1. Introduction
Crown group Galliformes are a very familiar clade, with members such as megapodes, chachalacas, guineafowl, quail, and pheasants (including the domestic chicken) that are distributed on all continents, except Antarctica. The stem lineage leading to that diverse crown group (including several extinct groups within the total group Pangalliformes [
1,
2]) is documented by a wide variety of Eocene, Oligocene, and early Miocene fossils, and extinct taxa from Africa, South America, North America, Asia, and Europe (e.g., [
3,
4,
5,
6,
7,
8,
9,
10]). A recent reanalysis of
Sylviornis and related fossils suggests that at least one lineage of pangalliformes outside of Galliformes survived into the late Holocene of New Caledonia and Fiji [
11]. Aside from that Holocene material, the youngest records of stem galliforms are otherwise from the early Miocene (e.g., [
2,
7]).
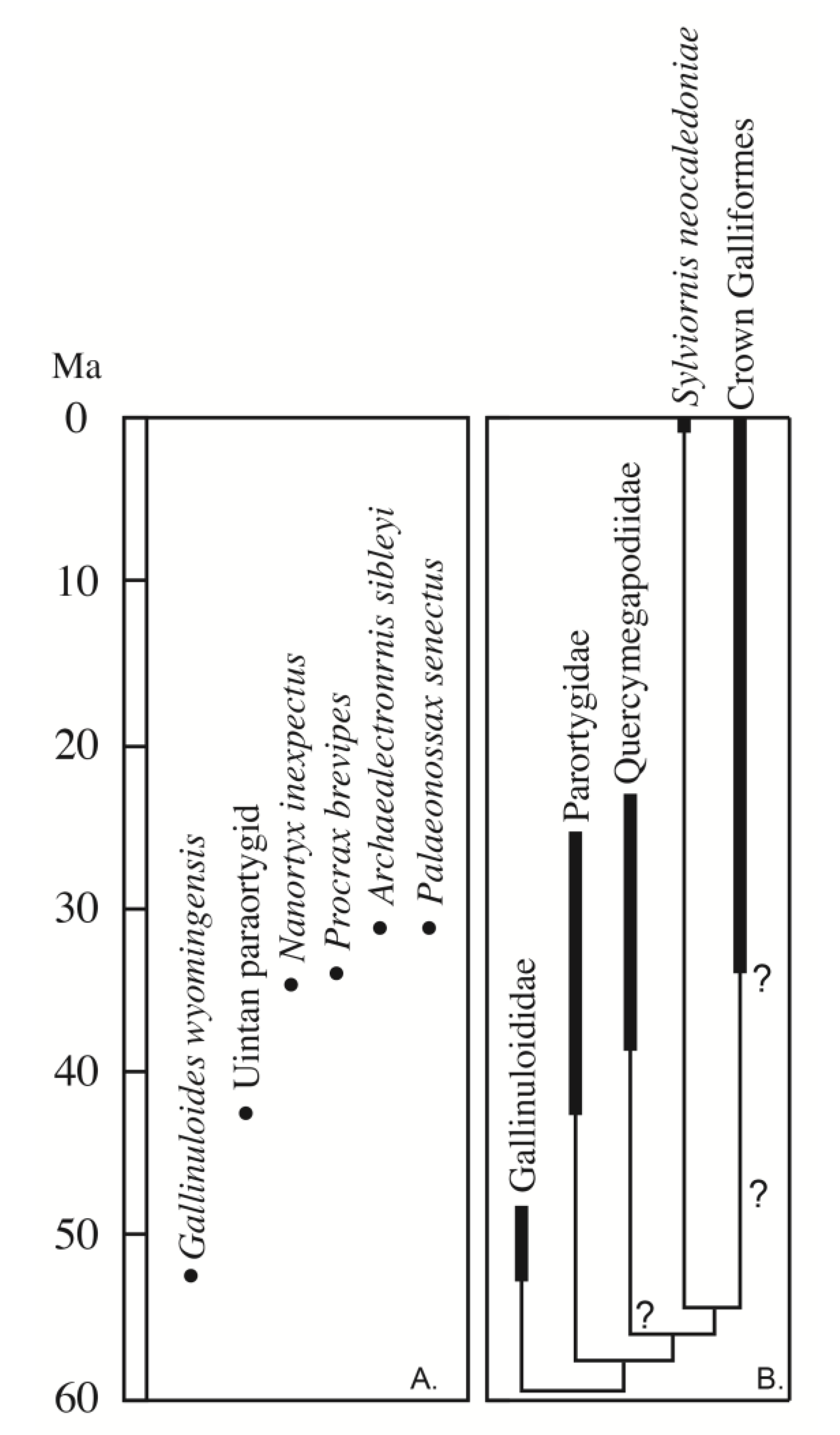
Despite the very wide geographic distribution of Paleogene stem galliforms (Asia, Africa, South America, North America, and Europe), the fossil record of stem galliforms is sparse in North America, and uncommon in the middle Eocene record around the world (
Figure 1). Potential records of stem galliforms have been described from the Cretaceous of the USA: a small galloanserine quadrate from the late Maastrichtian Lance Formation in eastern Wyoming [
12],
Austinornis from the Campanian of Texas [
1], and others [
13]. The oldest recognized records of stem galliform birds in North America are the specimens of
Gallinuloides wyomingensis (Gallinuloididae) from the early Eocene of Wyoming [
5,
14]. The extinct taxon
Amitabha urbsinterditensis from the early middle Eocene (Bridgerian North American Land Mammal Age, NALMA) of Wyoming was described originally as a crown group member of Galliformes, but more recent work demonstrates that it is not a galliform relative, being instead a gruiform related to Rallidae [
14]. Weigel [
15] described
Nanortyx inexpectus as a small species of New World quail (Odontophoridae) from the late Eocene (~35 Ma) Calf Creek fauna of the Cypress Hills Formation of Saskatchewan, Canada, based on a partial tarsometatarsus and coracoid. That material has not been reassessed in detail (including the two additional coracoid fragments mentioned, but not described or illustrated by Weigel [
15]).
Nanortyx is likely a stem galliform, but not a member of Paraortygidae, based on the reported presence of a circular and cup-like scapular cotyle [
16], which contrasts with the flattened scapular cotyle among crown galliforms and the elliptical cotyle in paraortygids (see below). While the late Eocene (Chadronian NALMA) and early Oligocene (Orellan NALMA) fossils from South Dakota and Nebraska named
Procrax brevipes,
Archaealectrornis sibleyi, and
Palaeonossax senectus were published originally [
17,
18,
19] as Galliformes, more recent data from Mayr [
2,
20] support not only their position outside of the crown group, but also their likely synonymy with one another. However, see Mayr and Weidig [
5] for a hypothesis about a crown position for
Procrax. Those fossils likely represent one or more non-crown group species that are approximately the size or slightly larger than the extant ~500 gram Chaco Chachalaca (
Ortalis canicollis) [
20,
21] and are much larger than
Nanortyx [
15]. If Mayr’s more recent hypothesis about a non-crown phylogenetic position is correct, then it is possible that only three named species of Paleogene stem galliforms are known from North America (i.e., the early Eocene
Gallinuloides wyomingensis, the late Eocene
Nanortyx inexpectus, and the late Eocene-Oligocene
Palaeonossax senectus, and its potential junior synonyms). A fossil originally published as the oldest odontophorid from the early Oligocene (Orellan NALMA) of Colorado [
18] may represent another non-crown member of the galliform clade, but that partial tarsometatarsus needs to be reexamined. Given the published fossil record, there is a gap in the pangalliform fossil record in North America, more than 15 million years long (between specimens of
Gallinuloides in the early Eocene and the holotypes of
Nanortyx and
Procrax, in the late Eocene;
Figure 1).
Increasing the geographic examination of Paleogene pangalliforms, we see that this middle Eocene gap is global in its scope, though smaller than the North American gap (>5–10 million years vs. >15 million years). This temporal dearth of specimens occurs between the youngest specimens of Gallinuloididae (
Paraortygoides) from the early middle Eocene of Europe, and the oldest records of Quercymegapodiidae and Paraortygidae in the late Eocene of Europe [
2,
6,
22]. The African record has a similar pattern with stem galliforms of unknown family clade affiliation, known from the late early or early middle Eocene of Tunisia (
Chambiortyx cristata [
8]) and early middle Eocene (early Lutetian, ~47–49 Ma) of Namibia (
Namaortyx sperrgebietenesis [
23]). The Asian stem galliform record currently is restricted to the early Eocene [
4,
9] and one other younger specimen [
10]. Potentially narrowing this global gap is
Scopelortyx klinghardtensis assigned to the Paraortygidae from the late middle Eocene (Bartonian; 38–41.3 Ma) of Namibia [
16], and the paraortygid
Xorazmortyx turkestanensis [
10] from the latest Lutetian or Bartonian of Uzbekistan. Thus, the size of the global middle Eocene gap is dependent on the age of the specimens of
Scopelortyx and
Xorazmortyx. Another published specimen hypothesized to be a pangalliform at the edges of this gap is
Telecrex grangeri from the Irdin Manha Formation of Inner Mongolia, China.
Telecrex was first published as an early rallid, but Olson [
24] reassessed the fossil femur as the oldest known guineafowl (Numididae). Its phylogenetic position among pangalliforms has been questioned [
6]. It appears that the late Eocene age estimate for the Irdin Manha Formation, repeated by many avian paleontological authors (e.g., [
9,
10,
20,
24]), has been revised to early middle Eocene (~48–49 Ma) [
25]. That age suggests the oldest known crown galliform (if
Telecrex is a member of Numididae) is middle Eocene (
Figure 1).
This gap in our knowledge of the evolution of early pangalliforms is interesting because Mayr [
2] has hypothesized that the shift to a large crop (and gastroliths in a gizzard) associated with a greater dietary component of seeds, and coarse plant matter likely occurred after the origin of Gallinuloididae in the early Eocene and before its arguable presence among later Eocene lineages (Quercymegapodiidae). Thus, the middle Eocene may have been a time of an increase in dietary breadth and a shift in pangalliform ecology. However, if the taxonomic allocation of the early Eocene (~55 Ma) pangalliform from Mongolia to the Quercymegapodiidae is correct [
9], then those features of the digestive track may have originated even earlier (i.e., Paleocene).
Expanding this meager fossil record in the middle Eocene is the discovery of a new coracoid fragment from an extinct paraortygid pangalliform from the middle Eocene Uinta Formation (Uintan NALMA, Lutetian), Uinta Basin of Utah (
Figure 2). This fossil documents a new Paleogene species of small-bodied pangalliform that derives from a temporal gap in their North American (and global) history between the early Eocene
Gallinuloides wyomingensis and late Eocene
Nanortyx inexpectus. This new fossil possesses apparently unique morphological features among pangalliforms and derives from a bird comparable in size to the smallest extant galliforms.
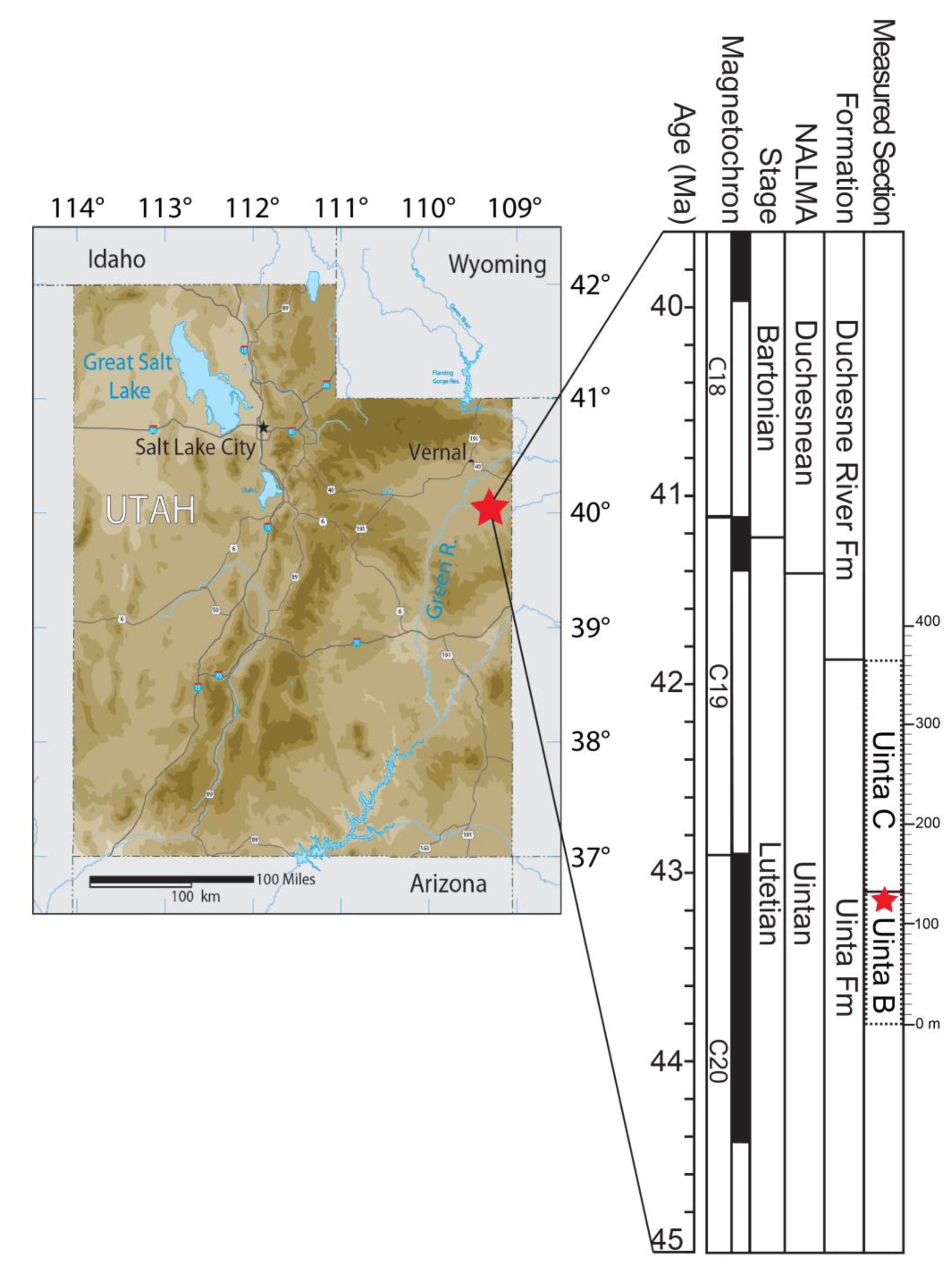
The Uinta Formation is exposed south of the Uinta Mountains in the Uinta Basin, an east-west trending synclinal basin in northeastern Utah [
28]. The formation is approximately 1298 m thick and has a discontinuous outcrop that is traceable for approximately 145 km from the edge of the Utah-Colorado state line to the western edge of the Uinta Basin [
29,
30,
31,
32,
33,
34,
35]. The Uinta Formation was deposited under fluvial conditions resulting from numerous streams and rivers feeding into the receding lake (Lake Uinta) that helped to form the underlying Green River Formation [
36].
The Uinta Formation has been divided formally into the lower Wagonhound and upper Myton Members, yet most workers refer to the formation using a modified tripartite division of Osborn’s [
35] stratigraphic nomenclature, from lowest to highest: Uinta A; Uinta B; and Uinta C [
25,
32,
37,
38]. The lowest unit, Uinta A, intertongues with the Parachute Creek Member of the Green River Formation and is composed of medium to massive resistant fine-grained sandstones that are yellowish-brown and yellowish-gray, and about 226 m in thickness [
32,
39]. Uinta B sediments are dominated by non-resistant slopes composed of light-gray, light green-gray, light-brown, light-purple mudstones, and claystones [
26,
32]. The muds and clays are interbedded by green-gray, yellow, and brown fine-grained sandstone beds, as well as gilsonite veins in the lower intervals of this unit [
26,
32,
40]. In the eastern part of the basin, Uinta B is capped by the massive gray and brown
Amynodon sandstone that spans 1.6 km in length, and that sandstone was the original boundary between the Uinta B and Uinta C horizons [
26,
35,
41]. Uinta C beds are light green, light gray, and light brown mudstones and claystones in the lower intervals, and the upper part is composed of mainly deep red-orange, red, dark brown, grayish-purple, and yellow muds and claystones with small green fine-grained sandstone beds interspersed throughout [
26,
32].
The Uinta Formation yields the type fauna for the Uintan NALMA, and recent fossil collecting efforts along with stratigraphic studies have made it possible to define biochronologic units and associated stratotype sections for this NALMA as Ui1, Ui2, and Ui3 [
41]. The greatest faunal diversity is found in the Ui2 biochron, or early Uintan, which corresponds to the Uinta B sediments [
42]. The locality where the pangalliform fossil was discovered is UMNH.VP.LOC.2734 (field locality number WU-117), and is at a 123 meter level of the stratigraphic section of Townsend et al. [
26] considered to be Uinta B sediments (
Figure 2) by those authors and Uinta C sediments by Sprinkel [
32]. The locality is near the biochronological transition from Ui2 to Ui3, but clearly within the Ui2 biochron [
42]. The typical Ui2 fauna includes multiple primate species, small bunodont artiodactyls, marsupials, leporids, micro-rodents (
Microparamys, small sciuravids, cylindrodontids, and
Protoptychus), larger ischyromyid rodents, pantolestids, agriochoerid and protoceratid artiodactyls, and larger ungulates such as
Eobasileus,
Achaenodon,
Uintatherium, and brontotheriids. Non-mammalian vertebrates include a diverse array of turtles (carettochelyids, testudinids, baenids, and testudinoids), crocodiles, lizards, and fish. The incredible diversity of taxa typical of Ui2 is what is generally recovered in any given field season at UMNH.VP.LOC.2734 (WU-117).
Only two fossil birds are otherwise described from the Uinta Basin, both from the slightly younger Uinta C beds. The original identification of
Eonessa anaticula Wetmore [
43] from the Myton Pocket in the Myton Member of the Uinta Formation was as an anseriform. Later work by Olson and Feuduccia [
44] indicates that it is not anseriform, but possibly a member of the Gruiformes.
Eutreptornis uintae was described originally as a cariamiform bathornithid, but its relationship to other taxa placed with bathornithids is unclear [
45,
46]. Townsend et al. [
26] noted the presence of bird fossils at localities WU-170 and WU-117 (where the pangalliform specimen also was collected), but those specimens have yet to be studied.
3. Systematic Paleontology
Pangalliformes Clarke, 2004 [
1]
Paraortygidae Mourer-Chauviré, 1992 [
6]
Unnamed species
Specimen
UMNH.VP.30891 is an omal end of a left coracoid collected by PAH from locality UMNH.VP.LOC.2734 (field number 18-019, Washington University locality WU-117) in the middle Eocene Uinta Formation of Utah, USA (
Figure 2 and
Figure 3).
Diagnostic Features
The fossil shares with Paraortygidae a short procoracoid process and an elliptical outline of the scapular cotyle with a long axis oblique to the omal-sternal axis. The large procoracoid prominence with an elongate slope into the m. supracoracoideus fossa is shared among the paraortygids the Uintan paraortygid,
Xorazmortyx turkestanensis, and
Scopelortyx klinghardtensis (
Figure 3) [
10,
16], but the prominence extends medial to the medial edge of the acrocoracoid only in the Uintan paraortygid and
Xorazmortyx. Those three taxa also share a short, obliquely oriented acrocoracoid crest [
10]. The caudolateral margin of the humeral articular facet is nearly straight in the Uintan paraortygid, but concave in
Scopelortyx and the most concave in
Xorazmortyx. The Uintan paraortygid also differs from
Scopelortyx and
Xorazmortyx in having an acrocoracoid that overhangs the m. supracoracoideus fossa, a craniocaudally shorter m. supracoracoideus fossa, and the apex of the acrocoracoid (medial view) being in its ventral half. The medial margin of the coracoid shaft in the Uintan paraortygid does not bifurcate cranially as in
Xorazmortyx, but the area where a pit would be present in
Xorazmortyx is not preserved in the Uintan paraortygid specimen. The Uintan paraortygid has a straight dorsal margin to the humeral articular facet as in
Xorazmortyx, but not
Scopelortyx [
10].
Occurrence
The fossil was found at locality UMNH.VP.LOC.2734, which is at the 123 m level above the base of section C in the Uinta Formation, and that locality has produced other bird material along with primate fossils [
26]. UMNH.VP.LOC.2734 contains the highest stratigraphic records of the mammals
Epihippus gracilis,
Mesomeryx grangeri,
Ourayia uintensis,
Pareumys milleri, and
Protoptychus hatcheri [
26]. The mammalian fauna is consistent with a biostratigraphic position within the Uintan B (near its top;
Figure 2) or early Uintan Ui2 NALMA, and is likely ~43–44 Ma in age [
26]. The precise locality coordinates are not presented here, in compliance with the United States Paleontological Resources Preservation Act (PRPA) (PL 111-11), but are available to qualified researchers from the repository upon request.
Description
The coracoid fragment UMNH.VP.30891 is 7.0 mm long craniocaudally (omal-sternal). The humeral articular surface is approximately 5 mm long craniocaudally, and has a maximum mediolateral width of 2.8 mm. The scapular cotyle is deeply concave and ovoid in outline. The long axis of that ovoid is oblique (approximately 45 degrees) to the mediolateral (and omal-sternal) axis of the bone with its apices located craniomedial and caudolateral. Near the medial apex of the cotyle is the greatly reduced but present procoracoid process that is missing its tip. The procoracoid prominence (with the procoracoid process at its apex) is the mediolaterally widest part of the specimen. The cranial part of the prominence slopes into the craniocaudally short m. supracoracoideus fossa, resulting in a relatively craniocaudally short concave portion (dorsal view) of the fossa (in its cranial half). There is no evidence of any pneumatic foramina in the m. supracoracoideus fossa, and the most-cranial (omal) part of the fossa is relatively flat. On the dorsal surface immediately cranial to the scapular cotyle is a small shallow fossa (that does not appear to be the result of wear or breakage) that is subtriangular in outline, with small (possibly pneumatic) foramina near the corners of the triangle (
Figure 3). The humeral articular surface extends caudally to the caudolateral side of the cotyle, is concave over its surface, and widens cranially making its outline asymmetric. The dorsal surface of the coracoid shaft just caudal to the scapular cotyle is distinctly offset from the cotyle by a steep slope. The coracoid shaft is a bit narrower than the cotylar area.
The caudal part of the acrocoracohumeralis ligament scar is preserved, but much of the acrocoracoid is missing. The acrocoracoid projects medially, but does not extend as far medially as the apex of the procoracoid prominence (with the missing tip of the procoracoid process). While much of the furcular facet is missing, the dorsal tip of the facet is preserved indicating that the bone likely did not extend significantly more medially. The caudal margin of the acrocoracoid is approximately level with the cranial tip of the humeral articular facet. The acrocoracoid crest is short and oriented obliquely to the mediolateral axis much like the scapular cotyle.
In dorsal view, the procoracoid prominence extends medially about as far as the humeral articular surface projects laterally. There is no evidence of a procoracoid foramen or notch for the passage of the n. supracoracoidei on the preserved coracoid shaft. The preserved ventral margin of the bone is straight suggesting that the acrocoracoid did not extend (far) ventrally. The cranial apex of the acrocoracoid would have been in the ventral half of the process, based on its preserved base. In cranial view, the preserved acrocoracoid has a roughly rectangular outline (though it is crushed a bit).
Remarks
The asymmetric outline of the humeral articular surface with a wider cranial end, a craniocaudally short m. supracoracoideus fossa, and a highly reduced procoracoid process (lacking a n. supracoracoidei foramen) are all features of the pangalliform clade. The presence of a deeply concave scapular cotyle is a crown avian plesiomorphy and indicates a non-crown group phylogenetic status of the specimen since that feature is absent in all crown members of Galliformes (e.g., [
14]). The position of the small procoracoid process adjacent to the medial apex of the concave scapular cotyle is consistent with that seen in the extinct Paraortygidae, and differs somewhat from Galliformes, where the likely homologous apex of the (reduced) procoracoid prominence appears overall to be a bit more cranially positioned [
21]. The long axis of the concave scapular cotyle being offset from a mediolateral (or omal-sternal) orientation with the medial apex positioned omally relative to the sternally positioned lateral apex also is present in, and apparently diagnostic of Paraortygidae [
10,
16,
21]. The cotyle is a bit more rounded with a different orientation in the Quercymegapodiidae and other stem taxa [
6,
10,
16]. The presence of a short procoracoid process in the fossil is another diagnostic feature of Paraortygidae [
16].
The fossil also exhibits a potentially autapomorphic (likely pneumatic) roughly triangular fossa with foramina cranial to the concave scapular cotyle (between the dorsal edges of the m. supracoracoideus fossa and the humeral articular facet) that is not known in any non-crown pangalliform (or galliform)—except for a shallow triangular fossa (lacking foramina) in a specimen of
Paraortyx lorteti and a foramen (not within a fossa) in the most recent referred coracoid of
Scopelortyx [
10,
49]. A likely derived feature is the large relative size of the procoracoid prominence (occupying a larger proportion of the m. supracoracoideus fossa) that appears relatively larger than the state in other fossils (such as
Gallinuloides) except for
Scopelortyx and
Xorazmortyx (see
Figure 2 in [
10]). The new fossil is much smaller than the material referred to the other known North American stem galliforms
Gallinuloides, and
Palaeonossax and its possible synonyms, but is similar in size to the much younger
Nanortyx (with its circular rather than elliptical scapular cotyle), and the similarly-aged
Scopelortyx and
Xorazmortyx [
2,
5,
20].
No other known stem (or crown) galliform has the (possibly pneumatic) fossa with foramina on the dorsal side of the coracoid just cranial to the scapular cotyle. However, the presence of a single foramen in that position in the coracoid recently referred to
Scopelortyx [
49], and a specimen of
Paraortyx lorteti illustrated by Zelenkov and Panteleyev [
10] having a shallow fossa there could suggest that this possibly pneumatic feature is indicative of a pneumatic morphology occurring in paraortygids more broadly. Additional specimens are needed to assess the character’s variability and taxonomic utility. In addition, the apex of the enlarged procoracoid prominence (i.e. the procoracoid process) is positioned near the medial apex of the elongate scapular cotyle as in some basal pangalliforms such as Paraortygidae (
Scopelortyx from Namibia [
16], and
Xorazmortyx from Uzbekistan [
10]) but the sizes of that prominence and process are larger than in other stem galliforms. In crown galliforms, it appears that the apex of the procoracoid prominence is positioned a bit more towards the omal end of the bone relative to the lateral apex of the flattened scapular cotyle (and the sternal end of the humeral articular facet). The Utah fossil is smaller than the same element in
Gallinuloides,
Parortygoides (Gallinuloididae), and
Procrax (possibly a stem galliform), and the fossil appears to be within the size variation known among members of the non-crown Quercymegapodiidae, Paraortygidae, and
Nanortyx [
6,
9,
10,
15,
21].
Crown group galliforms lack several of the features present in the fossil including the concave scapular cotyle, procoracoid process, enlarged procoracoid prominence, and fossa cranial to the scapular cotyle. Megapodes (Megapodiidae) are the extant sister clade of all other crown galliforms, serving as a basis for (plesiomorphic) comparison among crown clade members. Megapodiids lack the large bulbous procoracoid prominence present in the fossil [
2,
14]. The relative craniocaudal length of the m. supracoracoideus fossa varies within Megapodiidae. The caudal edge of the acrocoracoid is distinctly cranial to the cranial edge of the humeral articular facet in
Megapodius laperouse (MVZ 95073) and
Aepypodius afrakanius (MVZ 149060), but
Megapodius freycinet (MVZ 90031) and
Alectura lathami (MVZ 137590) have a relatively craniocaudally shorter fossa. The morphology of the m. supracoracoideus fossa in
Alectura lathami differs from the fossil in that it is directed a bit dorsally rather than just medially. The area cranial to the scapular cotyle where the shallow fossa is in the fossil is a broad convex area in the megapodiid skeletons examined. The m. supracoracoideus fossa has pneumatic foramina in
Megapodius laperouse and
M. freycinet, but there is no evidence of pneumatization of the fossa in
Aepypodius afrakanius and
Alectura lathami. All specimens of megapodiids examined also are distinctly larger than the Uinta fossil specimen.
The Uintan paraortygid most closely resembles in morphology and size the material placed in
Scopelortyx and
Xorazmortyx. The sizes of the coracoids of all three species are very similar with the omal ends of the coracoids being well under one centimeter in length [
10,
16,
49]. They share the enlarged procoracoid prominence, approximate sizes of the procoracoid process, short, oblique acrocoracoid crests, and other aspects of their morphology, but they differ in several features (see diagnostic features above) that distinguish each as a different taxon. The addition of their similar ages (~38–44 Ma) to that morphological and size similarity could suggest a relatively close phylogenetic affinity among those three species within Paraortygidae.
4. Discussion
The Uintan paraortygid specimen has a smooth bone surface consistent with the coracoid deriving from an individual of adult size and morphology. Thus, this coracoid likely is from a relatively small sized individual on par with the smallest extant galliforms and similar in size to some stem galliforms (see above). The length of the omal end of the coracoid (scapular cotyle to cranial tip of the acrocoracoid, and related humeral articular facet length) are approximately the same in the Common Quail
Coturnix (MVZ 41337) and the fossil. However, the fossil is stouter (mediolaterally wider) than that of
Coturnix and the Taiwan Partridge
Arborophila crudigularis (MVZ 50531; which has a relatively longer omal end). The average body mass of those crown galliform taxa,
Coturnix (~90–100 grams) and
Arborophila crudigularis (~200–300 grams), provide an initial estimate of the mass for this stem group species, with the individual the fossil derived from likely being in the 100 to 200 gram range [
21].
The smallest extant galliforms are derived odontophorids and phasianids (like
Coturnix and
Arborphila above), and their small size along with their derived phylogenetic position is likely the result of body mass reduction in those groups rather than plesiomorphy for the crown clade. However, the small size of this fossil along with other smaller non-crown group specimens (e.g., [
6,
8,
10,
16,
21,
49]) is suggestive of a plesiomorphically smaller size in the basal part of the pangalliform clade with a potential body size increase (or series of increases) near the crown node with
Sylviornis [
11], extant megapodiids, and extant cracids as larger pangalliforms. This hypothesis potentially is supported by the somewhat older ages of the Uintan paraortygid,
Scopelortyx, and
Xorazmortyx (~38–44 Ma) relative to the larger late Eocene taxa.
If the Uintan paraortygid,
Scopelortyx, and
Xorazmortyx form a clade within, or paraphyletic assemblage at the base of Paraortygidae (given their geological ages), then that grouping suggests a relatively rapid and wide ranging intercontinental dispersal distributed across Namibia (Southern Africa, Southern Hemisphere), Uzbekistan (Central Asia, Eastern Hemisphere), and the United States (Western North America, Western Hemisphere). No mammalian taxon has that wide of a geographic distribution in the middle Eocene. Zelenkov and Panteleyev [
10] hypothesize that the morphology of
Xorazmortyx and
Scopelortyx (and paraortygids generally) may have allowed them to be better fliers, and thus better dispersers than other clades of pangalliforms. Without a phylogenetic hypothesis of paraortygid taxa, it is difficult to discuss the potential pattern of intercontinental dispersal that occurred. However, the slightly older age of the Uintan paraortygid relative to
Scopelortyx and
Xorazmortyx could suggest a pattern of rapid dispersal from North America to Asia to Africa. In addition, the presence of paraortygids in the Eocene of Europe could have occurred during known tetrapod dispersal events between Europe and North America, or through a less likely route (based on known mammalian dispersal patterns) from Asia to Europe.
Townsend et al. [
27] support a paleoenvironmental reconstruction of humid forest and woodland habitats during Uinta B times of the Uinta Formation, with a transition to more open and less wooded habitats in stratigraphically higher localities around the receding Lake Uinta. The small size of the fossil coracoid (and hence a small individual) likely relates to an ecological role on the floor of its forest/woodland habitat similar to extant galliforms of similar body size. Mayr [
50] and Mayr and Smith [
21] suggested that the large crop and ingestion of dietary grit associated with the gizzard (and its increased herbivorous dietary range including dry seeds) evolved along the stem lineage leading to Galliformes, with a large crop apparently absent in Paraortygidae and Gallinuloididae. The evolution of those anatomical features and the shift in dietary breadth might have occurred during the middle Eocene gap in the pangalliform record prior to the oldest fossils of Quercymegapodidae, making this interval of time of great interest to future evolutionary biology research. Mayr [
50] hypothesized that the shift in anatomy and diet to coarser material might be related to competition with mammalian herbivores and opening up of forest habitats in the Oligocene and Miocene, but with this record of Paraortygidae, the stem lineage leading to Quercymegapodidae and crown Galliformes (with their crops) also shifts earlier in time to ~44 Ma, not younger. In addition, the potential occurrence of a quercymegapodiid near the Paleocene-Eocene Boundary in Mongolia [
9] would shift that evolutionary event even older, possibly into the Paleocene. These older ages would support evolution of the enlarged crop and gizzard before the opening of terrestrial habitats, and perhaps its evolution was triggered by a new plant resource earlier in the Eocene (or Paleocene). Furthermore, the smaller size of this Uintan pangalliform as compared to
Gallinuloides and
Procrax likely points to a difference in dietary items consumed and an overall wider ecological role of stem galliforms in North America.
The fossils attributed to
Scopelortyx, while recovered from tufa limestone from a spring, have been interpreted as occurring in a semi-arid or arid steppe habitat [
16]. The holotype of
Xorazmortyx was found in near shore marine sediments with sharks and other fish [
10]. Pangalliforms are not aquatic, and clearly the ocean was not that individual’s primary habitat. With the Uintan paraortygid deriving from an interior forested habitat (in a wider lake basin), it would seem that the paleoecology of these small-bodied paraortygid birds included occupying a diversity of habitats (coastal, humid forest, and arid steppe). Their small body size and likely lack of a large crop and gizzard in these different settings suggests that their biology was flexible with a diet that was either broad, or focused on a similarly widely-available and abundant food resource.
Today, the galliform fauna of North America comprises some species of Cracidae, members of the New World quail clade Odontophoridae, and members of two clades of related phasianids, turkeys (Meleagridinae) and grouse (Tetraoninae). Mayr [
2] states that the published records of late Eocene and early Oligocene records of putative odontophorids need to be verified. Those fossils include
Nanortyx and another specimen referred to Odonotphoridae from Colorado [
15,
18]. Their small size is roughly equivalent to extant odontophorids (except that
Nanortyx was described as being smaller than living species) as well as the Utah specimen. It is possible that the previously published specimens lie outside the crown clade, and their small size could indicate phylogenetic affinity or possibly similarity in ecology to the Uintan paraortygid. These small fossils might be additional records of paraortygids from North America, as hinted at for the larger
Palaeonossax,
Procrax, and
Archaealectrornis [
2]. No Paleogene records of Cracidae are known [
20]. The oldest published records of the other galliform clades in North America are from the Neogene [
2]. The oldest global records of crown Galliformes are from the Oligocene, but the problematic holotype of
Telecrex grangeri is middle Eocene in age (see above) [
2]. We know that representatives of stem lineages and the crown clade co-occurred (and potentially competed) in places like Europe (e.g., [
2,
6,
7]), but the pattern and timing of galliform faunal change from stem galliform dominated to exclusively galliform composition in North America is unknown. Certainly, both the New World Cracidae and Odontophoridae had to have been present during the Oligocene, but currently we have no definitive records of either group before the Neogene. With the likely origin of the crown group of Phasianidae in Asia, the timing of dispersal of Tetraoninae and Melagridinae into North America is not well constrained beyond their supposed oldest records in the Miocene [
51,
52]. However, a reexamination of published fossils (in a phylogenetic context) and the discovery of new specimens, such as the fossil from Utah, are needed to better understand the establishment of the modern North American galliform fauna.