DNA Barcoding for Delimitation of Putative Mexican Marine Nematodes Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Nematode Sampling and Identification
2.2. DNA Extraction, PCR Amplification, and DNA Sequencing
2.3. Data Analysis
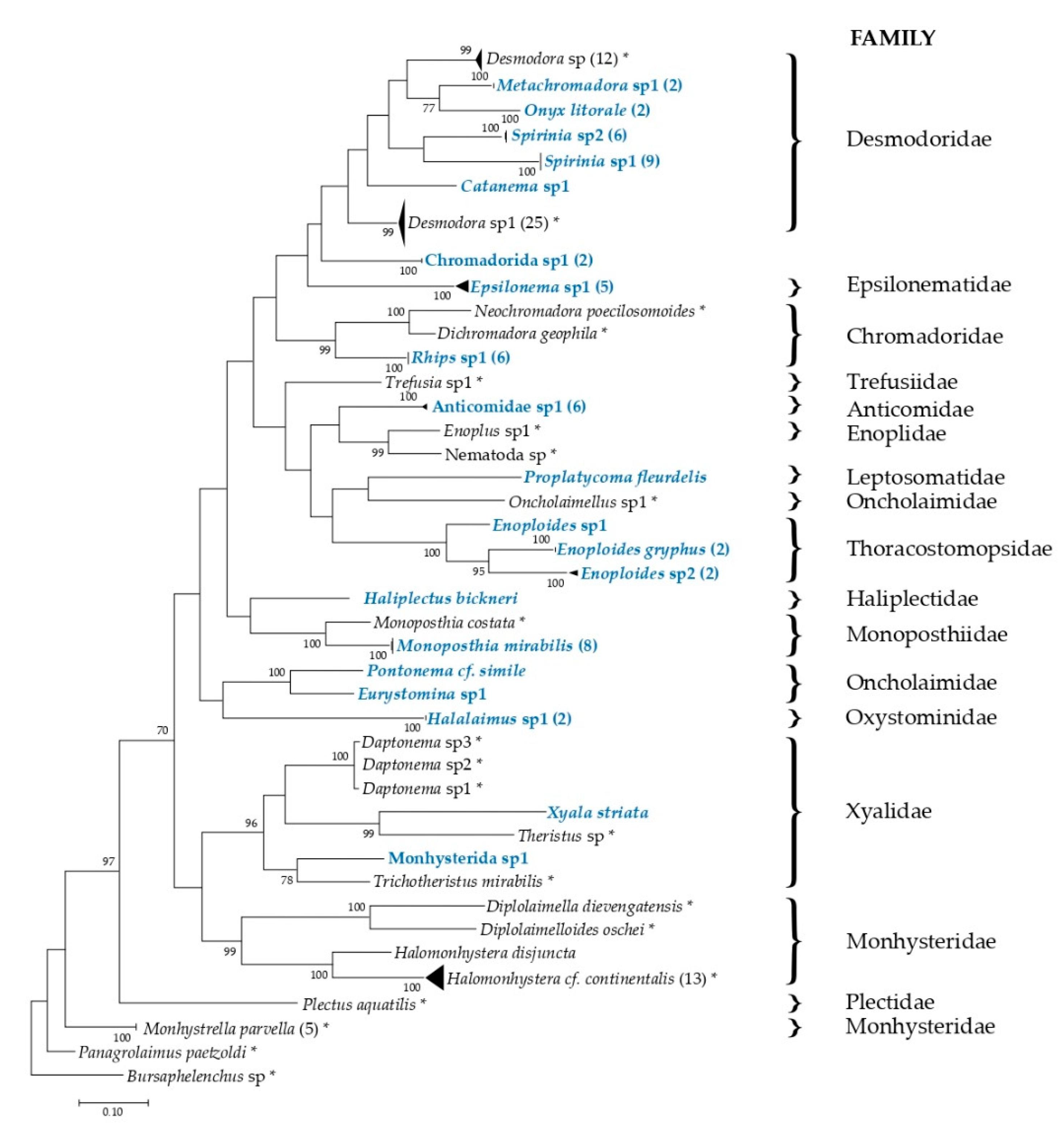
2.4. Phylogenetic Tree Reconstruction and DNA Taxonomy
3. Results
3.1. Morphological Identification
3.2. Amplification and Sequencing
3.3. Phylogenetic Analysis and DNA Taxonomy
4. Discussion
4.1. Diversity and Distribution of Meiofaunal Marine Nematodes in Mexico
4.2. COI Amplification
4.3. Integrative Taxonomy
4.4. Application of Species Delineation Models to Disentangle Diversity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Den Hoogen, J.; Geisen, S.; Routh, D.; Ferris, H.; Traunspurger, W.; Wardle, D.A.; de Goede, R.G.M.; Adams, B.J.; Ahmad, W.; Andriuzzi, W.S.; et al. Soil nematode abundance and functional group composition at a global scale. Nature 2019, 572, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Heip, C.; Vincx, M.; Vranken, G. Ecolgy of marine nematodes. Oceanogr. Mar. Biol. Annu. Rev. 1985, 23, 399–489. [Google Scholar]
- Vermeeren, H.; Vanreusel, A.; Vanhove, S. Species distribution within the free-living marine nematode genus Dichromadora in the Weddell Sea and adjacent areas. Deep. Res. Part II-Top. Stud. Ocean. 2004, 51, 1643–1664. [Google Scholar] [CrossRef]
- Boufahja, F.; Vitiello, P.; Aissa, P. More than 35 years of studies on marine nematodes from Tunisia: A checklist of species and their distribution. Zootaxa 2014, 3786, 269–300. [Google Scholar] [CrossRef] [PubMed]
- Ansari, K.G.M.T.; Pattnaik, A.K.; Rastogi, G.; Bhadury, P. An inventory of free-living marine nematodes from Asia’s largest coastal lagoon, Chilika, India. Wetl. Ecol. Manag. 2015, 23, 881–890. [Google Scholar] [CrossRef]
- Semprucci, F. Marine nematodes from the shallow subtidal coast of the Adriatic Sea: Species list and distribution. Int. J. Biodivers. 2013, 2013, 1–9. [Google Scholar] [CrossRef]
- Semprucci, F.; Balsamo, M.; Frontalini, F. La comunidad de nematodos de una laguna costera (Laguna de Varano, Italia meridional): Patrones de la ecología y la biodiversidad. Sci. Mar. 2014, 78, 579–588. [Google Scholar] [CrossRef]
- De Oliveira, D.A.S.; Derycke, S.; Da Rocha, C.M.C.; Barbosa, D.F.; Decraemer, W.; Dos Santos, G.A.P. Spatiotemporal variation and sediment retention effects on nematode communities associated with Halimeda opuntia (Linnaeus) Lamouroux (1816) and Sargassum polyceratium Montagne (1837) seaweeds in a tropical phytal ecosystem. Mar. Biol. 2016, 163, 1–13. [Google Scholar] [CrossRef]
- Diane, Z.M.; Gouvello, L.; Nel, R.; Harris, L.R.; Bezuidenhout, K. The response of sandy beach meiofauna to nutrients from sea turtle eggs. J. Exp. Mar. Bio. Ecol. 2017, 487, 94–105. [Google Scholar] [CrossRef]
- Ball, S.L.; Hebert, P.D.N.; Burian, S.K.; Webb, J.M. Biological identifications of mayflies (Ephemeroptera) using DNA barcodes. J. N. Am. Benthol. Soc. 2006, 24, 508–524. [Google Scholar] [CrossRef]
- Bonaglia, S.; Nascimento, F.J.A.; Bartoli, M.; Klawonn, I.; Brüchert, V. Meiofauna increases bacterial denitrification in marine sediments. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- World Register of Marine Species. Available online: www.marinespecies.org (accessed on 13 January 2017).
- Hugot, J.P.; Baujard, P.; Morand, S. Biodiversity in helminths and nematodes as a field of study: An overview. Nematology 2001, 3, 199–208. [Google Scholar] [CrossRef]
- Lambshead, P.J.D. Marine nematode biodiversity. In Nematology: Advances and perspectives Nematode Morphology, Physiology and Ecology; Chen, Z.X., Chen, S.Y., Dickson, D.W., Eds.; CABI Publishing: Wallingford, UK, 2004; pp. 436–467. [Google Scholar]
- Coomans, A. Present status and future of nematode systematics. Nematology 2002, 4, 573–582. [Google Scholar] [CrossRef]
- Castillo-Fernandez, D.; Lambshead, P.J.D. Revision of the genus Elzalia Gerlach, 1957 (Nematoda: Xyalidae) including three new species from an oil producing zone in the Gulf of Mexico, with a discussion of the sibling species problem. Bull. Br. Museum Natural Hist. Zool. Ser. 1990, 56, 63–71. [Google Scholar]
- De Jesús-Navarrete, A.; Herrera-Gómez, J. Vertical distribution and feeding types of nematodes from Chetumal Bay, Quintana Roo, Mexico. Estuaries 2002, 25, 1131–1137. [Google Scholar] [CrossRef]
- De Jesús-Navarrete, A. Diversity of nematoda in a Caribbean atoll: Banco Chinchorro, Mexico. Bull. Mar. Sci. 2003, 73, 47–56. [Google Scholar]
- De Jesús-Navarrete, A. Littoral free living nematode fauna of Socorro Island, Colima, Mexico. Hidrobiologica 2007, 17, 61–66. [Google Scholar]
- De Jesús-Navarrete, A. Nematodos de los arrecifes de Isla Mujeres y Banco Chinchorro, Quintana Roo, México. Rev. Biol. Mar. Oceanogr. 2007, 42, 193–200. [Google Scholar] [CrossRef]
- De Jesús-Navarrete, A. Distribución, abundancia y diversidad de los nematodos (Phylum Nematoda) bénticos de la Sonda de Campeche, México. Enero 1987. Rev. Biol. Trop. 1993, 41, 57–63. [Google Scholar]
- Comision Nacional para el Conocimiento y Uso de la Biodiversidad. Available online: https://www.gob.mx/conabio (accessed on 20 January 2020).
- De Jesús-Navarrete, A.; Herrera-Gómez, J. Nematofauna asociada a la zona urbana de la bahía de Chetumal, Quintana Roo, México. Rev. Biol. Trop. 1999, 47, 867–875. [Google Scholar]
- Gingold, R.; Mundo-Ocampo, M.; Holovachov, O.; Rocha-Olivares, A. The role of habitat heterogeneity in structuring the community of intertidal free-living marine nematodes. Mar. Biol. 2010, 157, 1741–1753. [Google Scholar] [CrossRef] [PubMed]
- Holovachov, O.; Tandingan De Ley, I.; Mundo-Ocampo, M.; Baldwin, J.G.; Rocha-Olivares, A.; De Ley, P. Nematodes from the Gulf of California. Part 1. The genera Ceramonema Cobb, 1920, Pselionema Cobb in Cobb, 1933 and Pterygonema Gerlach, 1954 (Nematoda: Ceramonematidae). Nematology 2008, 10, 347–373. [Google Scholar]
- Holovachov, O.; Mundo-Ocampo, M.; De Ley, I.T.; De Ley, P. Nematodes from the gulf of California. Part 2. Ceramonema nasobema sp. n. (nematoda: Ceramonematidae). Nematology 2008, 10, 835–844. [Google Scholar]
- Mundo-Ocampo, M.; Lambshead, P.J.D.; Debenham, N.; King, I.W.; De Ley, P.; Baldwin, J.G.; De Ley, I.T.; Rocha-Olivares, A.; Waumann, D.; Thomas, W.K.; et al. Biodiversity of littoral nematodes from two sites in the Gulf of California. Hydrobiologia 2007, 586, 179–189. [Google Scholar] [CrossRef]
- De Ley, P. Lost in worm space: Phylogeny and morphology as road maps to nematode diversity. Nematology 2002, 2, 9–16. [Google Scholar] [CrossRef]
- Powers, T. Nematode molecular diagnostics: From bands to barcodes. Annu. Rev. Phytopathol. 2004, 42, 367–383. [Google Scholar] [CrossRef]
- Bik, H.M. Let’s rise up to unite taxonomy and technology. PLoS Biol. 2017, 15, 4–7. [Google Scholar] [CrossRef]
- Leasi, F.; Sevigny, J.L.; Laflamme, E.M.; Artois, T.; Curini-Galletti, M.; de Jesus Navarrete, A.; Di Domenico, M.; Goetz, F.; Hall, J.A.; Hochberg, R.; et al. Biodiversity estimates and ecological interpretations of meiofaunal communities are biased by the taxonomic approach. Commun. Biol. 2018, 1, 1–12. [Google Scholar] [CrossRef]
- Derycke, S.; Remerie, T.; Vierstraete, A.; Backeljau, T.; Vanfleteren, J.; Vincx, M.; Moens, T. Mitochondrial DNA variation and cryptic speciation within the free-living marine nematode Pellioditis marina. Mar. Ecol. Prog. Ser. 2005, 300, 91–103. [Google Scholar] [CrossRef]
- De Ley, P.; Tandingan De Ley, I.; Morris, K.; Abebe, E.; Mundo-Ocampo, M.; Yoder, M.; Heras, J.; Waumann, D.; Rocha-Olivares, A.; Burr, A.H.J.; et al. An integrated approach to fast and informative morphological vouchering of nematodes for applications in molecular barcoding. Philos. Trans. R. Soc. London. Ser. B 2005, 360, 1945–1958. [Google Scholar] [CrossRef]
- Da Silva, N.R.R.; Da Silva, M.C.; Genevois, V.F.; Esteves, A.M.; De Ley, P.; Decraemer, W.; Rieger, T.T.; Dos Santos Correia, M.T. Marine nematode taxonomy in the age of DNA: The present and future of molecular tools to assess their biodiversity. Nematology 2010, 12, 661–672. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Gregory, T.R. The promise of DNA barcoding for taxonomy. Syst. Biol. 2005, 54, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Bhadury, P.; Austen, M.C.; Bilton, D.T.; Lambshead, P.J.D.; Rogers, A.D.; Smerdon, G.R. Development and evaluation of a DNA-barcoding approach for the rapid identification of nematodes. Mar. Ecol. Prog. Ser. 2006, 320, 1–9. [Google Scholar] [CrossRef]
- Bhadury, P.; Austen, M.C. Barcoding marine nematodes: An improved set of nematode 18S rRNA primers to overcome eukaryotic co-interference. Hydrobiologia 2010, 641, 245–251. [Google Scholar] [CrossRef]
- Fontaneto, D.; Flot, J.F.; Tang, C.Q. Guidelines for DNA taxonomy, with a focus on the meiofauna. Mar. Biodiv. 2015, 45, 433–451. [Google Scholar] [CrossRef]
- Blaxter, M.L. The promise of a DNA taxonomy. Philos. Trans. R. Soc. B 2004, 359, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Dayrat, B. Towards integrative taxonomy? Biol. J. Linn. Soc. 2005, 85, 407–415. [Google Scholar] [CrossRef]
- Pereira, T.J.; Fonseca, G.; Mundo-Ocampo, M.; Guilherme, B.C.; Rocha-Olivares, A. Diversity of free-living marine nematodes (Enoplida) from Baja California assessed by integrative taxonomy. Mar. Biol. 2010, 157, 1665–1678. [Google Scholar] [CrossRef] [PubMed]
- Leasi, F.; Andrade, S.C.D.S.; Norenburg, J. At least some meiofaunal species are not everywhere. Indication of geographic, ecological and geological barriers affecting the dispersion of species of Ototyphlonemertes (Nemertea, Hoplonemertea). Mol. Ecol. 2016, 25, 1381–1397. [Google Scholar] [CrossRef] [PubMed]
- Armenteros, M.; Rojas-Corzo, A.; Ruiz-Abierno, A.; Derycke, S.; Backeljau, T.; Decraemer, W. Systematics and DNA barcoding of free-living marine nematodes with emphasis on tropical desmodorids using nuclear SSU rDNA and mitochondrial COI sequences. Nematology 2014, 16, 979–989. [Google Scholar] [CrossRef]
- Macheriotou, L.; Guilini, K.; Bezerra, T.N.; Tytgat, B.; Nguyen, D.T.; Phuong Nguyen, T.X.; Noppe, F.; Armenteros, M.; Boufahja, F.; Rigaux, A.; et al. Metabarcoding free-living marine nematodes using curated 18S and CO1 reference sequence databases for species-level taxonomic assignments. Ecol. Evol. 2019, 9, 1211–1226. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Alcántara-Rodríguez, J.A.; Ciros-Pérez, J.; Gómez, A.; Hagiwara, A.; Galindo, K.H.; Jersabek, C.D.; Malekzadeh-Viayeh, R.; Leasi, F.; Lee, J.S.; et al. Fifteen species in one: Deciphering the Brachionus plicatilis species complex (Rotifera, Monogononta) through DNA taxonomy. Hydrobiologia 2017, 796, 39–58. [Google Scholar] [CrossRef]
- Tang, C.Q.; Leasi, F.; Obertegger, U.; Kieneke, A.; Barraclough, T.G.; Fontaneto, D. The widely used small subunit 18S rDNA molecule greatly underestimates true diversity in biodiversity surveys of the meiofauna. Proc. Natl. Acad. Sci. USA 2012, 109, 16208–16212. [Google Scholar] [CrossRef] [PubMed]
- Derycke, S.; Vanaverbeke, J.; Rigaux, A.; Backeljau, T.; Moens, T. Exploring the use of cytochrome oxidase c subunit 1 (COI) for DNA barcoding of free-living marine nematodes. PLoS ONE 2010, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bhadury, P.; Austen, M.C.; Bilton, D.T.; Lambshead, P.J.D.; Rogers, A.D.; Smerdon, G.R. Evaluation of combined morphological and molecular techniques for marine nematode (Terschellingia spp.) identification. Mar. Biol. 2008, 154, 509–518. [Google Scholar] [CrossRef]
- Floyd, R.; Abebe, E.; Papert, A.; Blaxter, M. Molecular barcodes for soil nematodes identification. Mol. Ecol. 2002, 11, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Derycke, S.; De Ley, P.; Tandingan De Ley, I.; Holovachov, O.; Rigaux, A.; Moens, T. Linking DNA sequences to morphology: Cryptic diversity and population genetic structure in the marine nematode Thoracostoma trachygaster (Nematoda, Leptosomatidae). Zool. Scr. 2010, 39, 276–289. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Ratnasingham, S.; DeWaard, J.R. Barcoding animal life: Cytochrome c oxidase subunit 1 divergences among closely related species. Proc. R. Soc. London. Ser. B 2003, 270, 96–99. [Google Scholar] [CrossRef]
- Kekkonen, M.; Hebert, P.D.N. DNA barcode-based delineation of putative species: Efficient start for taxonomic workflows. Mol. Ecol. Resour. 2014, 14, 706–715. [Google Scholar] [CrossRef]
- Puillandre, N.; Lambert, A.; Brouillet, S.; Achaz, G. ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Mol. Ecol. 2012, 21, 1864–1877. [Google Scholar] [CrossRef]
- Ratnasingham, S.; Hebert, P.D.N. A DNA-Based Registry for All Animal Species: The Barcode Index Number (BIN) System. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kapli, P.; Pavlidis, P.; Stamatakis, A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics 2013, 29, 2869–2876. [Google Scholar] [CrossRef] [PubMed]
- Traunspurger, W. The biology and ecology of lotic nematodes. Freshw. Biol. 2000, 44, 29–45. [Google Scholar] [CrossRef]
- Yoder, M.; Tandingan De Ley, I.; King, I.W.; Mundo-Ocampo, M.; Mann, J.; Blaxter, M.; Poiras, L.; De Ley, P. DESS: A versatile solution for preserving morphology and extractable DNA of nematodes. Nematology 2006, 8, 1–10. [Google Scholar] [CrossRef]
- Somerfield, P.J.; Warwick, R.M.; Moens, T. Meiofauna Techniques. In Methods for the study of marine benthos; Wiley Online Books: Chichester, UK, 2005; pp. 229–272. ISBN 9780470995129. [Google Scholar]
- Platt, H.M.; Warwick, R.M. Freeliving Marine Nematodes. Part 1: British Enoplids. Pictorial Key to World Genera and Notes for the Identification of British Species; Cambridge University Press, for the Linnean Society of London and the Estuarine and Brackish-water Sciences Association: Cambridge, UK, 1983; ISBN 0521254221. [Google Scholar]
- Platt, H.M.; Warwick, R.M. Free Livng Marine Nematodes: Part II British Chromadorids. Pictoral Key to World Genera and Notes for the Identification of British Species. Synopses of the British Fauna; Brill, E.J., Backhuys, W., Eds.; Linnean Society of London and the Estuarine and Brackish-water Sciences Association: Leiden, NY, USA, 1988. [Google Scholar]
- Warwick, R.M.; Platt, H.M.; Somerfield, P.J. Free Living Marine Nematodes: Pictorial Key to World Genera and Notes for the Identification of British Species. Part III, Monhysterids; Bames, R.S.K., Crothers, J.H., Eds.; Linnean Society of London and the Estuarine and Coastal Sciences Association by Field Studies Council: Dorchester, UK, 1998. [Google Scholar]
- Montero-Pau, J.; Gómez, A.; Muñoz, J. Application of an inexpensive and high-throughput genomic DNA extraction method for the molecular ecology of zooplanktonic diapausing eggs. Limnol. Oceanogr. Methods 2008, 6, 218–222. [Google Scholar] [CrossRef]
- Ivanova, N.V.; Grainger, C. Increased DNA barcode recovery using Platinum taq. CCDB Adv.: Methods Release 2006, 2, 1. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Hajibabaei, M.; DeWaard, J.R.; Ivanova, N.V.; Ratnasingham, S.; Dooh, R.T.; Kirk, S.L.; Mackie, P.M.; Hebert, P.D.N. Critical factors for assembling a high volume of DNA barcodes. Philos. Trans. R. Soc. B 2005, 360, 1959–1967. [Google Scholar] [CrossRef]
- Ivanova, N.V.; Dewaard, J.R.; Hebert, P.D.N. An inexpensive, automation-friendly protocol for recovering high-quality DNA. Mol. Ecol. Notes 2006, 6, 998–1002. [Google Scholar] [CrossRef]
- Ewing, B.; Green, P. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Res. 1998, 8, 186–194. [Google Scholar] [CrossRef]
- Ratnasingham, S.; Hebert, P.D.N. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Richly, E.; Leister, D. NUMTs in sequenced eukaryotic genomes. Mol. Biol. Evol. 2004, 21, 1081–1084. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Buhay, J.E.; Whiting, M.F.; Crandall, K.A. Many species in one: DNA barcoding overestimates the number of species when nuclear mitochondrial pseudogenes are coamplified. Proc. Natl. Acad. Sci. USA 2008, 105, 13486–13491. [Google Scholar] [CrossRef]
- Altschul, S.; Madden, T.; Schaffer, A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D. Gapped blast and psi-blast: A new generation of protein database search programs. FASEB J. 1998, 12, 3389–3402. [Google Scholar] [CrossRef]
- Hazir, C.; Giblin-Davis, R.M.; Keskin, N.; Ye, W.; Hazir, S.; Scheuhl, E.; Thomas, W.K. Diversity and distribution of nematodes associated with wild bees in Turkey. Nematology 2010, 12, 65–80. [Google Scholar]
- Jukes, T.H.; Cantor, C. Evolution of Protein Molecules. In Mammalian Protein Metabolism; Munro, H.N.E., Ed.; Academic Press: New York, NY, USA, 1969; pp. 21–132. [Google Scholar]
- Southern, R. Nemathelmaia, Kinorhyncha, and Chaetognatha. Proc. R. Irish Acad. 1914, 31, 1–80. [Google Scholar]
- Filipjev, I.N. Free-living marine nematodes of the Sevastopol area. Trudy Osob. Zool. Lab. Sebastop. Biol. Sta. 1921, II, 351–614. [Google Scholar]
- Velasco-Castrillón, A.; Stevens, M.I. Morphological and molecular diversity at a regional scale: A step closer to understanding Antarctic nematode biogeography. Soil Biol. Biochem. 2014, 70, 272–284. [Google Scholar] [CrossRef]
- Hauquier, F.; Leliaert, F.; Rigaux, A.; Derycke, S.; Vanreusel, A. Distinct genetic differentiation and species diversification within two marine nematodes with different habitat preference in Antarctic sediments. BMC Evol. Biol. 2017, 17, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Armenteros, M.; Ruiz-Abierno, A.; Decraemer, W. Revision of Desmodorinae and Spiriniinae (Nematoda: Desmodoridae) with redescription of eight known species. Eur. J. Taxon. 2014, 96, 1–32. [Google Scholar] [CrossRef]
- Castillo-Fernandez, D.; Decraemer, W. Cheironchus paravorax n. sp. and Cheironchus vorax Cobb, 1917 from the Campeche Sound, an oil producing zone in the Gulf of Mexico (Nemata: Selachinematidae). Bull. l’Institut R. des Sci. Nat. Belgique 1993, 63, 55–64. [Google Scholar]
- Holovachov, O.; De Ley, I.T.; Mundo-Ocamnpo, M.; Gingold, R.; De Ley, P. Nematodes from the Gulf of California. Part 3. Three new species of the genus Diplopeltoides Gerlach, 1962 (Nematoda: Diplopeltoididae) with overviews of the genera Diplopeltis Gerlach, 1962 and Diplopeltula Gerlach, 1950. Russ. J. Nematol. 2009, 17, 43–57. [Google Scholar]
- Witt, C.C.; Maliakal-Witt, S. Why are diversity and endemism Linked on islands? Ecography 2007, 30, 331–333. [Google Scholar] [CrossRef]
- Avó, A.P.; Daniell, T.J.; Neilson, R.; Oliveira, S.; Branco, J.; Adão, H. DNA Barcoding and morphological identification of benthic nematodes assemblages of estuarine intertidal sediments: Advances in molecular tools for biodiversity assessment. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Okimoto, R.; Macfarlene, J.L.; Clary, D.O.; Wolstenholme, D.R. The mitochondrial genomes of two nematodes, Caenorhabditis elegans and Ascaris suum. Genetics 1992, 130, 471–498. [Google Scholar]
- Ekrem, T.; Willassen, E.; Stur, E. A comprehensive DNA sequence library is essential for identification with DNA barcodes. Mol. Phylogenet. Evol. 2007, 43, 530–542. [Google Scholar] [CrossRef]
- Elsasser, S.C.; Floyd, R.; Hebert, P.D.N.; Schulte-Hostedde, A.I. Species identification of North American guinea worms (Nematoda: Dracunculus) with DNA barcoding. Mol. Ecol. Resour. 2009, 9, 707–712. [Google Scholar] [CrossRef]
- Prosser, S.W.J.; Velarde-Aguilar, M.G.; León-Règagnon, V.; Hebert, P.D.N. Advancing nematode barcoding: A primer cocktail for the cytochrome c oxidase subunit I gene from vertebrate parasitic nematodes. Mol. Ecol. Resour. 2013, 13, 1108–1115. [Google Scholar] [CrossRef]
- Leasi, F.; Norenburg, J.L. The necessity of DNA taxonomy to reveal cryptic diversity and spatial distribution of meiofauna, with a focus on nemertea. PLoS ONE 2014, 9, e104385. [Google Scholar] [CrossRef] [PubMed]
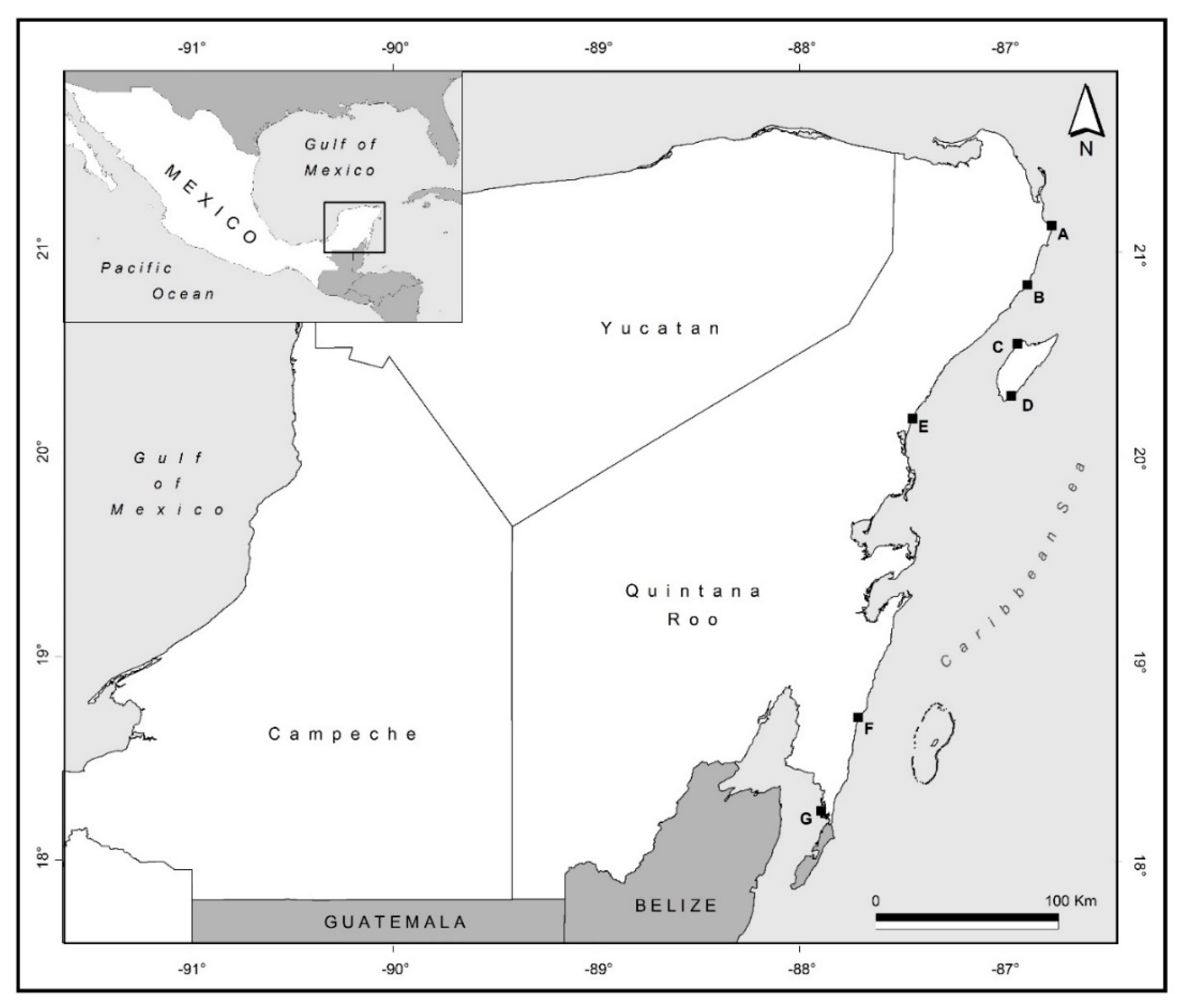
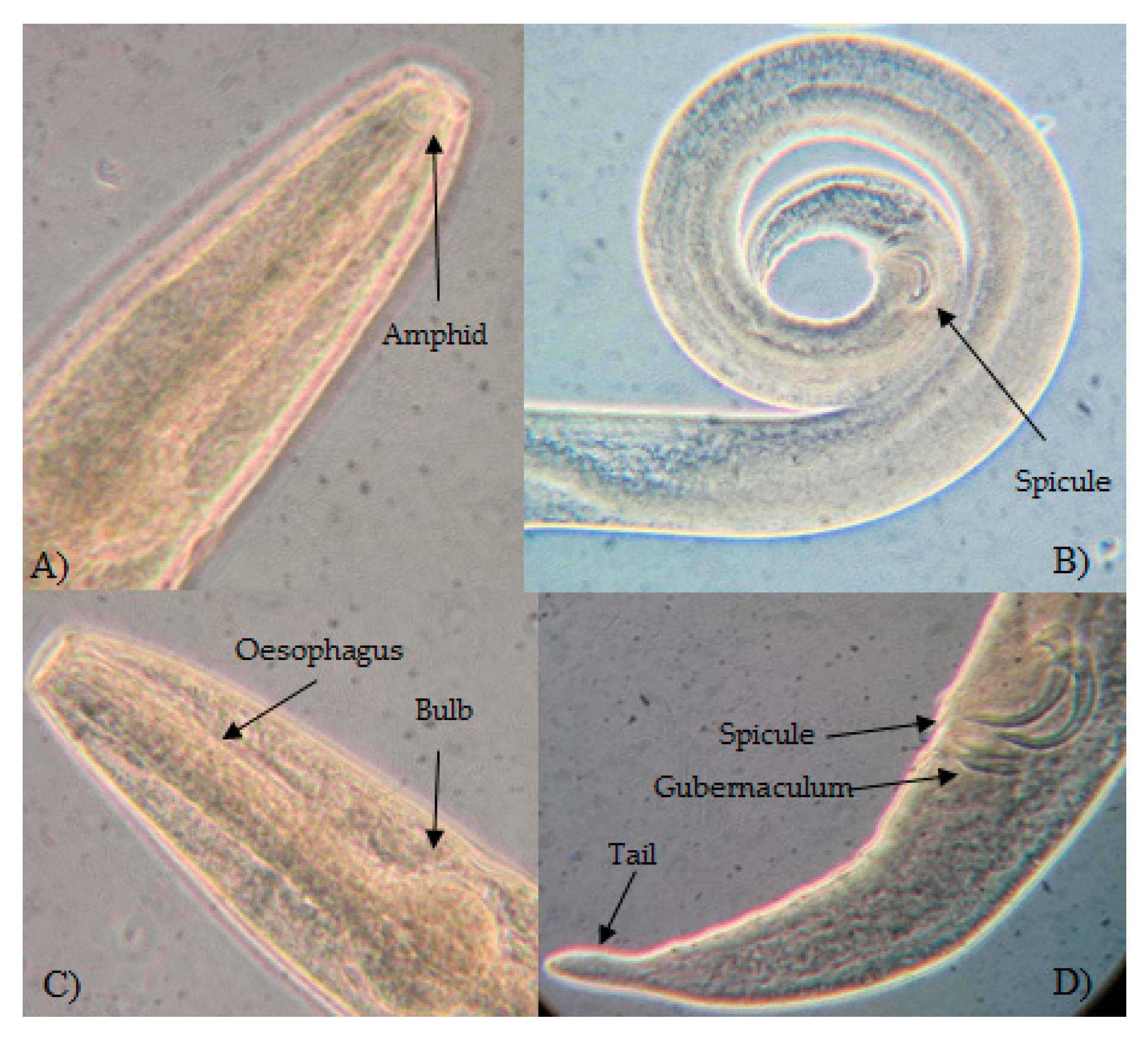
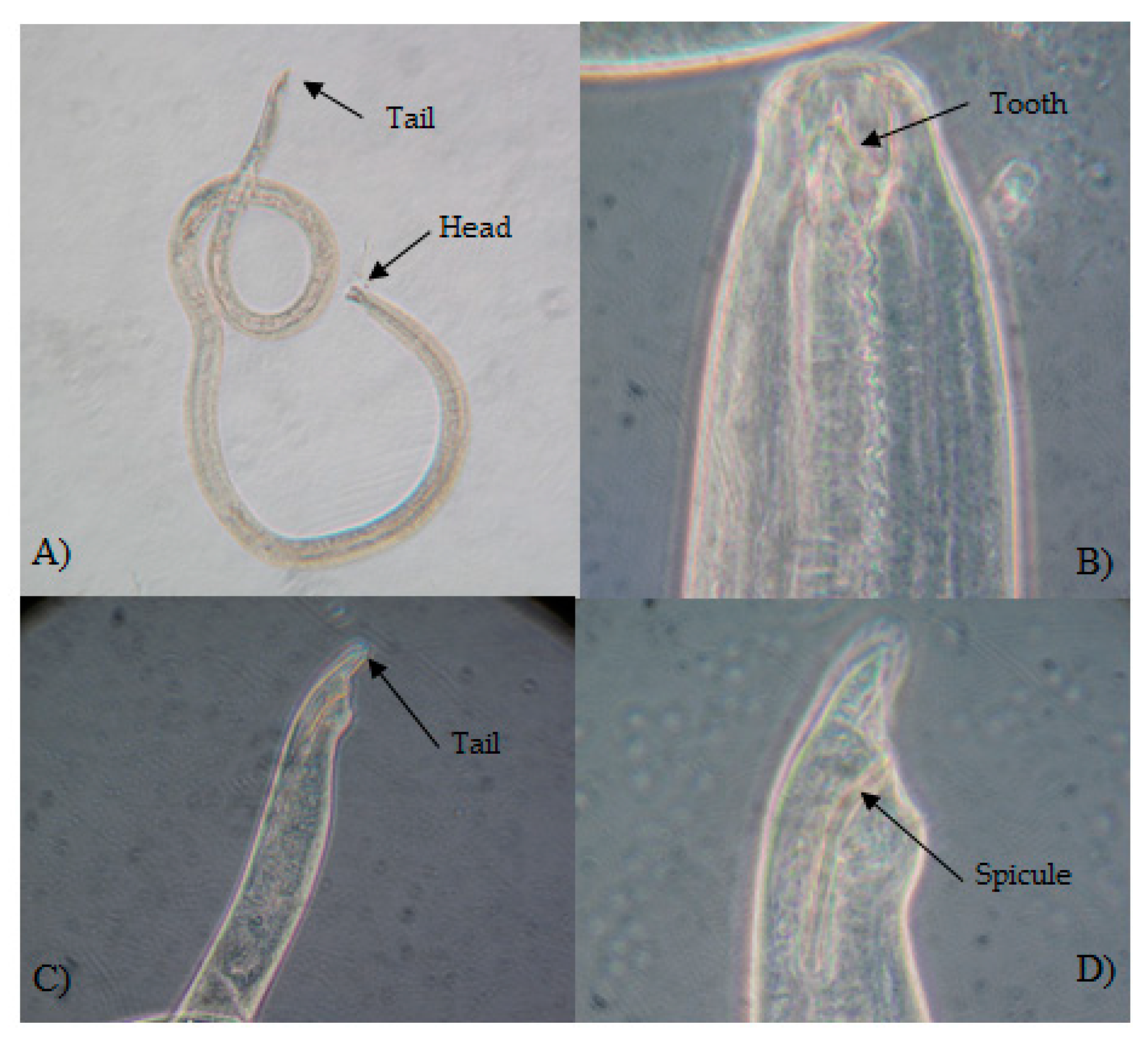
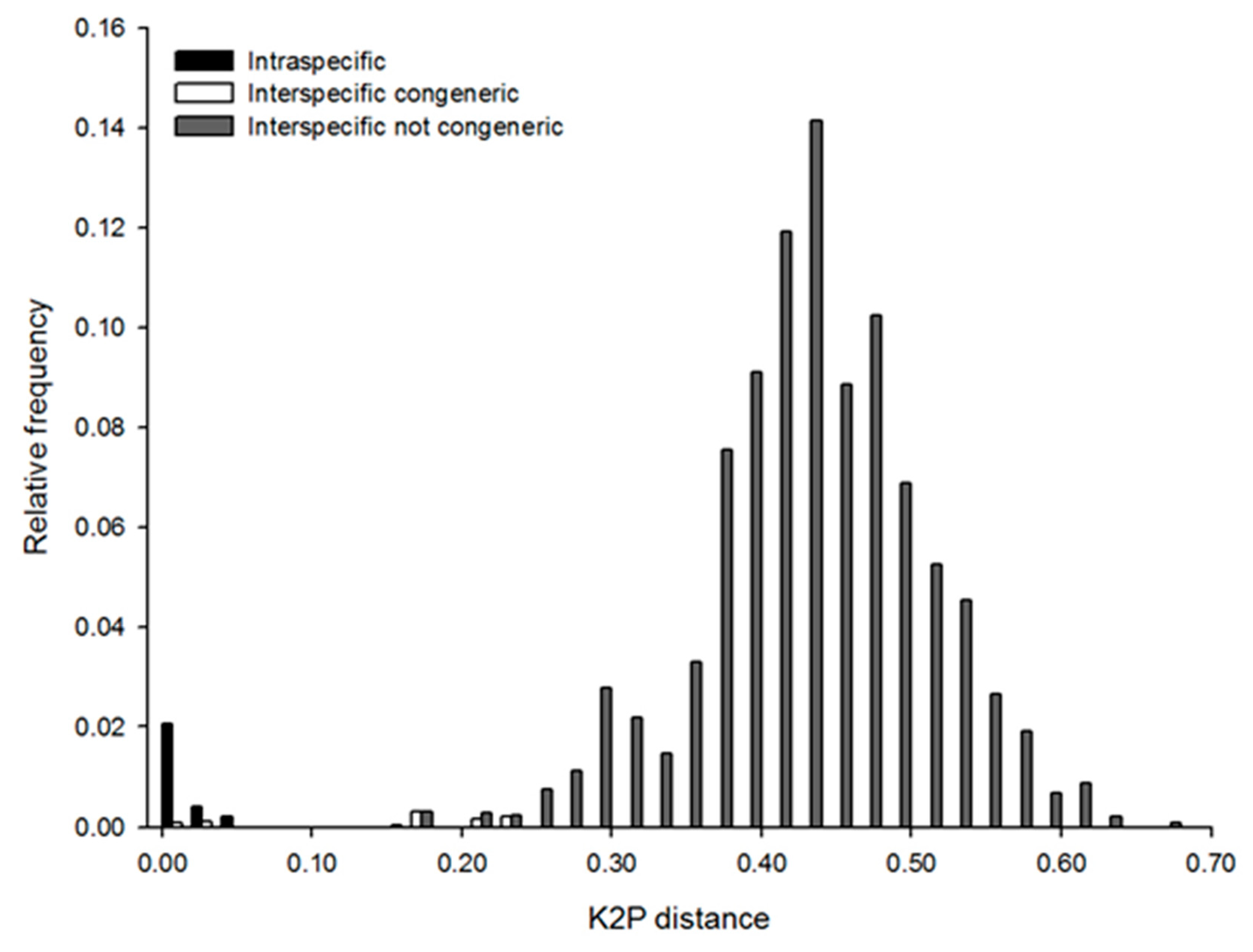
| Sites | Site ID | Lat | Long | Date of Sampling mm/yy/2011 |
|---|---|---|---|---|
| Cancún | A | 21.140 | −86.677 | 03/11 |
| Puerto Morelos | B | 20.508 | −86.525 | 03/11 |
| Cozumel (Playa Azul) | C | 20.548 | −86.929 | 03/11 |
| Cozumel (Punta sur) | D | 20.291 | −86.959 | 03/11 |
| Tulum | E | 20.395 | −87.315 | 03/11 |
| Mahahual | F | 18.708 | −87.712 | 09/11 |
| Xcalak | G | 18.2475 | −87.8944 | 09/11 |
| Taxonomy | Identified Specimens (n) | Sites | Sequences (n) | |
|---|---|---|---|---|
| Araeolaimida | ||||
| Odontophora bermudensis (Jensen & Gerlach, 1976) | 2 | D | 0 | |
| Chromadorida | ||||
| Actinonema sp.1 | 1 | B | 0 | |
| Chromadoridae sp.1 | 1 | G | 0 | |
| Chromadoridae sp.2 | 1 | E | 0 | |
| Chromadorida sp.1 | 2 | F | 2 | |
| Chromadorita sp.1 | 1 | D | 0 | |
| Rhips sp.1 | 9 | A; B | 6 | |
| Prochromadorella sp.1 | 4 | E | 0 | |
| Desmodorida | ||||
| Catanema sp.1 | 1 | D | 1 | |
| Epsilonema sp.1 | 6 | C; E | 5 | |
| Metachromadora sp.1 | 2 | F | 2 | |
| Monoposthia mirabilis (Schulz, 1932) | 10 | A; B; D | 8 | |
| Onyx litorale (Schulz, 1938) | 2 | D; F | 2 | |
| Spirinia sp.1 | 11 | D; F; G | 9 | |
| Spirinia sp.2 | 7 | G | 6 | |
| Enoplida | ||||
| Anticomidae sp.1 | 3 | B | 3 | |
| Eurystomina sp.1 | 1 | D | 1 | |
| Enoploides sp.1 | 1 | D | 1 | |
| Enoploides sp.2 | 3 | C | 2 | |
| Enoploides gryphus (Wieser& Hopper, 1967) | 2 | E | 2 | |
| Epacanthion sp.1 | 5 | D | 0 | |
| Halalaimus sp.1 | 6 | B | 2 | |
| Metaparoncholaimus sp.1 | 1 | E | 0 | |
| Oncholaimus sp.1 | 9 | E | 0 | |
| Pontonema cf. simile (Southern, 1914) Filipjev, 1921 | 1 | E | 1 | |
| Proplatycoma fleurdelis (Hope, 1988) | 10 | C; B; E | 1 | |
| Monhysterida | ||||
| Monhysteridae sp.1 | 1 | F | 1 | |
| Xyala striata (Cobb, 1920) | 2 | D | 1 | |
| Terschellingia longicaudata (de Man, 1907) | 1 | G | 0 | |
| Plectida | ||||
| Haliplectus bickneri (Chitwood, 1956) | 1 | B | 1 | |
| ALGORITHMIC APPROACHES | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BIN System | mPTP Model | ABGD | ||||||||||||||||||||||||
| p-value | 0.001 | |||||||||||||||||||||||||
| Subst. model | K2P | JC69 | ||||||||||||||||||||||||
| Prior intraspecific divergence (P) | 0.0010 | 0.0017 | 0.0028 | 0.0046 | 0.0077 | 0.129 | 0.0010 | 0.0017 | 0.0028 | 0.0046 | 0.0077 | 0.129 | ||||||||||||||
| Partition | I | R | I | R | I | R | I | R | I | R | I | R | I | R | I | R | I | R | I | R | I | R | I | R | ||
| EiEs | 22 | 20 | 20 | 25 | 20 | 21 | 20 | 21 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 25 | 20 | 21 | 20 | 21 | 20 | 20 | 20 | 20 | 20 | 20 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Arce, A.; De Jesús-Navarrete, A.; Leasi, F. DNA Barcoding for Delimitation of Putative Mexican Marine Nematodes Species. Diversity 2020, 12, 107. https://doi.org/10.3390/d12030107
Martínez-Arce A, De Jesús-Navarrete A, Leasi F. DNA Barcoding for Delimitation of Putative Mexican Marine Nematodes Species. Diversity. 2020; 12(3):107. https://doi.org/10.3390/d12030107
Chicago/Turabian StyleMartínez-Arce, Arely, Alberto De Jesús-Navarrete, and Francesca Leasi. 2020. "DNA Barcoding for Delimitation of Putative Mexican Marine Nematodes Species" Diversity 12, no. 3: 107. https://doi.org/10.3390/d12030107
APA StyleMartínez-Arce, A., De Jesús-Navarrete, A., & Leasi, F. (2020). DNA Barcoding for Delimitation of Putative Mexican Marine Nematodes Species. Diversity, 12(3), 107. https://doi.org/10.3390/d12030107






