The Minute Alga Schizocladia ischiensis (Schizocladiophyceae, Ochrophyta) Isolated by Germling Emergence from 24 m Depth off Rhodes (Greece)
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Habitat
3.2. Accompanying Species
3.3. Sequences
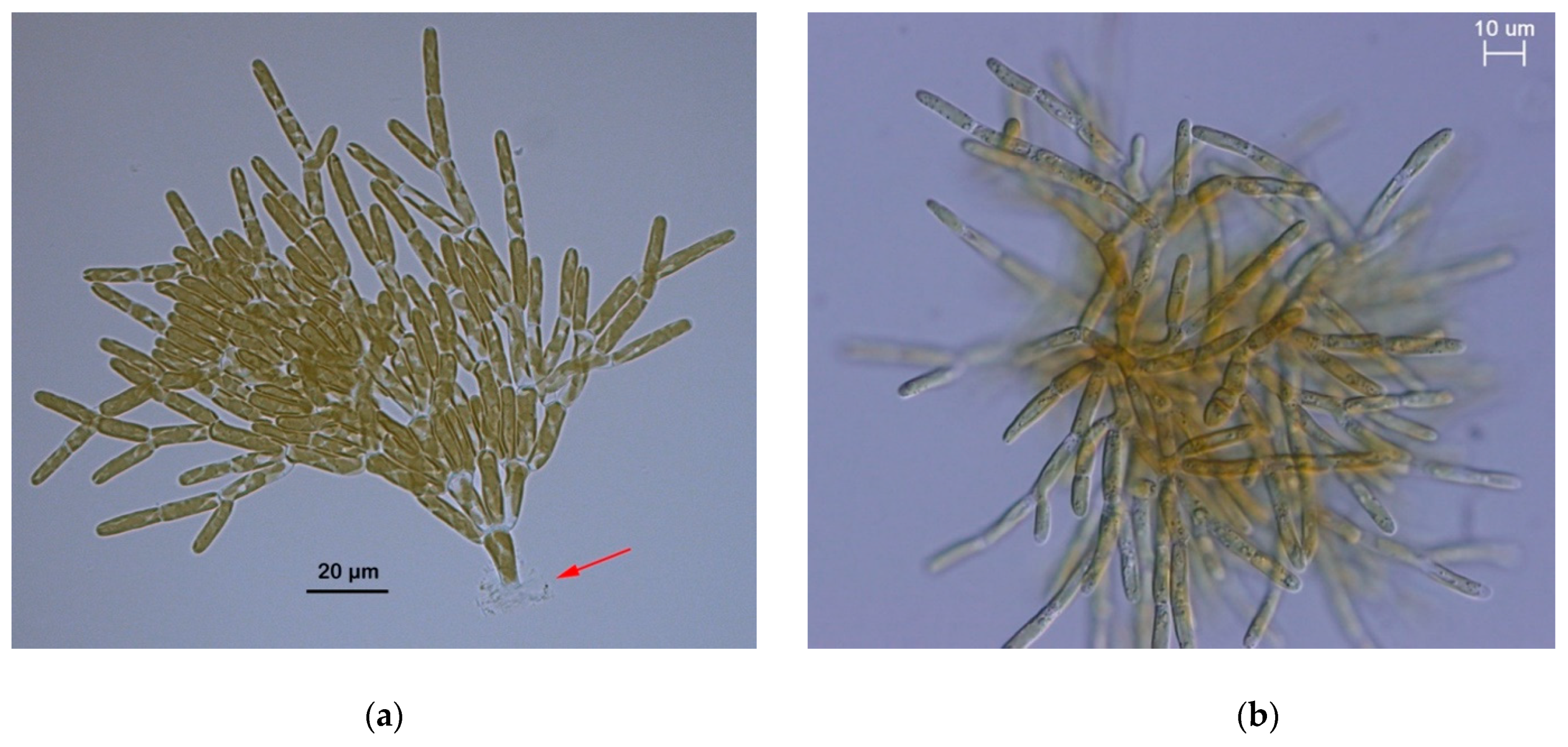
3.4. Morphology
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Lasram, F.B.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediterranean Sea: Estimates, patterns, and threats. PloS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [PubMed]
- Bartolo, A.; Zammit, G.; Peters, A.F.; Küpper, F.C. DNA barcoding of macroalgae in the Mediterranean Sea. Bot. Mar. 2020, (in press).
- De Jode, A.; David, R.; Haguenauer, A.; Cahill, A.E.; Erga, Z.; Guillemain, D.; Sartoretto, S.; Rocher, C.; Selva, M.; Le Gall, L.; et al. From seascape ecology to population genomics and back. Spatial and ecological differentiation among cryptic species of the red algae Lithophyllum stictiforme/L. cabiochiae, main bioconstructors of coralligenous habitats. Mol. Phylogenet. Evol. 2019, 137, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Pezzolesi, L.; Pena, L.V.; Le Gall, P.W.; Gabrielson, S.; Kaleb, J.R.; Hughey, G.; Rodondi, J.J.; Hernandez-Kantun, A.; Falace, D.; Basso, D.; et al. Mediterranean Lithophyllum stictiforme (Corallinales, Rhodophyta) is a genetically diverse species complex: Implications for species circumscription, biogeography and conservation of coralligenous habitats. J. Phycol. 2019, 55, 473–492. [Google Scholar] [CrossRef]
- Tsiamis, K.; Panayotidis, P. Seaweeds of the Greek coasts. Rhodophyceae: Ceramiales. Acta Adriat. 2016, 57, 227–250. [Google Scholar]
- Ballesteros, E. Mediterranean coralligenous assemblages: A synthesis of present knowledge. In Oceanography and Marine Biology - An Annual Review, Vol 44; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; Crc Press-Taylor & Francis Group: Boca Raton, FL, USA, 2006; pp. 123–195. [Google Scholar]
- Basso, D.; Babbini, L.; Ramos-Espla, A.A.; Salomidi, M. Mediterranean Rhodolith Beds. In Rhodolith/Maerl Beds: A Global Perspective; RiosmenaRodriguez, R., Nelson, W., Aguirre, J., Eds.; Springer International Publishing Ag: Cham, the Netherlands, 2017; pp. 281–298. [Google Scholar]
- Hereu, B.; Mangialajo, L.; Enric, B.; Thibaut, T. On the occurrence, structure and distribution of deep-water Cystoseira (Phaeophyceae) populations in the Port-Cros National Park (northwestern Mediterranean). Eur. J. Phycol. 2008, 43, 263–273. [Google Scholar] [CrossRef]
- Thibaut, T.; Pinedo, S.; Torras, X.; Ballesteros, E. Long-term decline of the populations of Fucales (Cystoseira spp. and Sargassum spp.) in the Albères coast (Frame, North-western Mediterranean). Mar. Pollu. Bull. 2005, 50, 1472–1489. [Google Scholar] [CrossRef]
- Joher, S.; Ballesteros, E.; Cebrian, E.; Sanchez, N.; Rodriguez-Prieto, C. Deep-water macroalgal-dominated coastal detritic assemblages on the continental shelf off Mallorca and Menorca (Balearic Islands, Western Mediterranean). Bot. Mar. 2012, 55, 485–497. [Google Scholar] [CrossRef]
- Žuljevic, A.; Peters, A.F.; Nikolic, B.; Antolic, V.; Despalatovic, M.; Cvitkovic, I.; Isajlovic, I.; Mihanovic, H.; Matijevic, S.; Shewring, D.M.; et al. The Mediterranean deep-water kelp Laminaria rodriguezii is an endangered species in the Adriatic Sea. Mar. Biol. 2016, 163, 69. [Google Scholar]
- Küpper, F.C.; Tsiamis, K.; Johansson, N.R.; Peters, A.F.; Salomidi, M.; Manousakis, L.; Kallergis, M.; Graham, M.H.; Kinlan, B.; Mystikou, A.; et al. New records of the rare deep-water alga Sebdenia monnardiana (Rhodophyta) and the alien Dictyota cyanoloma (Phaeophyceae ) and the unresolved case of deep-water kelp in the Ionian and Aegean Seas (Greece). Bot. Mar. 2019, 62, 577–586. [Google Scholar] [CrossRef]
- Aplikioti, M.; Louizidou, P.; Mystikou, A.; Marcou, M.; Stavrou, P.; Kalogirou, S.; Tsiamis, K.; Panayotidis, P.; Küpper, F.C. Further expansion of the alien seaweed Caulerpa taxifolia var. distichophylla (Sonder) Verlaque, Huisman & Procacini (Ulvophyceae, Bryopsidales) in the Eastern Mediterranean Sea. Aquat. Invasions 2016, 11, 11–20. [Google Scholar]
- Kawai, H.; Hanyuda, T.; Draisma, S.G.A.; Müller, D.G. Molecular phylogeny of Discosporangium mesarthrocarpum (Phaeophyceae) with a reinstatement of the order Discosporangiales. J. Phycol. 2007, 43, 186–194. [Google Scholar] [CrossRef]
- Alongi, G.; Cormaci, M.; Furnari, G. Verosphacela silvae sp. nov. (Onslowiaceae, Phaeophyceae) from the Mediterranean Sea. Phycol. Res. 2007, 55, 42–46. [Google Scholar] [CrossRef]
- Peters, A.F.; Couceiro, L.; Tsiamis, K.; Küpper, F.C.; Valero, M. Barcoding of cryptic stages of marine brown algae isolated from incubated substratum reveals high diversity in Acinetosporaceae (Ectocarpales). Cryptogam. Algol. 2015, 36, 3–29. [Google Scholar] [CrossRef]
- Montecinos, A.E.; Couceiro, L.; Peters, A.F.; Desrut, A.; Valero, M.; Guillemin, M.L. Species delimitation and phylogeographic analyses in the Ectocarpus subgroup siliculosi (Ectocarpales, Phaeophyceae). J. Phycol. 2017, 53, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Müller, D.G.; Ramírez, M.E. Filamentous brown algae from the Juan Fernandez Archipelago (Chile): Contribution of laboratory culture techniques to a phytogeographic survey. Bot. Mar. 1994, 37, 205–211. [Google Scholar] [CrossRef]
- Ramirez, M.E.; Müller, D.G. New records of benthic marine algae from Easter Island. Bot. Mar. 1991, 34, 133–137. [Google Scholar] [CrossRef]
- Robuchon, M.; Couceiro, L.; Peters, A.F.; Destombe, C.; Valero, M. Examining the bank of microscopic stages in kelps using culturing and barcoding. Eur. J. Phycol. 2014, 49, 128–133. [Google Scholar] [CrossRef]
- West, J.A.; Scott, J.L.; West, K.A.; Karsten, U.; Clayden, S.L.; Saunders, G.W. Rhodachlya madagascarensis gen. et sp nov.: A distinct acrochaetioid represents a new order and family (Rhodachlyales ord. nov., Rhodachlyaceae fam. nov.) of the Florideophyceae (Rhodophyta). Phycologia 2008, 47, 203–212. [Google Scholar] [CrossRef]
- Couceiro, L.; Le Gac, M.; Hunsperger, H.M.; Mauger, S.; Destombe, C.; Cock, J.M.; Ahmed, S.; Coelho, S.M.; Valero, M.; Peters, A.F. Evolution and maintenance of haploid-diploid life cycles in natural populations: The case of the marine brown alga Ectocarpus. Evolution 2015, 69, 1808–1822. [Google Scholar] [CrossRef]
- Küpper, F.C.; Peters, A.F.; Shewring, D.M.; Sayer, M.D.J.; Mystikou, A.; Brown, H.; Azzopardi, E.; Dargent, O.; Strittmatter, M.; Brennan, D.; et al. Arctic marine phytobenthos of northern Baffin Island. J. Phycol. 2016, 52, 532–549. [Google Scholar]
- Muñoz, L.A.; Wynne, M.J.; Kamiya, M.; West, J.A.; Küpper, F.C.; Ramirez, M.E.; Peters, A.F. Reinstatement of Phrix (Delesseriaceae, Rhodophyta) based on DNA sequence analyses and morpho-anatomical evidence. Phytotaxa 2018, 334, 215–224. [Google Scholar] [CrossRef]
- Kawai, H.; Maeba, S.; Sasaki, H.; Okuda, K.; Henry, E.C. Schizocladia ischiensis: A new filamentous marine chromophyte belonging to a new class, Schizocladiophyceae. Protist 2003, 154, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Coelho, S.M.; Scornet, D.; Rousvoal, S.; Peters, N.; Dartevelle, L.; Peters, A.F.; Cock, J.M. How to cultivate Ectocarpus. Cold Spring Harb. Protoc. 2012, 2012, 258–261. [Google Scholar] [CrossRef] [PubMed]
- KU-MACC. Kobe University Macro-Algal Culture Collection. 2020. Available online: http://ku-macc.nbrp.jp/ (accessed on 13 March 2020).
- BM. British Museum - Natural History. Herbarium. 2020. Available online: https://www.nhm.ac.uk/research-curation/scientific-resources/collections/botanical-collections/index.html (accessed on 13 March 2020).
- PC. Muséum National d’Histoire Naturelle, Paris - Cryptogamie. 2020. Available online: https://www.mnhn.fr/en/collections/collection-groups/botany/algae (accessed on 13 March 2020).
- SAP, Herbarium, Graduate School of Science, Hokkaido University. 2020. Available online: http://sap.museum.hokudai.ac.jp/ (accessed on 13 March 2020).
- ABDUK., Herbarium of the Museum of the University of Aberdeen. 2020. Available online: https://www.abdn.ac.uk/museums/collections/natural-history.php#herbarium (accessed on 13 March 2020).
- HCMR., Hellenic Centre for Marine Research. 2020. Available online: www.hcmr.gr (accessed on 13 March 2020).
- Gachon, C.M.M.; Strittmatter, M.; Müller, D.G.; Kleinteich, J.; Küpper, F.C. Differential host susceptibility to the marine oomycete pathogen Eurychasma dicksonii detected by real time PCR: Not all algae are equal. Appl. Environ. Microbiol. 2009, 75, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A. Se-Al Alignment Editor; University of Oxford: Oxford, UK, 2002. [Google Scholar]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 2000, 7, 203–214. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide To Methods And Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Peters, A.F.; Burkhardt, E. Systematic position of the kelp endophyte Laminarionema elsbetiae (Ectocarpales sensu lato, Phaeophyceae) inferred from nuclear ribosomal DNA sequences. Phycologia 1998, 37, 114–120. [Google Scholar] [CrossRef]
- Yang, E.C.; Peters, A.F.; Kawai, H.; Stern, R.; Hanyuda, T.; Barbara, I.; Muller, D.G.; Strittmatter, M.; van Reine, W.F.P.; Küpper, F.C. Ligulate Desmarestia (Desmarestiales, Phaeophyceae) revisited: D. japonica sp. nov. and D. dudresnayi differ from D. ligulata. J. Phycol. 2014, 50, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Peters, A.F.; Scornet, D.; Müller, D.G.; Kloareg, B.; Cock, J.M. Inheritance of organelles in artificial hybrids of the isogamous multicellular chromist alga Ectocarpus siliculosus (Phaeophyceae). Eur. J. Phycol. 2004, 39, 235–242. [Google Scholar] [CrossRef]
- Peters, A.F.; Ramírez, M.E. Molecular phylogeny of small brown algae, with special reference to the systematic position of Caepidium antarcticum (Adenocystaceae, Ectocarpales). Cryptogam. Algol. 2001, 22, 187–200. [Google Scholar] [CrossRef]
- Yoon, H.S.; Hackett, J.D.; Bhattacharya, D. A single origin of the peridinin- and fucoxanthin-containing plastids in dinoflagellates through tertiary endosymbiosis. PNAS 2002, 99, 11724–11729. [Google Scholar] [CrossRef] [PubMed]
- Draisma, S.G.A.; van Reine, W.F.P.; Kawai, H. A revised classification of the Sphacelariales (Phaeophyceae) inferred from a psbC and rbcL based phylogeny. Eur. J. Phycol. 2010, 45, 308–326. [Google Scholar] [CrossRef]
- AlgaeBase. Available online: http://www.algaebase.org (accessed on 29 January 2020).
- Kawai, H.; Hanyuda, T.; Yamagishi, T.; Kai, A.; Lane, C.; McDevit, D.; Küpper, F.C.; Saunders, G.W. Reproductive morphology and DNA sequences of the brown alga Platysiphon verticillatus support the new combination P. glacialis. J. Phycol. 2015, 51, 910–917. [Google Scholar] [CrossRef] [PubMed]
- West, J.A.; Hansen, G.I.; Hanyuda, T.; Zuccarello, G.C. Flora of drift plastics: A new red algal genus, Tsunamia transpacifica (Stylonematophyceae) from Japanese tsunami debris in the northeast Pacific Ocean. Algae 2016, 31, 289–301. [Google Scholar] [CrossRef]
- Hamsher, S.E.; Saunders, G.W. A floristic survey of marine tube-forming diatoms reveals unexpected diversity and extensive co-habitation among genetic lines of the Berkeleya rutilans complex (Bacillariophyceae). Eur. J. Phycol. 2014, 49, 47–59. [Google Scholar] [CrossRef]
- Ravizza, M.; Hallegraeff, G. Environmental conditions influencing growth rate and stalk formation in the estuarine diatom Licmophora flabellata (Carmichael ex Greville) C. Agardh. Diatom Res. 2015, 30, 197–208. [Google Scholar] [CrossRef]
- Zuccarello, G.C.; Yoon, H.S.; Kim, H.; Sun, L.; de Goer, S.L.; West, J.A. Molecular phylogeny of the upright Erythropeltidales (Compsogonophyceae, Rhodophyta): Multiple cryptic lineages of Erythrotrichia carnea. J. Phycol. 2011, 47, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Tsirintanis, K.; Sini, M.; Doumas, O.; Trygonis, V.; Katsanevakis, S. Assessment of grazing effects on phytobenthic community structure at shallow rocky reefs: An experimental field study in the North Aegean Sea. J. Exp. Mar. Biol. Ecol. 2018, 503, 31–40. [Google Scholar] [CrossRef]
- Garbary, D.J.; Kim, K.Y.; Klinger, T.; Duggins, D. Red algae as hosts for endophytic kelp gametophytes. Mar. Biol. 1999, 135, 35–40. [Google Scholar] [CrossRef]
- Chapman, A.R.O. Reproduction, recruitment and mortality in 2 species of Laminaria in southwest Nova Scotia. J. Exp. Mar. Biol. Ecol. 1984, 78, 99–109. [Google Scholar] [CrossRef]


| Primer Name | Marker | Position (Last nt) | Direction | Sequence 5’-3’ | Reference |
|---|---|---|---|---|---|
| NS1F | nrSSU | 35 | F | GTAGTCATATGCTTGTCTC | [36] |
| NS4R | nrSSU | 1108 | R | CTTCCGTCAATTCCTTTAAG | [36] |
| AFP2F | nrSSU | 1268 | F | AGCTCTTTCTTGATTCTATG | [37] |
| AFP1R | nrSSU | 1796 | R | GGTAATGATCCTTCCGCAG | New primer |
| rbcL77F | rbcL | 95 | F | TGGGNTAYTGGGATGCTGA | [38] |
| rbcL952R | rbcL | 952 | R | CATACGCATCCATTTACA | [14], as Ral-R952 |
| rbcLP1F | rbcL | 965 | F | GKGTWATTTGTAARTGGATGCG | [14] |
| rbcL1273F | rbcL | 1273 | F | GTGCGACAGCTAACCGTG | [39] |
| rbcS139R | rbcS | 139 | R | AGACCCCATAATTCCCAATA | [40] |
| psaA130F | psaA | 130 | F | AAACWACWACTTGGATTTGGAA | [41] |
| psaA940R | psaA | 940 | R | TATGDCCAATWCCCCAATT | [41] |
| psaA870F | psaA | 870 | F | GGNGGWYTATGGTTAAGTGA | [41] |
| psaA1760R | psaA | 1760 | R | CCTCTWCCWGGWCCATCRCAWGG | [41] |
| psbCF | psbC | 37 | F | GTGGAAACGCCCTTTAATA | [42] |
| psbCmidR | psbC | 710 | R | CCYCCWACDARATCTTCCATATTATC | [42] |
| Marker | Length of Examined Sequence (bp) | Identity with Closest Hit (%) | Query Cover (%) | Identity (bp) | Not Identical (bp) | Indels (bp) | Accession of Closest Hit | Comment |
|---|---|---|---|---|---|---|---|---|
| SSU-5P | 845 | 100 | 100 | 845/845 | 0 | 0 | MN994274 | - |
| SSU-3P | 488 | 100 | 100 | 488/488 | 0 | 0 | MN994274 | - |
| rbcL | 1372 | 99.42 | 100 | 1364/1372 | 8 | 0 | MN996275 | 7 synonymous, 1 nonsynonymous Ser > Ala |
| Rubisco spacer | 80 | 98 | 100 | 80/82 | 2 | 2 | AB085615 | An indel of 2 bp length |
| psaA | 1566 | 99.23 | 100 | 1554/1566 | 12 | 0 | AB908091 | 11 synonymous, 1 nonsynonymous Ile > Leu |
| psbC-5P | 616 | 99.84 | 100 | 615/616 | 1 | 0 | AB908094 | 1 synonymous |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizouli, A.; Küpper, F.C.; Louizidou, P.; Mogg, A.O.M.; Azzopardi, E.; Sayer, M.D.J.; Kawai, H.; Hanyuda, T.; Peters, A.F. The Minute Alga Schizocladia ischiensis (Schizocladiophyceae, Ochrophyta) Isolated by Germling Emergence from 24 m Depth off Rhodes (Greece). Diversity 2020, 12, 102. https://doi.org/10.3390/d12030102
Rizouli A, Küpper FC, Louizidou P, Mogg AOM, Azzopardi E, Sayer MDJ, Kawai H, Hanyuda T, Peters AF. The Minute Alga Schizocladia ischiensis (Schizocladiophyceae, Ochrophyta) Isolated by Germling Emergence from 24 m Depth off Rhodes (Greece). Diversity. 2020; 12(3):102. https://doi.org/10.3390/d12030102
Chicago/Turabian StyleRizouli, Anastasia, Frithjof C. Küpper, Paraskevi Louizidou, Andrew O. M. Mogg, Elaine Azzopardi, Martin D. J. Sayer, Hiroshi Kawai, Takeaki Hanyuda, and Akira F. Peters. 2020. "The Minute Alga Schizocladia ischiensis (Schizocladiophyceae, Ochrophyta) Isolated by Germling Emergence from 24 m Depth off Rhodes (Greece)" Diversity 12, no. 3: 102. https://doi.org/10.3390/d12030102
APA StyleRizouli, A., Küpper, F. C., Louizidou, P., Mogg, A. O. M., Azzopardi, E., Sayer, M. D. J., Kawai, H., Hanyuda, T., & Peters, A. F. (2020). The Minute Alga Schizocladia ischiensis (Schizocladiophyceae, Ochrophyta) Isolated by Germling Emergence from 24 m Depth off Rhodes (Greece). Diversity, 12(3), 102. https://doi.org/10.3390/d12030102






