Vespa velutina: An Alien Driver of Honey Bee Colony Losses
Abstract
1. Introduction
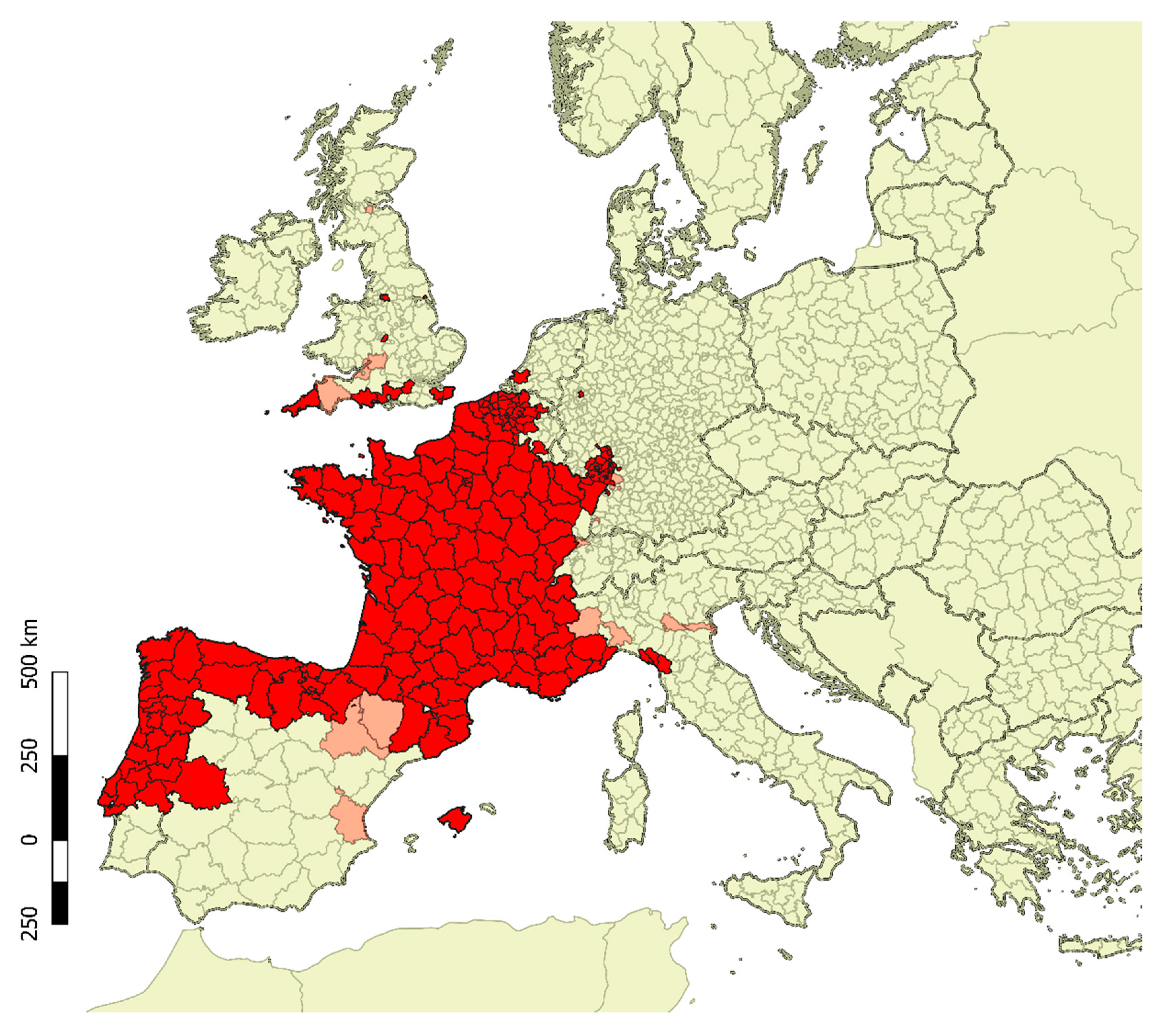
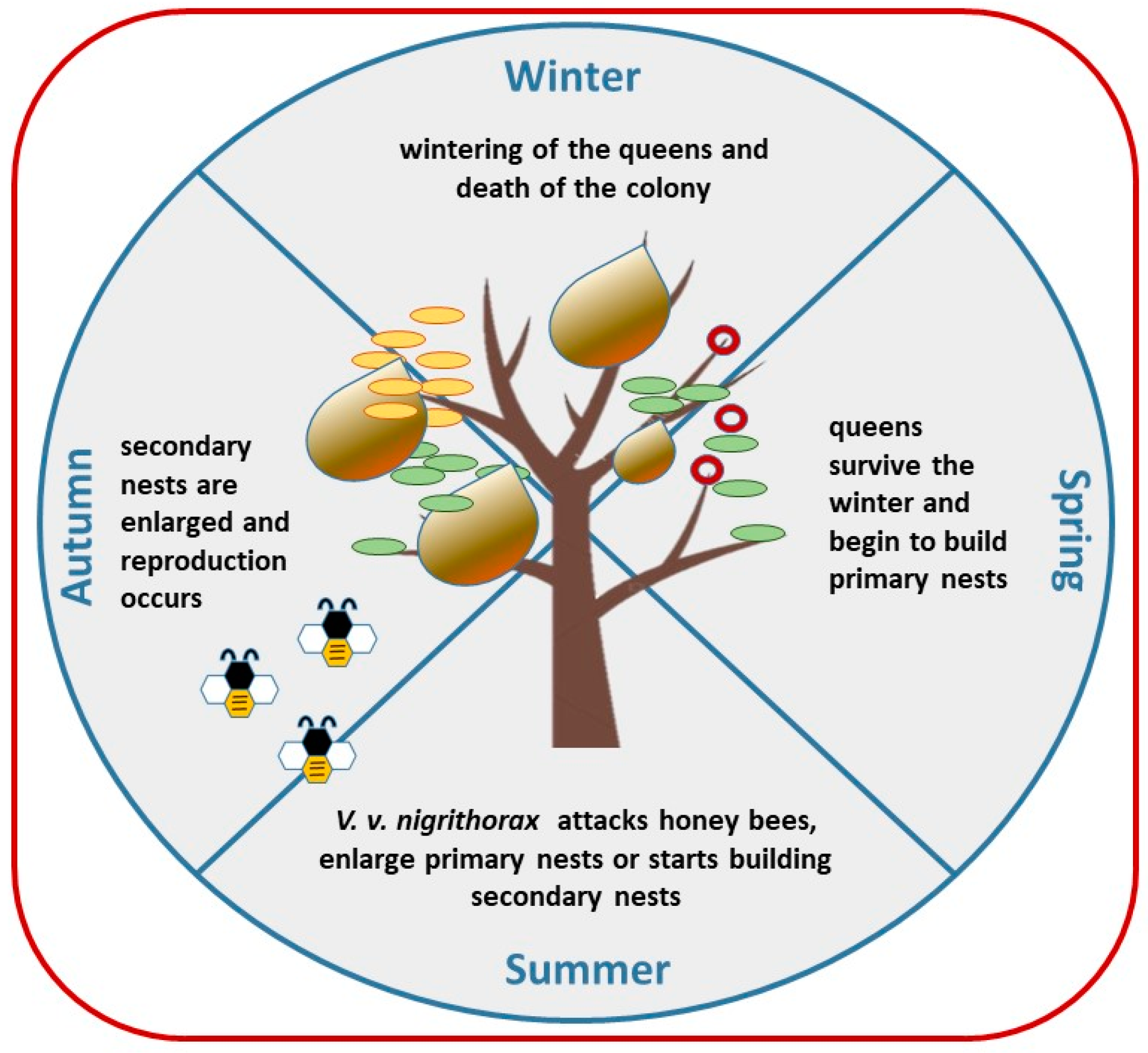
2. Biology
3. Impacts
- Economic threats: Loss of honey bee colonies and decreasing of beekeeping products; cost for control activities and nests removal;
- Ecological threats: Impacts on biodiversity; impacts on pollination activity; competition with native species;
- Public health threats: Potential risk for citizens.
3.1. Damage to Beekeeping
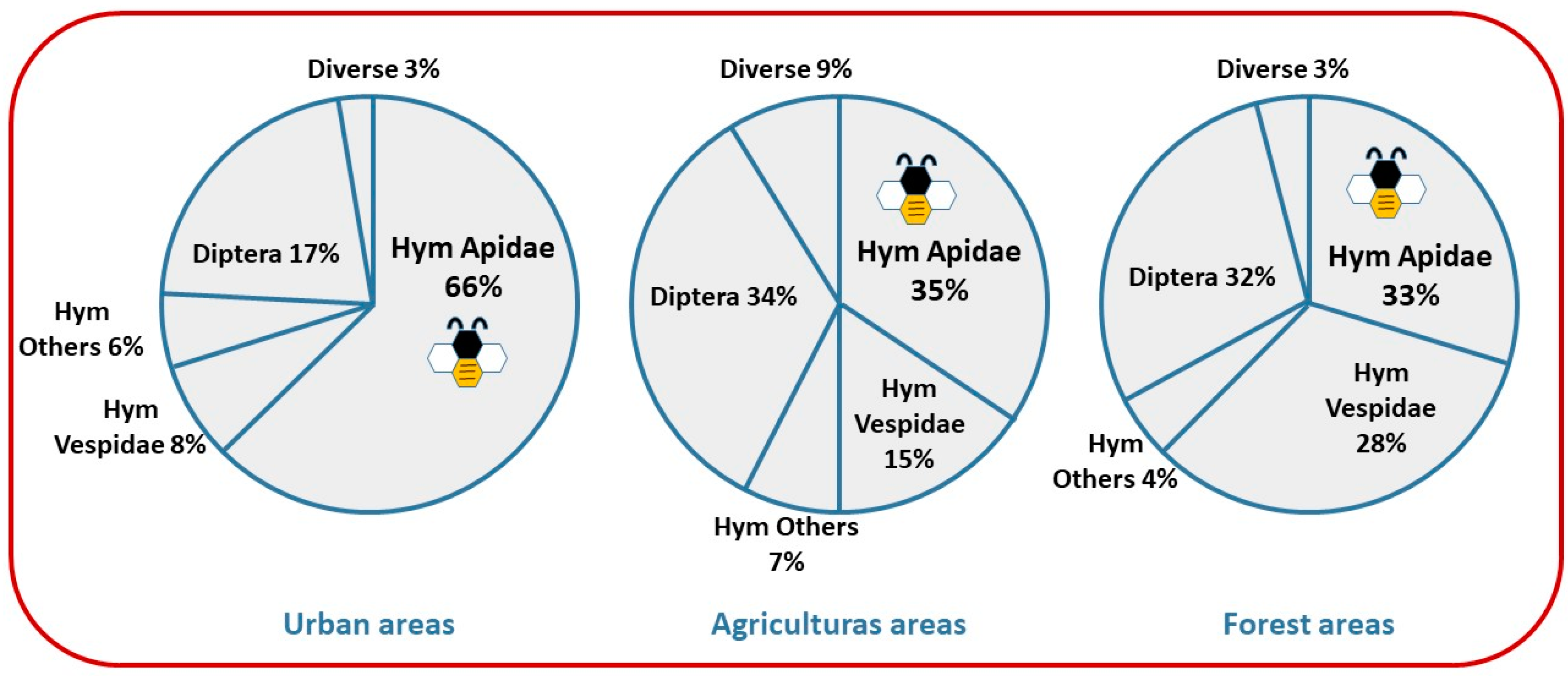
3.2. Impact on Ecosystem
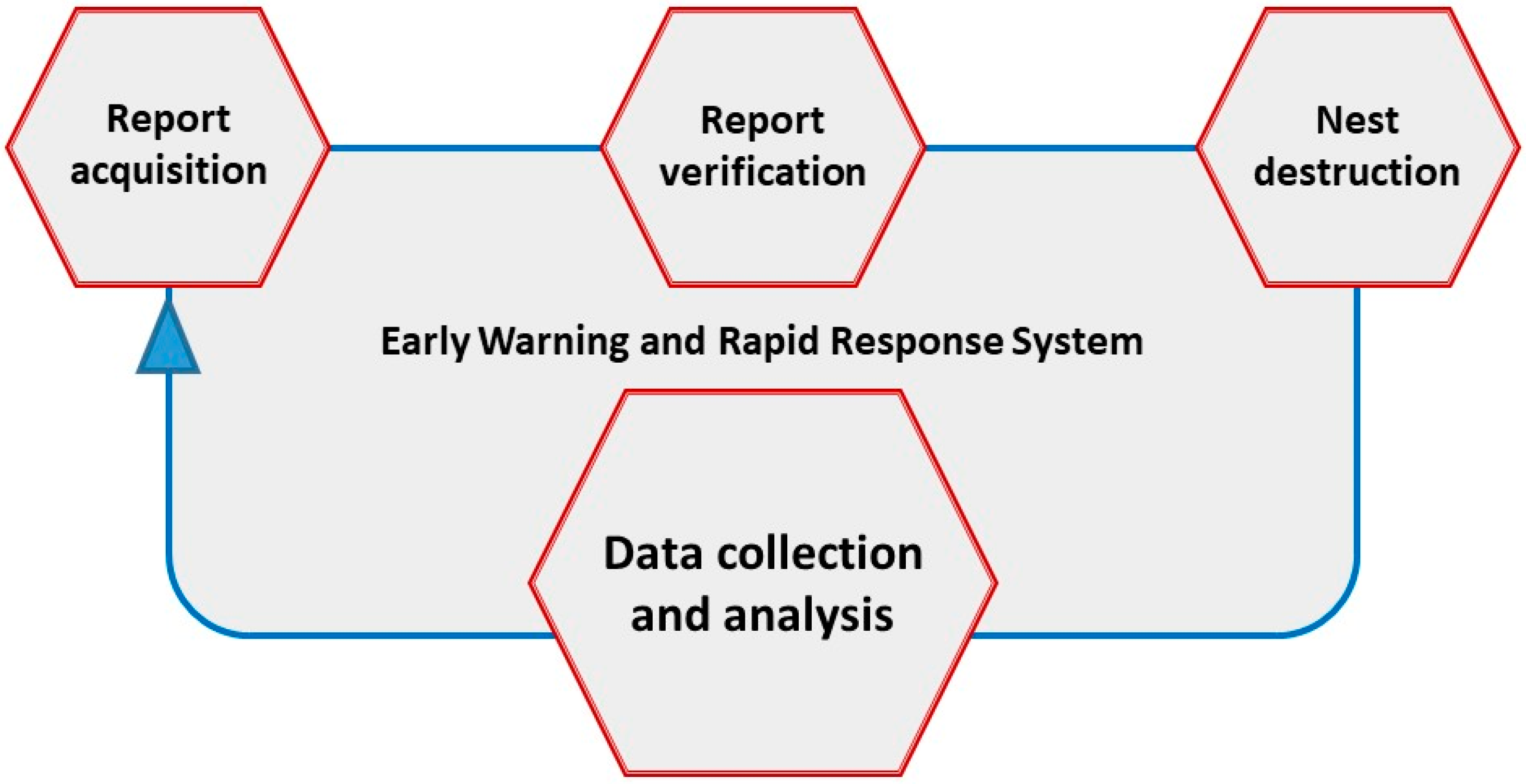
4. Monitoring and Surveillance Systems
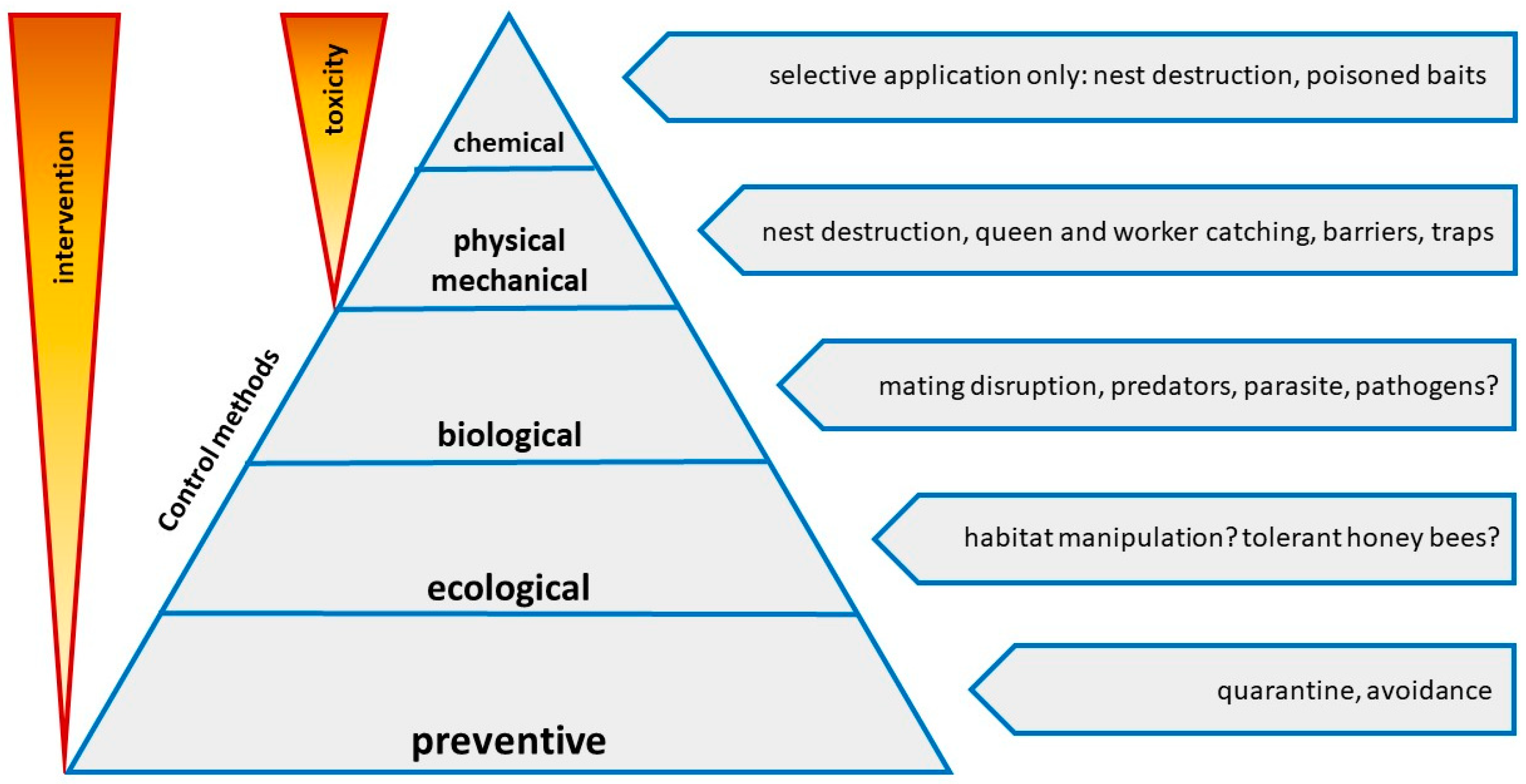
5. Control
5.1. Nest Detection
5.2. Biological Control
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Atkinson, I.A.E. Introductions of wildlife as a cause of species extinctions. Wildl. Biol. 1996, 2, 135–141. [Google Scholar] [CrossRef]
- Gandhi, K.J.K.; Herms, D.A. Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol. Invasions 2010, 12, 389–405. [Google Scholar] [CrossRef]
- Lever, C. Silent Summer: The State of Wildlife in Britain and Ireland—Vertebrate Animal Introductions; Maclean, N., Ed.; Cambridge University Press: Cambridge, UK, 2010; Volume 4, pp. 36–52. [Google Scholar]
- Van der Vecht, J. The Vespinae of the Indo Malayan and Papuan areas (Hymenoptera, Vespidae). Zool. Verh. 1957, 34, 1–83. [Google Scholar]
- Van der Vecht, J. Notes on Oriental Vespinae, including some species from China and Japan (Hymenoptera, Vespidae). Zool. Meded. 1959, 36, 205–232. [Google Scholar]
- Archer, M.E. Taxonomy, distribution and nesting biology of the Vespa bicolor group (Hym., Vespinae). Entomol. Mon. Mag. 1994, 130, 149–158. [Google Scholar]
- Perrard, A.; Arca, M.; Rome, Q.; Muller, F.; Tan, J.; Bista, S.; Ho, H.N.; Baudoin, R.; Baylac, M.; Silvain, J.F.; et al. Geographic variation of melanisation patterns in a hornet species: Genetic differences, climatic pressures or aposematic constraints? PLoS ONE 2014, 9, e94162. [Google Scholar] [CrossRef]
- Archer, M.E. Vespine Wasps of the World. Behaviour, Ecology and Taxonomy of the Vespinae; Monograph Series; Siri Scientific Press: Manchester, UK, 2012; Volume 4, pp. 1–352. ISBN 978-0-9567795-7-1. [Google Scholar]
- Choi, M.B.; Martin, S.J.; Lee, J.W. Distribution, spread, and impact of the invasive hornet Vespa velutina in South Korea. J. Asia Pac. Entomol. 2012, 15, 473–477. [Google Scholar] [CrossRef]
- Sakay, Y.; Takahashi, J. Discovery of a worker of Vespa velutina (Hymenoptera: Vespidae) from Tsushima Island, Japan. Jpn. J. Entomol. 2014, 17, 32–36. [Google Scholar]
- Ueno, T. Establishment of the invasive hornet Vespa velutina (Hymenoptera: Vespidae) in Japan. Int. J. Chem. Environ. Biol. Sci. 2014, 2, 220–222. [Google Scholar]
- Minoshima, Y.N.; Yamane, S.; Ueno, T. An invasive alien hornet, Vespa velutina nigrithorax du Buysson (Hymenoptera, Vespidae), found in Kitakyushu, Kyushu Island: A first record of the species from mainland Japan. Jpn. J. Syst. Entmol. 2015, 21, 259–261. [Google Scholar]
- Takahashi, J.; Okuyama, H.; Kiyoshi, T.; Takeuchi, T.; Martin, S.J. Origins of Vespa velutina hornets that recently invaded Iki Island, Japan and Jersey Island, UK. Mitochondrial DNA 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Haxaire, J.; Bouguet, J.P.; Tamisier, J.P. Vespa velutina Lepeletier, 1836, une redoutable nouveauté pour la faune de France (Hym., Vespidae). Bull. Soc. Entomol. Fr. 2006, 111, 194. [Google Scholar]
- Villemant, C.; Haxaire, J.; Streito, J.L. Premier bilan de l’invasion de Vespa velutina Lepeletier en France (Hymenoptera, Vespidae). Bull. Soc. Entomol. Fr. 2006, 111, 535–538. [Google Scholar]
- Castro, L.; Pagola-Carte, S. Vespa velutina Lepeletier, 1836 (Hymenoptera: Vespidae) recolectada en la Penınsula Iberica. Heteropterus Rev. Entomol. 2010, 10, 193–196. [Google Scholar]
- Lopéz, S.; Gonzalez, M.; Goldarazena, A. Vespa velutina Lepeletier, 1836 Hymenoptera: Vespidae): First records in Iberian Peninsula. Bull. OEPP/EPPO Bull. 2011, 41, 439–441. [Google Scholar] [CrossRef]
- Rodríguez-Flores, M.S.; Seijo-Rodríguez, A.; Escuredo, O.; Seijo-Coello, M.C. Spreading of Vespa velutina in northwestern Spain: Influence of elevation and meteorological factors and effect of bait trapping on target and non-target living organisms. J. Pest Sci. 2019, 92, 557–565. [Google Scholar] [CrossRef]
- Pujade-Villar, J.; Torrel, A.; Rojo, M. Confrimada la presència a CAtalunyad’una vespa originària d’Asia molt perillosa per als ruscs. Butll. Inst. Cat. Hist. Nat. 2012, 77, 173–176. [Google Scholar]
- Leza, M.; Miranda, M.A.; Colomar, V. First detection of Vespa velutina nigrithorax (Hymenoptera: Vespidae) in Balearic Islands (Western Mediterranean): A challenging study case. Biol. Invasions 2018, 1–7. [Google Scholar] [CrossRef]
- Leza, M.; Herrera, C.; Marques, A.; Roca, P.; Sastre Serra, J.; Pons, D.G. The impact of the invasive species Vespa velutina on honeybees: A new approach based on oxidative stress. Sci. Total Environ. 2019, 689, 709–715. [Google Scholar] [CrossRef]
- Grosso-Silva, J.M.; Maia, M. Vespa velutina Lepeletier, 1836 (Hymenoptera, Vespidae), new species for Portugal. AEGA 2012, 6, 53–54. [Google Scholar]
- Rome, Q.; Muller, F.; Villemant, C. Expansion en 2011 de Vespa velutina Lepeletier en Europe (Hym., Vespidae). Bull. Soc. Entomol. Fr. 2012, 117, 114. [Google Scholar]
- Demichelis, S.; Manino, A.; Minuto, G.; Mariotti, M.; Porporato, M. Social wasp trapping in north west Italy: Comparison of different bait-traps and first record of Vespa velutina Lepeletier (Hymenoptera: Vespidae). Bull. Insectol. 2014, 67, 307–317. [Google Scholar] [CrossRef]
- Porporato, M.; Manino, A.; Laurino, D.; Demichelis, S. Vespa velutina Lepeletier (Hymenoptera Vespidae): A first assessment two years after its arrival in Italy. Redia 2014, 97, 189–194. [Google Scholar]
- Bertolino, S.; Lioy, S.; Laurino, D.; Manino, A.; Porporato, M. Spread of the invasive yellow legged hornet Vespa velutina (Hymenoptera: Vespidae) in Italy. Appl. Entomol. Zool. 2016, 51, 589–597. [Google Scholar] [CrossRef]
- Lioy, S.; Manino, A.; Porporato, M.; Laurino, D.; Romano, A.; Capello, M.; Bertolino, S. Establishing surveillance areas for tackling the invasion of Vespa velutina in outbreaks and over the border of its expanding range. Neobiota 2019, 46, 51–69. [Google Scholar] [CrossRef]
- Witt, R. Erstfund eines Nestes der Asiatischen Hornisse Vespa velutina Lepeletier, 1838 in Deutschland und Details zum Nestbau (Himonoptera, Vespinae). Ampulex 2015, 7, 42–53. [Google Scholar]
- UK National Bee Unit. 2016. Available online: http://nationalbeeunit.com/ (accessed on 9 April 2019).
- Budge, G.E.; Hodgetts, J.; Jones, E.P.; Ostojà-Starzewski, J.C.; Tomkies, V.; Semmence, N.; Brown, M.; Stainton, K. The invasion provenance and diversity of Vespa velutina Lepeletier (Hymenoptera: Vespidae) in Great Britain. PLoS ONE 2017, 12, 1–12. [Google Scholar] [CrossRef]
- Villemant, C.; Barbet-Massin, M.; Perrard, A.; Muller, F.; Gargominy, O.; Jiguet, F.; Rome, Q. Predicting the invasion risk by the alien bee-hawking Yellow-legged hornet Vespa velutina nigrithorax across Europe and other continents with niche models. Biol. Conserv. J. 2011, 144, 2142–2150. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Rome, Q.; Muller, F.; Perrard, A.; Villemant, C. Climate change increases the risk of invasion by the Yellow-legged hornet. Biol. Conserv. 2013, 157, 4–10. [Google Scholar] [CrossRef]
- Kishi, S.; Goka, K. Review of the invasive yellow-legged hornet, Vespa velutina nigrithorax (Hymenoptera: Vespidae), in Japan and its possible chemical control. Appl. Entomol. Zool. 2017, 52, 361–368. [Google Scholar] [CrossRef]
- Rome, Q.; Muller, F.J.; Touret-Alby, A.; Darrouzet, E.; Perrard, A.; Villemant, C. Caste differentiation and seasonal changes in Vespa velutina (Hym.: Vespidae) colonies in its introduced range. J. Appl. Entomol. 2015, 139, 771–782. [Google Scholar] [CrossRef]
- Monceau, K.; Bonnard, O.; Thiery, D. Vespa velutina: A new invasive predator of honeybees in Europe. J. Pest Sci. 2014, 87, 1–16. [Google Scholar] [CrossRef]
- Wang, Z.; Qu, Y.; Dong, S.; Wen, P.; Li, J.; Tan, K.; Menzel, R. Honey bees modulate their olfactory learning in the presence of hornet predators and alarm component. PLoS ONE 2016, 11, 1–12. [Google Scholar] [CrossRef]
- Couto, A.; Monceau, K.; Bonnard, O.; Thiéry, D.; Sandoz, J.C. Olfactory attraction of the hornet Vespa velutina to honeybee colony odors and pheromones. PLoS ONE 2014, 9, 1–19. [Google Scholar] [CrossRef]
- Laurino, D.; Porporato, M. Vespa Velutina—Conoscerla e Prepararsi ad Affrontare il Pericolo, 1st ed.; Edizioni Montaonda: San Godenzo, Italy, 2017; p. 67. ISBN 9-78898-186228. [Google Scholar]
- Wang, Z.W.; Chen, G.; Tan, K. Both olfactory and visual cues promote the hornet Vespa velutina to locate its honeybee prey Apis Cerana. Insectes Soc. 2014, 61, 67–70. [Google Scholar] [CrossRef]
- Turchi, L.; Derijard, B. Options of the biological and physical control of Vespa velutina nigrithorax (Hym.: Vespidae) in Europe: A review. J. Appl. Entomol. 2018, 1, 10. [Google Scholar] [CrossRef]
- Couto, A.; Lapeyre, B.; Thiery, D.; Sandoz, J.C. Olfactory pathway of the hornet Vespa velutina: New insights into the evolution of the hymenopteran antennal lobe. J. Comp. Neurol. 2016, 524, 2335–2359. [Google Scholar] [CrossRef]
- Wen, P.; Cheng, Y.N.; Dong, S.H.; Wang, Z.W.; Tan, K.; Nieh, J.C. The sex pheromone of a globally invasive honey bee predator, the Asian eusocial hornet, Vespa velutina. Sci. Rep. 2017, 7, 12956. [Google Scholar] [CrossRef]
- Cheng, Y.; Wen, P.; Dong, S.; Tan, K.; Nieh, J.C. Poison and alarm: The Asian hornet Vespa velutina uses sting venom volatiles as alarm pheromone. J. Exp. Biol. 2017, 220, 645–651. [Google Scholar] [CrossRef]
- Fournier, A.; Barbet-Massin, M.; Rome, Q.; Courchamp, F. Predicting species distribution combining multi-scale drivers. Glob. Ecol. Conserv. 2017, 12, 215–226. [Google Scholar] [CrossRef]
- Robinet, C.; Suppo, C.; Darrouzet, E. Rapid spread of the invasive yellow-legged hornet in France: The role of human-mediated dispersal and the effects of control measures. J. Appl. Ecol. 2017, 54, 205–215. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Rome, Q.; Villemant, C.; Courchamp, F. Can species distribution models really predict the expansion of invasive species? PLoS ONE 2018, 13, e0193085. [Google Scholar] [CrossRef] [PubMed]
- Beggs, J.R.; Brockerhoff, E.G.; Corley, J.C.; Kenis, M.; Masciocchi, M.; Muller, F.; Rome, Q.; Villemant, C. Ecological effects and management of invasive alien Vespidae. BioControl 2011, 56, 505–526. [Google Scholar] [CrossRef]
- Monceau, K.; Maher, N.; Bonnard, O.; Thiery, D. Predation pressure dynamics study of the recently introduced honeybee killer Vespa velutina: Learning from the enemy. Apidologie 2013, 44, 209–221. [Google Scholar] [CrossRef]
- Monceau, K.; Bonnard, O.; Moreau, J.; Thiéry, D. Spatial distribution of Vespa velutina individuals hunting at domestic honeybee hives: Heterogeneity at a local scale. Insect Sci. 2014, 21, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Moller, H. Lessons for invasion theory from social insects. Biol. Conserv. 1996, 78, 125–142. [Google Scholar] [CrossRef]
- Martin, S.J. The Asian hornet: Threats, Biology & Expansion; The International Bee Research Association: England and Wales, 2017; p. 106. ISBN 9780860982814. [Google Scholar]
- De Haro, L.; Labadie, M.; Chanseau, P.; Cabot, C.; Blanc-Brisset, I.; Penouil, F. Medical consequences of the Asian black hornet (Vespa velutina) invasion in Southwestern France. Toxicon 2010, 55, 650–652. [Google Scholar] [CrossRef]
- Feàs Sanchez, X.; Charles, R.J. Notes on the Nest Architecture and Colony Composition in Winter of the Yellow-Legged Asian Hornet, Vespa velutina Lepeletier 1836 (Hym.: Vespidae), in Its Introduced Habitat in Galicia (NW Spain). Insects 2019, 10, 237. [Google Scholar] [CrossRef]
- Liu, Z.R.; Chen, S.G.; Zhou, Y.; Xie, C.H.; Zhu, B.F.; Zhu, H.M.; Liu, S.P.; Wang, W.; Chen, H.Z.; Ji, Y.H. Deciphering the Venomic Transcriptome of Killer-Wasp Vespa velutina. Sci. Rep. 2015, 5, 9454. [Google Scholar] [CrossRef]
- Tabar, A.I.; Chugo, S.; Joral, A.; Lizaso, M.T.; Lizarza, S.; Alvarez-Puebla, M.J.; Arroabarren, E.; Vela, C.; Lombardero, M. Vespa velutinanigrithorax: A new causative agent for anaphylaxis. Clin. Transl. Allergy 2015, 5, 43. [Google Scholar] [CrossRef]
- Sumner, S.; Law, G.; Cini, A. Why we love bees and hate wasps. Ecol. Entomol. 2018, 43, 836–845. [Google Scholar] [CrossRef]
- Requier, F.; Rome, Q.; Chiron, G.; Decante, D.; Marion, S.; Menard, M.; Muller, F.; Villemant, C.; Henry, M. Predation of the invasive Asian hornet affects foraging activity and survival probability of honey bees in Western Europe. J. Pest Sci. 2019, 92, 567–578. [Google Scholar] [CrossRef]
- Monceau, K.; Thiery, D. Vespa velutina: Current situation and perspectives. Atti Accad. Naz. Ital. Entomol. 2016, 64, 137–142. [Google Scholar]
- Helmer, S.H.; Kerbaol, A.; Aras, P.; Jumarie, C.; Boily, M. Effects of realistic doses of atrazine, metolachlor, and glyphosate on lipid peroxidation and diet-derived antioxidants in caged honey bees (Apis mellifera). Environ. Sci. Pollut. Res. 2015, 22, 8010–8021. [Google Scholar] [CrossRef]
- Rome, Q.; Perrard, A.; Muller, F.; Villemant, C. Monitoring and control modalities of a honeybee predator, the Yellow-legged hornet Vespa velutina nigrithorax (Hymenoptera: Vespidae). Aliens Invasive Spec. Bull. 2011, 31, 7–15. [Google Scholar]
- Ken, T.; Hepburn, H.R.; Radloff, S.E.; Yusheng, Y.; Yiqiu, L.; Danyin, Z.; Neumann, P. Heat-balling wasps by honeybees. Naturwissenschaften 2005, 92, 492–495. [Google Scholar] [CrossRef]
- Genovesi, P.; Scalera, R.; Brunel, S.; Solarz, W.; Roy, D. Towards an Early Warning and Information System for Invasive Alien Species (IAS) Threatening Biodiversity in Europe; Tech. Report 5/2010; European Environment Agency: Copenhagen, Denmark, 2010; 52p. [Google Scholar] [CrossRef]
- Rojas-Nossa, S.V.; Novoa, N.; Serrano, A.; Calviño-Cancela, M. Performance of baited traps used as control tools for the invasive hornet Vespa velutina and their impact on non-target insects. Apidologie 2018, 49, 872–885. [Google Scholar] [CrossRef]
- Blot, J. Localisation et destrution des nids de frelons asiatiques. Bull. Tech. Apic. 2008, 35, 95–100. [Google Scholar]
- Jeffs, K. Wild China BBC, Hornet Sequence between 42′ and 45′. 2008. Available online: http://www.bbc.co.uk/iplayer/episode/b==brvjx/wild-china-2-shangrila; https://vimeo.com/7877242 (accessed on 15 October 2019).
- Reynaud, B.; Guérin-Lassous, I. Design of a force-based controlled mobility on aerial vehicles for pest management. Ad Hoc Netw. 2016, 53, 41–52. [Google Scholar] [CrossRef]
- Semmence, N. Asian Hornet Update from the National Bee Unit. Incorporating the British Bee J. BBKA News, November 2018; 270–272. [Google Scholar]
- Kennedy, P.; Ford, S.M.; Poidatz, J.; Thiéry, D.; Osborne, J.L. Searching for nests of the invasive Asian hornet (Vespa velutina) using radio-telemetry. Commun. Biol. 2018, 1, 88. [Google Scholar] [CrossRef]
- Bortolotti, L.; Cervo, R.; Felicioli, A.; Quaranta, M.; Salvetti, O.; Berton, A. Progetto Velutina: La ricerca italiana a caccia di soluzioni. Atti Accad. Naz. Ital. Entomol. 2016, 64, 143–149. [Google Scholar]
- Milanesio, D.; Saccani, M.; Maggiora, R.; Laurino, D.; Porporato, M. Design of an harmonic radar for the tracking of the Asian yellow-legged hornet. Ecol. Evol. 2016, 6, 2170–2217. [Google Scholar] [CrossRef] [PubMed]
- Milanesio, D.; Saccani, M.; Maggiora, R.; Laurino, D.; Porporato, M. Recent upgrades of the harmonic radar for the tracking of the Asian yellow-legged hornet. Ecol. Evol. 2017, 7, 4599–4606. [Google Scholar] [CrossRef] [PubMed]
- Maggiora, R.; Saccani, M.; Milanesio, D.; Porporato, M. An Innovative Harmonic Radar to Track Flying Insects: The Case of Vespa velutina. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Yamane, S. New taxa of the genius Bareogonalos from Asia with further information on the tribe Nomadinini (Hymenoptera, Trigonalidae). Halteres 2014, 5, 17–31. [Google Scholar]
- Darrouzet, E.; Gérvar, J.; Dupont, S. A scientific note about a parasitoid that can parasitize the yellow-legged hornet, Vespa velutina nigrithorax, in Europe. Apidologie 2014, 46, 130–132. [Google Scholar] [CrossRef]
- Spradbery, J.P. Chap 6: Feeding and Foraging. In Wasps. An Account of the Biology and Natural History of Solitary and Social Wasps; Sidgwick & Jackson: London, UK, 1973; pp. 128–157. [Google Scholar]
- Villemant, C.; Zuccon, D.; Rome, Q.; Muller, F.; Poinar, G.O., Jr.; Justine, J.L. Des parasites peuvent-ils stopper l’invasion? Des nématodes mermithidés parasitent le frelon asiatique à pattes jaunes en France. Peer J. 2015, 947. [Google Scholar] [CrossRef]
- Harris, R.; Harcourt, S.J.; Glare, T.R.; Rose, E.A.F.; Nelson, T.J. Susceptibility of Vespula vulgaris (Hymenoptera: Vespidae) to Generalist Entomopathogenic Fungi and their potential for wasp control. J. Invertebr. Pathol. 2000, 75, 251–258. [Google Scholar] [CrossRef]
- Poidatz, J.; López Plantey, R.; Thiéry, D. Indigenous strains of Beauveria and Metharizium as potential biological control agents against the invasive hornet Vespa velutina. J. Invertebr. Pathol. 2018, 153, 180–185. [Google Scholar] [CrossRef]
- Macià, F.X.; Menchetti, M.; Corbella, C.; Grajera, J.; Vila, R. Exploitation of the invasive Asian Hornet Vespa velutina by the European Honey Buzzard Pernis apivorus. Bird Study 2019, 66, 425–429. [Google Scholar] [CrossRef]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laurino, D.; Lioy, S.; Carisio, L.; Manino, A.; Porporato, M. Vespa velutina: An Alien Driver of Honey Bee Colony Losses. Diversity 2020, 12, 5. https://doi.org/10.3390/d12010005
Laurino D, Lioy S, Carisio L, Manino A, Porporato M. Vespa velutina: An Alien Driver of Honey Bee Colony Losses. Diversity. 2020; 12(1):5. https://doi.org/10.3390/d12010005
Chicago/Turabian StyleLaurino, Daniela, Simone Lioy, Luca Carisio, Aulo Manino, and Marco Porporato. 2020. "Vespa velutina: An Alien Driver of Honey Bee Colony Losses" Diversity 12, no. 1: 5. https://doi.org/10.3390/d12010005
APA StyleLaurino, D., Lioy, S., Carisio, L., Manino, A., & Porporato, M. (2020). Vespa velutina: An Alien Driver of Honey Bee Colony Losses. Diversity, 12(1), 5. https://doi.org/10.3390/d12010005






