Filling the Gap and Improving Conservation: How IUCN Red Lists and Historical Scientific Data Can Shed More Light on Threatened Sharks in the Italian Seas
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
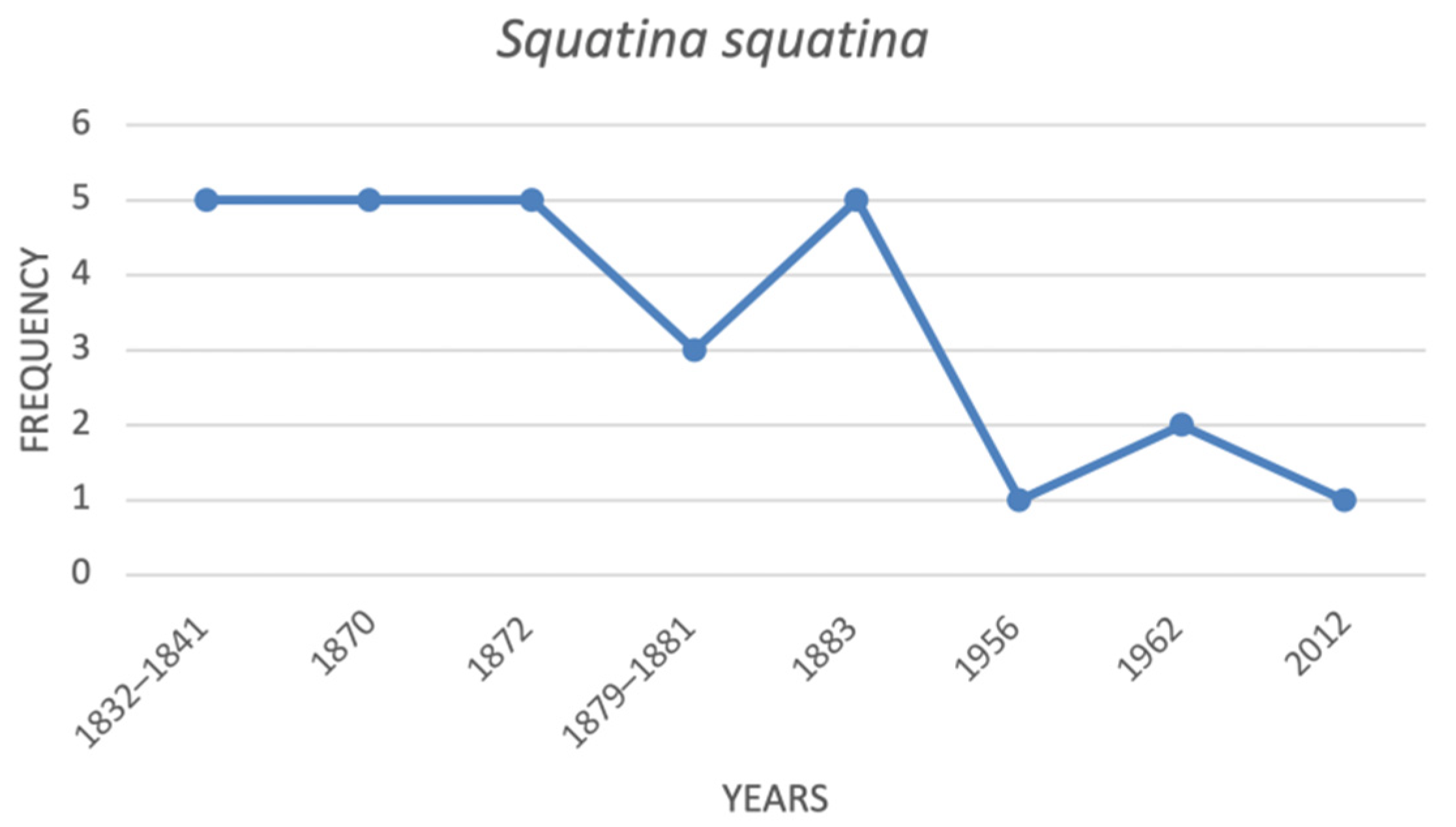
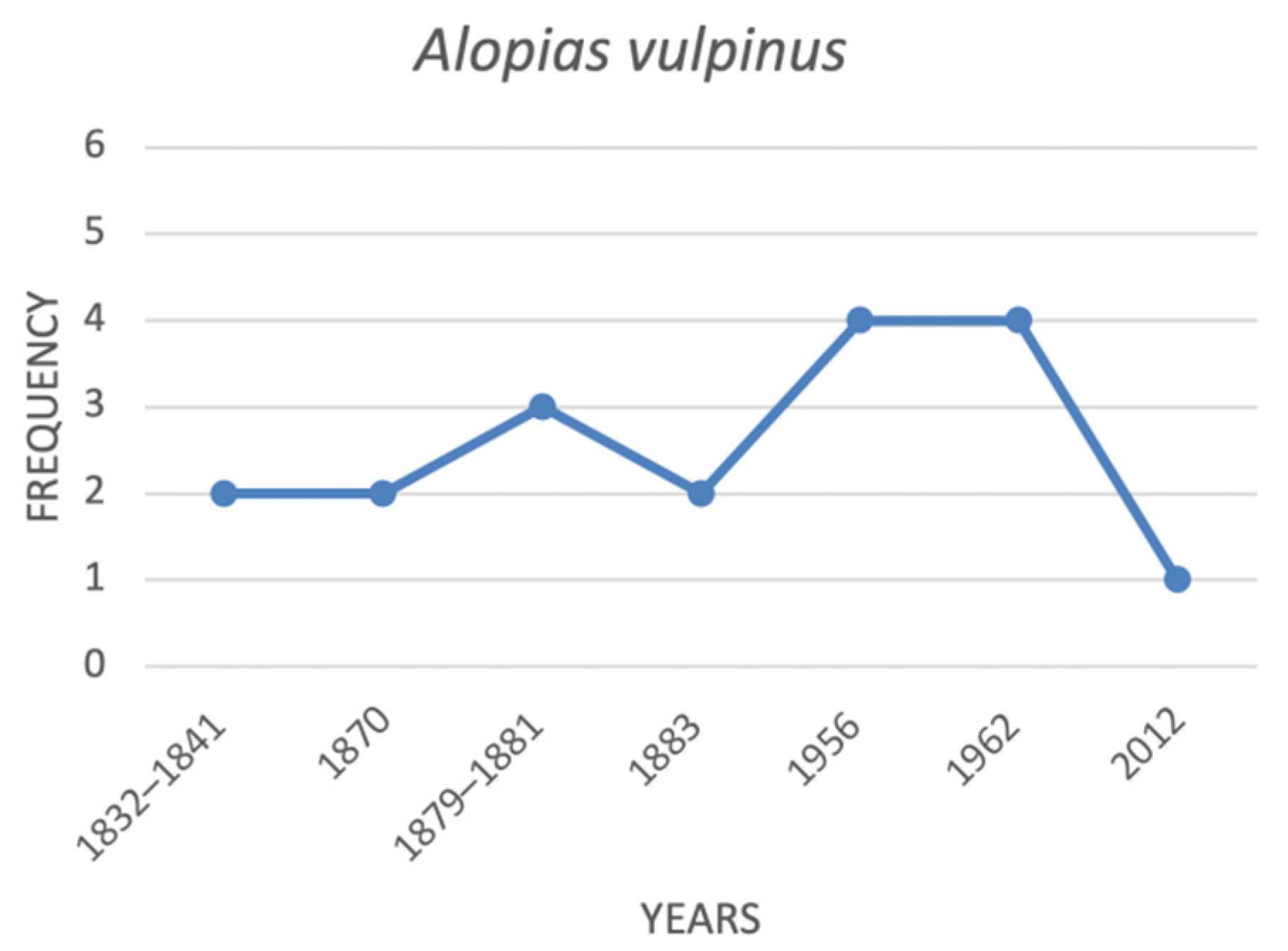
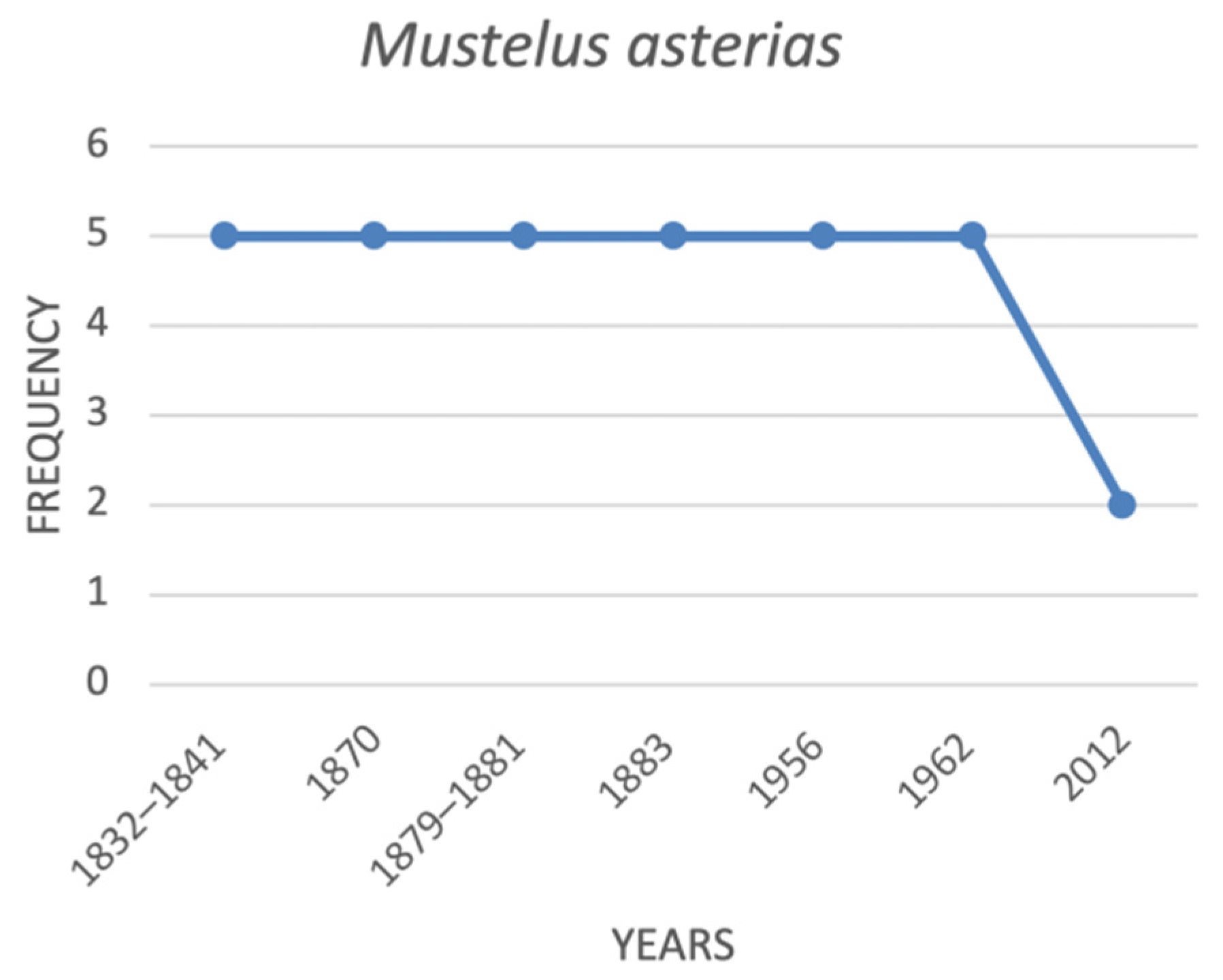
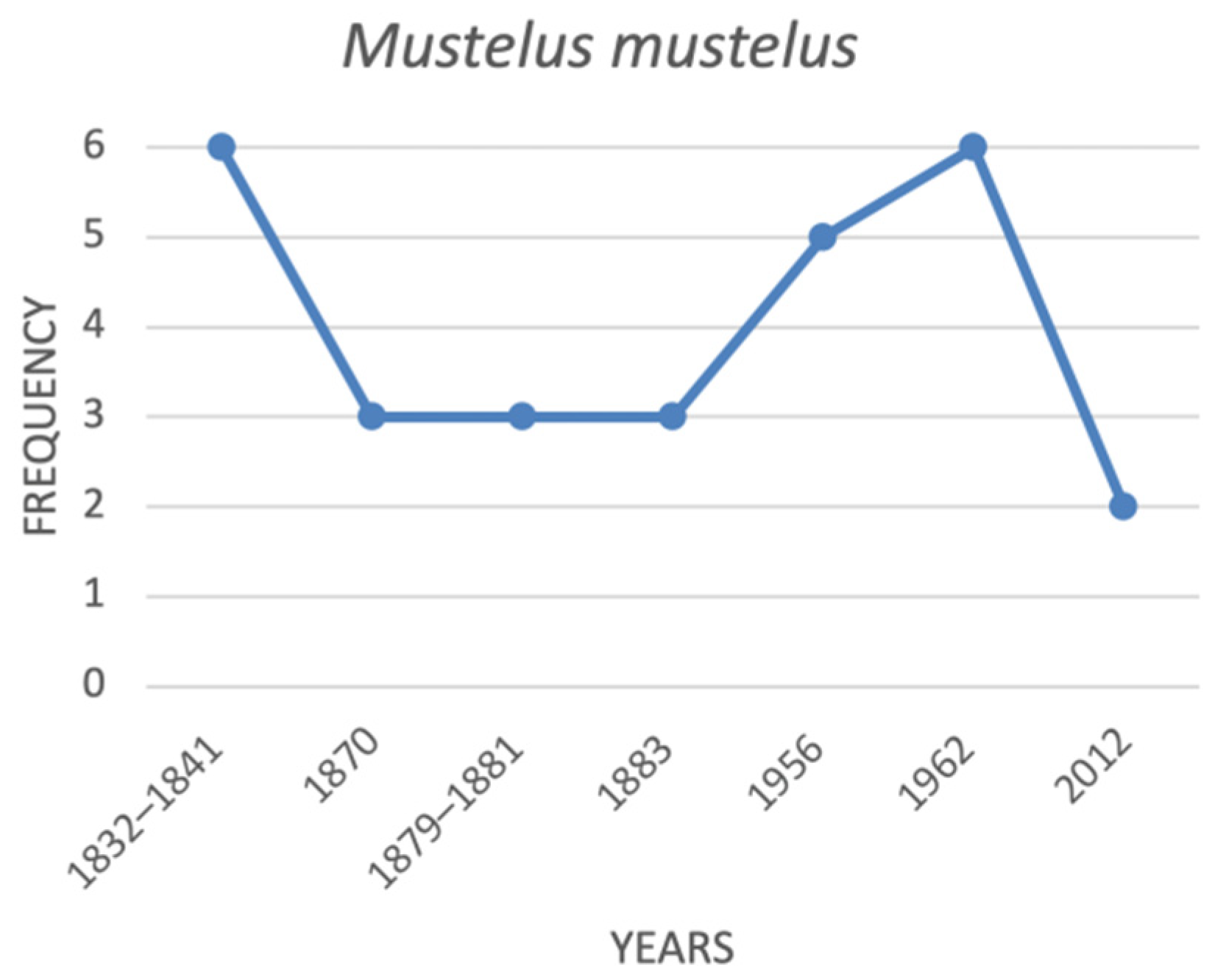
4.1. Historical Trends of the Threatened Shark Species in the Italian Seas
4.2. Increasing Historical Knowledge to Improve Current Conservation
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dulvy, N.K.; Fowler, S.L.; A Musick, J.; Cavanagh, R.D.; Kyne, P.M.; Harrison, L.R.; Carlson, J.K.; Davidson, L.N.; Fordham, S.V.; Francis, M.P.; et al. Extinction risk and conservation of the world’s sharks and rays. eLife 2014, 3, e00590. [Google Scholar] [CrossRef]
- Simpfendorfer, C.A.; Kyne, P.M. Limited potential to recover from overfishing raises concerns for deep-sea sharks, rays and chimaeras. Environ. Conserv. 2009, 36, 97–103. [Google Scholar] [CrossRef]
- Queiroz, M.M.; Pereira, S.C.F. Intention to adopt big data in supply chain management: A Brazilian perspective. Rev. Adm. Empresas 2019, 59, 389–401. [Google Scholar] [CrossRef]
- Simpfendorfer, C.A.; Dulvy, N.K. Bright spots of sustainable shark fishing. Curr. Biol. 2017, 27, R97–R98. [Google Scholar] [CrossRef]
- Cortés, E.; Domingo, A.; Miller, P.; Forselledo, R.; Mas, F.; Arocha, F.; Campana, S.E.; Coelho, R.; da Silva, C.; Hazin, F.; et al. Expanded ecological risk assessment of pelagic sharks caught in Atlantic pelagic longline fisheries. Collect. Vol. Sci. Pap. ICCAT 2015, 71, 2637–2688. [Google Scholar]
- Dulvy, N.K.; Simpfendorfer, C.A.; Davidson, L.N.; Fordham, S.V.; Bräutigam, A.; Sant, G.; Welch, D.J. Challenges and Priorities in Shark and Ray Conservation. Curr. Biol. 2017, 27, R565–R572. [Google Scholar] [CrossRef] [PubMed]
- Pauly, D. Anecdotes and the shifting baseline syndrome of fisheries. Trends Ecol. Evol. 1995, 10, 430. [Google Scholar] [CrossRef]
- Sáenz-Arroyo, A.; Roberts, C.M.; Torre, J.; Cariño-Olvera, M. Using fishers’ anecdotes, naturalists’ observations and grey literature to reassess marine species at risk: The case of the Gulf grouper in the Gulf of California, Mexico. Fish Fish. 2005, 6, 121–133. [Google Scholar] [CrossRef]
- Osio, G.C.; Orio, A.; Millar, C.P. Assessing the vulnerability of Mediterranean demersal stocks and predicting exploitation status of un-assessed stocks. Fish. Res. 2015, 171, 110–121. [Google Scholar] [CrossRef]
- Dulvy, N.K.; Allen, D.J.; Ralph, G.M.; Walls, R.H.L. The Conservation Status of Sharks, Rays and Chimaeras in the Mediterranean Sea [Brochure]; IUCN Centre for Mediterranean Cooperation: Malaga, Spain, 2016; 14p. [Google Scholar]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Lasram, F.B.R.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediterranean Sea: Estimates, patterns, and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [PubMed]
- Katsanevakis, S.; Levin, N.; Coll, M.; Giakoumi, S.; Shkedi, D.; Mackelworth, P.; Levy, R.; Velegrakis, A.; Koutsoubas, D.; Carić, H.; et al. Marine conservation challenges in an era of economic crisis and geopolitical instability: The case of the Mediterranean Sea. Mar. Policy 2015, 51, 31–39. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Morri, C. Global sea warming and “tropicalization” of the Mediterranean Sea: Biogeographic and ecological aspects. Biogeogr. J. Integr. Biogeogr. 2003, 24. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Morri, C. Marine biodiversity of the Mediterranean Sea: Situation, problems and prospects for future research. Mar. Pollut. Bull. 2000, 40, 367–376. [Google Scholar] [CrossRef]
- Guidetti, P.; Milazzo, M.; Bussotti, S.; Molinari, A.; Murenu, M.; Pais, A.; Spanò, N.; Balzano, R.; Agardy, T.; Boero, F.; et al. Italian marine reserve effectiveness: Does enforcement matter? Biol. Conserv. 2008, 141, 699–709. [Google Scholar] [CrossRef]
- Szabó, P. Historical ecology: Past, present and future. Biol. Rev. 2015, 90, 997–1014. [Google Scholar] [CrossRef]
- Balée, W. The Research Program of Historical Ecology. Annu. Rev. Anthropol. 2006, 35, 75–98. [Google Scholar] [CrossRef]
- McClenachan, L.; Cooper, A.B.; McKenzie, M.G.; Drew, J.A. The importance of surprising results and best practices in historical ecology. BioScience 2015, 65, 932–939. [Google Scholar] [CrossRef]
- Lotze, H.K.; Hoffmann, R.; Erlandson, J. Lessons from historical ecology and management. Mar. Ecosyst. Based Manag. Sea Ideas Observ. Prog. Study Seas 2014, 16, 17–55. [Google Scholar]
- Engelhard, G.H.; Lynam, C.P.; Garcia-Carreras, B.; Dolder, P.J.; Mackinson, S. Effort reduction and the large fish indicator: Spatial trends reveal positive impacts of recent European fleet reduction schemes. Environ. Conserv. 2015, 42, 227–236. [Google Scholar] [CrossRef]
- Sanchez, G.M.; Gobalet, K.W.; Jewett, R.; Cuthrell, R.Q.; Grone, M.; Engel, P.M.; Lightfoot, K.G. The historical ecology of central California coast fishing: Perspectives from Point Reyes National Seashore. J. Archaeol. Sci. 2018, 100, 1–15. [Google Scholar] [CrossRef]
- Trindade-Santos, I.; Moyes, F.; Magurran, A.E. Global change in the functional diversity of marine fisheries exploitation over the past 65 years. Proc. R. Soc. B 2020, 287, 20200889. [Google Scholar] [CrossRef] [PubMed]
- Guidetti, P.; Micheli, F. Ancient art serving marine conservation. Front. Ecol. Environ. 2011, 9, 374–375. [Google Scholar] [CrossRef]
- Lotze, H.K.; Coll, M.; Magera, A.M.; Ward-Paige, C.; Airoldi, L. Recovery of marine animal populations and ecosystems. Trends Ecol. Evol. 2011, 26, 595–605. [Google Scholar] [CrossRef]
- Fortibuoni, T.; Libralato, S.; Raicevich, S.; Giovanardi, O.; Solidoro, C. Coding early naturalists’ accounts into long-term fish community changes in the Adriatic Sea (1800–2000). PLoS ONE 2010, 5, e15502. [Google Scholar] [CrossRef] [PubMed]
- McClenachan, L.; Ferretti, F.; Baum, J.K. From archives to conservation: Why historical data are needed to set baselines for marine animals and ecosystems. Conserv. Lett. 2012, 5, 349–359. [Google Scholar] [CrossRef]
- Ferretti, F.; Crowder, L.B.; Micheli, F. Using Disparate Datasets to Reconstruct Historical Baselines of Animal Populations. In Marine Historical Ecology in Conservation: Applying the Past to Manage for the Future; University of California Press: Oakland, CA, USA, 2014; pp. 63–85. [Google Scholar]
- Colloca, F.; Enea, M.; Ragonese, S.; Di Lorenzo, M. A century of fishery data documenting the collapse of smooth-hounds (Mustelus spp.) in the Mediterranean Sea. Aquat. Conserv. Mar. Freshw. Ecosyst. 2017, 27, 1145–1155. [Google Scholar] [CrossRef]
- Mojetta, A.R.; Travaglini, A.; Scacco, U.; Bottaro, M. Where sharks met humans: The Mediterranean Sea, history and myth of an ancient interaction between two dominant predators. Reg. Stud. Mar. Sci. 2018, 21, 30–38. [Google Scholar] [CrossRef]
- Bargnesi, F.; Gridelli, S.; Cerrano, C.; Ferretti, F. Reconstructing the history of the sand tiger shark (Carcharias taurus) in the Mediterranean Sea. Aquat. Conserv. Mar. Freshw. Ecosyst. 2020, 30, 915–927. [Google Scholar] [CrossRef]
- Rondinini, C.; Battistoni, A.; Peronace, V.; Teofili, C. Lista Rossa IUCN dei Vertebrati Italiani; Comitato Italiano IUCN e Ministero dell’Ambiente e della Tutela del Territorio e del Mare: Roma, Italy, 2012; Volume 56. [Google Scholar]
- Bonaparte, C.L. Iconografia della fauna italica per le quattro classi degli animali vertebrati. Tomo III. Pesci. Roma Fasc 1832, 1, 1–6. [Google Scholar]
- Ninni, A.P. Enumerazione dei Pesci delle Lagune e Golfo di Venezia con Note per il Dott. Alessandro Ninni; Soliani: Modena, Italy, 1870. [Google Scholar]
- Doderlein, P. Manuale Ittiologico del Mediterraneo: Ossia Sinossi Metodica delle Varie Specie di Pesci Riscontrate nel Mediterraneo ed in Particolare nei Mari di Sicilia; Tip. del Giornale di Sicilia: Palermo, Italy, 1879. [Google Scholar]
- Faber, G.L. The Fisheries of the Adriatic and the Fish Thereof; Bernard Quaritch: London, UK, 1883. [Google Scholar]
- Lo Bianco, S. Biological notice with special reference to the period of sexual maturity of the animals of the Bay of Naples. Mitth. Zool. Stat. Neapel 1909, 19, 513–761. [Google Scholar]
- Tortonese, E. Fauna d’Italia: Vol. II—Leptocardia Ciclostomata Selachi; Editoriale Calderini: Bologna, Italy, 1956. [Google Scholar]
- Bini, G. Atlante Dei Pesci Delle Coste Italiane; Mondo Sommerso; Editoriale Olimpia: Firenze, Italy, 1962. [Google Scholar]
- Jackson, J.B.C.; Alexander, K.E.; Sala, E. Shifting Baselines: The Past and the Future of Ocean Fisheries; Island Press: Washington, DC, USA, 2011; p. 312. [Google Scholar]
- Caswell, B.; Klein, E.S.; Alleway, H.K.; Ball, J.E.; Botero, J.; Cardinale, M.; Eero, M.; Engelhard, G.H.; Fortibuoni, T.; Giraldo, A.-J.; et al. Something old, something new: Historical perspectives provide lessons for blue growth agendas. Fish Fish. 2020, 21, 774–796. [Google Scholar] [CrossRef]
- Baum, J.K.; Myers, R.A.; Kehler, D.G.; Worm, B.; Harley, S.J.; Doherty, P.A. Collapse and Conservation of Shark Populations in the Northwest Atlantic. Science 2003, 299, 389–392. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.A.; Worm, B. Rapid worldwide depletion of predatory fish communities. Nature 2003, 423, 280–283. [Google Scholar] [CrossRef]
- Myers, R.A.; Worm, B. Extinction, survival or recovery of large predatory fishes. Philos. Trans. R. Soc. B Biol. Sci. 2005, 360, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, F.; Worm, B.; Britten, G.L.; Heithaus, M.R.; Lotze, H.K. Patterns and ecosystem consequences of shark declines in the ocean. Ecol. Lett. 2010, 13, 1055–1071. [Google Scholar] [CrossRef] [PubMed]
- Roff, G.; Brown, C.J.; Priest, M.A.; Mumby, P.J. Decline of coastal apex shark populations over the past half century. Commun. Boil. 2018, 1, 223. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Candelas, I.A.; Pérez-Jiménez, J.C.; Espinoza-Tenorio, A.; McClenachan, L.; Méndez-Loeza, I. Use of historical data to assess changes in the vulnerability of sharks. Fish. Res. 2020, 226, 105526. [Google Scholar] [CrossRef]
- Ferretti, F.; Myers, R.A.; Serena, F.; Lotze, H.K. Loss of large predatory sharks from the Mediterranean Sea. Conserv. Biol. 2008, 22, 952–964. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, F.; Morey Verd, G.; Seret, B.; Sulić Šprem, J.; Micheli, F. Falling through the cracks: The fading history of a large iconic predator. Fish Fish. 2016, 17, 875–889. [Google Scholar] [CrossRef]
- Fortibuoni, T.; Libralato, S.; Arneri, E.; Giovanardi, O.; Solidoro, C.; Raicevich, S. Fish and fishery historical data since the 19th century in the Adriatic Sea, Mediterranean. Sci. Data 2017, 4, 170104. [Google Scholar] [CrossRef]
- Barausse, A.; Correale, V.; Curkovic, A.; Finotto, L.; Riginella, E.; Visentin, E.; Mazzoldi, C. The role of fisheries and the environment in driving the decline of elasmobranchs in the northern Adriatic Sea. ICES J. Mar. Sci. 2014, 71, 1593–1603. [Google Scholar] [CrossRef]
- Mazzoldi, C.; Sambo, A.; Riginella, E. The Clodia database: A long time series of fishery data from the Adriatic Sea. Sci. Data 2014, 1, 140018. [Google Scholar] [CrossRef] [PubMed]
- Fortibuoni, T.; Borme, D.; Franceschini, G.; Giovanardi, O.; Raicevich, S. Common, rare or extirpated? Shifting baselines for common angelshark, Squatina squatina (Elasmobranchii: Squatinidae), in the Northern Adriatic Sea (Mediterranean Sea). Hydrobiologia 2016, 772, 247–259. [Google Scholar] [CrossRef]
- Psomadakis, P.N.; Maio, N.; Vacchi, M. The chondrichthyan biodiversity in the Gulf of Naples (SW Italy, Tyrrhenian Sea): An historical overview. Cybium 2009, 33, 199–209. [Google Scholar]
- Targioni-Tozzetti, A. La Pesca in Italia (Vol. 2); R. Istituto Sordo-Muti: Genoa, Italy, 1874. [Google Scholar]
- Alverson, D.L.; Stansby, M.E. The Spiny Dogfish (Squalus acanthias) in the Northeastern Pacific (No. 447); US Department of the Interior, Fish and Wildlife Service, Bureau of Commercial Fisheries: Washington, DC, USA, 1963.
- Alonso, M.K.; Crespo, E.A.; García, N.A.; Pedraza, S.N.; Mariotti, P.A.; Mora, N.J. Fishery and ontogenetic driven changes in the diet of the spiny dogfish, Squalus acanthias, in Patagonian waters, Argentina. Environ. Biol. Fish. 2002, 63, 193–202. [Google Scholar] [CrossRef]
- King, J.R.; McFarlane, G.A. Trends in Abundance of Spiny Dogfish in the Strait of Georgia, 1980–2005. In Biology and Management of Dogfish Sharks; American Fisheries Society: Bethesda, MD, USA, 2009; pp. 89–100. [Google Scholar]
- Belleggia, M.; Figueroa, D.E.; Sánchez, F.; Bremec, C. Long-term changes in the spiny dogfish (Squalus acanthias) trophic role in the southwestern Atlantic. Hydrobiologia 2012, 684, 57–67. [Google Scholar] [CrossRef]
- Fordham, S.; Dolan, C. A case study in international shark conservation: The convention on international trade in endangered species and the spiny dogfish. Gold. Gate UL Rev. 2004, 34, 531. [Google Scholar]
- Fowler, S.; Raymakers, C.; Grimm, U. Trade in and Conservation of Two Shark Species, Porbeagle (Lamna nasus) and Spiny Dogfish (Squalus acanthias); Bundesamt für Naturschutz (BfN): Bonn, Germany, 2004. [Google Scholar]
- MAFMC. Mid-Atlantic Fishery Management Council. In Spiny Dogfish Fishery Management Plan; MAFMC: Dover, DE, USA, 1999. [Google Scholar]
- Dell’Apa, A.; Bangley, C.W.; Rulifson, R.A. Who let the dogfish out? A review of management and socio-economic aspects of spiny dogfish fisheries. Rev. Fish Biol. Fish. 2015, 25, 273–295. [Google Scholar] [CrossRef]
- Bellodi, A.; Porcu, C.; Cau, A.; Marongiu, M.F.; Melis, R.; Mulas, A.; Pesci, P.; Follesa, M.C.; Cannas, R. Investigation on the genus Squalus in the Sardinian waters (Central-Western Mediterranean) with implications on its management. Mediterr. Mar. Sci. 2018, 19, 256–272. [Google Scholar] [CrossRef]
- Bonanomi, S.; Pulcinella, J.; Fortuna, C.M.; Moro, F.; Sala, A. Elasmobranch bycatch in the Italian Adriatic pelagic trawl fishery. PLoS ONE 2018, 13, e0191647. [Google Scholar] [CrossRef]
- Bargione, G.; Donato, F.; La Mesa, M.; Mazzoldi, C.; Riginella, E.; Vasapollo, C.; Virgili, M.; Lucchetti, A. Life-history traits of the spiny dogfish Squalus acanthias in the Adriatic Sea. Sci. Rep. 2019, 9, 14317. [Google Scholar] [CrossRef] [PubMed]
- Lawson, J.M.; Pollom, R.A.; Gordon, C.A.; Barker, J.; Meyers, E.K.; Zidowitz, H.; Ellis, J.R.; Bartolí, Á.; Morey, G.; Fowler, S.L.; et al. Extinction risk and conservation of critically endangered angel sharks in the Eastern Atlantic and Mediterranean Sea. ICES J. Mar. Sci. 2020, 77, 12–29. [Google Scholar] [CrossRef]
- Ragonese, S.; Vitale, S.; Dimech, M.; Mazzola, S. Abundances of demersal sharks and chimaera from 1994–2009 scientific surveys in the central Mediterranean Sea. PLoS ONE 2013, 8, e74865. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, M.; Ammendolia, G.; Navarra, E. Finding of a rare Squatina squatina (Linnaeus, 1758) (Chondrichthyes: Squatinidae) along the Tyrrhenian coast of the Strait of Messina and its maintenance in an aquarium. Mar. Biodivers. Rec. 2015, 8. [Google Scholar] [CrossRef]
- Zava, B.; Fiorentino, F.; Serena, F. Occurrence of juveniles Squatina oculata Bonaparte, 1840 (Elasmobranchii: Squatinidae) in the Strait of Sicily (Central Mediterranean). Cybium Int. J. Ichthyol. 2016, 1840, 341–343. [Google Scholar]
- Serena, F. Field Identification Guide to the Sharks and Rays of the Mediterranean and Black Sea; Food and Agriculture Organization of the United Nations: Rome, Italy, 2005. [Google Scholar]
- Gordon, C.A.; Hood, A.R.; Al Mabruk, S.A.A.; Barker, J.; Bartolí, A.; Ben Abdelhamid, S.; Bradai, M.N.; Dulvy, N.K.; Fortibuoni, T.; Giovos, I.; et al. Mediterranean Angel Sharks: Regional Action Plan; The Shark Trust: Plymouth, UK, 2019. [Google Scholar]
- Giovos, I.; Stoilas, V.; Al-Mabruk, S.A.; Doumpas, N.; Marakis, P.; Maximiadi, M.; Moutopoulos, D.K.; Kleitou, P.; Keramidas, I.; Tiralongo, F.; et al. Integrating local ecological knowledge, citizen science and long-term historical data for endangered species conservation: Additional records of angel sharks (Chondrichthyes: Squatinidae) in the Mediterranean Sea. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 881–890. [Google Scholar] [CrossRef]
- Smith, S.E.; Rasmussen, R.C.; Ramon, D.A.; Cailliet, G.M. The Biology and Ecology of Thresher Sharks (Alopiidae). In Sharks of the Open Ocean: Biology, Fisheries and Conservation; Wiley: Hoboken, NJ, USA, 2008; pp. 60–68. [Google Scholar]
- Megalofonou, P.; Yannopoulos, C.; Damalas, D.; De Metrio, G.; De Florio, M. Incidental catch and estimated discards of pelagic sharks from the swordfish and tuna fisheries in the Mediterranean Sea. Fish. Bull. 2005, 103, 620–634. [Google Scholar]
- Garibaldi, F. By-catch in the mesopelagic swordfish longline fishery in the Ligurian Sea (Western Mediterranean). Collect. Vol. Sci. Pap. ICCAT 2015, 71, 1495–1498. [Google Scholar]
- Panayiotou, N.; Porsmoguer, S.B.; Moutopoulos, D.Κ.; Lloret, J. Offshore recreational fisheries of large vulnerable sharks and teleost fish in the Mediterranean Sea: First information on the species caught. Mediterr. Mar. Sci. 2020, 21, 222–227. [Google Scholar] [CrossRef]
- Watson, J.W.; Kerstetter, D.W. Pelagic longline fishing gear: A brief history and review of research efforts to improve selectivity. Mar. Technol. Soc. J. 2006, 40, 6–11. [Google Scholar] [CrossRef]
- Sassu, N.; Cannas, A.; Ferretti, M. Gli Attrezzi da Pesca in Uso nelle Marinerie Italiane; UNIMAR: Rome, Italy, 2001; Volume 16, p. 81. [Google Scholar]
- Ferretti, M. Classificazione e Descrizione Degli Attrezzi da Pesca in Uso Nelle Marinerie Italiane con Particolare Referimento al Loro Impatto Ambientale; ICRAM: Rome, Italy, 2002. [Google Scholar]
- Cataudella, S.; Spagnolo, M. Lo Stato della Pesca e Dell’Acquacoltura nei Mari Italiani; Ministero delle Politiche Agricole Alimentari e Forestali: Rome, Italy, 2011; 877p. [Google Scholar]
- Preti, A.; Smith, S.E.; Ramon, D.A. Diet differences in the thresher shark (Alopias vulpinus) during transition from a warm-water regime to a cool-water regime off California-Oregon, 1998–2000. Calif. Cooper. Ocean. Fish. Investig. Rep. 2004, 45, 118. [Google Scholar]
- Preti, A.; Soykan, C.U.; Dewar, H.; Wells, R.D.; Spear, N.; Kohin, S. Comparative feeding ecology of shortfin mako, blue and thresher sharks in the California Current. Environ. Biol. Fish. 2012, 95, 127–146. [Google Scholar] [CrossRef]
- Balestra, V.; Boero, F.; Carli, A. Andamento del pescato della tonnarella di Camogli dal 1950 al 1974. Valutazioni bio-statistiche. Boll. Pesca Piscic. Idrobiol. 1976, 31, 2. [Google Scholar]
- Cattaneo-Vietti, R. Structural changes in Mediterranean marine communities: Lessons from the Ligurian Sea. Rend. Lincei. Sci. Fis. Nat. 2018, 29, 515–524. [Google Scholar]
- Finotto, L.; Barausse, A.; Mazzoldi, C. In search of prey: The occurrence of Alopias vulpinus (Bonnaterre, 1788) in the northern Adriatic Sea and its interactions with fishery. Acta Adriat. 2016, 57, 295–304. [Google Scholar]
- Sguotti, C.; Lynam, C.P.; García-Carreras, B.; Ellis, J.R.; Engelhard, G.H. Distribution of skates and sharks in the North Sea: 112 years of change. Glob. Chang. Boil. 2016, 22, 2729–2743. [Google Scholar] [CrossRef]
- D’Ancona, U. Dell’Influenza della Stasi Peschereccia del Periodo 1914–1918 sul Patrimonio Ittico Dell’Alto Adriatico. In Regio Comitato Talassografico Italiano, Memoria CXXVI; Officine Grafiche Carlo Ferrari: Venice, Italy, 1926; pp. 1–95. [Google Scholar]
- D’Ancona, U. Rilievi statistici sulla pesca nell’alto Adriatico. Istit. Ven. Sci. Lett. Arti 1949, 108, 41–53. [Google Scholar]
- Di Lorenzo, M.; Vizzini, S.; Signa, G.; Andolina, C.; Palo, G.B.; Gristina, M.; Mazzoldi, C.; Colloca, F. Ontogenetic trophic segregation between two threatened smooth-hound sharks in the Central Mediterranean Sea. Sci. Rep. 2020, 10, 11011. [Google Scholar] [CrossRef]
- Last, P.R.; Stevens, J.D. Sharks and Rays of Australia, 2nd ed.; CSIRO Publishing: Melbourne, Australia, 2009. [Google Scholar]
- Ebert, D.A.; Fowler, S.L.; Compagno, L.J. Sharks of the World: A Fully Illustrated Guide; Wild Nature Press: Princeton, NJ, USA, 2013. [Google Scholar]
- Ovenden, J.R.; Kashiwagi, T.; Broderick, D.; Giles, J.; Salini, J. The extent of population genetic subdivision differs among four co-distributed shark species in the Indo-Australian archipelago. BMC Evol. Boil. 2009, 9, 40. [Google Scholar] [CrossRef]
- King, J.R.; Wetklo, M.; Supernault, J.; Taguchi, M.; Yokawa, K.; Sosa-Nishizaki, O.; Withler, R.E. Genetic analysis of stock structure of blue shark (Prionace glauca) in the north Pacific ocean. Fish. Res. 2015, 172, 181–189. [Google Scholar] [CrossRef]
- Leone, A.; Urso, I.; Damalas, D.; Martinsohn, J.; Zanzi, A.; Mariani, S.; Sperone, E.; Micarelli, P.; Garibaldi, F.; Megalofonou, P.; et al. Genetic differentiation and phylogeography of Mediterranean-North Eastern Atlantic blue shark (Prionace glauca, L. 1758) using mitochondrial DNA: Panmixia or complex stock structure? PeerJ 2017, 5, e4112. [Google Scholar] [CrossRef] [PubMed]
- Veríssimo, A.; Sampaio, Í.; McDowell, J.R.; Alexandrino, P.; Mucientes, G.; Queiroz, N.; Da Silva, C.; Jones, C.S.; Noble, L.R. World without borders—Genetic population structure of a highly migratory marine predator, the blue shark (Prionace glauca). Ecol. Evol. 2017, 7, 4768–4781. [Google Scholar]
- Bailleul, D.; MacKenzie, A.; Sacchi, O.; Poisson, F.; Bierne, N.; Arnaud-Haond, S. Large-scale genetic panmixia in the blue shark (Prionace glauca): A single worldwide population, or a genetic lag-time effect of the “grey zone” of differentiation? Evol. Appl. 2018, 11, 614–630. [Google Scholar] [CrossRef]
- Campana, S.E.; Marks, L.; Joyce, W.; Kohler, N.E. Effects of recreational and commercial fishing on blue sharks (Prionace glauca) in Atlantic Canada, with inferences on the North Atlantic population. Can. J. Fish. Aquat. Sci. 2006, 63, 670–682. [Google Scholar] [CrossRef]
- Froese, R.; Garilao, C.; Winker, H.; Coro, G.; Demirel, N.; Tsikliras, A.; Dimarchopoulou, D.; Scarcella, G.; Sampang-Reyes, A. Exploitation and Status of European Stocks; Oceana: Washington, DC, USA, 2016. [Google Scholar]
- Biton-PorSmoguer, S.; LLoret, J. Potentially unsustainable fisheries of a critically-endangered pelagic shark species: The case of the Blue shark (Prionace glauca) in the Western Mediterranean Sea. Cybium 2018, 42, 299–302. [Google Scholar]
- Storelli, M.M.; Barone, G.; Storelli, A.; Marcotrigiano, G.O. Levels and congener profiles of PCBs and PCDD/Fs in blue shark (Prionace glauca) liver from the South-Eastern Mediterranean Sea (Italy). Chemosphere 2011, 82, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, I.; Garibaldi, F.; Canesi, L.; Fossi, M.C.; Baini, M. First data on plastic ingestion by blue sharks (Prionace glauca) from the Ligurian Sea (North-Western Mediterranean Sea). Mar. Pollut. Bull. 2018, 135, 303–310. [Google Scholar] [CrossRef]
- Colloca, F.; Scannela, D.; Geraci, M.L.; Falsone, F.; Giusto, B.; Vitale, S.; Di Lorenzo, M.; Bono, G. British sharks in Sicily: Records of long-distance migration of tope shark (Galeorhinus galeus) from the north-eastern Atlantic to the Mediterranean Sea. Mediterr. Mar. Sci. 2019, 20, 309–313. [Google Scholar] [CrossRef]
- United Nations Environment (UNEP). Proposal for the Inclusion of the Tope Shark (Galeorhinus galeus) in Appendix II of the Convention. In Proceedings of the 4th Meeting of the Sessional Committee of the CMS Scientific Council (ScC-SC4), Bonn, Germany, 12–15 November 2019. UNEP/CMS/COP13/Doc. 27.1.10. [Google Scholar]
- D’ancona, U.; Razzauti, A. Pesci e Pesca nelle acque dell’Arcipelago Toscano. Comm. Int. Exp. Sci. Médit. Rapp. Pro. Verb. 1937, 11, 129. [Google Scholar]
- Relini, G.; Mannini, A.; De Ranieri, S.; Bitetto, I.; Follesa, M.C.; Gancitano, V.; Manfredi, C.; Casciaro, L.; Sion, L. Chondrichthyes caught during the medits surveys in Italian waters. Biol. Mar. Mediterr. 2010, 17, 186–204. [Google Scholar]
- Ferretti, F.; Myers, R.A.; Sartor, P.; Serena, F. Long term dynamics of the chondrichthyan fish community in the upper Tyrrhenian Sea. ICES CM 2005, 25, 1–34. [Google Scholar]
- Ferretti, F.; Curnick, D.; Liu, K.; Romanov, E.V.; Block, B.A. Shark baselines and the conservation role of remote coral reef ecosystems. Sci. Adv. 2018, 4, eaaq0333. [Google Scholar] [CrossRef] [PubMed]
- Drew, J.; Philipp, C.; Westneat, M.W. Shark Tooth Weapons from the 19th Century Reflect Shifting Baselines in Central Pacific Predator Assemblies. PLoS ONE 2013, 8, e59855. [Google Scholar] [CrossRef] [PubMed]
- Moro, S.; Jona-Lasinio, G.; Block, B.; Micheli, F.; De Leo, G.; Serena, F.; Bottaro, M.; Scacco, U.; Ferretti, F. Abundance and distribution of the white shark in the Mediterranean Sea. Fish Fish. 2020, 21, 338–349. [Google Scholar] [CrossRef]
- Burgman, M. Risk and Decisions for Conservation and Environmental Management; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Gallagher, A.J.; Kyne, P.M.; Hammerschlag, N. Ecological risk assessment and its application to elasmobranch conservation and management. J. Fish Biol. 2012, 80, 1727–1748. [Google Scholar] [CrossRef]
- Milton, D.A. Assessing the susceptibility to fishing of populations of rare trawl bycatch: Sea snakes caught by Australia’s Northern Prawn Fishery. Biol. Conserv. 2001, 101, 281–290. [Google Scholar] [CrossRef]
- Chin, A.; Kyne, P.M.; Walkers, T.I.; McAucley, R.B. An integrated risk assessment for climate change: Analyzing the vulnerability of sharks and rays on Australia’s Great Barrier Reef. Glob. Chang. Biol. 2010, 16, 1936–1953. [Google Scholar] [CrossRef]
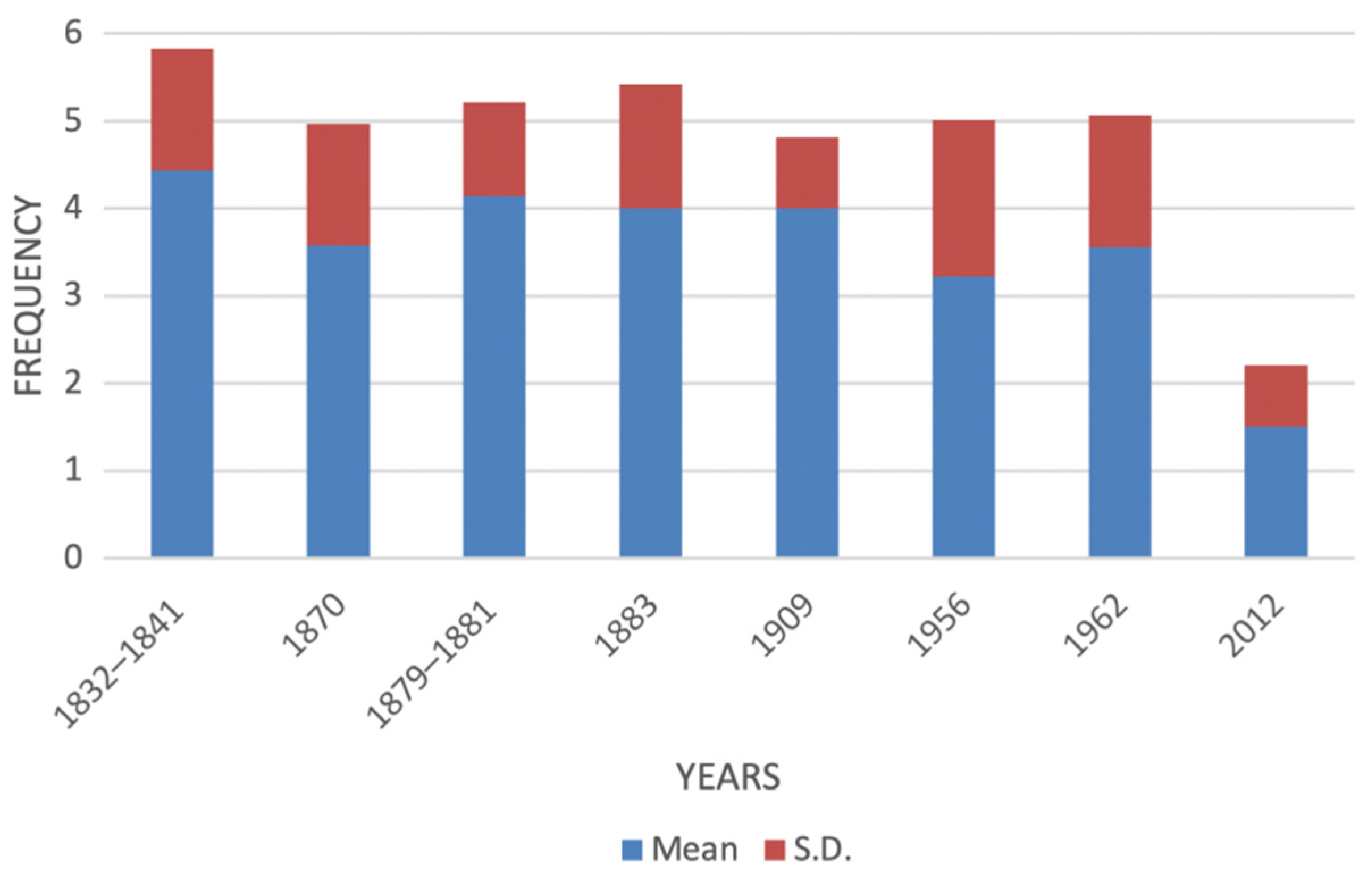
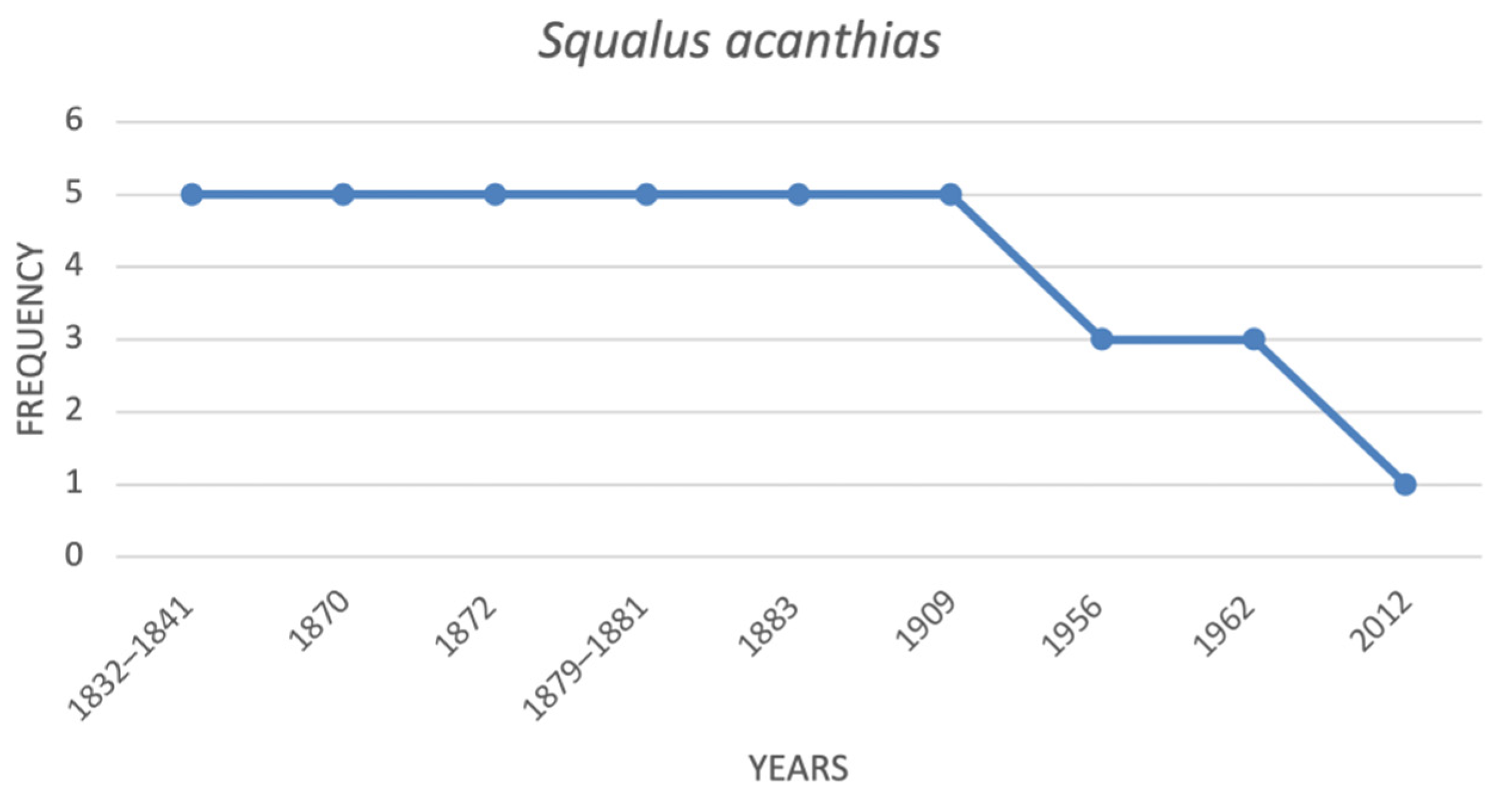
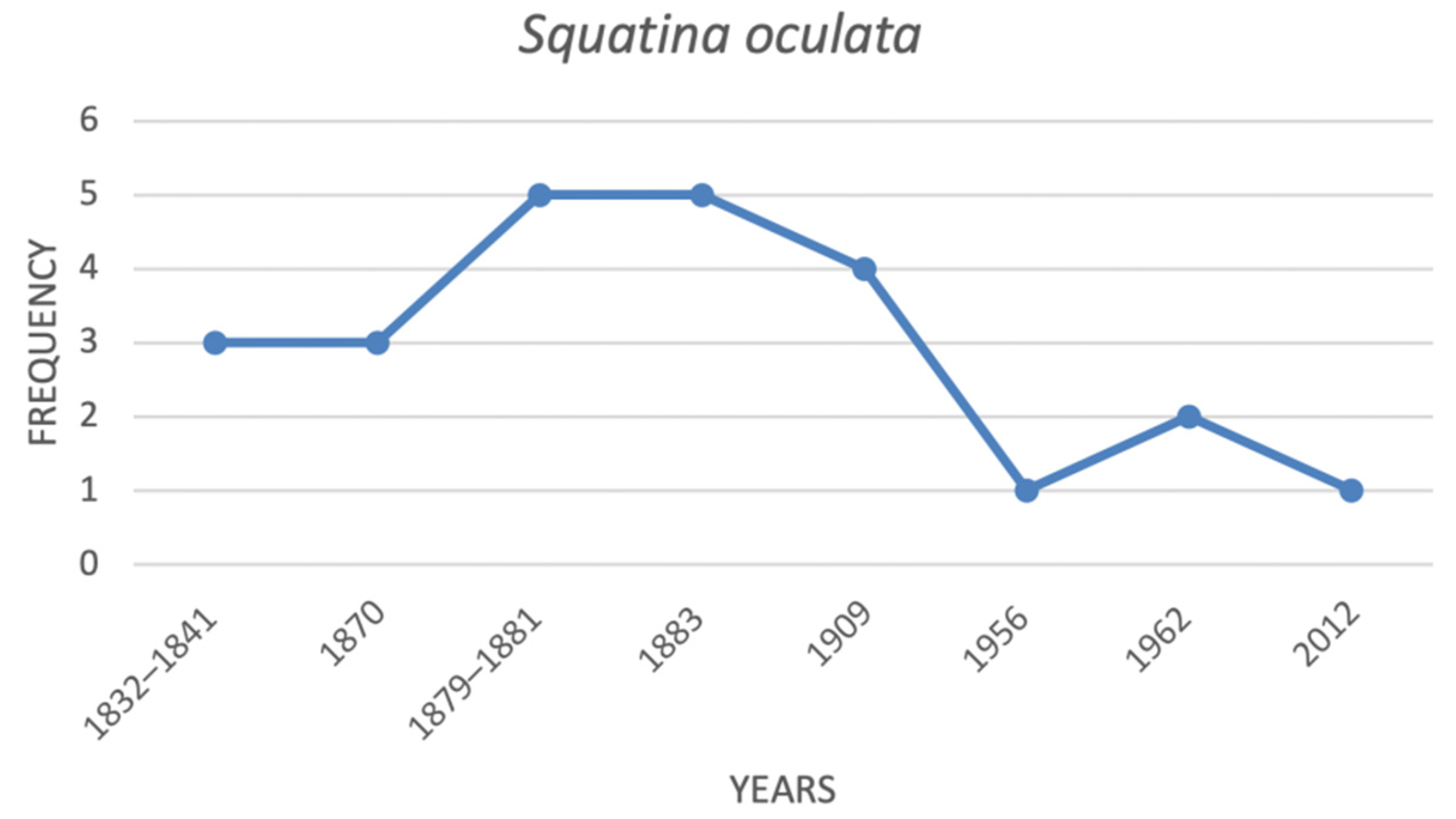
| Species | Common Name | ItalianIUCN Red List (2012) | Mediterranean IUCN Red List (2016) |
|---|---|---|---|
| Squalus acanthias | spiny dogfish | CR | EN |
| Squatina aculeata | sawback angel shark | CR | CR |
| Squatina oculata | smoothback angel shark | CR | CR |
| Squatina squatina | angel shark | CR | CR |
| Alopias vulpinus | common thresher shark | CR | EN |
| Mustelus asterias | starry smoothhound | EN | VU |
| Mustelus mustelus | common smoothhound | EN | VU |
| Mustelus punctulatus | blackspotted smoothhound | EN | VU |
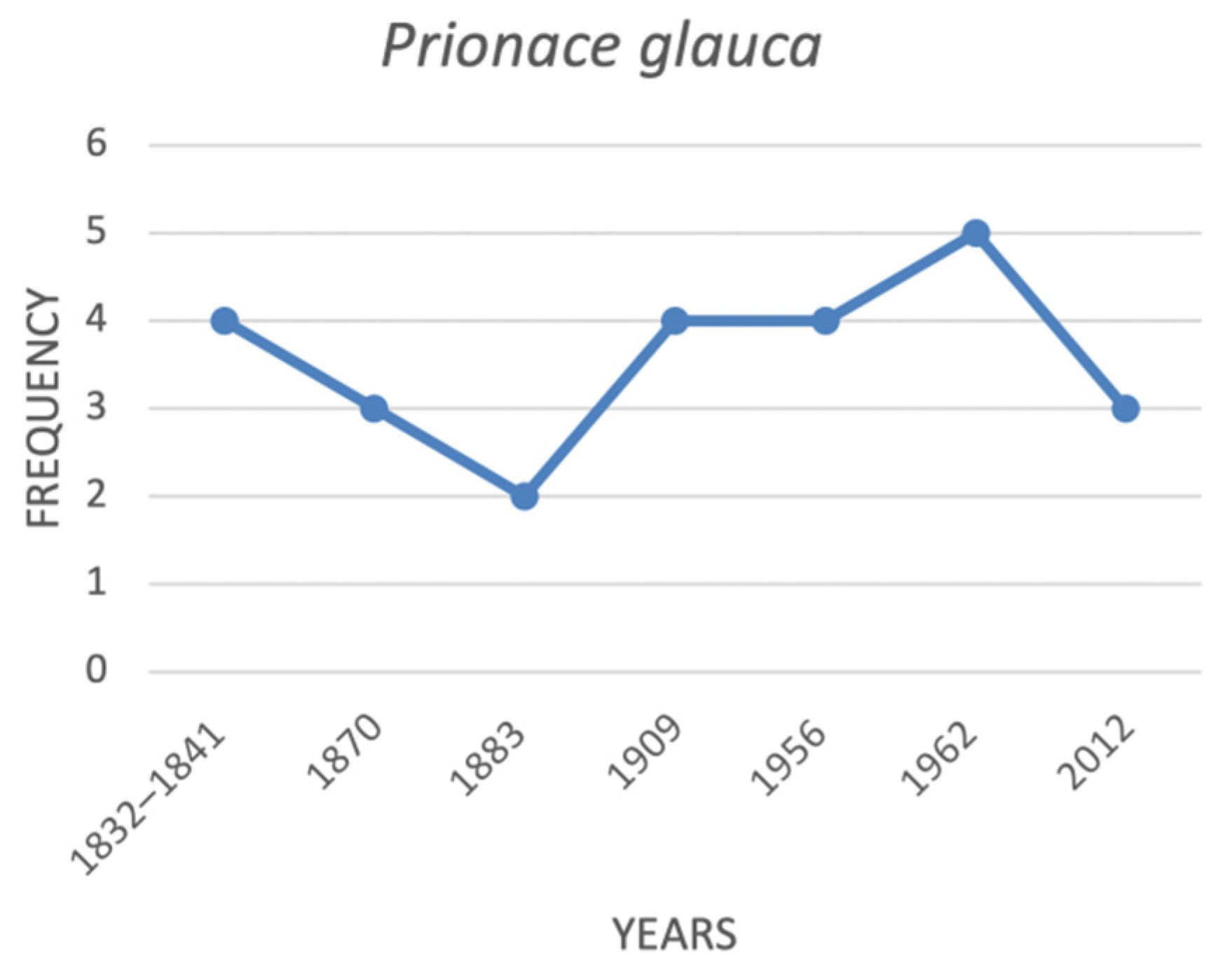
| Prionace glauca | blue shark | VU | CR |
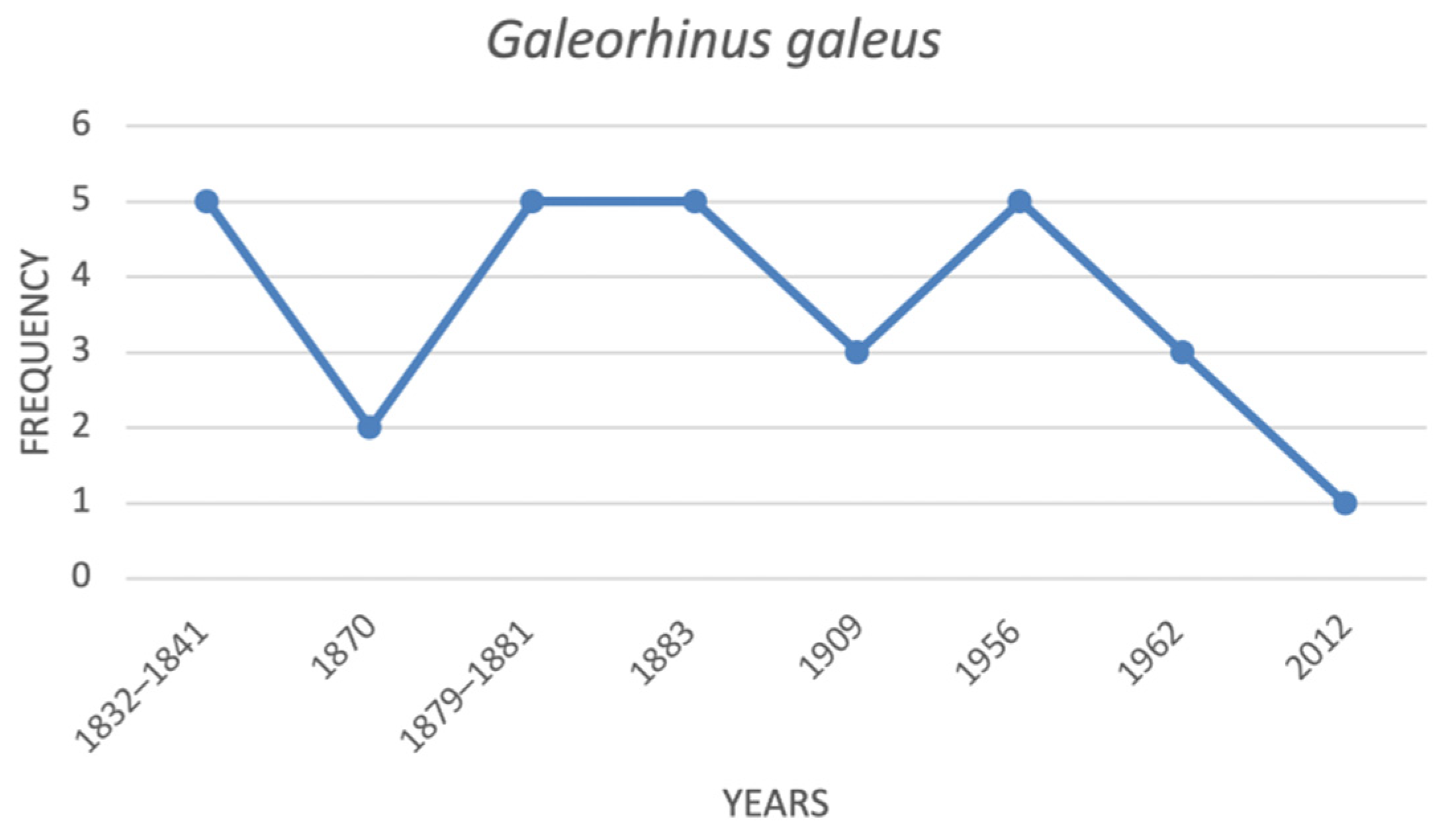
| Galeorhinus galeus | tope shark | CR | VU |
| Reported Frequency/IUCN Category | Very Rare/CR | Rare/EN | Uncommon/VU | Frequent/NT | Common/LC | Very Common/LC |
|---|---|---|---|---|---|---|
| Assigned value | 1 | 2 | 3 | 4 | 5 | 6 |
| Historical Periods, Related Maritime Sectors and References | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Species | Common name | 1832–1841, Italian Seas [32] | 1870, Adriatic Sea [33] | 1879–1881, Sicilian Seas [34] | 1883, Adriatic Sea [35] | 1909, Central Thyrrenian Sea [36] | 1956, Italian Seas [37] | 1962, Italian Seas [38] | 2012, Italian Seas [31] |
| Squalus acanthias | spiny dogfish | 5 | 5 | 5 | 5 | 5 | 3 | 3 | 1 |
| Squatina aculeata | sawback angel shark | na | na | na | na | na | 1 | 2 | 1 |
| Squatina oculata | smoothback angel shark | 3 | 3 | 5 | 5 | 4 | 1 | 2 | 1 |
| Squatina squatina | angel shark | 5 | 5 | 3 | 5 | na | 1 | 2 | 1 |
| Alopias vulpinus | common thresher shark | 2 | 2 | 3 | 2 | na | 4 | 4 | 1 |
| Mustelus asterias | starry smoothhound | 5 | 5 | 5 | 5 | na | 5 | 5 | 2 |
| Mustelus mustelus | common smoothhound | 6 | 3 | 3 | 3 | na | 5 | 6 | 2 |
| Mustelus punctulatus | blackspotted smoothhound | na | na | na | na | na | na | na | 2 |
| Prionace glauca | blue shark | na | 3 | na | 2 | 4 | 4 | 5 | 3 |
| Galeorhinus galeus | tope shark | 5 | 2 | 5 | 5 | 3 | 5 | 3 | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonetti, F.L.; Sperone, E.; Travaglini, A.; Mojetta, A.R.; Signore, M.; Psomadakis, P.N.; Dinkel, T.M.; Bottaro, M. Filling the Gap and Improving Conservation: How IUCN Red Lists and Historical Scientific Data Can Shed More Light on Threatened Sharks in the Italian Seas. Diversity 2020, 12, 389. https://doi.org/10.3390/d12100389
Leonetti FL, Sperone E, Travaglini A, Mojetta AR, Signore M, Psomadakis PN, Dinkel TM, Bottaro M. Filling the Gap and Improving Conservation: How IUCN Red Lists and Historical Scientific Data Can Shed More Light on Threatened Sharks in the Italian Seas. Diversity. 2020; 12(10):389. https://doi.org/10.3390/d12100389
Chicago/Turabian StyleLeonetti, Francesco Luigi, Emilio Sperone, Andrea Travaglini, Angelo R. Mojetta, Marco Signore, Peter N. Psomadakis, Thaya M. Dinkel, and Massimiliano Bottaro. 2020. "Filling the Gap and Improving Conservation: How IUCN Red Lists and Historical Scientific Data Can Shed More Light on Threatened Sharks in the Italian Seas" Diversity 12, no. 10: 389. https://doi.org/10.3390/d12100389
APA StyleLeonetti, F. L., Sperone, E., Travaglini, A., Mojetta, A. R., Signore, M., Psomadakis, P. N., Dinkel, T. M., & Bottaro, M. (2020). Filling the Gap and Improving Conservation: How IUCN Red Lists and Historical Scientific Data Can Shed More Light on Threatened Sharks in the Italian Seas. Diversity, 12(10), 389. https://doi.org/10.3390/d12100389





