2-Cyclopropyl-6-phenyl-2,3-dihydrothieno[3,2-d][1,3,2]diazaborinin-4(1H)-one
Abstract
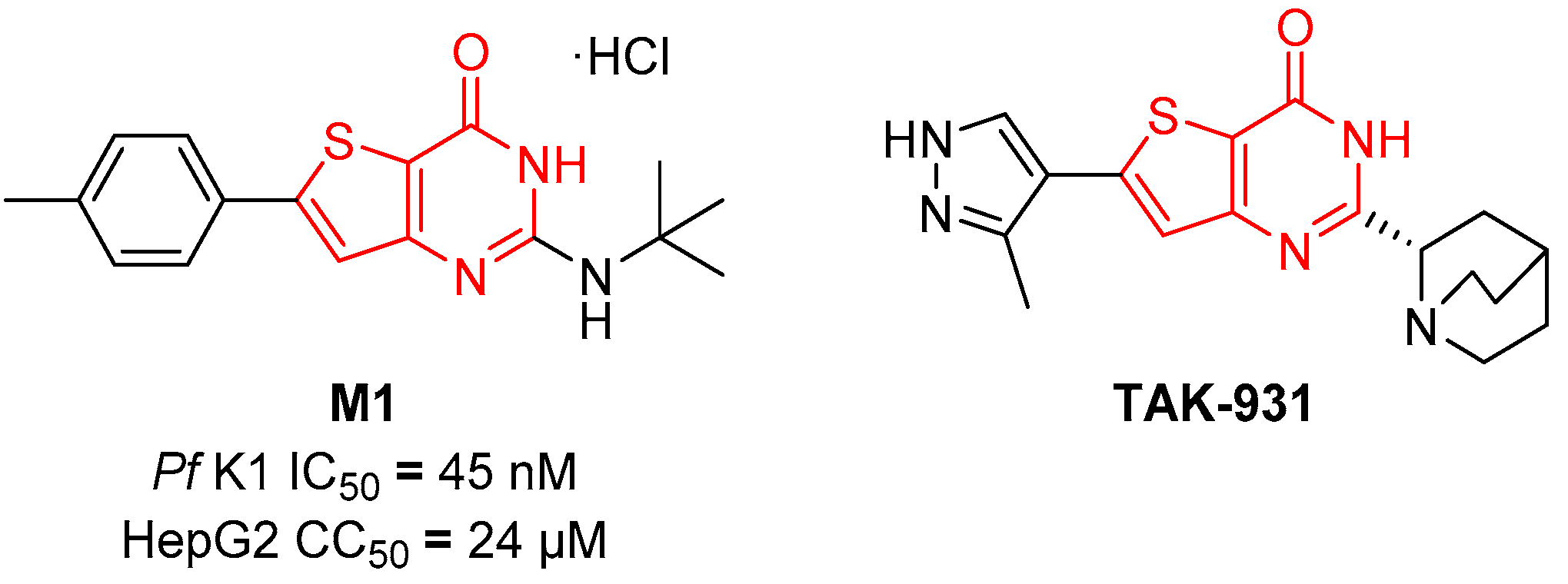
:1. Introduction
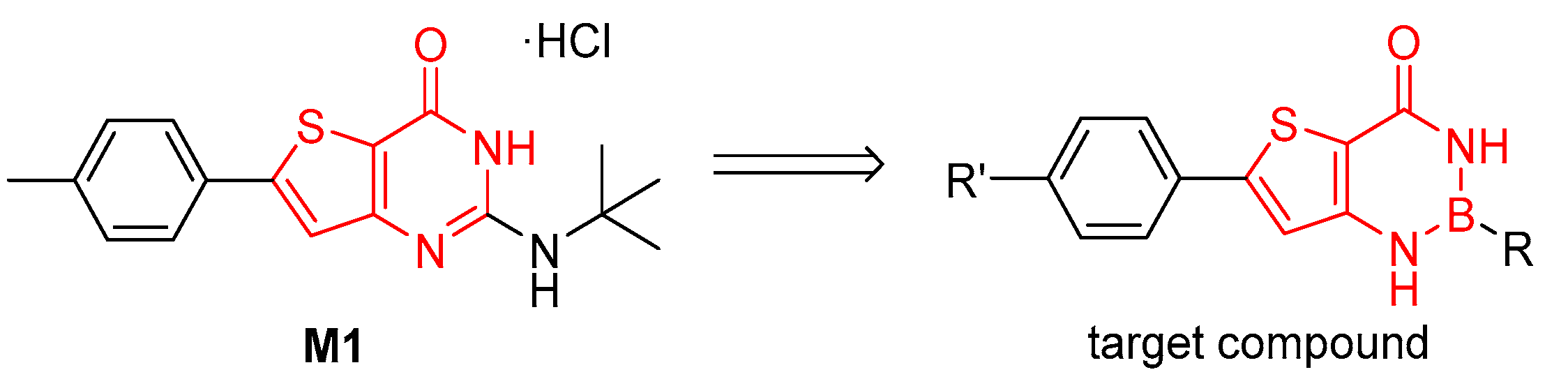
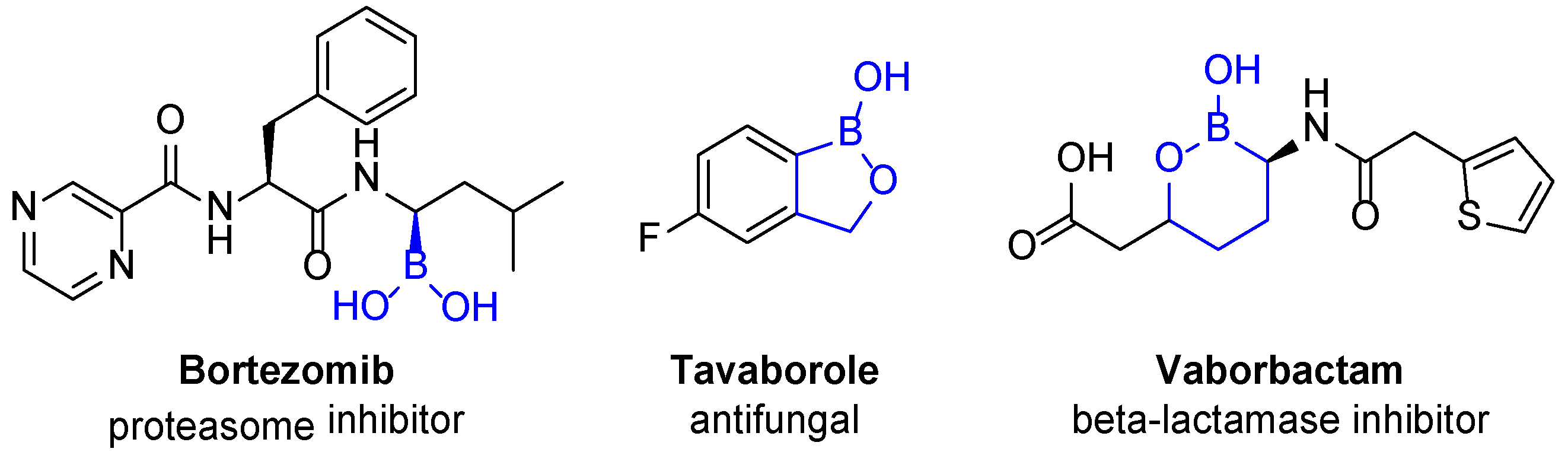
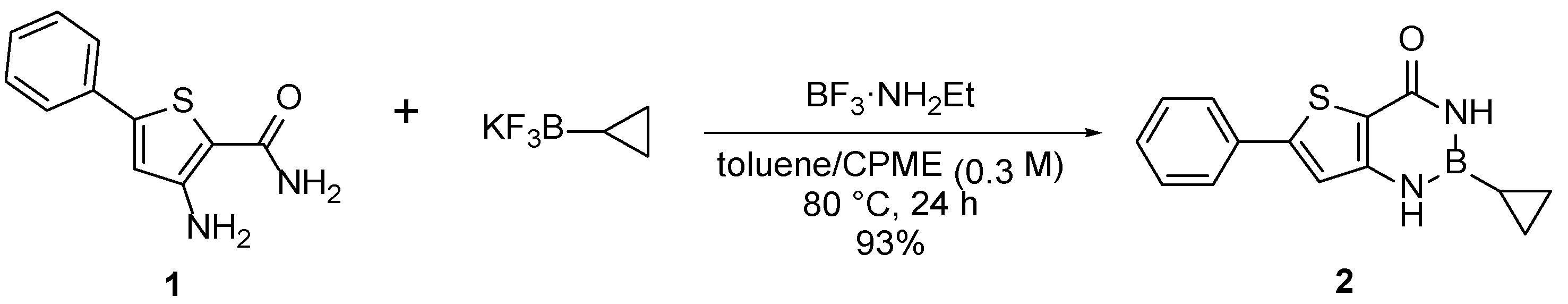
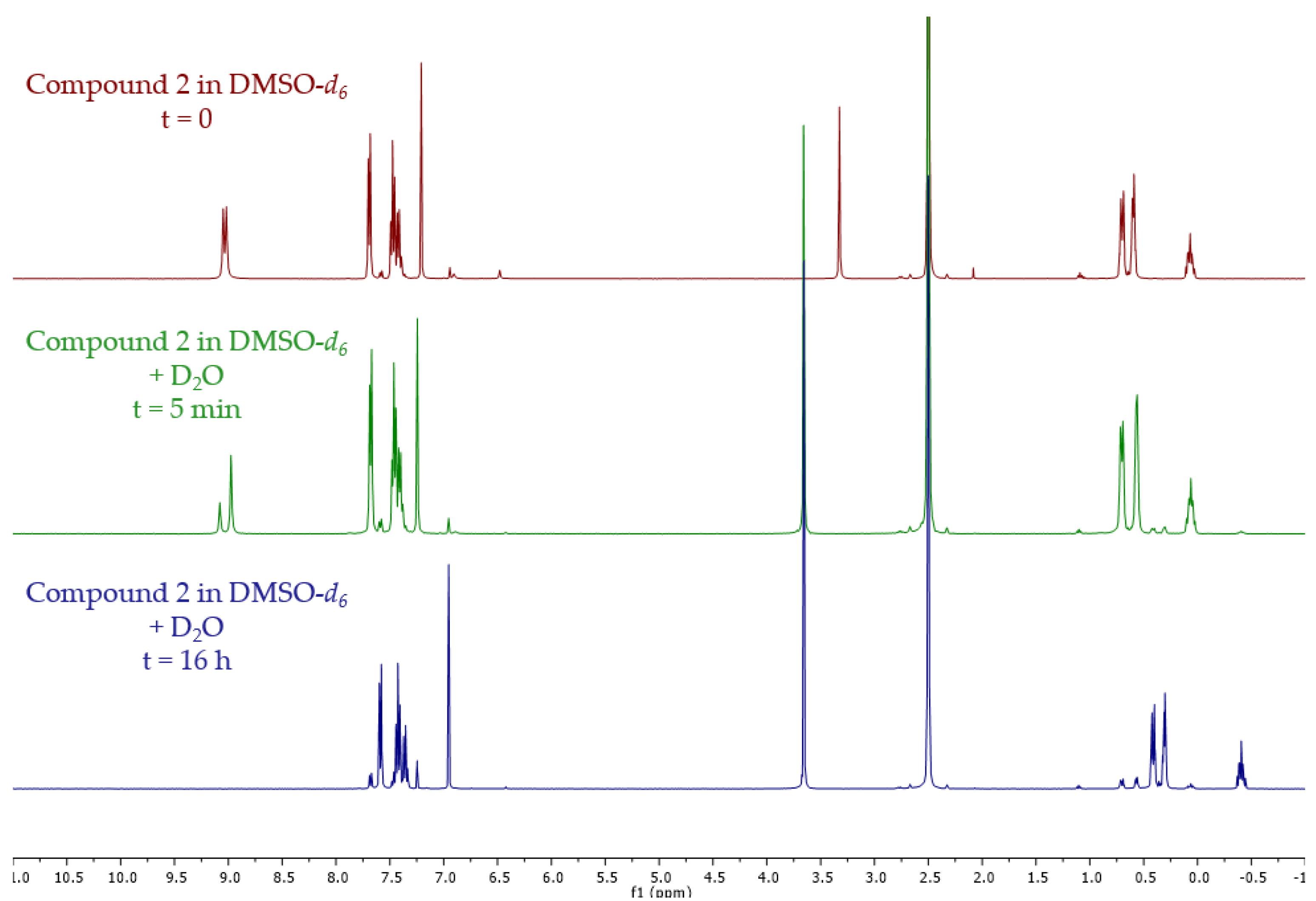
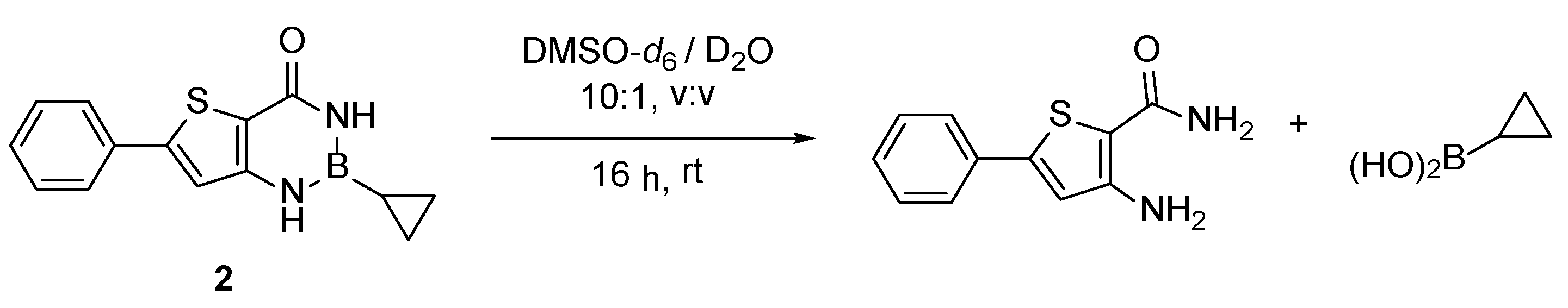
2. Results
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. World Malaria Report 2020: 20 Years of Global Progress and Challenges; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-001579-1. [Google Scholar]
- Straimer, J.; Gnadig, N.F.; Witkowski, B.; Amaratunga, C.; Duru, V.; Ramadani, A.P.; Dacheux, M.; Khim, N.; Zhang, L.; Lam, S.; et al. K13-propeller mutations confer artemisinin resistance in Plasmodium falciparum clinical isolates. Science 2015, 347, 428–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Pluijm, R.W.; Imwong, M.; Chau, N.H.; Hoa, N.T.; Thuy-Nhien, N.T.; Thanh, N.V.; Jittamala, P.; Hanboonkunupakarn, B.; Chutasmit, K.; Saelow, C.; et al. Determinants of dihydroartemisinin-piperaquine treatment failure in Plasmodium falciparum malaria in Cambodia, Thailand, and Vietnam: A prospective clinical, pharmacological, and genetic study. Lancet Infect. Dis. 2019, 19, 952–961. [Google Scholar] [CrossRef] [Green Version]
- Uwimana, A.; Legrand, E.; Stokes, B.H.; Ndikumana, J.-L.M.; Warsame, M.; Umulisa, N.; Ngamije, D.; Munyaneza, T.; Mazarati, J.-B.; Munguti, K.; et al. Emergence and clonal expansion of in vitro artemisinin-resistant Plasmodium falciparum kelch13 R561H mutant parasites in Rwanda. Nat. Med. 2020, 26, 1602–1608. [Google Scholar] [CrossRef] [PubMed]
- Uwimana, A.; Umulisa, N.; Venkatesan, M.; Svigel, S.S.; Zhou, Z.; Munyaneza, T.; Habiman, R.M.; Rucogoza, A.; Moriarty, L.F.; Sandford, R.; et al. Association of Plasmodium falciparum kelch13 R561H genotypes with delayed parasite clearance in Rwanda: An open-label, single-arm, multicentre, therapeutic efficacy study. Lancet Infect. Dis. 2021, 8. [Google Scholar] [CrossRef]
- Cohen, A.; Suzanne, P.; Lancelot, J.-C.; Verhaeghe, P.; Lesnard, A.; Basmaciyan, L.; Hutter, S.; Laget, M.; Dumètre, A.; Paloque, L.; et al. Discovery of new thienopyrimidinone derivatives displaying antimalarial properties toward both erythrocytic and hepatic stages of Plasmodium. Eur. J. Med. Chem. 2015, 95, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.M.H.; Abdel-Maksoud, M.S.; Oh, C.-H. Thieno[2,3-d]pyrimidine as a promising scaffold in medicinal chemistry: Recent advances. Bioorganic Med. Chem. 2019, 27, 1159–1194. [Google Scholar] [CrossRef] [PubMed]
- Kurasawa, O.; Miyazaki, T.; Homma, M.; Oguro, Y.; Imada, T.; Uchiyama, N.; Iwai, K.; Yamamoto, Y.; Ohori, M.; Hara, H.; et al. Discovery of a novel, highly potent, and selective thieno[3,2-d]pyrimidinone-based Cdc7 inhibitor with a quinuclidine moiety (TAK-931) as an orally active investigational antitumor agent. J. Med. Chem. 2020, 21, 1084–1104. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zhu, M.; Zhang, J.; Zhou, H. Synthesis of biologically active boron-containing compounds. MedChemComm 2018, 11, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Davies, G.H.M.; Mukhtar, A.; Saeednia, B.; Sherafat, F.; Kelly, C.B.; Molander, G.A. Azaborininones: Synthesis and structural analysis of a carbonyl-containing class of azaborines. J. Org. Chem. 2017, 82, 5380–5390. [Google Scholar] [CrossRef] [PubMed]
- Koyanagi, M.; Eichenauer, N.; Ihara, H.; Yamamoto, T.; Suginome, M. Anthranilamide-masked o-iodoarylboronic acids as coupling modules for iterative synthesis of ortho-linked oligoarenes. Chem. Lett. 2013, 42, 541–543. [Google Scholar] [CrossRef] [Green Version]
- Kamio, S.; Kageyuki, I.; Osaka, I.; Hatano, S.; Abe, M.; Yoshida, H. Anthranilamide (aam)-substituted diboron: Palladium-catalyzed selective b(aam) transfer. Chem. Commun. 2018, 54, 9290–9293. [Google Scholar] [CrossRef] [PubMed]
- Kamio, S.; Kageyuki, I.; Osaka, I.; Yoshida, H. Anthranilamide (aam)-substituted arylboranes in direct carbon–carbon bond-forming reactions. Chem. Commun. 2019, 55, 2624–2627. [Google Scholar] [CrossRef] [PubMed]
- Morwick, T.; Berry, A.; Brickwood, J.; Cardozo, M.; Catron, K.; DeTuri, M.; Emeigh, J.; Homon, C.; Hrapchak, M.; Jacober, S.; et al. Evolution of the thienopyridine class of inhibitors of IκB kinase-β: Part I: Hit-to-lead strategies. J. Med. Chem. 2006, 49, 2898–2908. [Google Scholar] [CrossRef] [PubMed]
- Knapp, D.M.; Gillis, E.P.; Burke, M.D. A general solution for unstable boronic acids: Slow-release ross-coupling from air-stable MIDA boronates. J. Am. Chem. Soc. 2009, 131, 6961–6963. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mustière, R.; Vanelle, P.; Primas, N. 2-Cyclopropyl-6-phenyl-2,3-dihydrothieno[3,2-d][1,3,2]diazaborinin-4(1H)-one. Molbank 2021, 2021, M1221. https://doi.org/10.3390/M1221
Mustière R, Vanelle P, Primas N. 2-Cyclopropyl-6-phenyl-2,3-dihydrothieno[3,2-d][1,3,2]diazaborinin-4(1H)-one. Molbank. 2021; 2021(2):M1221. https://doi.org/10.3390/M1221
Chicago/Turabian StyleMustière, Romain, Patrice Vanelle, and Nicolas Primas. 2021. "2-Cyclopropyl-6-phenyl-2,3-dihydrothieno[3,2-d][1,3,2]diazaborinin-4(1H)-one" Molbank 2021, no. 2: M1221. https://doi.org/10.3390/M1221
APA StyleMustière, R., Vanelle, P., & Primas, N. (2021). 2-Cyclopropyl-6-phenyl-2,3-dihydrothieno[3,2-d][1,3,2]diazaborinin-4(1H)-one. Molbank, 2021(2), M1221. https://doi.org/10.3390/M1221







