Significance of Mangrove Biodiversity Conservation in Fishery Production and Living Conditions of Coastal Communities in Sri Lanka
Abstract
1. Introduction
2. Materials and Methods
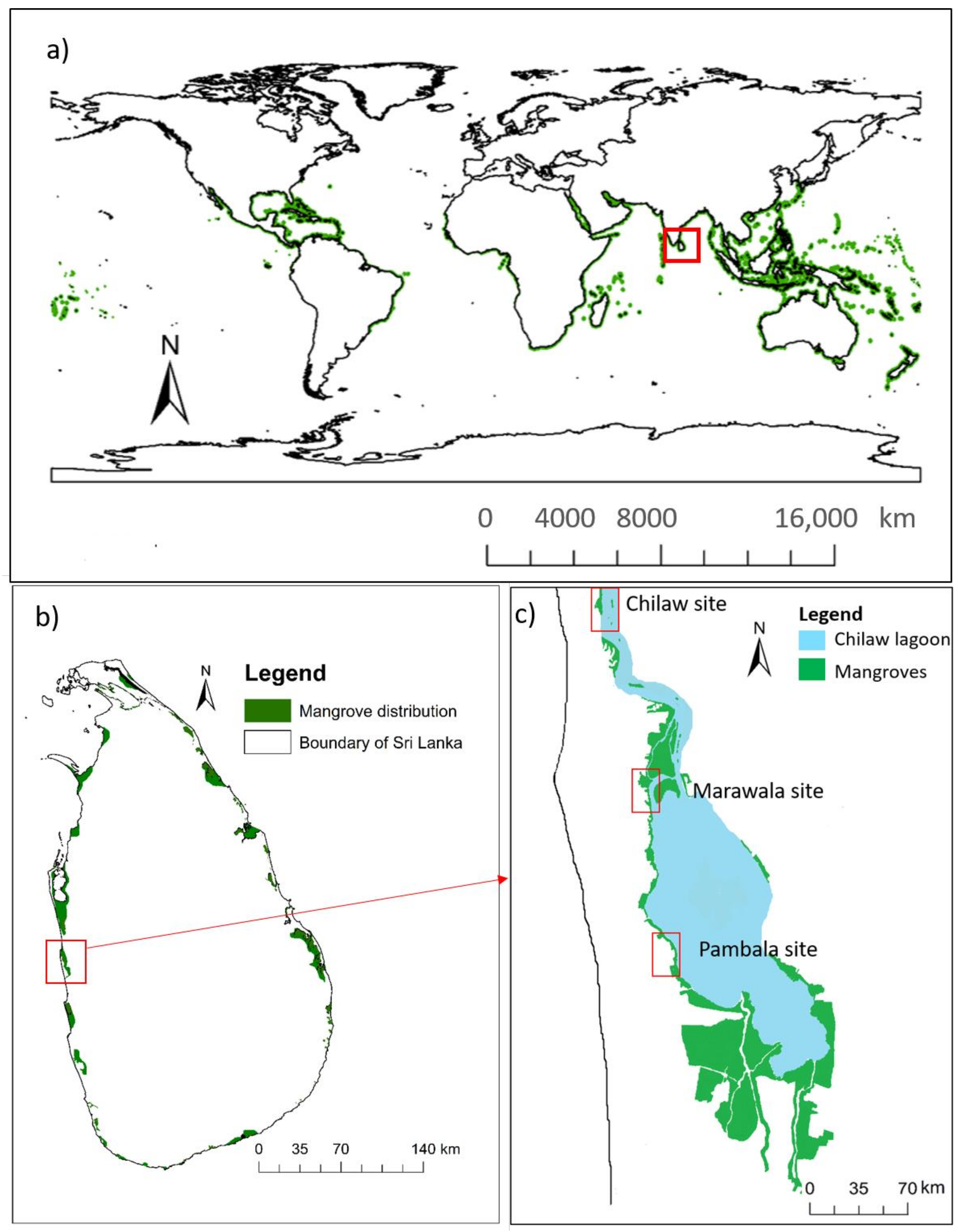
2.1. Study Site
2.2. Field Data Collection
2.2.1. Mangrove Floral and Faunal Surveys
2.2.2. Fishery Production Surveys
2.2.3. Socio-Economic Surveys
2.3. Data Analysis
2.3.1. Floral and Faunal Diversity and Fishery Catches
2.3.2. Species Compositions
2.3.3. Fishery Occupation and Mangrove Diversity
3. Results
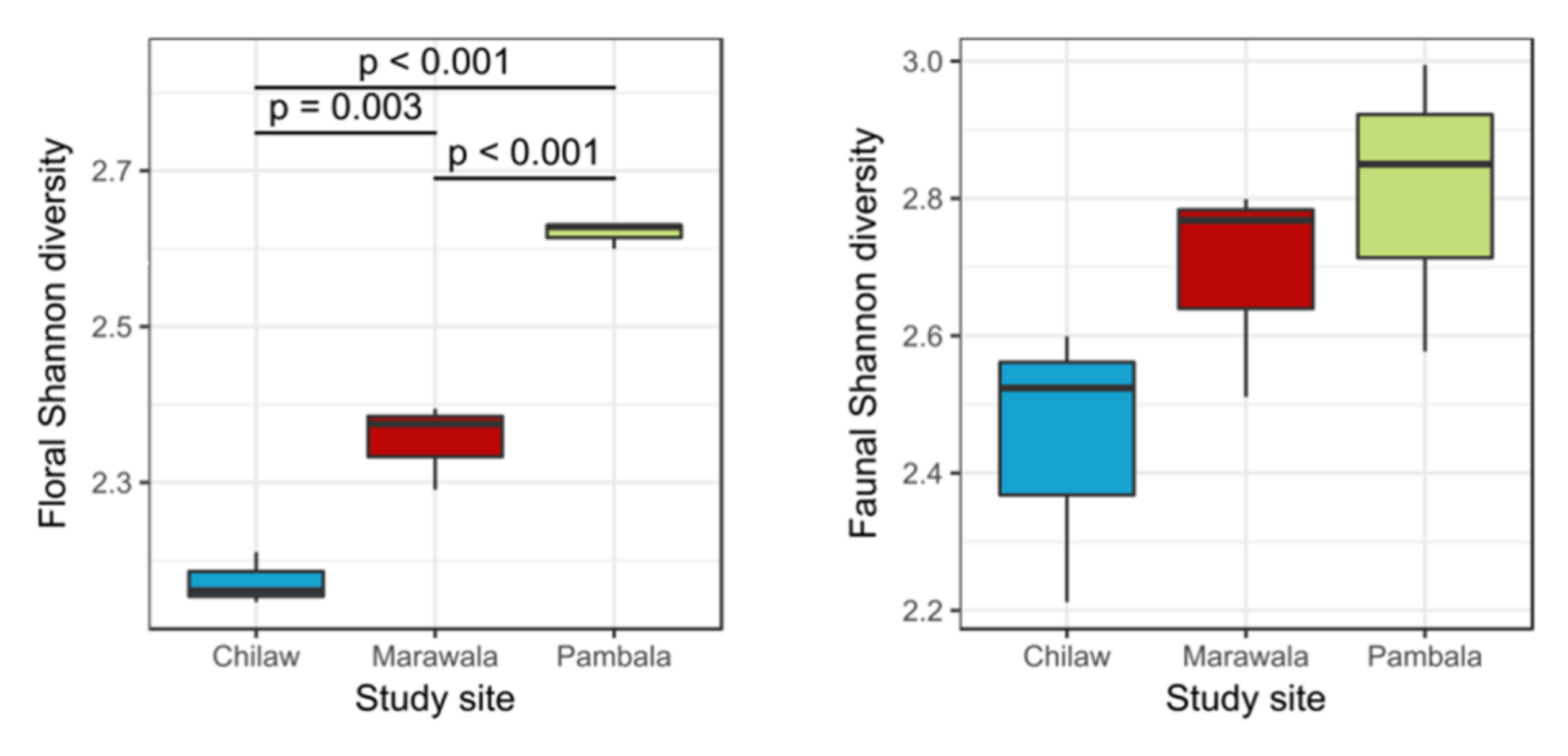
3.1. Mangrove Floral and Faunal Surveys
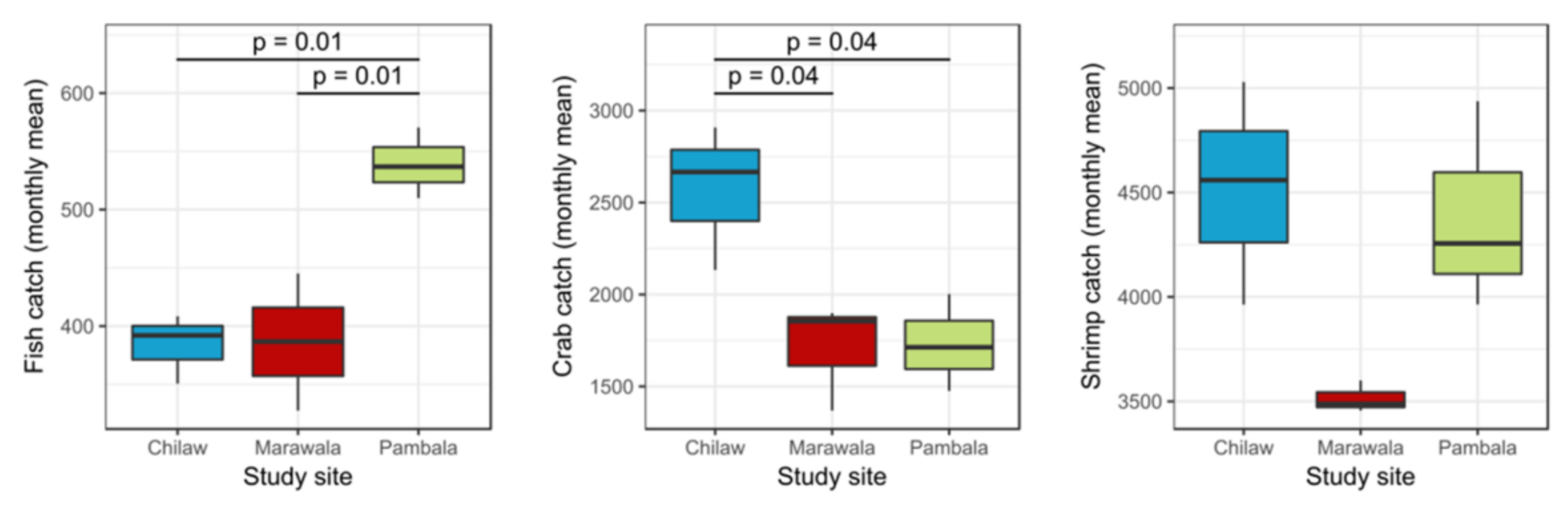
3.2. Floral and Faunal Diversity and Fishery Catches
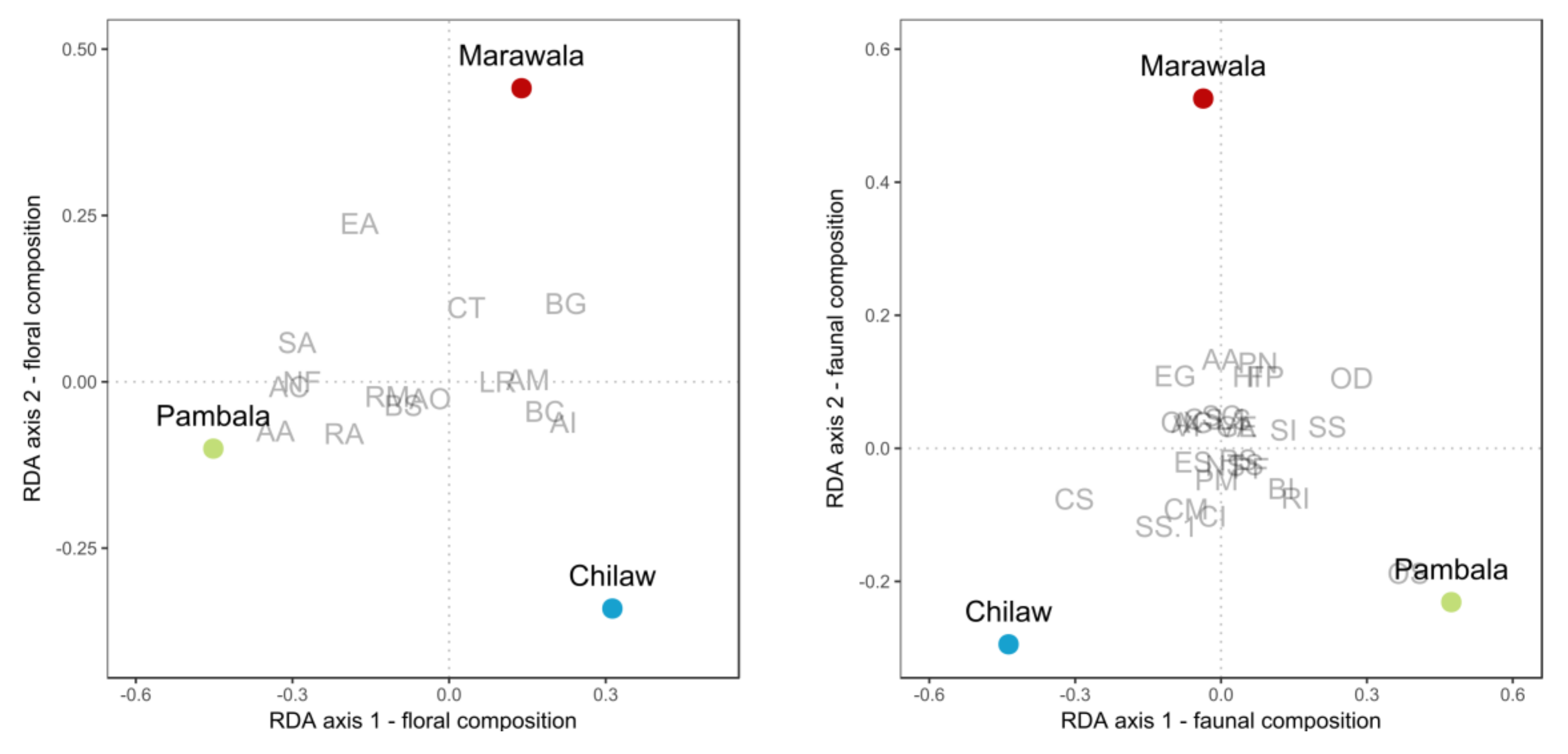
3.3. Species Compositions
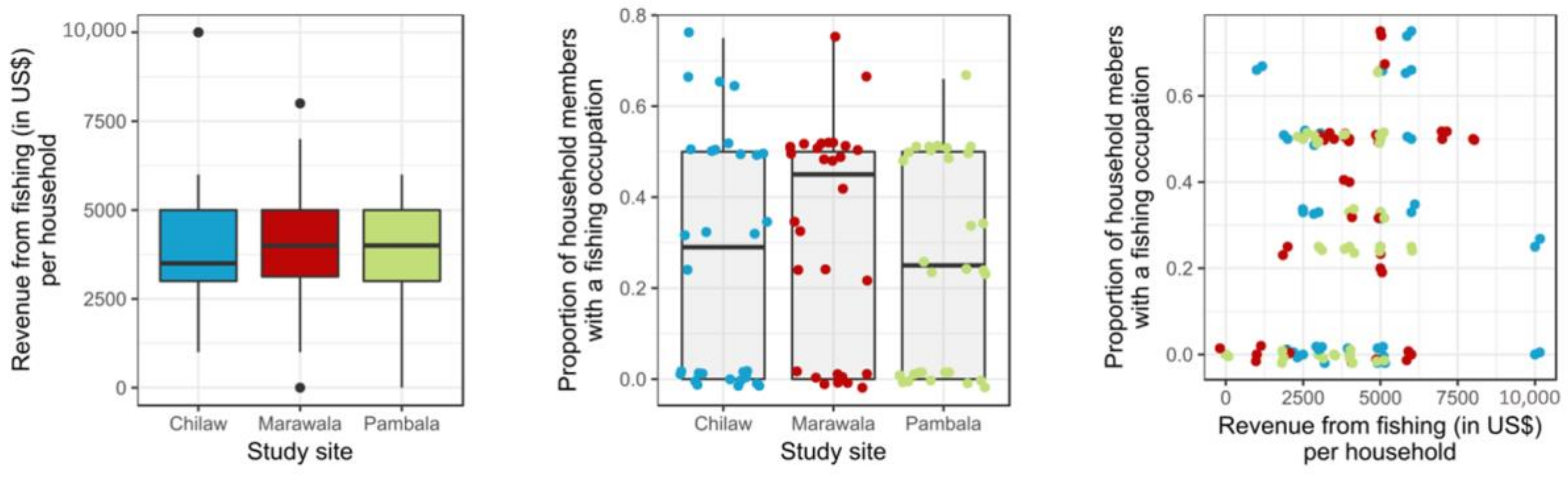
3.4. Fishery Occupation
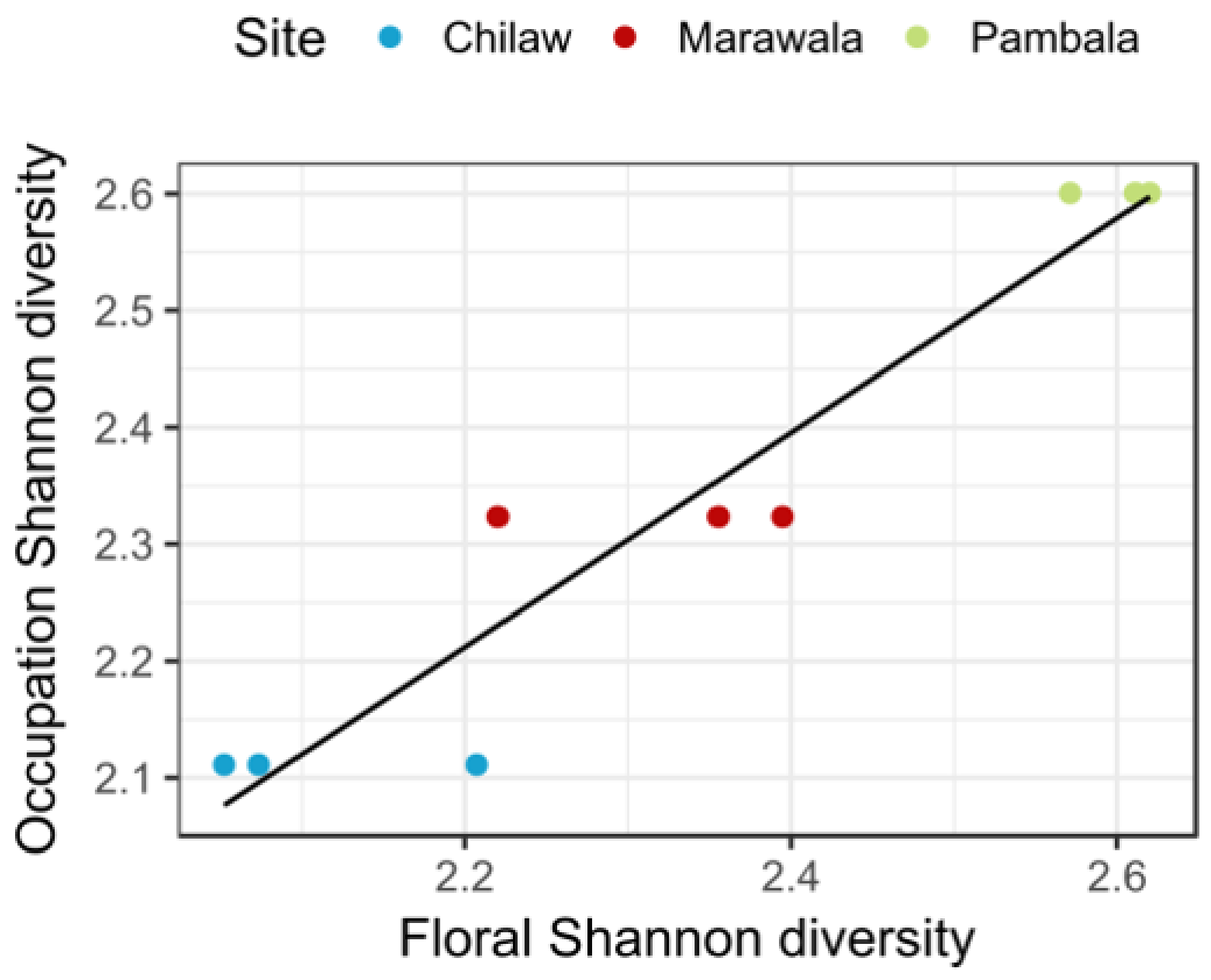
3.5. Mangrove Diversity and Occupational Diversity
4. Discussion
4.1. Relationship between Mangrove Diversity and Socioeconomic Conditions
4.2. The Current Condition of the Fishery Industry
4.3. Increasing the Sustainability and Livelihoods of Local Fishing Communities
4.4. Implications of Mangrove Biodiversity Conservation in Development Planning
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Polanía, J.; Urrego, L.E.; Agudelo, C.M. Recent advances in understanding Colombian mangroves. Acta Oecol. 2015, 63, 82–90. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Status and Trends in Mangrove Area Extent Worldwide; Forest Resources Assessment Working Paper No. 63; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2003. [Google Scholar]
- Ronnback, P. The ecological basis for economic value of seafood production supported by mangrove ecosystems. Ecol. Econ. 1999, 29, 235–252. [Google Scholar] [CrossRef]
- Duke, N.C.; Meynecke, J.-O.; Dittmann, S.; Ellison, A.M.; Anger, K.; Berger, U.; Cannicci, S.; Diele, K.; Ewel, K.C.; Field, C.D.; et al. A World without Mangroves? Science 2007, 317, 41–42. [Google Scholar] [CrossRef] [PubMed]
- Ellison, A.M.; Farnsworth, E.J. Anthropogenic Disturbance of Caribbean Mangrove Ecosystems: Past Impacts, Present Trends, and Future Predictions. Biotropica 1996, 28, 549–565. [Google Scholar] [CrossRef]
- Cunha-Lignon, M.; Kampel, M.; Menghini, R.P.; Novelli, Y.S.; Cintrón, G.; Guebas, F.D. Mangrove Forests Submitted to Depositional Processes and Salinity Variation Investigated using satellite images and vegetation structure surveys. J. Coast. Res. 2011, 1, 344–348. [Google Scholar]
- Harkes, I.H.T.; Drengstig, A.; Kumara, M.P.; Jayasinghe, J.M.P.K.; Huxham, M. Shrimp aquaculture as a vehicle for Climate Compatible Development in Sri Lanka. The case of Puttalam Lagoon. Mar. Policy 2015, 61, 273–283. [Google Scholar] [CrossRef]
- Jayawickrema, S.J.C. Status of the fishery in the Chilaw lagoon. J. Natl. Sci. Found. Sri Lanka 1992, 20, 199–207. [Google Scholar] [CrossRef][Green Version]
- Wijeratne, E.M.S.; Rydberg, L. Modelling and observations of tidal wave propagation, circulation and residence times in Puttalam Lagoon, Sri Lanka. Estuar Coast Shelf Sci. 2007, 74, 611–622. Available online: https://www.sciencedirect.com/science/article/pii/S0272771407001588 (accessed on 23 December 2017). [CrossRef]
- Jayatissa, L.P. Present Status of Mangroves in Sri Lanka, The National Read Data List of Sri Lanka: Conservation Status of Fauna and Flora; Weerakoon, D., Wijesundara, S., Eds.; Ministry of Environment: Colombo, Sri Lanka, 2012; pp. 197–199. Available online: https://www.scribd.com/doc/117262646/The-National-Red-List-2012-Sri-Lanka (accessed on 23 March 2017).
- Karunathilake, K.M.B.C. Status of Mangroves in Sri Lanka. J. Coast. Dev. 2003, 7, 5–9. [Google Scholar]
- De Silva, M.; de Silva, P.K. Status, diversity and conservation of the mangrove forests of Sri Lanka. J. South Asian Nat. Hist. 1998, 3, 79–102. [Google Scholar]
- Joseph, L. Coastai Fisheries and Brackishwater Aquaculture in Sri Lanka; Coastal Resources Management Project of the University of Rhode Island, The Government of Sri Lanka: Colombo, Sri Lanka, 1993.
- Ministry of Fisheries. Fish Production by Fishing Sub-Sectors, 1992–2013; Ministry of Fisheries and Aquatic Resources Development: Colombo, Sri Lanka, 2015.
- Saenger, P. Mangrove Ecology, Silviculture and Conservation; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Walters, B.B.; Ronnback, P.; Kovacs, J.M.; Crona, B.; Hussain, S.A.; Badola, R.; Primavera, J.H.; Barbier, E.; Dahdouh-Guebas, F. Ethnobiology, socio-economics and management of mangrove forests: A review. Aquat. Bot. 2008, 89, 220–236. [Google Scholar] [CrossRef]
- Satyanarayana, B.; Bhanderi, P.; Debry, M.; Maniatis, D.; Fore, F.; Badgie, D.; Jammeh, K.; Vanwing, T.; Farcy, C.; Koedam, N.; et al. A socio-ecological assessment aiming at improved forest resource management and sustainable ecotourism development in the mangroves of Tanbi Wetland National Park, the Gambia, West Africa. AMBIO 2012, 41, 513–526. [Google Scholar] [CrossRef] [PubMed]
- Boon, J.M. A socio-economic analysis of mangrove degradation in Samoa. Geogr. Rev. Jpn. 2001, 74, 159–186. [Google Scholar] [CrossRef][Green Version]
- Feka, N.Z.; Manzano, M.G.; Dahdouh-Guebas, F. The effects of different gender harvesting practices on mangrove ecology and conservation in Cameroon. Int. J. Biodivers. Sci. Ecosyst. Serv. Manag. 2011, 7, 108–121. [Google Scholar] [CrossRef][Green Version]
- Islam, M.M.; Shamsuzzaman, M.M.; Sunny, A.R.; Islam, N. Understanding fishery conflicts in hilsa sanctuaries of Bangladesh. In Inter-Sectoral Governance of Inland Fisheries; Song, A.M., Bower, S.D., Onyango, P., Cooke, S.J., Chuenpagdee, R., Eds.; TBTI Publication Series; TBTI: St John’s, NL, Canada, 2016; Available online: https://www.researchgate.net/publication/304589663 (accessed on 20 October 2017).
- Satyanarayana, B.; Mulder, S.; Jayatissa, L.P.; Dahdouh-Guebas, F. Are the mangroves in the Galle-Unawatuna area (Sri Lanka) at risk? A social-ecological approach involving local stakeholders for a better conservation policy. Ocean Coast. Manag. 2013, 71, 225–237. [Google Scholar] [CrossRef]
- Jayasuriya, P.M.A. The distribution and the species composition of the existing mangrove vegetation around Chilaw lagoon. Vidyodaya J. Sci. 1991, 3, 103–113. [Google Scholar]
- Satyanarayana, B.; Koedam, N.; De Smet, K.; Di Nitto, D.; Bauwens, M.; Jayatissa, L.P.; Cannicci, S.; Dahdouh-Guebas, F. Long-term mangrove forest development in Sri Lanka: Early predictions evaluated against outcomes using VHR remote sensing and VHR ground-truth data. Mar. Ecol. Prog. Ser. 2011, 443, 51–63. [Google Scholar] [CrossRef]
- Bournazel, J.; Kumara, M.P.; Jayatissa, L.P.; Viergever, K.; Morel, V.; Huxham, M. The impacts of shrimp farming on land-use and carbon storage around Puttalam lagoon, Sri Lanka. Ocean Coast. Manag. 2015, 113, 18–28. [Google Scholar] [CrossRef]
- Department of Census. Population by Divisional Secretariat Division and Sex, Census 2012. Available online: http://www.statistics.gov.lk/page.asp?page=Population and Housing (accessed on 8 June 2017).
- Ranasinghe, T. Sustainable Financing and Benefit—Sharing Strategy for Conservation and Management of Puttalam Lagoon; For the Project: Ecologically and Socio Economically Sound Coastal Ecosystem Rehabilitation and Conservation in Tsunami Affected Countries of the Indian Ocean; International Union for Conservation of Nature (IUCN): Gland, Switzerland, 2010. [Google Scholar]
- Department of Meteorology. Annual and Monthly Monthly Rainfall and Temperature at Observation Stations, 2010—2015. Colombo, Sri Lanka, 2016. Available online: http://www.statistics.gov.lk/Abstract2016/CHAP1/1.6.pdf (accessed on 10 July 2017).
- Dahdouh-Guebas, F.; Zetterström, T.; Rönnbäck, P.; Troell, M.; Wickramasinghe, A.; Koedam, N. Recent changes in land-use in the Pambala-Chilaw Lagoon complex (Sri Lanka) investigated using remote sensing and GIS: Conservation of mangroves vs. development of shrimp farming. Environ. Dev. Sustain. 2001, 4, 185–200. [Google Scholar] [CrossRef]
- International Union for Conservation of Nature (IUCN). Conservation Management Plan for Ten Selected Mangrove Sites in Northwestern Sri Lanka; International Union for Conservation of Nature (IUCN): Gland, Switzerland, 1996. [Google Scholar]
- International Union for Conservation of Nature (IUCN). METADATA: Digital Distribution Maps on the IUCN Red List of Threatened Species; International Union for Conservation of Nature (IUCN): Gland, Switzerland, 2014; pp. 1–5. [Google Scholar]
- International Union for Conservation of Nature (IUCN). IUCN Sri Lanka and the Ministry of Environment and Natural Resources, the 2007 Red List of Threatened Fauna and Flora of Sri Lanka. Colombo, Sri Lanka, 2007. Available online: https://cmsdata.iucn.org/downloads/rl_548_7_003.pdf (accessed on 23 December 2017).
- Ministry of Fisheries. Basic Statistics of Marine Fisheries by Fishing District; Ministry of Fisheries and Aquatic Resources Development: Colombo, Sri Lanka, 2013.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: https://www.r-project.org/ (accessed on 28 September 2017).
- Tukey, J.W.; Cleveland, W.S. What Is the Tukey Test/Honest Significant Difference? Tukey-Kramer Method, 1st ed.; Brillinger, D.R., Ed.; Wadsworth Pub Co.: Belmont, CA, USA, 1984; Available online: https://www.amazon.com/Collected-Works-John-Tukey-Probability/dp/0534033032 (accessed on 8 June 2017).
- Kendall, M.G. Kendall Rank Correlation Coefficient. Rank Correlation. 1948. Available online: https://link.springer.com/content/pdf/10.1007/978-0-387-32833-1_211.pdf (accessed on 14 September 2017).
- Folke, C. Resilience: The emergence of a perspective for social-ecological systems analyses. Glob. Environ. Chang. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Clark, J.R. Coastal Zone Management Handbook; Lewis Publishers: New York, NY, USA, 1996; Available online: https://www.crcpress.com/Coastal-Zone-Management-Handbook/Clark/p/book/9781566700924 (accessed on 25 Augest 2017).
- Jalaldeen, M.H.; Periyathamby, V. Alternative Resilient Livelihood Options for Fisher Folk Tsunami Victims: Special Reference to Batticaloa District of Sri Lanka; Department of Zoology, Faculty of Science, Eastern University: Sri Lanka, 2010; Available online: https://www.yumpu.com/en/document/view/37052698/alternative-resilient-livelihood-option-for-fisher-folks-tsunami-victims (accessed on 30 October 2017).
- Huxham, M. How Shrimp Farming Wreaked Havoc on Sri Lanka’s Coasts. The Conversation. 2015. Available online: http://theconversation.com/how-shrimp-farming-wreaked-havoc-on-sri-lankas-coasts-44933 (accessed on 20 November 2017).
| Family Name | Species Name | Conservation Status * | Percentage Trees per Transect % | ||
|---|---|---|---|---|---|
| Pambala | Marawala | Chilaw | |||
| Rhizophoraceae | Rhizophora mucronata Lam. | LC | 11.2 | 6.4 | 6.1 |
| Rhizophora apiculata Blume. | NT | 7.7 | 2.1 | 2.6 | |
| Bruguiera cylindrica (L.) Blume | EN | 3.9 | 7.9 | 12.2 | |
| Bruguiera gymnorrhiza (L.) Lam | VU | 3.4 | 14.3 | 10.4 | |
| Bruguiera sexangula (Lour.) Poir | VU | 9.4 | 5.7 | 6.1 | |
| Ceriops tagal (Perr.) C.B.Rob. | NT | 5.6 | 10.0 | 6.1 | |
| Acanthaceae | Avicennia officinalis L. | NT | 9.0 | 7.1 | 7.8 |
| Avicennia marina (Forssk.) Vierh. | LC | 5.6 | 10.7 | 12.2 | |
| Combretaceae | Lumnitzera racemosa Willd. | NT | 3.9 | 6.4 | 7.0 |
| Primulaceae | Aegiceras corniculatum (L.) Blanco | LC | 5.6 | 0.7 | 0.0 |
| Arecaceae | Nypa fruticans Wurmb | VU | 4.7 | 0.7 | 0.0 |
| Pteridaceae | Acrostichum aureum L. | NE | 5.6 | 0.0 | 0.0 |
| Lythraceae | Sonneratia alba Sm. | EN | 5.6 | 1.4 | 0.0 |
| Euphorbiaceae | Excoecaria agallocha L. | LC | 5.6 | 7.1 | 0.9 |
| Acanthaceae | Acanthus illicifolius L. | NE | 11.6 | 19.3 | 28.7 |
| Meliaceae | Xylocarpus granatum J.Koenig | LC | 1.7 | 0.0 | 0.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarathchandra, C.; Kambach, S.; Ariyarathna, S.C.; Xu, J.; Harrison, R.D.; Wickramasinghe, S. Significance of Mangrove Biodiversity Conservation in Fishery Production and Living Conditions of Coastal Communities in Sri Lanka. Diversity 2018, 10, 20. https://doi.org/10.3390/d10020020
Sarathchandra C, Kambach S, Ariyarathna SC, Xu J, Harrison RD, Wickramasinghe S. Significance of Mangrove Biodiversity Conservation in Fishery Production and Living Conditions of Coastal Communities in Sri Lanka. Diversity. 2018; 10(2):20. https://doi.org/10.3390/d10020020
Chicago/Turabian StyleSarathchandra, Chaya, Stephan Kambach, Sameera Chathuranga Ariyarathna, Jianchu Xu, Rhett D. Harrison, and Sriyani Wickramasinghe. 2018. "Significance of Mangrove Biodiversity Conservation in Fishery Production and Living Conditions of Coastal Communities in Sri Lanka" Diversity 10, no. 2: 20. https://doi.org/10.3390/d10020020
APA StyleSarathchandra, C., Kambach, S., Ariyarathna, S. C., Xu, J., Harrison, R. D., & Wickramasinghe, S. (2018). Significance of Mangrove Biodiversity Conservation in Fishery Production and Living Conditions of Coastal Communities in Sri Lanka. Diversity, 10(2), 20. https://doi.org/10.3390/d10020020






