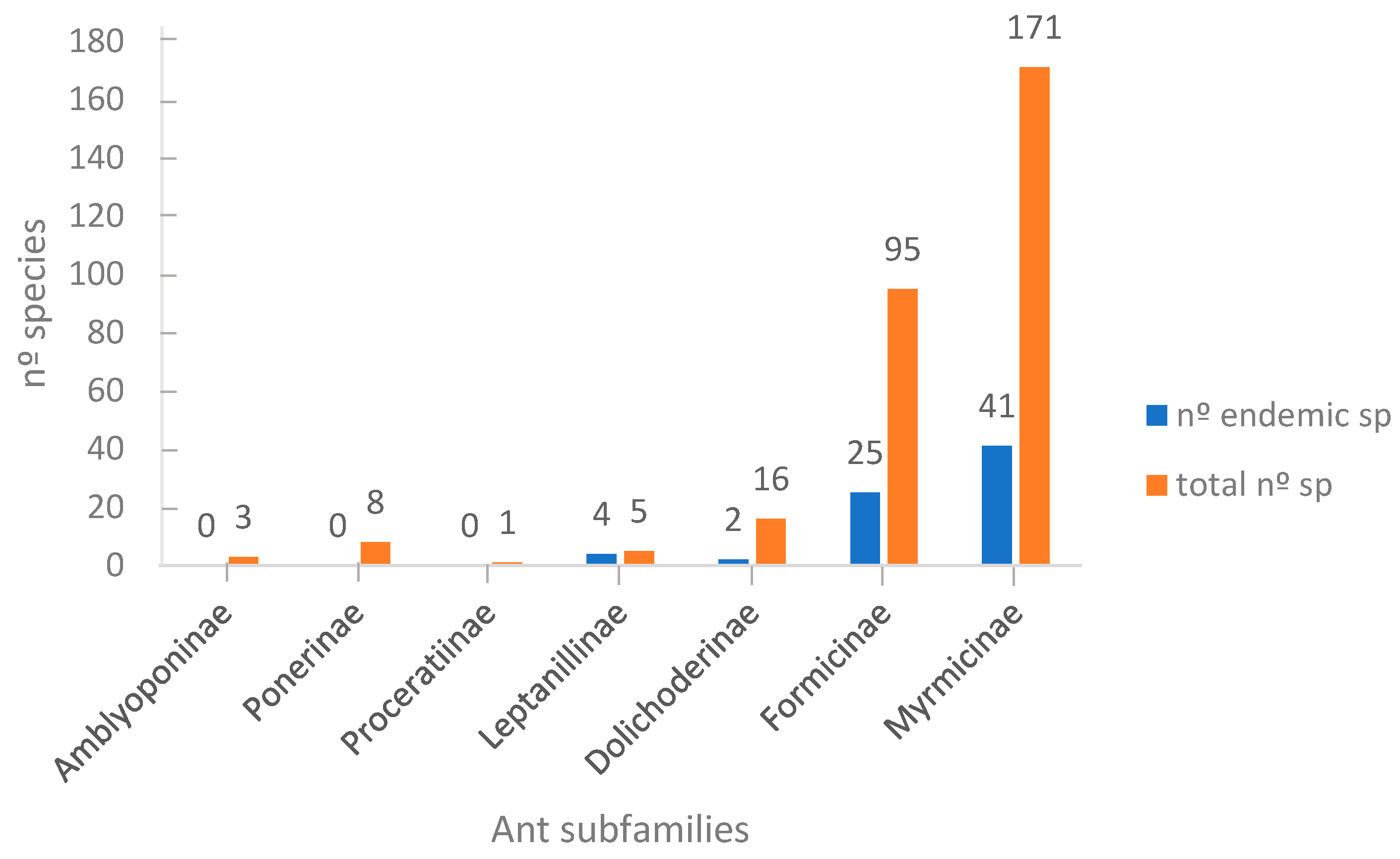
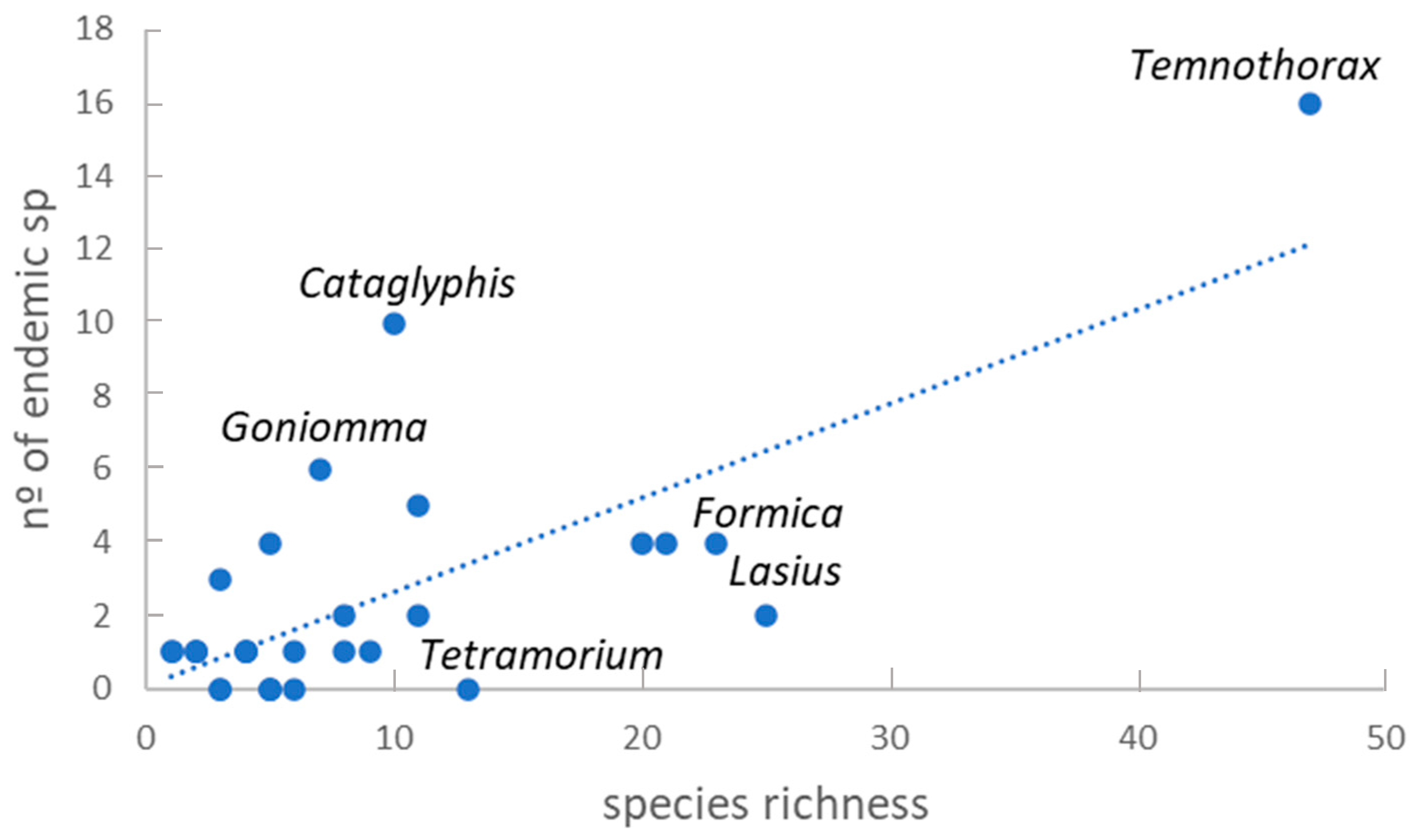
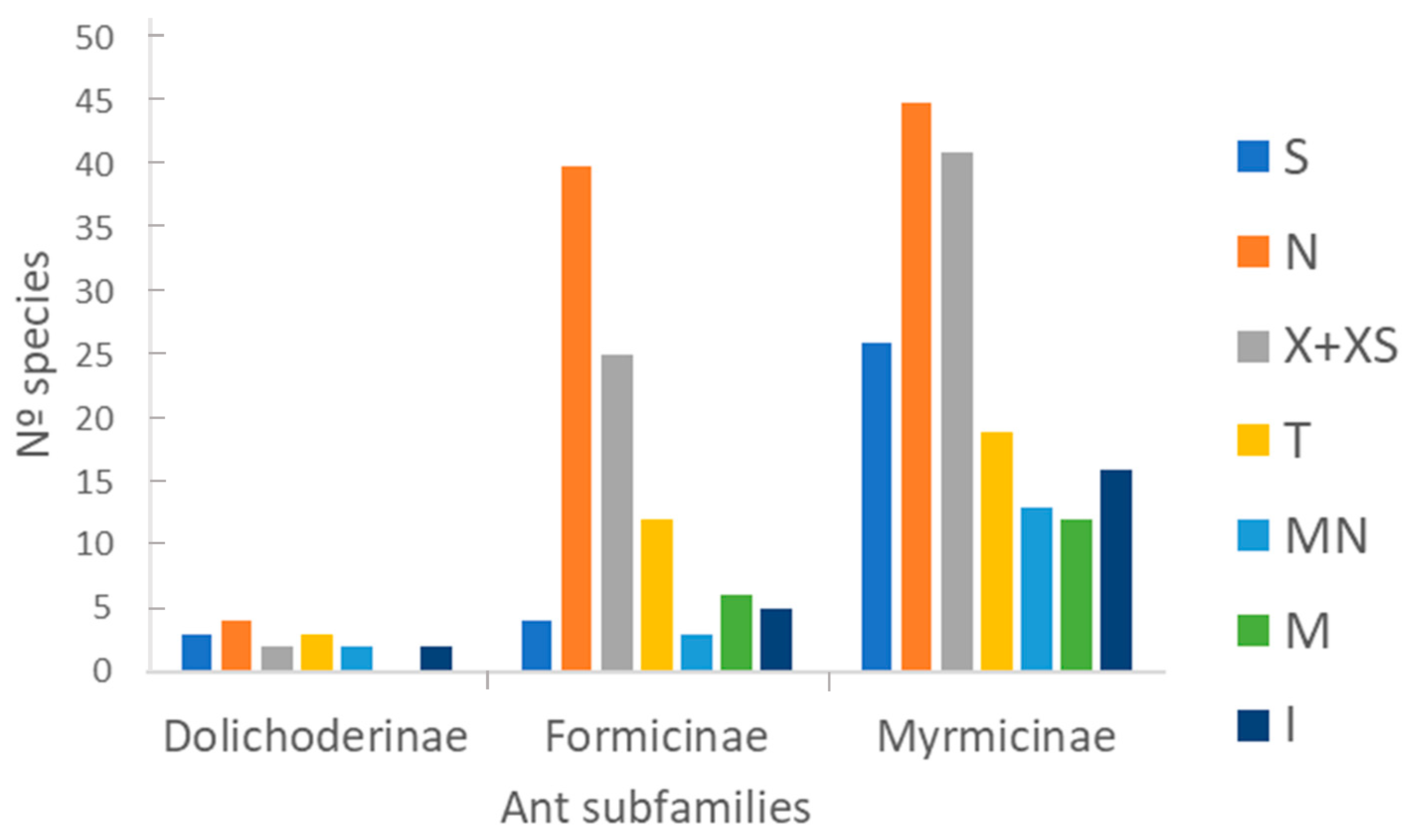
3.1. Taxonomic Richness
3.2. Biogeographical Analysis
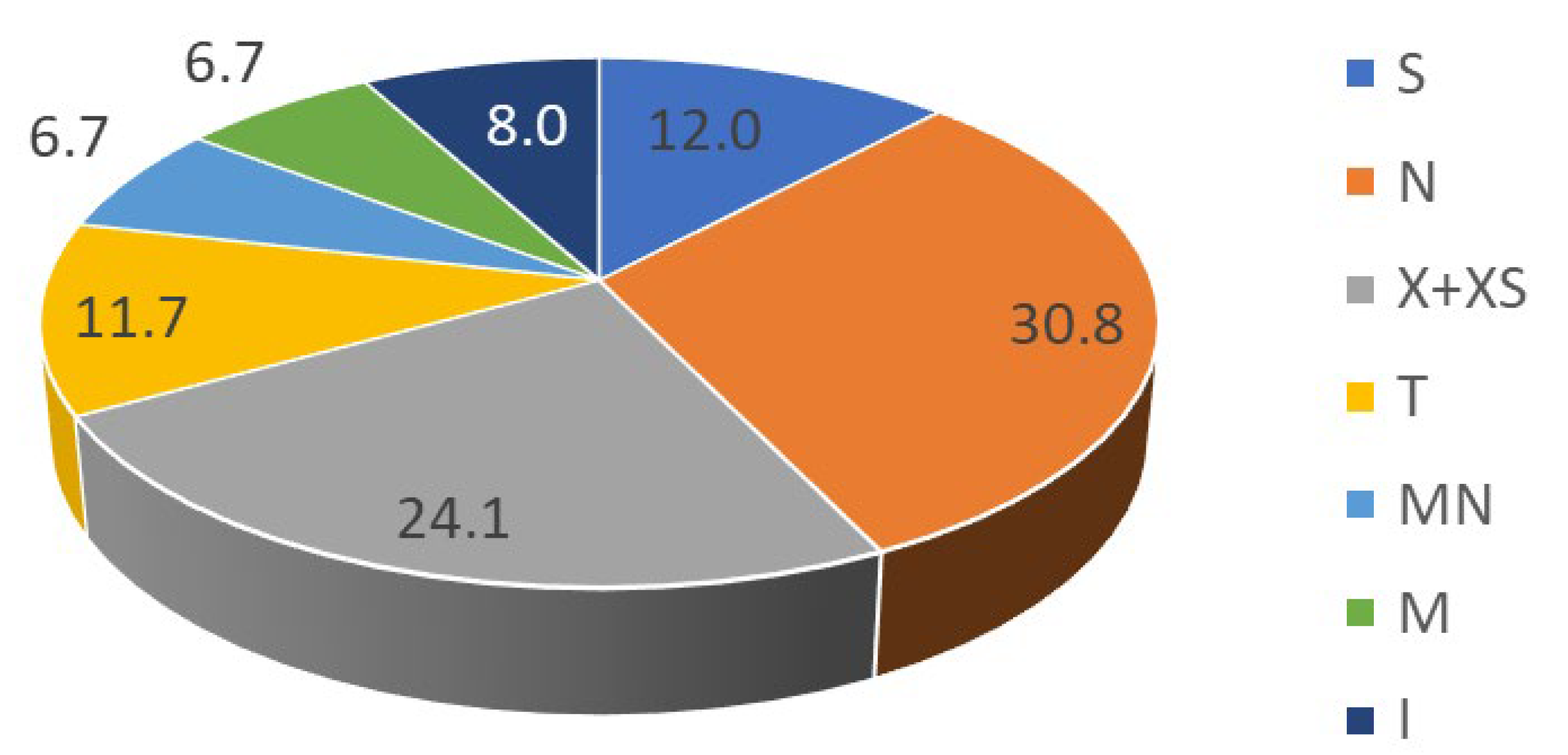
3.2.1. Chorology
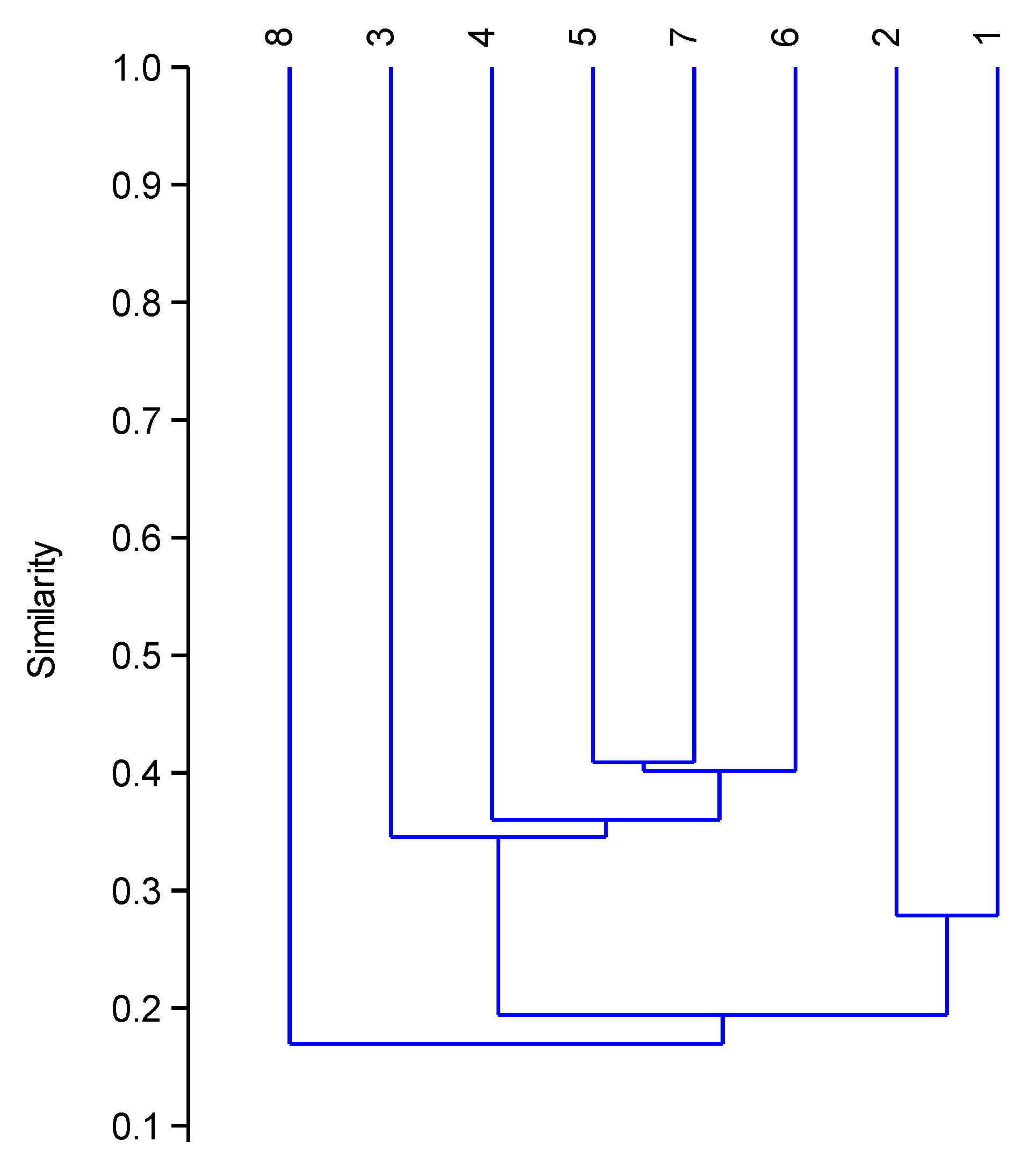
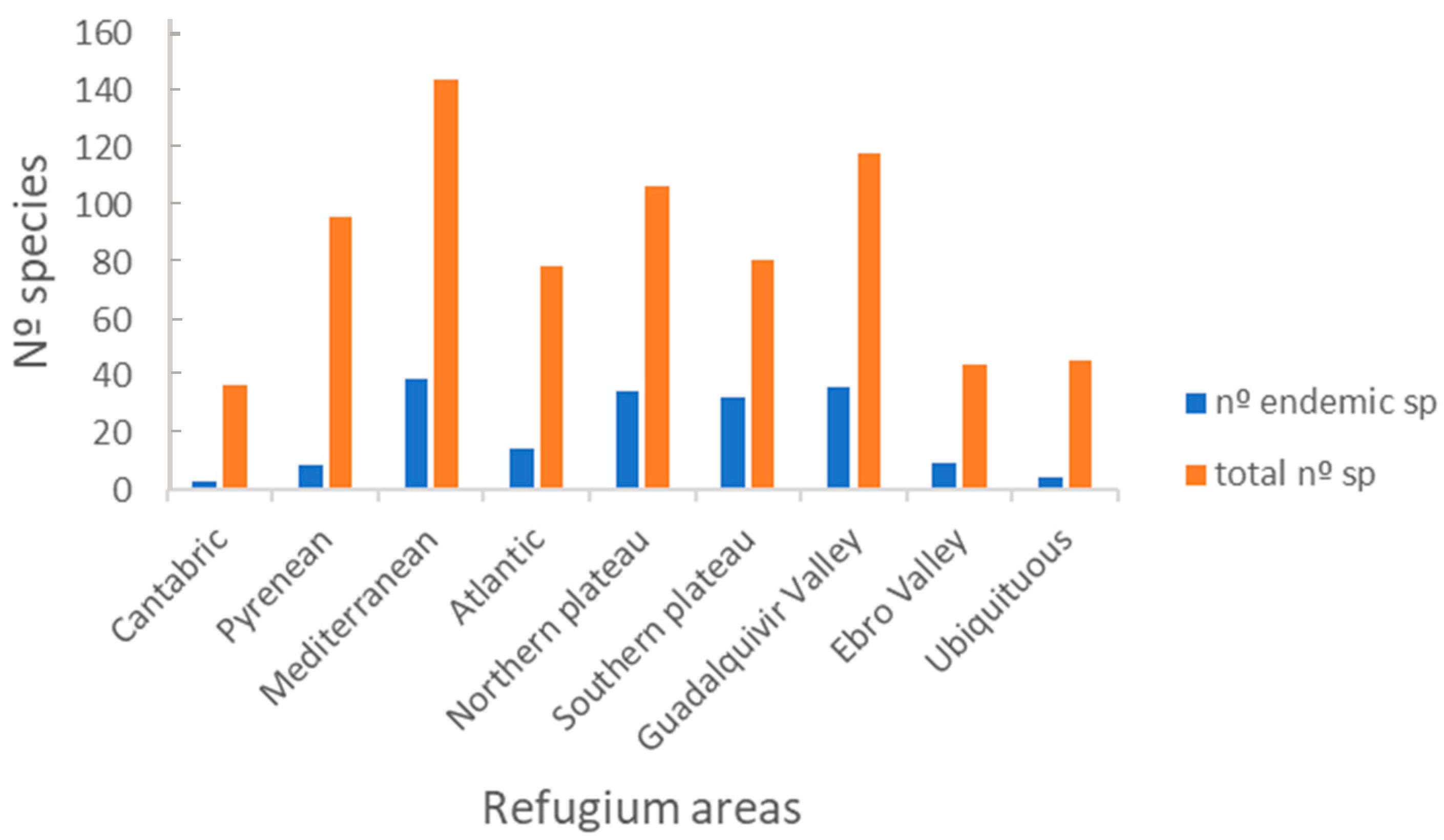
3.2.2. Distribution Range
4. Discussion
4.1. Taxonomic Richness
4.2. Biogeographical Analysis
4.2.1. Chorology
4.2.2. Distribution Ranges
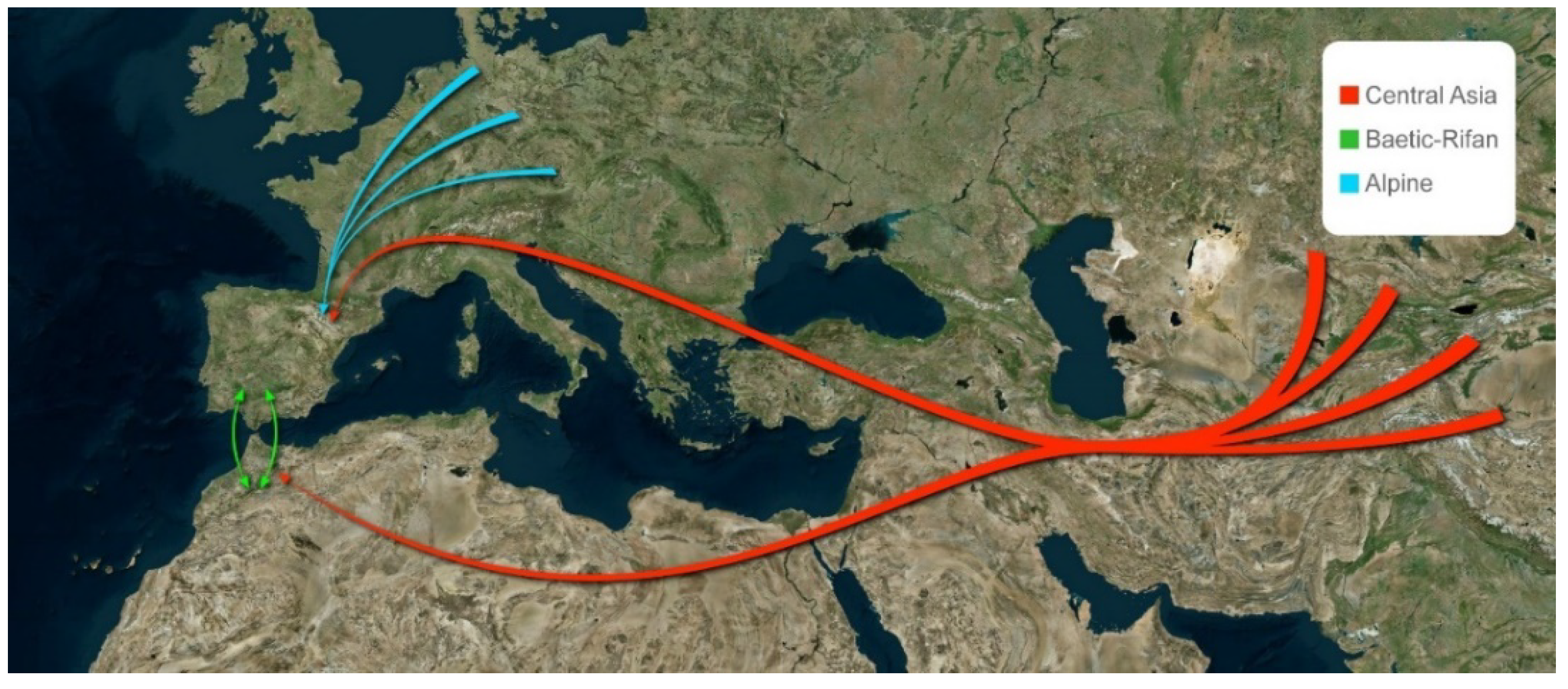
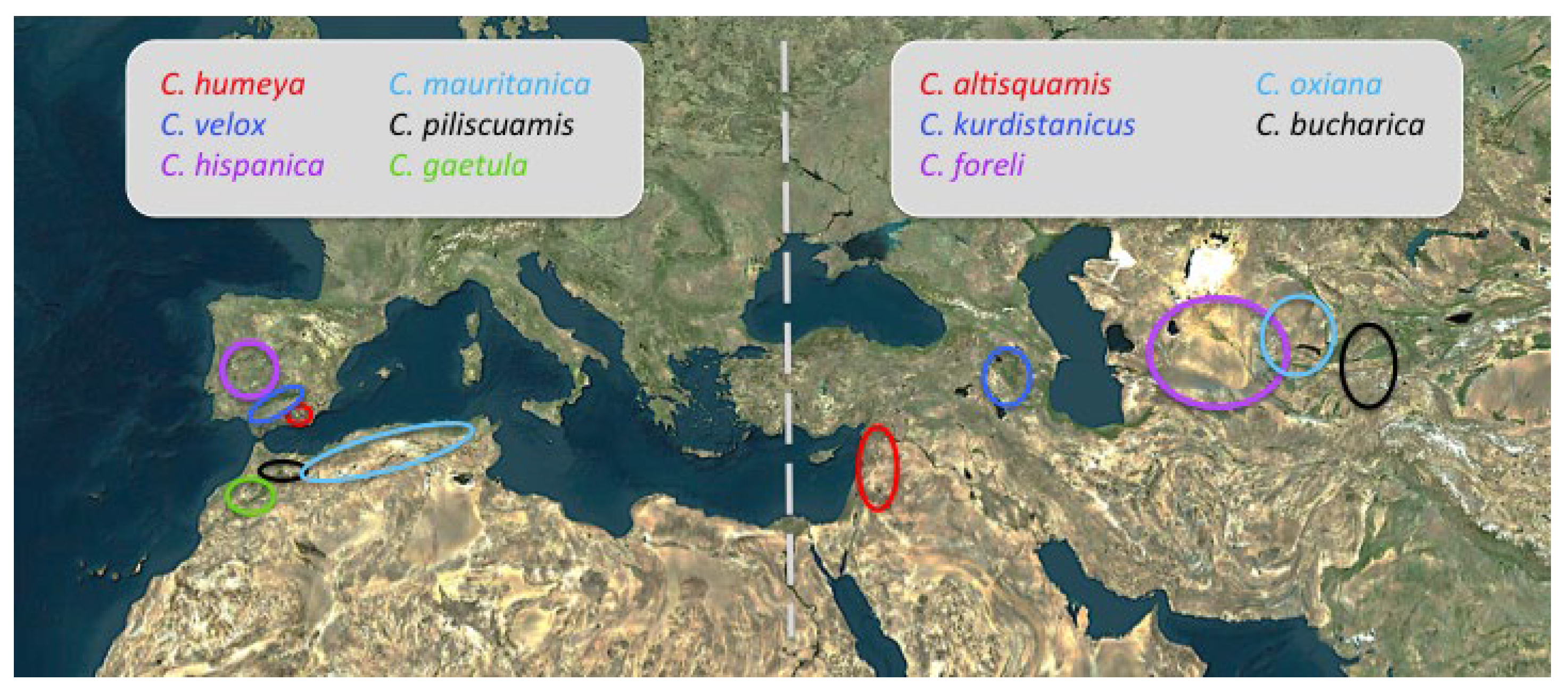
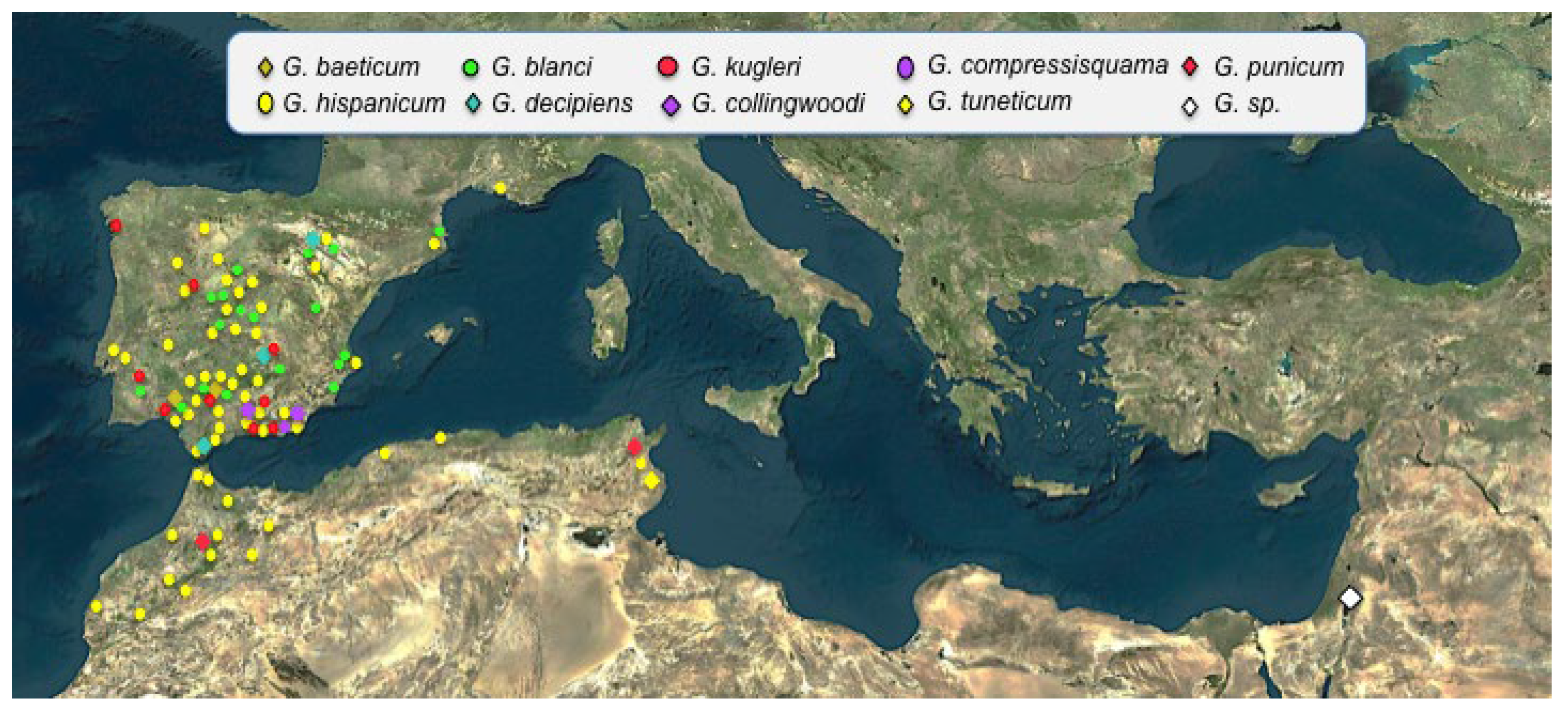
4.2.3. Origin and Dispersal Routes
Relict Species
Asian-IP Disjunct Species
Baetic-Rifan Species
Alpine Species
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jenkins, C.N.; Sanders, N.J.; Andersen, A.N.; Arnan, X.; Brühl, C.A.; Cerda, X.; Ellison, A.M.; Fisher, B.L.; Fitzpatrick, M.C.; Gotelli, N.J.; et al. Global diversity in light of climate change: The case of ants. Divers. Distrib. 2011, 17, 652–662. [Google Scholar] [CrossRef]
- Andersen, A.N.A.N. Ant megadiversity and its origins in arid Australia. Austral Entomol. 2016, 55, 132–137. [Google Scholar] [CrossRef]
- Economo, E.P.; Narula, N.; Friedman, N.R.; Weiser, M.D.; Guénard, B. Macroecology and macroevolution of the latitudinal diversity gradient in ants. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hakala, S.M.; Seppä, P.; Helanterä, H. Evolution of dispersal in ants (Hymenoptera: Formicidae): A review on the dispersal strategies of sessile superorganisms. Myrmecol. News 2019, 29, 35–55. [Google Scholar]
- Dunn, R.R.; Agosti, D.; Andersen, A.N.; Arnan, X.; Bruhl, C.A.; Cerdá, X.; Ellison, A.M.; Fisher, B.L.; Fitzpatrick, M.C.; Gibb, H.; et al. Climatic drivers of hemispheric asymmetry in global patterns of ant species richness. Ecol. Lett. 2009, 12, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.L. Biogeography. In Ant Ecology; Lach, L., Parr, C.L., Abbott, K.L., Eds.; Oxford University Press: Oxford, UK, 2010; pp. 18–37. [Google Scholar]
- Guénard, B.; Perrichot, V.; Economo, E.P. Integration of global fossil and modern biodiversity data reveals dynamism and stasis in ant macroecological patterns. J. Biogeogr. 2015, 42, 2302–2312. [Google Scholar] [CrossRef]
- Catarineu, C.; Barberá, G.G.; Reyes-López, J.L. Zoogeography of the ants (Hymenoptera: Formicidae) of the Segura River Basin. Sociobiology 2018, 65, 383–396. [Google Scholar] [CrossRef]
- Jowers, M.J.; Taheri, A.; Reyes-López, J. The ant Anochetus ghilianii (Hymenoptera, Formicidae), not a Tertiary relict, but an Iberian introduction from North Africa: Evidence from mtDNA analyses. Syst. Biodivers. 2015, 13, 865–874. [Google Scholar] [CrossRef]
- Villalta, I.; Amor, F.; Galarza, J.A.; Dupont, S.; Ortega, P.; Hefetz, A.; Dahbi, A.; Cerdá, X.; Boulay, R. Origin and distribution of desert ants across the Gibraltar Straits. Mol. Phylogenet. Evol. 2018, 118, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Sanllorente, O.; Lorite, P.; Ruano, F.; Palomeque, T.; Tinaut, A. Phylogenetic relationships between the slave-making ants Rossomyrmex and their Proformica hosts in relation to other genera of the ant tribe Formicini (Hymenoptera: Formicidae). J. Zool. Syst. Evol. Res. 2018, 56, 48–60. [Google Scholar] [CrossRef]
- Ruano, F.; Devers, S.; Sanllorente, O.; Errard, C.; Tinaut, A.; Lenoir, A. A geographical mosaic of coevolution in a slave-making host-parasite system. J. Evol. Biol. 2011, 24, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Sanllorente, O.; Ruano, F.; Tinaut, A. Large-scale population genetics of the mountain ant Proformica longiseta (Hymenoptera: Formicidae). Popul. Ecol. 2015, 57, 637–648. [Google Scholar] [CrossRef]
- Sánchez-Vialas, A.; García-París, M.; Ruiz, J.L.; Recuero, E. Patterns of morphological diversification in giant Berberomeloe blister beetles (Coleoptera: Meloidae) reveal an unexpected taxonomic diversity concordant with mtDNA phylogenetic structure. Zool. J. Linn. Soc. 2020, 189, 1249–1312. [Google Scholar] [CrossRef]
- Mas-Peinado, P.; Buckley, D.; Ruiz, J.L.; GarcÍa-ParÍs, M. Recurrent diversification patterns and taxonomic complexity in morphologically conservative ancient lineages of Pimelia (Coleoptera: Tenebrionidae). Syst. Entomol. 2018, 125, 331–348. [Google Scholar] [CrossRef]
- Condamine, F.L.; Sperling, F.A.H.; Wahlberg, N.; Rasplus, J.Y.; Kergoat, G.J. What causes latitudinal gradients in species diversity? Evolutionary processes and ecological constraints on swallowtail biodiversity. Ecol. Lett. 2012, 15, 267–277. [Google Scholar] [CrossRef]
- Herrera-Russert, J.; López-López, A.; Serrano, J.; Matalin, A.; Galián, J. Influence of the Mediterranean basin history on the origin and evolution of the halophile tiger beetle genus Cephalota (Coleoptera: Cicindelidae). Ann. Soc. Entomol. Fr. 2020, 56, 447–454. [Google Scholar] [CrossRef]
- Ribera, I.; Castro, A.; Díaz, J.A.; Garrido, J.; Izquierdo, A.; Jäch, M.A.; Valladares, L.F. The geography of speciation in narrow-range endemics of the “Haenydra” lineage (Coleoptera, Hydraenidae, Hydraena). J. Biogeogr. 2011, 38, 502–516. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeler, R.A.; Mittermeler, C.G.; Da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Araújo, M.B.; Lobo, J.M.; Moreno, J.C. The effectiveness of Iberian protected areas in conserving terrestrial biodiversity. Conserv. Biol. 2007, 21, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Medail, F.; Quezel, P. Biodiversity hotspots in the Mediterranean Basin: Setting global conservation priorities. Conserv. Biol. 1999, 13, 1510–1513. [Google Scholar] [CrossRef]
- Hewitt, G.M. Mediterranean Peninsulas: The Evolution of Hotspots. In Biodiversity Hotspots; Springer: Berlin/Heidelberg, Germany, 2011; pp. 123–147. [Google Scholar]
- Martín-Piera, F.; Sanmartín, I. Biogeografía de áreas y biogeografía de artrópodos holárticos y mediterráneos. Bol. Soc. Entomol. Aragon. 1999, 26, 535–560. [Google Scholar]
- Braga, J.C.; Martín, J.M. Los arrecifes del Mioceno de Almería. Investig. Gest. 1997, 1, 5–19. [Google Scholar]
- Arribas, A.; Garrido, G.; Viseras, C.; Soria, J.M.; Pla, S.; Solano, J.G.; Garcés, M.; Beamud, E.; Carrión, J.S. A mammalian lost world in Southwest Europe during the late pliocene. PLoS ONE 2009, 4, e7127. [Google Scholar] [CrossRef]
- Martín Martín, J.M.; Braga Alarcón, J.C.; Gómez Pugnaire, M.T. Itinerarios Geológicos por Sierra Nevada; de Andalucía, J., Ed.; Consejería de Medio Ambiente: Seville, Spain, 2008. [Google Scholar]
- Masseti, M.; Mazza, P.P.A. Western European Quaternary lions: New working hypotheses. Biol. J. Linn. Soc. 2013, 109, 66–77. [Google Scholar] [CrossRef]
- Arribas, A.; Riquelme, J.A.; Palmqvist, P.; Garrido, G.; Hernández, R.; Laplana, C.; Soria, J.M.; Viseras, C.; Durán, J.J.; Gumiel, P.; et al. Un nuevo yacimiento de grandes mamíferos villafranquienses en la Cuenca de Guadix-Baza (Granada): Fonelas P-1, primer registro de una fauna próxima al límite Plio-Pleistoceno en la Península Ibérica. Bol. Geol. Min. 2001, 112, 3–34. [Google Scholar]
- Arribas, A.; Baeza, E.; Bermúdez, D.; Blanco, S.; Durán, J.J.; Garrido, G.; Gumiel, J.C.; Hernández, R.; Soria, J.M.; Viseras, C. Nuevos registros paleontológicos de grandes mamíferos en la Cuenca de Guadix-Baza (Granada): Aportaciones del Proyecto Fonelas al conocimiento sobre las faunas continentales del Plioceno-Pleistoceno europeo. Bol. Geol. Min. 2004, 115, 567–582. [Google Scholar]
- Garrido, G. Generalidades Sobre Los Carnívoros Del Villafranquiense Superior En Relación Con El Registro Fósil De Fonelas P-1 Reflections on the Carnivores of the Upper Villafranchian Represented in the Fossil Record of the Fonelas P-1 Site. In Vertebrados del Plioceno Superior Terminal en el Suroeste de Europa: Fonelas P-1 y el Proyecto Fonelas; Arribas, A., Ed.; IGME: Madrid, Spain, 2008; pp. 85–146. ISBN 9788478407644. [Google Scholar]
- Centeno-Cuadros, A.; Delibes, M.; Godoy, J.A. Phylogeography of Southern Water Vole (Arvicola sapidus): Evidence for refugia within the Iberian glacial refugium? Mol. Ecol. 2009, 18, 3652–3667. [Google Scholar] [CrossRef] [PubMed]
- Gómez, A.; Lunt, D.H. Refugia within Refugia: Patterns of Phylogeographic Concordance in the Iberian Peninsula. In Phylogeography of Southern European Refugia: Evolutionary Perspectives on the Origins and Conservation of European Biodiversity; Springer: Dordrecht, The Netherlands, 2007; pp. 155–188. ISBN 9781402049040. [Google Scholar]
- Lobo, J.M.; Moreno, J.C. Spatial and environmental determinants of vascular plant species richness distribution in the Iberian Peninsula and Balearic Islands. Biol. J. Linn. Soc. 2001, 73, 233–253. [Google Scholar] [CrossRef]
- Miraldo, A.; Hewitt, G.M.; Paulo, O.S.; Emerson, B.C. Phylogeography and demographic history of Lacerta lepida in the Iberian Peninsula: Multiple refugia, range expansions and secondary contact zones. BMC Evol. Biol. 2011, 11, 170. [Google Scholar] [CrossRef]
- Hewitt, G.M. Genetic consequences of climatic oscillations in the Quaternary. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2004, 359, 183–195. [Google Scholar] [CrossRef]
- Hewitt, G.M. Post-glacial re-colonization of European biota. Biol. J. Linn. Soc. 1999, 68, 87–112. [Google Scholar] [CrossRef]
- Schmitt, T. Molecular biogeography of Europe: Pleistocene cycles and postglacial trends. Front. Zool. 2007, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Galkowski, C. Quelques fourmis nouvelles ou intéressantes pour la faune de France (Hymenoptera, Formicidae). Bull. Soc. Linn. Bordx. Tome 2008, 143, 423–433. [Google Scholar]
- Galkowski, C. Une liste des fourmis (Hymenoptera; Formicidae) récoltées dans la région de Grasse, avec la mention d’une nouvelle espèce de la faune de France. Bull. Soc. linn. Provence 2011, 62, 1–4. [Google Scholar]
- Steiner, F.M.; Csősz, S.; Markó, B.; Gamisch, A.; Rinnhofer, L.; Folterbauer, C.; Hammerle, S.; Stauffer, C.; Arthofer, W.; Schlick-Steiner, B.C. Turning one into five: Integrative taxonomy uncovers complex evolution of cryptic species in the harvester ant Messor “structor”. Mol. Phylogenet. Evol. 2018, 127, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Seifert, B.; Buschinger, A.; Aldawood, A.; Antonova, V.; Bharti, H.; Borowiec, L.; Dekoninck, W.; Dubovikoff, D.; Espadaler, X.; Flegr, J.; et al. Banning paraphylies and executing Linnaean taxonomy is discordant and reduces the evolutionary and semantic information content of biological nomenclature. Insectes Soc. 2016, 63, 237–242. [Google Scholar] [CrossRef]
- Salgueiro, J. Catálogo dos formicídeos de Portugal continental e ilhas. Bol. Soc. Entomol. Aragon. 2002, 31, 145–171. [Google Scholar]
- Collingwood, C.A.; Prince, A. A guide to ants of continental Portugal. Bol. Soc. Port. Entomol. 1998, Suplement. [Google Scholar]
- Boeiro, M.; Espadaler, X.; Azedo, A.R.; Collingwood, C.; Serrano, A.R.M. One genus and three ants species new to Portugal (Hymenoptera, Formicidae). Bol. Soc. Entomol. Aragon. 2009, 45, 515–517. [Google Scholar]
- Gonçalves, C.; Espadaler, X.; Pereira, J.A.; Santos, S.; Patanita, M.I. Primeiros registos das espécies Strongylognathus caeciliae Forel, 1897 e Temnothorax tyndalei (Forel, 1909)(Hymenoptera, Formicidae) em Portugal Continental. Bol. Soc. Entomol. Aragon. 2014, 54, 402. [Google Scholar]
- Henin, J.M.; Collingwood, C.; Paiva, M.R. Synonymy between Leptothorax caparica Henin, Paiva & Collingwood, 2001 and Cardiocondyla mauritanica. Bol. Soc. Port. Entomol. 2003, 211, 377–378. [Google Scholar]
- Sánchez-García, D.; Cuesta-Segura, D.A.; Trigos-Peral, G.; Arcos, J.; Catarineu, C.; García-García, F.; Herraiz, J.A.; Espadaler, X.; Gómez, K.; Tinaut, A. Checklist of the Iberian Myrmecofauna, In preparation.
- Tinaut, A.; Reyes-lópez, J. Descripción de una nueva especie para la península ibérica: Temnothorax alfacarensis n. sp. (Hymenoptera, Formicidae). Boln. Asoc. Esp. Ent 2020, 44, 359–378. [Google Scholar]
- Casiraghi, A.; Espadaler, X.; Hidalgo, N.P.; Gómez, K. Two additions to the Iberian myrmecofauna: Crematogaster inermis Mayr, 1862, a newly established, tree-nesting species, and Trichomyrmex mayri (Forel, 1902), an emerging exotic species temporarily nesting in Spain (Hymenoptera, Formicidae). J. Hymenopt. Res. 2020, 78, 57–68. [Google Scholar] [CrossRef]
- Bernadou, A.; Fourcassié, V.; Espadaler, X. A preliminary checklist of the ants Hymenoptera, Formicidae) of Andorra. Zookeys 2013, 277, 13–23. [Google Scholar] [CrossRef]
- Guillem, R.; Bensusan, K. Technomyrmex vexatus (Santschi, 1919) from Gibraltar (Hymenoptera: Formicidae): A new ant species for Europe and genus for Iberia. Myrmecol. News 2008, 11, 21–23. [Google Scholar]
- Guillem, R.; Bensusan, K. Tetramorium parvioculum sp. n. (Formicidae: Myrmicinae) a new species of the T. simillimum group from Gibraltar. Bol. Soc. Entomol. Aragon. 2009, 45, 157–161. [Google Scholar]
- Guillem, R.; Bensusan, K. Two new species of ants (Hymenoptera: Formicidae) for Europe from southern Iberia. Rev. Soc. Gaditana Hist. Nat. 2019, 13, 5–10. [Google Scholar]
- Casevitz-Weulersse, J.; Galkowski, C. Liste actualisée des Fourmis de France (Hymenoptera: Formicidae). Bull. Soc. Entomol. Fr. 2009, 114, 475–510. [Google Scholar]
- Monnin, T.; Espadaler, X.; Lenoir, A.; Peeters, C. Guide des Fourmis de France; Belin: Paris, France, 2013. [Google Scholar]
- Seifert, B. A taxonomic revision of the Palaearctic members of the subgenus Lasius s.str. Soil Org. 2020, 92, 15–86. [Google Scholar]
- Cagniant, H. Liste actualisee des fourmis du Maroc (Hymenoptera: Formicidae ). Myrmecol. Nachr. 2006, 8, 193–200. [Google Scholar]
- Galkowski, C.; Cagniant, H. Contribution à la connaissance des fourmis du groupe angustulus dans le genre Temnothorax (Hymenoptera, Formicidae). Rev. Assoc. Roussill. Entomol. 2017, 26, 180–191. [Google Scholar]
- Taheri, A.; Reyes-López, J. Exotic Ants (Hymenoptera: Formicidae) in Morocco: Checklist, Comments and New Faunistic Data. Trans. Am. Entomol. Soc. 2018, 144, 99–107. [Google Scholar] [CrossRef]
- Taheri, A.; Reyes-López, J.L. Five new records of ants (Hymenoptera: Formicidae) from Morocco. J. Insect Sci. 2015, 15, 37. [Google Scholar] [CrossRef]
- Delabie, J.H.C. Présence de Pheidole teneriffana Forel, 1893, au Maroc (Hym., Formicidae, Myrmicinae). Bull. Soc. Entomol. Fr. 2007, 112, 288. [Google Scholar]
- Cagniant, H. Le Genre Cataglyphis Foerster, 1850 au Maroc (Hyménoptères Formicidae). Orsis 2009, 24, 41–71. [Google Scholar]
- Taheri, A.; Reyes-López, J.; Espadaler, X. Citas nuevas o interesantes de hormigas (Hymenoptera, Formicidae) para Marruecos. Bol. Soc. Entomol. Aragon. 2010, 47, 299–300. [Google Scholar]
- Taheri, A.; Reyes-López, J. Primera cita de Pyramica membranifera (Emery, 1869) (Hymenoptera, Formicidae) y listado actualizado de hormigas alóctonas para Marruecos (Norte de África). Bol. Soc. Entomol. Aragon. 2011, 49, 363. [Google Scholar]
- Guillem, R.; Bensusan, K.; Taheri, A. First record of the ant subfamily Cerapachyinae Forel, 1893 (Hymenoptera: Formicidae) from Morocco. Bull. l’Inst. Sci. 2012, 34, 121–123. [Google Scholar]
- Guillem, R.; Bensusan, K.; Taheri, A. New data on genus Dorylus Fabricius, 1793 (Formicidae, Dorylinae) in Morocco. Bull. l’Inst. Sci. 2015, 37, 47–51. [Google Scholar]
- Borowiec, L. Catalogue of ants of Europe, the Mediterranean Basin and adjacent regions (Hymenoptera: Formicidae); Polish Taxonomical Society: Wroclaw, Poland, 2014; Volume 25, ISBN 9788361764496. [Google Scholar]
- Antwiki. Available online: http://antwiki.org/ (accessed on 20 November 2020).
- Agosti, D.; Collingwood, C.A. A provisional list of the Balkan ants (Hym., Formicidae) and a key to the worker caste. I. Synonymic List. Bull. Soc. Entomol. Fr. 1987, 60, 51–62. [Google Scholar]
- Wagner, H.C.; Seifert, B.; Borovsky, R.; Paill, W. Insight into the ant diversity of the Vjosa valley, Albania (Hymenoptera: Formicidae). Acta ZooBot Austria 2018, 155, 315–321. [Google Scholar]
- Petrov, I.Z. A list of currently known ant species (Formicidae, Hymenoptera) of Serbia. Arch. Biol. Sci. 2004, 56, 121–125. [Google Scholar] [CrossRef]
- Bracko, G. Review of the ant fauna (Hymenoptera: Formicidae) of Croatia. Acta Entomol. Slov. 2006, 14, 131–156. [Google Scholar]
- Bracko, G.; Wagner, H.C.; Schulz, A.; Gioahin, E.; Maticic, J.; Tratnik, A. New investigation and a cheklist of the ants (Hymenoptera: Formicidae) of the republic of Macedonia. North West. J. Zool. 2014, 10, 10–24. [Google Scholar]
- Borowiec, L.; Salata, S. Ants of Greece-additions and corrections (Hymenoptera: Formicidae). Genus 2012, 24, 335–401. [Google Scholar]
- Kiran, K.; Karaman, C. First annotated cheklist of the ant fauna of Turkey (Hymenoptera. Formicidae). Zootaxa 2012, 3548, 1–38. [Google Scholar] [CrossRef]
- Vonshak, M.; Ionescu-Hirsch, A. A checklist of the ants of Israel (Hymenoptera: Formicidae). Isr. J. Entomol. 2009, 39, 33–55. [Google Scholar]
- Mohamed, S.; Zalat, S.; Fadi, H.; Gadalla, S.; Sharaf, M. Taxonomy of ant species (Hymenoptera: Formicidae) collected by pitfall traps from Sinai and Delta region, Egypt. Egypt. J. Nat. Hist. 2001, 3, 40–61. [Google Scholar] [CrossRef]
- Ribera, I. Biogeography and conservation of Iberian water beetles. Biol. Conserv. 2000, 92, 131–150. [Google Scholar] [CrossRef]
- Arnan, X.; Cerdá, X.; Retana, J. Relationships among taxonomic, functional, and phylogenetic ant diversity across the biogeographic regions of Europe. Ecography 2017, 40, 448–457. [Google Scholar] [CrossRef]
- Romo, H.; García-Barros, E. Biogeographic regions of the Iberian Peninsula: Butterflies as biogeographical indicators. J. Zool. 2010, 282, 180–190. [Google Scholar] [CrossRef]
- Carrascal, L.M.; Lobo, J.M. Respuestas a viejas preguntas con nuevos datos: Estudio de los patrones de distribución de la avifauna española y consecuencias para su conservación. In Atlas de las Aves Reproductoras de España; Martí, R., Del Moral, J.C., Eds.; Dirección General de la Conservación de la Naturaleza y Sociedad española de Ornitología: Madrid, Spain, 2003; pp. 651–668. [Google Scholar]
- antmaps.org. Available online: https://antmaps.org/? (accessed on 2 December 2020).
- Sokal, R.R.; Rohlf, F.J. Biometry, 3rd ed.; Freeman and Company: New York, NY, USA, 1995; ISBN 0-7167-2411-1. [Google Scholar]
- Moreno Saiz, J.C.; Donato, M.; Katinas, L.; Crisci, J.V.; Posadas, P. New insights into the biogeography of south-western Europe: Spatial patterns from vascular plants using cluster analysis and parsimony. J. Biogeogr. 2013, 40, 90–104. [Google Scholar] [CrossRef]
- Hammer, D.A.T.; Ryan, P.D.; Hammer, Ø.; Harper, D.A.T. Past: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 178. [Google Scholar]
- Real, R. Tables of significant values of Jaccard’s index of similarity. Miscel-lania Zool. 1999, 22, 29–40. [Google Scholar]
- Baroni-Urbani, C.; Buser, M.W. Similarity of binary data. Syst. Zool. 1976, 25, 251–259. [Google Scholar] [CrossRef]
- Brosens, D.; Vankerhoven, F.; Ignace, D.; Wegnez, P.; Noé, N.; Heughebaert, A.; Borteis, J.; Dekoninck, W. FORMIDABEL: The Belgian Ants Database. Zookeys 2013, 306, 59–70. [Google Scholar]
- Ødegaard, F. New and little known ants (Hymenoptera, Formicidae) in Norway. Nor. J. Entomol. 2013, 60, 172–175. [Google Scholar]
- Moreau, C.S.; Bell, C.D. Testing the museum versus cradle tropical biological diversity hypothesis: Phylogeny, diversification, and ancestral biogeographical range evolution of the ants. Evolution 2013, 67, 2240–2257. [Google Scholar] [CrossRef]
- Mittelbach, G.G.; Schemske, D.W.; Cornell, H.V.; Allen, A.P.; Brown, J.M.; Bush, M.B.; Harrison, S.P.; Hurlbert, A.H.; Knowlton, N.; Lessios, H.A.; et al. Evolution and the latitudinal diversity gradient: Speciation, extinction and biogeography. Ecol. Lett. 2007, 10, 315–331. [Google Scholar] [CrossRef]
- Willig, M.R.; Kaufman, D.M.; Stevens, R.D. Latitudinal Gradients of Biodiversity: Pattern, Process, Scale, and Synthesis. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 273–309. [Google Scholar] [CrossRef]
- Gómez, K.; Lorite, P.; García, F.; Tinaut, A.; Espadaler, X.; Palomeque, T.; Sanllorente, O.; Trager, J. Differentiating Iberoformica and Formica (Serviformica) with description of the sexual castes of Formica (Serviformica) gerardi Bondroit, 1917 stat. rev. Sociobiology 2018, 65, 463–470. [Google Scholar] [CrossRef]
- O’Regan, H.J. The Iberian Peninsula—Corridor or cul-de-sac? Mammalian faunal change and possible routes of dispersal in the last 2 million years. Quat. Sci. Rev. 2008, 27, 2136–2144. [Google Scholar] [CrossRef]
- Lomolino, M.V.; Riddle, B.R.; Whittaker, R.J. Biogeography: Biological Diversity across Space and Time; Sinauer Associates: Sun-derland, MA, USA, 2017; ISBN 9781605354729. [Google Scholar]
- Jenkins, D.G.; Rinne, D. Red herring or low illumination? The peninsula effect revisited. J. Biogeogr. 2008, 35, 2128–2137. [Google Scholar] [CrossRef]
- González-Taboada, F.; Nores, C.; Álvarez, M.Á. Breeding bird species richness in Spain: Assessing diversity hypothesis at various scales. Ecography 2007, 30, 241–250. [Google Scholar] [CrossRef]
- Martin, J.; Gurrea, P. The Peninsular Effect in Iberian Butterflies (Lepidoptera: Papilionoidea and Hesperioidea). J. Biogeogr. 1990, 85–96. [Google Scholar] [CrossRef]
- Abellán, P.; Svenning, J.-C. Refugia within refugia-patterns in endemism and genetic divergence are linked to Late Quaternary climate stability in the Iberian Peninsula. Biol. J. Linn. Soc. 2014, 113, 13–28. [Google Scholar] [CrossRef]
- Pie, M.R.; Feitosa, R.M. Relictual ant lineages (Hymenoptera: Formicidae) and their evolutionary implications. Myrmecol. News 2016, 22, 55–58. [Google Scholar]
- Puente, A.I.; Altonaga, K.; Prieto, C.E.; Rallo, A. Delimitation of biogeographical areas in the Iberian Peninsula on the basis of Helicoidea species (Pulmonata: Stylommatophora). Glob. Ecol. Biogeogr. Lett. 1998, 7, 97–113. [Google Scholar] [CrossRef]
- Molina-Venegas, R.; Aparicio, A.; Lavergne, S.; Arroyo, J. Climatic and topographical correlates of plant palaeo- and neoendemism in a Mediterranean biodiversity hotspot. Ann. Bot. 2017, 119, 229–238. [Google Scholar] [CrossRef]
- Rahbek, C.; Borregaard, M.K.; Antonelli, A.; Colwell, R.K.; Holt, B.G.; Nogues-Bravo, D.; Rasmussen, C.M.Ø.; Richardson, K.; Rosing, M.T.; Whittaker, R.J.; et al. Building mountain biodiversity: Geological and evolutionary processes. Science 2019, 365, 1114–1119. [Google Scholar] [CrossRef]
- Fernández Escudero, I.; Tinaut, A.; Ruano, F. Ovarian Maturation under Cold Winter Conditions in a High-Mountain Ant (Hymenoptera: Formicidae). Environ. Entomol. 1997, 26, 1373–1377. [Google Scholar] [CrossRef]
- Catarineu, C.; Barberá, G.G.; Reyes-López, J.L. A new ant species, Temnothorax ansei sp.n. (Hymenoptera: Formicidae) from the arid environments of South-Eastern Spain. Sociobiology 2017, 64, 138–145. [Google Scholar] [CrossRef][Green Version]
- Tinaut, A. Teleutomyrmex kutteri, spec. nov. A new species from Sierra Nevada (Granada, Spain). Spixiana 1990, 13, 201–208. [Google Scholar]
- Reyes Lopez, J.; Benavente Martínez, A. Nueva cita de Teleutomyrmex kutteri Tinaut, 1990 (Hym. Formicidae) para la Península Ibérica. Bol. Soc. Entomol. Aragon. 2011, 49, 206. [Google Scholar]
- Lebas, C.; Galkowski, C.; Wegnez, P.; Espadaler, X.; Blatrix, R. The exceptional diversity of ants on mount Coronat (Pyrénées-Orientales), and Temnothorax gredosi (Hymenoptera, Formicidae) new to France. Rev. Assoc. Roussill. Entomol. 2015, 24, 24–33. [Google Scholar]
- García García, F.; Espadaler Gelabert, X.; Cuesta-Segura, A.D.; Sánchez-García, D. Primera cita ibérica para Temnothorax conatensis Galkowski & Lebas, 2016, y actualización de la distribución para Temnothorax grouvellei (bondroit, 1918) (Hymenoptera: Formicidae). Iberomyrmex 2018, 10, 22–27. [Google Scholar]
- Azcárate, F.M.; Rota, C.; Hevia, V.; Silvestre, M.; Tinaut, A.; Ruano, F.; Seoane, J.; Martín Azcárate, F.; Rota, C.; Hevia, V.; et al. Primera cita de Rossomyrmex minuchae Tinaut, 1981 (Hymenoptera, Formicidae) en el Sistema Central (España). Bol. Asoc. Esp. Entomol. 2016, 40, 535–537. [Google Scholar]
- Reyes-López, J.L.; Obregón Romero, R.; López Tirado, J. Nuevo registro de Myrmoxenus bernardi (Espadaler, 1982) (Hymenoptera: Formicidae) para la península ibérica. Bol. Assoc. Esp. Entomol. 2012, 36, 427–432. [Google Scholar]
- Tinaut, A.; Ruano, F.; Martínez, M.D. Biology, distribution and taxonomic status of the parasitic ants of the Iberian Peninsula (Hymenoptera: Formicidae, Myrmicinae). Sociobiology 2005, 46, 1–42. [Google Scholar]
- Ortuño, V.M.; Gilgado, J.D.; Tinaut, A. Subterranean ants: The case of Aphaenogaster cardenai (Hymenoptera: Formicidae). J. Insect Sci. 2014, 14, 212. [Google Scholar] [CrossRef]
- Tinaut, A.; Barranco, P.; Fernández, D. Nuevas localidades para Aphaenogaster cardenai Espadaler, 1981 (Hymenoptera, Formicidae). Bol. Asoc. Esp. Entomol. 2015, 39, 421–424. [Google Scholar]
- Blaimer, B.B.; Brady, S.G.; Schultz, T.R.; Lloyd, M.W.; Fisher, B.L.; Ward, P.S. Phylogenomic methods outperform traditional multi-locus approaches in resolving deep evolutionary history: A case study of formicine ants. BMC Evol. Biol. 2015, 15, 1–14. [Google Scholar] [CrossRef]
- Radchenko, A.G.; Khomych, M.R. Ants of the extinct genus Cataglyphoides Dlussky, 2008 (Hymenoptera: Formicidae: Formicinae) from the late Eocene European ambers. Invertebr. Zool. 2020, 17, 154–161. [Google Scholar] [CrossRef]
- La Polla, J.S.; Dlussky, G.M.; Perrichot, V. Ants and the fossil record. Annu. Rev. Entomol. 2013, 58, 609–630. [Google Scholar] [CrossRef]
- Prebus, M. Insights into the evolution, biogeography and natural history of the acorn ants, genus Temnothorax Mayr (hymenoptera: Formicidae). BMC Evol. Biol. 2017, 17, 250. [Google Scholar] [CrossRef]
- Jansen, G.; Savolainen, R.; Vepsäläinen, K. Phylogeny, divergence-time estimation, biogeography and social parasite-host relationships of the Holarctic ant genus Myrmica (Hymenoptera: Formicidae). Mol. Phylogenet. Evol. 2010, 56, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Branstetter, M.G. Origin and diversification of the cryptic ant genus Stenamma Westwood (Hymenoptera: Formicidae), inferred from multilocus molecular data, biogeography and natural history. Syst. Entomol. 2012, 37, 478–496. [Google Scholar] [CrossRef]
- Todisco, V.; Gratton, P.; Cesaroni, D.; Sbordoni, V. Phylogeography of Parnassius apollo: Hints on taxonomy and conservation of a vulnerable glacial butterfly invader. Biol. J. Linn. Soc. 2010, 101, 169–183. [Google Scholar] [CrossRef]
- Alonso, J. El yacimiento de ámbar cretácico de Peñacerrada (Álava). Una extraordinaria puerta de acceso al Aptiense (112-114 m.a.). Rev. PH 1999, 29, 142–147. [Google Scholar] [CrossRef]
- Peñalver, E.; Martínez Delclòs, X. Importancia Patrimonial del Arroyo de la Pascueta, un yacimiento de ámbar Cretácico con insectos fósiles en Rubielos de Mora. In El patrimonio paleontológico de Teruel; Meléndez Hevia, G., Mollá Peñalver, E., Eds.; Instituto de Estudios Turolenses: Teruel, Spain, 2002; pp. 201–208. [Google Scholar]
- Peñalver, E.; Martínez-Delclòs, X.; Arillo, A. Yacimientos con insectos fósiles en España. Rev. Esp. Paleontol. 1999, 14, 231–245. [Google Scholar]
- Peñalver, E.; Martínez Delclòs, X. Insectos del Mioceno inferior de Ribesalbes (Castellón, España). Hymenoptera. Treb. Mus. Geol. Barc. 2000, 9, 97–153. [Google Scholar]
- Braga, J.C.; Martín, J.M.; Quesada, C. Patterns and average rates of late Neogene- Recent uplift of the Betic Cordillera, SE Spain. Geomorphology 2003, 50, 3–26. [Google Scholar] [CrossRef]
- Agustí, J.; Antón, M. Memoria de la tierra: Vertebrados fósiles de la Península Ibérica; Ediciones del Serbal: Barcelona, Spain, 1997; ISBN 978-84-7628-195-6. [Google Scholar]
- Goropashnaya, A.V.; Fedorov, V.B.; Seifert, B.; Pamilo, P. Phylogenetic relationships of Palaearctic Formica species (hymenoptera, Formicidae) based on mitochondrial cytochrome b sequences. PLoS ONE 2012, 7, e41697. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goropashnaya, A.V.; Fedorov, V.B.; Pamilo, P. Recent speciation in the Formica rufa group ants (Hymenoptera, Formicidae): Inference from mitochondrial DNA phylogeny. Mol. Phylogenet. Evol. 2004, 32, 198–206. [Google Scholar] [CrossRef]
- Gaudeul, M.; Véla, E.; Rouhan, G. Eastward colonization of the Mediterranean Basin by two geographically structured clades: The case of Odontites Ludw. (Orobanchaceae). Mol. Phylogenet. Evol. 2016, 96, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Inda, L.A.; Sanmart, I.; Buerki, S.; Catal, P. Mediterranean origin and Miocene–Holocene Old World diversification of meadow fescues and ryegrasses (Festuca subgenus Schedonorus and Lolium). J. Biogeogr. 2014, 41, 600–614. [Google Scholar] [CrossRef]
- Wheeler, W.M. The Ants of the Baltic Amber; Teubner, B.G., Ed.; BG Teubner: Berlin, Germany, 1915. [Google Scholar]
- Dlussky, G.M. Genera of ants (Hymenoptera: Formicidae) from Baltic amber (in russian). Paleontol. J. 1997, 6, 50–62. [Google Scholar]
- Rust, J.; Andersen, N.M. Giant ants from the Paleogene of Denmark with a discussion of the fossil history and early evolution of ants (Hymenoptera: Formicidae). Zool. J. Linn. Soc. 1999, 125, 331–348. [Google Scholar] [CrossRef]
- de Andrade, M.L. Fossil Odontomachiti Ants from the Dominican republic (Amber Collection Stuttgart: Hymenoptera, Formicidae. VII: Odontomachiti); Staatliches Museum für Naturkunde: Berlin, Germany, 1994. [Google Scholar]
- Tinaut, A. El género Amblyopone Erichson en la península ibérica (Hymenoptera, Formicidae). Miscel.lania Zool. 1988, 12, 189–193. [Google Scholar]
- Tinaut, A.; Bensusan, K. Second record of Amblyopone impressifrons (Emery, 1869) (Formicidae, Amblyoponinae) for Iberia, with some coomments on the genus. Bol. Assoc. Esp. Entomol. 2011, 35, 509–514. [Google Scholar]
- Rodríguez-Sánchez, F.; Pérez-Barrales, R.; Ojeda, F.; Vargas, P.; Arroyo, J. The Strait of Gibraltar as a melting pot for plant biodiversity. Quat. Sci. Rev. 2008, 27, 2100–2117. [Google Scholar] [CrossRef]
- Ros-Montoya, S. El Padul (Granada): Presencia de Mamut Lanudo (Mammuthus primigenius Blumenbach) en el Sur de España. Misc. Paleontológica 2005, 6, 275–292. [Google Scholar]
- Ward, P.S.; Brady, S.G.; Fisher, B.L.; Schultz, T.R. Phylogeny and biogeography of dolichoderine ants: Effects of data partitioning and relict taxa on historical inference. Syst. Biol. 2010, 59, 342–362. [Google Scholar] [CrossRef]
- Tinaut, A. Situación taxonómica del género Cataglyphis Förster, 1850 en la Península Ibérica III. El grupo de C. velox Santschi, 1929 y descripción de Cataglyphis humeya, sp. n. (Hymenoptera, Formicidae). Eos 1991, 66, 215–227. [Google Scholar]
- Espadaler, X. Les formigues granivores de la mediterránia Occidental. Treb. Inst. Catalana Hist. Nat. 1981, 9, 39–44. [Google Scholar]
- Salata, S.; Borowiec, L. A taxonomic revision of the genus Oxyopomyrmex André, 1881 (Hymenoptera: Formicidae). Zootaxa 2015, 4025, 1–66. [Google Scholar] [CrossRef] [PubMed]
- Kryukov, A.; Iwasa, M.A.; Kakizawa, R.; Suzuki, H.; Pinsker, W.; Haring, E. Synchronic east-west divergence in azure-winged magpies (Cyanopica cyanus) and magpies (Pica pica). J. Zool. Syst. Evol. Res. 2004, 42, 342–351. [Google Scholar] [CrossRef]
- Tinaut, A. A new species of the genus Rossomyrmex Arnoldi, 1928 from Turkey (Hymenoptera. Formicidae). Graellsia 2007, 63, 135–142. [Google Scholar] [CrossRef]
- Ribera, I.; Blasco-Zumeta, J. Biogeographical links between steppe insects in the Monegros region (Aragon, NE Spain), the eastern Mediterranean, and central Asia. J. Biogeogr. 1998, 25, 969–986. [Google Scholar] [CrossRef]
- Sánchez Piñero, F.; Tinaut, A.; Aguirre-Segura, A.; Miñano, J.; Lencina, J.L.; Ortiz-Sánchez, F.J.; Pérez-López, F.J. Terrestrial arthropod fauna of arid areas of SE Spain: Diversity, biogeography, and conservation | Elsevier Enhanced Reader. J. Arid Environ. 2011, 75, 1321–1332. [Google Scholar] [CrossRef]
- Olivares, F.J.; Barea-Azcón, J.M.; Pérez-López, F.J.; Tinaut, A.; Henares, I. Las mariposas diurnas de Sierra Nevada; de Andalucía, J., Ed.; Consejería de Medio Ambiente: Seville, Spain, 2011. [Google Scholar]
- Tinaut, A. Taxonomic situation of the genus Cataglyphis Förster, 1850 in the Iberian Peninsula II. New position for C. viatica (Fabricius, 1787) and redescription of C. velox Santschi, 1929 stat. n.(Hymenoptera, Formicidae). Eos 1990, 66, 49–59. [Google Scholar]
- Martín, J.M.; Puga-Bernabéu, Á.; Aguirre, J.; Braga, J.C. Miocene Atlantic-Mediterranean seaways in the Betic cordillera (Southern Spain). Rev. Soc. Geol. Esp. 2014, 27, 175–186. [Google Scholar]
- Tinaut, A.; Heinze, J. Wing reduction in ant queens from arid habitats. Naturwissenschaften 1992, 79, 84–85. [Google Scholar] [CrossRef]
- Cerdá, X.; Arnan, X.; Retana, J. Is competition a significant hallmark of ant (Hymenoptera: Formicidae) ecology? Myrmecol. News 2013, 18, 131–147. [Google Scholar]
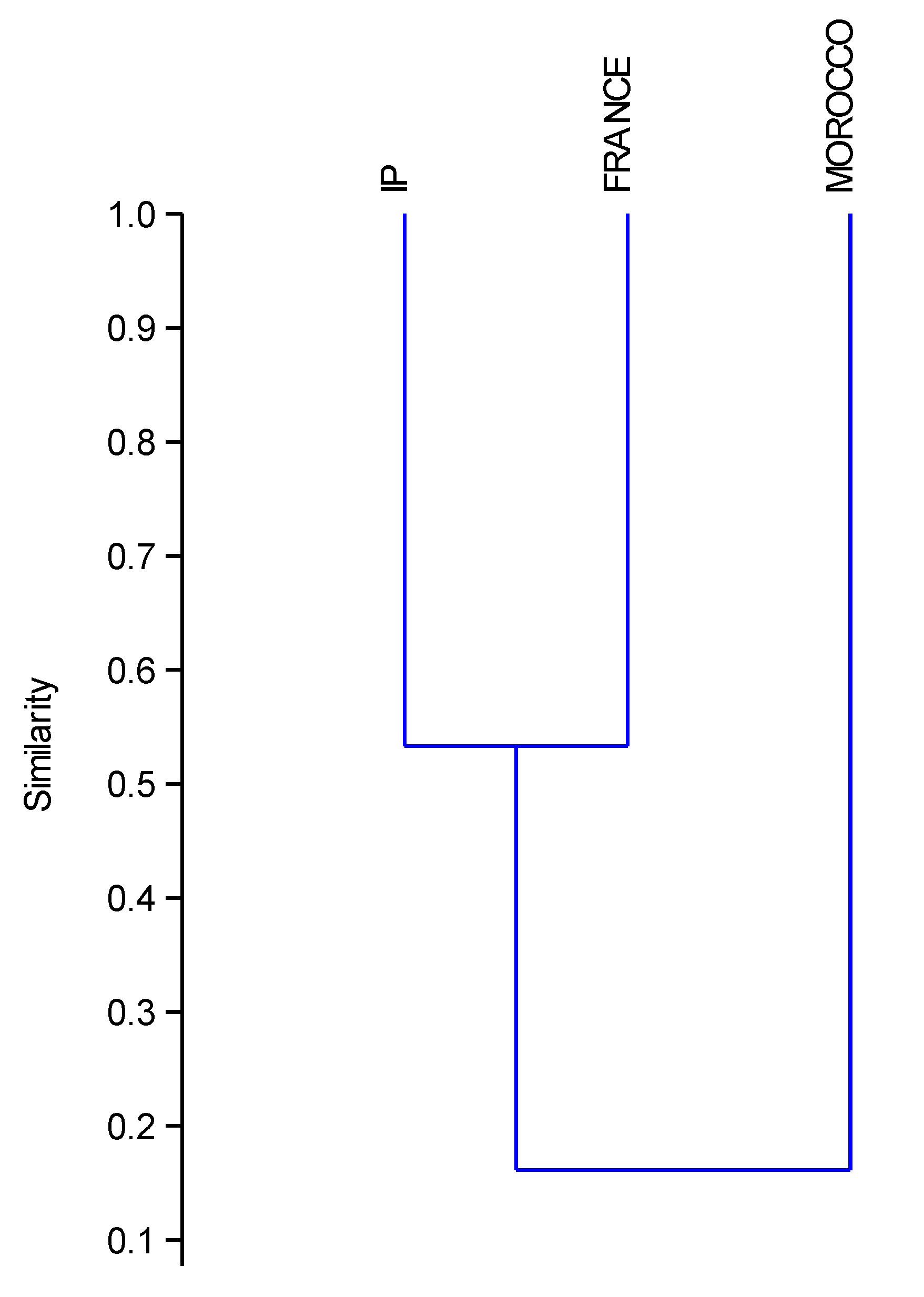
| Country/Region | Nº Species | Reference |
|---|---|---|
| EUROPE | 622 | [67] |
| IP | 299 | present paper |
| FRANCE | 211 | present paper |
| ITALY | 187 | [68] |
| BALKANS | 319 | [69] |
| ALBANIA | 79 | [70] |
| SERBIA | 116 | [71] |
| CROATIA | 140 | [72] |
| YUGOSLAVIA (former) | 171 | [69] |
| MACEDONIA | 80 | [73] |
| GREECE | 259 | [74] |
| TURKEY | 286 | [75] |
| ISRAEL | 203 | [76] |
| EGYPT | 97 | [77] |
| TUNISIA & ALGERIA | 180 | [57] |
| MOROCCO | 233 | present paper |
| Subfamily | Total Shared sp/% Fr-IP | Total Shared sp/% Fr-Mo | Total Shared sp/%IP-Fr | Total Shared sp/% IP-Mo | Total Shared sp/% Mo-Fr | Total Shared sp/% Mo/IP |
|---|---|---|---|---|---|---|
| Amblyoponinae | 0/0% | 0/0% | 0/0% | 2/67% | 0/0% | 2/67% |
| Cerapachyinae | 0/0% | 0/0% | 0/0% | 0/0% | 0/0% | 0/0% |
| Dorylinae | 0/0% | 0/0% | 0/0% | 0/0% | 0/0% | 0/0% |
| Ponerinae | 7/87.5% | 4/50% | 7/87.5% | 5/63% | 4/80% | 5/100% |
| Proceratiinae | 1/100% | 0/0% | 1/100% | 0/0% | 0/0% | 0/0% |
| Leptanillinae | 1/100% | 1/100% | 1/20% | 1/20% | 1/33% | 1/33% |
| Dolichoderinae | 10/100% | 3/30% | 10/62% | 4/25% | 3/37% | 3/37% |
| Formicinae | 69/90% | 13/17% | 68/72% | 19/20% | 13/21% | 19/31% |
| Myrmicinae | 89/79% | 28/25% | 89/52% | 57/33% | 28/19% | 45/38% |
| Total shared species | 178/84% | 49/23% | 176/59% | 88/29% | 49/21% | 87/37% |
| Total unshared species | 33/16% | 162/77% | 123/41% | 211/17% | 184/79% | 146/63% |
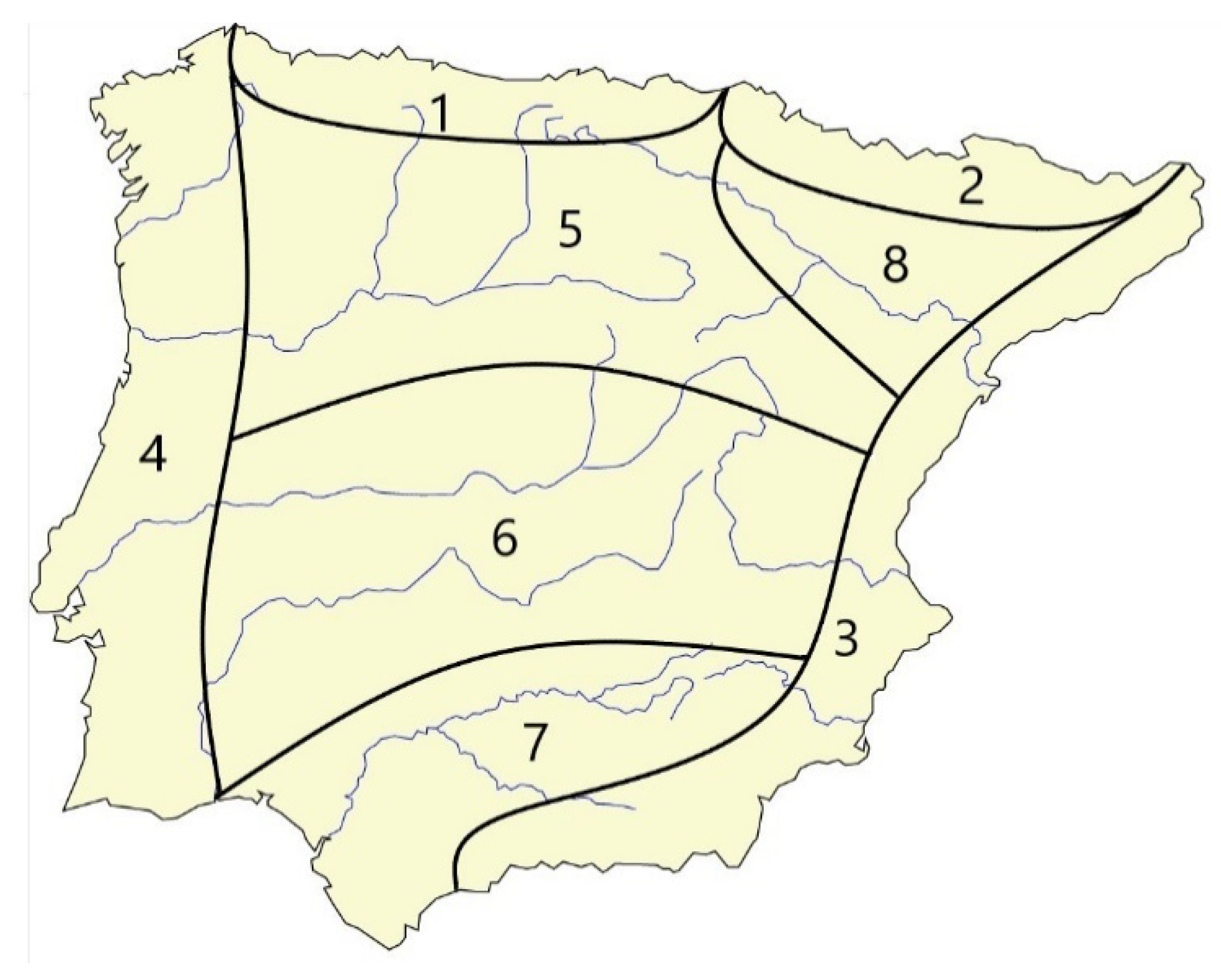
| Cantabric | Pyrenees | Mediterranean | Atlantic | Northern Plateau | Southern Plateau | Guadalquivir Valley | Ebro Valley | |
|---|---|---|---|---|---|---|---|---|
| Cantabric | 1 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 |
| Pyrenees | 0.28 | 1 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 |
| Mediterranean | 0.11 | 0.26 | 1 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 |
| Atlantic | 0.12 | 0.25 | 0.33 | 1 | <0.01 | <0.01 | <0.01 | <0.01 |
| Northern plateau | 0.23 | 0.32 | 0.35 | 0.36 | 1 | 0.01 | <0.01 | <0.01 |
| Southern plateau | 0.1 | 0.22 | 0.3 | 0.37 | 0.4 | 1 | <0.01 | <0.01 |
| Guadalquivir Valley | 0.1 | 0.21 | 0.4 | 0.35 | 0.4 | 0.4 | 1 | <0.01 |
| Ebro Valley | 0.1 | 0.25 | 0.09 | 0.17 | 0.21 | 0.2 | 0.15 | 1 |
| Nº Occupied Refugium Areas | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Nº species | 86 | 46 | 35 | 32 | 31 | 16 | 6 | 46 |
| Nº endemic sp | 24 | 12 | 10 | 10 | 10 | 2 | 0 | 5 |
| Pyrenean Area | |
| Myrmicinae | - |
| Myrmica lemasnei | Bernard, 1967 |
| Mediterranean Area | |
| Leptanillinae | - |
| Leptanilla theryi | Forel, 1903 |
| Formicinae | - |
| Camponotus amaurus | Espadaler, 1997 |
| Cataglyphis gadeai | De Haro & Collingwood, 2003 |
| Cataglyphis humeya | Tinaut, 1991 |
| Myrmicinae | - |
| Goniomma collingwoodi | Espadaler, 1997 |
| Goniomma compressisquama | Tinaut, 1994 |
| Messor timidus | Espadaler, 1997 |
| Teleutomyrmex kutteri | Tinaut, 1990 |
| Temnothorax ansei | Catarineu, Barberá & Reyes-López, 2017 |
| Temnothorax crepuscularis | (Tinaut, 1994) |
| Northern Plateau Area | |
| Leptanillinae | - |
| Leptanilla charonea | López, Martínez & Barandica, 1994 |
| Myrmicinae | - |
| Aphaenogaster ulibeli | Gómez & Espadaler, 2018 |
| Myrmoxenus bernardi | (Espadaler, 1982) |
| Southern Plateau Area | |
| Leptanillinae | - |
| Leptanilla plutonia | López, Martínez y Barandica, 1994 |
| Myrmicinae | - |
| Crematogaster fuentei | Menozzi, 1922 |
| Oxyopomyrmex magnus | Salata & Borowiec, 2015 |
| Guadalquivir Valley Area | |
| Formicinae | - |
| Cataglyphis floricola | Tinaut, 1993 |
| Cataglyphis tartessica | Amor & Ortega, 2014 |
| Myrmicinae | - |
| Goniomma baeticum | Reyes & Rodríguez, 1987 |
| Ebro Valley Area | |
| Myrmicinae | - |
| Temnothorax caesari | (Espadaler, 1997) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinaut, A.; Ruano, F. Biogeography of Iberian Ants (Hymenoptera: Formicidae). Diversity 2021, 13, 88. https://doi.org/10.3390/d13020088
Tinaut A, Ruano F. Biogeography of Iberian Ants (Hymenoptera: Formicidae). Diversity. 2021; 13(2):88. https://doi.org/10.3390/d13020088
Chicago/Turabian StyleTinaut, Alberto, and Francisca Ruano. 2021. "Biogeography of Iberian Ants (Hymenoptera: Formicidae)" Diversity 13, no. 2: 88. https://doi.org/10.3390/d13020088
APA StyleTinaut, A., & Ruano, F. (2021). Biogeography of Iberian Ants (Hymenoptera: Formicidae). Diversity, 13(2), 88. https://doi.org/10.3390/d13020088






