Abstract
Balkan chamois (Rupicapra rupicapra balcanica) is a protected species with an Inadequate-Bad (U2) conservation status in Greece. Our study explores its seasonal range use pattern, demography and habitat selection in a site of the Natura 2000 network, Timfi Mountain. To this aim, we examined 1168 observations obtained from six seasonal surveys (2002: four seasons, 2014 and 2017: autumn) and performed an ecological-niche factor analysis (ENFA), using 16 environmental and human-disturbance variables. The species had an annual range of 6491 ha (25% of the study area), followed the typical range-use pattern, and presented the minimum core area during the rutting season (autumn). Timfi Mt hosted 469 individuals in 2017 (the largest population in Greece), increasing by 3.55 times since 2002. The species selected higher altitudes during summer and autumn, pinewoods over broad-leaved woods as winter grounds, and it avoided south-facing slopes. Our results supported the anthropogenic risk avoidance hypothesis; the species always selected remote areas away from roads, human settlements, and hunting grounds. In Greece, 40% of its distribution area falls within hunting ban areas (16.5% of the country). A national conservation policy is needed towards maintaining and increasing roadless areas and hunting-ban areas within Balkan chamois range nationwide.
Keywords:
climate change; conservation; demography; disturbance; ENFA; habitat selection; hunting; poaching; seasonal range; roadless areas 1. Introduction
Wildlife shares the planet with an exploding human population [1] that transforms the land in such a way that further rapid wildlife habitat loss is expected, leading to even higher species extinction risks [2]. Although humans can use technological means to colonize and adapt to every corner of the Earth, complex evolutionary and ecological processes interact to determine the distribution patterns of wildlife species. Habitat selection is a fundamental spatiotemporal ecological process: animals select habitats that maximize their fitness, in a continuous trade-off between selecting the best foraging and breeding habitats that would increase their survival probability and reproduction success, while minimizing environmental stresses and anthropogenic risks [3]. The challenge, therefore, lies in pinpointing the underlying factors that restrict animal distribution, assessing in particular the role of human-induced factors as compared to natural ones in the habitat selection process, under the scope to minimize negative anthropogenic impacts through effective conservation management actions and policy decisions [4]. This challenge has to be met in a species-by-species approach at the local scale, where conservation action usually applies, rendering the conservation endeavour highly demanding. Such local-scale conservation actions can act synergistically and gradually lead to the elimination of the extinction risk of the target species globally.
The target species of the current work is the Balkan chamois (Rupicapra rupicapra balcanica), one of the most poorly studied sub-species of the Northern chamois R. rupicapra. In winter, it lives in forests adjoining cliffs and rocky slopes at medium altitudes and in summer it approaches mountainous grasslands above the treeline, in proximity with steep rocky slopes [5]. It remains there also during the rutting season in autumn, when mixed groups of females, males, yearlings and kids are formed, and males compete for mating. Its distribution range covers nine countries in South-Eastern Europe (population estimate of 9100–10,285 individuals) [6], often forming small and isolated populations, with varying conservation statuses and population trends [7]. In Greece, its overall population size is reported to have been doubled during the last two decades, reaching a national population size of 1180–1605 individuals [8] and 1330–1765 according to the most recent assessment of the IUCN red list of threatened species [6]. The species exhibits a long-lasting fragmented distribution pattern of 30 distinct sub-populations, grouped into six main populations [6]. The Greek legislation has forbidden hunting of the Balkan chamois since 1969. The Balkan chamois is, furthermore, protected under European legislation (listed in Annexes II, IV of the Habitat Directive) [9].
However, it features an Inadequate-Bad (U2) conservation status according to assessment categories of the Habitats Directive, with poaching, disturbance from hunting other game species, and road infrastructure reported as the highest impact threats for the species nationwide [10]. Pastoral farming and parasitic diseases were considered till now of secondary importance [5,8]. The new national policies of fast wind farm development in the high mountains to combat global warming under the Paris agreement [11,12] as well as the current shift of traditional sheep-goat extensive grazing to intensive cattle grazing appear as imminent threats that may result in habitat loss and degradation in the near future. Poaching attitudes have deep social and cultural roots in local Greek communities [13,14], but reliable data on poaching intensity, as well as on the proportion of hunters that systematically or occasionally exercise poaching are largely lacking. Hunting activity is a popular and widespread activity in the Greek countryside, recognized by the Greek State as a tool for forest management in the recent (2018) national forest strategy. According to the Greek legislation, hunting is freely allowed all over the country, including the protected areas of the Natura 2000 network or private lands when not fenced, but it is not allowed in several nature reserves and in wildlife refuges, which function as set-asides for the enhancement of wildlife populations. However, there is a gap of knowledge on the exact area of the Greek hunting grounds, and on the spatiotemporal use of natural ecosystems by hunters. Furthermore, the road network in Greece is extensive, resulting in severe problems of landscape fragmentation, with roadless areas (>1 km2 from the closest roads) accounting for far less than 24% of terrestrial land, as compared with the European (42%) and global (80%) averages [15,16]. As global road length is anticipated to increase dramatically (by 60% until 2050) [17], road sprawling is perhaps the most crucial global environmental threat that opens the Pandora’s box of profuse environmental problems, exacerbating ecosystem degradation, overexploitation of wildlife and natural resources and disruption of ecosystem functionality [15,18,19].
The present research attempts to explore the impact of such large-scale problems in a local context, taking as a case study the largest population of Balkan chamois in Greece, in Timfi Mt [8]. This is the first study on the habitat selection of the Balkan chamois, aiming at providing an integrated ecological overview of the species, under a conservation perspective. We attempted to answer the following research questions for the chamois population in Timfi Mt: (a) What is its seasonal range-use pattern? (b) What is its demographic structure and trend? (c) Which factors drive its habitat selection process? (d) Does the species avoid human presence? (e) Since disturbance from hunting is considered as a high-impact threat for the species at national scale, we also estimated the degree to which the chamois distribution area coincides with hunting ban areas nationwide. The ultimate scope is to provide a science-based conservation and policy guideline as a paradigm fostering conservation action for the Balkan chamois in Greece and in the Balkans.
2. Materials and Methods
2.1. Study Area
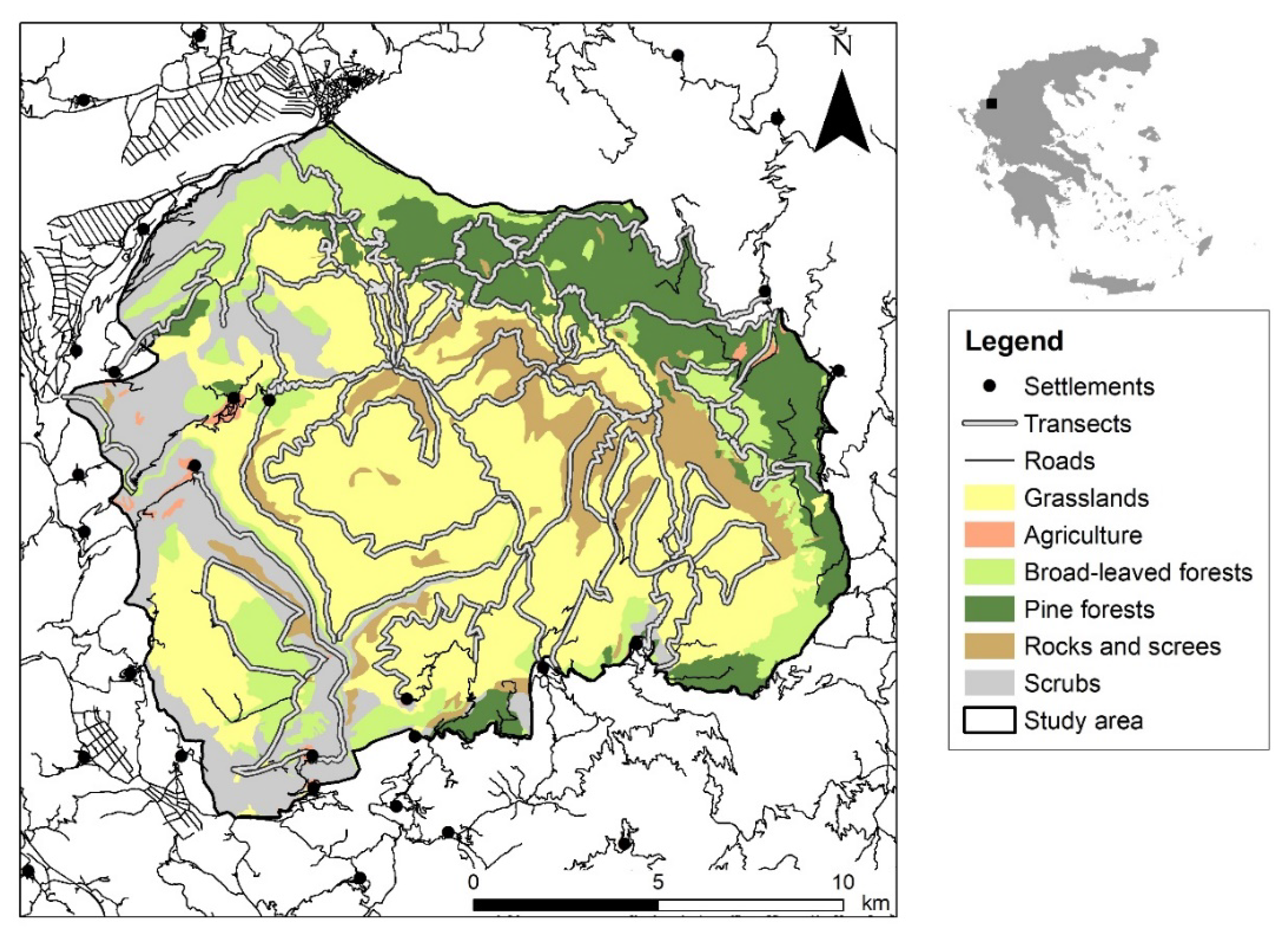
The study area is located in Timfi Mt (39.877347 Ν, 20.784790 Ε) at the north-western part of Pindus mountain range in Greece, covering 25,896 ha (Figure 1). The climate is intermediate between the Mediterranean and the Continental type: total annual precipitation of 1560 mm, average monthly temperature from 2.5 °C (January) to 24.4 °C (July), with a minimum in February (−8.6 °C) and a maximum in August (36 °C) (Data from Papingo Meteorological Station, 920 m: 2010–2013). It is a mountainous area (50% above 1000 m) within an elevation range between 412 m and 2497 m; the geological bedrock consists of limestone and flysch. The dominant habitat types in the study area are mountainous grasslands, pine forests (endemic black and endemic Mesogean pines), broadleaved forests (beech, mixed fir-beech, oak and mixed thermophilous forests), and screes (Table S1).
Figure 1.
Chamois field survey transects across the main habitat types in the study area (Natura 2000 site “Oros Timfi”: GR2130009), noting settlements and the existing road network. Habitat types described in Table S1.
The study area coincides with the Natura 2000 site “Oros Timfi” (GR2130009), covering parts of two more overlapping Natura 2000 sites: “Vikos-Aoos National Park” (GR2130001:87%) and “Kentriko Tmima Zagoriou” (GR2130004:8%), managed by the Management Authority of Northern Pindus National Park. It is a sparsely populated area, including six settlements. The main human activities include livestock breeding, hunting and tourism. Transhuman livestock breeding takes place from June until October above 1300 m, but permanent breeding is confined around the settlements. Tourism is an essential component of the local economy including mountaineering, climbing and hiking, which take place mainly in the Vikos-Aoos National Park. Chamois presence was documented in the study area since the Late Upper Pleistocene, playing a crucial role as a food resource to prehistoric human communities [20]. The population in Timfi Mt is the largest population in the country, being one of the 12 sub-populations that form the northern population block of the Pindus mountain range [6]. Pindus is the backbone of Greece, including three chamois population blocks that share similar genetic characteristics [21]. Chamois natural predators in the area are currently grey wolf, bear, red fox and golden eagle.
2.2. Chamois Surveys
We conducted 47 transects (mountaineering trails) of 205 km length in total, in conjunction with a set of vantage points along them (winter 2001 to spring 2002) (Figure 1), in order to cover all chamois habitats across the elevation gradient. Chamois surveys were evenly repeated by conducting 47 field days per season as follows: winter (cold period: 10 December–9 March), spring (parturition: 10 March–9 June), summer (warm period: 10 June–9 September), and autumn (rutting period: 10 September–9 December) [22]. The dataset included direct (animal sightings) and indirect observations (chamois tracks and droppings found in a belt of 1 m on either side of the transects; older droppings that might refer to a previous season were not considered). A group of animals was considered as one direct observation when the distance from a neighboring group was greater than the average inter-individual distance within the group [23].
To estimate chamois population size and trend, we counted animals using the pointage flash method [24] during the rutting period (three successive days at the end of October) for the years: 2002, 2014, 2017. The same transects were walked by seven groups of two researchers, to scan the study area uniformly. We noted the coordinates, date, time, group size and demographic composition for each direct observation, to be able to detect and exclude duplicate observations. We recorded the age and sex of each animal as follows: kids (<1 year), yearlings (between 1–2 years), adult females (>2 years) and adult males (>2 years) [25].
2.3. Environmental Variables
We overlaid a grid of 100 m × 100 m in the study area and calculated the average values, or the percent frequency of occurrence of the following 16 environmental variables per grid cell. We used a digital elevation model (DEM) of 20 m resolution, provided by Anavasi Editions Company. The DEM was reclassified to match the resolution of the grid, to calculate seven topographic variables (elevation, slope, curvature and aspect). We used the available habitat map of the Natura 2000 site of Timfi Mt provided by the Management Agency of Northern Pindus National Park (year 2008). We considered six broader habitat types (grasslands, pine and broadleaved forests, rocks, scrubs, agricultural land), and calculated their cover (%) per grid cell (Table S1). We finally calculated three human disturbance variables: the hunting activity (binary), the distance from settlements and the distance from the nearest road per grid cell. Distances were computed accounting for the curvature of the surface. Spatial data for hunting ban areas (wildlife refuges, including the strict nature reserves of Northern Pindus national park), and settlements were openly available [26]. The spatial file of the road network for the broader study area was provided by Anavasi Editions Company in 2001 and it was cross-checked for 2014 and 2017 using aerial photography. Calculations were performed using ArcGIS 10.7 [27].
2.4. Data Analysis
2.4.1. Annual and Seasonal Ranges
Using the data from the annual survey 2001–2002, we determined chamois seasonal ranges by employing the 95% fixed kernel density estimator (probability of species occurrence in its range greater than 95%) [28] and the extension “Animal Movement” [29] of ArcView. To define the core area as the area that is used more heavily in every season, excluding any random effect, we plotted the area of animal range (%) with the probability of area use (%) [30], as indicated in the upper right corner of Figure S1: the straight line reflects a random use of space, while the curve that sags below the straight line reflects a clumped use of space. The point where the curve’s tangent becomes parallel to the line of random use is the threshold to define the core area. After defining the seasonal ranges and respective core areas, we merged them to define the annual range and core area, respectively. We calculated seasonal ranges overlap, using ArcGIS 10.7 [27].
2.4.2. Population Demography
We assessed the annual chamois population density for the period 2001–2002 by dividing the estimated population size with the annual range. We considered only the autumn surveys (2002, 2014, and 2017) to compare the demographic characteristics of the population in terms of population structure and trend.
2.4.3. Habitat Selection
We performed an ecological-niche factor analysis (ENFA) [31] to explore the habitat selection by the species versus available environment [32] for the six survey datasets, namely the four seasons in 2002 and in autumn 2014 and 2017. In the case of direct observations, the absolute number of animals counted was assigned to each grid cell, whereas in the case of indirect observations, we assigned a value of one. ENFA assesses habitat selection by decomposing the ecological niche into two components within the ecological space: marginality and specialization. Τhe marginality measures how different are the average conditions in the habitat used compared to those that are available in the environment. Τhe specialization estimates habitat selection on a particular direction of the ecological space, i.e., the niche breadth of the habitat used. In the absence of high correlation among the candidate explanatory variables (Spearman’s |r| ≤ 0.75), we performed ENFA considering all the 16 environmental variables, by contrasting the environmental conditions between the occupied grid cells with presence data and the available grid cells across the study area [33].
We first extracted one axis of marginality and then used the broken-stick method [34] to define the number of specialization axes on the basis of their eigenvalues, which express the amount of specialization they account for [31]. We then identified the degree to which candidate explanatory variables contributed to the marginality and specialization in terms of their respective contribution score to the corresponding axes, noting that high scores mean strong habitat selection and narrow niche. Finally, we tested the statistical significance of the above scores, using a Monte-Carlo randomization procedure (1000 iterations). All statistical analyses were conducted using R 3.6.1 [35] and the R-package adehabitatHS (v.0.3.14) [36].
2.4.4. Chamois Distribution and Hunting Ban Areas of Greece
As disturbance from hunting other game species is considered as high-impact threat for the Balkan chamois, we assessed the spatial overlap of the chamois distribution with the hunting ban area in Greece. According to the Greek legislation, hunting is allowed all over the country, except from the following cases: for reasons of public safety, hunting is banned at a distance less than 250 m of cities and villages, and 100 m from single houses, as well as at a distance less than 500 m from the eastern borderline, in archaeological sites and monuments, and in camping sites when operating. To estimate this hunting ban area approximately, we used Corine land-cover data [37], applied a buffer zone of 250 m around the polygons classified under the broad category of artificial surfaces, and calculated the buffer area of 500 m from the eastern borderline using open data. We also calculated the hunting ban areas for wildlife conservation, namely the area of wildlife refuges, core zones of the 10 old-standing national parks of the country, strict nature reserves and nature reserves, by merging the respective calculated areas, using open data from the European Environmental Agency. Using the same source of open data, we further estimated the merged area of the game breeding stations and the controlled hunting areas of Greece, where hunting is banned or controlled for game management reasons. We merged all the above areas to calculate the permanent hunting ban area, excluding overlapping areas. Finally, we screened all ministerial decisions temporarily banning hunting in specific areas for a given period (game protection, burnt areas etc.) and we considered their overall area for 2020 (37 decisions since 2016). It was not possible to estimate the further hunting ban areas, such as fenced private land or private agricultural land where hunting harms the harvest crop. We then considered the most recent distribution map of Balkan chamois, provided by the Greek Ministry of Environment and Energy, which is used in the action plan for the species that is currently under development, and we calculated the proportion of the distribution area that falls within the hunting ban area nationwide. Calculations were performed using ArcGIS 10.7, and percentages were calculated considering the area of Greece (132,029 km2) and the distribution area of Balkan chamois according to the action plan (2706.67 km2).
3. Results
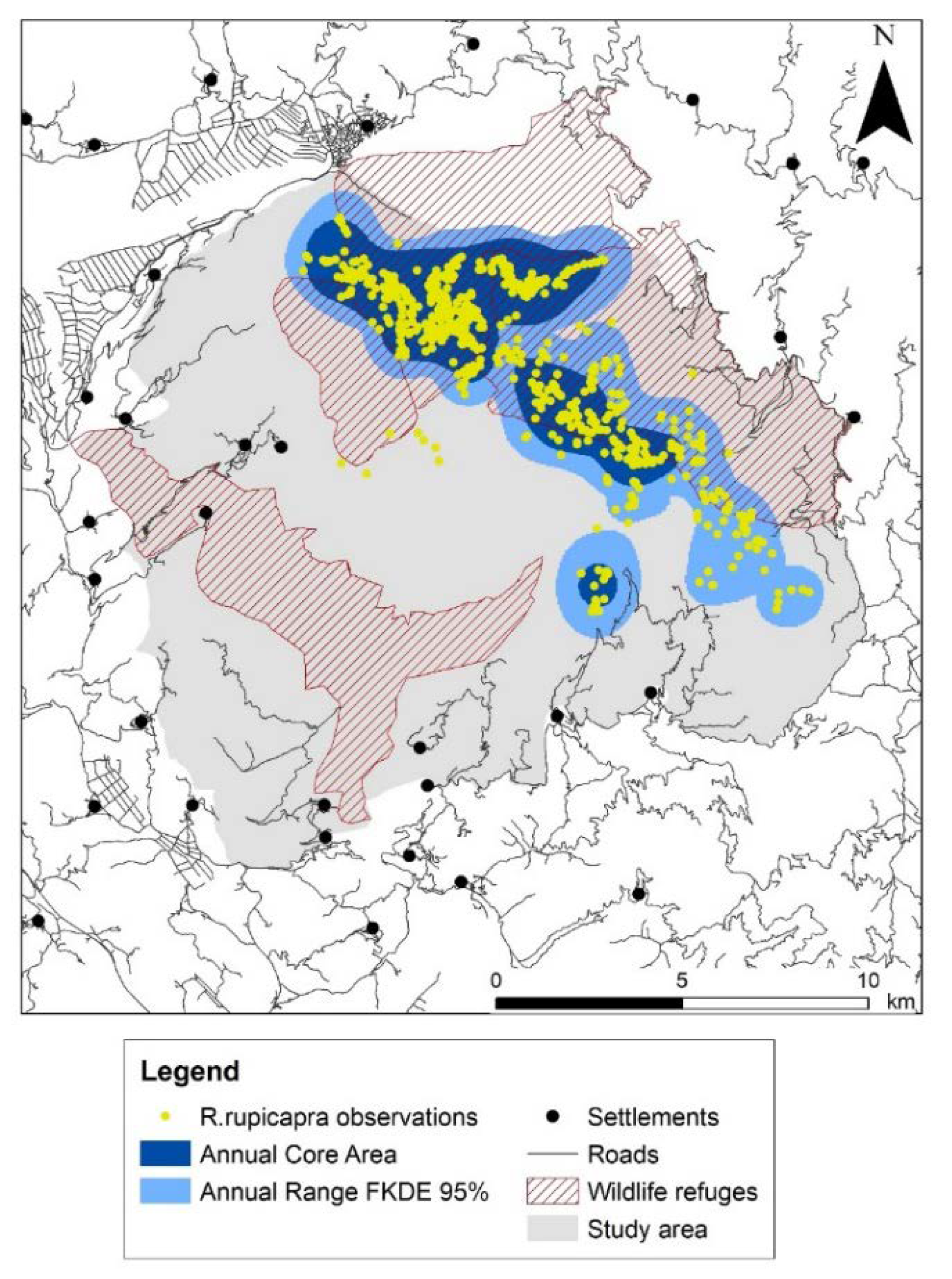
Mountainous grasslands covered a great part of the study area (46%) and extended above the tree-line together with rocky habitats and screes (9%). The forest zone included pinewoods (15%) and broadleaved woods (14%), whilst scrubs (15%) and agricultural areas (<1%) lay in the lower altitudes (Figure 1, Table S1). The overall road length was 174 km in the study area and has not changed since 2001. Hunting activity was excluded in 48% of the study area, namely in the core area of Vikos gorge and three wildlife refuges (Figure 2). We collected 1168 observations overall during the six surveys. For the annual survey of 2001–2002, 36% were direct observations, and 47% and 17% were chamois droppings and tracks respectively (Table S2).
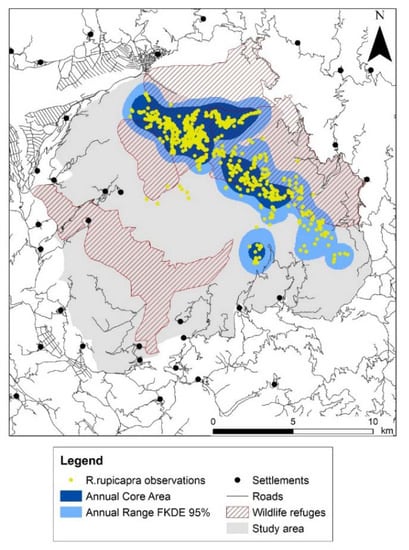
Figure 2.
Annual range and core area of Balkan chamois in the study area deriving from merging seasonal ranges and core areas, respectively; human settlements, roads and wildlife refuges (hunting ban areas) are indicated.
3.1. Annual and Seasonal Ranges
Seasonal ranges presented a minimum in winter and a maximum in summer, with the maximum aggregation within the core area observed in autumn (Table 1). We found an important overlap (above 49%) among seasonal ranges. Two main seasonal patterns emerged (Figure S1). First, the summer–autumn pattern consisted of a single large range area and two core areas; the two ranges coincided to a great extent but less so their respective core areas. Second, the winter–spring pattern consisted of four range areas and one or two core areas; the core areas coincided to a great extent and less so their respective ranges (Table 1). The annual range of the Balkan chamois in Timfi Mt covered an area of 6491 ha, accounting for 25% of the study area; the annual core area was about half of the annual range (Figure 2, Table 1).

Table 1.
Seasonal ranges and core areas (ha) of the Balkan chamois population in Timfi Mt (2001-2002) and respective proportions (Range: seasonal range/annual range, Core: core seasonal range/seasonal range), and overlap of the seasonal ranges and respective core areas (in parenthesis).
3.2. Population Demography
In 2002, the population density of the species was estimated to be 2 individuals/100 ha. The population has shown an important increasing trend of the order of 9.5% per year, increasing from 132 individuals in 2002 to 469 individuals in 2017 (Table 2). Kids were about one fourth of the overall population, the sex ratio was always in favour of females, and the fecundity rate remained high (0.6 to 0.8).

Table 2.
Trend and demographic characteristics of the Balkan chamois population in Timfi Mt (2002–2017).
3.3. Habitat Selection
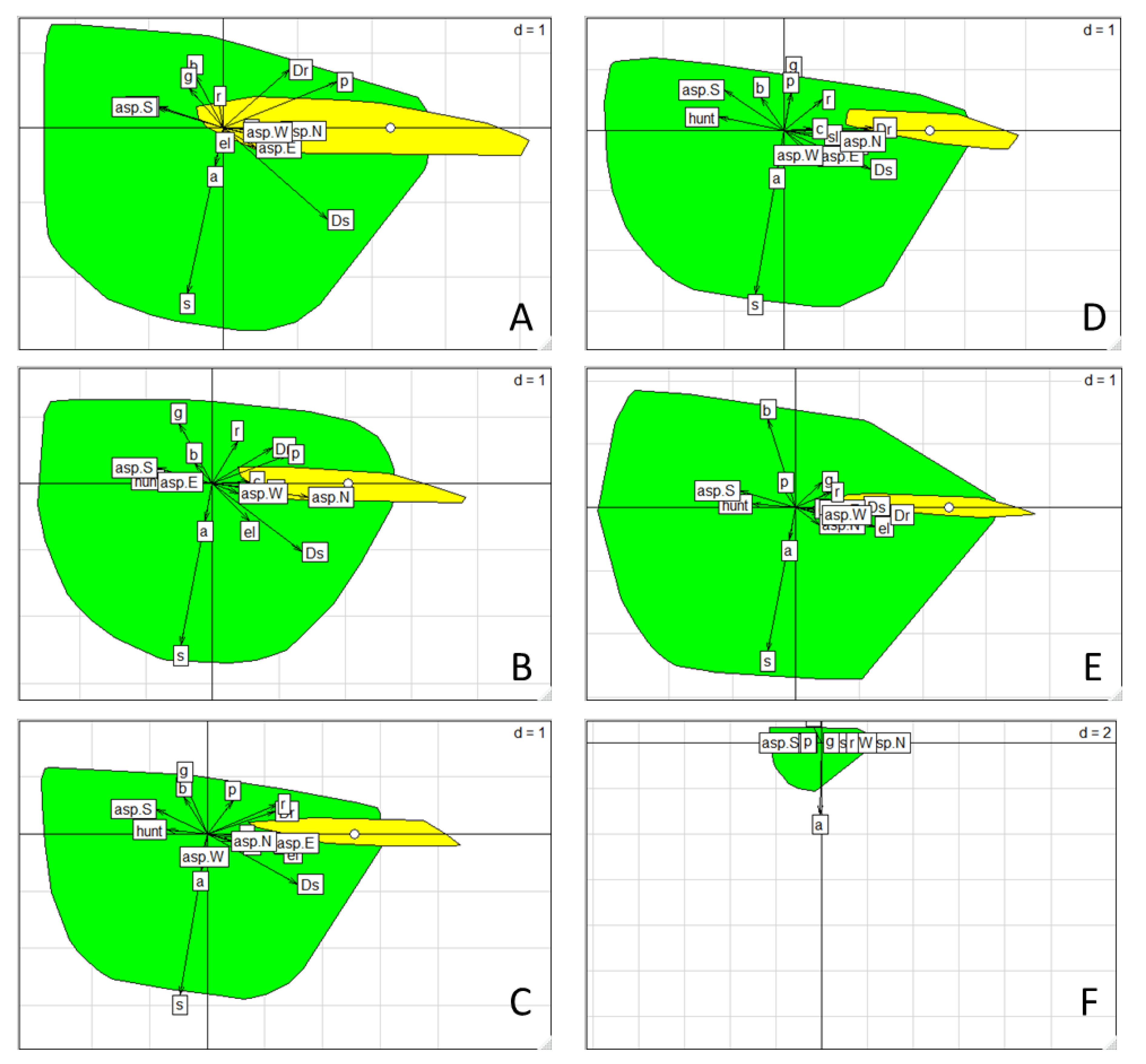
The ENFA results indicated a pronounced habitat selection behaviour by chamois, as both marginality and specialization scores were statistically significant (Monte Carlo, p < 0.001). The environmental conditions in which chamois were found showed a clear deviation from the average conditions in the available grids of the study area, as indicated by the high marginality scores obtained (Table S3). The species was recorded in a rather narrow interval of environmental conditions, being sensitive to their change, as the specialization scores showed a much greater variance of the environmental conditions in the study area (50–126 times) than within the chamois ranges. The scatterplots showed that the position of the used habitat was clearly distinct from the available habitat across the seasons (Figure 3). Considering the environmental variables contributing most to the marginality axis we found that the chamois always selected areas away from the human settlements, roads, and areas where hunting was allowed, avoiding also south-facing slopes (Figure 3, Table 3 and Table S4). This pronounced avoidance pattern was persistent across seasons and years, although a few more factors contributed to the seasonal habitat selection. In winter and spring, chamois selected north-facing slopes in pine forests. In summer and autumn, it preferred higher elevations with a substantial cover of rocks and screes, favouring east-facing or north-facing slopes, respectively. On the basis of the specialization axis, chamois did not tolerate any cover of scrub or agricultural and anthropogenic land in its habitat (Figure 3, Table 3 and Table S4).
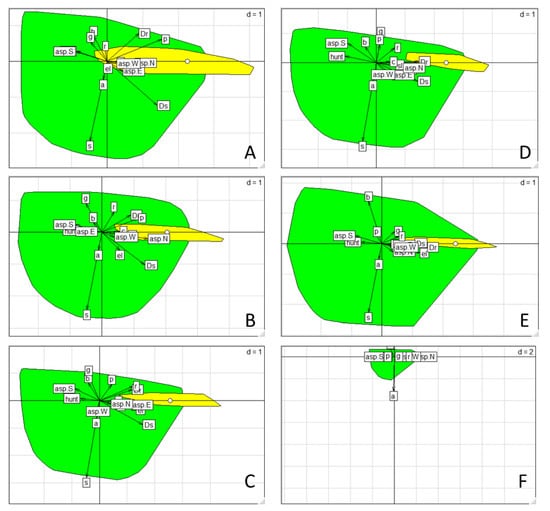
Figure 3.
Biplots representing the projection of the explanatory variables in the two-dimensional environmental space, formed by the marginality axis (x axis) and the first specialization axis (y axis). The position of the available (green) and used (yellow) resource units along the two axes are enclosed by minimum convex hull polygons. The white dot corresponds to the centroid of the used habitat. (A) Winter 2002, (B) Spring 2002, (C) Summer 2002, (D) Autumn 2002, (E) Autumn 2014, (F) Autumn 2017. See Table 3 for variable abbreviations and Table S4 for variable coefficients.

Table 3.
Average values (A) or percent frequency of occurrence (F) of the environmental variables in the occupied grid cells (100 m × 100 m) by chamois across the six surveys, and in the available grid cells of the study area. Shaded cells indicate the top five variables with the highest marginality coefficients (Table S4).
3.4. Chamois Distribution and Hunting Ban Area of Greece
The first rough estimation for the overall hunting ban area in Greece in 2019 was 21,842.37 km2 accounting for 16.54% of Greece. The area where hunting was banned for reasons of wildlife conservation was 9.16% of Greece. About 40% of the Balkan chamois distribution area falls within the hunting ban area of Greece (Table 4).

Table 4.
Percentage of the distribution area of Balkan chamois that lies within the hunting ban zones of Greece (2020), according to the legal rules relevant to hunting prohibition or control *. Percentages were calculated considering the Greek land area (132,029 km2) and the chamois distribution area (2706.67 km2).
4. Discussion
4.1. Range Pattern
Our results demonstrated that the Balkan chamois population in Timfi Mt was confined to a small part of the study area (25%), despite the large availability of typical chamois habitats there, such as mountainous grasslands, rocks and pine forests [5]. It exhibited two distinct seasonal spatial patterns (winter–spring and summer–autumn), presenting a minimum range in winter, a maximum range in summer and a minimum core area range in autumn. The species also seems to follow the “typical range-use pattern”: the animals restrict to a small winter range and start to move from spring to summer grounds expanding gradually their range [38,39,40,41], while in autumn they aggregate again due to rutting. This pattern is well known for the chamois population in cooler climates, where winter is the main stress period for animal survival, due mainly to harsher climate and to restricted food resources [42,43,44,45,46]. We note, however, that in the southernmost limit of Balkan chamois distribution, Giona Mt in Greece, chamois follow exactly the inverse pattern (Mediterranean range use pattern) from the Timfi population; it exhibits a minimum range and core area in the hot and dry stress period of summer, possibly due to global warming that further restricts fresh palatable food resources [47].
4.2. Population Trend and Demography
According to our findings, Timfi Mt hosts a large population, substantially increasing during the last two decades, and accounting for about one third of the national population. The former population density (2002) was particularly low (2 individuals/100 ha), whilst the current population density is unknown. If we assume that the annual range remained stable, the population density would reach 7 individuals/100 ha in 2017, which is comparable to those in other European mountains, usually lying between 6 and 10 individuals/100 ha, and in some cases reaching up to 24 or 40 individuals/100 ha [48,49,50,51]. The current population density should be lower than 7 individuals/100ha, as the population increase is expected to result in gradual range expansion.
The population seems to be healthy in terms of its demographic characteristics. The fecundity rate found of the study population falls within the usual rate of other European populations (0.55 to 0.85) [52,53]. However, we found a sex ratio in favour of females. This could be explained by the theory of higher male mortality due to high competition for mating [54,55], but in other areas, similar survival rates of the two sexes in Rupicapra rupicapra are reported [42,56]. One possible explanation for our study area could be the selective poaching towards males, under the scope to maintain females for next year’s harvest, or missing males that preferred to remain in lower forested habitats during the rutting season, and hence be less detectable during autumn surveys.
4.3. Seasonal Habitat Selection
Chamois selected higher altitudes in Timfi Mt during summer and autumn. They followed the typical circular seasonal migration pattern: moving to higher altitudes in spring as the snow recedes in search of sprouts and fresh palatable grass [22,57], occupying gradually the highest available habitats during summer and autumn, and moving slowly towards the lower altitudes in winter. We argue that it is possible that global warming will modify the above typical circular pattern in the future. For instance, we have recently noticed that some individuals remain at lower forested habitats during summer, as already reported in other cases [57,58,59,60]. In our case, the animals were observed drinking water in the rivers at the bottom of gorges, from 2017 onwards, either in the study area (Aoos gorge) or in the vicinal Tzoumerka Mt to the south (Aracthos gorge), potentially in search of water resources and cooler microhabitats. We have also noticed in the year 2017 a substantial delay in some cases in leaving the high-altitude autumn grounds of the study area towards winter grounds, potentially due to the milder weather conditions prevailing. Although the impact of global warming on chamois habitat selection and behaviour was not among the objectives of our study, this issue needs further investigation, in particular for the more vulnerable chamois populations of southern Europe.
Furthermore, the species avoided south-facing slopes across all seasons, preferring eastern-facing slopes in summer and mostly northern-facing slopes in winter and spring. In other parts of Europe, animals tend to stay less often on slopes with a southerly aspect [61], or avoid them only in summer and prefer them in winter to cope with the harsh weather conditions [57,62], which does not seem to be the case in the milder winter conditions of Timfi Mt. We argue that the strong avoidance pattern of southern-facing slopes could be attributed to global warming, as the animals may search cooler microhabitats throughout seasons. Such responses are of local character and largely depend on prevailing environmental contexts. For instance, the aspect did not seem to play an important role for the most southern population of Balkan chamois in Giona Mt, where the population employed a more dynamic strategy to adapt to the arid stressful conditions of summer (Mediterranean range pattern) rather than using different aspects of the mountain [47]. However, we do not attribute the selection of norther-facing slopes to global warming, but rather to the availability of escape terrains mostly in the northern parts of the mountains, as these microhabitats can function as adequate escape terrains for the species to evade human disturbance [63]. In our study area, steep slopes lie mainly in the northern aspects of Timfi Mt and extend even at lower altitudes in the pine zone, and less in the broadleaved forest zone that is characterized by milder topography. The greater availability of such escape terrain microhabitats in the pinewood zone could also explain why chamois selected pinewoods over broadleaved woods as winter grounds, a preference that is known from the southwestern Pyrenees [60]. Therefore, we argue that the availability of escape terrains may play an important role in the habitat selection process, as an underlying driver explaining the selection of northern aspects and pinewoods in the study area, rendering this issue worthy of further investigation.
4.4. Anthropogenic Risk-Avoidance Pattern
Our results supported the anthropogenic risk-avoidance hypothesis, as the species clearly selected remote areas away from roads, human settlements, and hunting zones. The pattern was strong and permanent in time. When even a small new road penetrates into wilderness or previous remote areas, it can still trigger a cascade of disturbances and impairments of ecosystems, a phenomenon known as the “contagious effect” of roads [64]. The impact of roads on wildlife is negative, reducing habitat quality, whilst ungulates, including chamois, show a general avoidance pattern to road infrastructure in different ecosystems of the world [65,66,67,68]. However, the small forest roads in mountainous ecosystems cannot be considered detrimental per se for chamois. They cannot really act as barriers for animal dispersion and its subsequent free gene flow, and the collision-mortality problem is reported to be minimal [69], presumably concerning only paved roads. However, roads are surrogates of human disturbance, and more specifically of poaching and hunting activity. The animals in Timfi Mt avoid roads in order to fend off poachers and hunters.
This is in line with the national report on the conservation status of chamois in Greece, highlighting roads, poaching and hunting as the main high-impact threats to the species nationwide [10]. Such interconnection of roads to hunting and poaching is widely recognized, because increased public access provided by roads increases the intensity of hunting and poaching of ungulates in several parts of the world [66,70,71]. Poaching is considered a common phenomenon in the country, enhanced by the poor law enforcement and poaching control by the Greek State [8,72]. For instance, the rangers of the Epirus hunting association denounced to the Court of Justice a poaching incidence (six chamois shot in the study area in February 2017), where barcoding techniques were also used to confirm the crime [73]. The litigation is still ongoing and if it ends up in a conviction of the defendants, this would be the first such case in Greece. Furthermore, our results support that legal hunting constitutes a pressure to the species, a fact that could be attributed to the disturbance from shooting and hunting dogs.
4.5. Chamois Distribution and Hunting Grounds
According to our results, the distribution area of Balkan chamois is quite well covered (40%) by hunting ban areas and particularly by wildlife refuges (29%). Given the negative impact of hunting disturbance on the chamois population, the designation of such refuges and reserves has been quite successful for chamois population conservation. Further research is recommended for the effectiveness of those hunting ban areas to benefit other game and non-game species, since the impact of hunting in terms of harvest or disturbance on other wildlife populations is still poorly studied [74,75]. At national scale, the hunting grounds of Greece were estimated to be about 83% of the country. This percentage is similar with other countries in Europe, where the hunting system is completely different, including hunting grounds leasing and the implementation of adequate local hunting management plans [76], which is not the case in Greece. Our findings include a degree of uncertainty. This is the first study that attempted to scan the current hunting legislation and estimate the hunting grounds in Greece, on the basis of which the overlapping distribution area of chamois was calculated. The legislation is scattered, and fragmented and spatial data are largely lacking for delineating the Greek hunting grounds accurately. The estimated hunting ban areas might be overestimated in our methodology, when considering the category of banning hunting for reasons of public safety, or underestimated, when considering the cases for which no data were available. Therefore, we suggest an update and improvement of the hunting legislation towards a single and explicit legal frame that would provide among others a clear delineation of the hunting grounds of Greece, to be available as open maps for hunters and the broad public.
4.6. Conservation Implications
We showed that road avoidance is a major determinant of the Balkan chamois space-use behaviour, which may apply to other populations in its distribution range in Greece and in the Balkans. Our findings, therefore, provide support for a national roadless policy, with special focus on the chamois distribution range. The policy should involve banning of road expansion in crucial chamois habitats and controlled access to existing mountainous roads, as road removal is a well-recognized strategy for restoring habitat for large mammals [77]. Such a roadless policy would be beneficial for chamois, largely diminishing the poaching pressure on the species, and fostering its fast population recovery at national scale. It would also benefit other protected large mammals that are negatively affected by roads in Greece [74], and would improve the environmental performance of the country, since the fragmentation pressure of Greece is high [16] and has been recently reported to be rapidly increasing [78]. Furthermore, our results highlighted the negative role of hunting disturbance on chamois space use in Timfi Mt but also showed that an important part of the national chamois population is protected within hunting ban areas. Therefore, we suggest the maintenance and expansion of the nature reserves and wildlife refuges all over the country, towards increasing their cover in chamois habitats. Such a policy would greatly contribute to the species population recovery, and it might be beneficial for other mountainous wildlife populations.
The two above policies should be implemented by priority in the Northern Pindus mountain range. Our study proved the importance of Timfi Mt for Balkan chamois population recovery at a national scale, hosting about one third of the national population. Roadless areas and new wildlife refuges or nature reserves should be established by priority in the mountains of the Northern Pindus population block, to link the respective chamois sub-populations through ecological corridors [21]. Located in the heart of the Northern Pindus mountain, the chamois population of Timfi Mt has the potential to recolonize previously occupied areas and secure gene flow towards vicinal small and declining populations, if such an upward trend persists [21], rendering the positive impact of the above two measures high for the national population. The animals have gradually started to recolonize adjacent areas, such as Vikos gorge in the study area or Mitsikeli Mt, where chamois was extinct.
Finally, our key research findings provide scientific evidence for measures and policies that could be integrated into the forthcoming national action plan for the Balkan chamois. The plan is under development by the Greek State and is expected to undertake a suite of well-coordinated actions and policies, in order to improve the conservation status of the Balkan chamois in the country, as an annexed species of the Habitats’ Directive.
Supplementary Materials
The following are available online at https://www.mdpi.com/1424-2818/12/4/124/s1: Table S1: Classification of the 25 habitat types of the study area into six broad habitat types. For each habitat type, we present its area (ha) and the respective cover (%) of the study area, as well as the habitat area (ha) that falls within chamois annual range and core area. The habitat typology follows the Habitats Directive (92/43/EC), noting priority habitat types of Annex I (*). Rivers and lakes considered in the broadleaved woods category, settlements considered under the agriculture category, Table S2: Number of observations by type collected in Timfi Mountain in the field surveys: 20021, 2014, 2017, Table S3: The scores of the marginality axis and the eigenvalue of the first axis of specialization of the ENFA models for the seasonal surveys of the 2002 (all observations) and for 2014 and 2017 (direct observations only), Table S4: Coefficients of the explanatory environmental variables in the 2D environmental space formed by the marginality axis (M) and the first specialization axis (S) of the ecological-niche factor analysis (ENFA) across the six surveys. Τhe top five variables with the highest marginality scores and the variable with the highest specialization score are shaded. Abbreviations as in Table 3, Figure S1: Balkan chamois seasonal ranges (Fixed Kernel Density Estimator: 95% probability of occurrence) and seasonal core areas respectively in Timfi Mountain in 2002. The upper right corner presents the method of defining the probability of occurrence in the core areas (80% in winter, 85% in spring, 70% in summer, and 75% in autumn).
Author Contributions
Conceptualization, H.P. and V.K.; Methodology: V.K., C.K., D.V. and H.P.; Formal analysis: C.K. and D.V.; Investigation: H.P.; Resources: V.K.; writing-original draft preparation: V.K., H.P., C.K.; writing-review and editing: V.K., C.K., D.V., H.P.; visualization: C.K.; project administration: V.K., funding acquisition: V.K. and H.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the GREEN FUND OF GREECE, in the frame of the measure “Innovative actions with citizens” – project title “Roadless areas and sustainable development in Greece”. HP has received funding by the WWF-HELLAS and the MANAGEMENT AGENCY OF NORTHERN PINDUS NATIONAL PARK to collect field data during the surveys of 2002 and 2014 respectively.
Acknowledgments
We are grateful to Mathieu Basille for providing technical advice in ENFA analysis. We also thank Anavasi Editions Company for providing digital data, the Management Agency of the Northern Pindus National Park for providing the habitat map of the study area, as well as to numerous volunteers for enthusiastically taking part in chamois surveys.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Wilson, E.O. Half-Earth: Our Planet’s Fight for Life; Liveright Publishing Corporation: New York, NY, USA, 2016; p. 272. [Google Scholar]
- Powers, R.P.; Jetz, W. Global habitat loss and extinction risk of terrestrial vertebrates under future land-use-change scenarios. Nat. Clim. Chang. 2019, 9, 323–329. [Google Scholar] [CrossRef]
- Morrison, M.L.; Marcot, B.G.; Mannan, R.W. Wildlife-habitat Relationships: Concepts and Applications, 3rd ed.; Island Press: Washington, DC, USA, 2006; p. 493. [Google Scholar]
- Krausman, P.R.; Cain, J.W. Wildlife Management and Conservation; The Johns Hopkins University Press: Baltimore, MD, USA, 2013; p. 342. [Google Scholar]
- Papaioannou, H.I.; Kati, V.I. Current status of the Balkan chamois (Rupicapra rupicapra balcanica) in Greece: Implications for conservation. Belg. J. Zool. 2007, 137, 33–39. [Google Scholar]
- Anderwald, P.; Ambarlı, H.; Avramov, S.; Ciach, M.; Corlatti, L.; Farkas, A.; Jovanovic, M.; Papaioannou, H.; Peters, W.; Sarasa, M.; et al. Rupicapra rupicapra. IUCN Red List Threat. Species 2018, (in press).
- Apollonio, M.; Andersen, R.; Putman, R. European Ungulates and Their Management in the 21th Century; Cambridge University Press: Cambridge, UK, 2010; p. 604. [Google Scholar]
- Papaioannou, H. Current status and conservation management of Balkan chamois (Rupicapra rupicapra balcanica) in Greece. In Chamois International Congress Proceedings, Lama dei Peligni—Majella National Park, Italy, 17–19 June 2014; Antonucci, A., Di Domenico, G., Eds.; Majambiente Edizioni: Lama dei Peligni, Italy, 2015; pp. 111–122. [Google Scholar]
- EEC. Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. 1992. Available online: http://data.europa.eu/eli/dir/1992/43/2013–07–01 (accessed on 20 February 2020).
- EIONET. Annex B—Report Format on the ‘Main Results of the Surveillance Under Article 11’ for Annex II, IV & V Species. Available online: http://cdr.eionet.europa.eu/gr/eu/art17/envxi9xsq (accessed on 2 March 2020).
- National Plan for Energy and Climate. Available online: http://www.et.gr/index.php/ (accessed on 1 March 2020).
- UNFCCC. United Nations Framework Convention on Climate Change. Decision 1/CP.2. Adoption of the Paris Agreement. Available online: https://unfccc.int/resource/docs/2015/cop21/eng/l09r01.pdf (accessed on 20 February 2020).
- Bell, S.; Hampshire, K.; Topalidou, S. The political culture of poaching: A case study from northern Greece. Biodivers. Conserv. 2007, 16, 399–418. [Google Scholar] [CrossRef]
- Karris, G.; Martinis, A.; Kabassi, K.; Dalakiari, A.; Korbetis, M. Changing social awareness of the illegal killing of migratory birds in the Ionian Islands, western Greece. J. Biol. Educ. 2018. [Google Scholar] [CrossRef]
- Ibisch, P.L.; Hoffmann, M.T.; Kreft, S.; Pe’Er, G.; Kati, V.; Biber-Freudenberger, L.; Della Sala, D.A.; Vale, M.M.; Hobson, P.R.; Selva, N. A global map of roadless areas and their conservation status. Science 2016, 354, 1423–1427. [Google Scholar] [CrossRef]
- Kati, V. Fragmentation of natural and semi-natural areas—SEBI 13. In Greece: State of the Environment. Summary/2018; National Center of Environment and Sustainable Development (NCESD): Athens, Greece, 2018; pp. 430–433. [Google Scholar]
- Dulac, J. Global Land Transport Infrastructure Requirements: Estimating Road and Railway Infrastructure Capacity and Costs to 2050; International Energy Agency: Paris, France, 2013. [Google Scholar]
- Laurance, W.F.; Clements, G.R.; Sloan, S.; O’Connell, C.S.; Mueller, N.D.; Goosem, M.; Venter, O.; Edwards, D.P.; Phalan, B.; Balmford, A.; et al. A global strategy for road building. Nature 2014, 513, 229–232. [Google Scholar] [CrossRef]
- Trombulak, S.C.; Frissell, C.A. Review of ecological effects of roads on terrestrial and aquatic communities. Conserv. Biol. 2000, 14, 18–30. [Google Scholar] [CrossRef]
- Kotjabopoulou, E. The mountainscapes of upper Palaeolithic epirus in NW Greece: A view from the bones. In The Palaeolithic of the Balkans, Proceedings of the XV World Congress, Lisbon, Portugal, 4–9 September 2006; Darlas, A., Mihailovic, D., Eds.; British Archaeological Reports, International Series 1819: Lisbon, Portugal, 2008; Volume 17, pp. 21–31. [Google Scholar]
- Papaioannou, H.; Fernández, M.; Pérez, T.; Domínguez, A. Genetic variability and population structure of chamois in Greece (Rupicapra rupicapra balcanica). Conserv. Genet. 2019, 20, 939–945. [Google Scholar] [CrossRef]
- Pepin, D.; Menaut, P.; Desneux, L.; Cargnelutti, B. Seasonal changes in the use of space by Isards (Rupicapra pyrenaica) in a protected area. In Ongules/Ungulates 91; Spitz, F., Janeau, G., Gonzales, G., Aulagnier, S., Eds.; IRGM-INRA: Toulouse, France, 1992; pp. 327–330. [Google Scholar]
- Clutton-Brock, T.H.; Guinness, F.E.; Albon, S.D. Red Deer: Behavior and Ecology of Two Sexes; Edinburgh University Press: Edinburgh, UK, 1982; p. 400. [Google Scholar]
- Houssin, H.; Loison, A.; Jullien, J.-M.; Gaillard, J.-M.; Taran, E.; Ebner, Ü.K. Validité de la méthode du pointage-flash pour l’estimation des effectifs de chamois (Rupicapra rupicapra). Gibier Faune Sauvage 1994, 11, 287–298. [Google Scholar]
- Catusse, M.; Corti, R.; Cugnasse, J.M.; Dubray, D.; Gibert, P.; Michallet, J. La Grande Faune de Montagne; Hatier Litterature Generale: Paris, France, 1996; p. 260. [Google Scholar]
- GEODATA.gov.gr. Available online: http://geodata.gov.gr/ (accessed on 23 February 2020).
- ESRI. ArcGISDesktop; Environmental Systems Research Institute: Redlands, CA, USA, 2018. [Google Scholar]
- Worton, B.J. Kernel methods for estimating the utilization distribution in home range studies. Ecology 1989, 70, 164–168. [Google Scholar] [CrossRef]
- Hooge, P.N.; Eichenlaub, B. Animal Movement Extension to Arcview, Ver. 1.1. Geological Survey; Alaska Science Center—Biological Science Office, USA Geological Survey: Anchorage, AK, USA, 1997. [Google Scholar]
- Powell, R.A. Animal home ranges and territories and home range estimators. In Research Techniques in Animal Ecology: Controversies and Consequences; Boitani, L., Fuller, T., Eds.; Columbia University Press: New York, NY, USA, 2000; pp. 65–110. [Google Scholar]
- Hirzel, A.H.; Hausser, J.; Chessel, D.; Perrin, N. Ecological-niche factor analysis: How to compute habitat-suitability maps without absence data? Ecology 2002, 83, 2027–2036. [Google Scholar] [CrossRef]
- Basille, M.; Calenge, C.; Marboutin, E.; Andersen, R.; Gaillard, J.M. Assessing habitat selection using multivariate statistics: Some refinements of the ecological-niche factor analysis. Ecol. Model. 2008, 211, 233–240. [Google Scholar] [CrossRef]
- Thomas, D.; Taylor, E. Study designs and tests for comparing resource use and availability. J. Wildl. Manag. 1990, 54, 322–330. [Google Scholar] [CrossRef]
- Jackson, D.A. Stopping rules in principal components analysis: A comparison of heuristical and statistical approaches. Ecology 1993, 74, 2204–2214. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 15 December 2019).
- Calenge, C. The package “adehabitat” for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 2006, 197, 516–519. [Google Scholar] [CrossRef]
- CLC 2018. Available online: https://land.copernicus.eu/pan-european/corine-land-cover/clc2018 (accessed on 20 February 2020).
- Crampe, J.P.; Bon, R.; Gerard, J.F.; Serrano, E.; Caens, P.; Florence, E.; Gonzalez, G. Site fidelity, migratory behaviour, and spatial organization of female isards (Rupicapra pyrenaica) in the Pyrenees National Park, France. Can. J. Zool. 2007, 85, 16–25. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, R.; Hidalgo, R.; Ameztoy, J.M.; Herrero, J. Census, population structure and habitat use of a Chamois population in Ordesa, N.P. living in sympatry with Pyrenean wild goat In Ongules/Ungulates 91; Spitz, F., Janeau, G., Gonzales, G., Aulagnier, S., Eds.; IRGM—INRA: Toulouse, France, 1992; pp. 321–325. [Google Scholar]
- Lovari, S.; Cosentino, R. Seasonal habitat selection and group size of the Abruzzo Chamois (Rupicapra pyrenaica ornata). Boll. Zool. 1986, 53, 73–78. [Google Scholar] [CrossRef]
- Nesti, I.; Posillico, M.; Lovari, S. Ranging behaviour and habitat selection of Alpine chamois. Ethol. Ecol. Evol. 2010, 22, 215–231. [Google Scholar] [CrossRef]
- Bocci, A.; Canavese, G.; Lovari, S. Even mortality patterns of the two sexes in a polygynous, near-monomorphic species: Is there a flaw? J. Zool. 2010, 280, 379–386. [Google Scholar] [CrossRef]
- Gonzalez, G.; Crampe, J.-P. Mortality patterns in a protected population of isards (Rupicapra pyrenaica). Can. J. Zool. 2001, 79, 2072–2079. [Google Scholar] [CrossRef]
- Jonas, T.; Geiger, F.; Jenny, H. Mortality pattern of the Alpine chamois: The influence of snow-meteorological factors. Ann. Glaciol. 2008, 49, 56–62. [Google Scholar] [CrossRef]
- Rughetti, M.; Festa-Bianchet, M. Seasonal changes in sexual size dimorphism in northern chamois. J. Zool. 2011, 284, 257–264. [Google Scholar] [CrossRef]
- Willisch, C.S.; Bieri, K.; Struch, M.; Franceschina, R.; Schnidrig-Petrig, R.; Ingold, P. Climate effects on demographic parameters in an unhunted population of Alpine chamois (Rupicapra rupicapra). J. Mammal. 2013, 94, 173–182. [Google Scholar] [CrossRef]
- Papaioannou, H.; Sgardelis, S.; Chondropoulos, B.; Vassilakis, D.; Kati, V.; Dimopoulos, P. Demographic characteristics, seasonal range and habitat topography of Balkan chamois population in its southernmost limit of its distribution (Giona mountain, Greece). J. Nat. Hist. 2015, 49, 327–345. [Google Scholar] [CrossRef]
- Boschi, C.; Nievergelt, B. The spatial patterns of Alpine chamois (Rupicapra rupicapra rupicapra) and their influence on population dynamics in the Swiss National Park. Mamm. Biol. 2003, 68, 16–30. [Google Scholar] [CrossRef]
- Corlatti, L.; Caroli, M.; Pietrocini, V.; Lovari, S. Rutting behaviour of territorial and nonterritorial male chamois: Is there a home advantage? Behav. Process. 2013, 92, 118–124. [Google Scholar] [CrossRef]
- Mustoni, A.; Pedrotti, L.; Zanon, E.; Tosi, G. Ungulati delle Alpi. Biologia-Riconoscimento—Gestione; Nitida Immagine Editrice: Cles, Italy, 2002; p. 539. [Google Scholar]
- Unterthiner, S.; Ferretti, F.; Rossi, L.; Lovari, S. Sexual and seasonal differences of space use in Alpine chamois. Ethol. Ecol. Evol. 2012, 24, 257–274. [Google Scholar] [CrossRef]
- Allaine, D.; Houssin, H.; Gaillard, J.M. Étude de la variabilité spatio-temporelle d’un indice de reproduction dans une population de chamois (Rupicapra rupicapra). Gibier Faune Sauvage 1990, 7, 85–94. [Google Scholar]
- Tosi, G.; Pedrotti, L.; Monaco, A.; Scherini, G. Progetto Camoscio Monte Baldo; Universita degli studi di Milano: Varese, Italy, 1996; p. 276. [Google Scholar]
- Schröder, W. Untersuchungen zur Ökologie des Gamswildes (Rupicapra rupicapra L.) in einem Vorkommen der Alpen—II. Teil. Zeitschrift Jagdwissenschaft 1971, 17, 197–235. [Google Scholar] [CrossRef]
- Schröder, W. Untersuchungen zur Ökologie des Gamswildes (Rupicapra rupicapra L.) in einem Vorkommen der Alpen. I, Teil. Zeitschrift Jagdwissenschaft 1971, 17, 113–168. [Google Scholar] [CrossRef]
- Corlatti, L.; Lebl, K.; Filli, F.; Ruf, T. Unbiased sex-specific survival in Alpine chamois. Mamm. Biol. 2012, 77, 135–139. [Google Scholar] [CrossRef]
- Hamr, J. Seasonal home range size and utilisation by female chamois (Rupicapra rupicapra L.) in Northern Tyrol. In The Biology and Management of Mountain Ungulates; Lovari, S., Ed.; Croom Helm: London, UK, 1985; Volume 12, pp. 106–116. [Google Scholar]
- Lovari, S.; Sacconi, F.; Trivellini, G. Do alternative strategies of space use occur in male Alpine chamois? Ethol. Ecol. Evol. 2006, 18, 221–231. [Google Scholar] [CrossRef]
- Shank, C.C. Inter-and intra-sexual segregation of chamois (Rupicapra rupicapra) by altitude and habitat during summer. Zeitschrift Säugetierkunde 1985, 50, 117–125. [Google Scholar]
- Herrero, J.; Garin, I.; García-Serrano, A.; García-González, R. Habitat use in a Rupicapra pyrenaica pyrenaica forest population. Ecol. Manag. 1996, 88, 25–29. [Google Scholar] [CrossRef]
- Ciach, M.; Peksa, Ł. Human-induced environmental changes influence habitat use by an ungulate over the long term. Curr. Zool. 2018, 65, 129–137. [Google Scholar] [CrossRef]
- Bačkor, P. Altitudinal distribution and morphological attributes of chamois (Rupicapra rupicapra tatrica) habitat in the Western Carpathians. Acta Zool. Lit. 2010, 20, 162–167. [Google Scholar] [CrossRef]
- Hamr, J. Disturbance behaviour of chamois in an Alpine tourist area of Austria. Mt. Res. Dev. 1988, 8, 65–73. [Google Scholar] [CrossRef]
- Selva, N.; Switalski, A.; Kreft, S.; Ibisch, P.L. Why keep areas road-free? The importance of roadless areas. In Handbook of Road Ecology; van Der Ree, R., Smith, D.J., Grilo, C., Eds.; John Wiley and Sons, Ltd.: Oxford, UK, 2015; pp. 10–26. [Google Scholar]
- Bleich, V.; Davis, J.; Marshal, J.; Torres, S.; Gonzales, B. Mining activity and habitat use by mountain sheep (Ovis canadensis). Eur. J. Wildl. Res. 2009, 55, 183–191. [Google Scholar] [CrossRef]
- Laurance, W.F.; Croes, B.M.; Tchignoumba, L.; Lahm, S.A.; Alonso, A.; Lee, M.E.; Campbell, P.; Ondzeano, C. Impacts of roads and hunting on central African rainforest mammals. Conserv. Biol. 2006, 20, 1251–1261. [Google Scholar] [CrossRef]
- Lian, X.; Li, X. Avoidance distances of four ungulates from roads in Kekexili and related protection suggestions. Chin. J. Ecol. 2012, 31, 81–86. [Google Scholar]
- Trepet, S.A.; Eskina, T.G. The influence of environmental factors on the dynamics of the size and spatial structure of the chamois (Rupicapra rupicapra caucasica) population on the Caucasian Reserve. Biol. Bull. 2013, 40, 698–707. [Google Scholar] [CrossRef]
- Putzu, N.; Bonetto, D.; Civallero, V.; Fenoglio, S.; Meneguz, P.G.; Preacco, N.; Tizzani, P. Temporal patterns of ungulate-vehicle collisions in a subalpine Italian region. Ital. J. Zool. 2014, 81, 463–470. [Google Scholar] [CrossRef]
- Gadd, M.E. Expected effects of a road across the serengeti. In Handbook of Road Ecology; van Der Ree, R., Smith, D.J., Grilo, C., Eds.; John Wiley and Sons, Ltd.: Oxford, UK, 2015; pp. 455–464. [Google Scholar]
- Slaght, J.C.; Milakovsky, B.; Maksimova, D.A.; Seryodkin, I.V.; Zaitsév, V.A.; Panichev, A.M.; Miquelle, D.G. Anthropogenic influences on the distribution of a Vulnerable coniferous forest specialist: Habitat selection by the Siberian musk deer Moschus moschiferus. Oryx 2019, 53, 174–180. [Google Scholar] [CrossRef]
- Papaioannou, H. Ungulates and their management in Greece. In European Ungulates and Their Management in the 21st Century; Apollonio, M., Andersen, R., Putman, R., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 540–562. [Google Scholar]
- Bounas, A.; Siarabi, S.; Toli, E.-A.; Sotiropoulos, K. DNA barcoding against poaching of Chamois (Rupicapra rupicapra), two confirmed cases from Greece. J. Wildl. Biodiv. 2018, 2, 1–5. [Google Scholar] [CrossRef]
- Iliopoulos, Y.; Youlatos, D.; Sgardelis, S. Wolf pack rendezvous site selection in Greece is mainly affected by anthropogenic landscape features. Eur. J. Wildl. Res. 2014, 60, 23–34. [Google Scholar] [CrossRef]
- Sokos, C.K.; Birtsas, P.K.; Connelly, J.W.; Papaspyropoulos, K.G. Hunting of migratory birds: Disturbance intolerant or harvest tolerant? Wildl. Biol. 2013, 19, 113–125. [Google Scholar] [CrossRef]
- Analysis of legislation and practice of hunting in some EU countries. Available online: http://www.enpi-fleg.org (accessed on 11 March 2020).
- Switalski, T.A.; Nelson, C.R. Efficacy of road removal for restoring wildlife habitat: Black bear in the Northern Rocky Mountains, USA. Biol. Conserv. 2011, 144, 2666–2673. [Google Scholar] [CrossRef]
- EEA. The European environment—State and outlook 2020. In Knowledge for Transition to a Sustainable Europe; Publications Office of the European Union: Luxembourg; Brussels, Belgium, 2019; p. 496. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).