The First Fossil Owl (Aves, Strigiformes) From the Paleogene of Africa
Abstract
1. Introduction
2. Materials and Methods
2.1. Osteological and Geologic Conventions
2.2. Institutional Abbreviations
3. Systematic Paleontology
3.1. Referred Specimen
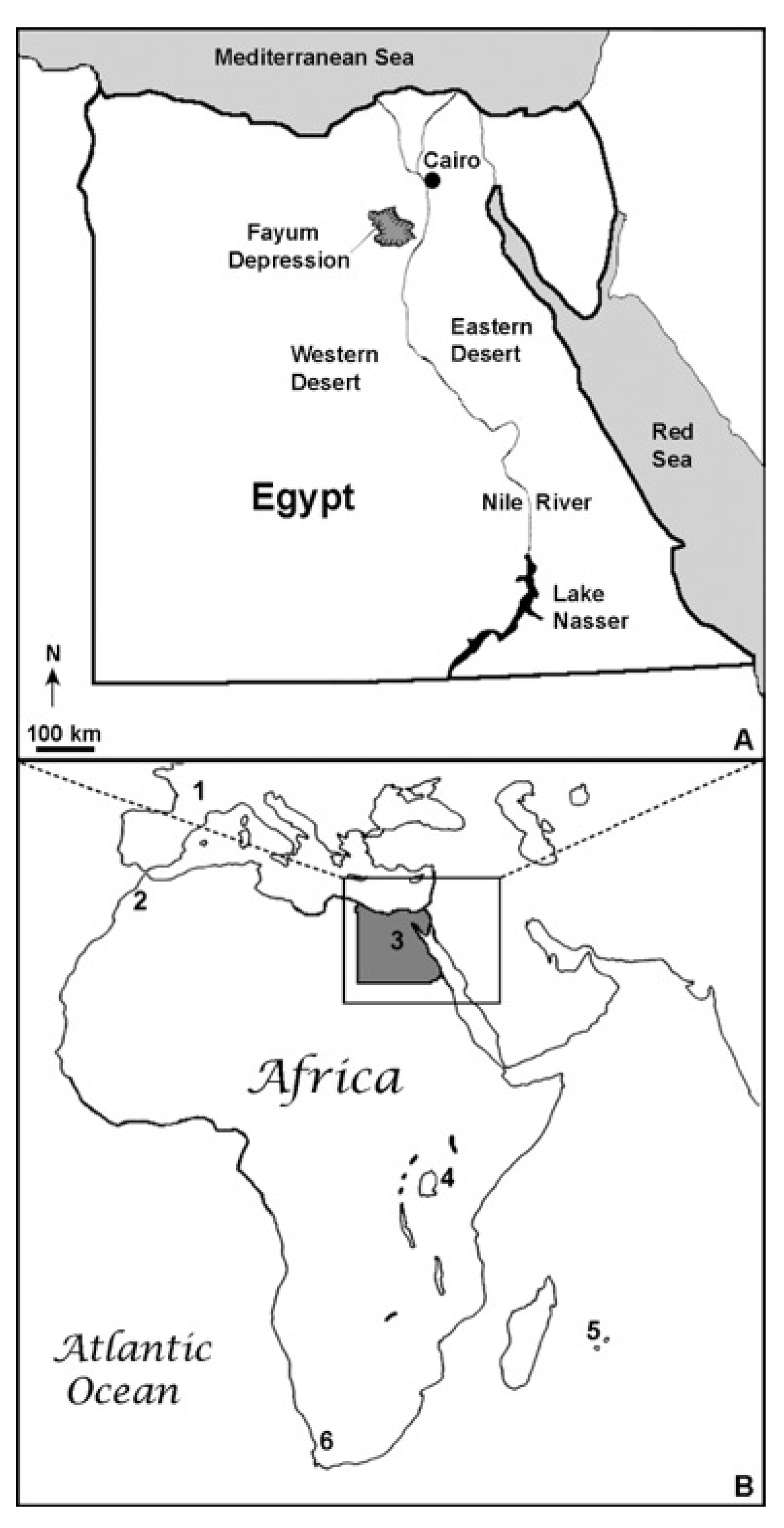
3.2. Locality
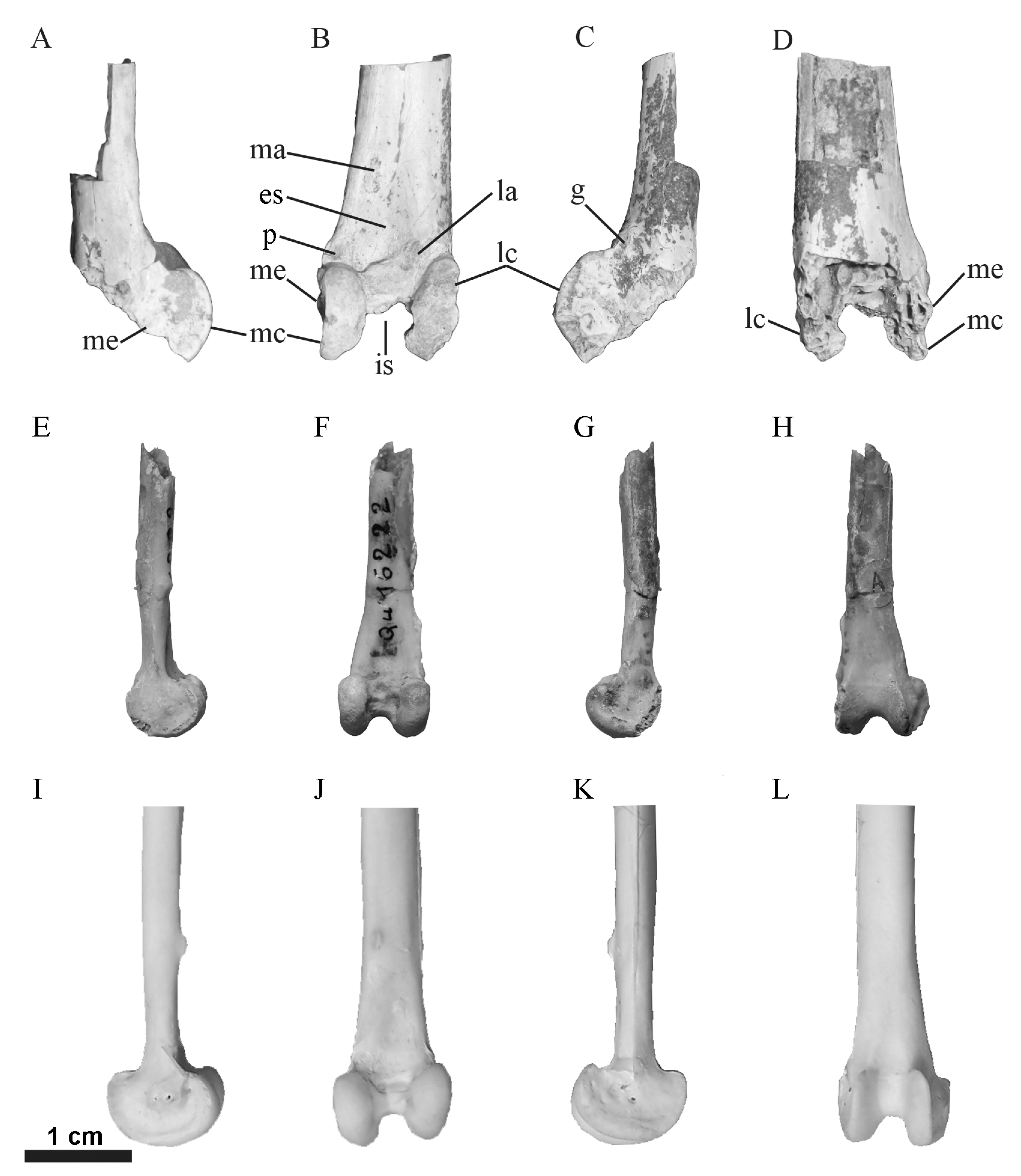
3.3. Anatomical Description
3.4. Taxonomic Comparisons
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barn-Owls to Hummingbirds; Del Hoyo, J.; Elliott, A.; Sargatal, J. (Eds.) Handbook of the Birds of the World; Lynx Edicions: Barcelona, Spain, 1999; Volume 5, p. 759. [Google Scholar]
- Kurochkin, E.N.; Dyke, G.J. The First Fossil Owls (Aves: Strigiformes) from the Paleogene of Asia and a Review of the Fossil Record of Strigiformes. Paleontol. J. 2011, 45, 445–458. [Google Scholar] [CrossRef]
- Mayr, G. Avian Evolution: The Fossil Record of Birds and its Paleobiological Significance; Wiley Blackwell: Chichester, UK, 2017; p. 312. [Google Scholar]
- Mayr, G. Paleogene Fossil Birds; Springer: Berlin, Germany, 2009; p. 262. [Google Scholar]
- Chiappe, L.M. Fossil Birds from the Miocene Pinturas Formation of Southern Argentina. J. Vertebr. Paleontol. 1991, 11, 21–22. [Google Scholar]
- Pavia, M.; Manegold, A.; Haarhoff, P. New Early Pliocene Owls from Langebaanweg, South Africa, With First Evidence of Athene South of the Sahara and a New Species of Tyto. Acta Palaeontol. Pol. 2014, 60, 815–828. [Google Scholar]
- Walker, C.A.; Dyke, G.J. New Records of Fossil Birds of Prey from the Miocene of Kenya. Hist. Biol. 2006, 18, 95–98. [Google Scholar] [CrossRef]
- Milne-Edwards, A. Recherches Sur La Faune Ancienne Des Iles Mascareignes. Ann. Sci. Nat. 1873, 2, 1–1931. [Google Scholar]
- Newton, E.; Gadow, H. On Additional Bones of the Dodo and Other Extinct Birds of Mauritius Obtained by Mr. Theodore Sauzier. Trans. Zool. Soc. Lond. 1893, 13, 281–302. [Google Scholar] [CrossRef]
- Rich, P.V. Preliminary Report on the Fossil Avian Remains from Late Tertiary Sediments at Langebaanweg (Cape Province), South Africa. S. Afr. J. Sci. 1980, 76, 166–170. [Google Scholar]
- Brodkorb, P. Fossil Owls from Early Man Sites of Olduvai Gorge, Tanzania. Ostrich 1984, 54, 17–27. [Google Scholar] [CrossRef]
- Cowles, G.S. A New Genus, Three New Species and Two New Records of Extinct Holocene Birds from Réunion Island, Indian Ocean. Geobios 1993, 27, 87–93. [Google Scholar] [CrossRef]
- Mourer-Chauvire, C.; Bour, R.; Moutou, F.; Ribes, S. Mascarenotus nov. gen. (Aves: Strigiformes), Extinct Endemic Genus from the Mascarene Islands, and M. Grucheti n. sp., Extinct Species De La Reunion. Comptes Rendus De L’académie Des Science De Paris (Iia) 1994, 318, 1699–1706. [Google Scholar]
- Mourer-Chauviré, C.; Geraads, D. The Upper Pliocene Avifauna of Ahl Al Oughlam, Morocco: Systematics and Biogeography. Rec. Aust. Mus. 2010, 62, 157–184. [Google Scholar] [CrossRef]
- Brunet, J. Oiseaux. Le gisement de Vertébrés Miocènes de Beni Mellal (Maroc). Notes Et Mémoires Du Service Géologique (Maroc) 1961, 155, 105–108. [Google Scholar]
- Brunet, J. Oiseaux Miocènes De Beni Mellal (Maroc); Un Complément À Leur Étude. Notes Et Mémoires Du Service Géologique Du Maroc 1971, 237, 109–111. [Google Scholar]
- Stewart, J.R. The Evidence for the Timing of Speciation of Modern Continental Birds and the Taxonomic Ambiguity of the Quaternary Fossil Record. In Proceedings of the 5th Symposium of the Society of Avian Paleontology and Evolution, Beijing, China, 1–4 June 2000; pp. 261–282. [Google Scholar]
- Stewart, J.R.; Beech, M. The Miocene Birds of Abu Dhabi (United Arab Emirates) With a Discussion of the Age of Modern Species and Genera. Hist. Biol. 2006, 18, 107–117. [Google Scholar] [CrossRef]
- Pavia, M.; Mourer-Chauviré, C. Redescription of Tyto sanctialbani Lydekker, 1893 (Aves, Strigiformes), from Its Type Locality of La Grive-Saint-alban (Middle Miocene, France). J. Vertebr. Paleontol. 2011, 31, 1093–1101. [Google Scholar] [CrossRef]
- Roberts, D.L.; Matthews, T.; Herries, A.I.R.; Boulter, C.; Scott, L.; Dondo, C.; Mtembi, P.; Browning, C.; Smith, R.M.H.; Haarhoff, P.; et al. Regional and Global Context of the Late Cenozoic Langebaanweg (Lbw) Palaeontological Site: West Coast of South Africa. Earth-Sci. Rev. 2011, 106, 191–214. [Google Scholar] [CrossRef]
- Baumel, J.; Witmer, L.; Baumel, J.J.; King, A.S.; Breazile, J.E.; Evans, H.E.; VandenBerge, J.C. (Eds.) Handbook of Avian Anatomy: Nomina Anatomica Avium; Nuttall Ornithological Club, 23: Cambridge, MA, USA, 1993; pp. 45–132. [Google Scholar]
- Von Den Driesch, A. A Guide to the Measurement of Animal Bones from Archaeological Sites. Peabody Mus. Bull. 1976, 1, 103–129. [Google Scholar]
- International Committee on Stratigraphy. International Chronostratigraphic Chart. 2017. Available online: https://www.iugs.org/ics (accessed on 13 February 2019).
- Linnaeus, C.V. Systema Naturae Per Regna Tria Naturae, Secundum Classes, Ordines, Genera, Species, Cum Characteribus, Differtiis, Synonymis, Locis. Ed. DecimaReformata 1758, 1–4, 824. [Google Scholar]
- Wagler, J.G. Natürliches System Der Amphibien Mit Vorangehender Classification Der Saügthiere Und Vögel: Ein Beitrag Zur Vergleichemden Zoologie. Cotta 1830, 1, 354. [Google Scholar]
- Mourer-Chauvire, C. Les Strigiformes (Aves) Des Phosphorites Du Quercy (France): Systématique, Biostratigraphie Et Paléobiogéographie. Documents Des Laboratoires De Géologie De Lyon 1987, 99, 89–127. [Google Scholar]
- Rasmussen, D.T.; Olson, S.L.; Simons, E.L. Fossil Birds from the Oligocene Jebel Qatrani Formation, Fayum Province, Egypt. Smithson. Contrib. Paleobiol. 1987, 61, 1–20. [Google Scholar] [CrossRef]
- Bown, T.M.; Kraus, M.J. Geology and Paleoenvironment of the Oligocene Jebel Qatrani Formation and Adjacent Rocks, Fayum Depression, Egypt. U. S. Geol. Surv. Prof. Pap. 1987, 1452, 1–60. [Google Scholar]
- Seiffert, E.R. Revised Age Estimates for the Later Paleogene Mammal Faunas of Egypt and Oman. Proc. Natl. Acad. Sci. USA 2006, 103, 5000–5005. [Google Scholar] [CrossRef] [PubMed]
- Said, E.R. Cenozoic. In The Geology of Egypt; Said, R., Ed.; AA Balkema: Brookfield, WA, USA, 1990; pp. 451–486. [Google Scholar]
- Chapman, W.L., Jr. Appearance of Ossification Centers and Epiphysial Closures as Determined by Radiographic Techniques. J. Am. Vet. Med Assoc. 1965, 147, 138. [Google Scholar]
- Watanabe, J.; Matsuoka, H. Ontogenetic Change of Morphology and Surface Texture of Long Bones in the Gray [sic] Heron (Ardea cinerea, Ardeidae). In Paleornithological Research; Goelich, U., Kroh, A., Eds.; Verlag Naturhistorisches Museum Wien: Wein, Austria, 2013; pp. 279–306. [Google Scholar]
- Fowler, D.W.; Freeedman, E.A.; Scannella, J.B. Predatory Functional Morphologyin Raptors: Interdigital Variation in Talon Size is related to Prey Restraint and Immobilisation Technique. PLoS ONE 2009, 4, e7999. [Google Scholar] [CrossRef]
- Mayr, G.; Clarke, J.A. The Deep Divergences of Neornithine Birds: A Phylogenetic Analysis of Morphological Characters. Cladistics 2003, 19, 527–553. [Google Scholar] [CrossRef]
- Stidham, T.A.; Smith, N.A. An Ameghinornithid-Like Bird (Aves, Cariame, ?Ameghinornithidae) from the Early Oligocene of Egypt. Palaeontol. Electron. 2015, 18, 1–8. [Google Scholar]
- Mayr, G. A Small Loon and a Large Species of Owl from the Rupelian of Belgium (Aves, Gaviiformes, Strigiformes). Palaontol. Z. 2009, 83, 247–254. [Google Scholar] [CrossRef]
- Rich, P.V.; Bohaska, D.J. The world’s oldest owl: A new strigiform from the Paleocene of southwestern Colorado. In Smithsonian Contributions to Paleobiology; Olson, S.L., Ed.; Smithsonian Institution: Washington DC, USA, 1976; Volume 27, pp. 87–93. [Google Scholar]
- Pavia, M. A new large barn owl (Aves, Strigiformes, Tytonidae) from the Middle Pleistocene of Sicily, Italy, and its taphonomical significance. Geobios 2004, 37, 631–641. [Google Scholar] [CrossRef]
- Miller, E.R.; Rasmussen, D.T.; Simons, E.L. Fossil Storks (Ciconiidae) From the Late Eocene and Early Miocene of Egypt. Ostrich 1997, 68, 23–26. [Google Scholar] [CrossRef]
- Smith, N.A. Avian Fossils from the Early Miocene Moghra Formation of Egypt. Ostrich 2013, 84, 181–189. [Google Scholar] [CrossRef]
- Dyke, G.J.; Walker, C.A. New Records of Fossil ‘Waterbirds’ From the Miocene of Kenya. Am. Mus. Novit. 2008, 3610, 1–10. [Google Scholar] [CrossRef][Green Version]
- Olson, S.L.; Rasmussen, D.T. Paleoenvironment of the Earliest Hominids: New Evidence from the Oligocene Avifauna of Egypt. Science 1986, 233, 1202–1204. [Google Scholar] [CrossRef]
- Dunning, J.B.J. CRC Handbook of Avian Body Masses, 2nd ed.; CRC press: Boca Raton, FL, USA, 2008; p. 655. [Google Scholar]
- Louchart, A. Integrating the Fossil Record in the Study of Insular Body Size Evolution: Example of Owls (Aves, Strigiformes). In Proceedings of the International Symposium “Insular Vertebrate Evolution: The Palaeontological Approach”, Mallorca, Spain, 16–19 September 2005; Monografies de la Societat d’Història de las Balears. Fundación Dialnet: Logroño, Spain, 2005; Volume 12, pp. 155–174. [Google Scholar]
- Gunnell, G.F.; Miller, E.R. Origin of Anthropoidea: Dental Evidence and Recognition of Early Anthropoids in the Fossil Record, With Comments on the Asian Anthropoid Radiation. Am. J. Phys. Anthropol. 2001, 114, 177–191. [Google Scholar] [CrossRef]
- Perry, J.M.G.; Cooke, S.B.; Runestead-Connour, J.A.; Burgess, M.L.; Ruff, C.B. Articular Scaling and Body Mass Estimation in Platyrrhines and Catyrrhines: Modern Variation and Application to Fossil Anthropoids. J. Hum. Evol. 2018, 115, 20–35. [Google Scholar] [CrossRef] [PubMed]
- Sanders, W.J.; Trapani, J.; Mitani, J.C. Taphonomic Aspects of Crowned Hawk-Eagle Predation on Monkeys. J. Hum. Evol. 2003, 44, 87–105. [Google Scholar] [CrossRef]
- Terry, R.C. Quantifying the Digestive Fingerprints of Predators on the Bones of Their Prey Using Scanning Electron Microscopy. Palaios 2018, 33, 487–497. [Google Scholar] [CrossRef]
- Berger, L.R.; Clarke, R.J. Eagle Involvement in Accumulation of the Taung Child Fauna. J. Hum. Evol. 1995, 29, 275–299. [Google Scholar] [CrossRef]
- Jarvis, E.D.; Mirarab, S.; Aberer, A.J.; Li, B.; Houde, P.; Li, C.; Ho, S.Y.W.; Faircloth, B.C.; Nabholz, B.; Howard, J.T. Whole-Genome Analyses Resolve Early Branches in the Tree of Life of Modern Birds. Science 2014, 346, 1320–1331. [Google Scholar] [CrossRef]
- Prum, R.O.; Berv, J.S.; Dornburg, A.; Field, D.J.; Townsend, J.P.; Lemmon, E.M.; Lemmon, A.R. A Comprehensive Phylogeny of Birds (Aves) Using Targeted Next-Generation DNA Sequencing. Nature 2015, 526, 569–573. [Google Scholar] [CrossRef]
- Ksepka, D.T.; Clarke, J.A. Affinities of Palaeospiza Bella and the Phylogeny and Biogeography of Mousebirds (Coliiformes). Auk 2009, 126, 245–259. [Google Scholar] [CrossRef]
- Smith, K.T.; Bhullar, B.S.; Holroyd, P.A. Earliest African Record of the Varanus Stem-Clade (Squamata: Varanidae) From the Early Oligocene of Egypt. J. Vertebr. Paleontol. 2008, 28, 909–913. [Google Scholar] [CrossRef]
- Holroyd, P.A.; Parham, J.F. The Antiquity of African Tortoises. J. Vertebr. Paleontol. 2003, 23, 688–690. [Google Scholar] [CrossRef]
- Grohé, C.; Morlo, M.; Chaimanee, Y.; Blondel, C.; Coster, P.; Valentin, X.; Salem, M.; Bilal, A.A.; Jaeger, J.J.; Brunet, M. New Apterodontinae (Hyaenodontida) From the Eocene Locality of Dur At-Talah (Libya): Systematic, Paleoecological and Phylogenetical Implications. PLoS ONE 2012, 7, e49054. [Google Scholar] [CrossRef]
- Holroyd, P.A.; Maas, M.C. Anthropoid Origins; Fleagle, J.G., Kay, R.F., Eds.; Plenum Press: New York, NY, USA, 1994; pp. 297–334. [Google Scholar]
- De Pietri, V.L.; Mourer-Chauvire, C.; Menkveld-Gfeller, U.; Meyer, C.A.; Costeur, L. An Assessment of the Cenozoic Avifauna of Switzerland, With a Description of Two Fossil Owls (Aves, Strigiformes). Swiss J. Geosci. 2013, 106, 187–197. [Google Scholar] [CrossRef]
- Mayr, G.; De Pietri, V.L.; Scofield, R.P.; Smith, T. A Fossil Heron from the Early Oligocene of Belgium: The Earliest Temporally Well-Constrained Record of the Ardeidae. IBIS 2017, 161, 79–90. [Google Scholar] [CrossRef]
- Stidham, T.A.; Wang, Y.Q. An Ameghinornithid-Like Bird (Aves: Cariamae: Ameghinornithidae?) From the Middle Eocene of Nei Mongol, China. Vertebr. Palasiat. 2017, 55, 218–226. [Google Scholar]
- Zelenkov, N.V.; Panteleyev, A.V. A Small Stem-Galliform Bird (Aves: Parortygidae) From the Eocene of Uzbekistan. Comptes Rendus Paleovol 2019, 18, 517–523. [Google Scholar] [CrossRef]
- Liu, Z.; Pagani, M.; Zinniker, D.; Deconto, R.; Huber, M.; Brinkhuis, H.; Shah, S.R.; Leckie, R.M.; Pearson, A. Global Cooling During the Eocene-Oligocene Climate Transition. Science 2009, 323, 1187. [Google Scholar] [CrossRef]
- Vantuinen, M.; Stidham, T.A.; Hadly, E.A. Tempo and Mode of Modern Bird Evolution Observed with Large-Scale Taxonomic Sampling. Hist. Biol. 2006, 18, 205–221. [Google Scholar]
- Brown, J.W.; Payne, R.B.; Mindell, D.P. Nuclear DNA Does Not Reconcile ‘Rocks’ and ‘Clocks’ in Neoaves: A Comment on Ericson Et Al. Biol. Lett. 2007, 3, 257–260. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Palmqvist, P.; Vizcaíno, S.F. Ecological and Reproductive Constraints of Body Size in the Gigantic Argentavis Magnificens (Aves, Theratornithidae [Sic]) From the Miocene of Argentina. Ameghiniana 2003, 40, 379–385. [Google Scholar]
- Fox-Dobbs, K.; Stidham, T.A.; Bowen, G.J.; Emslie, S.D.; Koch, P.L. Dietary Controls on Extinction Versus Survival Among Avian Megafauna in the Late Pleistocene. Geology 2006, 34, 685–688. [Google Scholar] [CrossRef]
- Facenna, C.; Glisovic, P.; Forte, A.; Becker, T.W.; Garzanti, E.; Sembroni, A.; Gvirtzman, Z. Role of Dynamic Topography in Sustaining the Nile River Over 30 Million Years. Nat. Geosci. 2019, 12, 1012–1017. [Google Scholar] [CrossRef]
| Species | n | tl | tsw | tdw |
|---|---|---|---|---|
| Bubo bubo | 3 | 138.0 | 5.3 | 17.5 |
| Bubo scandiaca | 5 | 117.6 | 5.1 | 15.8 |
| Bubo virginianus | 10 | 124.3 | 5.2 | 15.6 |
| Strix nebulosa | 5 | 118.7 | 4.9 | 14.5 |
| Strix occidentalis | 4 | 94.9 | 3.7 | 10.7 |
| Strix otus | 4 | 77.2 | 2.8 | 8.5 |
| Strix varia | 10 | 106.0 | 3.8 | 12.0 |
| Aegolius acadicus | 4 | 46.9 | 1.8 | 5.5 |
| Asio flammeus | 4 | 86.2 | 3.0 | 8.9 |
| Micrathene whitneyi | 3 | 38.3 | 1.8 | 4.0 |
| Otus asio | 9 | 54.1 | 2.2 | 6.5 |
| Tyto alba | 6 | 106.4 | 4.2 | 10.6 |
| †Ornimegalonyx oteroi | 1 | 250.0 | 13.0 | - |
| †Tyto gigantea | 1 | 195.0 | 9.7 | - |
| †Tyto noeli | 1 | 147.0 | 6.4 | - |
| †Necrobyas arvernensis | 1 | 77.9 | 4.3 | 10.0 |
| †Grallistrix auceps | 1 | 110.8 | 4.9 | 10.2 |
| †Grallistrix orion | 1 | 101.9 | 4.2 | 9.6 |
| †Grallistrix erdmani | 1 | 96.0 | 4.6 | 9.2 |
| †Eostrix mimica | 1 | - | 6.0 | 11.4 |
| †cf Selenornithinae (Jebel Owl) | 1 | - | 9.7 | 13.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, N.A.; Stidham, T.A.; Mitchell, J.S. The First Fossil Owl (Aves, Strigiformes) From the Paleogene of Africa. Diversity 2020, 12, 163. https://doi.org/10.3390/d12040163
Smith NA, Stidham TA, Mitchell JS. The First Fossil Owl (Aves, Strigiformes) From the Paleogene of Africa. Diversity. 2020; 12(4):163. https://doi.org/10.3390/d12040163
Chicago/Turabian StyleSmith, N. Adam, Thomas A. Stidham, and Jonathan S. Mitchell. 2020. "The First Fossil Owl (Aves, Strigiformes) From the Paleogene of Africa" Diversity 12, no. 4: 163. https://doi.org/10.3390/d12040163
APA StyleSmith, N. A., Stidham, T. A., & Mitchell, J. S. (2020). The First Fossil Owl (Aves, Strigiformes) From the Paleogene of Africa. Diversity, 12(4), 163. https://doi.org/10.3390/d12040163





