Modern Benthic Foraminiferal Diversity: An Initial Insight into the Total Foraminiferal Diversity along the Kuwait Coastal Water
Abstract
1. Introduction
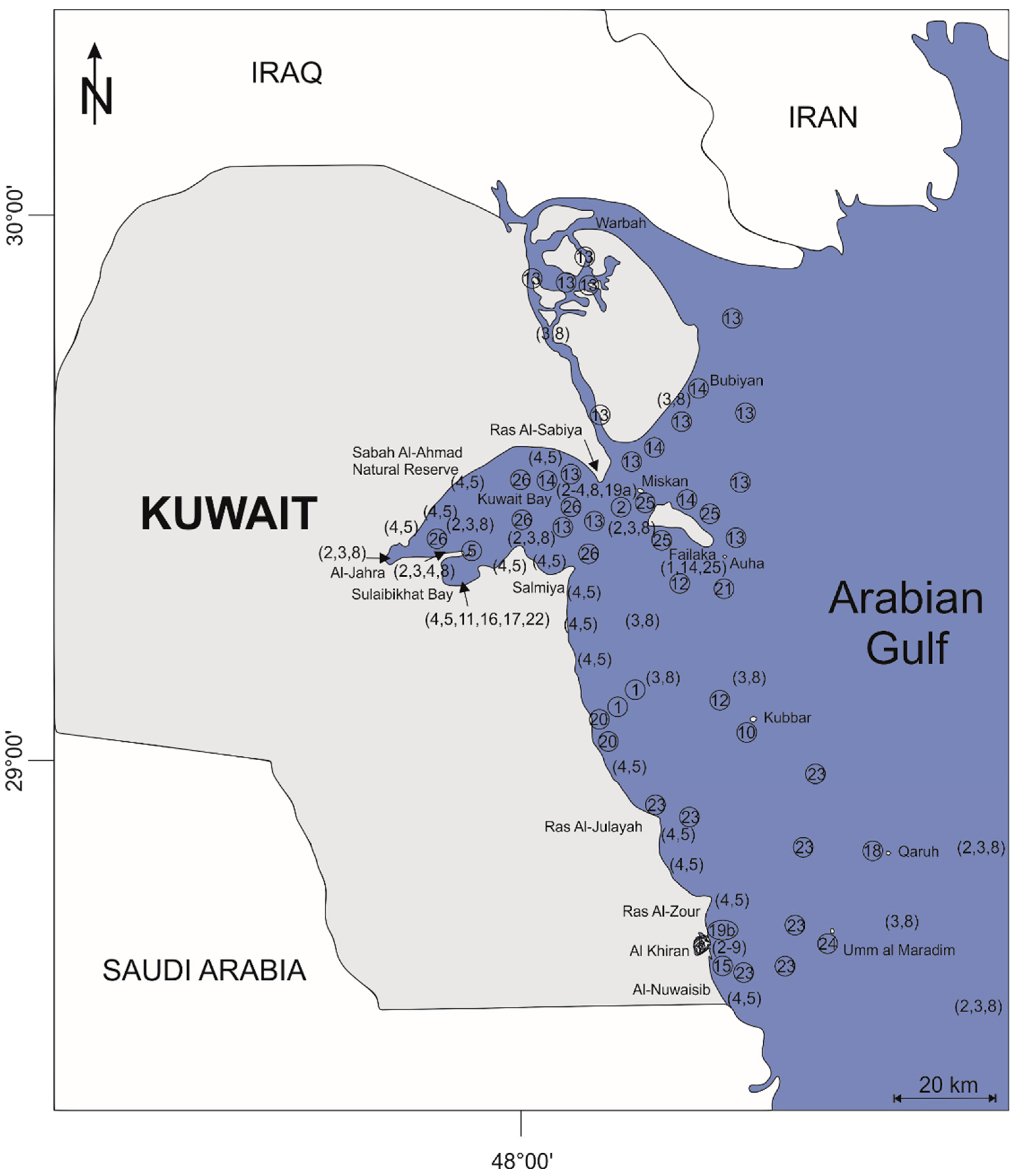
2. Geography, Hydrology and Study Area
3. Previous Benthic Foraminiferal Studies within the Kuwaiti Territorial Waters
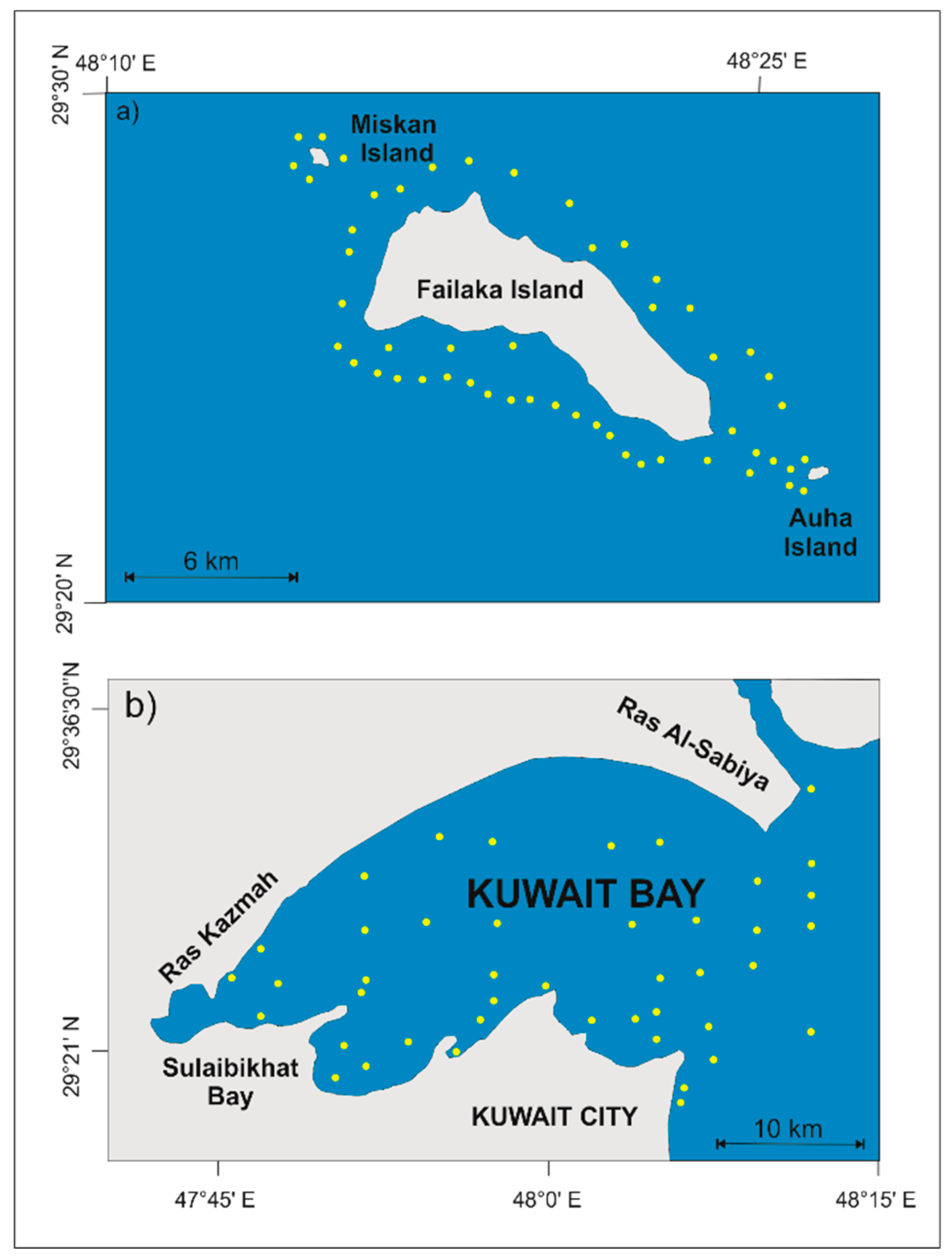
4. Materials and Methods
5. Results
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, N.Y.; Munawar, M.; Price, A.R.G. The Gulf Ecosystem: Health and Sustainability, 1st ed.; Backhuys Publishers: Leiden, The Netherlands, 2002; pp. 1–509. [Google Scholar]
- Riegl, B.M.; Purkis, S.J. Coral Reefs of the Gulf: Adaptation to Climatic Extremes in the World’s Hottest Sea, 1st ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 1–379. [Google Scholar]
- Sheppard, C.; Al-Husiani, M.; Al-Jamali, F.; Al-Yamani, F.; Baldwin, R.; Bishop, J.; Benzoni, F.; Dutrieux, E.; Dulvy, N.K.; Durvasula, S.R.; et al. The Gulf: A young sea in decline. Mar. Pollut. Bull. 2010, 60, 13–38. [Google Scholar] [CrossRef] [PubMed]
- Noori, R.; Tian, F.; Berndtsson, R.; Abbasi, M.R.; Naseh, M.V.; Modabberi, A.; Soltani, A.; Kløve, B. Recent and future trends in sea surface temperature across the Persian Gulf and Gulf of Oman. PLoS ONE 2019, 14, e0212790. [Google Scholar] [CrossRef]
- Khan, N.Y. Physical and human geography. In The Gulf Ecosystem: Health and Sustainability, 1st ed.; Khan, N.Y., Munawar, M., Price, A.R.G., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2002; pp. 3–21. [Google Scholar]
- Al-Bakri, D. Environmental Assessment of the Intertidal Zone of Kuwait; Kuwait Institute for Scientific Research Report; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 1984; pp. 1–429. [Google Scholar]
- Al-Yamani, F.Y.; Bishop, J.; Ramadhan, E.; Al-Husaini, M.; Al-Ghadban, A.N. Oceanographic Atlas of Kuwait’s Waters, 1st ed.; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2004; pp. 1–203. [Google Scholar]
- Bishop, J.M. History and current checklist of Kuwait’s ichthyofauna. J. Arid Environ. 2003, 54, 237–256. [Google Scholar] [CrossRef]
- Al-Yamani, F.Z.; Skryabin, V.A. Identification Guide for Protozoans from Kuwait’s Waters. Coastal Planktonic Ciliates: I. Tintinnids, 1st ed.; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2006; pp. 1–109. [Google Scholar]
- Al-Yamani, F.Z.; Saburova, M.A. Illustrate Guide on the Flagellates of Kuwait’s Intertidal Soft Sediments, 1st ed.; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2010; pp. 1–197. [Google Scholar]
- Al-Yamani, F.Z.; Prusova, I. Common Copepods of the Northwestern Arabian Gulf: Identification Guide, 1st ed.; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2003; pp. 1–162. [Google Scholar]
- Al-Kandari, M.; Al-Yamani, F.Y.; Al-Rifaie, K. Marine Phytoplankton Atlas of Kuwait’s Waters, 1st ed.; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2009; pp. 1–354. [Google Scholar]
- Murray, J.W. Ecology and Applications of Benthic Foraminifera, 2nd ed.; Cambridge University Press: Cambridge, UK, 2006; pp. 1–426. [Google Scholar]
- Lee, J.L.; Anderson, O.R. Symbiosis in Foraminifera. In Biology of Foraminifera; Lee, J.L., Anderson, O.R., Eds.; Elsevier Science Publishing Co. Inc.: San Diego, CA, USA, 1991; pp. 157–220. [Google Scholar]
- Moodley, L.; Middelburg, J.J.; Boschker, H.T.S.; Duineveld, G.; Pel, R.; Herman, P.M.J.; Heip, C.H.R. Bacteria and Foraminifera: Key players in a short-term deep-sea benthic response to phytodetritus. Mar. Ecol. Prog. Ser. 2002, 236, 23–29. [Google Scholar] [CrossRef]
- Jorissen, F.J. Benthic foraminifera from the Adriatic Sea; principles of phenotypic variation. Utrecht Micropaleontol. Bull. 1988, 37, 1–174. [Google Scholar]
- Sen Gupta, B.K.; Smith, L.E. Modern Benthic Foraminifera of the Gulf of Mexico: Census report. J. Foraminifer. Res. 2010, 40, 247–256. [Google Scholar] [CrossRef]
- Milker, Y.; Schmiedl, G. A taxonomic guide to modern benthic shelf foraminifera of the western Mediterranean Sea. Palaeontol. Electron. 2012, 15, 1–134. [Google Scholar] [CrossRef]
- Dorst, D.; Schönfeld, J. Diversity of benthic Foraminifera on the shelf and slope of the NE Atlantic: Analysis of datasets. J. Foraminifer. Res. 2013, 43, 238–254. [Google Scholar] [CrossRef]
- Martins, M.V.; Frontalini, F.; Laut, L.L.; Silva, F.S.; Moreno, J.; Sousa, S.; Zaaboub, N.; El Bour, M.; Rocha, F. Foraminiferal Biotopes and their Distribution Control in Ria de Aveiro (Portugal): A multiproxy approach. Environ. Monit. Assess. 2014, 186, 8875–8897. [Google Scholar] [CrossRef]
- Frontalini, F.; Kaminski, M.A.; Mikellidou, I.; du Châtelet, E.A. Checklist of benthic foraminifera (class Foraminifera: D’Orbigny 1826; phylum Granuloreticulosa) from Saros Bay, northern Aegean Sea: A biodiversity hotspot. Mar. Biodivers. 2015, 45, 549–567. [Google Scholar] [CrossRef]
- Kim, S.; Frontalini, F.; Martins, M.V.; Lee, W. Modern benthic foraminiferal diversity in Jeju Island and first insights into the total diversity of Korea. Mar. Biodivers. 2016, 46, 337–354. [Google Scholar] [CrossRef]
- Cherif, O.H.; Al-Ghadban, A.N.; Al-Rifaiy, I.A. Distribution of foraminifera in the Arabian Gulf. Micropaleontology 1997, 43, 253–280. [Google Scholar] [CrossRef]
- Arslan, M.; Kaminski, M.A.; Tawabini, B.S.; Ilyas, M.; Babalola, L.O.; Frontalini, F. Seasonal variations, environmental parameters and standing crop assessment of benthic foraminifera in eastern Bahrain. Arabian Gulf. Geol. Q. 2016, 60, 24–35. [Google Scholar] [CrossRef]
- Arslan, M.; Kaminski, M.A.; Tawabini, B.S.; Ilyas, M.; Frontalini, F. Benthic foraminifera in sandy (siliciclastic) coastal sediments of the Arabian Gulf (Saudi Arabia): A technical report. Arab. J. Geosci. 2016, 9, 7. [Google Scholar] [CrossRef]
- Amao, A.O.; Kaminski, M.A.; Qurban, M.A.; Thadikal, J.; Frontalini, F. A baseline investigation of benthic foraminifera in relation to marine sediments parameters in western parts of the Arabian Gulf. Mar. Pollut. Bull. 2019, 146, 751–766. [Google Scholar] [CrossRef] [PubMed]
- Amao, A.O.; Kaminski, M.A.; Asgharian Rostami, M.; Gharaie, M.H.M.; Lak, R.; Frontalini, F. Distribution of benthic foraminifera along the Iranian Coast. Mar. Biodivers. 2019, 49, 933–946. [Google Scholar] [CrossRef]
- Al-Enezi, E.; Al-Ghadban, A.N.; Al-Refai, I.; Pieretti, N.; Frontalini, F. Living benthic foraminifera around the unique Umm al Maradim Island (Kuwait). Kuwait J. Sci. 2019, 46, 59–66. [Google Scholar]
- Al-Zamel, A.; Al-Sarawi, M.; Khader, S.; Al-Rifaiy, I. Benthic foraminifera from polluted marine environment of Sulaibikhat Bay (Kuwait). Environ. Monit. Assess. 2009, 149, 395–409. [Google Scholar] [CrossRef]
- Al-Enezi, E.; Frontalini, F. Benthic foraminifera and environmental quality: The case study of Sulaibikhat Bay (Kuwait). Arab. J. Geosci. 2015, 8, 8527–8538. [Google Scholar] [CrossRef]
- Neelamani, S.; Al-Salem, K.; Rakha, K. Extreme waves for Kuwaiti territorial waters. Ocean Eng. 2007, 34, 1496–1504. [Google Scholar] [CrossRef]
- Al-Bakri, D.; El-Sayed, M.I. Mineralogy and provenance of the elastic deposits of the modern intertidal environment of the northern Arab. Gulf. Mar. Geol. 1991, 97, 121–135. [Google Scholar] [CrossRef]
- Kassler, P. The structural and geomorphic evolution of the Persian Gulf. In The Persian Gulf. Holocene Carbonate Sedimentation and Diagenesis in a Shallow Epicontinental Sea, 1st ed.; Purser, B.H., Ed.; Springer: Berlin, Germany, 1973; pp. 11–32. [Google Scholar]
- Al-Sarawi, M.A.; Gundlach, E.; Baca, B.J. Coastal geomorphology and resources in terms of sensitivity to oil spill in Kuwait. J. Univ. Kuwait (Sci.) 1988, 15, 141–184. [Google Scholar]
- Riegl, B.; Poiriez, A.; Janson, X.; Bergman, K.L. The gulf: Facies belts, physical, chemical, and biological parameters of sedimentation on a carbonate ramp. In Carbonate Depositional Systems: Assessing Dimensions and Controlling Parameters, 1st ed.; Westphal, H., Riegl, B., Eberli, G.P., Eds.; Springer Science & Business Media: New York, NY, USA, 2010; pp. 145–213. [Google Scholar]
- Khalaf, F.; Al-Bakri, D.; Al-Ghadban, A. Sedimentological characteristics of the surficial sediments of the Kuwaiti marine environment, northern Arabian Gulf. Sedimentology 1984, 31, 531–545. [Google Scholar] [CrossRef]
- Reynolds, R.M. Physical oceanography of the Gulf, Strait of Hormuz, and the Gulf of Oman—Results from the Mt Mitchell expedition. Mar. Pollut. Bull. 1993, 27, 35–59. [Google Scholar] [CrossRef]
- Jones, D.A.; Price, A.R.G.; Al-Yamani, F.Y.; Al-Zaidan, A. Coastal and marine ecology. In The gulf Ecosystem: Health and Sustainability, 1st ed.; Khan, N.Y., Munawar, M., Price, A.R.G., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2002; pp. 65–103. [Google Scholar]
- Peiris, N.I. Recent Foraminifera and Ostracoda from the Persian Gulf. Master’s Thesis, University College of Wales, Aberystwyth, UK, 1969. [Google Scholar]
- Anber, S.A. Foraminifera from the Offshore Area of Kuwait (Northern Arabian Gulf). Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 1974. [Google Scholar]
- Shublaq, W. Foraminiferida from Bottom Sediments in the Northwestern Part of the Arabian Gulf (Offshore Kuwait and Saudi Arabia—Kuwait Portioned Zone). Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 1977. [Google Scholar]
- Al-Abdul Razzaq, S.; Khalaf, F.; Al-Bakri, D.; Shublaq, W.; Al-Sheikh, Z. Marine Sedimentology and Benthic Ecology of Kuwait Environment; Report No. KISR 694; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 1982. [Google Scholar]
- Al-Abdul Razzaq, S.; Shublaq, W.; Al-Sheikh, Z. The marine benthic microfauna of the tidal flats of Kuwait. J. Kuwait Univ. 1983, 10, 101–109. [Google Scholar]
- Al-Abdul-Razzaq, S.; Bhalla, S. On the genus Cribrospirlina Haman, 1972 (Foraminifera). J. Micropaleontol. 1987, 6, 63–64. [Google Scholar] [CrossRef]
- Al-Abdul-Razzaq, S.; Bhalla, S. Microfauna from Al-Khiran area, Southern Kuwait. Rev. Paléobiol. 1987, 6, 139–142. [Google Scholar]
- Shublaq, W. Microfauna of Kuwait’s Marine Environment (Foraminifera and Ostracoda); Phase I. Final Report, Restricted, EES-108; Reports No. KISR 2812; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 1988. [Google Scholar]
- Al-Zamel, A.; Cherif, O.; Al-Rifaiy, I. Tidal creeks foraminiferal distribution in Khor Al-Mufateh and Khor Al-Malaha, Khiran area, southeast Kuwait. Rev. Micropalaeontol. 1996, 39, 3–26. [Google Scholar]
- Al-Shuaibi, A.A.; Cherif, O.H.; Al-Sarawi, M.A. Recent Subtidal Foraminiferal Distribution and Water Quality of Kubbar Island, Kuwait. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 1997. [Google Scholar]
- Khader, S. Coastal Geomorphology and Environmental Assessment of Sulaibikhat Bay. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 1997, unpublished. [Google Scholar]
- Al-Zamel, Z.; Cherif, O. Subtidal foraminiferal assemblages of the western part of the Shatt Al-Arab Delta, Kuwait, Arabian Gulf. J. Foraminifer. Res. 1998, 28, 327–344. [Google Scholar] [CrossRef]
- Al-Ghadban, A.N. The Potential Impact of Draining the Southern Iraqi Marshes on the Sediment Budget and Associated Pollutants in the Northern Arabian Gulf; Kuwait Institute for Scientific Research: Al-Ahmadi, Kuwait, 2000; pp. 1–230. [Google Scholar]
- Al-Enezi, E.H. Environmental Factors Controlling the Distribution of Foraminifera in the Umm Al-Maradem Island and Khor Iskandar Southern Part of Kuwait. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 2002. [Google Scholar]
- Al-Kandari, A.J.H. Trace Metals Concentration and Their Effects on Foraminifera at Coastal Area Adjacent to Kuwait University Campus at Shuwaikh. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 2008. [Google Scholar]
- Al-Ammar, M. Distribution of Recent Benthic Foraminifera around the Coral Reefs of Quran Island, Southern Kuwait: Variability and Biodiversity. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 2011. [Google Scholar]
- Al Theyabi, N.T. Effect of Thermal Pollution on Microfauna “Foraminifera” a Case Study of Ras Al-Zour and Ras Al-Subiya Power Stations, State of Kuwait. Master’s Thesis, Kuwait University, Kuwait City, Kuwait, 2012. [Google Scholar]
- Uddin, S.; Al-Ghadban, A.N.; Pokavanich, T.; Al Banna, K.; Al-Enezi, E.; Karam, Q.; Al-Shamroukh, D.; Al-Khabbaz, A.; Al- Yaegoub, A. Marine Survey and Dispersion Modeling for Environmental Aspect at MAA and MAB Refineries; Technical Report No. KISR 11496; Kuwait Institute for Scientific Research: Safat, Kuwait, 2013. [Google Scholar]
- Nabavi, S.M.B.; Moosapanah, S.G.R.; Ghatrami, E.R.Z.; Ashrafi, M.G.; Nabavi, S.N. Distribution, Diversity and Abundance of Benthic Foraminifera of the Northwestern Persian Gulf. J. Persian Gulf (Mar. Sci.) 2014, 5, 15–26. [Google Scholar]
- Parker, J.H.; Gischler, E. Modern and relict foraminiferal biofacies from a carbonate ramp, offshore Kuwait, northwest Persian Gulf. Facies 2015, 61, 1–22. [Google Scholar] [CrossRef]
- Khader, S. Benthic Foraminifera Response to Environmental Stress around Three Northern Kuwaiti Islands (Failaka, Auha and Miskan Islands), NW Kuwait, Arabian Gulf. Ph.D. Thesis, Alexandria University, Alexandria, Egypt, 2020, unpublished. [Google Scholar]
- Loeblich, A.R.; Tappan, H. Foraminiferal Genera and Their Classification; Van Nostrand Reinhold: New York, NY, USA, 1987; pp. 1–970. [Google Scholar]
- Cimerman, F.; Langer, M.R. Mediterranean Foraminifera; Slovenska Akademija Znanosti in Umetnosti, Akademia Scientiarium et Artium Slovenica: Ljubljana, Slovenia, 1991; Volume 30, pp. 1–119. [Google Scholar]
- Hottinger, L.; Halicz, E.; Reiss, Z. Recent Foraminifera from the Gulf of Aqaba, Red Sea, 1st ed.; Slovenska Akademija Znanosti Umetnosti: Ljubljana, Slovenia, 1993; pp. 1–179. [Google Scholar]
- Sgarella, F.; Moncharmont Zei, M. Benthic foraminifera of the Gulf of Naples (Italy): Systematics and autoecology. Bull. Soc. Paleontol. Ital. 1993, 32, 145–264. [Google Scholar]
- Jones, R.W. The Challenger Foraminifera, 1st ed.; Oxford University Press: Oxford, UK, 1994; pp. 1–149. [Google Scholar]
- Debenay, J.P. A Guide to 1,000 Foraminifera from Southwestern Pacific, New Caledonia, 1st ed.; IRD, Ed.; Publications Scientifiques du Museum; Museum National d’Histoire Naturelle: Paris, France, 2013; pp. 1–384. [Google Scholar]
- Amao, A.O. Benthic Foraminiferal Taxonomy, Distribution and Ecology in the Arabian Gulf. Ph.D. Thesis, King Fahd University of Petroleum and Minerals, Dhahran, Saudi Arabia, 2016. [Google Scholar]
- World Register of Marine Species. Available online: http://www.marinespecies.org/aphia.php?p=taxdetails&id=1410 (accessed on 14 February 2020).
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Whittaker, R.H. Vegetation of the Siskiyou mountains, Oregon and California. Ecol. Monogr. 1960, 30, 279–338. [Google Scholar] [CrossRef]
- Amao, A.O.; Kaminski, M.A. A new foraminiferal species Pseudotriloculina hottingeri n. sp. from the Arabian Gulf. J. Foraminifer. Res. 2017, 47, 366–371. [Google Scholar] [CrossRef]
- Amao, A.O.; Kaminski, M.A. Pseudonubeculina arabica n. gen. n. sp., a new Holocene benthic foraminifer from the Arabian Gulf. Micropalaeontology 2016, 62, 81–86. [Google Scholar]
- Amao, A.O.; Kaminski, M.A. Falsonubeculina, a replacement name for Pseudonubeculina Amao and Kaminski 2016 (preoccupied). Micropaleontology 2019, 65, 544. [Google Scholar]
- Lecroq, B.; Lejzerowicz, F.; Bachar, D.; Christen, R.; Esling, P.; Baerlocher, L.; Østerås, M.; Farinelli, L.; Pawlowski, J. Ultra-deep sequencing of foraminiferal microbarcodes unveils hidden richness of early monothalamous lineages in deep-sea sediments. Proc. Natl. Acad. Sci. USA 2011, 108, 13177–13182. [Google Scholar] [CrossRef]
- Al-Enezi, E.; Al-Hazeem, S. Benthic Foraminifera as Proxies for the Environmental Quality Assessment of the Kuwait Bay: Morphological and Environmental DNA Approaches; Technical report KISR No. EM084C; Kuwait Institute of Scientific Research: Safat, Kuwait, 2019. [Google Scholar]
- Loeblich, A.R.; Tappan, H. Foraminifera of the Sahul Shelf and Timor Sea. Cushm. Found. Foram. Res. Spec. Publ. 1994, 31, 1–661. [Google Scholar]
- Langer, M.R.; Weinmann, A.E.; Lötters, S.; Bernhard, J.M.; Rödder, D. Climate-driven range extension of Amphistegina (Protista, Foraminiferida): Models of current and predicted future ranges. PLoS ONE 2013, 8, e54443. [Google Scholar] [CrossRef]
- Guastella, R.; Marchini, A.; Caruso, A.; Cosentino, C.; Evans, J. “Hidden invaders” conquer the Sicily Channel and knock on the door of the Western Mediterranean Sea. Estuar. Coast. Shelf Sci. 2019, 225, 106234. [Google Scholar] [CrossRef]
- Gischler, E.; Lomando, A.J. Offshore sedimentary facies of a modern carbonate ramp, Kuwait, northwestern Arabian-Persian Gulf. Facies 2005, 50, 443–462. [Google Scholar] [CrossRef]
- Triantaphyllou, M.V.; Dimiza, M.D.; Koukousioura, O.; Hallock, P. Observations on the life cycle of the symbiont-bearing foraminifer Amphistegina lobifera Larsen, an invasive species in coastal ecosystems of the aegean sea (Greece, E. Mediterranean). J. Foraminifer. Res. 2012, 42, 143–150. [Google Scholar] [CrossRef]
- Langer, M.R. Assessing the contribution of foraminiferan protists to global ocean carbonate production. J. Eukaryot. Microbiol. 2008, 55, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Förderer, M.; Rödder, D.; Langer, M.R. Patterns of species richness and the center of diversity in modern Indo-Pacific larger foraminifera. Sci. Rep. 2018, 8, 8189. [Google Scholar] [CrossRef] [PubMed]
| Order | Superfamily | Family | Number of Genera | Number of Species |
|---|---|---|---|---|
| Lituolida | Lituoloidea | Lituolidae | 3 | 6 |
| Verneuilinoidea | Prolixoplectidae | 1 | 2 | |
| Verneuilinidae | 1 | 1 | ||
| Trochamminoidea | Trochamminidae | 3 | 3 | |
| Textulariida | Textularioidea | Textulariidae | 4 | 25 |
| Olgiidae | 1 | 1 | ||
| Eggerelloidea | Valvulinidae | 1 | 1 | |
| Hormosinoidea | Reophacidae | 1 | 1 | |
| Miliolida | Cornuspiroidea | Cornuspiridae | 1 | 1 |
| Milioloidea | Cribrolinoididae | 1 | 10 | |
| Hauerinidae | 35 | 165 | ||
| Spiroloculinidae | 1 | 18 | ||
| Miliolacea | Riveroinidae | 1 | 1 | |
| Nubecularioidea | Fischerinidae | 2 | 2 | |
| Ophthalmidiidae | 3 | 7 | ||
| Nubeculariidae | 3 | 3 | ||
| Soritoidea | Peneroplidae | 4 | 7 | |
| Soritidae | 1 | 2 | ||
| Miliamminidae | 1 | 2 | ||
| Squamulinoidea | Squamulinidae | 1 | 1 | |
| Spirillinida | Spirillinidae | 1 | 2 | |
| Patellinidae | 1 | 1 | ||
| Lagenida | Nodosarioidea | Lagenidae | 3 | 17 |
| Nodosariidae | 5 | 5 | ||
| Vaginulinida | Vaginulinidae | 1 | 1 | |
| Robertinida | Robertinoidea | Robertinidae | 1 | 1 |
| Rotaliida | Buliminoidea | Buliminidae | 1 | 1 |
| Reussellidae | 2 | 6 | ||
| Trimosinidae | 2 | 2 | ||
| Cassidulinoidea | Bolivinitidae | 7 | 8 | |
| Tortoplectellidae | 1 | 1 | ||
| Siphogenerinoididae | 1 | 1 | ||
| Chilostomelloidea | Trichohyalidae | 1 | 1 | |
| Anomalinidae | 1 | 1 | ||
| Fursenkoinoidea | Fursenkoinidae | 1 | 2 | |
| Turrilinoidea | Turrilinidae | 1 | 1 | |
| Stainforthiidae | 2 | 2 | ||
| Bolivinoidea | Bolivinidae | 1 | 10 | |
| Discorboidea | Rosalinidae | 3 | 10 | |
| Eponididae | 2 | 3 | ||
| Cancrisidae | 2 | 3 | ||
| Discorbidae | 1 | 1 | ||
| Heleninidae | 1 | 1 | ||
| Discorbinelloidea | Discorbinellidae | 2 | 4 | |
| Pseudoparrellidae | 1 | 1 | ||
| Planorbulinoidea | Cibicididae | 4 | 7 | |
| Planorbulinidae | 2 | 2 | ||
| Cymbaloporidae | 3 | 6 | ||
| Nonionoidea | Nonionidae | 3 | 7 | |
| Glabratelloidea | Glabratellidae | 1 | 1 | |
| Rotalioidea | Elphidiidae | 4 | 21 | |
| Elphidiellidae | 1 | 1 | ||
| Haynesinidae | 1 | 2 | ||
| Ammoniidae | 6 | 25 | ||
| Notorotaliidae | 1 | 1 | ||
| Acervulinoidea | Acervulinidae | 1 | 2 | |
| Asterigerinoidea | Amphisteginidae | 1 | 1 | |
| Epistomariidae | 1 | 1 | ||
| Asterigerinatidae | 1 | 1 | ||
| Nummulitoidea | Nummulitidae | 2 | 6 | |
| Murrayinellidae | Murrayinellidae | 1 | 2 | |
| Polymorphinida | Polymorphinoidea | Glandulinidae | 2 | 3 |
| Polymorphinidae | 4 | 6 | ||
| Ellipsolagenidae | 3 | 11 |
| Global β Diversities | Northern Island | Southern Island | Tidal Flat | Southern Coast | Kuwait Bay | ||
|---|---|---|---|---|---|---|---|
| Whittaker (βw) | 1.12 | Northern Island | 0.00 | ||||
| Southern Island | 0.84 | 0.00 | |||||
| Tidal flat | 1.80 | 1.69 | 0.00 | ||||
| Southern coast | 0.89 | 0.82 | 1.79 | 0.00 | |||
| Kuwait Bay | 0.84 | 0.86 | 1.69 | 0.85 | 0.00 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Enezi, E.; Khader, S.; Balassi, E.; Frontalini, F. Modern Benthic Foraminiferal Diversity: An Initial Insight into the Total Foraminiferal Diversity along the Kuwait Coastal Water. Diversity 2020, 12, 142. https://doi.org/10.3390/d12040142
Al-Enezi E, Khader S, Balassi E, Frontalini F. Modern Benthic Foraminiferal Diversity: An Initial Insight into the Total Foraminiferal Diversity along the Kuwait Coastal Water. Diversity. 2020; 12(4):142. https://doi.org/10.3390/d12040142
Chicago/Turabian StyleAl-Enezi, Eqbal, Sawsan Khader, Eszter Balassi, and Fabrizio Frontalini. 2020. "Modern Benthic Foraminiferal Diversity: An Initial Insight into the Total Foraminiferal Diversity along the Kuwait Coastal Water" Diversity 12, no. 4: 142. https://doi.org/10.3390/d12040142
APA StyleAl-Enezi, E., Khader, S., Balassi, E., & Frontalini, F. (2020). Modern Benthic Foraminiferal Diversity: An Initial Insight into the Total Foraminiferal Diversity along the Kuwait Coastal Water. Diversity, 12(4), 142. https://doi.org/10.3390/d12040142






