Threats Posed to the Rediscovered and Rare Salvia ceratophylloides Ard. (Lamiaceae) by Borer and Seed Feeder Insect Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Species
2.2. Sampling Sites
2.3. Data Collection
2.3.1. Study Methods and Field Observations
2.3.2. Borers and Their Natural Enemies
2.3.3. Seed Feeders and Their Natural Enemies
2.3.4. Herbivores and Natural Enemies’ Identification
2.4. Data Analyses
2.5. Molecular Identification
DNA Extraction, Amplification, and Sequencing
3. Results
3.1. Field Observations
3.2. Complex of Herbivores and Their Natural Enemies on Salvia ceratophylloides Plants
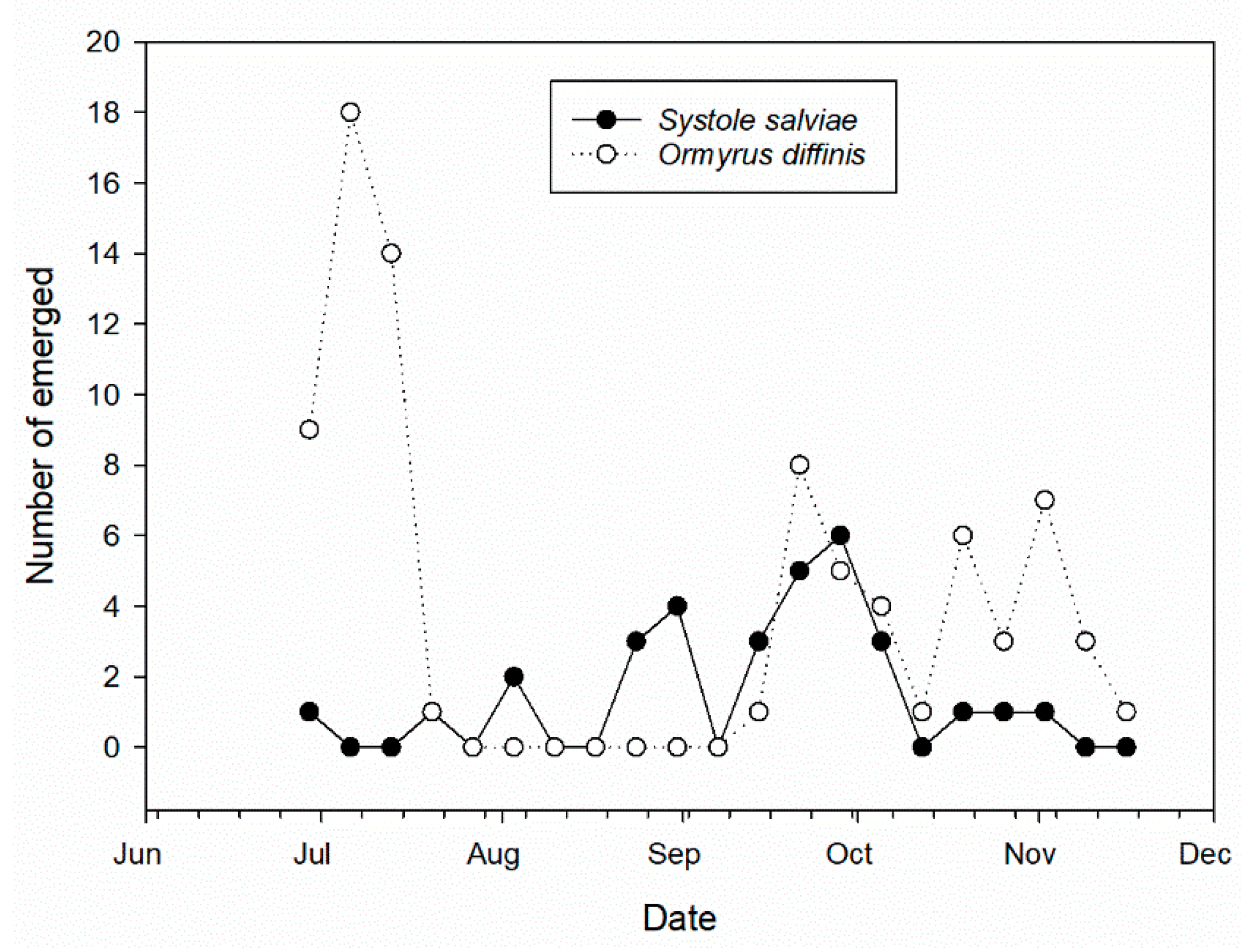
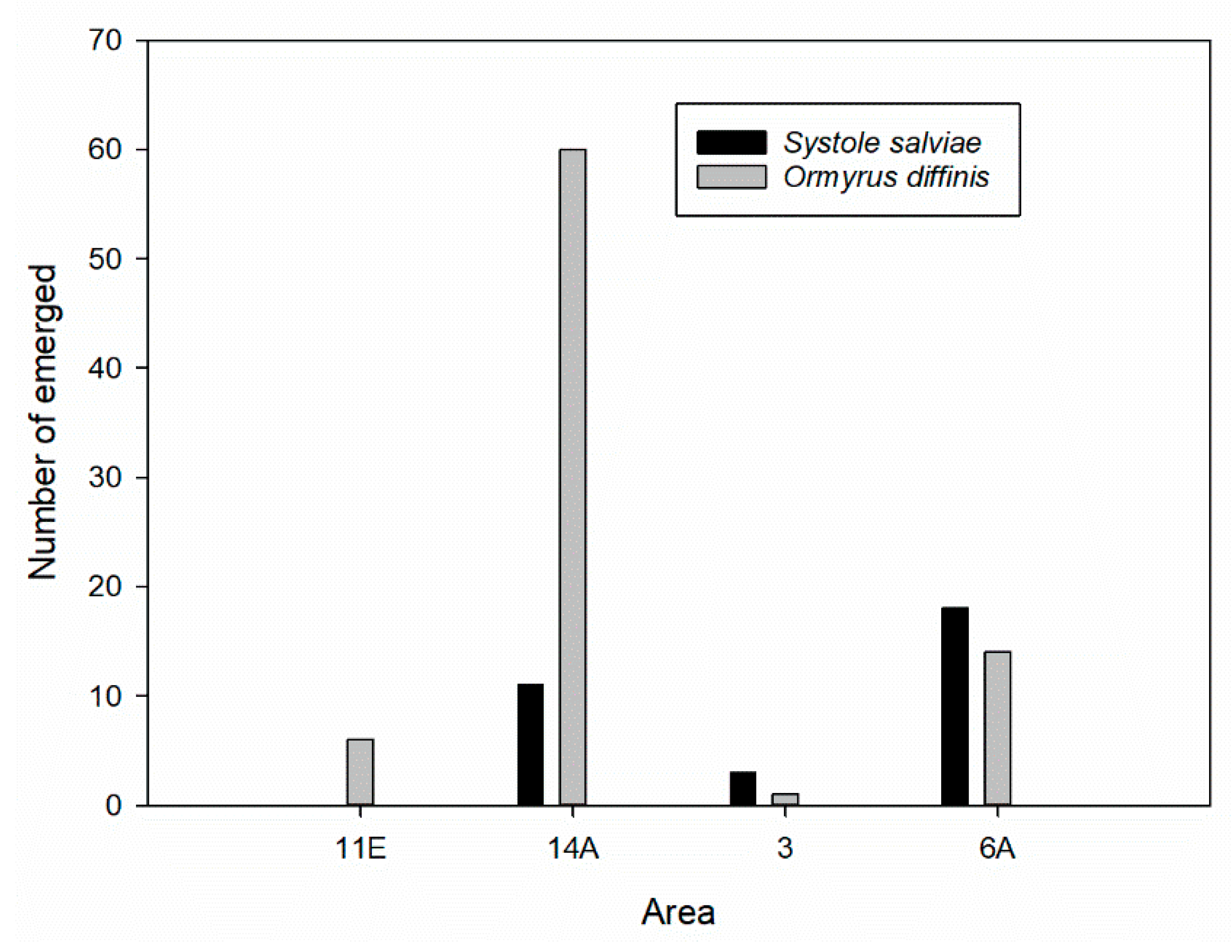
3.3. Salvia ceratophylloides Seed Herbivores and Their Natural Enemies
3.4. Molecular Identification of the Species
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Menges, E.S. Habitat preferences and response to disturbance for Dicerandra frutescens, a lake wales ridge (Florida) endemic plant. Bull. Torrey Bot. Club 1992, 119, 308. [Google Scholar] [CrossRef]
- Kaye, T.N.; Kirkland, M. Effects of timber harvest on Cimicifuga elata, a rare plant of western forests. Northwest Sci. 1999, 73, 159–167. [Google Scholar]
- Corlett, R.T. Plant diversity in a changing world: Status, trends, and conservation needs. Plant Divers. 2016, 38, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Wagner, D.L.; Van Driesche, R.G. Threats posed to rare or endangered insects by invasions of nonnative species. Annu. Rev. Èntomol. 2010, 55, 547–568. [Google Scholar] [CrossRef]
- Souza, L.; Zelikova, T.J.; Sanders, N.J. Bottom-up and top-down effects on plant communities: Nutrients limit productivity, but insects determine diversity and composition. Oikos 2015, 125, 566–575. [Google Scholar] [CrossRef]
- Maron, J.L.; Baer, K.C.; Angert, A.L. Disentangling the drivers of context-dependent plant-animal interactions. J. Ecol. 2014, 102, 1485–1496. [Google Scholar] [CrossRef]
- Brigham, C.A. Factors affecting persistence in formerly common and historically rare plants. In Population Viability in Plants. Ecological Studies (Analysis and Synthesis); Brigham, C.A., Schwartz, M.W., Eds.; Springer: Berlin/Heidelberg, Germany, 2003; Volume 165. [Google Scholar]
- Ancheta, J.; Heard, S.B. Impacts of insect herbivores on rare plant populations. Biol. Conserv. 2011, 144, 2395–2402. [Google Scholar] [CrossRef]
- Jorge, A.; Loureiro, J.; Castro, S. Flower biology and breeding system of Salvia sclareoides Brot. (Lamiaceae). Plant Syst. Evol. 2014, 301, 1485–1497. [Google Scholar] [CrossRef]
- Myers, J.H.; Sarfraz, R.M. Impacts of insect herbivores on plant populations. Annu. Rev. Èntomol. 2017, 62, 207–230. [Google Scholar] [CrossRef]
- Barrett, S.C.H. Understanding plant reproductive diversity. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 99–109. [Google Scholar] [CrossRef]
- Charlesworth, D. Evolution of plant breeding systems. Curr. Biol. 2006, 16, R726–R735. [Google Scholar] [CrossRef] [PubMed]
- Schöps, K. Local and regional dynamics of a specialist herbivore: Overexploitation of a patchily distributed host plant. Oecologia 2002, 132, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Kenis, M.; Auger-Rozenberg, M.-A.; Roques, A.; Timms, L.; Péré, C.; Cock, M.J.W.; Settele, J.; Augustin, S.; Lopez-Vaamonde, C. Ecological effects of invasive alien insects. Biol. Invasions 2008, 11, 21–45. [Google Scholar] [CrossRef]
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef]
- Oostermeijer, J.G.B. Threats to rare plant persistence. In Population Viability in Plants: Conservation, Management and Modeling of Rare Plants; Brigham, C., Schwartz, M., Eds.; Springer: Heidelberg, Germany, 2003; pp. 17–58. [Google Scholar]
- Brullo, S.; Scelsi, F.; Spampinato, G. La vegetazione dell’Aspromonte. Studio Fitosociologico; Laruffa Editore: Reggio Calabria, Italy, 2001; pp. 1–370. ISBN 88-7221-160-3. [Google Scholar]
- Spampinato, G. Guida Alla Flora dell’Aspromonte; Laruffa Editore: Reggio Calabria, Italy, 2014; ISBN 978-88-7221-707-8. [Google Scholar]
- Del Carratore, F.; Garbari, F. Indagini biosistematiche sul genere Salvia L. Sect. Plethiosphace Benyham (Labiatae). Inf. Bot. Ital. 1997, 29, 297–299. [Google Scholar]
- Conti, F.; Manzi, A.; Pedrotti, F. Liste rosse Regionali Delle Piante d’Italia; Dipartimento di Botanica ed Ecologia, Università degli Studi di Camerino: Camerino, Italy, 1997. [Google Scholar]
- Conti, F.; Abbate, G.; Alessandrini, A.; Blasi, C. An Annotated Checklist of the Italian Vascular Flora; Palombi Editori: Roma, Italy, 2005; p. 420. [Google Scholar]
- Scoppola, A.; Spampinato, G. Atlante delle specie a rischio di estinzione. In Stato Delle Conoscenze Sulla Flora Vascolare d’Italia; Scoppola, A., Blasi, C., Eds.; Palombi Editori: Roma, Italy, 2005. [Google Scholar]
- Lacaita, C. Addenda et emendanda ad floram italicam. Bull. Della Soc. Bot. Ital. 1921, 28, 18–19. [Google Scholar]
- Spampinato, G. Guida alla Flora dell’Aspromonte; Laruffa Editore: Reggio Calabria, Italy, 2002; ISBN 88-7221-189-1. [Google Scholar]
- Spampinato, G.; Crisafulli, A. Struttura Delle Popolazioni e Sinecologia di Salvia ceratophylloides (Lamiaceae) Specie Endemica Minacciata di Estinzione; Spamipanato, G.G., Signorino, G., Eds.; Atti 103° Congr Società Botanica Italiana; 17–19 Seet: Reggio Calabria; Kalìt Editrice: Catania, Italy, 2008; p. 56. [Google Scholar]
- Crisafulli, A.; Cannavò, S.; Maiorca, G.; Musarella, C.M.; Signorino, G.; Spampinato, G. Aggiornamenti floristici per la Calabria. Inf. Bot. Ital. 2010, 42, 431–442. [Google Scholar]
- Spampinato, G.; Crisafulli, A.; Marino, A.; Signorino, G. Salvia ceratophylloides Ard. Inf. Bot. Ital. 2011, 43, 381–458. [Google Scholar]
- Laface, V.L.A.; Musarella, C.M.; Spampinato, G. Conservation status of the Aspromontana flora: Monitoring and new stations of Salvia ceratophylloides Ard. (Lamiaceae) endemic species in Reggio Calabria (Southern Italy). In Proceedings of the 113° Congresso Della Società Botanica Italiana. V International Plant Science Conference (IPSC), Fisciano, Italy, 12–15 September 2018; p. 96, ISBN 978-88-85915-22-0. [Google Scholar]
- Spampinato, G.; Araniti, F.; Vescio, R.; Musarella, C.M.; Di Iorio, A.; Abenavoli, M.R.; Sorgonà, A. VOCs composition of Salvia ceratophylloides Ard. (Lamiaceae), a Calabrian endangered species, through targeted and untargeted metabolomic analyses. In Proceedings of the 113° Congresso Della Società Botanica Italiana. V International Plant Science Conference (IPSC), Fisciano, Italy, 12–15 September 2018; p. 33, ISBN 978-88-85915-22-0. [Google Scholar]
- Spampinato, G.; LaFace, V.L.A.; Ortiz, A.C.; Canas, R.Q.; Musarella, C.M. Salvia ceratophylloides Ard. (Lamiaceae): A rare endemic species of Calabria (Southern Italy). In Endemic Species; Cano Carmona, E., Musarella, C.M., Cano Ortiz, A., Eds.; IntechOpen: London, UK, 2019. [Google Scholar]
- FAO-Unesco. Soil Map of the World, Revised Legend; FAO: Rome, Italy, 1990. [Google Scholar]
- Climate-Data.org© / AM OP / OpenStreetMap Contributors USt-IdNR: DE279133320. 2020. Available online: https://it.climate-data.org/europa/italia/calabria/reggio-di-calabria-1147/ (accessed on 24 April 2020).
- Hegland, S.J.; Van Leeuwen, M.; Oostermeijer, J.G.B. Population structure of Salvia pratensis in relation to vegetation and management of Dutch dry floodplain grasslands. J. Appl. Ecol. 2001, 38, 1277–1289. [Google Scholar] [CrossRef]
- Wanat, M. New and little known Squamapion species (Coleoptera: Apionidae) from Western Paleartic. Ann. Zool. 1997, 47, 285–295. [Google Scholar]
- Wanat, M. New records of the Apionidae (Coleoptera: Curculionoidea) from NE Poland. Wiad. Entomol. 1998, 17, 49–53. [Google Scholar]
- Alonso-Zarazaga, M.A. Revision of the supraspecific taxa in the palaearctic Apionidae Schoenherr, 1823. 1. Introduction and subfamily Nanophyinae Seidlitz, 1891 (Coleoptera, Curculionoidea). Fragm. Entomol. 1989, 21, 205–262. [Google Scholar]
- Alonso-Zarazaga, M.A. Revision of the supraspecific taxa in the Palaearctic Apionidae Schoenherr, 1823 (Coleoptera, Curculionoidea) 2 Subfamily Apioninae Schoenherr, 1823: Instruction, keys and descriptions. Graellsia 1990, 46, 19–156. [Google Scholar]
- Baur, H.; Muller, F.; Gibson, G.; Mason, P.; Kuhlmann, U. A review of the species of Mesopolobus (Chalcidoidea: Pteromalidae) associated with Ceutorhynchus (Coleoptera: Curculionidae) host-species of European origin. Bull. Èntomol. Res. 2007, 97, 387–397. [Google Scholar] [CrossRef]
- Muller, F.J.; Baur, H.; Gibson, G.A.; Mason, P.G.; Kuhlmann, U. Review of the species of Trichomalus (Chalcidoidea: Pteromalidae) associated with Ceutorhynchus (Coleoptera: Curculionidae) host species of European origin. Can. Èntomol. 2007, 139, 643–657. [Google Scholar] [CrossRef][Green Version]
- Freude, H.; Harde, K.W.; Lohse, G.A. Die Käfer Mitteleuropas, Band 11; Goecke and Evers Verlag: Krefeld, Germany, 1983. [Google Scholar]
- Zerova, M.D. New species of systole walk (Hymenoptera, Eurytomidae) from the seeds of Salvia spp. Vest. Zoolog. 1968, 4, 22–26. (In Russian) [Google Scholar]
- Zerova, M.D. Parasitic Hymenoptera—Eurytominae and Eudecatominae of Palearctics; Schmalhausen, I.I., Ed.; The National Academy of Sciences of Ukraine, Institute of Zoology: Kyiv, Russian, 1995. [Google Scholar]
- Zerova, M.D. The Parasitic Hymenoptera–Subfamilies Eurytominae and Eudecatominae (Chalcidoidea, Eurytomidae) of the Palaearctics; Naukova Dumka Publishers: Kiev, Ukraine, 1995. (In Russian) [Google Scholar]
- Popescu, I.E. Eurytomid Wasps (Hymenoptera, Chalcidoidea, Eurytomidae) New for Romanian Fauna (III). Analele Ştiinţifice ale Universităţii “AL. I. CUZA” Iaşi, s. Biologie Animală 2006, 139, 155–161. [Google Scholar]
- IBM Corp. Released: IBM SPSS Statistics for Windows, Version 23.0; IBM Corp.: Armonk, NY, USA, 2015. [Google Scholar]
- Sigmaplot 13.0. Systat Copyright © Systat Software; Systat Software Inc.: San Jose, CA, USA, 2018. [Google Scholar]
- Vono, G.; Bonsignore, C.P.; Gullo, G.; Marullo, R. Olive production threatened by a resurgent pest Liothrips oleae (Costa, 1857) (Thysanoptera: Phlaeothripidae) in Southern Italy. Insects 2020, 11, 887. [Google Scholar] [CrossRef]
- Simon, C.; Frati, F.; Beckenbach, A.; Crespi, B.J.; Liu, H.; Flook, P. Evolution, Weighting, and Phylogenetic Utility of Mitochondrial Gene Sequences and a Compilation of Conserved Polymerase Chain Reaction Primers. Ann. Èntomol. Soc. Am. 1994, 87, 651–701. [Google Scholar] [CrossRef]
- Campbell, B.C.; Steffen-Campbell, J.D.; Werren, J.H. Phylogeny of the Nasonia species complex (Hymenoptera: Pteromalidae) inferred from an internal transcribed spacer (ITS2) and 28S rDNA sequences. Insect Mol. Biol. 1994, 2, 225–237. [Google Scholar] [CrossRef]
- Madeira, F.; Park, Y.M.; Lopez, R.; Lee, J.; Buso, N.; Gur, T.; Madhusoodanan, N.; Basutkar, P.; Tivey, A.R.N.; Potter, S.C.; et al. The EMBL-EBI search and sequence analysis tools APIs in 2019. Nucleic Acids Res. 2019, 47, W636–W641. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Beckenbach, A.T. Evolution of the mitochondrial cytochrome oxidase II gene among 10 orders of insects. Mol. Phylogenet. Evol. 1992, 1, 41–52. [Google Scholar] [CrossRef]
- Alonso-Zarazaga, M.A. Apionidae. In Catalogue of Palaearctic Coleoptera; Löbl, I., Smetana, A., Eds.; Apollo Books: Stenstrup, Denmar, 2011; Volume 7, p. 373. [Google Scholar]
- Łętowski, J.; Pawlęga, K.; Ścibior, R.; Rojek, K. The morphology of the preimaginal stages of Squamapion elongatum (Germar, 1817) (Coleoptera, Curculionoidea, Apionidae) and notes on its biology. ZooKeys 2015, 519, 101–115. [Google Scholar] [CrossRef] [PubMed]
- Cmoluch, Z. Badania nad fauną ryjkowców (Coleoptera, Curculionidae) roślinnych zespołów kserotermicznych południowo-wschodniej części Wyżyny Lubelskiej. Ann. Univ. Mariae Curie-Skłodowska Sectio C 1962, 17, 1–75. [Google Scholar]
- Lopes, M.F.R. Estudo da Entomofauna Presente em Alguns Habitats da Salvia sclerioides Brot. Master’s Thesis, Polytechnic Institute of Santarém, Santarém, Portugal, 2013. [Google Scholar]
- Zarkani, A.; Turanli, F. Insect pests complex of common Sage (Salvia officinalis L.) (Lamiaceae) and their natural enemies. J. Agric. Sci. 2019, 29, 34–42. [Google Scholar]
- Hendrich, L.; Morinière, J.; Haszprunar, G.; Hebert, P.D.N.; Hausmann, A.; Köhler, F.; Balke, M. A comprehensive DNA barcode database for Central European beetles with a focus on Germany: Adding more than 3500 identified species to BOLD. Mol. Ecol. Resour. 2014, 15, 795–818. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information (NCBI). Bethesda, M.D. National Library of Medicine (US), National Center for Biotechnology Information. Available online: https://www.ncbi.nlm.nih.gov (accessed on 25 April 2020).
- Woodcock, T.S.; Boyle, E.E.; Roughley, R.E.; Kevan, P.G.; Labbee, R.N.; Smith, A.B.; Goulet, H.; Steinke, D.; Adamowicz, S.J. The diversity and biogeography of the Coleoptera of Churchill: Insights from DNA barcoding. BMC Ecol. 2013, 13, 40. [Google Scholar] [CrossRef]
- Pentinsaari, M.; Hebert, P.D.N.; Mutanen, M. Barcoding Beetles: A Regional Survey of 1872 Species Reveals High Identification Success and Unusually Deep Interspecific Divergences. PLoS ONE 2014, 9, e108651. [Google Scholar] [CrossRef]
- Kazachkova, N.; Meijer, J.; Ekbom, B. Genetic diversity in European pollen beetle (Meligethes aeneus) populations assessed using AFLP analysis. Eur. J. Entomol. 2008, 105, 807–814. [Google Scholar] [CrossRef]
- Hairston, N.G.; Smith, F.E.; Slobodkin, L.B. Community Structure, Population Control, and Competition. Am. Nat. 1960, 94, 421–425. [Google Scholar] [CrossRef]
- Wetzel, W.C.; Kharouba, H.M.; Robinson, M.; Holyoak, M.; Karban, R. Variability in plant nutrients reduces insect herbivore performance. Nat. Cell Biol. 2016, 539, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Koussoroplis, A.M.; Klauschies, T.; Pincebourde, S.; Giron, D.; Wacker, A. A comment on “Variability in plant nutrients reduces insect herbivore performance”. Rethink. Ecol. 2019, 4, 79–87. [Google Scholar] [CrossRef]
- Ford, C.R.; Elliott, K.J.; Clinton, B.D.; Kloeppel, B.D.; Vose, J.M. Forest dynamics following eastern hemlock mortality in the southern Appalachians. Oikos 2011, 121, 523–536. [Google Scholar] [CrossRef]
- Poland, T.M.; McCullough, D.G. Emerald ash borer: Invasion of the urban forest and the threat to North America’s ash resource. J. For. 2006, 104, 118–124. [Google Scholar]
- Haye, T.; Olfert, O.; Weiss, R.; Mason, P.G.; Gibson, G.; Gariepy, T.D.; Gillespie, D.R. Bioclimatic analyses of Trichomalus perfectus and Mesopolobus morys (Hymenoptera: Pteromalidae) distributions, two potential biological control agents of the cabbage seedpod weevil in North America. Biol. Control. 2018, 124, 30–39. [Google Scholar] [CrossRef]
- Lotfalizadeh, H.; Askew, R.R.; Fuentes-Utrilla, P.; Tavakoli, M. The species of Ormyrus Westwood (Hymenoptera: Ormyridae) in Iran with description of an unusual new species. Zootaxa 2012, 3300, 34–44. [Google Scholar] [CrossRef]
- Askew, R.R.; Nieves-Aldrey, J.-L. Eupelmidae de Iberia y las Islas Canarias: Check list comentada de las especies, incluyendo la descripción de una nueva especie de Calosota Curtis, 1836 y de los machos previamente no reconocidos de algunas especies. Graellsia 2017, 73, 065. [Google Scholar] [CrossRef]
- Fusu, L An integrative taxonomic study of European Eupelmus (Macroneura) (Hymenoptera: Chalcidoidea: Eupelmidae), with a molecular and cytogenetic analysis of Eupelmus (Macroneura) vesicularis: Several species hiding under one name for 240 years. Zool. J. Linn. Soc. 2017, 181, 519–603. [CrossRef]
- Bonsignore, C.P.; Vono, G.; Bernardo, U. Environmental thermal levels affect the phenological relationships between the chestnut gall wasp and its parasitoids. Physiol. Entomol. 2019, 44, 87–98. [Google Scholar] [CrossRef]
- Crawley, M.J.; Ross, G.J.S. The population dynamics of plants. Philos. Trans. R. Soc. B Biol. Sci. 1990, 330, 125–140. [Google Scholar] [CrossRef]
- Alsendi, A.; Adelimaneshy, H.; Sadeghi, S.E. Report of some Eurytomid wasps (Hymenoptera: Eurytomidae) and their associated host plants from two provinces of Iran. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Karbala, Iraq, 17–18 November 2019; Volume 388, p. 012018. [Google Scholar]
- Askew, R.R.; Plantard, O.; Gómez, J.F.; Nieves, M.H.; Nieves-Aldrey, J.L. Catalogue of parasitoids and inquilines in galls of Aylacini, Diplolepidini and Pediaspidini (Hym., Cynipidae) in the West Palaearctic. Zootaxa 2006, 1301, 1–60. [Google Scholar] [CrossRef]
- Gomez, J.F.; Nieves, M.H.; Gayubo, S.F.; Nieves-Aldrey, J.L. Terminal-instar larval systematics and biology of west European species of Ormyridae associated with insect galls (Hymenoptera, Chalcidoidea). ZooKeys 2017, 644, 51–88. [Google Scholar] [CrossRef] [PubMed]
- Bonsignore, C.P.; Bernardo, U. Effects of environmental parameters on the chestnut gall wasp and its complex of indigenous parasitoids. Naturwissenschaften 2018, 105, 20. [Google Scholar] [CrossRef]
- Ferracini, C.; Bertolino, S.; Bernardo, U.; Bonsignore, C.P.; Faccoli, M.; Ferrari, E.; Lupi, D.; Maini, S.; Mazzone, L.; Nugnes, F.; et al. Do Torymus sinensis (Hymenoptera: Torymidae) and agroforestry system affect native parasitoids associated with the Asian chestnut gall wasp? Biol. Control 2018, 121, 36–43. [Google Scholar] [CrossRef]
- Bonsignore, C.P.; Manti, F.; Castiglione, E. Interactions between pupae of the pine processionary moth (Thaumetopoea pityocampa) and parasitoids in a Pinus forest. Bull. Entomol. Res. 2015, 105, 621–628. [Google Scholar] [CrossRef]
- Fernández-Martínez, M.; Pearse, I.; Sardans, J.; Sayol, F.; Koenig, W.D.; Lamontagne, J.M.; Bogdziewicz, M.; Collalti, A.; Hacket-Pain, A.; Vacchiano, G.; et al. Nutrient scarcity as a selective pressure for mast seeding. Nat. Plants 2019, 5, 1222–1228. [Google Scholar] [CrossRef]
- Espelta, J.M.; Arias-LeClaire, H.; Fernández-Martínez, M.; Doblas-Miranda, E.; Muñoz, A.; Bonal, R. Beyond predator satiation: Masting but also the effects of rainfall stochasticity on weevils drive acorn predation. Ecosphere 2017, 8, e01836. [Google Scholar] [CrossRef]




| Station ID | Subsite | Date | Surface Subsite (m2) | Non-Breeding Plants | Breeding Plants | Total Number of Plants | Infested Plants |
|---|---|---|---|---|---|---|---|
| 1 | 1° | 17.06.2019 | 16 | 29 | 14 | 43 | 5 |
| 1B | 6 | 5 | 11 | 16 | 3 | ||
| 2 | 2A | 17.06.2019 | 12 | 2 | 18 | 20 | 18 |
| 2B | 2 | 0 | 2 | 2 | 2 | ||
| 2C | 1 | 0 | 1 | 1 | 1 | ||
| 3 | 3A | 25.06.2019 | 36 | 15 | 46 | 61 | 35 |
| 3B | 12 | 0 | 7 | 7 | 4 | ||
| 4 | 4 | 22.06.2019 | 15 | 1 | 17 | 18 | 0 |
| 5 | 5A | 22.06.2019 | 10 | 5 | 12 | 17 | 0 |
| 5B | 1200 | 36 | 236 | 272 | 0 | ||
| 6 | 6A | 19.06.2019 | 80 | 0 | 46 | 46 | 15 |
| 6B | 265 | 52 | 41 | 93 | 8 | ||
| 6C | 168 | 14 | 78 | 92 | 30 | ||
| 6D | 1 | 0 | 1 | 1 | 0 | ||
| 6E | 6 | 0 | 6 | 6 | 2 | ||
| 6F | 1 | 0 | 1 | 1 | 0 | ||
| 7 | 7A | 22.06.2019 | 3 | 0 | 8 | 8 | 4 |
| 7B | 25 | 50 | 21 | 71 | 16 | ||
| 8 | 8 | 22.06.2019 | 8 | 4 | 9 | 13 | 1 |
| 9 | 9 | 22.06.2019 | 10 | 4 | 5 | 9 | 2 |
| 10 | 10 | 22.06.2019 | 120 | 6 | 120 | 126 | 20 |
| 11 | 11A | 19.06.2019 | 1 | 0 | 3 | 3 | 2 |
| 11B | 175 | 12 | 19 | 31 | 3 | ||
| 11C | 8 | 11 | 12 | 23 | 0 | ||
| 11D | 9 | 4 | 0 | 4 | 0 | ||
| 11E | 9 | 39 | 10 | 49 | 1 | ||
| 11F | 35 | 3 | 26 | 29 | 0 | ||
| 11G | 2 | 1 | 2 | 3 | 0 | ||
| 11H | 1 | 0 | 1 | 1 | 0 | ||
| 11I | 15 | 15 | 7 | 22 | 0 | ||
| 12 | 12A | 17.06.2019 | 50 | 0 | 25 | 25 | 6 |
| 12B | 3 | 4 | 4 | 8 | 2 | ||
| 13 | 13 | 17.06.2019 | Extinct | 0 | 0 | 0 | 0 |
| 14 | 14A | 23.06.2019 | 20 | 4 | 15 | 19 | 1 |
| 14B | 40 | 1 | 8 | 9 | 2 | ||
| 15 | 15A | 22.06.2019 | 10 | 0 | 55 | 55 | 2 |
| 15B | 1 | 0 | 5 | 5 | 0 | ||
| 16 | 16 | 22.06.2019 | 3 | 2 | 16 | 18 | 0 |
| 17 | 17 | 22.06.2019 | 25 | 2 | 10 | 12 | 2 |
| 18 | 18 | 22.06.2019 | 4 | 0 | 3 | 3 | 0 |
| Name | Sequence 5′–3′ | Fragment | Source | PCR Cycle | ||
|---|---|---|---|---|---|---|
| T (°C) | Time | N of Cycle | ||||
| LCO-1490 | GGTCAACAAATCATAAAGATATTGG | COI | [48] | 95 | 1′ | |
| 94 | 30” | 40 | ||||
| 48 | 1′30” | |||||
| HCO-2198 | GTAAATATATGRTGDGCTC | 72 | 1′ | |||
| 72 | 7” | |||||
| D2F | CGTGTTGCTTGATAGTGCAGC | 28S-D2 | [49] | 95 | 3′ | |
| 94 | 45” | 35 | ||||
| 55 | 45” | |||||
| D2R | TTGGTCCGTGTTTCAAGACGGG | 72 | 1′ | |||
| 72 | 7” | |||||
| Response Variable | Source | Df | Wald Chi-Square | p |
|---|---|---|---|---|
| Plants infested | Intercept | 1 | 0.91 | 0.340 |
| Site | 12 | 19.34 | 0.081 | |
| Reproductive plants (RPs) | 1 | 12.04 | 0.001 | |
| Growing plants (GPs) | 1 | 0.50 | 0.468 | |
| RP * GP | 1 | 0.392 | 0.081 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonsignore, C.P.; Laface, V.L.A.; Vono, G.; Marullo, R.; Musarella, C.M.; Spampinato, G. Threats Posed to the Rediscovered and Rare Salvia ceratophylloides Ard. (Lamiaceae) by Borer and Seed Feeder Insect Species. Diversity 2021, 13, 33. https://doi.org/10.3390/d13010033
Bonsignore CP, Laface VLA, Vono G, Marullo R, Musarella CM, Spampinato G. Threats Posed to the Rediscovered and Rare Salvia ceratophylloides Ard. (Lamiaceae) by Borer and Seed Feeder Insect Species. Diversity. 2021; 13(1):33. https://doi.org/10.3390/d13010033
Chicago/Turabian StyleBonsignore, Carmelo Peter, Valentina Lucia Astrid Laface, Gregorio Vono, Rita Marullo, Carmelo Maria Musarella, and Giovanni Spampinato. 2021. "Threats Posed to the Rediscovered and Rare Salvia ceratophylloides Ard. (Lamiaceae) by Borer and Seed Feeder Insect Species" Diversity 13, no. 1: 33. https://doi.org/10.3390/d13010033
APA StyleBonsignore, C. P., Laface, V. L. A., Vono, G., Marullo, R., Musarella, C. M., & Spampinato, G. (2021). Threats Posed to the Rediscovered and Rare Salvia ceratophylloides Ard. (Lamiaceae) by Borer and Seed Feeder Insect Species. Diversity, 13(1), 33. https://doi.org/10.3390/d13010033










