The Afro–Oriental Genus Yaeprimus Sasa et Suzuki (Diptera: Chironomidae: Chironomini): Phylogeny, New Species and Expanded Diagnoses
Abstract
1. Introduction
2. Materials and Methods
2.1. Morphology
2.2. Molecular Work
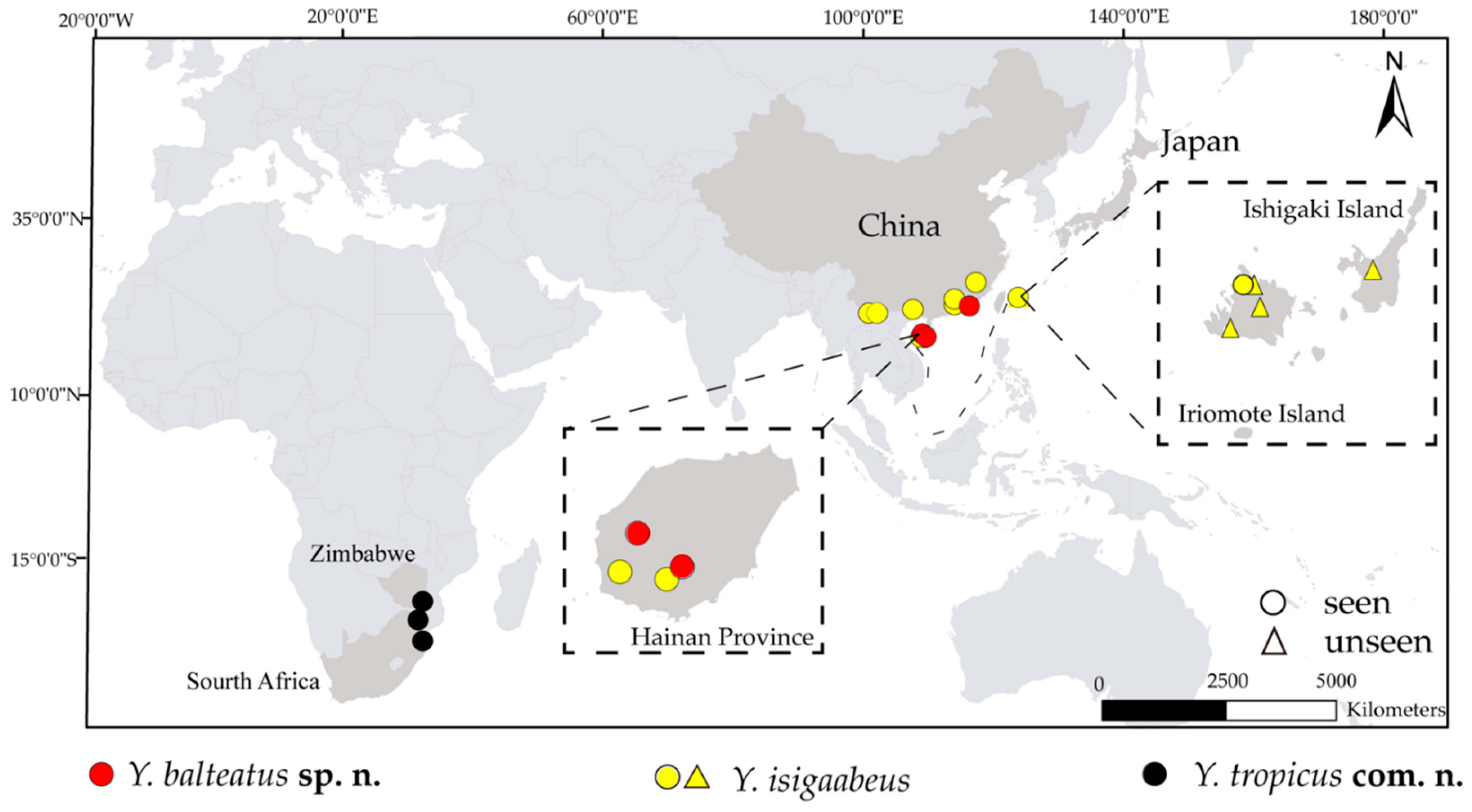
2.3. Mapping
2.4. Abrreviation
3. Results
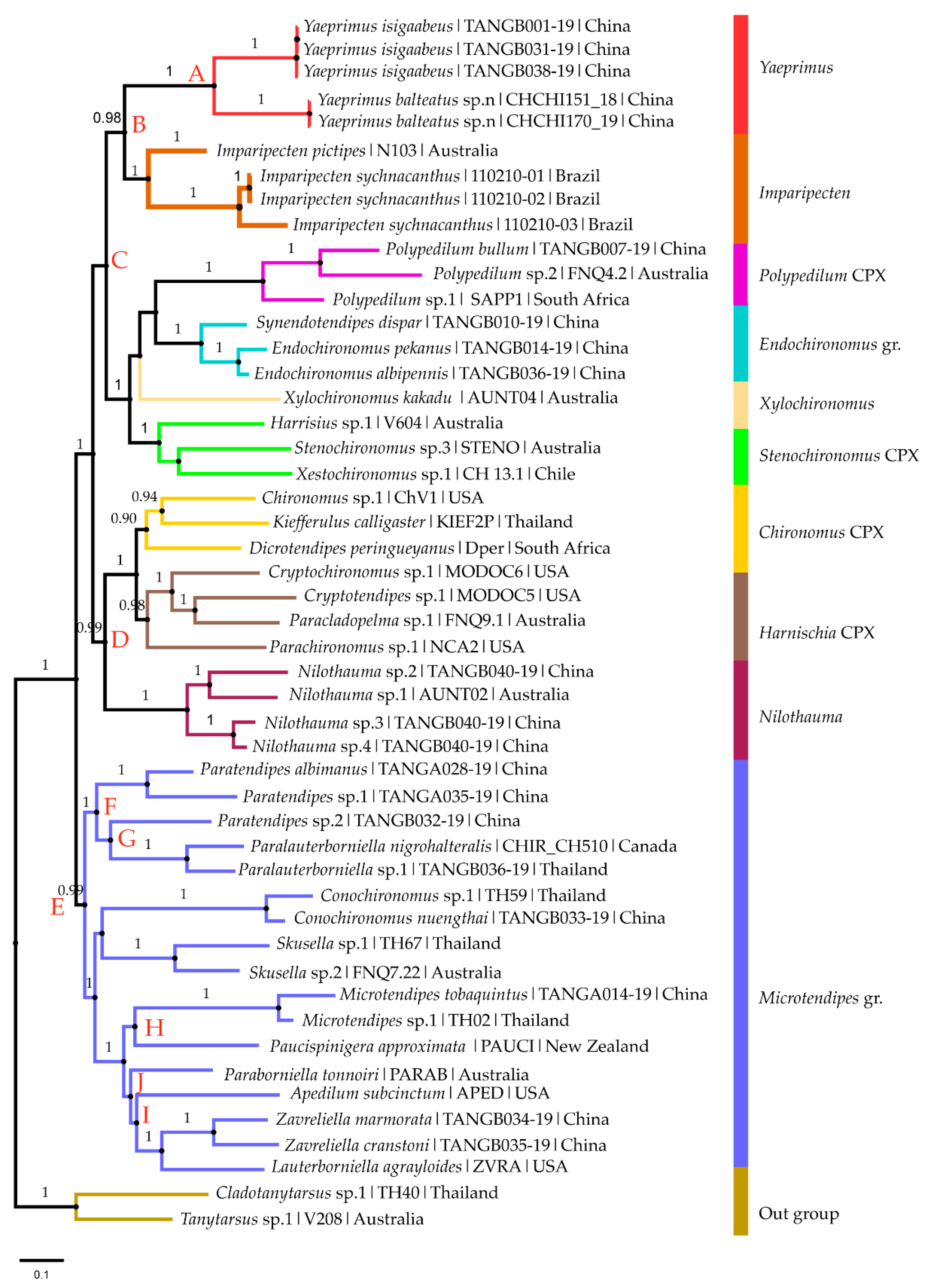
3.1. Molecular Analysis
3.1.1. DNA Barcode
3.1.2. Phylogenetic Analysis
3.2. Morphology
3.2.1. Generic Diagnosis Emendation
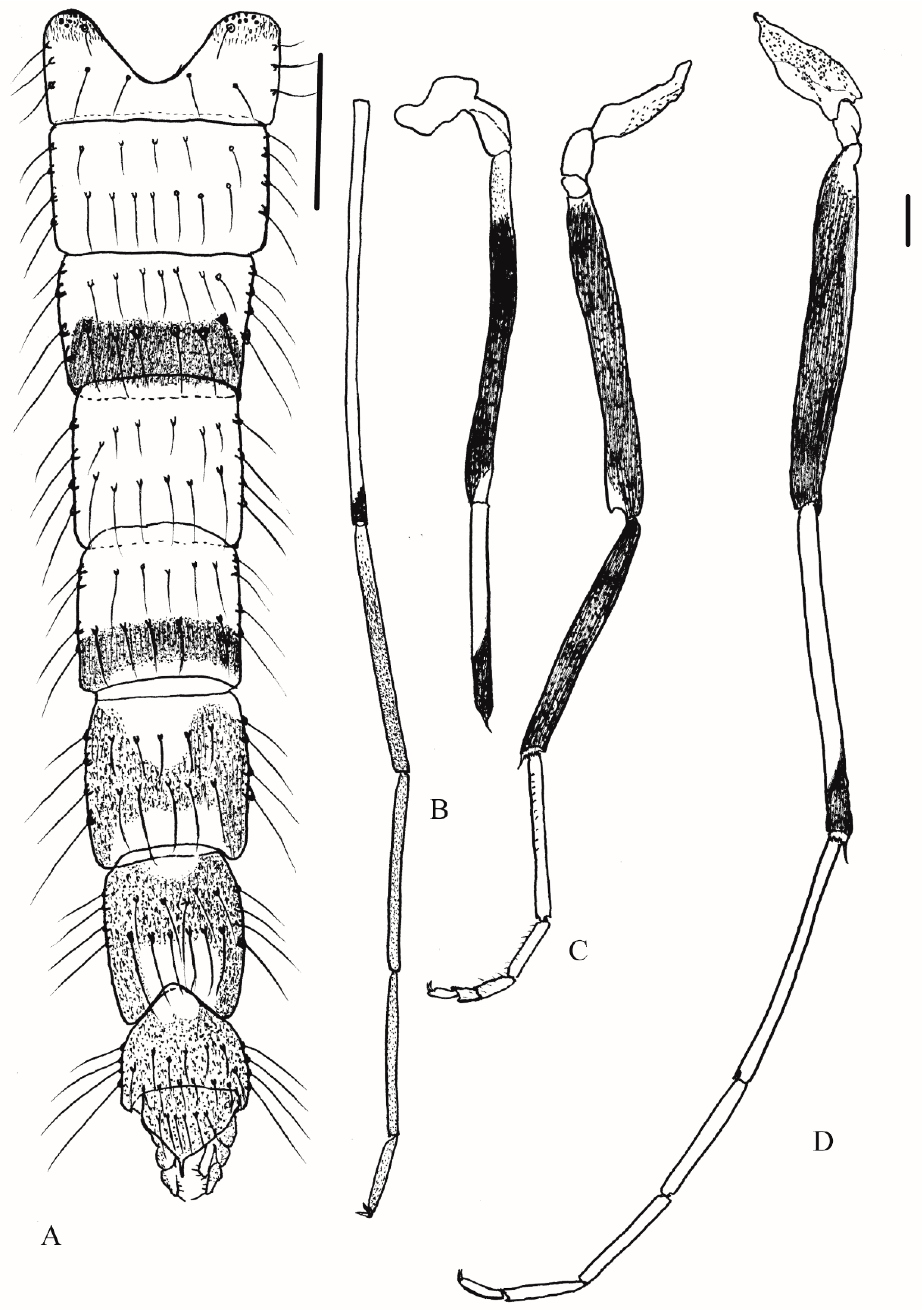
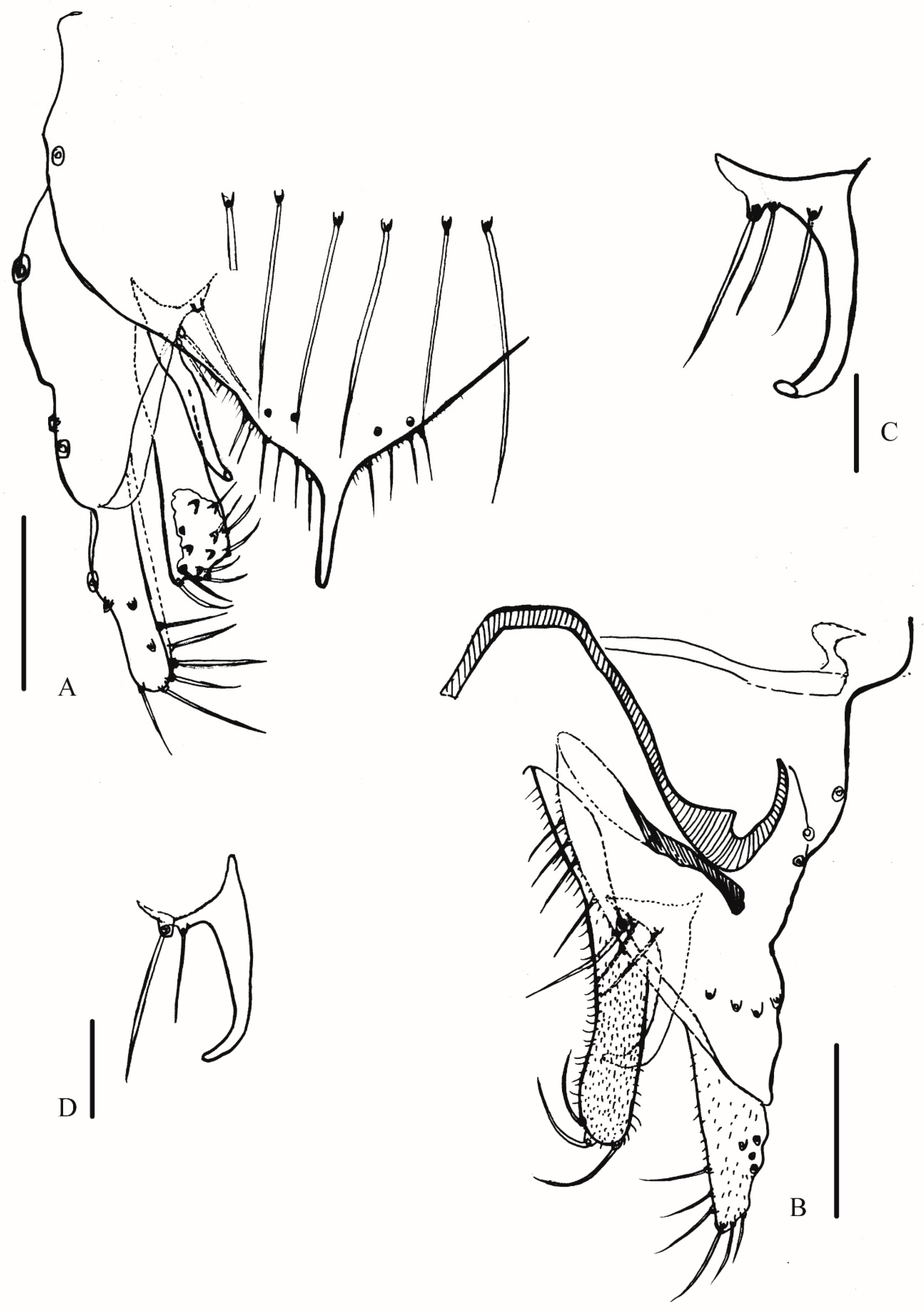
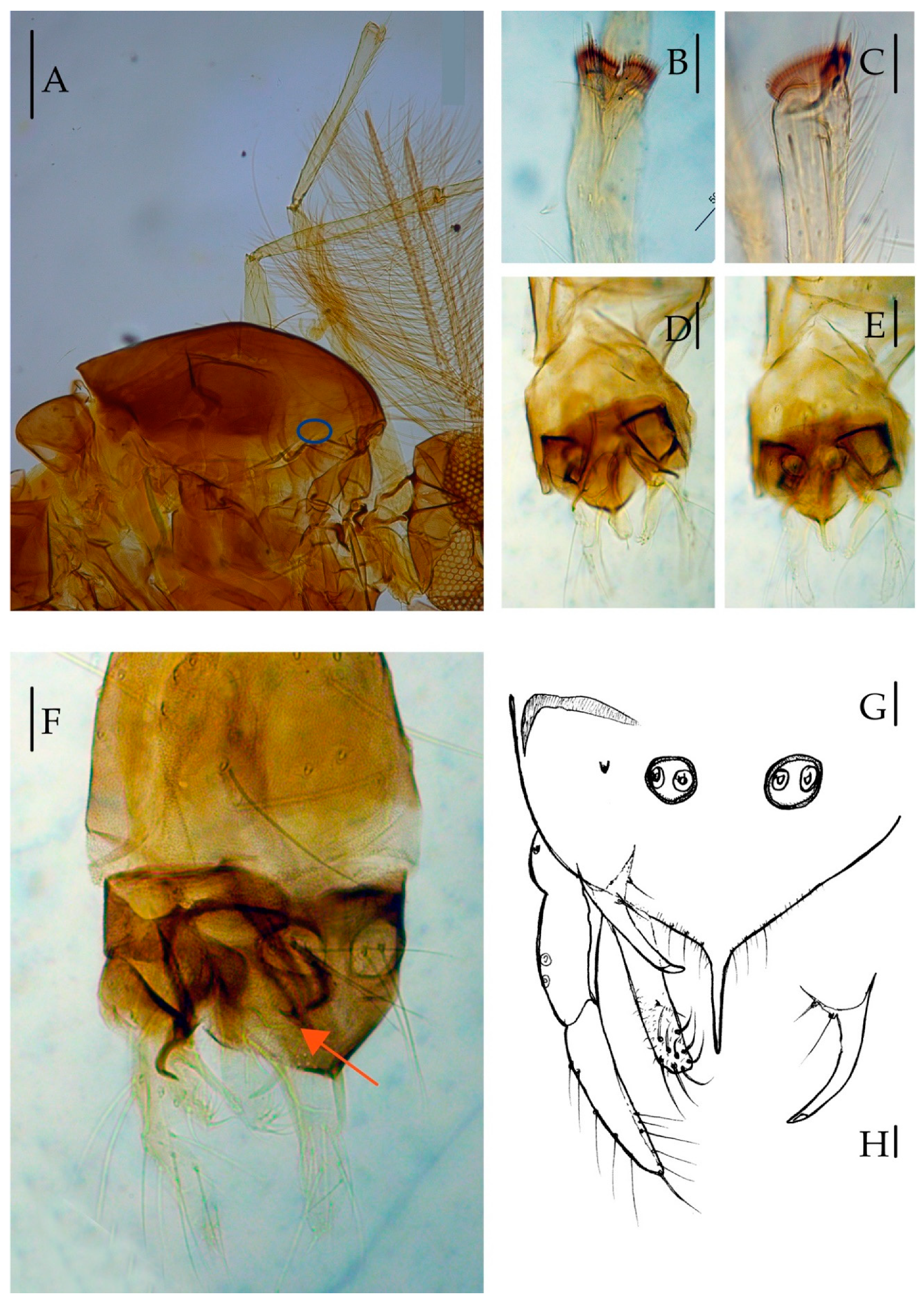
3.2.2. Description of Species
Yaeprimus balteatus Han et Tang, sp. n.
Yaeprimus isigaabeus Sasa et Suzuki
Yaeprimus tropicus comb. n.
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Gene | Name | Sequence | Reference |
|---|---|---|---|
| 18S rDNA | 18S | CCTGAGAAACGGCTACCACATC | Whiting et al. (1997) [52] |
| 18S rDNA | 18S | GAGTCTCGTTCGTTATCGGA | Whiting et al. (1997) [52] |
| 28S rDNA | S3660 | GAGAGTTMAASAGTACGTGAAAC | Morse and Normark (2006) [53] |
| 28S rDNA | A335 | TCGGAAGGAACCAGCTACTA | Whiting et al. (1997) [52] |
| CAD1 | 54F | GTNGTNTTYCARACNGGNATGGT | Moulton and Wiegmann (2004) [54] |
| CAD1 | 405R | GCNGTRTGYTCNGGRTGRAAYTG | Moulton and Wiegmann (2004) [54] |
| CAD4 | 787F | GGDGTNACNACNGCNTGYTTYGARCC | Moulton and Wiegmann (2004) [54] |
| CAD4 | 1098R | TTNGGNAGYTGNCCNCCCAT | Moulton and Wiegmann (2004) [54] |
| COI-5P | LCO1490 | GGTCAACAAATCATAAAGATATTGG | Folmer et al. (1994) [55] |
| COI-5P | HCO2198 | TAAACTTCAGGGTGACCAAAAAATCA | Folmer et al. (1994) [55] |
| COI-3P | S2183 | CAACATTTATTTTGATTTTTTGG | Simon et al. (1994) [56] |
| COI-3P | A3014 | TCCAATGCACTAATCTGCCATATTA | Simon et al. (1994) [56] |
| Species Name | ID | 28S | COI–3P | 18S | CADIV | CADI | COI–5P |
|---|---|---|---|---|---|---|---|
| Apedilum subcinctum | APED | HQ440708 | HQ440869 | HQ440558 | HQ440416 | HQ440230 | N |
| Chironomus sp. 1 | ChV1 | HQ440719 | HQ440883 | HQ440572 | HQ440428 | HQ440256 | N |
| Cladotanytarsus sp. 1 | TH40 | HQ440720 | HQ440884 | HQ440573 | HQ440429 | HQ440257 | N |
| Conochironomus tobaterdecimus | TH59 | HQ440724 | HQ440888 | HQ440577 | HQ440433 | HQ440261 | N |
| Cryptochironomus sp. 1 | MODOC6 | HQ440729 | HQ440893 | HQ440582 | HQ440438 | HQ440266 | N |
| Cryptotendipes sp. 1 | MODOC5 | HQ440730 | HQ440894 | HQ440583 | HQ440439 | HQ440267 | N |
| Dicrotendipes peringueyanus | Dper | HQ440732 | HQ440896 | HQ440585 | HQ440440 | HQ440270 | N |
| Harrisius sp. 1 | V604 | HQ440754 | HQ440916 | HQ440603 | HQ440456 | HQ440288 | N |
| Imparipecten pictipes | N103 | HQ440759 | HQ440921 | HQ440608 | HQ440461 | HQ440294 | N |
| Imparipecten sychnacanthus | 110210-01 | MH131689 | MH602431 | N | MH602428 | MH558540 | N |
| Imparipecten sychnacanthus | 110210-02 | MH131690 | MH602432 | N | MH602429 | MH558541 | N |
| Imparipecten sychnacanthus | 110210-03 | MH131691 | MH602433 | N | MH602430 | MH558542 | N |
| Kiefferulus calligaster | KIEF2P | HQ440763 | HQ440924 | HQ440611 | HQ440464 | HQ440298 | N |
| Lauterborniella agrayloides | ZVRA | HQ440766 | HQ440927 | HQ440613 | HQ440467 | HQ440301 | N |
| Microtendipes sp. 1 | TH02 | HQ440776 | HQ440937 | HQ440622 | N | N | N |
| Nilothauma sp. 1 | AUNT02 | HQ440782 | HQ440945 | HQ440629 | HQ440481 | HQ440316 | N |
| Paucispinigera approximate | PAUCI | HQ440806 | HQ440969 | HQ440649 | HQ440500 | HQ440338 | N |
| Paraborniella tonnoiri# | PARAB | HQ440789 | HQ440952 | HQ440789 | HQ440485 | N | N |
| Parachironomus sp. 1 | NCA2 | HQ440791 | HQ440954 | HQ440635 | HQ440486 | HQ440323 | N |
| Paracladopelma sp. 1 | FNQ9.1 | HQ440793 | HQ440956 | HQ440637 | HQ440488 | HQ440325 | N |
| Polypedilum sp. 1 | SAPP1 | HQ440813 | HQ440977 | HQ440657 | HQ440506 | HQ440346 | N |
| Polypedilum sp. 2 | FNQ4.2 | HQ440815 | HQ440979 | HQ440659 | HQ440508 | HQ440349 | N |
| Skusella sp. 1 | TH67 | HQ440831 | HQ440994 | HQ440672 | N | HQ440367 | N |
| Skusella sp. 2 | FNQ7.22 | HQ440832 | HQ440995 | HQ440673 | HQ440525 | HQ440368 | N |
| Stenochironomus sp. 3 | STENO | HQ440837 | HQ441000 | HQ440677 | HQ440529 | HQ440372 | N |
| Tanytarsus sp. 1 | V208 | HQ440846 | HQ441009 | HQ440686 | HQ440537 | HQ440382 | N |
| Xestochironomus sp. 1 | CH 13.1 | HQ440861 | HQ441023 | HQ440700 | HQ440549 | HQ440401 | N |
| Xylochironomus Kakadu | AUNT04 | HQ440863 | HQ441025 | HQ440702 | HQ440551 | HQ440403 | N |
| Conochironomus nuengthai * | TANGB033-19 | Y | Y | Y | N | N | Y |
| Endochironomus albipennis * | TANGB036-19 | Y | N | Y | Y | Y | Y |
| Endochironomus pekanus * | TANGB014-19 | Y | Y | Y | Y | Y | Y |
| Microtendipes tobaquintus * | TANGA014-19 | Y | Y | Y | Y | Y | Y |
| Nilohtauma sp. 4 * | TANGB042-19 | Y | Y | N | Y | N | Y |
| Nilothauma sp. 2 * | TANGB040-19 | Y | Y | N | Y | Y | Y |
| Nilothauma sp. 3 * | TANGB041-19 | Y | Y | N | Y | N | Y |
| Paralauterborniella nigrohalteralis * | CHIR_CH510 | Y | Y | Y | Y | Y | N |
| Paratendipes albimanus * | TANGA028-19 | Y | Y | Y | Y | Y | Y |
| Paratendipes alpinus * | TANGA035-19 | Y | Y | N | Y | Y | Y |
| Paratendipes sp. 2 * | TANGA032-19 | Y | N | Y | Y | Y | Y |
| Polypedilum bullum | TANGB007-19 | Y | Y | Y | Y | N | Y |
| Polypedilum bullum * | TANGB007-19 | Y | N | N | Y | Y | Y |
| Syendotendipes dispar * | TANGB010-19 | Y | Y | Y | Y | Y | Y |
| Yaeprimus balteatus sp. n. * | CHCHI170-19 | Y | Y | Y | Y | Y | N |
| Yaeprimus balteatus sp. n. * | CHCHI151-18 | Y | Y | N | N | Y | N |
| Yaeprimus balteatus sp. n. * | CHCHI185-19 | Y | N | N | N | N | N |
| Yaeprimus isigaabeus * | TANGB031-19 | Y | Y | N | N | N | Y |
| Yaeprimus isigaabeus * | TANGB001-19 | Y | Y | Y | Y | N | Y |
| Yaeprimus isigaabeus * | TANGB038-19 | Y | Y | N | Y | N | Y |
| Yaeprimus isigaabeus * | TANGB032-19 | Y | Y | Y | Y | N | N |
| Zavreliella cranstoni * | TANGB035-19 | N | Y | Y | Y | N | Y |
| Zavreliella marmorata * | TANGB034-19 | N | Y | Y | N | Y | Y |
References
- Sasa, M.; Suzuki, H. Stuidies on the chironomid species collected on Ishigaki and Iriomote Islands, Southwestern Japan. Trop. Med. 2000, 42, gz7. [Google Scholar]
- Yamamoto, M.; Yamamoto, N. A review of Yaeprimus isigaabeus Sasa et Suzuki, 2000 (Diptera: Chironomidae), with taxonomic notes on the genera distributed in Yaeyama Island. In Proceedings of the 17th Intertional Symposium on Chironomidae, Nankai University, Tianjin, China, 6–10 July 2009; Wang, X.H., Liu, W., Eds.; Nankai University Press: Tianjin, China, 2009; pp. 224–239. [Google Scholar]
- Cranston, P.S.; Hardy, N.B.; Morse, G.E. A dated molecular phylogeny for the Chironomidae (Diptera). Syst. Entomol. 2012, 37, 172–188. [Google Scholar] [CrossRef]
- Harrison, A.D. Four new genera and species of Chironomidae (Diptera) from southern Africa. Aquat. Insects 2000, 22, 219–236. [Google Scholar] [CrossRef]
- Tang, H.Q. (Jinan University, Guangzhou, Guangdong, China); Helen, B.J. (Albany Museum, Grahamstown, South Africa). Personal communication, 2019.
- Tang, H.Q. (Jinan University, Guangzhou, Guangdong, China); Martin, S. (Zoologische Staatssammlung München, Munich, Germany). Personal communication, 2019.
- Tang, H.Q.; Jinan University, Guangzhou, Guangdong, China; Zhang, R.L.; Shanghai Ocean University, Shanghai, China); Lin, X.L. (Nankai University, Tianjin, China. Personal communication, 2019.
- Tang, H.Q. Paralauterborniella from Oriental China (Diptera: Chironomidae). Orient. Insects 2016, 50, 160–170. [Google Scholar] [CrossRef]
- Tang, H.Q.; Cranston, P.S. A new tribe in the Chironominae (Diptera: Chironomidae) validated by first immature stages of Xiaomyia Sæther & Wang and a phylogenetic review. Raffles B. Zool. 2019, 67, 684–693. [Google Scholar] [CrossRef]
- Wu, H.; Tang, H.Q. Phylogeny of marine Ainuyusurika tuberculata (Tokunaga) (Diptera: Chironomidae: Chironominae), with description of the immature stages. Zootaxa 2019, 4695, 131–147. [Google Scholar] [CrossRef]
- Niitsuma, H.; Tang, H.Q. Taxonomic review of Ablabesmyia Johannsen (Diptera: Chironomidae: Tanypodinae) from Oriental China, with descriptions of six new species. Zootaxa 2019, 4564, 248–270. [Google Scholar] [CrossRef]
- Cranston, P.S. Electronic Identification Guide to the Australian Chironomidae. Available online: http://entomology/ucdavis.edu/chiropage/index.html (accessed on 30 October 2019).
- Cranston, P.S.; Dillon, M.E.; Pinder, L.C.V.; Reiss, F. The adult males of Chironominae (Diptera: Chironomidae) of the Holarctic region—Keys and diagnoses. In Chironomidae of the Holarctic Region; Wiederholm, T., Ed.; Keys and Diagnoses, Part 3, Adult Males; Entomological Society of Lund: Lund, Sweden, 1989; Volume 34, pp. 353–502. [Google Scholar]
- Langton, P.H. A Key to Pupal Exuaviae of West Palaearctic Chironomidae; P. Langton: Swanage, UK, 1991; pp. 222–311. [Google Scholar]
- Saether, O.A. Glossary of chironomid morphology terminology (Diptera: Chironomidae). Entomol. Scand. 1980, 14, 1–51. [Google Scholar]
- Cranston, P.S. Riethia (Kieffer 1917)(Diptera: Chironomidae) revised for the Austro–Pacific region. Zootaxa 2019, 4646, 461–500. [Google Scholar] [CrossRef]
- Epler, J.H. Biosystematics of the genus Dicrotendipes Kieffer, 1913 (Diptera: Chironomidae) of the world. Mem. Am. Entomol. Soc. 1988, 36, 1–214. [Google Scholar]
- Lin, X.L.; Stur, E.; Ekrem, T. Molecular phylogeny and temporal diversification of Tanytarsus van der Wulp (Diptera: Chironomidae) support generic synonymies, a new classification and centre of origin. Syst. Entomol. 2018, 43, 659–677. [Google Scholar] [CrossRef]
- Lin, X.L.; Stur, E.; Ekrem, T. Exploring species boundaries with multiple genetic loci using empirical data from non-biting midges. Zool. Scr. 2018, 47, 325–341. [Google Scholar] [CrossRef]
- Lin, X.L.; Stur, E.; Ekrem, T. DNA barcodes and morphology reveal unrecognized species in Chironomidae (Diptera). Insect. Syst. Evol. 2017, 49, 329–398. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Ratnasingham, S.; De Waard, J.R. Barcoding animal life: Cytochrome c oxidase subunit 1 divergences among closely related species. Proc. Biol. Sci. 2003, 270 (Suppl. 1), S96–S99. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with improved accuracy and speed. In Proceedings of the 2004 IEEE Computational Systems Bioinformatics Conference, Stanford, CA, USA, 19 August 2004; IEEE: Piscataway, NJ, USA, 2004; pp. 728–729. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Rogers, J.; Wall, R. A mechanism for RNA splicing. Proc. Natl. Acad. Sci. USA 1980, 77, 1877–1879. [Google Scholar] [CrossRef]
- Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 2000, 17, 540–552. [Google Scholar] [CrossRef]
- Talavera, G.; Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 2007, 56, 564–577. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, F.; Li, W.; Jakovlić, I.; Zou, H.; Zhang, J.; Wang, G.T. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol. Ecol. Resour. 2018. [Google Scholar] [CrossRef]
- Lanfear, R.; Calcott, B.; Ho, S.Y.W.; Guindon, S. PartitionFinder: Combined Selection of Partitioning Schemes and Substitution Models for Phylogenetic Analyses. Mol. Biol. Evol. 2012, 29, 1695–1701. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ–TREE: A fast and effective stochastic algorithm for estimating maximum–likelihood phylogenies. Mol. Biol. Evol. 2014, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Hohna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Rambaut, A.; Suchard, M.A.; Xie, D.; Drummond, A.J. Tracer v1.6. Available online: http://beast.bio.ed.ac.uk/Tracer (accessed on 30 October 2019).
- Ratnasingham, S.; Hebert, P.D.N. BOLD: The Barcode of Life Data System (http://www/. barcodinglife. org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Ratnasingham, S.; Hebert, P.D.N. A DNA–Based Registry for All Animal Species: The Barcode Index Number (BIN) System. PLoS ONE 2013, 8, e66213. [Google Scholar] [CrossRef] [PubMed]
- Maguire, D.J. ArcGIS: General purpose GIS software system. Encycl. GIS 2008, 25–31. [Google Scholar]
- Sæther, O.A. Female Genitalia in Chironomidae and Other Nematocera: Morphology, Phylogenies, Keys; Stevenson, J.C., Ed.; Bulletin of the Fisheries Research Board of Canada: Ottawa, ON, Canada, 1997; p. 139. [Google Scholar]
- Huang, D.W.; Cheng, L.N. The flightless marine midge Pontomyia (Diptera: Chironomidae): Ecology, distribution, and molecular phylogeny. Zool. J. Linn. Soc. 2011, 162, 443–456. [Google Scholar] [CrossRef]
- Lin, X.L.; Qi, X.; Zhang, R.L.; Wang, X.H. A new species of Polypedilum (Uresipedilum) Oyewo & Sæther, 1998 from Zhejiang Province of Oriental China (Diptera, Chironomidae). Zookeys 2013, 320, 43–49. [Google Scholar] [CrossRef]
- Yamamoto, M.; Yamamoto, N.; Tang, H.Q. Two new chironomids bearing peculiar morphological features from Japan and China (Diptera: Chironomidae). J. Limnol. 2018, 77, 40–49. [Google Scholar] [CrossRef]
- Tang, H.Q. (Jinan University, Guangzhou, Guangdong, China); Yamamoto, M. (Kankyou–kagakuK abushiki–gaisha, Naka–sakurazuka, Toyonaka, Osaka, Japan). Personal communication, 2018.
- Zorina, O.V. Non–biting midges of the genus Paracladopelma Harnisch (Diptera: Chironomidae) from the Russian Far East. Zootaxa 2006, 1134, 29–49. [Google Scholar] [CrossRef]
- Donato, M.; Paggi, A.C.; Epler, J.H. New record, geographic variation and redescription of Apedilum elachistus Townes (Diptera: Chironomidae: Chironominae). Stud. Neotrop. Fauna. E. 2008, 43, 125–134. [Google Scholar] [CrossRef]
- Epler, J.H. A reconsideration of the genus Apedilum Townes, 1945. Spixiana 1988, 14, 105–116. [Google Scholar]
- Pinho, L.C.; Mendes, H.F.; Andersen, T. Revision of Beardius Reiss et Sublette, 1985 (Diptera: Chironomidae), with the description of twenty new species. Zootaxa 2013, 3742, 1–78. [Google Scholar] [CrossRef] [PubMed]
- Grodhaus, G. Endochironomus Kieffer, Tribelos Townes, Synendotendipes, n. gen., and Endotribelos, n. gen.(Diptera: Chironomidae) of the Nearctic region. J. Kansas Entomol. Soc. 1987, 60, 167–247. [Google Scholar]
- Silva, F.L. Procladius travassosi (Oliveira, Messias Silva–Vasconcelos, 1992) regarded as a new junior synonym of Procladius stroudi Roback 1982 (Diptera: Chironomidae: Tanypodinae). Zootaxa 2019, 4555, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Niitsuma, H. A New Species of the Genus Coffmania (Insecta: Diptera: Chironomidae) from Japan. Species Divers. 2008, 13, 123–131. [Google Scholar] [CrossRef][Green Version]
- Wiedenbrug, S.; Andersen, T. New Species of Parakiefferiella Thienemann, 1936 from South America (Chironomidae, Orthocladiinae). Stud. Neotrop. Fauna Environ. 2002, 37, 119–132. [Google Scholar] [CrossRef]
- Cranston, P.S.; Saether, O.A. Rheosmittia (Diptera: Chironomidae): A generic validation and revision of the western Palaerctic species. J. Nat. Hist. 1986, 20, 31–51. [Google Scholar] [CrossRef]
- Reiss, F. Irmakia, ein neues Subgenus von Demicryptochironomus LENZ, 1941. mit der Beschreibung von vier neuen Arten (Diptera, Chironomidae). Spixiana 1988, 11, 1–12. [Google Scholar]
- Cranston, P.S.; Martin, J.; Spies, M. Cryptic species in the nuisance midge Polypedilum nubifer (Skuse (Diptera: Chironomidae) and the status of Tripedilum Kieffer. Zootaxa 2016, 4079, 429–447. [Google Scholar] [CrossRef]
- Fusari, L.M.; Dantas, G.P.; Hamada, N.; Andrade–Souza, V.; Lima, K.M.; Silva, J.G. Not endemic after all: Imparipecten Freeman, 1961 (Diptera: Chironomidae) described from the Neotropical Region. Zootaxa 2018, 4532, 396–406. [Google Scholar] [CrossRef]
- Whiting, M.F.; Carpenter, J.C.; Wheeler, Q.D.; Wheeler, W.C. The Strepsiptera problem: Phylogeny of the holometabolous insect orders inferred from 18S and 28S ribosomal DNA sequences and morphology. Syst. Biol. 1997, 46, 1–68. [Google Scholar] [CrossRef] [PubMed]
- Morse, G.E.; Normark, B.B. A molecular phylogenetic study of armoured scale insects (Hemiptera: Diaspididae). Syst. Entomol. 2006, 31, 338–349. [Google Scholar] [CrossRef]
- Moulton, J.K.; Wiegmann, B.M. Evolution and phylogenetic utility of CAD (rudimentary) among Mesozoic–aged Eremoneuran Diptera (Insecta). Mol. Phylogenet. Evol. 2004, 31, 363–378. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Simon, C.; Frati, F.; Beckenbach, A.; Crespi, B.; Liu, H.; Flook, P. Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann. Entomol. Soc. Am. 1994, 87, 651–701. [Google Scholar] [CrossRef]



| Features of male | Y. balteatus sp. n. | Y. isigaabeus | Y. tropicus com. n. |
|---|---|---|---|
| Scutum | hump, with tubercle | hump, without tubercle | normal, without tubercle |
| Tibial comb | separated | fused | separated |
| Anal tergite band | absent | absent | reduced |
| Anal median setae | 5–6 setae, one row | two pairs, grouped | two pairs, grouped |
| TIX oval concavity | absent | present | present |
| Gonostylus | reduced | reduced | normal |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, W.; Wei, J.; Lin, X.; Tang, H. The Afro–Oriental Genus Yaeprimus Sasa et Suzuki (Diptera: Chironomidae: Chironomini): Phylogeny, New Species and Expanded Diagnoses. Diversity 2020, 12, 31. https://doi.org/10.3390/d12010031
Han W, Wei J, Lin X, Tang H. The Afro–Oriental Genus Yaeprimus Sasa et Suzuki (Diptera: Chironomidae: Chironomini): Phylogeny, New Species and Expanded Diagnoses. Diversity. 2020; 12(1):31. https://doi.org/10.3390/d12010031
Chicago/Turabian StyleHan, Wu, Jinxing Wei, Xiaolong Lin, and Hongqu Tang. 2020. "The Afro–Oriental Genus Yaeprimus Sasa et Suzuki (Diptera: Chironomidae: Chironomini): Phylogeny, New Species and Expanded Diagnoses" Diversity 12, no. 1: 31. https://doi.org/10.3390/d12010031
APA StyleHan, W., Wei, J., Lin, X., & Tang, H. (2020). The Afro–Oriental Genus Yaeprimus Sasa et Suzuki (Diptera: Chironomidae: Chironomini): Phylogeny, New Species and Expanded Diagnoses. Diversity, 12(1), 31. https://doi.org/10.3390/d12010031






