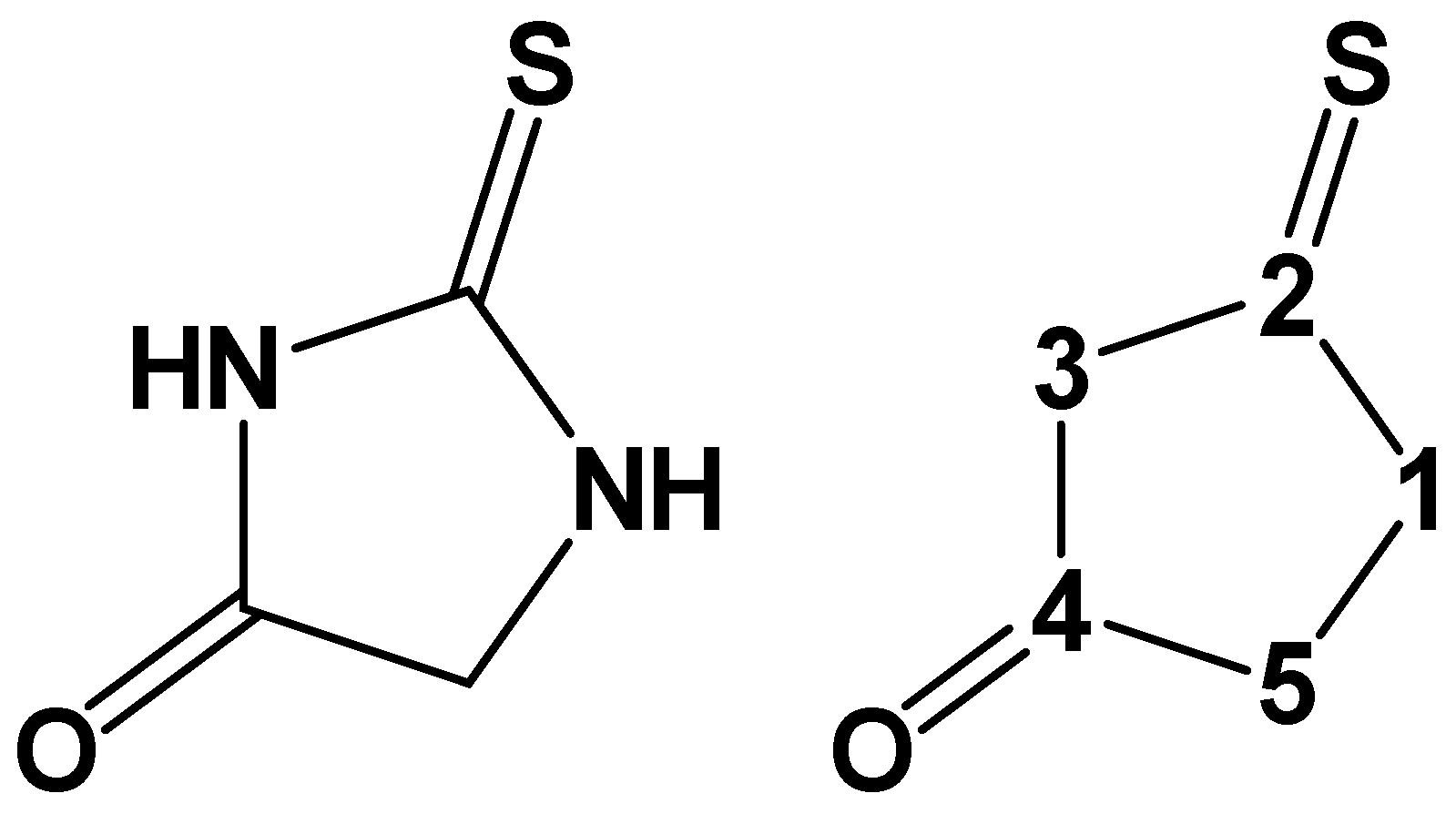
(Z)-2′-((Adamantan-1-yl)thio)-1,1′-dimethyl-2′,3′-dihydro-[2,4′-biimidazolylidene]-4,5,5′(1H,1′H,3H)-trione
Abstract
1. Introduction
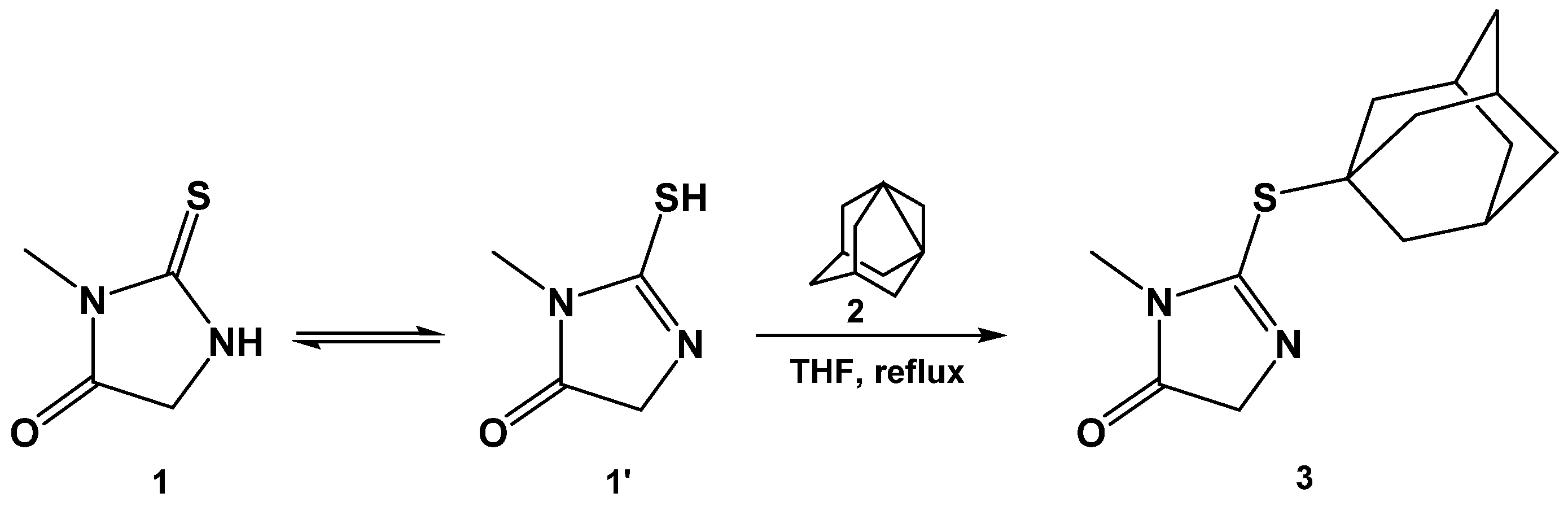
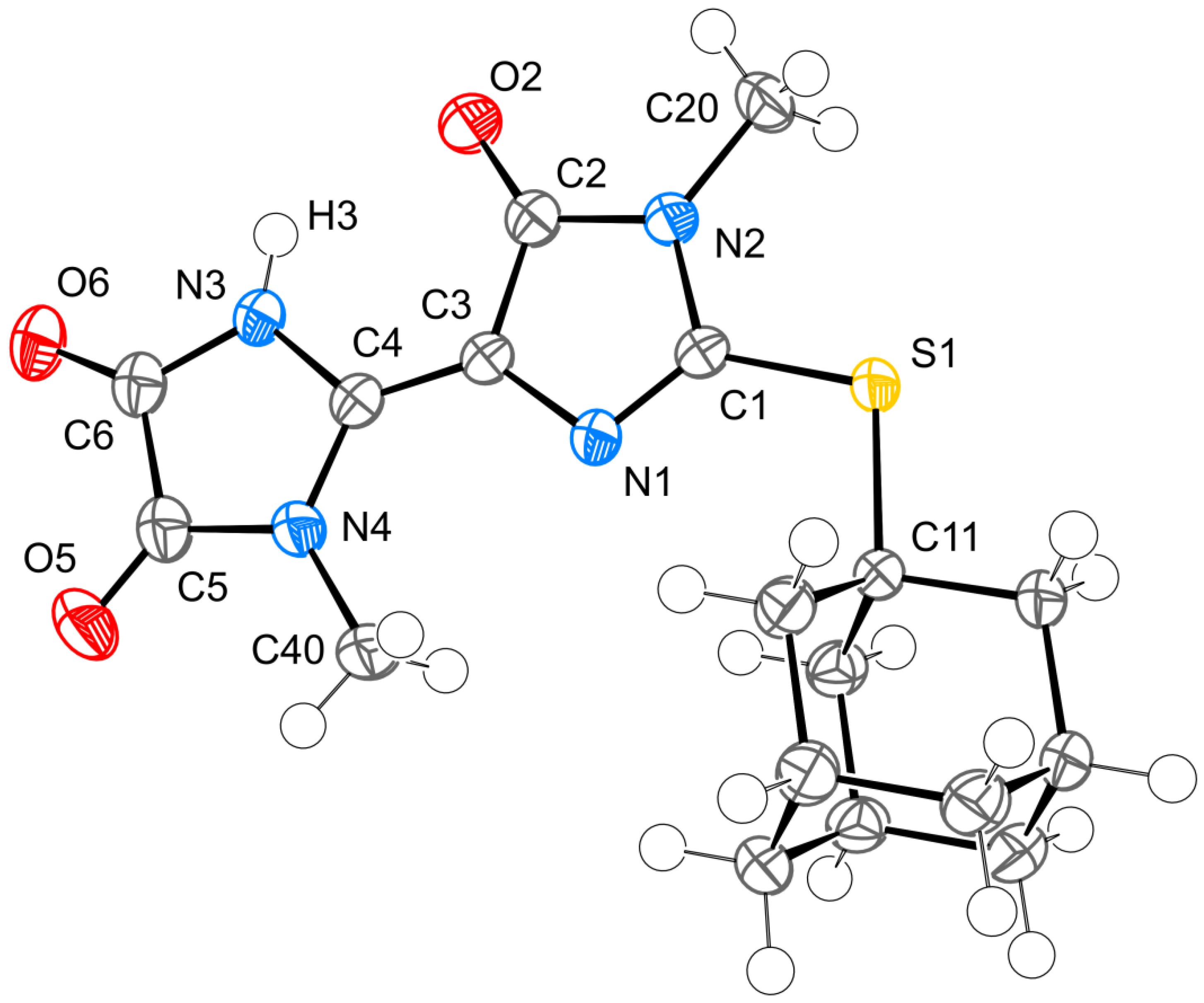
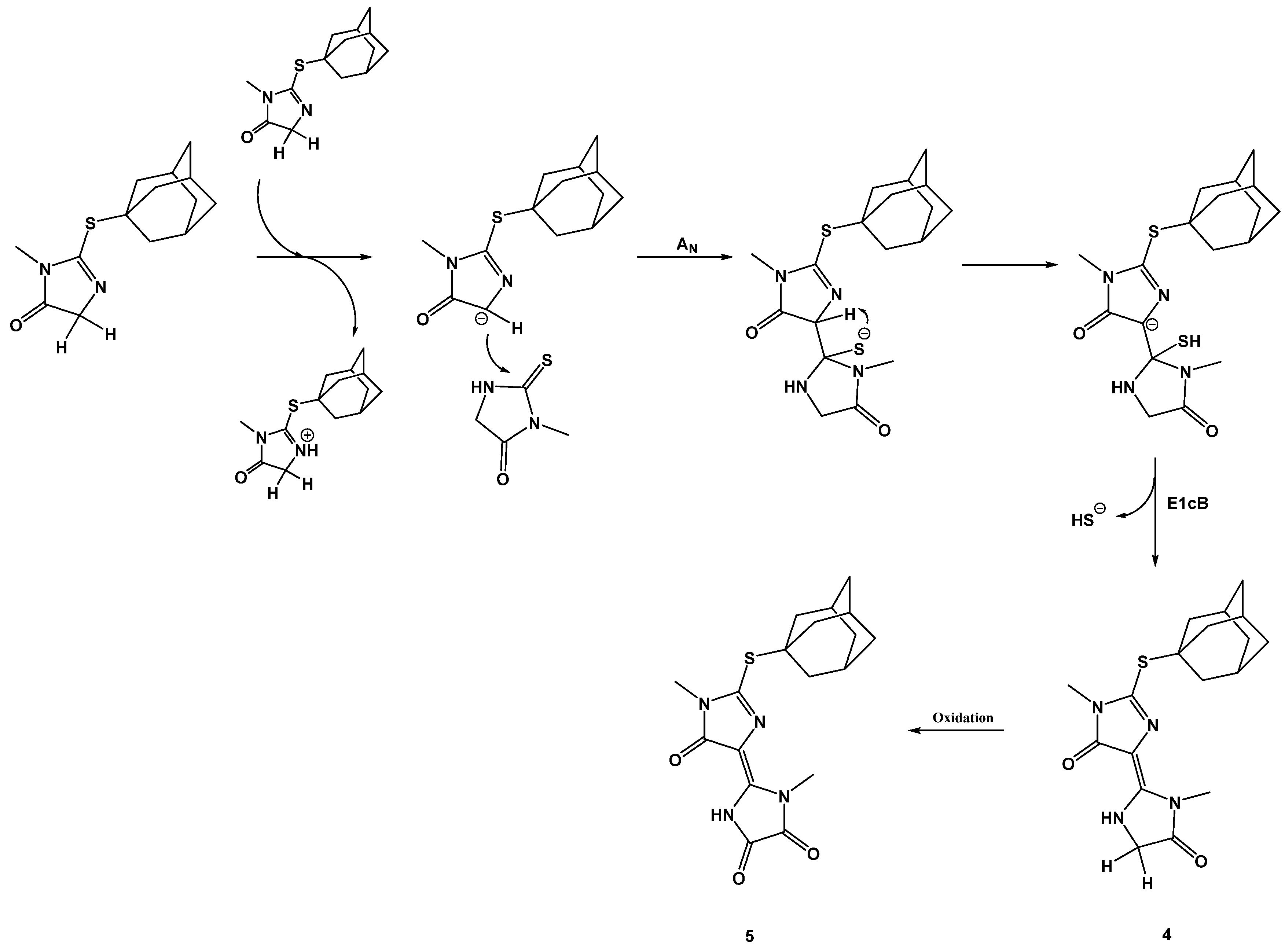
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wu, F.; Jiang, H.; Zheng, B.; Kogiso, M.; Yao, Y.; Zhou, C.; Li, X.; Song, Y. Inhibition of Cancer-Associated Mutant Isocitrate Dehydrogenases by 2-Thiohydantoin Compounds. J. Med. Chem. 2015, 58, 6899–6908. [Google Scholar] [CrossRef] [PubMed]
- Gosling, S.; Rollin, P.; Tatibouët, A. Thiohydantoins: Selective N- and S-Functionalization for Liebeskind–Srogl Reaction Study. Synthesis 2011, 2011, 3649–3660. [Google Scholar]
- Jha, S.; Silversides, J.D.; Boyle, R.W.; Archibald, S.J. Hydrogen bonded dimers vs. one-dimensional chains in 2-thiooxoimidazolidin4-one (thiohydantoin) drug derivatives. Cryst. Eng. Comm. 2010, 12, 1730–1739. [Google Scholar] [CrossRef]
- Abou El-Regal, M.K.; Abdalha, A.A.; El-Kassaby, M.A.; Ali, A.T. Synthesis of New Thiohydantoin Derivatives Under Phase Transfer Catalysis. Phosphorus Sulfur Silicon 2007, 182, 845–851. [Google Scholar] [CrossRef]
- Renault, S.; Bertrand, S.; Carreaux, F.; Bazureau, J.P. Parallel Solution-Phase Synthesis of 2-Alkylthio-5-arylidene-3,5-dihydro-4H-imidazol-4-one by One-Pot Three-Component Domino Reaction. J. Comb. Chem. 2007, 9, 935–942. [Google Scholar] [CrossRef]
- Burmistrov, V.V.; Pitushkin, D.A.; Vasipov, V.V.; D’yachenko, V.S.; Butov, G.M. Synthesis of 3-adamantylated hydantoins and their 2-thio(seleno) analogs. Chem. Heterocycl. Comp. 2019, 55, 619–622. [Google Scholar] [CrossRef]
- Burmistrov, V.; Saxena, R.; Pitushkin, D.; Butov, G.M.; Chung, F.-L.; Aggarwal, M. Adamantyl Isothiocyanates as Mutant p53 Rescuing Agents and Their Structure−Activity Relationships. J. Med. Chem. 2021, 64, 6621–6633. [Google Scholar] [CrossRef]
- Beloglazkina, E.K.; Majouga, A.G.; Yudin, I.V.; Frolova, N.A.; Zyk, N.V.; Dolzhikova, V.D.; Moiseeva, A.A.; Rakhimov, R.D.; Butin, K.P. 5-(Pyridylmethylidene)-substituted 2-thiohydantoins and their complexes with CuII, NiII, and CoII: Synthesis, electrochemical study, and adsorption on the cystamine-modified gold surface. Russ. Chem. Bull. 2006, 55, 1015–1027. [Google Scholar] [CrossRef]
- Sirivolu, V.R.; Vernekar, S.K.V.; Marchand, C.; Naumova, A.; Chergui, A.; Renaud, A.; Stephen, A.G.; Chen, F.; Sham, Y.Y.; Pommier, Y.; et al. 5-Arylidenethioxothiazolidinones as Inhibitors of Tyrosyl–DNA Phosphodiesterase I. J. Med. Chem. 2012, 55, 8671–8684. [Google Scholar] [CrossRef]
- Dang, P.; Madan, A.K. Structure-Activity Study on Anticonvulsant (Thio) Hydantoins Using Molecular Connectivity Indices. J. Chem. Inf. Comput. Sci. 1994, 34, 1162–1166. [Google Scholar] [CrossRef]
- Lindel, T.; Hoffmann, H. Synthesis of dispacamide from the marine sponge agelas dispar. Tetrahedron Lett. 1997, 38, 8935–8938. [Google Scholar] [CrossRef]
- Yadav, L.D.S.; Shukla, S. Synthesis of new peptidyl imidazodithi(and -thiadi)azoles as potential fungicides. J. Agric. Food Chem. 1995, 43, 2526–2529. [Google Scholar] [CrossRef]
- Marton, J.; Enisz, J.; Hosztafi, S.; Timar, T. Preparation and fungicidal activity of 5-substituted hydantoins and their 2-thio analogs. J. Agric. Food Chem. 1993, 41, 148–152. [Google Scholar] [CrossRef]
- Yong, X.; Su, M.; Wan, W.; You, W.; Lu, X.; Qu, J.; Liu, R. 2-Thiohydantoin containing OH and NH recognition subunits: A fluoride ion selective colorimetric sensor. New J. Chem. 2013, 37, 1591–1594. [Google Scholar] [CrossRef]
- El-Barbary, A.A.; Khodair, A.I.; Pedersen, E.B.; Nielsen, C. S-Glucosylated hydantoins as new antiviral agents. J. Med. Chem. 1994, 37, 73–77. [Google Scholar] [CrossRef]
- Takahashi, A.; Matsuoka, H.; Ozawa, Y.; Uda, Y. Antimutagenic Properties of 3,5-Disubstituted 2-Thiohydantoins. J. Agric. Food Chem. 1998, 46, 5037–5042. [Google Scholar] [CrossRef]
- Froelich, E.; Fruehan, A.; Jackman, M.; Kirchner, F.K.; Alexander, E.J.; Archer, S. 5-Heptyl-2-Thiohydantoin, a New Antitubercular Agent. J. Am. Chem. Soc. 1954, 76, 3099–3100. [Google Scholar] [CrossRef]
- Blanc, M.; Cussac, M.; Boucherle, A.; Leclerc, G. Synthesis and immunomodulating activity of 1-amino-2-thiohydantoin derivatives. Eur. J. Med. Chem. 1992, 27, 839–843. [Google Scholar] [CrossRef]
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; Wongvipat, J.; Smith-Jones, P.M.; Yoo, D.; Kwon, A.; et al. Development of a Second-Generation Antiandrogen for Treatment of Advanced Prostate Cancer. Science 2009, 324, 787–790. [Google Scholar] [CrossRef]
- Taplin, M.-E.; Balk, S.P. Androgen receptor: A key molecule in the progression of prostate cancer to hormone independence. J. Cell. Biochem. 2004, 91, 483–490. [Google Scholar] [CrossRef]
- Harrison, C. New approaches to anti-androgen activities. Nat. Rev. Drug Discov. 2009, 8, 452–453. [Google Scholar] [CrossRef]
- Shen, H.C.; Balk, S.P. Development of androgen receptor antagonists with promising activity in castration-resistant prostate cancer. Cancer Cell 2009, 15, 461–463. [Google Scholar] [CrossRef]
- Wanka, L.; Iqbal, K.; Schreiner, P.R. The Lipophilic Bullet Hits the Targets: Medicinal Chemistry of Adamantane Derivatives. Chem. Rev. 2013, 113, 3516–3604. [Google Scholar] [CrossRef] [PubMed]
- Mokhov, V.M.; Butov, G.M. Adamantylation of Carbonitriles with 1,3-Dehydroadamantane and Its Homologs. Russ. J. Org. Chem. 2014, 50, 1279–1282. [Google Scholar] [CrossRef]
- Butov, G.M.; Mokhov, V.M.; Parshin, G.Y.; Panyushkina, O.A. Adamantylation of Azoles by 1,3-Dehydroadamantane: I. N-Adamantylation of Imidazoles by 1,3-Dehydroadamantane. Russ. J. Org. Chem. 2009, 45, 1732–1733. [Google Scholar] [CrossRef]
- Butov, G.M.; Mokhov, V.M. Reaction of 1,3-Dehydroadamantane with Dicarboxylic Acid Imides. Russ. J. Org. Chem. 2013, 49, 1403–1404. [Google Scholar] [CrossRef]
- No, B.I.; Mokhov, V.M.; Vishnevetskii, E.N. Preparation of Adamantyl-substituted Amino Acids Lactams. Russ. J. Org. Chem. 2003, 39, 1193–1194. [Google Scholar] [CrossRef]
- Butov, G.M.; Mokhov, V.M.; Burmistrov, V.V.; Saad, K.R.; Pitushkin, D.A. Reactions of 1,3-dehydroadamantane with inorganic oxygen-free acids. Russ. J. Org. Chem. 2014, 50, 1276–1278. [Google Scholar] [CrossRef]
- Dürüst, Y.; Nohut, F. A New And Convenient Synthesis Of Some Substituted Thiohydantoins. Synth. Commun. 1999, 29, 1997–2005. [Google Scholar] [CrossRef]
- Shteingolts, S.A.; Stash, A.I.; Tsirelson, V.G.; Fayzullin, R.R. Orbital-Free Quantum Crystallographic View on Noncovalent Bonding: Insights into Hydrogen Bonds, π∙∙∙π and Reverse Electron Lone Pair∙∙∙π Interactions. Chem. Eur. J. 2021, 27, 7789–7809. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burmistrov, V.; Mokhov, V.; Fayzullin, R.R.; Butov, G.M. (Z)-2′-((Adamantan-1-yl)thio)-1,1′-dimethyl-2′,3′-dihydro-[2,4′-biimidazolylidene]-4,5,5′(1H,1′H,3H)-trione. Molbank 2023, 2023, M1585. https://doi.org/10.3390/M1585
Burmistrov V, Mokhov V, Fayzullin RR, Butov GM. (Z)-2′-((Adamantan-1-yl)thio)-1,1′-dimethyl-2′,3′-dihydro-[2,4′-biimidazolylidene]-4,5,5′(1H,1′H,3H)-trione. Molbank. 2023; 2023(1):M1585. https://doi.org/10.3390/M1585
Chicago/Turabian StyleBurmistrov, Vladimir, Vladimir Mokhov, Robert R. Fayzullin, and Gennady M. Butov. 2023. "(Z)-2′-((Adamantan-1-yl)thio)-1,1′-dimethyl-2′,3′-dihydro-[2,4′-biimidazolylidene]-4,5,5′(1H,1′H,3H)-trione" Molbank 2023, no. 1: M1585. https://doi.org/10.3390/M1585
APA StyleBurmistrov, V., Mokhov, V., Fayzullin, R. R., & Butov, G. M. (2023). (Z)-2′-((Adamantan-1-yl)thio)-1,1′-dimethyl-2′,3′-dihydro-[2,4′-biimidazolylidene]-4,5,5′(1H,1′H,3H)-trione. Molbank, 2023(1), M1585. https://doi.org/10.3390/M1585








